Abstract
The objective of this study was to determine the effects of investigational vaccine candidates on acute-phase proteins (APPs) as determined in GLP toxicology studies. Sixty-four GLP toxicity studies, which were submitted to the Food and Drug Administration from 2008 to 2012 in support of proposed clinical investigations, were reviewed and entered into a database. These studies employed the intramuscular route of injection and were conducted using New Zealand White rabbits. A retrospective review of these GLP toxicity studies was conducted to evaluate the changes in plasma levels of C-reactive protein (CRP), fibrinogen, and albumin as APPs following the administration of various investigational vaccines. The incidence and intensity of responses associated with acute-phase responses both positive and negative were observed to increase in animals when treated with vaccines containing more potent immunological components such as novel adjuvants that activate Toll-like receptors in the investigational vaccine products. Changes in plasma levels of CRP were prominent among these responses and provided a basis to propose a classification scheme of H, M, L, and N responding groups. These changes in plasma proteins reflect an activation of the acute-phase response and indicate increasing levels of systemic inflammation, which potentially may be correlated with important clinical adverse events.
Introduction
Vaccines activate the innate immune system as a first step in producing an efficacious response to various antigens alone or in combination with adjuvants. The proinflammatory response that follows the administration of a vaccine occurs with the release of a variety of cytokines, such as interleukin 6 and tumor necrosis factor α and involves different cellular components, such as antigen-presenting cells and neutrophils that operate through receptor-mediated mechanisms, such as Toll-like receptors. The release and subsequent actions of proinflammatory cytokines produce local reactogenic effects and have the potential to exert systemic effects. One consequence of initiating the acute-phase response is a change in the serum concentration of several plasma proteins collectively referred to as the acute-phase proteins (APPs). 1 –6 Given the potential of investigational vaccines to induce various adverse events that are associated with the release of systemic cytokines and the subsequent expression of APP, the relationship of changes in levels of APPs to investigational vaccines are likely to provide information useful in assessing clinical safety. This article is a retrospective analysis of nonclinical GLP toxicology studies as reported to the FDA from 2008 to 2011 that meet standard criteria for inclusion in the study. The purpose of the study was to assess changes in C-reactive protein (CRP) plasma levels following the intramuscular administration of different investigational vaccines and the relationship between levels of CRP and statistically significant changes in both positive and negative APPs. The ultimate goal of the study was to contribute to a better understanding of the potential of plasma CRP levels that were observed in rabbits in toxicology studies of investigational vaccines as biomarkers of potential, adverse clinical events. Recently, Destexhe et al reported on the utility of CRP as a biomarker of local inflammatory changes after the administration of diphtheria–tetanus–whole pertussis (DTPw) or model vaccine adjuvants. 7
Materials and Methods
Study Population
Data from individual New Zealand White rabbits were taken from the toxicology study reports that were provided in original Investigational New Drug Application (INDs) to support various, initial clinical investigations and transcribed into an independent database. Toxicity studies that were considered qualified for inclusion in the database fulfilled a standard set of criteria. The study design included a saline-treated arm, and the investigational vaccines were administered by intramuscular injection using needle and syringe. Studies were performed in a manner compliant with GLP and as a minimum collected data on blood levels of CRP, fibrinogen (FIB), albumin, total protein (TP), and rabbit heterophils, which are homologous to neutrophils in the rabbit, as part of the evaluation for clinical chemistry in the study animals. The CRP levels were determined using a commercial high-sensitivity enzyme-linked immunosorbent assay (ELISA) method with an interassay variability of <10% which was validated under GLP. The remaining variables were also measured by validated techniques that meet standards for GLP. The minimum group size was 5 animals per sex per group, and data from both males and females were combined after a statistical test was performed which failed to detect differences between sexes. Individual data provided in toxicology study reports on pertinent variables were reentered into a new database and statistically analyzed to ensure a consistent statistical approach and methodology. Only the data collected prior to and during the first interval of dosing was analyzed to avoid the potential for interactions between successive doses. The toxicology studies, which used the intramuscular route of administration, were submitted in 21 INDs. These studies yielded 64 different treatment groups, which composed various arms of the toxicology studies. Treatment groups consisted of various combinations of adjuvants and antigens or a saline control arm. In instances where multiple levels of antigen and/or adjuvant were studied, the highest combination of both the antigen and the adjuvant was used to avoid redundancies in the data set that had the potential of confounding an analysis of relationship between plasma levels of CRP and changes in other APPs (FIB, ALB, and total globulins).
