Abstract
Aristolochic acid (AA) can accumulate in the tubulointerstitium and cause kidney-specific injuries. However, the mechanism by which AA induces nephropathy remains largely unknown. This study explored the effect of AA-I on tight junctions (TJs), and the fence function in a renal epithelial cell (REC). NRK-52E cells were exposed to different concentrations of AA-I for 4 h or 25 μM AA-I for different time. Cell viability was detected by MTT, cell apoptosis by flow cytometric analysis, the expression of zonula occludens-1 (ZO-1), E-cadherin and polarity scaffold (Par3) by western blot and immunofluorescence, cell membrane permeability by transepithelial electrical resistance (TEER). It was found that AA-I reduced the expression of ZO-1, E-cadherin, and Par3 in a concentration- and time-dependent fashion, and altered the distribution of ZO-1 and Par3 from cell membrane to cell plasma. In parallel to the reduced expression of TJ proteins, TEER exhibited a significant reduction in response to AA-I treatment in a time- and concentration-dependent manner. Meanwhile, α-SMA expression in cells was increased following AA-I treatment. In contrast, cell viability and apoptosis were unaltered with the doses of AA-I tested. Our findings show for the first time that AA-I treatment in cultured RECs induced a rapid disruption of TJ and the fence function preceding apoptosis, which indicated that aberrant expression of TJ proteins within RECs may be involved in initiating the renal tubulointerstitial disorders.
Introduction
Aristolochic acids (AAs) are a family of structurally related nitrophenanthrene carboxylic acids contained in the plant extract of the Aristolochia species such as
The epithelial tight junction (TJ) is one of the epithelial cell–cell junctional complexes which is responsible for the control of paracellular transport between epithelial cells (‘gate’ function) and maintenance of apical/basolateral polarity by preventing the diffusion of membrane lipids and/or proteins from one surface domain to another (‘fence’ function). 9 In the renal tubular epithelia, the TJ is formed by the organization of a number of specific proteins including occludin, zonula occludens (ZO-1), claudins and junctional adhesion molecules. 10 The TJ proteins that regulate epithelial polarity also control cell proliferation and differentiation and participate in EMT. 11 Alteration of the TJ complex in renal epithelial cells (RECs) can affect renal barrier function and perturb normal kidney homeostasis. Transforming growth factor-β1 (TGF-β1)-induced EMT is associated with reduced expression of ZO-1 and E-cadherin. 12 Cyclosporine A (CsA) treatment could cause an alteration of the TJ complex, resulting in changes in transepithelial permeability in Madin-Darby canine kidney cell line (MDCK) distal tubular/collecting duct cells. 13 The potential effect of AA on the cell junctions and transepithelial permeability in renal tubular epithelia is unknown. The aim of this study was to explore the effect of AA-I on the expression and distribution of ZO-1, E-cadherin and Par3 as well as the function of TJs in NRK-52E cell line.
Materials and methods
Antibody and reagents
Rabbit anti-ZO-1 polyclonal antibody, Alexa Fluor 546/633-conjugated donkey anti-rabbit, Alexa Fluor 488-conjugated donkey anti-mouse and DAPI were purchased from Invitrogen (Carlsbad, CA, USA). Rabbit anti-Par3 antibody and mouse anti-αSMA from Upstate (Millipore, Bedford, MA, USA). Mouse anti-E-cadherin monoclonal antibody from BD Biosciences (San Jose, CA, USA). AA-I from Guangzhou Institute for Drug Control. Dulbecco’s modified eagle’s medium (DMEM)/F12, fetal calf serum (FCS) from Gibco-BRL (Grand Island, NY, USA, Cat.No. 26140-079, with endotoxin level ≤50 EU/ml, levels routinely ≤ 10 EU/ml). Dimethylsulfoxide (DMSO) and Annexin V-FTIC Apoptosis Detection Kit (50 assay) from Sigma Chemicals Co. (St Louis, MO, USA).
