Abstract
Introduction
Inflammatory bowel disease (IBD) and ulcerative colitis (UC) generally start in young adulthood and lasts throughout life, as chronic disabling diseases.1,2 The typical symptoms of UC are abdominal pain and diarrhea combined with mucus and blood. 3 Although efforts have been made, the etiology and pathogenesis of UC remain unclear. 4 Some scholars believe that UC is caused by immune dysfunction and environmental factors that affect genetically susceptible people.5,6 In contrast, nitrosative and oxidative stress and intestinal epithelial barrier dysfunction are the main reasons.7,8 Clinical drug treatment for UC largely depends on the corticosteroids, 9 immune-suppressants, and aminosalicylates. 10 Among them, salazosulfapyridine (SASP) and mescaline are the first-line drugs to manage UC.11,12 Although the above medicines have attained excellent achievements in dealing with UC, problems such as high cost and adverse events still exist. Cases of treatment failure due to the development of disease-related complications have also been reported. 13 Thus, searching for new potential alternative medicine with reasonable cost and less side effects may effectively extend our strategies to deal with UC. In recent years, oxymatrine (OMT), a quinolizidine alkaloid extracted from the roots of traditional Chinese herb Sophora flavescens Aiton has received more and more attention on treating various diseases due to its pharmacological activities. 14 For example, OMT reduces expression of programmed death-ligand 1 by promoting DNA demethylation in colorectal cancer cells. 15 OMT exerts a protective effect in myocardial ischemia/reperfusion–induced acute lung injury by inhibiting autophagy in diabetic rats. 16 Interestingly, several studies indicated that the efficacy of OMT (like anti-inflammation, apoptosis promotion, anti-fibrosis, and immune regulation) might promise the therapeutic effect of OMT on UC.17–19 For instance, OMT protects against DSS-induced colitis via inhibiting the PI3K/AKT signaling pathway. 20 However, other molecular mechanisms of OMT on UC are still ambiguous.
The Toll-like receptor (TLR) family is a pattern recognition receptor group that belongs to the pathogen-associated molecular patterns (PAMPs). 21 Generally, TLRs can control innate immunity and critically regulate subsequent adaptive immunity.22,23 Among them, TLR 9 owns an irreplaceable position due to its explicit identification of unmethylated cytidine phosphate guanosine (CPG), which is the main immunostimulatory component in viral and bacterial DNA. 24 Studies have found that TLR9 played an essential role in the immune response in the pathogenesis of UC.25,26 However, as far as the authors know, few studies have concentrated on the impact of OMT on the TLR9 signal pathway in UC. Thus, to better understand the relieving mechanism of OMT on UC, it is necessary to figure out the potential influence of OMT on this signal pathway.
In this article, SASP was chosen as the positive drug of TNBS-induced colitis due to its non-obsolescent therapeutic effect. The potential influence of OMT on TNBS-induced colitis, especially the impact on TLR9 signal pathway and the tight junction protein, was explored by introducing TNBS-induced colitis rat models. By so doing, we hope better to figure out the therapeutic mechanism of OMT on UC.
Materials and methods
Animals
60 Male SD rats (body weight: 190 ± 20g) without any specific pathogen are recruited from the experimental animal centre of Nanjing University (Nanjing, China). They are maintained in pathogen-free conditions and allowed ad libitum access to rodent chow and water. Standard conditions (22°C, 45–55% humidity) and light/dark cycles (12/12 h) are maintained. Before commencing experiments, animals had a right to acclimatize for 2 weeks. Animal experiments are conducted strictly under the guidelines of the Animal Research Committee of Nanjing University of Chinese Medicine. All the animal experiments have attained the approval of the Institutional Animal Care and Use Committee before proceeding. Besides, the experimental animal procedures also complied with the ARRIVE guidelines from NC3Rs and had to be carried out according to the EU Directive 2010/63/EU.