Analysis
Various criteria were established for the inclusion of a toxicity study in the database and subsequent analysis. Criteria pertaining to study design and execution included the following: the investigational vaccine was administered by needle and syringe using an intramuscular route of injection. The total injection volume of the investigational vaccine was 0.5 mL per administration; and the New Zealand White rabbit was selected as the animal model. All statistical evaluations were performed using JMP (SAS Institute Inc, Cary, NC, USA). Summary statistics were computed to derive group averages using the study arm as the basic unit of analysis. For animals used in the toxicology studies, male rabbits weighed 1.8 to 3.6 kg and females weighed 2.0 to 3.6 kg. All animals were between 10 and 26 weeks of age. The findings presented in this retrospective study represent the results obtained from approximately 1180 animals. Blood chemistry values included the measurement of the following factors by commercially available, automated clinical chemistry analyzers that were validated under GLP: FIB, creatine kinase (CK), TP, and ALB. Globulin levels were determined by the subtraction of TP levels by ALB levels in accordance with the standard practice used in toxicology studies, and ALB–globulin ratios (A:G) were then computed. Plasma CRP was measured by a high-sensitivity ELISA method which was validated under GLP. The heterophil count for each rabbit was measured by automated hematology analyzer. Since blood heterophil count is not classified as a positive or negative responder to the release of APPs, statistical significant changes in heterophil count was categorized as increased, decreased, or not changed. An analysis of the CRP data was performed by pooling data from 230 saline-treated control rabbits prior to their injection and subsequently analyzing the distribution of CRP plasma levels. To establish an upper limit for the normal range of CRP levels, a 90th percentile level was calculated from the groups assigned to receive saline injections. The distribution of the population of CRP levels from all predosed saline controls in terms of their cumulative percentiles was compiled, and a plasma level corresponding to the upper 90th percentile level of the observed CRP plasma levels was selected as a critical threshold value. The threshold value was subsequently used to group different study arms based on the percentage of animals in a study arm that exceeded the threshold value. Percentages of ≥80%, <80% to ≥40%, <40% to ≥10%, and <10% of animals in a group that exceeded the threshold value were then used to assign that arm of the toxicology study to 1 of 4 corresponding classifications of high (H), moderate (M), low (L), or nonresponse (N), respectively. Using each study as a statistical unit and the saline-treated arm as a standard control arm, statistical comparisons were made between different arms of a study for the outcome variables CRP, FIB, ALB globulin, ALB–globlin ratio, and heterophil count. Using the plasma levels within individual arms of the toxicity studies, both negative and positive APPs and heterophil count were statistically analyzed using Dunnett test (P < .05) for differences relative to the concurrent saline-treated control group. Since some APPs are anticipated to increase and other decrease in a manner that is consistent with the APR, the agreement or concordance between positive and negative responses was determined relative to increases in CRP levels. The percentage of concordance was defined as the frequency of agreement relative to the anticipated change in APP based on their classification as either negative or positive APPs in accordance with Gruyse et al. 8 Using this classification scheme, ALB is a negative APP because its levels decrease in response whereas FIB is a positive APP as its levels increase. Concordance was summated by groups in accordance with their classifications for H, M, L, and N response. Similarly, changes in the direction of the heterophil count was summarized in terms of increases, decreases, and no change for all studies based on the results of individual groups, which comprised a single study arm after their classified as H, M, L, and N response.