Cell culture and treatment
NRK-52E cells were obtained from American Type Culture Collection (Manassas, VA, USA). The cells were maintained in cell culture flasks in DMEM-F12 containing 10% fetal bovine serum at 37°C with 5%CO2. AA-I was prepared as a stock solution of 8.3 μg/ml in 100% DMSO. After incubation with serum-free for 24 h, NRK-52E cells were exposed to different concentrations of AA-I (0, 5, 10, 25, 50, and 100 μM) for 4 h or 25 μM for different times (0, 2, 4, 6, 8, 12, 16, and 24 h), respectively. Cell viability was detected by MTT; cell apoptosis rate by flow cytometric analysis; cell membrane permeability by transepithelial electrical resistance (TEER); the expression of ZO-1, E-cadherin, and Par3 by western blot; the expression of those proteins mentioned above as well as α-SMA was detected by immunofluorescence.
Detection of cell viability by MTT
Cytotoxicity was determined using MTT assay as described by Mossman 14 with minor modification. In brief, NRK-52E cells were seeded at 10 5 cells/ml in 96-well plates. Twenty-four hours after plating, the cells were incubated of serum-free DMEM/F12 for another 24 h, and then exposed to 0, 5, 10, 25, 50, 100 μM of AA-I dissolved in serum-free medium for 4 h. The cells were exposed to 25 μM of AA-I for 0, 2, 4, 8, 12, 16, and 24 h, respectively, after incubation for appropriate time periods, MTT was added and cells were incubated for additional 4 h. The absorbance was then measured at 570 nm on a spectramax plus 834 multiwell plate reader (Bio-Rad Model 550, USA). Relative cell viability (%) was calculated as (mean absorbance of sample/mean absorbance of vehicle control) × 100.
Detection of apoptotic rate by flow cytometric analysis
NRK-52E cells were seeded at 10 5 cells/ml in six-well plates and were treated in different concentration of AA-I (0, 5, 10, 25, 50, and 100 μM) for 4 h or 25 μM of AA-I for different times (0, 4, 8, 12, 16, and 24 h) as described above. The cells were harvested and rinsed with phosphate buffered saline (PBS). The apoptosis was detected according to the manufacturer’s instructions of the Annexin V-FTIC Apoptosis Detection Kit (Sigma, St louis, MO, USA).
Transepithelial electrical resistance
The intactness of paracellular pathways of the NRK-52E monolayer to small ions was monitored by measurement of TEER using a Millicell-ERS Electrical Resistance System (Millipore, Bedford, MA, USA). Cells were grown on Costar HTS-Transwell (Corning, NY, USA) cell culture inserts (pore size 0.4 μm). NRK-52E cells that were seeded at a density of 1 × 10 5 cells/ml were seen to reach a stable TEER, representing a confluent monolayer 5 days after seeding. The cells were exposed to treatment on the apical side only. The point of drug addition was taken as time 0, and TEER was monitored over a range of times. TEER was normalized to the area of the filter after removal of background resistance of a blank filter that contained only medium. TEER was expressed as the relative value to that at 0 min. 15
Western blotting
With the treatment described above, cells were scraped into 1× cell lysis buffer, which was diluted in 10× cell lysis buffer (Cell Signaling Technology, USA). Protein concentration was determined using a BCA protein assay kit (Pierce, Rockford, IL, USA). Equal amounts of cell extracts were electrophoresed on SDS-poly-acrylamide gels, and proteins were transferred to nitrocellulose membrane. Membranes were blocked with 5% (wt/vol) milk proteins/Tris-buffered saline and incubated overnight at 4°C with the primary antibody such as mouse anti-E-cadherin, rabbit anti-ZO-1 and rabbit anti-Par3. Bound antibody was detected with appropriate secondary antibodies and enhanced chemiluminescence.
Immunofluorescence co-staining of ZO-1, E-cadherin, Par3, and α-SMA
Coverslips with cells were fixed with methanol at −20°C and then washed three times with cold PBS. Cells were blocked with 5% goat serum. Cells were incubated with primary antibody: mouse anti-E-cadherin, rabbit anti-ZO-1, rabbit anti-Par3, or mouse anti α-SMA, respectively. Some coverslips were incubated with PBS alone in place of primary antibody as a negative control. After incubated overnight at 4°C, the cells were washed three times with PBS. Alexa-488 (546/633)-conjugated appropriate secondary antibody was added and incubated for 40 min at 37°C. Then the cells were washed three times with PBS. Cover slips were mounted using the Aqueous mounting medium (R&D). Signal was detected under LSM 510 confocal immunofluorescence (Carl Zeiss, Inc., Jena, Germany), images were obtained with LSM image browser.