Experimental
The rats are randomly divided into six groups: Control; TNBS; TNBS+OMT(L): TNBS+10 mg/kg OMT; TNBS+OMT(M): TNBS+30 mg/kg OMT; TNBS+OMT(H): TNBS+60 mg/kg OMT; TNBS+SASP., 4% w/v TNBS solution (1 volume of 4% w/v TNBS solution mixed with 1 volume of absolute ethanol) was induced in rats for 7 days to establish the models.27–29 From the first day of the model induction, the OMT groups are administered by an intraperitoneal injection of 10, 30, or 60 mg/kg OMT (A111285, Aladdin, China), respectively, daily for 7 days. Although SASP was commonly used as an oral drug for treating UC, the intraperitoneal injection was selected in this study to ensure the operation condition of SASP was consistent with that of OMT for comparison. Various dosages of OMT are dissolved by PBS and 30 mg/kg SASP was dissolved by DMSO solution. Control and TBNS groups are injected with an equal volume of PBS at the same time.
Evaluation of UC
Disease activity index (DAI) is defined as the summation of the stool consistency (0–3), fecal bleeding (0–3), and weight loss (0–4). 17 All the rats are anesthetized with 5% chloral hydrate (50 mg/kg) and euthanized on the eighth day when the experiment finished. Furthermore, also to ascertain whether the rat’s heart stops beating to verify death or not. Colons are dissected and collected for histological analysis. Part of the colony was fixed and stained with hematoxylin and eosin for histopathological analysis.
Histological assessment of colitis
Colon tissues are harvested and assessed as described previously. 30 In brief, the tissues are fixed in 4% buffered paraformaldehyde, dehydrated in graded ethanol, embedded in paraffin, and then sectioned into 4 µm thick slices. Sections are subsequently stained with hematoxylin and eosin (H&E) and then assessed and scored by a board certified veterinary pathologist blinded to the treatment groups. The severity was graded 1–5 scales as previously described, 31 semi-quantitatively as: grade 1: No infiltration; grade 2: Infiltration in basal layer of crypt; grade 3: Infiltration reaches the mucosal muscle layer; grade 4: Infiltration deep into the mucosal muscle layer, accompanied by mucosal thickening and edema; grade 5: Infiltration to the submucosa. 32 The magnification is 200X.
Immunohistochemistry (IHC) analysis
IHC studies are used to detect the expressions of TLR9, myeloid differentiation primary response protein-88 (Myd88), phosphorylated p nuclear factor κB (p-NF-κB/p-p65) as described previously. 33 Briefly, the sections are washed three times with PBS and endogenous peroxidases are removed by incubation for 10 min at room temperature. Afterward, the sections are incubated with primary antibodies Myd88 (Cat. ab133739, Abcam), TLR9 (Cat. ab37154, Abcam), p-NF-κB (Cat. ab239882, Abcam) overnight at 37°C and then incubated with the secondary Goat Anti-Rabbit IgG H&L (HRP) antibody (Cat. ab6721, Abcam) for 2 h at room temperature. The slides are prepared with DAB and analyzed by Olympus BX43 (X 200).
Western blot assay
The expression of specific proteins in colonic tissues was evaluated by Western blot. The colon samples are homogenized using RIPA lysis buffer equipped with a protease inhibitor cocktail (Roche, Basel, Switzerland), and concentrations of proteins are tested by the BCA protein assay kit (Goodbio Technology, Wuhan, China). SDS-PAGE gels are used to separate the protein extract, then electro-transferred to a polyvinylidene difluoride membrane. After that, the membranes are blocked with 5% nonfat skim milk and incubated subsequently at 4°C for 24 h with appropriate primary antibodies against TLR9 (Cat. ab37154, Abcam), Myd88 (Cat. ab133739, Abcam), NF-κB (p65) and p-NF-κB p65 (Cat. ab239882, Abcam), claudin-2 (Cat. ab53032, Abcam), occludin (Cat. ab216327, Abcam), zonula occluden (ZO)-1 (Cat. Ab276131, Abcam), respectively. All the membranes are washed carefully with TBST before being incubated again with a secondary Anti-Rabbit IgG H&L (HRP) antibody (Cat. ab6721, Abcam) conjugated with peroxidase at 24°C for 1 h. The protein bands of interest are then visualized and analyzed, and GAPDH (Cat. ab181602, Abcam) expression was used as a comparison.
