Abstract
Di(2-ethylhexyl) phthalate (DEHP) is a ubiquitous environmental contaminant and a well-known endocrine disruptor (ED) that interferes with the reproductive function in both humans and animals. This study aimed to find out the impact of lactational exposure of DEHP in testes of first filial generation (F1) progeny male rat postnatal day (PND)-60. Lactating dams were orally treated with DEHP (0, 1, 10 and 100 mg/kg body weight/day, respectively) from the PND-1 to PND-21. Rats were killed at PND 60. Testes were removed and used for histological analysis and for isolation of Sertoli cells (SCs). The histoarchitecture of DEHP-treated rats showed disturbed testicular structure. DEHP-treated rats also showed increased oxidative stress by decreasing antioxidant levels in the SCs; it disrupted SC tight junctional proteins occludin, claudin, junctional adhesion molecule, zona occludens protein-1 (ZO-1), zona occludens protein-2 (ZO-2), and afadin-6 (AF-6), increased apoptosis by altering the apoptotic genes Bax, cytochrome c, caspase-8, -9, -3 and antiapoptotic gene Bcl-2. It is concluded that early postnatal exposure to DEHP disturbs histoarchitecture of testis and SC function in pubertal Wistar rats.
Introduction
Endocrine disruptors (EDs) are a highly heterogeneous group of compounds that range from synthetic chemicals and pesticides to natural compounds found in foods which have been shown to impose long-term effects on animal health and development. 1 Phenols and phthalates are among the EDs that have impact on male fertility and several other pathologies are associated with developmental abnormalities. 2 Di(2-ethylhexyl) phthalate (DEHP) and its major metabolite mono(2-ethylhexyl) phthalate (MEHP) are found mainly in polycarbonate plastics, toys, dentist devices, food packaging, blood bags, and cosmetics. 3 Unfortunately, they can leach from the lining of plastic packages, cans and baby bottles, and pipe walls. In this way, the human body is exposed to concentrations of 3–30 μg/kg body weight/day. 4 They have been detected in human body samples such as serum, urine, amniotic fluid of pregnant women, breast milk and even in semen.5,6 Thus, experimental evidences clearly show that humans are exposed to phthalate, which may threaten normal physiology during development and adult life.
The development of rat testis during perinatal period is critical for the establishment of normal function during adult stage and also for the development of accessory male reproductive organs. For example, the final size of the Sertoli cell (SC) population must be appropriate for quantitatively normal spermatogenesis to occur, and the number of SC is determined during the perinatal period when they actively divide. 7 Thus, disruption of SC proliferation during the perinatal period will lead to reduced numbers of SC in the adult and a parallel decrease in sperm production. 8 The structure of SCs and the specialized junctions between them and the neighboring germ cells create a sophisticated microenvironment providing all the nutrients and growth factors required for the full development of spermatogenic cells and the blood testis barrier (BTB) which lies between the adjacent SC protecting the cells from getting exposed to the cytotoxic agents. Ultrastructurally, the BTB is composed of tight junctional proteins which are the integral membrane proteins like occludin, claudin, junctional adhesion molecule (JAM), and cytoplasmic accessory proteins like zona occludens protein-1 (ZO-1), zona occludens protein-2 (ZO-2), and afadin-6 (AF-6). 9 In addition to SC, germ cells also undergo critical development during the neonatal period. Since gonocytes in neonates are coupled to SC via gap junctions, they depend on SC-derived signals for normal function. 10 The maintenance of an appropriate relationship between these cell types in neonates is important for testicular development.
Exposure to environmental chemicals including but not limited to antiandrogens during fetal and childhood development may be partly responsible for the reported deterioration of reproductive health in human males. 11 For example, phthalate esters with alcohol moieties ranging from C4 to C10 are documented testicular toxicants, and it is known that young rats (14–35 days old) are more sensitive to phthalates than the adult. 12 Exposure of rat pups to di(n-butyl) phthalate (DBP) during the perinatal period causes permanent damage in male reproductive organs of the adult. This damage for the DBP at ≥250 mg/kg/day includes decreased testis weight, depletion of the seminiferous epithelium, and damage to androgen-dependent development of accessory male reproductive organs. 13
Exposure to DEHP during critical developmental period results in testicular dysgenesis and reduced production of testosterone. This implies that Leydig cells are also one among the targets. 14 The coculture study revealed that the exposure of neonatal SC and gonocytes to low levels of MEHP inhibits SC proliferation and causes detachment of gonocytes from the underlying SC monolayer. 15
However, the potential effect of DEHP, the parent compound of MEHP, to produce similar effects in vivo remains unexplored and the precise molecular mechanisms underlying any effects in vivo or in vitro have not yet been identified. Major study on the effects of DEHP shows that this compound induces apoptosis in the cells of various organs and perturbs their functional properties. 16 Apoptosis could occur extrinsically via the Fas ligand (FasL) or the tumor necrosis factor α (TNF-α) receptors. 17 On the other hand, oxidative stress resulting due to the imbalance of free radicals over the antioxidative system could also result in the apoptosis initiation. 18
Testes are more prone to oxidative stress as it is a sensitive organ requiring more oxygen.
