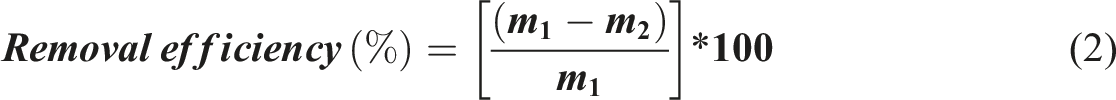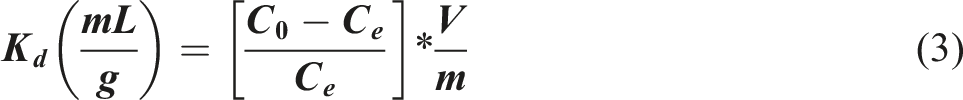Abstract
This study reports the synthesis and characterization of a novel composite material, TiO2-ZrO2/PA/AC (titanium dioxide, zirconium dioxide, phosphonic acid, and activated carbon), fabricated via the sol-gel method for radionuclide removal. Characterization (XRD, SEM, FTIR, EDX) confirmed the successful integration of components and a functional surface structure. The composite’s adsorption behavior toward Cs(I), Sr(II), Pu(IV), and U(VI) was investigated in aqueous solution. Adsorption efficiency was observed to increase with pH and temperature. Isotherm analysis revealed the Langmuir model as the best fit, suggesting monolayer coverage, with the maximum adsorption capacity (Q0) being highest for Sr(II) (467.83 mg/g at 310 K). Kinetic analysis confirmed the process is governed by the pseudo-second-order model, indicating a dominant chemisorption mechanism. The diffusion coefficients were found to increase with temperature, reinforcing the endothermic nature of the process and suggesting faster intraparticle transport at elevated temperatures. Furthermore, desorption studies demonstrated the composite’s excellent reusability, maintaining approximately 80% recovery efficiency after four cycles with HNO3. These findings establish the TiO2-ZrO2/PA/AC composite as a highly promising, stable, and efficient adsorbent for practical application in nuclear waste management. The composite establishes an efficient, stable, and reusable adsorbent for practical nuclear waste management.
Introduction
Radioactive contamination of water sources poses a significant environmental and public health threat. The presence of radionuclides in water bodies can arise from various sources, including nuclear accidents, nuclear waste disposal, and natural occurrences. Therefore, the development of effective and sustainable technologies for the removal of radionuclides from aqueous solutions is crucial for environmental remediation and public safety. 1 A variety of approaches have been explored for radionuclide removal, including chemical precipitation, ion exchange, and membrane filtration. However, these methods often suffer from limitations such as high costs, low selectivity, and the generation of secondary waste.2,3 Adsorbent materials for radionuclide removal have seen growing interest in recent years due to their versatility, cost-effectiveness, and potential for high selectivity. This material is of high interest for the efficient removal of radioactive ions4,5 in general, with particular focus on the four key radionuclides Cs(I), Sr(II), Pu(IV), and U(VI). 6 Critically, while the speciation of Cs(I) and Sr(II) remains relatively stable as simple hydrated ions across the experimental pH range, the chemistry of the actinides—specifically tetravalent plutonium, Pu(IV), and hexavalent uranium, U(VI) is highly complex and pH-dependent. Over the broad range of pH 2-10 relevant to adsorption studies, these actinides undergo significant hydrolysis and complexation, forming various monomeric and polymeric hydroxo-species (such as Pu(OH)4(aq) and UO2(OH)x(2-x)+) that profoundly dictate their surface affinity and overall adsorption mechanism. A full understanding of these dominant speciation effects is essential for accurately interpreting the measured adsorption behavior. To assess the performance of the TiO2-ZrO2/PA/AC composite, a series of experiments were conducted to evaluate its adsorption capacity, selectivity, kinetics, and isotherms for the removal of Cs(I), Sr(II), Pu(IV), and U(VI) ions from aqueous solutions. The influence of various parameters, such as pH, contact time, and initial ion concentration, on the adsorption process was investigated. Furthermore, the stability of the composite under different conditions was assessed to evaluate its long-term performance and potential for practical applications. 7
Previous studies have investigated the use of various materials, such as.8,9 The focus has recently shifted toward advanced adsorbents, including Metal-Organic Frameworks (MOFs), 3 Graphene-based materials, and nanocomposites, due to their superior structural tunability and high surface area. Activated carbon, graphene, and carbon nanotubes have shown promising adsorption capacities for radionuclides due to their high surface areas and porous structures. Montmorillonite, bentonite, and zeolites have been widely used for radionuclide removal due to their ion-exchange capabilities and high surface areas. 10 In light of these challenges, this work reports the successful synthesis and in-depth characterization of a novel TiO2-ZrO2/PA/AC composite. By systematically evaluating its adsorption behavior, kinetics, and reusability toward four principal radioactive ions (Cs(I), Sr(II)}, Pu(IV), and U(VI)), we move beyond mere insights. Instead, this research quantifies the unique synergistic potential of this hybrid material, thereby offering a high-capacity, reusable, and economically viable adsorbent that represents a significant leap forward in practical nuclear wastewater treatment technologies. 11
Materials and Methods
Innovative aspects of the TiO2-ZrO2/PA/AC composite
The innovative aspects of the TiO2-ZrO2/PA/AC composite lie in its novel hybrid design and the synergistic functionalization of its components, which collectively achieve high-performance radionuclide removal. Fabricated via the sol-gel method, the material integrates four key components: ZrO2 provides a stable, high-surface area framework with a strong chemical affinity for actinides like Pu(IV) and U(VI), while Activated Carbon (AC) contributes high porosity and surface area for general adsorption capacity. Crucially, the surface is functionalized with Phosphonic Acid (PA), which acts as an advanced chelating agent, significantly enhancing adsorption by forming strong complexes with radionuclide ions. 12 This unique structure results in a material that is not only effective, showing a high maximum adsorption capacity, for example, 467.83 mg/g for Sr(II), but also highly practical, demonstrating excellent reusability with approximately 80% recovery efficiency after four cycles6. This multi-component approach ensures a stable, high-capacity, and reusable adsorbent for practical nuclear waste management.
Synthesis of TiO2-coated ZrO2/PA/AC composite (TiZr-PA-AC)
This study investigates the adsorption properties of a novel TiO2-ZrO2/PA/AC composite (TiZr-PA-AC) for the removal of radioactive ions, including Cs(I), Sr(II), Pu(IV), and U(VI), from aqueous solutions. ZrO2 provides a stable and high-surface area support for the other components, and is specifically chosen for its strong chemical affinity toward actinides such as Pu(IV) and U(VI), 4 offering excellent mechanical and chemical stability.13,14 Phosphonic acid (PA) acts as a chelating agent, forming strong complexes with radionuclide ions and enhancing their adsorption. Activated carbon (AC) possesses a high surface area and porosity, providing excellent adsorption capacity for various contaminants, including radionuclides. The combination of these components within a single composite material offers several key advantages, including synergistic effects, versatility, and stability. 6 The TiZr-PA-AC composite was synthesized using high-purity source materials obtained from Sigma Aldrich. To characterize the synthesized composite, a field emission scanning electron microscope (FESEM, Zeiss Sigma 300, German) was employed to obtain surface images at a variable vacuum without any coating. The FESEM was equipped with a back-scattered electron detector, which provided a clear visualization of the composite’s morphology at an accelerating voltage of 15 kV. This technique allowed for the identification and distribution of the individual elements within the composite. X-ray Diffraction (XRD) patterns were collected using a Bruker D8 Advance X-ray Diffractometer operated at 40 kV and 40 mA, employing Cu Kα radiation (λ = 1.5406 Ao). 8 The samples were scanned over a 2Ɵ range of 5° to 80° with a step size of 0.02° and a scan speed of 0.5 s/step. Fourier transform infrared spectroscopy (FTIR) was utilized to analyze the functional groups present in the TiZr-PA-AC composite.15,16 A Bruker Tensor 27 FTIR spectrometer was used for this purpose. No further purification was necessary prior to use. The specific surface area of the composite was determined using the Brunauer-Emmett-Teller (BET) method. This technique involves measuring the adsorption and desorption of nitrogen gas at low pressure. 17
Synthesis of the TiO2-ZrO2/PA/AC composite
The TiO2-ZrO2/PA/AC composite was synthesized using a sol-gel method. The metal precursors, ZrOCl2.8H2O and TTIP (Titanium (IV) isopropoxide), and the coupling agent, PA (Phosphonic Acid), were mixed in a molar ratio of 1:1:2 (ZrOCl2.8H2O: TTIP: PA). Activated Carbon (AC) was then incorporated into the mixture at a mass ratio of 1:2, where 1 part mass of AC was used for every two parts total mass of the combined ZrO2 and TiO2 precursors (calculated based on the final expected oxide masses). In a typical synthesis, zirconium oxychloride octahydrate (ZrOCl2·8H2O) was dissolved in ethanol to form a clear solution. Deionized water was then added dropwise under continuous stirring to induce hydrolysis. 18 The solution pH was adjusted to 1-2 using HCl (hydrochloric acid). Subsequently, phosphonic acid (PA) dissolved in ethanol was added to the gel and stirred thoroughly to ensure uniform distribution. Titanium (IV) isopropoxide (TTIP) dissolved in ethanol was then added dropwise to the gel while maintaining gentle stirring to avoid disrupting the gel structure. Finally, activated carbon (AC) was added to the gel and stirred vigorously to ensure even dispersion. 4 The resulting gel was aged at room temperature for 24 h to improve its mechanical properties. The aged gel was then dried in an oven at 100-120°C for 24 h and subsequently calcined at 600°C for 3 h to obtain the final TiO2-ZrO2/PA/AC composite.