Quantitative real-time PCR (qRT-PCR)
RNA extraction from colonic tissues was performed by using TRIzol reagent. The reverse RNA transcription into cDNA was performed using PrimeScript 1st Strand cDNA synthesis kit (Takara, Otsu, Japan). The real-time quantitative PCR analyses are implemented to evaluate the expression levels of the target gene based on the 2−ΔΔCT method.
ELISA analysis
Enzyme-linked immunosorbent assay (ELISA) kit purchased from R&D Systems (Shanghai, China) was utilized to measure the serum levels of IL-1β, IL-6, TNF-α, IL-10 in the supernatants obtained from abdominal aorta blood. The detailed testing procedure was according to the manufacturer’s protocols.
Statistical analysis
The experiments are replicated three times. The data are presented as the mean values±standard deviation. Statistical analysis was conducted using GraphPad Prism 6 (GraphPad Software, Inc.), and differences between groups are assessed by one-way ANOVA followed by Dunnett’s multiple comparison analysis. p < 0.05 was considered statistically significant.
Results
Effect of OMT on the tissue damage of TNBS-induced colitis
The molecular formula of OMT is C15H24N2O2, and its structure is presented in Figure 1(a). Ulcerative models are created by TNBS to explore the role of OMT in UC rats. As shown in Figure 1(b), rats treated with TNBS suffer a dramatic loss of body weight, compared with that of control rats. Other groups include TNBS+OMT(L), TNBS+OMT(M), TNBS+OMT(H), and TNBS+SASP alleviate this symptom of losing weight, and TNBS+OMT(H)shows the best alleviating effect. TNBS caused by colitis is usually associated with severe reduction of colonic length, as indicated in Figure 1(b), and the reduction degree can reach as high as 50% compared with the controlled one. The TNBS+OMT and TNBS+SASP protect the colony from being downsized. Specifically, minor reduction (about 25%) in colon length is TNBS+OMT(H)group, while the group with TNBS+SASP presented a slightly higher reduction, about 27%. The effect of OMT on the UC rats induced by TNBS. (a) Chemical structure of OMT. (b) Daily changes of body weight. (c) DAI of rats. (d) The length of the colon. Data are presented as means ± SD (n = 8). **p < 0.01, versus control group. ##p < 0.01, versus model group.
The DAI score (Figure 1(c)) of each group shows no significant difference in the first few days, which may result from the slow absorption rate of those drugs at the beginning. After 3 days, the DAI score of the TNBS group displays an exponential increase and hit the highest point of 6 on day 7. Comparatively, the DAI score in TNBS+OMT and TNBS+SASP treated groups decrease linearly at the same period, and TNBS+OMT(H) and TNBS+SASP groups show minor DAI score of 1.6 and 1.7 on day 7, respectively. In a nutshell, the above analysis upheld the therapeutic effect of OMT on UC.