19
Free radicals and reactive oxygen species (ROS) like hydroxyl radical
It is therefore, hypothesized that the dose-dependent DEHP exposure during lactation period may affect SC function. This study aims to analyze the lactational exposure of DEHP on testicular architecture and expression of tight junctional and apoptotic proteins in SC of F1 pubertal rats.
Materials and methods
Chemicals
DEHP or dioctyl phthalate with ≥99.5% purity (D201154; CAS no. 117-81-7; 500 ml) was purchased from Sigma–Aldrich (St Louis, Missouri, USA). All other chemicals and reagents used in this study were of analytical grade (AR), and were purchased from Sigma–Aldrich, Amersham Biosciences Ltd. (UK), and Sisco Research Laboratories (Mumbai, Maharashtra, India). Antibodies were purchased from Santa Cruz Biotechnology Inc. (Santa Cruz, California, USA). β-Actin antibody was purchased from Sigma–Aldrich.
Animals
Animals were maintained as per the national guidelines and protocols approved by the Institutional Animal Ethical Committee (IAEC No. 01/01/10). Healthy adult female albino rats of Wistar strain (Rattus norvegicus), weighing 100–120 g, were used in this study. Animals were housed in polypropylene cages under specific humidity (65 ± 5%) and temperature (21° ± 2°C) with constant 12-h light/12-h dark schedule. They were fed with standard rat-pelleted diet (Lipton India, Mumbai, Maharashtra, India), and clean drinking water (reverse osmosis) was made available ad libitum.
Dose selection and treatment
Male rats were exposed to DEHP by maternal gavage during lactation. Nulliparous rats were mated, and the presence of sperm was taken as gestation day 0 (GD 0), and the dams was separated from male and maintained in individual cages in all cases. After the parturition, dams were divided into four groups, at postnatal day (PND)-1, the pups were culled to six numbers per each dam (four dams/group) and balanced for sex to the extent possible. The pups were culled according to Organisation for Economic Co-operation and Development guidelines. The lactating dams were administered DEHP in olive oil by gavage at dose of 0 (vehicle), 1, 10, and 100 mg/kg body weight/day from PND-1 to PND-21. The solutions were prepared fresh daily according to the weights of the dams. The dosing volume was 0.2 ml in all groups. The rats in the vehicle control group received olive oil in equal amounts as in experimental groups. The male rats were allowed to grow up until pubertal (PND-60) stage. The doses and duration of DEHP exposure were selected based on the study by Akingbemi et al. 21 in which pregnant rats were gavaged with 1, 10, 100 mg/kg/day DEHP from GDs 12 to 21 and lactation days PND-1–PND-21. In their study, serum testosterone and leutinizing hormone levels were significantly reduced in male offspring, compared with the control in a dose-dependent manner. The no observed effect level and lowest observed effect level were determined to be 1 mg/kg/day and 10 mg/kg/day, respectively, and previous study by Akingbemi et al. 22 reported that in utero exposures to DEHP (100 mg/kg) alone caused significant decreases of serum testosterone levels postnatally. At necropsy, the animals were weighed and killed under ether anesthesia followed by decapitation; trunk blood was collected in tubes and centrifuged at 3000g for 10 min at 4°C and stored at −80°C for the estimation of serum hormones and tissues (testes, epididymis, ventral prostate, and seminal vesicle) were excised immediately. The tissues were dissected and weighed in order to calculate the organ/body weight ratios for each animal. The organ weight was considered as absolute organ weight, whereas organ/body weight ratio was considered as relative organ weight. One testis from each rat was fixed in 10% formalin for histopathological evaluation, and the other one was used for isolation of SCs.
Histological studies
The testes of rats were fixed in 10% formalin for 2 weeks. Then, the testes were sliced into 0.5 cm3, further immersion in 10% formalin overnight at room temperature. Following fixation, testes were placed in 70% ethanol, the tissue was maintained at 37°C until routinely processed and embedded in paraffin. The paraffin blocks were cut into 7 µm thickness using rotary microtome. The sections were stained with hematoxylin and eosin and were analyzed by Nikon microscope Eclipse 80i (10×, 20×, 40× magnifications, Japan).
Isolation and purification of SCs
The SCs were isolated from rat testes based on the procedure described by Majumdar et al. 23 Briefly, testes are decapsulated and suspended in Dulbecco’s modified Eagle’s medium (DMEM) for 5 min. The seminiferous tubules are broken into smaller fragments and subjected to collagenase IV digestion (3 mg/30 ml DMEM) for 3 min at 37°C in a shaking water bath at 150–160 oscillations/min. The supernatant is separated and the dispersed tubules are washed twice with fresh DMEM. The dispersed tubules are shaken slightly keeping on ice and allowed to settle at unit gravity. The tubules are then treated with prewarmed collagenase solution for further digestion for 15 min. The peritubular clusters are removed using forceps, and the supernatant is collected and centrifuged at 500 r/min for 5 min at 4°C. The pellet obtained is washed twice with fresh DMEM and observed under the microscope for the presence of SCs. In order to check its purity, the isolated SCs are subjected to oil red O staining. Purity of SCs was greater than 90% on the basis of positive staining for oil red O. There was minor contamination by spermatogonia (5–7%), spermatocytes (2–4%), and peritubular myoid cells (1%). The viability of purified SCs was 90–95% as assessed by trypan blue exclusion.