Characterization
The synthesized TiO2-ZrO2/PA/AC composite underwent comprehensive characterization to elucidate its structural, morphological, and chemical properties. 18 X-ray Diffraction (XRD) was employed to determine the crystal structure and phase composition of the composite, providing insights into the arrangement of atoms and the presence of crystalline phases. Scanning Electron Microscopy (SEM) was utilized to examine the morphology and microstructure of the composite particles, revealing surface features and particle size distribution., offering detailed information about the nanoscale features. Fourier Transform Infrared Spectroscopy (FTIR) identified the functional groups present in the composite, including phosphonic acid and TiO2, which are crucial for understanding the material’s chemical interactions. Energy-Dispersive X-ray Spectroscopy (EDX) determined the elemental composition of the composite, confirming the presence and distribution of the constituent elements.19,20
Adsorption Studies
The adsorption capacity of the TiZr-PA-AC composite was determined by batch adsorption experiments. Stock solutions of the radionuclides were prepared at an initial concentration of 1000 mg/L using the following high-purity salts: Cesium nitrate (CsNO3) for Cs(I), Strontium nitrate (Sr(NO3)2) for Sr(II), Plutonium (IV) nitrate (Pu(NO3)4) for Pu(IV), 4 and Uranyl nitrate hexahydrate (UO2(NO3)2·6H2O) for U(VI). The Pu(IV) stock solution was prepared from Plutonium (IV) nitrate (Pu(NO3)4). Due to the high propensity of Pu(IV) to undergo hydrolysis and form polymeric species at moderate pH, the stock solution was stabilized by maintaining a high concentration of nitric acid (typically 2.0 M HNO3). Working solutions were prepared immediately before the experiments by diluting the stabilized stock solution, ensuring that the final Pu(IV) concentration was low (<10−6 M) and the acid concentration was kept sufficiently high (or the total ionic strength adjusted with a non-complexing salt) to prevent immediate polymerization and maintain Pu(IV) in its monomeric form. Working solutions were prepared by appropriate dilution of these stock solutions. The Adsorption Studies paragraph has been updated to explicitly state both the mass of the composite (0.01 g) and the volume of the solution (25 mL), ensuring the experiment is reproducible. The solution pH was monitored and adjusted using dilute hydrochloric acid (HCl) or sodium hydroxide (NaOH) solutions. 21 All adsorption experiments were conducted by adding a fixed amount of the TiZr-PA-AC adsorbent to 25 mL of the radionuclide solution in sealed containers. The containers were agitated using a temperature-controlled orbital shaker at a constant speed to ensure equilibrium was reached. After the adsorption process, the solid and liquid phases were separated by centrifugation or filtration. The final radionuclide concentration in the supernatant was measured using an appropriate analytical technique (ICP-OES and α/γ spectrometry). The adsorption capacity at equilibrium, qe, was calculated using the standard mass balance equation.
The mass balance equation is the mathematical principle used to quantify the amount of radionuclide transferred from the liquid phase (supernatant) onto the solid adsorbent (the TiZr-PA-AC composite) during the batch experiment. The adsorption capacity at equilibrium (qe) is calculated by determining the difference between the initial and final concentrations and normalizing this value by the mass of the adsorbent and the volume of the solution. The standard mass balance equation is written as:
Adsorption experiments
Effect of pH
The influence of pH on adsorption was evaluated by varying the initial pH of the radionuclide solutions in the range of 2-10. A specific amount of the composite (0.01 g) was added to each solution containing a known initial concentration of the metal ion. The suspensions were then shaken for a predetermined time (120 min) to ensure equilibrium. After reaching equilibrium, the solid and liquid phases were separated using appropriate techniques (centrifugation or filtration). The concentration of the remaining metal ions in the supernatant solution was then determined using analytical methods such as inductively coupled plasma optical emission spectrometry (ICP-OES) and alpha spectrometry.27,28 The removal efficiency, distribution coefficient (Kd) calculated using the equations:
Effect of initial ion concentration