Results
The levels of CRP were compared before and after the injection of saline in animals assigned to the control groups. In 230 rabbits from different controls groups, the median and average levels of CRP prior to dosing with saline were determined and found to be 1.05 and 4.92 μg/mL, whereas the maximum observed level of plasma CRP was 150.75 μg/mL. Following injection in the same group of saline control animals, the median and average postdose CRP levels were 4.72 μg/mL, respectively, and 12.2 μg/mL with an observed maximum of 230.4 μg/mL. Based on the classification scheme, 20 different arms of the various toxicology studies were assigned to the H category, 9 to the M category, 12 to the L category, and 23 to the N category. The average and median CRP levels for the different groups over the period of observation are presented in Table 1. Measurement of CRP levels in the plasma of rabbits following administration of a variety of novel vaccine candidates which included various adjuvants is based on the classification scheme of H, M, L, or N relative to the injection of saline in control groups. Arms of the various toxicology studies were assigned to the H category (N = 20), M category (N = 9), L category (N = 12), or N category (N = 23). The classification scheme was based on an upper limit (90th percentile level) for the normal range of CRP levels in rabbits given saline intramuscularly. Threshold values were subsequently determined using the percentage of animals in a study arm that exceeded the threshold value. Percentage cutoffs defining groups were ≥80%, <80% to ≥40%, <40% to ≥10%, and <10% of animals in a group that exceeded the threshold value determined the classification group. The observed values within each category are presented in the graph (Figure 1) in a whiskers and box plot. The top and lower limits of the box plot indicate the 75th and 25th percentiles of the populations of the observed levels in each category of response along with the median values. The upper and lower whiskers indicate the 90th and 10th percentiles for the observed values. Due to the nonparametric distribution of the observed values, a Wilcoxon pairwise comparison was performed at P < 0.05. This analysis indicated that each pairwise comparison by category of response was statistically different from each other with the exception of the pairwise comparison of M and L categories.
Quantitative Measures of CRP and Classification by Response Category.
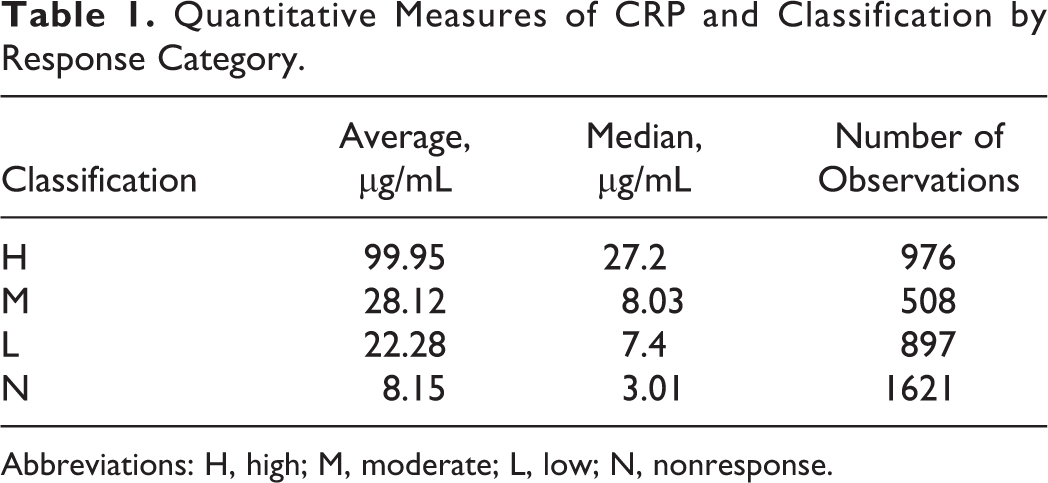
Abbreviations: H, high; M, moderate; L, low; N, nonresponse.