Biochemical analysis
In brief, isolated SCs were sonicated in ice-cold tris(hydroxymethyl)aminomethane–hydrochloric acid buffer (pH 7.2), centrifuged at 16,000g for 15 min at 4°C, and the supernatant was used for measuring the following biochemical parameters.
Oxidative stress markers
Estimation of LPO
Protein content was determined by the method of Lowry et al. 24 LPO was assayed spectrophotometrically as described by Devasagayam and Tarachand. 25 The results were expressed as nmoles of malondialdehyde (MDA) formed/mg protein.
Estimation of H2O2
H2O2 generation was assayed by the spectrophotometric method of Pick and Keisari. 26 Levels of H2O2 generation were expressed as nanomoles of H2O2 generated per minute per milligram protein.
Estimation of hydroxyl radicals
Hydroxyl radical
Antioxidant enzymes assay
Determination of SOD
The activity of superoxide dismutase (SOD) was assayed according to the method of Marklund and Marklund. 28 The enzyme activity is expressed as units per milligram protein.
Determination of CAT
The activity of catalase (CAT) was assayed spectrophotometrically according to the method of Sinha. 29 The activity of CAT is expressed as units per milligram protein (one unit is the amount of enzyme that utilizes 1 mmol H2O2 per minute).
Determination of GPx
The activity of glutathione peroxidase (GPx) was determined as described by the method of Rotruck et al. 30 The enzyme activity was expressed as units per milligram protein (one unit is the amount of enzyme that converts 1 mmol reduced glutathione (GSH) to glutathione disulfide (GSSG) in the presence of H2O2 per minute).
Determination of GR
The activity of glutathione reductase (GR) was assayed spectrophotometrically as described by Staal et al. 31 The activity of GR was expressed as micromolar of nicotinamide adenine dinucleotide phosphate oxidized per minute per milligram protein.
Determination of GST
The activity of glutathione-S-transferase (GST), a phase II enzyme with antioxidant potential, was assayed through the conjugation of GSH with 1-chloro-2,4-dinitrobenzene (CDNB) at 340 nm as described by Habig et al. 32 The activity of GST was expressed as units per milligram protein (one unit is the amount of enzyme that conjugates 1 nmol CDNB with GSH per minute).
RNA preparation
Total RNA from the isolated SC (1 × 106) was extracted using TRI reagent following the method of Chomczynski and Sacchi. 33 One microgram of total RNA was subjected to a two-step reverse transcription polymerase chain reaction (RT-PCR). First strand reaction: complementary DNA (cDNA) was made from messenger RNA (mRNA) template using deoxyribonucleotides and reverse transcriptase (Superscript-III Reverse Transcriptase, Invitrogen, Carlsbad, California, USA). The components were combined with a DNA primer in a reverse transcriptase buffer for 1 h at 37°C. Second strand reaction: after the RT-PCR was complete, cDNA has been generated from the original single-strand mRNA, standard PCR was initiated. PCR Ready Mix DNA polymerase was purchased from KAPA-Biosystem (Wilmington, Massachusetts, USA). PCR primers and conditions used in this study have been tabulated (see Table 1). RNA purity was checked by spectrophotometer. The total RNA was amplified, and the amplified products were separated by electrophoresis on 2% agarose gel and identified by ethidium bromide staining. Specificity was confirmed by the size of the amplified products with reference of 100 bp DNA ladder (Xcleris Genomics, Bangalore, Karnataka, India), and the band intensities were quantified by Quantity One Software, Bio-Rad (Berkeley, California, USA).
IPCR primers and conditions used in this study.