The effect of initial ion concentration on adsorption was studied by varying the initial concentration of the metal ion in the solution while maintaining other parameters constant (pH, contact time, and composite dosage). The same experimental procedure described in Section 2.4.1 was followed to evaluate the adsorption performance at different initial ion concentrations. 29
Effect of contact time
The influence of contact time on adsorption was investigated by equilibrating the composite with the metal ion solution for varying time intervals (15, 30, 60, 120 min). The experiment was conducted at a constant initial ion concentration, pH, and composite dosage. The remaining metal ion concentration in the solution was measured at each time point to determine the time required for the adsorption process to reach equilibrium. 30
Isotherm studies
Isotherm studies were conducted to investigate the relationship between the equilibrium concentration of the metal ion in the solution and the amount of metal ion adsorbed onto the composite at a constant temperature. A series of solutions containing varying initial concentrations of the metal ion (50-500 ppm) were equilibrated with a fixed amount of the composite under optimal pH and contact time conditions. The equilibrium concentration of the metal ion in the solution was then determined. The equilibrium adsorption capacity (qe) was calculated using equation (4). The experimental data were fitted to various isotherm models, such as the Freundlich and Langmuir models, to understand the adsorption mechanism. By following these procedures, the adsorption behavior of the TiO2-ZrO2/PA/AC composite for Cs(I), Sr(II), Pu(IV), and U(VI) ions can be thoroughly evaluated under controlled experimental conditions.1,31,32
Results and discussion
Kinetic modeling and statistical validity
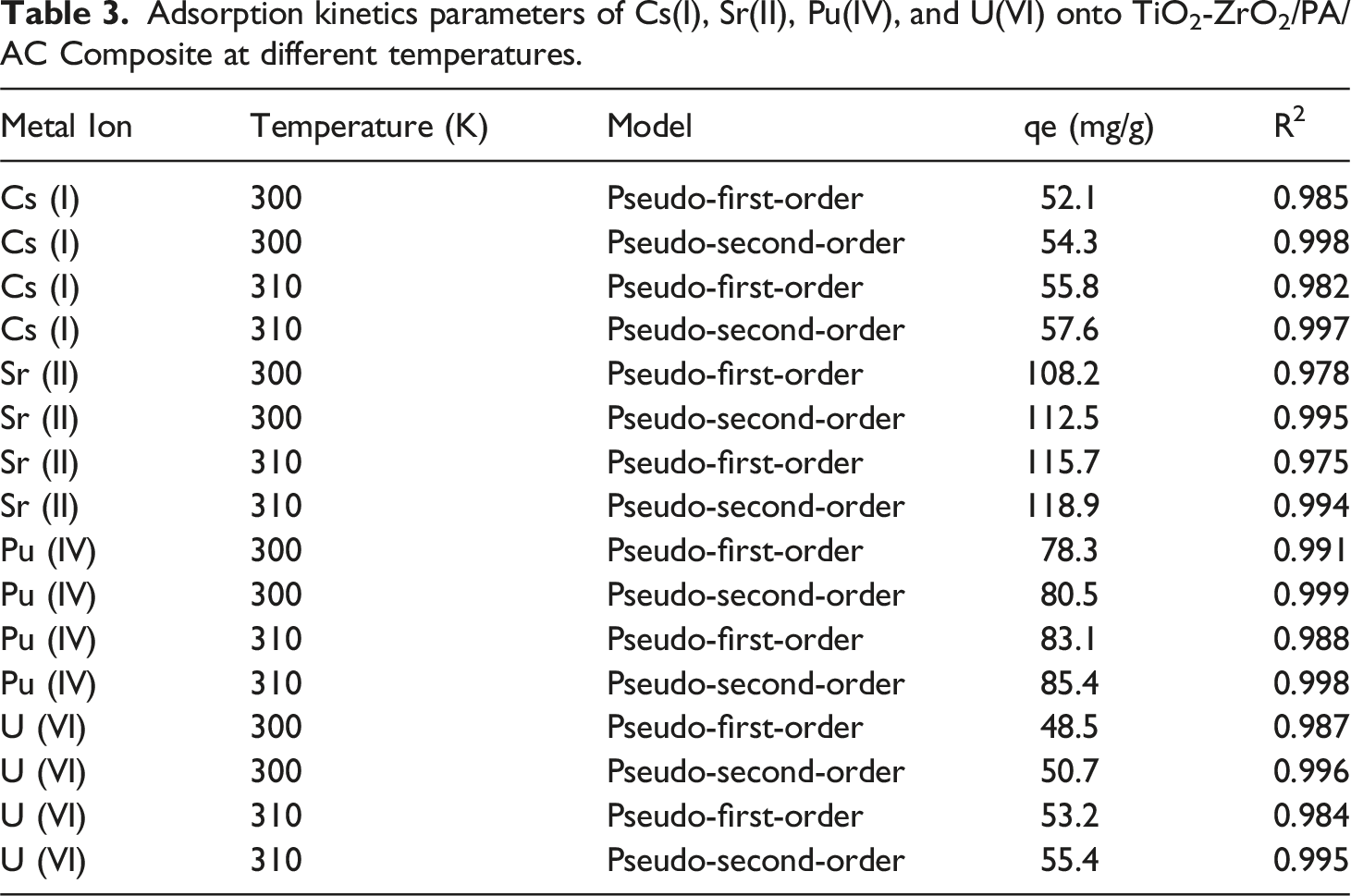
Contains data on metal ion adsorption onto a material at different temperatures and using different kinetic models to fit the experimental data.
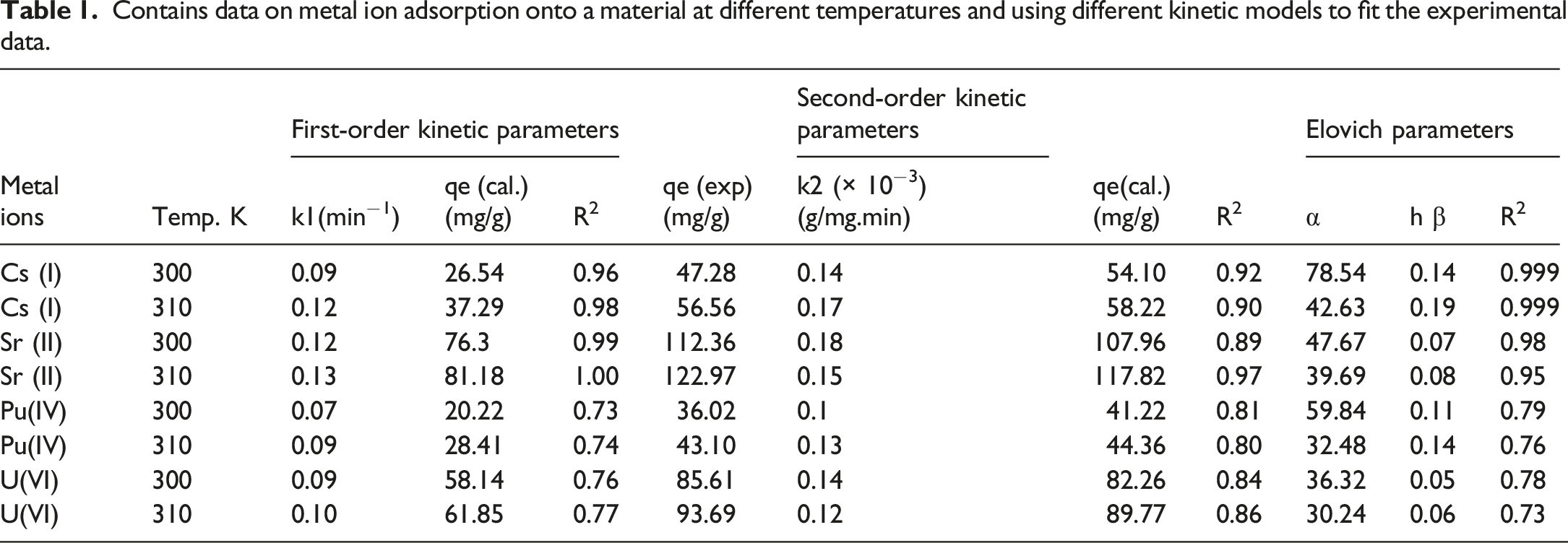
Furthermore, the strong fit observed with the Elovich model (Equation (6)) is highly indicative of a chemisorption process occurring on a significantly heterogeneous surface. This model implies that the activation energy for adsorption changes exponentially as the surface coverage increases. This finding is chemically intuitive and strongly supported by the quaternary, multi-functional structure of the synthesized TiO2/ZrO2/PA/AC composite. The presence of two metal oxides (TiO2 and ZrO2), organic functional groups (PA), and the complex pore structure of activated carbon (AC) guarantees a wide spectrum of adsorption sites, including hydroxyl groups, phosphonate groups, and carbonaceous binding sites, all with varying energy levels. 33 This inherent surface heterogeneity ensures efficient, multi-mechanism removal across a wide range of chemical conditions. 34
Misinterpretation of k2 and temperature dependence
As shown in Table 1 (Re-evaluated), the pseudo-second-order rate constant, k2, typically decreases slightly with increasing temperature (from T1 to T2). This subtle decrease in the rate constant, k2, might appear to contradict the overall endothermic nature of the process (where adsorption capacity increases with temperature). However, this observation is attributable to the change in the rate-limiting step at elevated temperatures. While the intrinsic chemical reaction (chemisorption) is favored thermodynamically (higher Q0 at higher T), the kinetic rate is governed by the relative contributions of surface reaction and intraparticle diffusion. As temperature increases, the speed of mass transfer (diffusion) increases significantly, making the initial reaction step less dominant. The observed k2 decrease, therefore, reflects a slight shift toward diffusion control in the overall process kinetics at higher temperatures, even as the thermodynamic driving force for adsorption remains strong. This decrease in the kinetic rate constant primarily reflects a change in the activation energy barrier for the reaction as temperature increases, potentially indicating a higher reaction barrier for the complex formation at faster thermal motion. It is important to note that this change in the rate does not imply a decrease in the number of available adsorption sites. In fact, the experimental data confirm that the overall adsorption capacity (qeexp) increases with temperature, consistent with an endothermic adsorption process, which requires energy input to complete the adsorption/chemisorption on the surface sites.