Statistical analysis
Data are expressed as means ± SD. Statistical analysis was performed by unpaired
Results
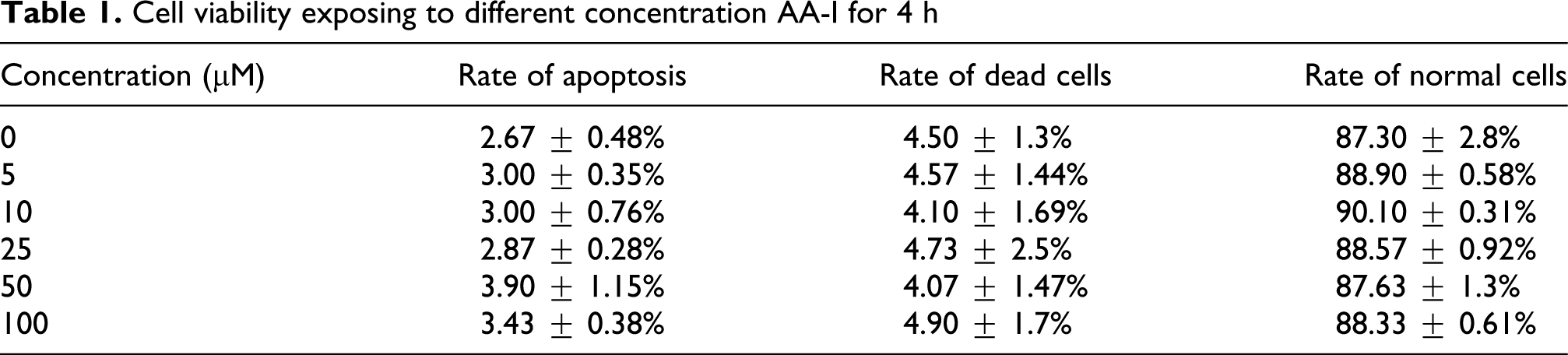
Effects of AA-I on cell viability
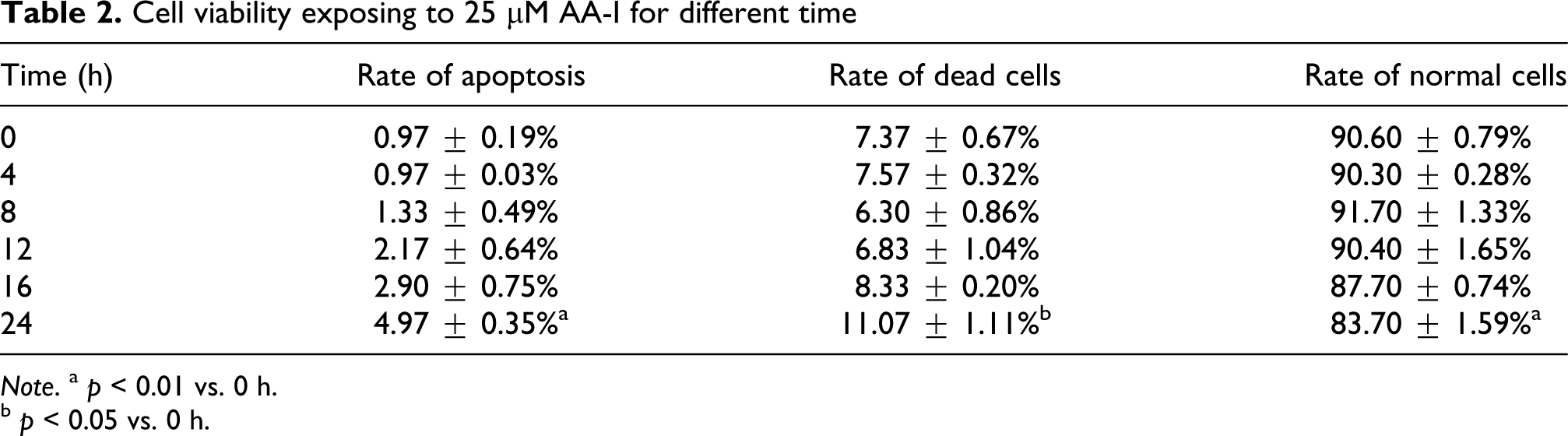
The viability of NRK-52E cells was analyzed after treatment with 25 μM AA-I for 0, 2, 4, 6, 8, 12, 16, and 24 h, respectively. The cellular viability was not significantly altered at the first 8 h when treated with 25 μM of AA-I. However, at 12, 16, and 24 h after treatment with AA-I, cell viability was reduced dramatically, with 78.8 ± 7.4%, 77.5 ± 14.8%, and 72.3 ± 11.7% compared to the control group (
Effects of AA-I on cell apoptosis
As exposing to 5, 10, 25, 50, and 100 μM AA-I for 4 h, respectively, cell apoptosis showed no significant difference compared to control group (Table 1). As exposing to 25 μM AA-I for 24 h, the value of cell apoptosis and death significantly increased as comparing with control group (Table 2 ).