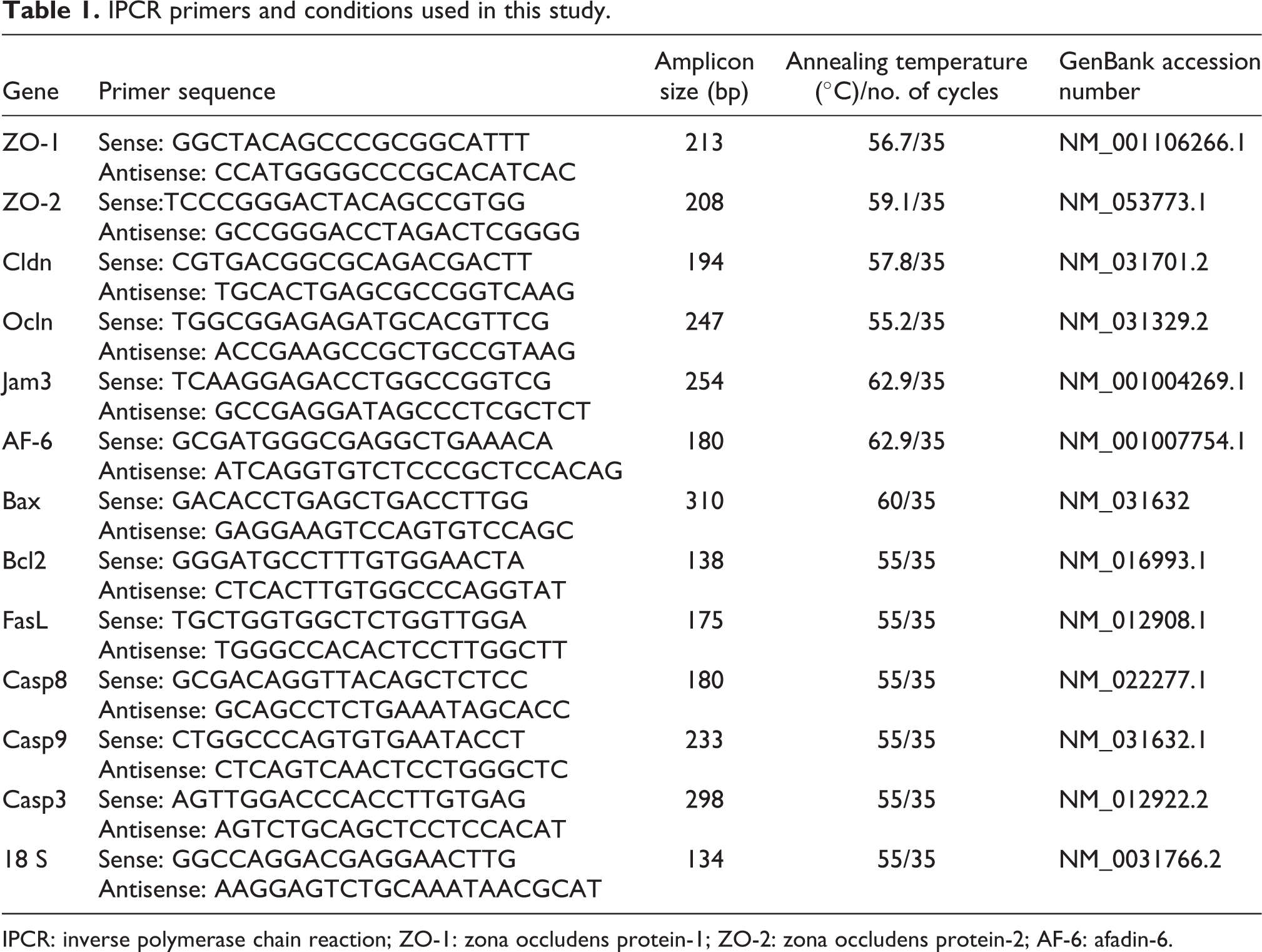
IPCR: inverse polymerase chain reaction; ZO-1: zona occludens protein-1; ZO-2: zona occludens protein-2; AF-6: afadin-6.
Immunoblot analysis
The SC (1 × 106) was lysed with radioimmunoprecipitation assay buffer with protease inhibitor cocktails. Proteins were quantified and then equal amounts of proteins were subjected to 10–12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE). Following electrophoresis, separated proteins on SDS-PAGE gels were transferred to polyvinylidene difluoride membrane (Millipore, Billerica, Massachusetts, USA). To block the nonspecific binding, the membranes were incubated with 5% skimmed milk for 2 h. Membranes were immunoblotted with primary antibodies occludin, claudin, Bax, Bcl2, cytochrome c, caspases-8, -9, and -3 (1:1000; Cell Signaling Technology, Beverly, Massachusetts, USA). The membranes were washed with phosphate-buffered saline (PBS) and incubated with horseradish peroxidase (HRP)-labeled antimouse Rabbit immunoglobulin G (IgG) or anti-rabbit mouse IgG antibody at a dilution of 1:10,000. The bands were developed by enhanced chemiluminescence (ECL) kit (Millipore) in chemi doc image scanner. The band intensity was quantified using Quantity one software (Bio-Rad). The membranes were stripped and reprobed for β-actin (1:5000) and HRP-conjugated secondary antibody (1:10,000) as an internal control.
Apoptosis detection by PI staining
The cells (SCs; 200–300 cells/sample) were placed on 12-mm coverslips. The monolayer of cells was washed with PBS and fixed with 3% paraformaldehyde for 10 min at room temperature. The fixed cells were permeabilized with 0.2% Triton X-100 in PBS for 10 min at room temperature and incubated with 0.5 μg/ml of propidium iodide (PI) for 5 min. The apoptotic nuclei (intensely stained, fragmented nuclei, and condensed chromatin) were viewed under a fluorescent microscope (Nikon) with an excitation at 359 nm and emission at 461 nm wavelengths.
Statistical analysis
The data were subjected to statistical analysis using one-way analysis of variance (ANOVA) followed by Student’s Newman Keul’s and Tukey’s multiple comparison posttest to assess the significance of individual variations between the control and treatment groups using a computer-based software (SPSS 7.5). GraphPad Prism 5 was used for statistical analyses and graphics (GraphPad Software, Inc., La Jolla, California, USA); the significance was considered at the level of p < 0.05.