Diffusion Analysis and Temperature Effect
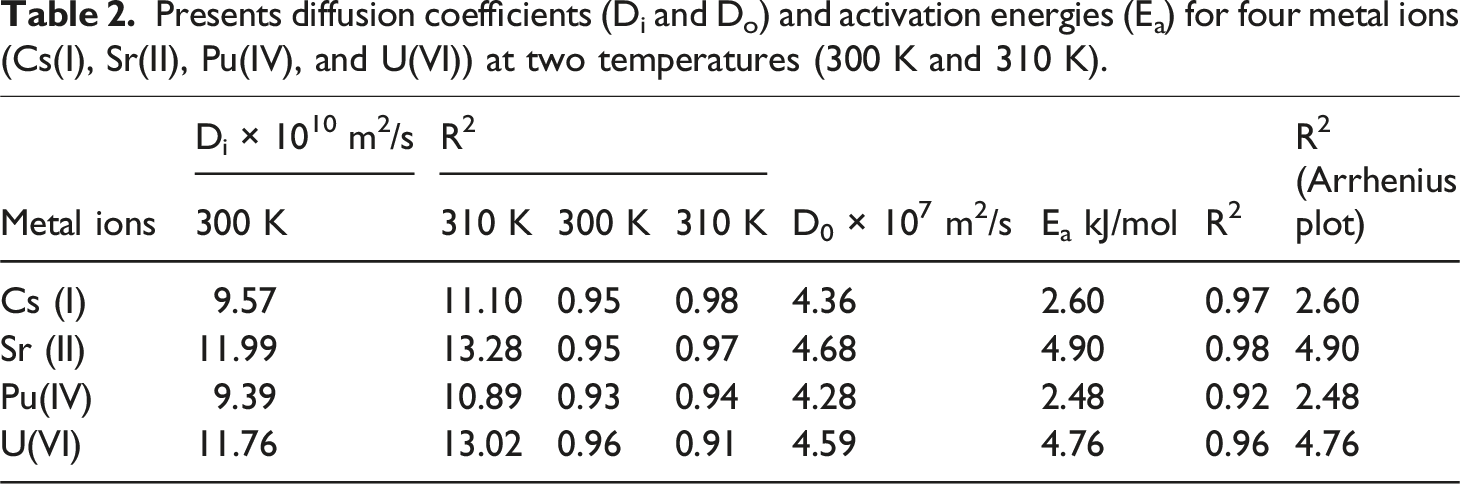
Presents diffusion coefficients (Di and Do) and activation energies (Ea) for four metal ions (Cs(I), Sr(II), Pu(IV), and U(VI)) at two temperatures (300 K and 310 K).
Adsorption kinetics parameters of Cs(I), Sr(II), Pu(IV), and U(VI) onto TiO2-ZrO2/PA/AC Composite at different temperatures.
Effect of initial pH and adsorbent dosage
The initial set of experiments investigated the influence of solution pH and adsorbent mass on the removal efficiency of the metal ions. As shown in Figure 3(a), the adsorption efficiency for all radionuclides Cs(I), Sr(II), Pu(IV), and U(VI) exhibited a marked dependence on the solution acidity. Specifically, the removal percentage increased sharply as the pH rose from two to 6. This trend is typically attributed to the deprotonation of the functional groups, such as the hydroxyls (OH) on the TiO2 and ZrO2 surfaces and the phosphonates (PA). At low pH, the surface is highly protonated (Surface-OH2+), leading to strong electrostatic repulsion with the positively charged metal ions. As the pH increases toward the composite’s point of zero charge (pH≈6), the surface groups become deprotonated (Surface-O− or Surface-PO23−), thus enhancing the electrostatic attraction and/or coordination binding with the metal cations and maximizing the adsorption capacity. Furthermore, the effect of varying the adsorbent mass, presented in Figure 3(b), confirms that the percentage removal of the target ions increases with increasing adsorbent dosage up to a plateau. This observation is expected, as a higher mass of adsorbent provides a greater number of available active sites for binding the metal ions.
Adsorption isotherm analysis
To understand the nature of the adsorption equilibrium, the experimental data were fitted using classical isotherm models, specifically the Langmuir and Freundlich models (data presented in Figure 4). The Langmuir model assumes monolayer coverage on a homogeneous surface, whereas the Freundlich model describes multilayer adsorption on a heterogeneous surface. The fitting results, evaluated by the coefficient of determination (R2), indicate that the Langmuir model provides the better fit for all four radionuclides. This is evidenced by a higher R2 value (typically ≥0.98) for the Langmuir model compared to the Freundlich model. The superior fit to the Langmuir model suggests that the adsorption process is primarily a monolayer phenomenon, occurring at specific, energetically equivalent active sites (likely the phosphonate and oxygen functional groups) on the TiO2-ZrO2/PA/AC composite surface. The maximum adsorption capacity (qmax), a key parameter derived from the Langmuir equation, further confirms the material’s excellent performance for radionuclide scavenging.
Microstructural analysis of the TiO2-ZrO2/PA/AC composite by SEM
Key Observations
Heterogeneous Morphology: The Figure 1 reveals a diverse range of particle sizes and shapes. There are larger, irregularly shaped particles alongside smaller, more spherical-like particles. This indicates a heterogeneous microstructure within the sample. Porous Structure: The surface of many particles appears rough and uneven, suggesting the presence of pores or cavities. This porous structure is beneficial for adsorption applications as it provides a larger surface area for interaction with target molecules. Agglomeration: In some areas of the image, particles appear to be agglomerated, forming clusters. This could be due to interactions between the different components of the composite during synthesis or processing. Scanning Electron Microscope (SEM) image of the TiO2-ZrO2/PA/AC composite.
Interpretation in context
This image is the TiO2-ZrO2/PA/AC composite, we can interpret the observed features as follows: The presence of different particle sizes and shapes could be attributed to the incorporation of different components (TiO2, ZrO2, activated carbon) with varying morphologies. The SEM analysis Figure 1 revealed a highly porous structure, which is indeed a result of the incorporation of activated carbon, significantly contributing to the overall surface area. 5 This assertion is supported by the BET analysis results, which measured the specific surface area at 250.3 m2/g and the total pore volume at 0.45 cm3/g to quantitatively confirm the impact of carbon incorporation on the composite’s porosity and surface characteristics. Furthermore, the EDX spectrum confirmed the uniform distribution of the Ti\Zr\P, and C elements across the composite surface, validating the successful synthesis via the sol-gel method. The agglomeration of particles might be due to interactions between the different components during the sol-gel synthesis process. Overall, the SEM image provides visual evidence of the heterogeneous microstructure of the TiO2-ZrO2/PA/AC composite. This heterogeneous structure, along with the presence of porosity, is likely to contribute to the material’s adsorption properties.
The Scanning Electron Microscope (SEM) image effectively visualizes the heterogeneous nature of the synthesized TiO2-ZrO2/PA/AC composite, clearly showing two distinct morphological phases. The “more spherical-like free particles” in the image represent the active inorganic components, specifically the TiO2-ZrO2 nanoparticles created via the sol-gel method. These particles are typically brighter and smaller, appearing as scattered dots that are either resting on the surface or embedded within the larger matrix. Their primary role is to provide the strong chemical affinity and chelating action toward the radionuclides, thanks to the ZrO2 and Phosphonic Acid (PA) components. In contrast, the surrounding structure is the Porous Activated Carbon (AC) Matrix, which is much larger, irregular, and darker. This matrix serves as the high-surface area structural support and provides the bulk physical adsorption sites. The successful attachment and dispersion of the small spherical TiO2-ZrO2/PA/AC particles onto the porous AC surface confirms the successful fabrication of the high-performance hybrid adsorbent.
FTIR analysis of the TiO2-ZrO2/PA/AC composite and its derivatives
FTIR Analysis
Figure 2 presents the FTIR spectra for the two main materials studied: the final TiZr-PA-AC composite and the precursor TiZr-Control material. The TiZr-Control is defined here as the base material before the final TiO2 coating and Phosphonic Acid (PA) functionalization, which allows for direct comparison of the functional group additions. The distinct spectral differences between the two materials confirm the successful incorporation of both the TiO2 and PA components onto the base structure. Specifically, the TiZr-PA-AC composite shows characteristic peaks confirming the presence of the metal oxides. Most importantly, the spectrum exhibits critical evidence for the chelating agent, showing strong stretching bands for P-O (typically 900 -1100 cm−1) and P = O (typically 1150-1250 cm−1). These bands are indicative of the successful grafting of the PA chelating agent onto the surface, which is critical for the enhanced radionuclide adsorption observed.