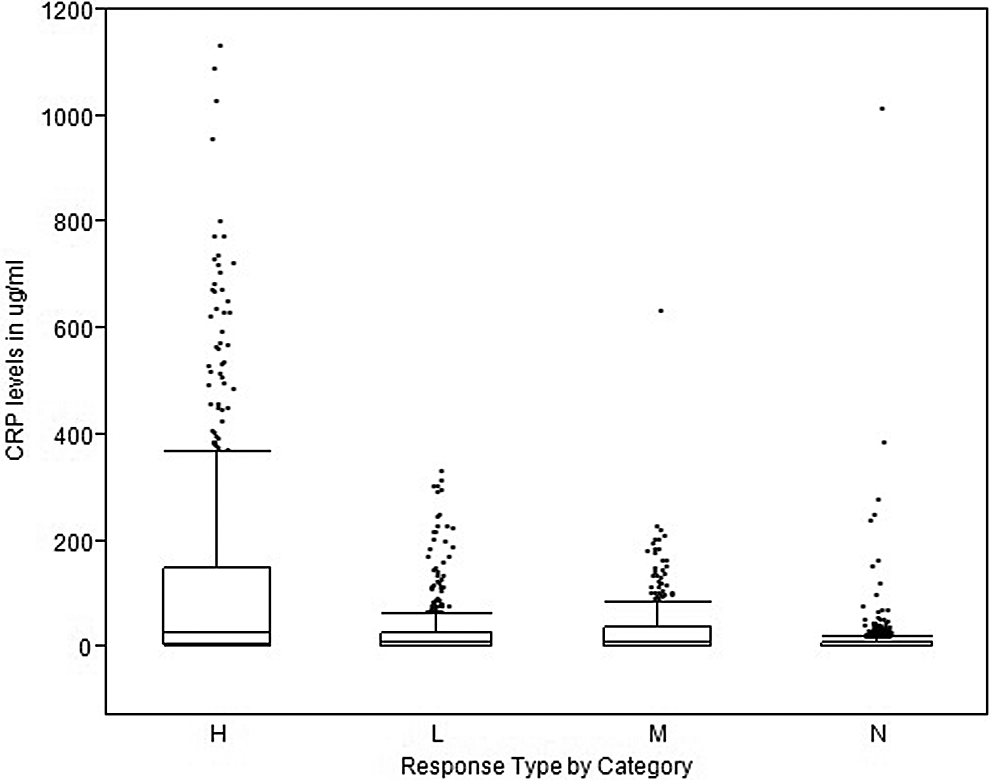
Box and whiskers plot of plasma levels for CRP (μg/mL) taken from toxicology studies submitted in 21 INDs and organized by the response categories of H, M, L, and N. H indicates high; M, moderate; L, low; N, nonresponse. The top and lower limits of the box plot are the 75th and 25th percentiles and the upper and lower whiskers indicate the 90th and 10th values for the observed levels in each category. These studies yielded 64 different treatment groups and composed the various arms of the toxicology studies which used the intramuscular route of administration.
Levels of CK were examined to determine whether local tissue damage that was observed following intramuscular injection was a significant factor contributing to differences in CRP levels. As a measure of tissue damage following intramuscular injection, CK values were analyzed in accordance with the different classifications and are presented in Table 2. Levels of CK remained relatively constant across the different classifications. Furthermore, although a 12-fold difference was observed between the average CRP levels for the highest and the lowest classification groups, only a 1.2-fold difference was found for CK levels between these 2 groups. Accordingly, the degree of trauma experienced at the site of injection as reflected by CK levels is 10-fold less than that shown by changes in CRP levels. Levels of CK remained relatively constant over a greater and 12-fold change in CRP levels between categories reflecting a lack of correspondence between local injury and systemic manifestation of the acute-phase response. Based on the anticipated patterns of change between various APPs and their associated measures, agreement in the pattern of acute-phase responses for various individual APP was denoted as having occurred when statistically significant changes (P < 0.05) were found between individual study arms comparing an investigational vaccine product and saline. These differences between arms of individual toxicity studies were determined using Dunnett statistical test, and the aggregate results were summated across all studies. The frequency of agreement in the anticipated change in plasma levels between positive and negative APP was summarized as a percentage and reported in Table 3. For example, in accordance with this scheme, statistically significant increases that were found between different groups in a study between CRP and FIB were considered as a positive concordance as were a decrease in ALB levels, since these changes would be consistent with anticipated changes in APPs. The highest degree of agreement in change occurred with total globulin and the ALB–globulin ratio in H response group. This may reflect not only the increased expression of CRP in this group but also the expression of other globulin components within the family of APPs such as serum amyloid A and α 2 macroglobulins. Fibrinogen levels, which rose 4.7-fold, also increased but in a nonproportionate manner to CRP levels. Presentation of the percentage of agreement of anticipated change consistent with the acute-phase response. The concordant and reciprocal nature of anticipated changes between plasma CRP levels and other APPs were examined. Changes in levels of FIB were anticipated to parallel those of CRP, whereas levels of ALB should be inverted to those of CRP. Levels of total globulins represent a mixture of responses, since it is an aggregate measure of several globular proteins including other acute-phase reactant proteins as well as immunoglobulins. Higher plasma levels of CRP as represented by their categorical grouping demonstrated a greater degree of agreement in association with another prominent acute-phase reactant protein, FIB. Hence, higher categorical grouping was generally associated with greater degrees of concordance. Besides changes in clinical chemistry end points, absolute neutrophil count was assessed as a responsive hematological end point that follows activation of the innate immune system. To assess changes in rabbit heterophils, which are the counterpart to neutrophils in other species, the reported blood levels across different toxicology studies were statistically analyzed on the basis of individual study arms during the first week that followed the initial dose of the investigational vaccines. Statistically significant increases or decreases were noted and grouped in accordance with the H, M, L, and N categories of response. The results are presented in Table 4 as the percentage of incidence of change by category of response. Within a category of response, a greater proportion of statistically significant increases in heterophile counts were observed with increasing average levels of CRP, which may be a reflection of the intensity of the acute-phase response. Percent change in blood levels of circulating rabbit heterophils by category of response is based on plasma levels of CRP from various arms of toxicology studies of novel vaccines including adjuvants. When represented by categories based on increases in plasma CRP levels, concomitant changes in heterophil levels are observed reflecting activation of an arm of the acute inflammatory response.