Results
Gross morphology and histopathological analysis
Figure 1(a) shows marked dose-dependent decrease in gross testis size. Histoarchitecture of testis showed distinct dose-dependent changes in their appearance when compared with control (Figure 1(b)). In control rats, the testis section shows normal seminiferous tubular epithelium with active spermatogenesis. The seminiferous tubules are with intact interstitium and intact SC anchorage and tubular lumen are filled with spermatozoa (Figure 1(b)). In 1 mg/kg body weight/day treatment group, the gross testicular section showed milled damage in seminiferous tubules; however, most of the tubules showed active spermatogenesis. Milder epithelial sloughing was seen (Figure 1(b)). In the testis section of 10 mg/kg body weight/day treatment group shows epithelial sloughing, damaged tubules at periphery and testicular lumen (L) occupied by detached spermatogenic cells from SC junctions which increased thickening of basement membrane were observed (Figure 1(b)). Vacuolization of SCs was seen at certain regions. In 100 mg/kg body weight/day treatment group, the gross testicular morphology was reduced. Damaged seminiferous tubules were observed with vacuolization (Figure 1(b)).

Effect of lactational exposure of di (2-ethylhexyl) phthalate on morphology (a), histology (×10, ×20, ×40) (b) of testicular sections—(PND-60) and propidium iodide staining of SC (20×) (c) of pubertal Wistar rats. Results were confirmed by three independent observations, representative images are included here (×10, ×20—100 µm; ×40—50 µm). Gross morphology of testis showed marked dose-dependent changes in their appearance when compared with control (a). Histoarchitecture of testis shows apparent dose–response effect such as shrinkage of the seminiferous tubules (*), vacuolization of SC, some tubules failed to show a distinct lumen (L). Lumen occupied by detached spermatogenic cells known as sloughing of germ cells to the lumen (L), damaged tubules at periphery and increased thickening of basement membrane is also observed when compared with the control (b). Propidium iodide staining of SC (c) shows the apoptotic nuclei (intensely stained, fragmented nuclei, and condensed chromatin) in the 10 and 100 mg groups compared with the control. (d) Effect of lactational exposure of DEHP on body weight-PND-60. Each bar represents the mean ± SEM of three observations representing six animals. Significance at p < 0.05. (a) Compared with control. (e) Effect of lactational exposure of DEHP on testes and accessory sex organ relative weight-PND-60. Each bar represents the mean ± SEM of three observations representing six animals. Significance at p < 0.05. (a) Compared with control; (b) compared with 1 mg DEHP. DEHP: di(2-ethylhexyl) phthalate; PND: postnatal day; SC: Sertoli cell.
PI staining of SCs
Figure 1(c) data provides PI staining of SCs which coherently resulted in the increase apoptotic nuclei (intensely stained) in the 10 and 100 mg/kg body weight/day groups compared with the control.
Body weight and accessory sex organ weight
Figure 1(d) depicts the effect of DEHP on animal body weight. A significant decrease in body weight was observed in 1, 10, and 100 mg/kg body weight/day DEHP treatment group. No significant change was observed between DEHP-treated groups. Figure 1(d) shows the effect of DEHP on testes and accessory sex organ weight. Dose-dependent decrease in relative organ weight was observed in testes, epididymis shows decreased relative organ weight in 10 and 100 mg DEHP treatment group, whereas in ventral prostate and seminal vesicle only 100 mg DEHP treatment shows decrease in relative organ weigh when compared with the control at PND-60.
mRNA and protein expression of junction proteins
To investigate the DEHP-mediated disruption of junctional proteins, we have seen the transcriptional expression of tight junctional proteins—the integral membrane proteins include occludin (ocldn), claudin (cldn), JAM (Figure 2(a), (b), and (c), respectively), and cytoplasmic accessory proteins such as ZO-1, ZO-2, and AF-6, (Figure 3(a), (b), and (c), respectively). We observed significantly decreased mRNA expression of both integral membrane proteins and cytoplasmic accessory proteins in 1, 10, and 100 mg/kg body weight/day compared with the control. The protein expression of occludin and claudin (Figure 2(d)) were also decreased in a dose-dependent manner.