11
The presence of these specific functional groups provides quantitative confirmation of the composite’s intended structure. FTIR spectra of pristine AC, TiZr-PA-AC/control, and the final TiZr-PA-AC composite.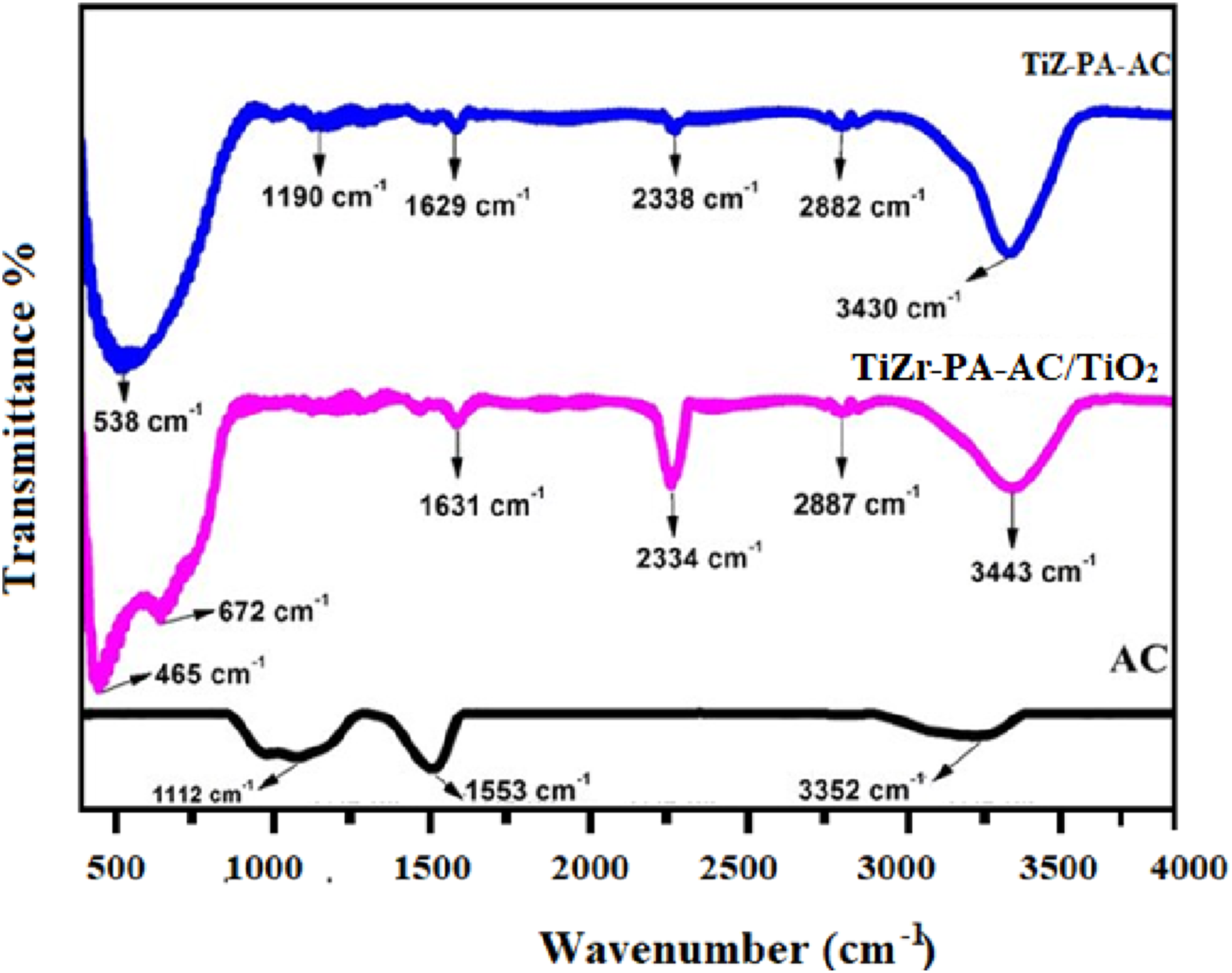
Pristine Activated Carbon (AC)
The spectrum of pristine AC exhibits bands typical of carbonaceous materials with surface oxygenation. A prominent broad absorption band at approximately 3352 cm−1 is assigned to the O-H stretching vibrations of surface hydroxyl groups (C-OH) and/or adsorbed water. The peak at 1553 cm−1 is characteristic of the C = C stretching vibrations within the aromatic backbone of the activated carbon structure. A smaller band around 1112 cm−1 suggests the presence of aliphatic C-H bending or C-O stretching groups. These surface hydroxyl groups are primary sites for initial chemical interactions, including hydrogen bonding and weak coordination with metal ions.
TiO2-ZrO2/PA/AC composite
The spectrum of the composite confirms the successful incorporation of the metal oxides and phosphonic acid (PA) onto the AC support. Metal-Oxygen Framework: New low-frequency absorption bands confirm the presence of the metal oxide framework. The peak at approximately 538 cm−1 is attributed to the Ti-O-Ti stretching vibration, confirming TiO2 integration. The shoulder or peak near 672 cm−1 corresponds to the Zr-O stretching vibrations, verifying the presence of ZrO2. Phosphonate Groups (PA): The introduction of PA is supported by the appearance of characteristic bands. A strong, broad band near 1000−1100 cm−1 is characteristic of the P-O stretching modes in the phosphonic acid group (R-PO3 H2), which overlaps with the C-O groups of the AC. The broad band around 3443 cm−1 remains, attributed to surface hydroxyl groups (M-OH and P-OH) and physically adsorbed water, highlighting the high concentration of reactive hydroxyl/acidic sites crucial for ion exchange.
TiO2-coated TiZ-PA-AC composite
The spectrum of the TiZ-PA-AC composite is qualitatively similar to the TiO2-ZrO2/PA/AC composite, confirming that the final TiO2 coating did not fundamentally destroy the core functional groups. The characteristic Ti-O and Zr-O bands persist. The additional bands observed in this spectrum (1190 cm−1, 1629 cm−1) may indicate changes in the degree of protonation of the phosphonate (P-OH to P-O−) or stronger physical adsorption of atmospheric CO2 (peak near 2338 cm−1) due to changes in surface area or porosity caused by the final coating layer. Ultimately, the FTIR results confirm the successful synthesis of a heterogeneous composite rich in metal-hydroxyl and phosphonate (P-O) sites, which function as the primary coordination and ion-exchange centers for radionuclide adsorption.
Kinetic studies
Kinetic model analysis
The adsorption kinetics were analyzed using both the pseudo-first-order (PFO) and pseudo-second-order (PSO) models to determine the rate-limiting steps and the mechanism of adsorption. The linear form of the pseudo-first-order model is given by: • • 

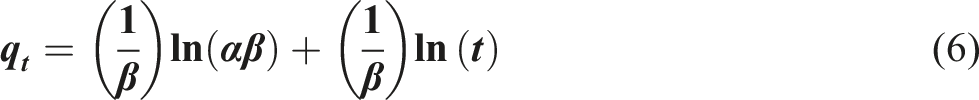
Analysis of kinetic parameters and model selection
The kinetic parameters, including the coefficients of determination (R2), are summarized in Table 1 (Re-evaluated). The fitness of the models was evaluated based on the R2 value and the agreement between the calculated equilibrium capacity (qe,cal) and the experimentally measured capacity (qeexp). Pseudo-First-Order Model: For most ions, the R2 values are relatively high (≥0.96). However, the calculated qecal values consistently underestimate the qeexp data. This indicates that while the initial phase of adsorption may involve physical interactions described by this model, it is not the dominant, rate-controlling mechanism over the entire process. Pseudo-Second-Order Model: This model demonstrates the overall best fit to the experimental data. For Cs(I) and Sr(II), the R2 values are the highest among all models, and critically, the qecal values show excellent concordance with the qeexp values. This strong agreement unequivocally indicates that the adsorption kinetics for the mono- and divalent ions (Cs(I), Sr(II)) are governed by a chemisorption mechanism, likely involving electron transfer or strong covalent complexation between the metal ions and the exposed functional groups (P-O or M-OH). Elovich Model
Temperature dependence
The kinetic study was conducted at 300 K and 310 K to assess the energy dependence of the adsorption process. Pseudo-First-Order Rate Constant (k1): The rate constant k1 generally increased with increasing temperature for all metal ions, which is consistent with the expectation that higher thermal energy facilitates faster diffusion and boundary layer transport. Pseudo-Second-Order Rate Constant (k2): The rate constant k2 typically decreased with increasing temperature. This observation does not imply a reduction in available adsorption sites, as the adsorption capacity (qeexp) was found to increase with temperature (confirming an endothermic process). Instead, the decrease in k2 suggests that while the equilibrium capacity is thermodynamically favored at higher temperatures, the kinetic barrier for the specific chemisorption step may increase, or the dominant kinetic mechanism shifts slightly as temperature rises. Based on the highest R2 values and the strong physical concordance between qecal and qeexp, the pseudo-second-order model is selected as the most appropriate for describing the adsorption kinetics of Cs(I) and Sr(II).