Cell viability exposing to different concentration AA-I for 4 h
Cell viability exposing to 25 μM AA-I for different time
b
Effect of AA-I on TEER
AA-I induced a significant decrease in TEER of cells in concentration- and time-dependent way. As exposing to AA-I with 5, 10, 25, 50, and 100 μM for 4 h, TEER reduced in different levels with 56.6 ± 10.7%, 47.0 ± 3.6%, 44.3 ± 3.5%, 37.6 ± 4.0%, and 32.1 ± 6.3% (
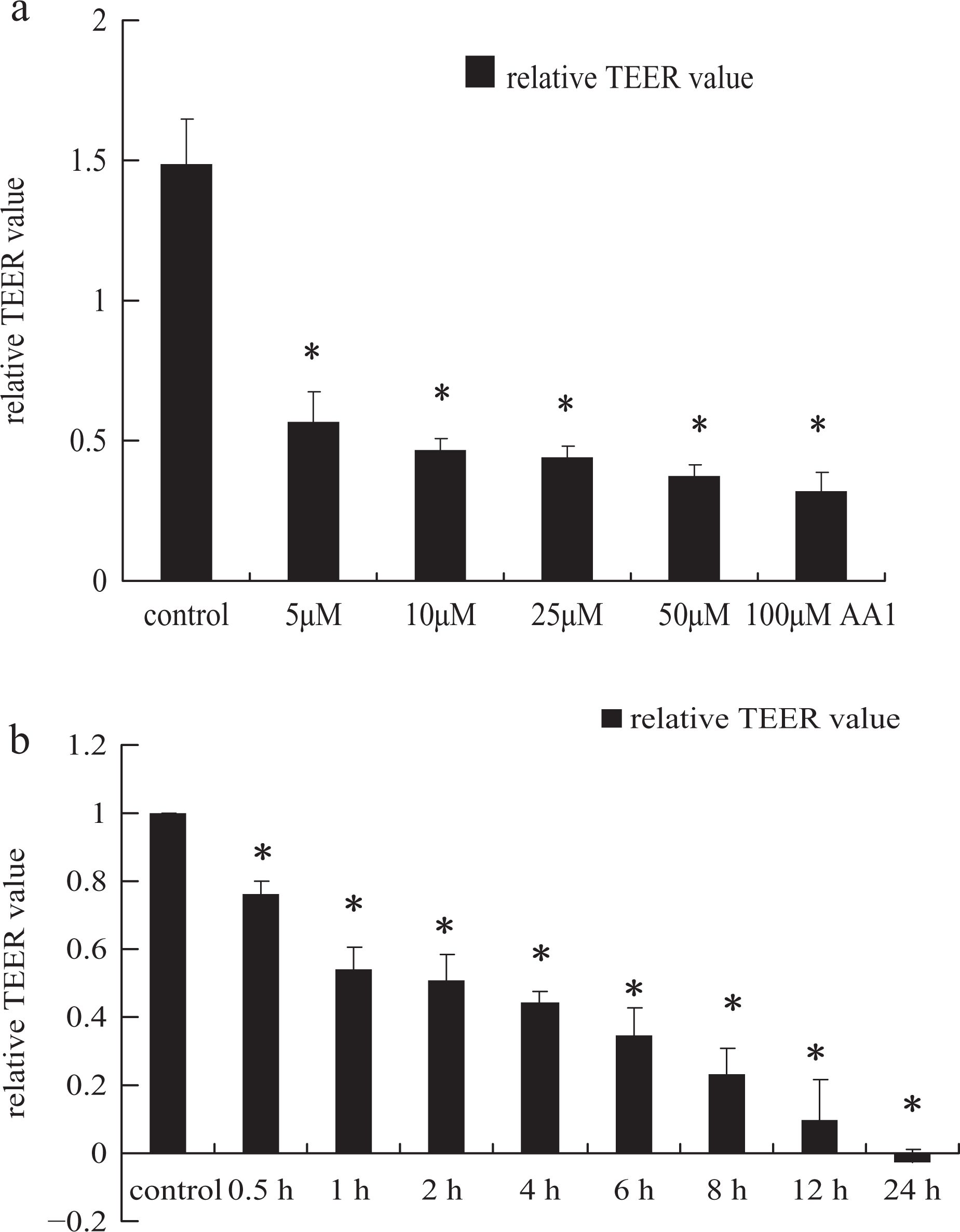
(a) Effect of different concentration of AA-I on transepithelial electrical resistance (TEER) of NRK-52E cells for 4 h. The columns represent the ratio of Δ
Effect of AA-I on expression of tight junction proteins
Compared to the control group, western blot analysis showed that AA-I induced a significant decrease in TJ and adheren junction protein expression of cells in concentration- and time-dependent way. On treatment with 25 μM AA-I for 2, 4, 8, 12, and 16 h, respectively, ZO-1 decreased to 93.2 ± 1.2%, 63.5 ± 5.3% (
On treatment with AA-I in concentration of 5, 10, 25, 50, and 100 μM for 4 h, ZO-1 decreased to 73.6 ± 16.9%, 64.1 ± 9.2%, 58.4 ± 12.6%, 54.9 ± 11.0%, and 36.0 ± 6.3% (

(a–c) Alteration in the protein expression of ZO-1 (a), E-cadherin (b), and Par3 (c) in NRK-52E cells after exposure to different concentration of AA-I for 4 h, respectively, (d–f) Alteration in the protein expression of ZO-1 (d), E-cadherin (e), and Par3 (f) in NRK-52E cells after exposure to 25 μM AA-I for 2, 4, 8, 12, and16 h. Cells incubated without AA-I were used as control. The relative expression of ZO-1, E-cadherin, and Par3 was determined by normalization to β-actin. The images (a–f) are representatives of the results obtained from three separate experiments. The columns in (a–c) represent the relative expression of ZO-1, E-cadherin, and Par3 and data are present as the mean ± SD (