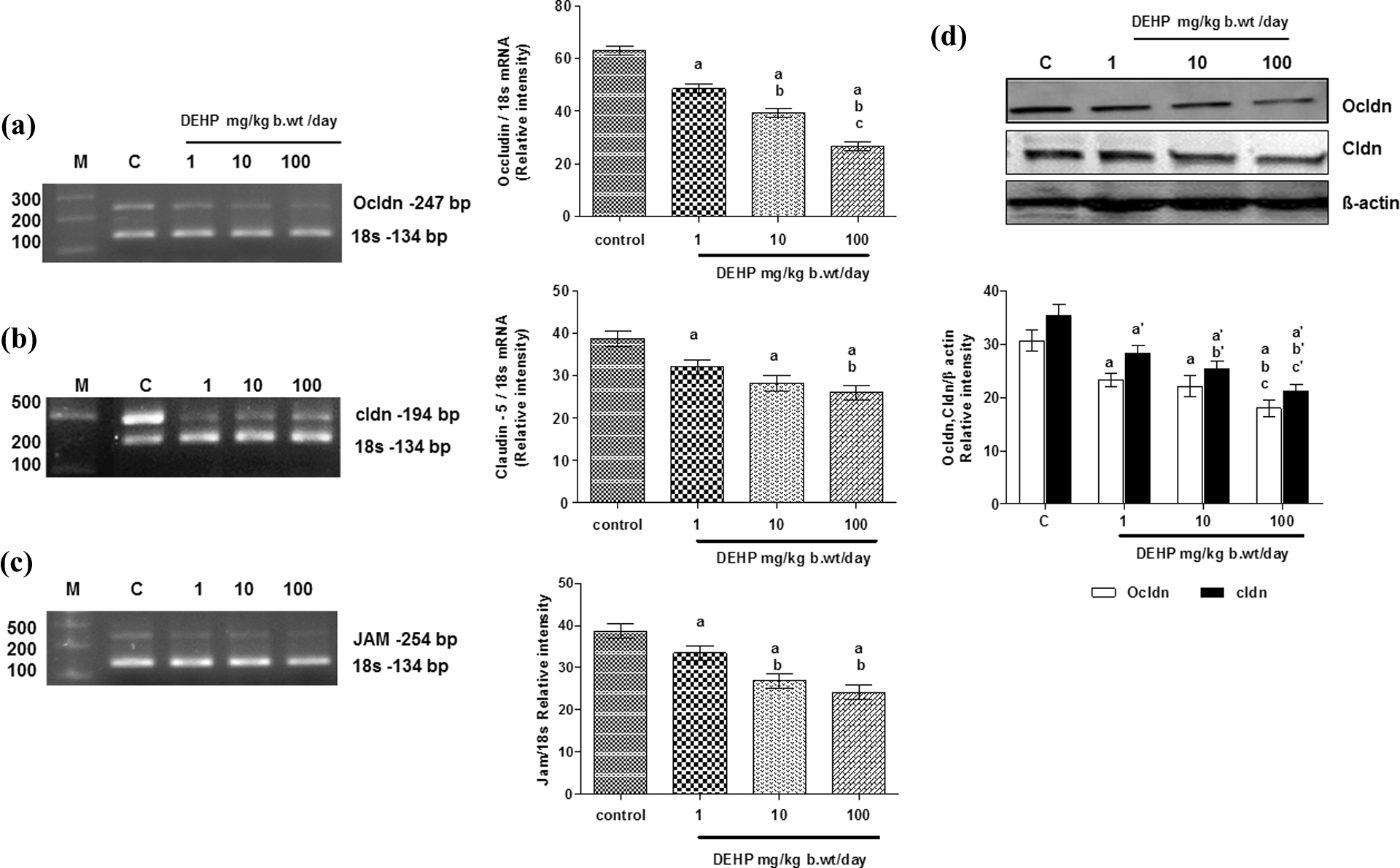
Effect of lactational exposure of DEHP on mRNA (a–c) and protein (d) expression of integral membrane proteins occludin, claudin, and JAM in the SC of pubertal Wistar rats. Each bar represents the mean ± SEM of three independent observations. Significance at p < 0.05. (a) Compared with control; (b) compared with 1 mg DEHP; (c) compared with 10 mg DEHP. DEHP: di(2-ethylhexyl) phthalate; JAM: junction adhesion molecule; SC: Sertoli cell.
Oxidative stress markers
Considering the results obtained from the biochemical analysis, Table 2 compares the levels of LPO and ROS, such as
Effect of lactational exposure of DEHP on oxidative stress markers H2O2, •OH, LPO and antioxidant enzymes SOD, CAT, GPx, GST, and GR in the SC of pubertal Wistar rats.a

DEHP: di(2-ethylhexyl) phthalate; MDA: malondialdehyde; NADPH: nicotinamide adenine dinucleotide phosphate; CDNB: 1-chloro-2,4-dinitrobenzene; GSH: glutathione; SOD: superoxide dismutase; CAT: catalase; GPx: glutathione peroxidase; GST: glutathione-S-transferase; GR: glutathione reductase; SC: Sertoli cell; LPO: lipid peroxidation; H2O2: hydrogen peroxide.
aEach value represents mean ± SEM of six animals. Statistical significance at p < 0.05.
bCompared with control.
cCompared with 1 mg DEHP.
dCompared with 10 mg DEHP.