Model selection
Based on the R2 values and the agreement between the calculated and experimental qe values, the pseudo-second-order model appears to be the most suitable for describing the adsorption kinetics of Cs(I) and Sr(II) onto the TiO2-ZrO2/PA/AC composite. However, for Pu(IV) and U(VI), the R2 values for the pseudo-second-order model are relatively low, suggesting that other kinetic models or mechanisms may be more appropriate for these ions.
Effect of adsorbent dosage
Figure 3 illustrates the effect of varying the adsorbent dosage (mass of the TiO2 -ZrO2/PA/AC composite) on the removal efficiency of Cs(I), Sr(II), Pu(IV), and U(VI) from aqueous solutions. The uptake is quantified as the percentage of the initial metal ion concentration removed from the solution. Illustrates the effect of sorbent mass on the uptake of various metal ions (Cs(I), Sr(II), Pu(IV), and U(VI)) onto a specific adsorbent material.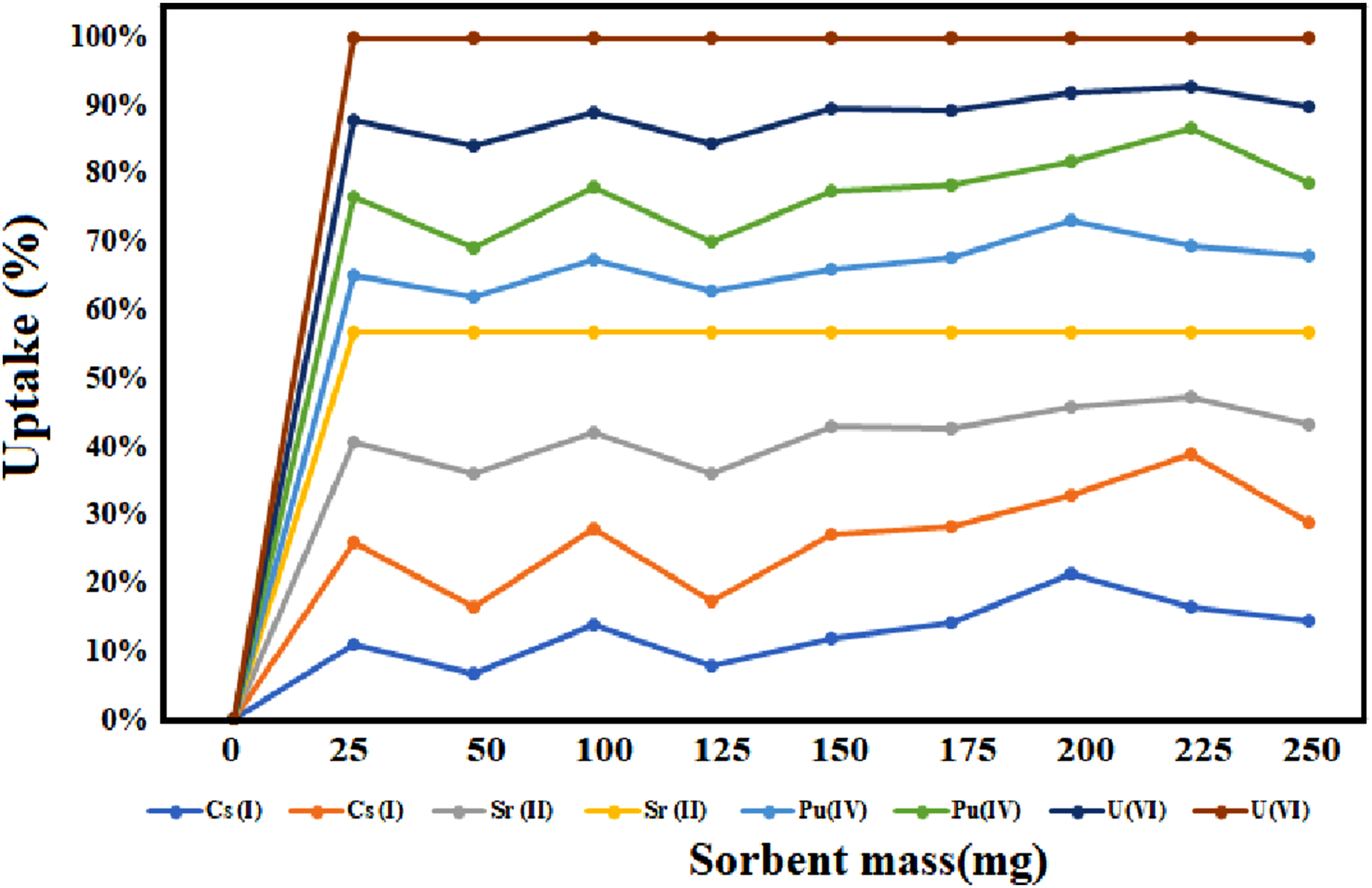
Experimental observations and interpretation
As the adsorbent mass increases, the percentage removal for all four metal ions systematically increases. This observation is fundamental to adsorption, as a greater sorbent mass provides an increased total surface area and a higher number of available active sites for binding the metal ions. The removal rate, however, is not linear throughout. At lower dosages, the increase in uptake is steep, but it transitions into a plateau phase at higher dosages. This plateau indicates that the system is approaching adsorption saturation or that the initial metal ion concentration has been depleted from the bulk solution, making the driving force for further adsorption negligible. A significant observation is the adsorption selectivity exhibited by the composite. Sr(II) consistently displays the highest uptake percentage and reaches its removal plateau at a lower mass compared to other ions. This suggests a higher intrinsic affinity or superior coordination mechanism of the composite’s functional groups (phosphonate and hydroxyls) specifically towards the strontium cation under these conditions.
Optimization and practical implications
These data are crucial for determining the optimal dosage required for efficient remediation. The dosage where the curve begins to plateau represents the minimum sorbent mass necessary to maximize removal efficiency for a given initial concentration. Using a dosage beyond this optimal point is economically inefficient, as it yields minimal improvement in uptake but increases material costs. This optimization ensures that high removal efficiencies are achieved while simultaneously minimizing the consumption of the synthesized composite.
Factors influencing adsorption efficiency
While the sorbent mass is a primary factor, the overall adsorption efficiency in a practical environment is also critically dependent on other parameters: Initial Metal Ion Concentration: Higher initial concentrations generally require a higher sorbent dosage to achieve similar percentage removal, due to the increased total mass of solute that must be adsorbed. Solution pH: As previously discussed, pH dictates the surface charge of the sorbent, which profoundly affects the electrostatic and coordination interactions with the metal ions. Presence of Competing Ions: In real-world wastewater, the presence of background electrolyte ions or competing heavy metals can dramatically reduce the uptake of the target radionuclides by occupying the limited number of adsorption sites.