Histopathological analysis of OMT on the TNBS-induced UC rats
The histological score of rats and colonic histopathological changes is shown in Figures 2(a) and (b), respectively. The highest score of the TNBS model group undoubtedly shows the harmfulness of UC, while the scores of groups treated with TNBS+OMT(H) and TNBS+SASP decreased obviously (score 2 and 2.2, respectively). Graphs of colonic histopathological changes further confirmed the efficacy of OMT. It is clearly shown that the introduction of TNBS causes severe damage to the colonic structure, including injury to the mucosa, the formation of ulceration, reduction in goblet cells, and infiltration of inflammatory cells. The injection of high doses of OMT (TNBS+OMT(H))or TNBS+SASP group substantially improve the health state of colonic tissue, which exhibit a more stable structure than that of the TNBS group. Histological analysis of colon samples. (a) Histological score. (b) Amplification, ×200. Data represent means ± SD (n = 8). **p < 0.01, versus control group. ##p < 0.01, versus model group.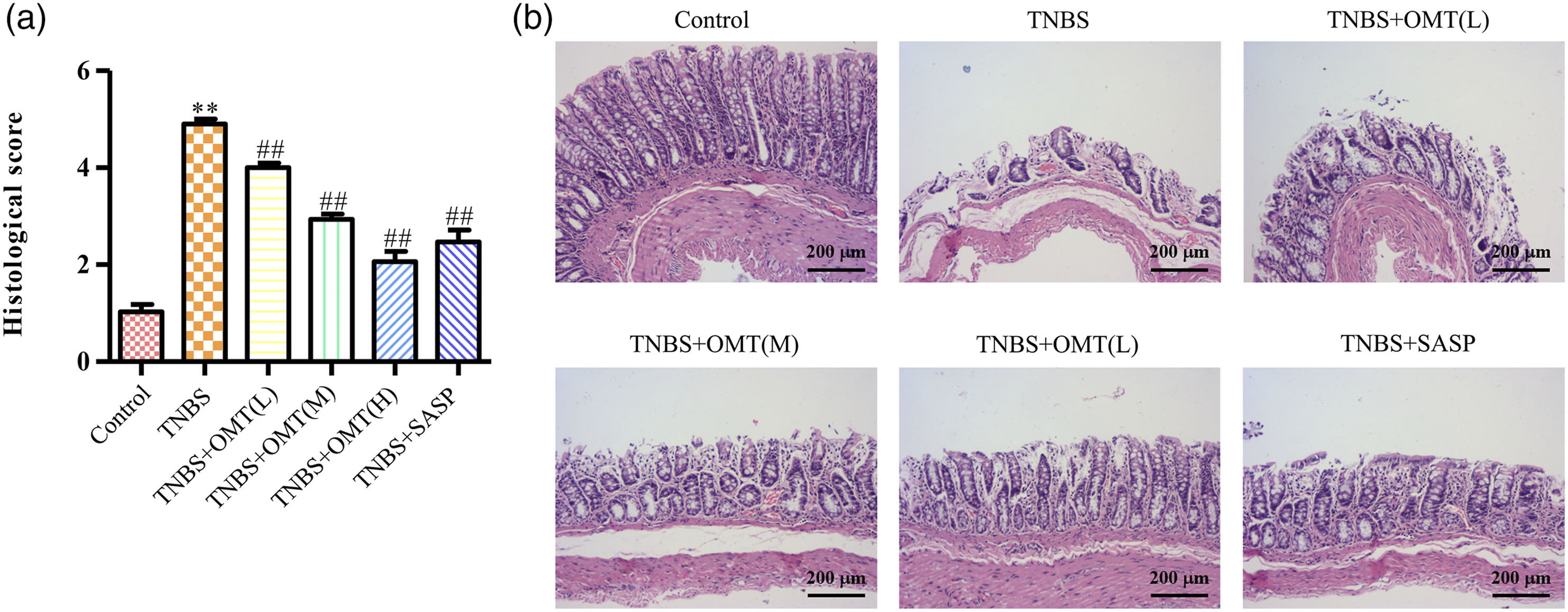
Effect of OMT on the expression of colonic tight junction protein
The intestinal epithelium is a dynamic barrier that is maintaining uniform uptake of nutrients and water. It is the most extensive interface between the host and many bacteria in the lumen. The defect of this barrier has been proved to significantly aggravate experimental colitis, although it is not the essential factor that induced spontaneous colitis. Tight junctions (TJs) like occludin, claudins, and zonula occludens (ZO) are the intestinal epithelial barrier’s essential components, which play essential roles in controlling the permeability and integrity of this barrier. Thus, it is a major interest to investigate the influence of OMT on the expression of those proteins. Figure 3 shows the mRNA and protein expression levels of ZO-1, occludin, and claudin-2 analyzed by qRT-PCR and Western blot. The results clearly showed that TNBS markedly inhibit the mRNA and protein expression levels of ZO-1, occludin, and claudin-2, which could be degraded to as low as 24%, 22%, and 20% of that in the control group, respectively. Intervention of increasing OMT dosage showed an escalating activation effect on those proteins, where the corresponding mRNA and protein expressive levels could be elevated almost equal to the levels of the control group. Our data suggest that OMT could effectively protect the integrity of the intestinal epithelial barrier. OMT regulates the mRNA and protein expression levels of TJ-associated factors. (a) The mRNA and protein levels of ZO-1, occludin, and claudin-2 are detected by qRT-PCR and Western blot. (b) Relative ratio levels are determined by densitometric analysis normalized to GAPDH. Data are presented as means ± SD (n = 8). **p < 0.01, versus control group. ##p < 0.01, versus model group.