Antioxidant enzymes
Table 2 provides data on cellular levels of antioxidant enzymes, such as SOD, CAT, GPx, GR, and GST in the SCs of DEHP-treated rats. In our results, the antioxidant enzymes, such as SOD, CAT, GPx, GR, and GST showed a dose-dependent decrease in the antioxidant enzyme activity when compared with the control group.
mRNA and protein expression of apoptotic and antiapoptotic genes
The mRNA expression of apoptotic and antiapoptotic genes were recorded similar to the genes of tight junctional proteins. The protein involved in the extrinsically mediated apoptotic pathway, FasL (Figure 4(a)) was observed for its transcriptional expression; it showed dose-dependent increase in DEHP-treated group when compared with the control which showed the basal state expression. Bax, the apoptotic protein also showed dose-dependent increase in DEHP-treated group when compared with the control (Figure 4(b)). The antiapoptotic gene, Bcl-2 (Figure 4(c)) showed decreased expression in the 1, 10, and 100 mg/kg body weight/day DEHP-treated group when compared with the control which support the data obtained for the apoptotic genes. The protein expression data of Bax and cytochrome c (Figure 4(d)) showed a significant dose-dependent increase in 1, 10, and 100 mg/kg body weight/day DEHP-treated group when compared with the control group. Whereas in contrast to the results obtained for the apoptosis promoting molecules, the antiapoptotic molecule, Bcl-2 (Figure 4(d)) showed a significant decrease in 1 and 100 mg/kg body weight/day DEHP-treated group when compared with the control. The immediate downstream protein of the apoptotic pathway, caspase-8, -9, and -3 showed dose-dependent increase in mRNA (Figure 5(a), (b), and (c), respectively) as well as protein expression (Figure 5(d)) in DEHP-treated groups when compared with the control.

Effect of lactational exposure of DEHP on mRNA (a–c) expression of cytoplasmic accessory proteins (ZO-1, ZO-2, and AF-6) in the SC of pubertal Wistar rats. Each bar represents the mean ± SEM of three independent observations. Significance at p < 0.05. (a) Compared with control; (b) compared with 1 mg DEHP. DEHP: di(2-ethylhexyl) phthalate; SC: Sertoli cell; ZO-1: zona occludens protein-1; ZO-2: zona occludens protein-2; AF-6: afadin-6; mRNA: messenger RNA.

Effect of lactational exposure of DEHP on mRNA expression of Fas (a), mRNA expressions of Bax, Bcl-2 (b, c), and protein expression of Bax, Bcl-2, Cyt-C (d) in the SC of pubertal Wistar rats. Each bar represents the mean ± SEM of three independent observations. Significance at p < 0.05. (a) Compared with control; (b) compared with 1 mg DEHP; (c) compared with 10 mg DEHP. DEHP: di(2-ethylhexyl) phthalate; SC: Sertoli cell; mRNA: messenger RNA.

Effect of lactational exposure of DEHP on mRNA and protein expressions of caspases-8, -9, and -3 (a–d) in the SC of pubertal Wistar rats. Each bar represents the mean ± SEM of three independent observations. Significance at p < 0.05. (a) Compared with control; (b) compared with 1 mg DEHP; (c) compared with 10 mg DEHP. DEHP: di(2-ethylhexyl) phthalate; SC: Sertoli cell.
Discussion
The molecular mechanism of DEHP toxicity has been attributed to the toxicological properties of its metabolite MEHP, formed by cytochrome P450 oxidation. There is a general agreement that male reproductive organs are particularly susceptible to the deleterious effects of ROS and LPO which ultimately lead to impaired fertility.34,35 In utero exposure to DEHP exerts both short-term and long-lasting suppressive effects on testosterone production in the rat. 36 Recently, it was stated that MEHP primary metabolite of DEHP affects the steroidogenesis in rat Leydig cells by provoking ROS perturbation. 37
Testes are vulnerable to oxidative damage. Phthalates induces oxidative stress and decreases activities of antioxidant enzymes and disrupts the junctional parameters in testicular Leydig cells and SCs.
38
The results of the biochemical analysis in this study showed an increased level of the oxidative stress markers like LPO, H2O2 consumption and
The findings also point out the disturbed gene expression at the pubertal stage of the male rats, leading to the disordered expression of the tight junctional and the apoptosis-related proteins. This disequilibrium of gene and protein products results in various deleterious morphological changes (Figure 6).

Schematic representation of effect of lactational exposure of DEHP on F1 pubertal rat testis. Schematic diagram depicts that DEHP acts probably through its primary metabolite MEHP, induces extrinsic- and intrinsic-mediated apoptosis, and disrupts tight junctional protein expression in SC of F1 progeny rats. DEHP: di(2-ethylhexyl) phthalate; MEHP: mono (2-ethylhexyl) phthalate; SC: Sertoli cell; F1: first filial generation.
DEHP is a reproductive toxicant that results in cryptorchidism, a testicular atrophy. 13 Oral administration of DEHP induces testicular toxicity characterized with detachment and sloughing of spermatogenic cells and atrophy in seminiferous tubules. 19 Recent studies have evidenced that MEHP treatment resulted in modification of expression of occludin and the appearance of gaps between SC. 39 In this study, histoarchitecture of testis showed sloughing of epithelial (germ) cells and detached spermatogenic cells from SC junctions, which are very much in correlation with the dose-dependent decreased gene and protein expression data of the occludin and claudin. These proteins are the integral membrane proteins responsible for the paracellular barrier with claudin providing the stability to these junctional strands. 40 The low expression of these proteins also led to the vacuolization of SC, damaged seminiferous tubules along with the basement membrane lined by myoid cells. Further, JAM, present in SC and round elongated spermatids, 41 was also found to show a decreased expression in the DEHP-treated group in comparison with the control explaining the morphological detachment of the germ cells into the lumen.
