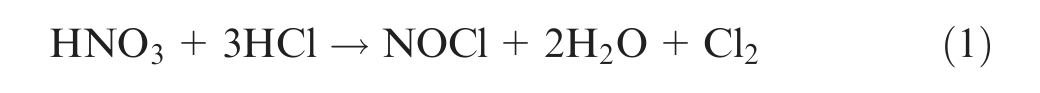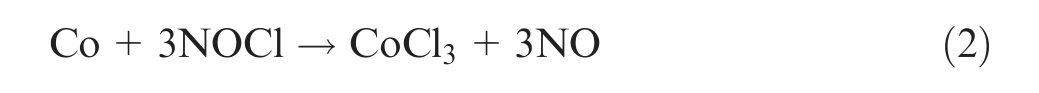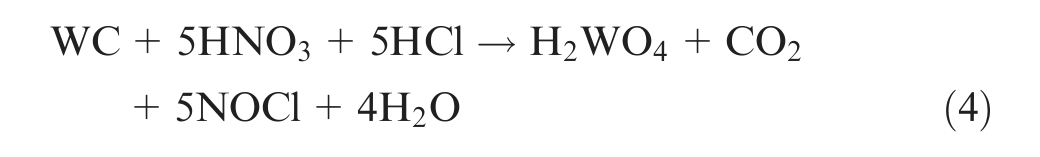Abstract
Due to their excellent hardness and toughness, diamond tools have been widely used as cutting tools for mining applications such as machining and/or processing of natural stones. A considerable amount of used sawblades with diamond grits are damaged, scrapped and wasted in the regions where machining and/or processing of ornamental stones are heavily conducted. Since a damaged and/or scrapped sawblade could still have diamonds grits, the recovery of diamond grits from sawblades has recently gained attention. The aim of this study was to investigate the recoverability of diamonds from damaged sawblades using hydrometallurgical processes, including aqua regia. Using scanning electron microscopy micrographs, the worn surfaces of diamonds were also examined before and after leaching. The results of the study represented that the diamonds from damaged sawblades can be successfully recovered by hydrometallurgical processes, including aqua regia, and the recovered diamonds could be reused in cutting tools after size classification. The results also revealed that among the damage types of diamond, the fractured and polished crystals resulted in diamond crystals with lower grain size.
Introduction
Due to diamond’s extreme hardness, diamond cutting tools govern cutting, grinding, drilling and polishing processes for hard materials and similar fields of application.1,2 Circular diamond sawblades containing diamond grits are one of the well-known tools used for stone cutting. A sawblade is basically composed of the steel disk and the diamond metal matrix segments. The diamond particles are normally embedded in the metal matrix by means of a combination of chemical and physical interactions. Then, the segments are attached to the steel disk by brazing or laser welding. 3 A typical segment contains cobalt, tungsten carbide, various alloys and up to 20% of diamond grits. The quantity of the diamond particles or grits depends strongly on the geometry and dimensions of the tool. 4 The diamond grits are the most important part of the sawblades since they are responsible for the cutting action.
As the sawblade cuts, the diamond particle is exposed freshly above the bond accompanying the wear of the matrix and will penetrate the material (e.g. stone) during the life of the tool. At the end of their useful lifetime and/or during the cutting processes, the sawblades may be damaged, scrapped and wasted. Being unable to use damaged sawblades means waste of an economically valuable source since a significant amount of the existing diamond grits (nearly 10%) could still remain in a damaged and/or scrapped sawblade as also indicated by Shedd. 5 That is why the recovery of diamonds from sawblades has recently gained attention. Since the recoverability has recently gained attention, studies focusing on the related subjects are required. Therefore, this study mainly focuses on the recoverability of diamonds from sawblades. A preliminary investigation on diamond recovery from damaged sawblades using hydrometallurgical process, including aqua regia, is presented. Additionally, before and after leaching, the worn surfaces of diamonds are examined using scanning electron microscopy (SEM) micrographs in this study.
Materials and method
The virgin sawblade having a total diameter of 40 cm includes 28 impregnated diamond segments (circumferential length 40 mm, width 3.5 mm and height 10 mm; see Figure 1(a)). A sawblade damaged during cutting process in the laboratory was used in this study. In order to make the procedure easy and to decrease the leaching time, segments were removed from the damaged sawblade body (Figure 1(b)–(d)). This procedure was applied for the first time in this study differing from the past studies where sawblade was vertically fixed in a rack and turned in a suitable time period in the solution. 4

Removing of segments from a sawblade, a) a virgin sawblade, b) damaged sawblade without segments c) an overview of segments and d) a closer look to a single segment.
Essentially, the diamond is recovered by dissolving metals and other materials inside the matrix to leave the diamond. This recovery process removes all the particles that may be of marginal quality. Hydrometallurgical processes for tungsten carbide (WC) and cobalt (Co) materials can be applied to extract cobalt in solution and to expose the diamond grains implanted into a Co-based matrix strengthened with WC. 6 The chemical dissolution methods with strong acid (HCl, H2SO4, H3PO4, H2Cr2O7) or alkaline (KOH, NaOH) solutions in the presence of oxidizing agents (NaNO3, HNO3) are known for etching and dissolving.6–8 The aqua regia (three parts of hydrochloric acid and one part of nitric acid) leaching of the WC–Co materials to extract cobalt and formation of tungstic acid can be mainly expressed as follows 9
The segments removed from the damaged sawblade were leached using aqua regia solution by freshly mixing 300 mL of concentrated HCl and 100 mL of concentrated HNO3 in a 1-L glass beaker by stirring continuously with a glass rod on a hot plate (Figure 2). The temperature of aqua regia leaching was varied in the range 25 °C–65 °C within ±4 °C by controlling with a thermometer to evaluate the effect of temperature. After leaching of all segments, the diamonds and liquid phases were separated by filtration using a filter paper and a glass funnel. The diamonds were then cleaned with a solution, containing 16 g of K2Cr2O7 and 50 mL of H2SO4, to dissolve any impurity materials. Finally, the separated diamonds were washed, dried and weighted. The pure diamond particles were granulometrically classified using standard sieves of sizes 300, 250 and 212 µm to determine the particle size distribution (Figure 3).