Influence of OMT on the secretion of colonic inflammatory factors
The changes of pro-inflammatory cytokines measured by ELISA are presented in Figure 4(a). The highest concentration of IL-6, about 950 pg/mL, was found in the TNBS group relative to the control one. Other groups included TNBS+OMT(L), TNBS+OMT(M), TNBS+OMT(H), and TNBS+SASP significantly lower the level of IL-6, which are lowered to around 450 pg/mL by the TNBS+OMT(H) group. A similar tendency can be found in the protein level of IL-1β, TNF-a, and IL-10, is TNBS+OMT(H) group also shows a solid inhibiting effect on the secretion of those proteins. Influences of OMT on the changes of inflammatory factors. (a) ELISA analysis of IL-1β, IL-6, TNF-a, and IL-10 in the colon. (b) The mRNA expression levels of IL-1β, IL-6, TNF-a, and IL-10 in the colon are quantified. Data are presented as means ± SD (n = 8). **p < 0.01, versus control group. ##p < 0.01, versus model group.
The mRNA level of IL-1β, IL-6, TNF-a, and IL-10 tested by RT-qPCR analysis is shown in Figure 4(b) A much more noticeable repressive effect of TNBS+OMT(H) group on the mRNA level of each protein is discovered, which is almost equal to the level in the control group. These findings indicated that OMT exerted a practical anti-inflammatory effect in the TNBS-induced acute intestinal inflammation rat model.
Effect of OMT on the TLR9/Myd88/NF-κB signal pathway
TLR9 is a vital receptor that participated in the innate immune response and is involved in specific immunity and immune tolerance. TLR9 can precisely identify the unmethylated cytidine phosphate guanosine, the main immunostimulatory component of viral and bacterial DNA. Completed recognition starts the transduction of specific chemical signals and subsequently results in the activation of nuclear factor-κB (NF-κB), which will further promote the release of downstream inflammatory factors, including interleukin, tumor necrosis factor and interferon. In recent years, TLR9/Myd88/NF-κB signal pathway has been found closely linked to the progress of UC. The effects of OMT on the TLR9/Myd88/NF-κB signal pathway are analyzed by immunohistochemistry (Figure 5(a)) and Western blot (Figure 5(b)) analysis. The introduction of TNBS caused a much higher expression level of TLR9, Myd88 and p-NF-κB P65 compared with the control