Summary of CK Levels by Response Category.a
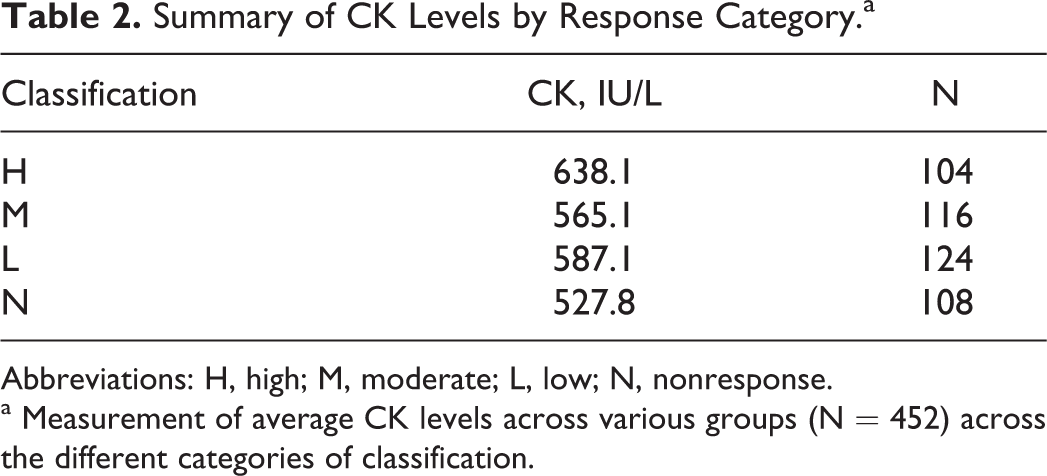
Abbreviations: H, high; M, moderate; L, low; N, nonresponse.
a Measurement of average CK levels across various groups (N = 452) across the different categories of classification.
Concordance Between Category of Response and Changes in Components of the Acute Phase Response.

Abbreviations: H, high; M, moderate; L, low; N, nonresponse.
Category of Response and Percent Change in Heterophil Level.
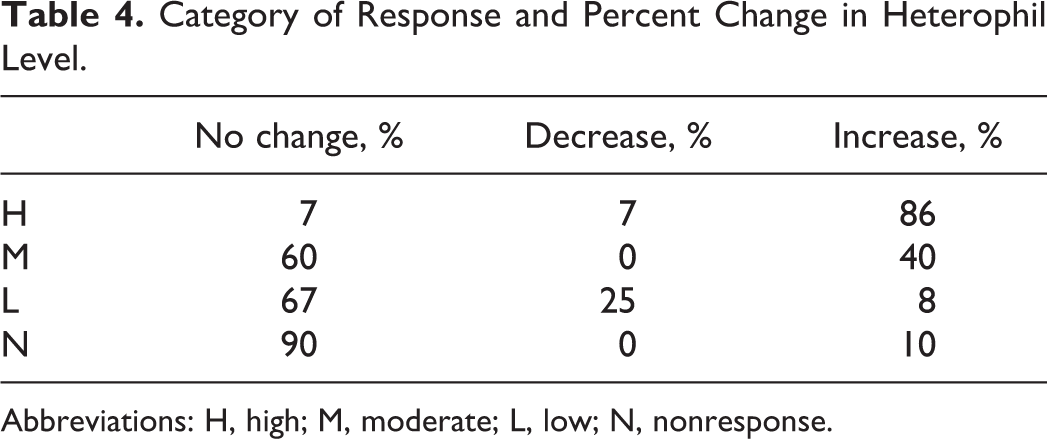
Abbreviations: H, high; M, moderate; L, low; N, nonresponse.