Effect of AA-I on distribution of tight junction
The effects of a 4-h treatment with different concentrations of AA-I on distribution of TJ and adheren junctions associated protein in cells were characterized. Co-staining of ZO-1, E-cadherin, and Par3 in control cells were predominantly limited to cell membrane of cell–cell contact. On treatment with 25 μM AA-I for 4 h, the weaker and tortuous fluorescence intensity of ZO-1, E-cadherin, and Par3 protein were observed; while on treatment with 50 and 100 μM AA-I treatment for 4 h, distinct breaks were observed in the lateral membrane co-staining for ZO-1, E-cadherin, and Par3, which suggested that TJ and adheren junction integrity was seriously disrupted and cell polarity became weakened or even broke down. As the reduction and rupture of ZO-1 staining in membrane, ZO-1 accumulation was observed in cell plasma with prolonged treatment. Similar alteration was detected in Par3 staining (Figure 3 ).

Effect of different concentration of AA-I on expression of TJ (ZO-1), AJ (E-cadherin), and polarity scaffold (Par3). (a) Without AA-I treatment, ZO-1, E-cadherin, and Par3 located on cell membrane and expressed as continuous, linear fluorescence staining. (b–d) As exposing to 25 μM (b), 50 μM (c), 100 μM (d) of AA-I, the fluorescence intensity of above three proteins became weaker and tortuous; ZO-1 and Par3 broke down from cell membrane and accumulated in cell plasma. As concentration increased, the damage became more and more severe.