Experimental set-up for the leaching of segments removed from a damaged sawblade.

The recovered diamonds.
Results and discussion
The temperature is an important parameter for the leaching rate. In general, a higher temperature results in faster leaching kinetics. The rate constant improves exponentially with increase in temperature in accordance with the Arrhenius’ equation for reaction-controlled leaching. 10 For this reason, the dissolution of segments was performed by heating. While a segment was entirely dissolved within 2 h at 25 °C temperature, the dissolution time for a segment was found to be a very short time such as 15 min at only 65 °C temperature. Additionally, it is known that the operations with acidic solutions such as in this study may create some environmental issues. For this reason, it is recommended that the waste acid solutions should be rehabilitated. The waste acid solutions can be destroyed by transforming them to stable salt compounds or products that can be used in the industry by acid–base neutralization, so that the pollution of chemical reagents can be minimized to low levels.
The particle size distribution of the recovered diamonds from the damaged sawblade exhibited that 69.3% of the diamonds are present in the range of −250 + 212 µm (Figure 4). Also, 32.3% of the diamonds are present in range of +300 and −300 + 250 µm. Any evidence of wear or fracture on the recovered diamonds for these fractions was not observed in SEM (Figure 5). However, some deformation formed during cutting was observed in 0.4% of the diamonds, which were in the range of −212 µm. These observations show that 99.6% of the diamonds in the damaged sawblade are still present in usable form.
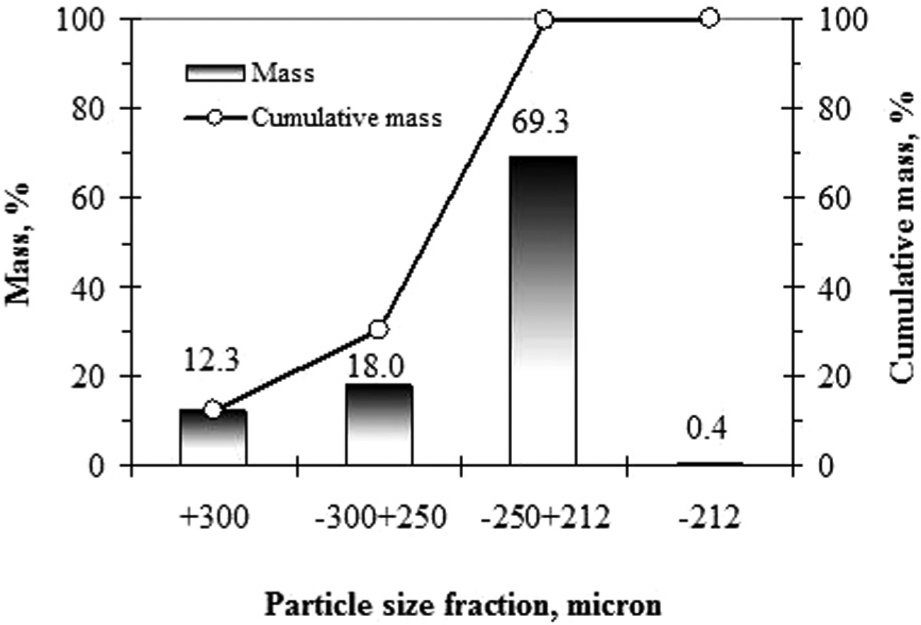
Particle size distribution of the recovered diamonds from the damaged sawblade.

Typical sample of diamonds recovered in the size range presented.
The SEM micrographs of the worn surfaces of segments before leaching were also examined to understand the damages limiting the reuse of diamonds. Similar to the results from other researchers,11–14 emerging, fractured, polished and pull-out crystals were observed in the wearing surfaces of the diamond grits (Figure 6). Among the damage types presented, pull-out crystals cannot be recovered since they were removed from the cutting area by a cooling fluid. Additionally, fractured and polished crystals resulted in diamond crystals with lower grain size. The emerging crystals were recovered effectively together with the other crystals that do not still start to cut the material.

The types of wear observed in worn surfaces of segments.
Conclusion
A preliminary investigation on the diamond recovery from damaged sawblades using hydrometallurgical process, including aqua regia, was experimentally presented in this study. This study revealed that the diamonds from damaged sawblades can be successfully recovered by the hydrometallurgical process presented, and the majority of the recovered diamonds are still undamaged. It was also concluded that with a suitable size classification, the recovered diamonds could be reused in cutting tools using diamonds. Additionally, the SEM micrographs showed that among the damage types of diamonds, the fractured and polished crystals resulted in diamond crystals with lower grain size. Furthermore, it is clearly demonstrated that the temperature is an important parameter for the leaching rate of used sawblades in aqua regia solution. The detailed leaching tests of segments should be considered by examining the effect of temperature to obtain more effective dissolving rate.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.