group, which suggested that TNBS was responsible for the activation of TLR9/Myd88/NF-κB. In contrast, both the phosphorylation of NF-κB and the expression level of TLR9 and Myd88 are downgraded with TNBS+OMT(L), TNBS+OMT(M), and TNBS+OMT(H) groups, where TNBS+OMT(H) group exhibits the most substantial repressive effect on the expression of TLR9 and consequently inhibits the release of Myd88 and the phosphorylation of NF-κB. In addition, colonic tissues of rats treated with TNBS+OMT(H) show a much more regular structure relative to that of the TNBS group. The results clarified that OMT could remedy UC by interrupting the signal transduction of the TLR9/Myd88/NF-κB pathway. The effect of OMT on the TLR9 pathway. (a) Expressions of p-NF-κB p65, Myd88, TLR9 in colon sections are assessed by IHC (magnification, ×200). (b) Total and representative protein levels of TLR9, Myd88, p-NF-κB p65, and NF-κB p65 are measured by Western blot. Data are presented as means ± SD (n = 8). **p < 0.01, versus control group. ##p < 0.01, versus model group.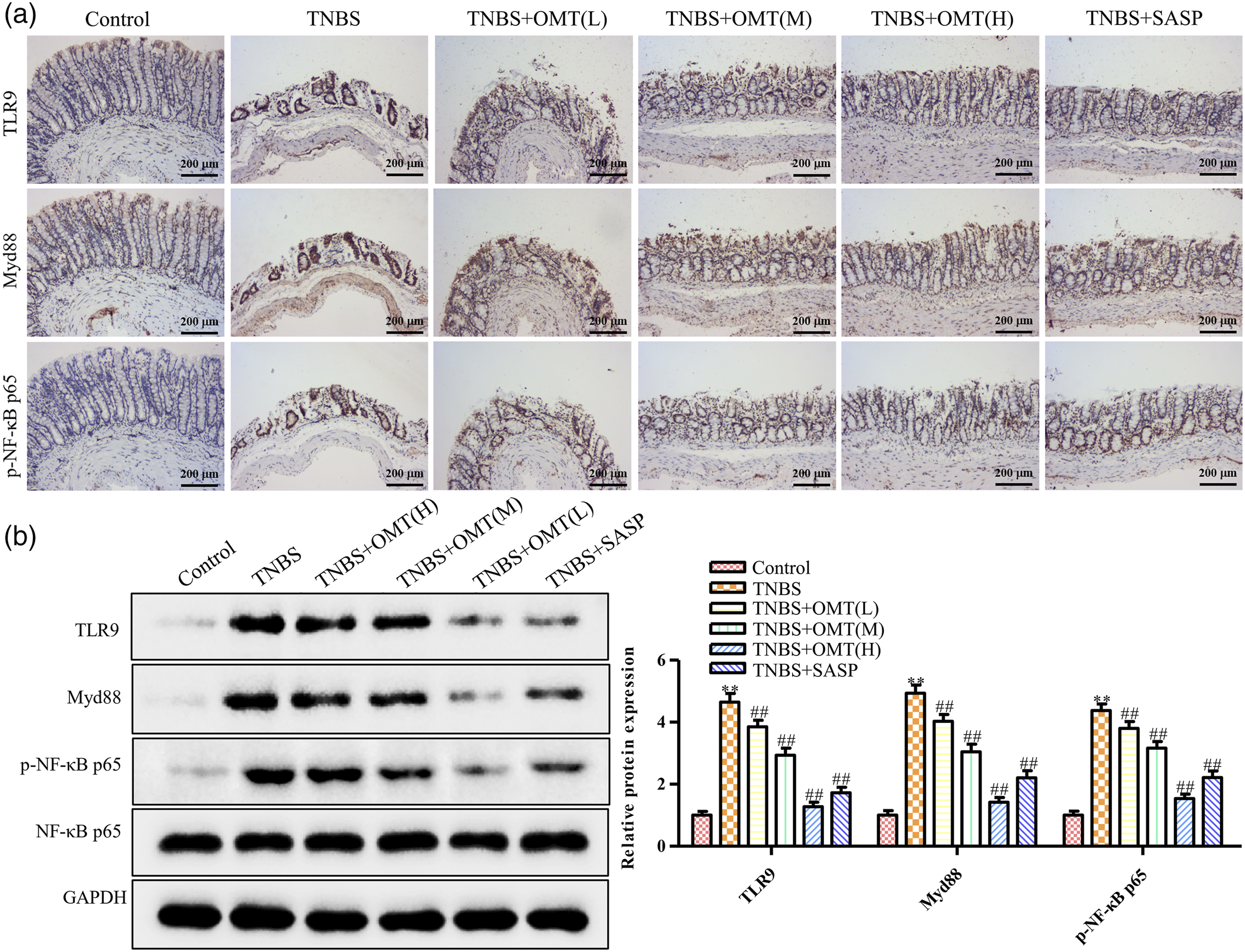
Discussion
As a nonspecific chronic inflammatory bowel disease, UC is characterized by chronic inflammation and ulcerative changes in the intestinal mucosa. 34 Lesions of UC were mainly located in the mucous membrane, submucosa of the colon and rectum. 35 The pathogenic mechanism of UC is still not precise. Some studies considered that the abnormal performance of the immune system is an essential factor in intestinal inflammation and tissue damage caused by UC, while others indicated that it might be related to environmental, genetic, infectious and immune factors.36–38 Clinical treatments for UC are usually accompanied by the use of corticosteroids, immune-suppressants, and aminosalicylates. Some drugs like SASP, mesalamine, and 5-aminosalicylates have been proved their efficacy in treating UC. 39 However, a lot of adverse events caused by those drugs have also been recorded. 13 Other newly developed specific antibodies such as infliximab, adalimumab, and CD54 antagonist ISIS 2302 have an excellent therapeutic effect on UC with fewer side effects,40–42 but the high prices have limited their wide application in clinical treatment. Thus, it is of profound significance to find drugs with few adverse reactions and low cost.