As the AA-I (25 μM) exposure of cells for 2 h, tortuous co-staining of ZO-1, E-cadherin, and Par3 were observed. The alteration became more pronounced as time prolonging. When the cells were treated with AA for 16 h, drastically damaged TJs of cell membrane and accumulation of Par3 in cell plasma were observed (Figure 4 ).

Effect of 25 μM AA-I treatment for different time points(0, 2, 4, 8, 12, and 16 h) on expression of TJ (ZO-1), AJ (E-cadherin),, and Par3 located on cell membrane and expressed as continuous, linear fluorescence staining. (b–f) As exposing to 25 μM AA-I for 2 h (b), 4 h (c), 8 h (d), 12 h (e), 16 h (f), the fluorescence intensity of above three proteins became weaker and tortuous; ZO-1 and Par3 broke down from cell membrane and accumulated in cell plasma. As time prolonged, damage became more pronounced.
Effect of AA-I on the expression of α-SMA
In control group, α-SMA expression was not detected in cultured cells. On treatment with AA-I (25 μM) for 4 h, expression of α-SMA was detected obviously in cell plasma (Figure 5 ).

The expression of α-SMA in NRK-52E cells treated with 25 μM AA-I for 4 h. Compared with untreated cells (a), the expression of α-SMA was significantly increased as exposing to AA-I for 4 h (b).
Discussion
AAs are one important group of naturally occurring toxic compounds causing renal fibrosis and urothelial carcinoma. It is considered that AAs can accumulate in the tubulointerstitium and thus cause kidney-specific injuries. 4 AAs induced cytotoxic effects in a dose-dependent and time-dependent manner.16,17 AA-I was found to be most toxic in MDCK cells upon exposure for 24, 48, and 72 h. The significant apoptosis in MDCK cells was observed with AA at as low as micromol/L following exposure for 24 h. 17 Our study found that as exposing to 5, 10, 25, 50, and 100 μM AA-I to NRK-52E cells for 4 h, cell apoptosis showed no significant difference compared to control group. As exposing to 25 μM AA-I for 24 h, the cell apoptosis and death significantly increased. In the present study, we try to observe the early alteration of cells in expression of ZO-1, E-cadherin, and Par3, as well as the fence function after relative lower dose of AA-I exposure or short period treatment before the appearance of the apoptosis and the changes of viability of cells. Based on the results of our preliminary experiments, NRK-52E cells were exposed to different concentrations of AA-I for 4 h or 25 μM AA-I for different time. The results demonstrated that complex changes in the TJs as well as the disruption of the fence function were induced even by low dose of AA-I (5 μM) treatment for short time (4 h) and happened before the apoptosis and the changes of viability of cells. These findings indicated that aberrant expression of TJ proteins may implicate in initiating the renal tubulointerstitial disorder.
Alteration of the TJ complex in RECs can affect renal barrier function and perturb normal kidney homeostasis.13,18 One of the major functions of the epithelial TJ is the gate function, which refers to the control of movement of molecules between epithelial cells via the paracellular route. TEER is a parameter of TJ permeability. If the gate function of renal tubular epithelial cells (RTEC) is impaired, compartments in the filtrate which are kept out of body will get into renal interstitial easily. Aberrant expression of individual claudin isoforms within the TJ of RECs has been implicated in renal diseases. CsA treatment caused an alteration of the TJ complex, resulting in changes in transepithelial permeability in MDCK distal tubular/collecting duct cells. 19 One mechanism for the reduction in glomerular filtration rate (GFR) in acute renal failure (ARF) is the ‘backleak’ of the filtrate across the damaged tubular epithelium and intratubular obstruction. Recent demonstration of TJ abnormalities such as altered cellular distribution of the TJ protein, and rapid internalization of E-cadherin as well as disruption of its interaction with the catenins provides the molecular and cellular basis for these observations. Reduced expression and redistribution of TJ proteins have also been documented in renal allografts with postischemic injury. 20 In primary culture of porcine thyroid epithelium treated with both TGF-β and epidermal growth factor (EGF), reduced expression of claudin-1 and occludin occurred in the first 24 h, accompanied by a loss of transepithelial resistance and a marked increase in the paracellular flux of [3H]inulin. 