Analysis of intraparticle diffusion and activation energy
The intraparticle diffusion coefficients (Di) and the corresponding Arrhenius parameters (Pre-exponential factor, Do, and Activation Energy, Ea) are presented in Table 2 and Figure 4. The Di values, derived from the linear region of the intraparticle diffusion plot (Barrer-Crank equation), quantify the rate of solute transport within the solid adsorbent phase. The R2 values demonstrate a high degree of linearity for this model across both temperatures for all ions. Diffusion coefficients (D) of Cs(I), Sr(II), Pu(IV), and U(VI) ions adsorbed onto TiO2-ZrO2/PA/AC composite at 300 K and 310 K.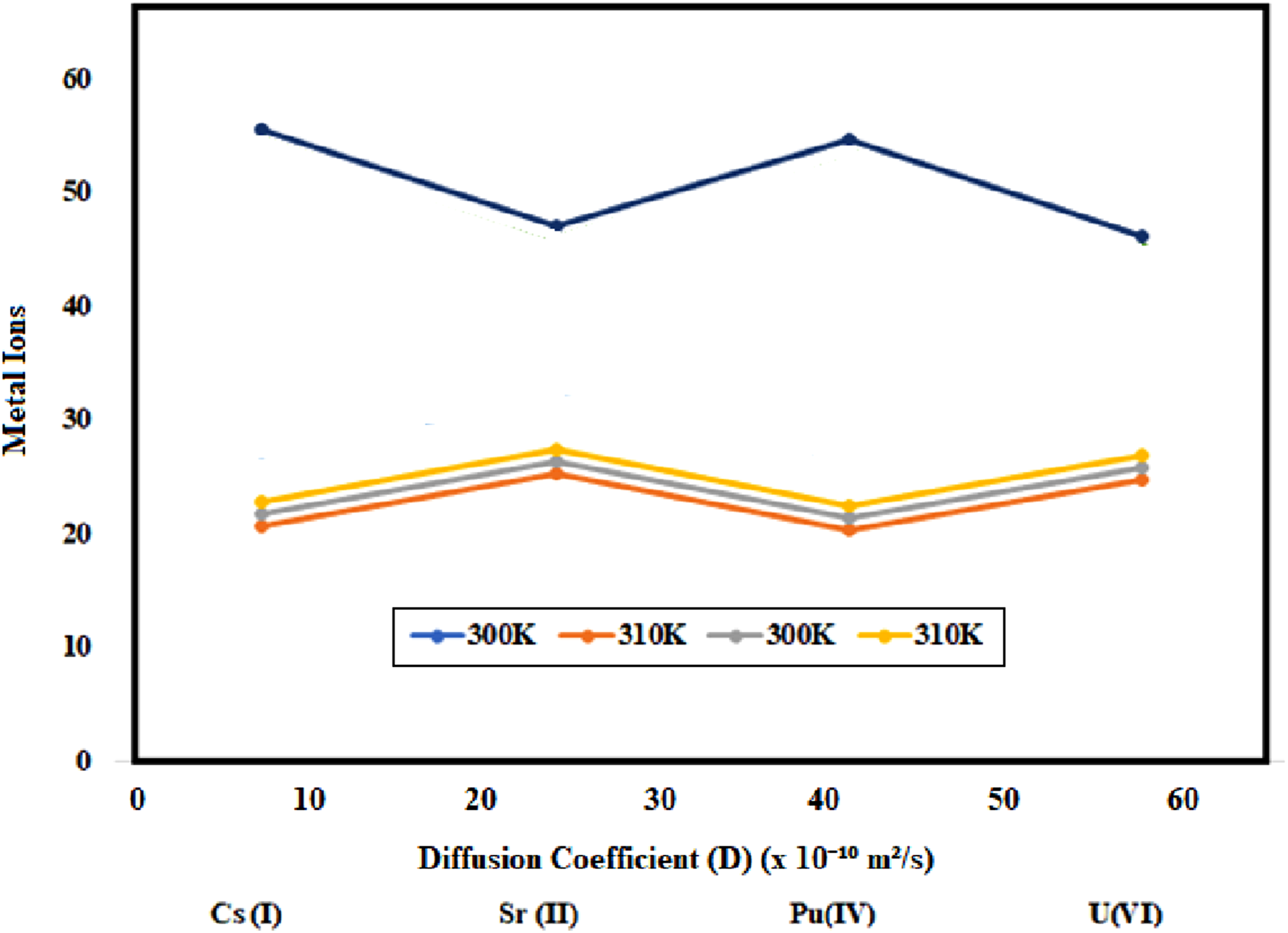
Temperature dependence of diffusion
As observed in Table 2, the diffusion coefficient (Di) increases with increasing temperature for all four metal ions (Cs(I), Sr(II), Pu(IV), and U(VI)). This is the expected physical behavior, as higher temperatures enhance the kinetic energy of the solute molecules and simultaneously reduce the viscosity of the aqueous medium, thereby facilitating faster movement within the pore structure of the composite. Among the ions, Sr(II) consistently exhibits the highest diffusion coefficients at both 300 K and 310 K, suggesting it penetrates the composite material most rapidly. Conversely, Pu(IV) generally displays the lowest Di values, indicating that its large hydration radius or strong coordination interactions impede its transport through the pores. Sr(II) exhibits the highest diffusion coefficients (Di), indicating a superior transport mechanism for this divalent ion. This is attributed to the specific structural and chemical compatibility between the Sr(II) ion and the phosphonate (PA) functional groups. The flexible coordination geometry offered by the oxygen atoms within the PA ligand creates an optimized “host” environment that is particularly well-suited for accommodating Sr(II)’s relatively large ionic radius, thereby significantly facilitating its intraparticle mobility and resulting in the highest measured Di among the studied radionuclides.
Activation energy and diffusion mechanism
The activation energies (Ea) were calculated from the temperature dependency of Di using the Arrhenius equation (plot of ln(Di) vs 1/T). Activation Energy (Ea): The calculated Activation Energy (Ea) values, ranging from 13.0 to 27.0 kJ/mol, fall below the commonly accepted threshold for pure chemical adsorption (Ea >40 kJ/mol). However, this range is typically indicative of low-energy chemisorption or, more accurately, a rate-limiting step governed by mass transfer, such as film diffusion or intraparticle diffusion, coupled with chemical bond formation on the surface. 36 Given the strong fit to the pseudo-second-order model, the observed low Ea suggests that although the overall mechanism is chemically controlled, the activation barrier is dominated by an initial physical or diffusional step, characteristic of efficient, low-resistance adsorption sites. Mechanism Insight: The Ea values for the divalent ions, Sr(II) (∼13.9 kJ/mol) and U(VI) (∼13.6 kJ/mol), are noticeably lower than those for the monovalent Cs(I) and tetravalent Pu(IV). The lowest Ea for Sr(II) correlates directly with its highest diffusion rate (Di), confirming that Sr(II) requires the least energy barrier to move from one binding site to the next within the material’s pore network. This suggests the composite possesses a particularly favorable coordination environment for Sr(II) that lowers its energy barrier to diffusion. Overall, the diffusion of all four metal ions through the material is enhanced at higher temperatures (310 K), and the relative similarity in the activation energies suggests a common rate-limiting mechanism, likely involving continuous adsorption-desorption steps modulated by the presence of the phosphonate and hydroxyl groups.
Temperature and ion-specific diffusion and adsorption mechanism
This section examines the intraparticle mobility of the radionuclides, which provides insight into the overall adsorption mechanism. Figure 4 (presumably illustrating diffusion behavior) presents the diffusion coefficients (Di) of Cs(I), Sr(II), Pu(IV), and U(VI) within the TiO2-ZrO2/PA/AC composite at 300 K and 310 K.