Discussion
Toxicology studies may be performed prior to conducting initial clinical studies of novel, investigational vaccines. The presently reported study, which summarizes a series of GLP toxicology studies in the rabbit, examines the feasibility of using outcome measures found in these studies that are associated with the expression of the acute-phase response. Levels of CRP were measured prior to and following intramuscular injection of saline to control rabbits, and the average, median, and observed Cmax CRP levels increased 1.5- to 4.5-fold across all these measures relative to saline-injected control groups. The increase in CRP levels is likely due to a response to the vaccine and/or adjuvant but also could conceivably include a response to the inflammatory events associated with the trauma of injection. However, the reported minimal inflammatory events that occur due to the trauma of injection suggest that it is unlikely to account for a significant portion of the large increases in CRP levels that were observed following the injection of saline. Furthermore, the trauma of injection was reported to result in only slight increases in plasma levels of CK, 9 –11 and this finding is consistent with those observed by Thuilliez et al that demonstrated the occurrence of slight local inflammatory reaction to the injection of normal saline. 12 Additionally, levels of serum CK across different groups did not meaningfully vary with increasing levels of CRP across these groups, which suggests that trauma at the site of injection contributed in limited and consistent manner and was not proportionate to increases in CRP levels. In contrast to the findings in groups of animals given normal saline, levels of CRP were elevated after the injection of candidate vaccines. Based on the 90th percentile level found in a population of preinjected saline controls, a classification scheme was devised of H, M, L, and N groupings. This classification scheme may be reflective of immunological potency based on the ability to stimulate the growth and differentiation of immune cells and their expression of immunoglobulins, since a review of animals with the highest levels of CRP from the different toxicology studies suggests an association with investigational vaccines containing Toll-like receptors, cytotoxic T-cell activations, or Th1 enhancers. Conversely, low levels of CRP were predominantly associated with treatments that were composed of antigens alone or combinations of antigens with alum, and nonresponsive groups were largely composed of saline-treated animals. Supportive of the changes in levels of CRP levels, the levels of FIB varied positively and ALB negatively in accordance with classification categories. Furthermore, globulin levels and ALB–globulin ratio changed in manner reflective of an acute-phase response. Dextexhe et al reported that CRP and FIB levels increased and were accompanied by recruitment of circulating heterophils and mixed inflammatory cell infiltrates at the site of injection following the injection of either DTPw–hepatitis B vaccine or 3 different adjuvant systems. The presently reported findings confirm and extend those of Destexhe et al 7 regarding increases in CRP and FIB following the injection of vaccines and adjuvants as the present findings studied the relationship of 21 unique and novel vaccine candidates and incorporates an extensive range of antigen and adjuvant combinations. In summary, this article investigates the relationship between candidate vaccines and changes in APPs using data collected during the course of GLP toxicology studies. The results suggest that plasma CRP levels are a promising biomarker in assessing the acute-phase response following the administration of novel, candidate vaccine products, which may be linked to the expression of clinical adverse events such as malaise, fatigue, and pain at the site of injection. In support of this concept, a limited assessment was conducted of the results of 9 phase 1 clinical studies, the findings suggest a useful correspondence may exists between elevations in CRP levels that were observed in rabbits when used in toxicology studies and clinical adverse events subsequently observed in clinical subjects during the course of initial clinical studies. This topic is the subject of an ongoing retrospective study into the intensity and frequency of the adverse clinical outcomes in early clinical studies and their relationship to the classification scheme proposed in this article.
Footnotes
Acknowledgments
Carol Yang and Liliana Yu are recognized for their assembly and analysis of the databases.
Author Contribution
Green contributed to conception and design, acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; and agreed to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