21 In our study, an early event of AA-induced renal tubular epithelial cell injure was investigated. AA-induced cell TJ function disturbance occurred at a very early period after exposure (30 min), although the significant structure alteration only could be observed after a later period (from 4 h). This observation is consistent with similar researches by others. 22 Function alteration, which occurs earlier than structure alteration, refers to the structure of TJ. As for the structure of TJ, junction proteins of adjacent cells fasten each other like a button. 23 When exposing to physiological or pathological factors, for example, a course of EMT, 24 TJ loosen this ‘button’ first, with or without redistribution and expression alteration. 25 Significant alteration of TJ protein expression could be also detected as exposed to a low-dose AA (5 μM) within a short period (for 4 h). TJ injury became more pronounced during prolonged exposure time or increasing concentration. Compared with 10 and 25 μM, the effect on TEER of treatment with AA at 5 μM seems to have a relative moderate reduction on TEER in early time period, like 30 min, 1 h, 2 h (this part of result was not given in text). However, after 4 h treatment, the effect on TEER of different concentration of AA seems similar, while the effect of the protein profile shown significant difference between these concentrations. This mismatch, once again, indicates that function disturbance occurs earlier than structure alteration. Our study demonstrated that treatment with AA results in loss of TJ integrity and an increase in paracellular permeability in NRK-52E cells. Although the molecular mechanisms involved in these processes were poorly understood in this experiment, AA-induced TJ dysfunction leads to a loss of barrier function, and increased paracellular permeability, which could allow the leakage of AA-contained glomerular filtrate (tubular fluid) across tubular epithelium and back to the renal interstitials. Accumulation of AA-I in renal interstitials may facilitate renal interstitial fibrosis in patients with AAN.
The TJ fence function refers to the maintenance of epithelial cell polarity. The Par3 is a polarity scaffold, which is a part of aPKC-Par3-Par6 polarity complex and plays a crucial role in formation of TJ and cell polarity in various cell types and organisms. 26 The TJ proteins that regulate epithelial polarity also control cell proliferation and differentiation and participate in EMT. EMT in epithelium is characterized by the disruption of epithelial junction complexes and the loss of cell polarity, transforming stationary epithelial cells into migratory mesenchymal fibroblast-like cells. The association between EMT and structural and functional alterations in TJ is well established. 11 In rat lung carcinogenesis, losses of ZO-1 and E-cadherin expression are early events that precede EMT, raising the possibility of a cause-and-effect relationship. 27 TGF-β initiates and maintains EMT, which is associated with reduced expression of ZO-1 and E-cadherin. The most common type of AAN is known to be a progressive renal fibrosis. α-SMA, a phenotypic marker of both dedifferentiation and transdifferentiation of cells, was highly expressed in RTEC of patients with AAN, which indicates that RTEC may undergo transdifferentiation to further initiate chronic disease progression in AAN. 7 In our study, the disassembly of Par3 from cell membrane has been observed as even exposure to a very low dose of AA, indicating the breakdown of cell polarity. We also found that AA-I (25 μM) could induce the expression of α-SMA in cell plasma. Structural and functional alterations in the TJ are important components in the genesis of EMT. The reduction in epithelial TJ protein expression and an augmentation of mesenchymal marker expression after the treatment with AA indicated that AA-I-induced TJ changes may involve in EMT. 28 Spatiotemporal studies indicate that loss of cell–cell adhesion and cell polarity precedes full-scale EMT.24,29
In conclusion, complex changes in the expression of TJ as well as the disruption of the fence function were induced even by low dose of AA-I treatment for short time and happened before the apoptosis and the changes of viability of cells. These findings indicated that aberrant expression of TJ proteins within the TJ of RECs may implicate in initiating the renal tubulointerstitial disorder. However, how could AA-specific modification of the renal epithelial TJ contribute to the adverse effect of nephrotoxicity and how could TJ be regulated by intracellular signaling cascades? Much work is needed to facilitate our conceptual understanding of the mechanistic links of tubulointerstitial fibrosis induced by AA.
Footnotes
This work is supported by the Chinese National Natural Science Research Grant (Grant number 30873432, 81073138).