ZO-1, ZO-2, and AF-6 are the cytoplasmic accessory proteins which act as a scaffolding complex; assisting the integral membrane proteins to anchor with the actin cytoskeleton in the cytoplasm of the SC. 42 ZO-1 functions in supporting the barrier permeability. Hence, any alteration in its expression could disrupt the permeability. This study has established that a dose-dependent decrease in the gene expression of these molecules could impair the SC anchoring function in F1 male rats.
Recent literature suggests that ROS can induce caspase family members that play an important role in spermatogenesis and apoptosis. 43 However, the perturbation and imbalance in the apoptotic and mitotic events lead to abnormalities. Oral administration of DEHP increases ROS in testes, and it has been observed to selectively promote apoptosis in spermatocytes. 19
This sequence of events culminating in the activation of caspases has been broadly categorized into two pathways: the extrinsic pathway characterized by the engagement of cell surface death receptors (DRS). Caspases-8, -9, and -10 are the initiators in extrinsic pathway and they share the same homology with dead effector domains 44 and the intrinsic pathway involving key mitochondrial events which involves anti-apoptotic protein Bcl2, Bcl-xL, and proapoptotic protein Bax, Bad and bid; 45 thus regulating the release of cytochrome c from mitochondria to cytosol. In both cases, an initiator caspase activates itself via its interaction with a specialized adaptor molecule; ultimately activating the downstream effectors or executioner caspases. It is the activity of these caspases that ensure destruction of the cell.
Fas receptors (DRS) are the cell surface receptors that transmit apoptotic signals initiated by specific ligand FasL, which activates caspase cascade.44,45 MEHP disturbs the quality of the germ cell anchoring to SC and germ cell apoptosis involving the Fas/FasL pathway. 46 Decline in germ cells, apoptosis was observed after MEHP treatment in FasL-deficient mice 47 substantiating the extrinsic-mediated apoptosis triggered by MEHP. Germ cell and spermatocyte apoptotic mechanism takes place through the Fas signaling pathway which is initiated after MEHP induces SC injury, with SC expressing the FasL and germ cells expressing its receptor, Fas. 48 In this study, DEHP increased FasL gene expression in the SC of pubertal rats. This upregulation could result in Fas/FasL-mediated induction of apoptosis in SC. The ligation of FasL to Fas triggers apoptotic death in a target cell leading to the activation of caspase-3.
Bcl-2 is an antiapoptotic factor that heterodimerizes with Bax and neutralizes the effects of the latter. When Bcl-2 is present in excess, cells are protected against apoptosis. When Bax is in excess and the homodimers of Bax dominates, cells are susceptible to programmed cell death. Therefore, it is the ratio of Bax to Bcl-2 which determines the fate of a cell. 45 The release of cytochrome c from mitochondria is regulated by the Bax to Bcl-2 family of proteins upstream of caspase activation. DEHP has been shown to generate ROS and selectively induce cytochrome P450Scc, a possible source of ROS. 19 The cytochrome c that is released from the intermembrane space of mitochondria is another responsible element to form complexes that mediated the intrinsic pathway of apoptosis.49,50 In this study, the protein expression of cytochrome c showed a significant increase in a dose-dependent manner. Bax gene expression also showed a dose-dependent increase. Whereas Bcl-2, an antiapoptotic biomolecule showed downregulation of gene and protein expression in SCs of 1, 10, 100 mg/kg body weight/day DEHP-treated group.
In addition, the upregulation of caspase-8 and -9 gene and protein expression in SCs of DEHP-treated animals confirmed induction of both extrinsic- and intrinsic-mediated apoptosis. Caspase-3 is a potent effector caspase which is triggered via several different pathways in a variety of mammalian cell types and is one of the most important caspases activated downstream of cytochrome c in the cytochrome-dependent apoptotic pathways. 51 The increase in gene and protein expression of caspase-3 protein in this study further supports apoptosis in the SC of DEHP-treated rats. Yet the study of its activity and levels of cleaved Poly (ADP-ribose) polymerase protein are warranted as it is involved in the downstream activation of genes responsible in cholesterol uptake, transport and normal physiology. 52 To confirm the data with respect to apoptosis occurrence in the SC, the PI staining established the nuclei fragments, the morphological event found at the time of cell death. These nuclear fragments were found to be more in the 10 and 100 mg/kg body weight/day DEHP-treated groups suggesting favorably to the evidences stated above.
Conclusion
The results of this study showed that lactational exposure of DEHP causes dose-dependent changes in testicular SC of male offspring through ROS-induced apoptosis and perturbation of the tight junctional proteins. Further to all the work recorded, molecular findings pertaining to gene expression regulation are needed to establish DEHP induced long-lasting male reproductive anomalies.
Footnotes
Conflict of interest
The authors declared no conflicts of interest.
Funding
Financial assistance from UGC under SAP-DRS-III program, UGC-BSR-RFSMS, New Delhi, India, is gratefully acknowledged.