OMT is an alkaloid extracted from plant S.ophora flavescens and has been proved to have multiple pharmacological effects. 43 Studies have been carried out to explore the influence of OMT on UC, where a favorable relieving effect of OMT on UC symptoms has been confirmed. 17 However, the underlying interaction mechanism is not precise. Our study first confirmed that OMT could primarily alleviate the ulcerative colitis caused by TNBS. The weight loss and reduction of colonic length of UC rats are restrained significantly by OMT. Pathomorphological observation showed that OMT reduced the inflammatory injury degree of the colon caused by TNBS in a dose-dependent manner. Those results are in line with other findings.17,18 Second, we try to explore more on the underlying therapeutic mechanism of OMT on UC. The intestinal epithelial barrier is the most critical part of the intestinal barrier, composed of colonic epithelial cells and intercellular connections. The defect of this barrier has been proved to significantly aggravate experimental colitis, although it is not the essential factor that induced spontaneous colitis. 44 From the top of the basement membrane, the intercellular junctions are tight, adhesive junctions, desmosomes, and gap junctions, among which tight junctions are the most important. Tight junctions (TJs) like occludin, claudins, and zonula occludens (ZO) are the essential components of the intestinal epithelial barrier, which plays essential roles in controlling the permeability and integrity of this barrier. Our results showed that the expression of TJ proteins in TNBS-induced colitis in rats was lower than that of the control group, indicating a clear relationship between UC and TJs. The expression of TJs in the OMT intervention group was much higher than that in the TNBS-induced model group, suggesting that OMT can promote the expression of TJ proteins and repaired the tight junction between intestinal epithelial cells, which may be one of the reasons why OMT relieved UC.
The impact of OMT on TLRs has attracted considerable attention. Dai et al. 45 observed that OMT possess anti-IAV and anti-inflammatory activities by inhibiting IAV-induced activations of TLR4, p38 MAPK, and NF-κB pathways. Xu et al. 46 found that OMT effectively attenuates LPS/D-GalN-induced acute liver failure by oxidative damage via activation of Nrf2/HO-1 and modulation of TLR4-dependent inflammatory signaling pathways. However, few works have verified the influence of OMT on TLR9 in UC models. In this study, our results indicated that OMT has an inhibitory effect on TLR9/Myd88/NF-κB pathway activation. Moreover, the expression of inflammatory factors downstream of this pathway was further downregulated. This exciting finding strongly indicated that OMT could relieve colitis by impacting TLR9/Myd88/NF-κB signal pathways, which, to the authors’ knowledge, are rarely addressed in other research work.
The intraperitoneal injection of OMT and SASP was applied in this article. However, in clinical practice, most therapeutic medications such as mesalazine and SASP are administered orally. For further analyzing the efficacy of OMT, oral delivery should be taken into account when treated with UC rat model. In addition, COX2 is an important factor of UC, it is a potentially meaningful downstream molecule of NF-κB, we will explore the molecular mechanisms between them to make a novel treatment strategy of UC more credible and complete.
In summary, OMT reversed the inhibition of TJ protein expression induced by TNBS. Activation of the TLR9/Myd88/NF-κB signal pathway and the expression of its downstream inflammatory factors are inhibited by OMT. These findings demonstrated that OMT could relieve colitis in rat models by impacting TJs’ TLR9/Myd88/NF-κB signal pathways and activity.
Footnotes
Authors’ contributions
Shengwei
Li and Fangshi
Zhu made majority contribution to the conception of this study, Guangqing
Feng and Xing Zhang carried out all of experiments, Jihong Lu prepared the first draft of this article. Fangshi
Zhu agreed the final design of this work and revised this article critically. Shengwei
Li and Chenyahui
Feng performed cell culture and Western blot. All authors have read and approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the The YangZhou’s 13th Five-plan for “KeJiaoQiangWei” (No.ZDRC201826, ZDRC201884).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