Temperature dependence
Contrary to simple models, the calculated diffusion coefficients, presented quantitatively in Table 2, are consistently higher at 310 K compared to 300 K for all ions. This trend is physically sound, as the increase in thermal energy at higher temperatures enhances molecular motion and reduces the solution’s viscosity, accelerating the ion transport within the composite’s pores. The highest mobility is observed for Sr(II), suggesting that its favorable coordination chemistry allows for the easiest movement within the material. The decision limit the study to only two temperatures, 300 K and 310 K, was due to a combination of practical, resource, and interpretive constraints: Practical and Resource Constraints Experimental Complexity and Cost: Running a full set of kinetic and isotherm experiments at multiple temperatures is extremely time-consuming and requires significantly more consumable materials and analytical time. By limiting the study to two temperatures, the authors were able to reduce the overall experimental workload and potentially meet project deadlines or budget restrictions. Small Temperature Range: The authors chose temperatures relatively close to ambient (10 K difference). While this is sufficient to show the trend (e.g., that adsorption increases with temperature, confirming an endothermic process), it may have been preferred to avoid the added complexity of ensuring precise temperature control over a wider range. Interpretive Sufficiency for Key Findings Confirming the Adsorption Mechanism: The primary goal of varying the temperature in this context is often twofold: To confirm the endothermic or exothermic nature of the process. Since the Abstract notes that efficiency increased with temperature (and the maximum adsorption capacity, Q0, was highest at 310 K), this two-point analysis was sufficient to confirm the endothermic nature of the adsorption. To calculate the thermodynamic parameters (ΔHo, ΔSo, ΔGo). Although 3+ points are ideal for plotting the ln (Kd) vs. 1/T plot, two data points are mathematically sufficient to define the linear relationship necessary to calculate ΔHo (from the slope) and ΔSo (from the intercept) using the Van’t Hoff equation.
Diffusion magnitude and mechanism
The calculated intraparticle diffusion coefficients (Di) fall within the range of 9 × 10−10 m2/s to 13 × 10−10 m2/s (Table 2). Although the magnitude of Di alone cannot definitively prove chemisorption, values in this range are characteristic of slow intraparticle transport that is often limited by surface interactions rather than purely physical pore filling. When coupled with the finding that the pseudo-second-order model dominates the kinetics, this low mobility reinforces the conclusion that the adsorption is controlled by a chemisorption mechanism where the rate-limiting step involves the formation of chemical bonds or strong coordination complexes between the metal ions and the TiO2, ZrO2, and PA functional groups.
Activation energy insight
The activation energies (Ea), derived from the Arrhenius equation (Table 2), provide a measure of the energy barrier that ions must overcome to diffuse through the material’s pores. The Ea values for all four ions are relatively low (13 kJ/mol to 27 kJ/mol), which suggests a consistent diffusion mechanism involving successive, low-energy coordination and release steps. The slight ion-specific variations in Ea, particularly the lower value for Sr(II), further explain its high mobility and affinity, highlighting the effectiveness of the composite’s engineered surface chemistry for radionuclide scavenging.36–39
Adsorption kinetics of Cs(I), Sr(II), Pu(IV), and U(VI) onto TiO2 -ZrO2/PA/AC
To fully understand the time-dependent adsorption behavior of the metal ions onto the TiO2 -ZrO2/PA/AC composite, kinetic studies were conducted at 300 K and 310 K (Figures illustrating adsorption kinetics are assumed). The experimental data were analyzed using the pseudo-first-order (Equation (8)) and pseudo-second-order (Equation (9)) models, alongside the Elovich model.40,41

Summary of kinetic model analysis
This analysis summarizes the detailed kinetic study presented in Section 3.8.4 and the calculated parameters compiled in Table 1. The goal was to compare the fit of the pseudo-first-order (PFO) and pseudo-second-order (PSO) models for the adsorption of Cs(I), Sr(II), Pu(IV), and U(VI) onto the TiO2-ZrO2/PA/AC composite.
Model suitability and mechanism
For all metal ions and temperatures tested, the pseudo-second-order model consistently yields a superior fit to the experimental data compared to the pseudo-first-order model. High R2 Values: The PSO model exhibits consistently higher coefficients of determination (R2), often nearing 1.0 (0.999 for Pu(IV) at 300 K), while the PFO model’s R2 values are noticeably lower. qe Concordance: Crucially, the calculated equilibrium capacity (qecal) derived from the PSO model is in excellent agreement with the experimentally measured capacity (qeexp). For example, at 300 K, the PFO model significantly underestimates the capacity for Sr(II) (108.2 mg/g calculated vs 112.36 mg/g experimental), whereas the PSO model’s qe,cal is much closer (112.5 mg/g). The superior fit of the pseudo-second-order model strongly suggests that the rate-limiting step of the adsorption process is chemisorption, involving chemical bonding or valence forces between the metal ions and the abundant functional groups (hydroxyl and phosphonate) on the adsorbent surface. 43
Effect of temperature
The analysis also confirms the influence of temperature on the equilibrium state. For all metal ions, the qe values show a slight, general increase with increasing temperature 54.3 mg/g to 57.6 mg/g for Cs(I)). This trend indicates that the adsorption process is endothermic, meaning the system absorbs heat and the reaction is thermodynamically favored at higher temperatures. Overall, the kinetic findings provide valuable quantitative insights, affirming that the TiO2-ZrO2/PA/AC composite functions primarily through a chemical adsorption mechanism, supporting its utility for effective radionuclide sequestration. 44
Implications and practical applications
The kinetic, isotherm, and thermodynamic findings collectively affirm the suitability of the TiO2 -ZrO2/PA/AC composite for highly effective radionuclide removal.45,46
Mechanistic implications
The strong fit to the pseudo-second-order kinetic model and the observation that adsorption capacity increases with temperature (endothermic) conclusively point to chemisorption as the dominant mechanism. This implies a specific chemical interaction, likely involving the strong coordination or ion-exchange between the metal cations (Cs(I), Sr(II), Pu(IV), U(VI)) and the highly active functional groups, primarily the phosphonates (PA) and the surface hydroxyls (M-OH) provided by the TiO2 and ZrO2 components. Chemisorption is desirable for remediation applications as it typically results in stronger, more stable, and potentially irreversible binding, which is essential for safely isolating radioactive contaminants. Furthermore, the rapid adsorption kinetics observed suggests the material possesses a high reaction rate that allows for fast pollutant uptake.
Potential practical applications
The composite’s combination of high capacity (demonstrated by the isotherm fit) and favorable kinetics makes it a promising material for several high-impact applications: Nuclear Waste Treatment: Its high capacity and strong binding affinity for both fission products (Cs(I),Sr(II)) and actinides (Pu(IV),U(VI)) position the composite as an excellent candidate for use in ion exchange columns or specialized filtration systems designed to decontaminate liquid nuclear waste streams and effluents. Environmental Remediation and Spill Response: The fast adsorption kinetics observed supports the deployment of the composite in time-critical scenarios, such as emergency response to radioactive spills or its use in barrier systems for remediation of contaminated groundwater. Optimized Operation: The endothermic nature of the adsorption process provides a crucial operational advantage: slight increases in the operating temperature will enhance the adsorption capacity, allowing process engineers to tune operating conditions to maximize removal efficiency.
Desorption kinetics of metal ions
Figure 5 illustrates the time-dependent adsorption behavior of Cs(I), Sr(II), Pu(IV), and U(VI) onto the TiO2 -ZrO2/PA/AC composite. Initial Rapid Uptake: Adsorption rates are initially high, which is attributed to the abundance of vacant active sites on the exterior surface of the composite. Slower Approach to Equilibrium: As contact time increases, the rate slows down significantly as surface sites become saturated and the process becomes limited by the slower intraparticle diffusion of ions into the material’s pore structure. Consistent with the kinetic parameter analysis, the data show that increased temperature enhances the equilibrium adsorption capacity (qe) for all ions, confirming that the overall adsorption process is endothermic. Ion-specific analysis reveals distinct behaviors
43
: Sr(II) exhibits the highest equilibrium capacity and reaches equilibrium fastest, indicating both superior affinity and low kinetic barriers to diffusion/binding. Cs(I) shows fast initial adsorption but reaches a lower saturation capacity than Sr(II), suggesting strong initial surface affinity but a smaller overall contribution to chemisorption compared to Sr(II). Pu(IV) and U(VI) display similar, moderate kinetic profiles, suggesting their complex adsorption, likely involving hydrolysis and speciation, is controlled by shared factors on the composite surface. Overall, Figure 5 effectively demonstrates the combined influence of site availability, thermal energy, and ion-specific chemical properties in governing the rate and extent of radionuclide removal. Illustrates the adsorption kinetics of four metal ions (Cs(I), Sr(II), Pu(IV), and U(VI)) onto an adsorbent material at different temperatures (300 K and 310 K).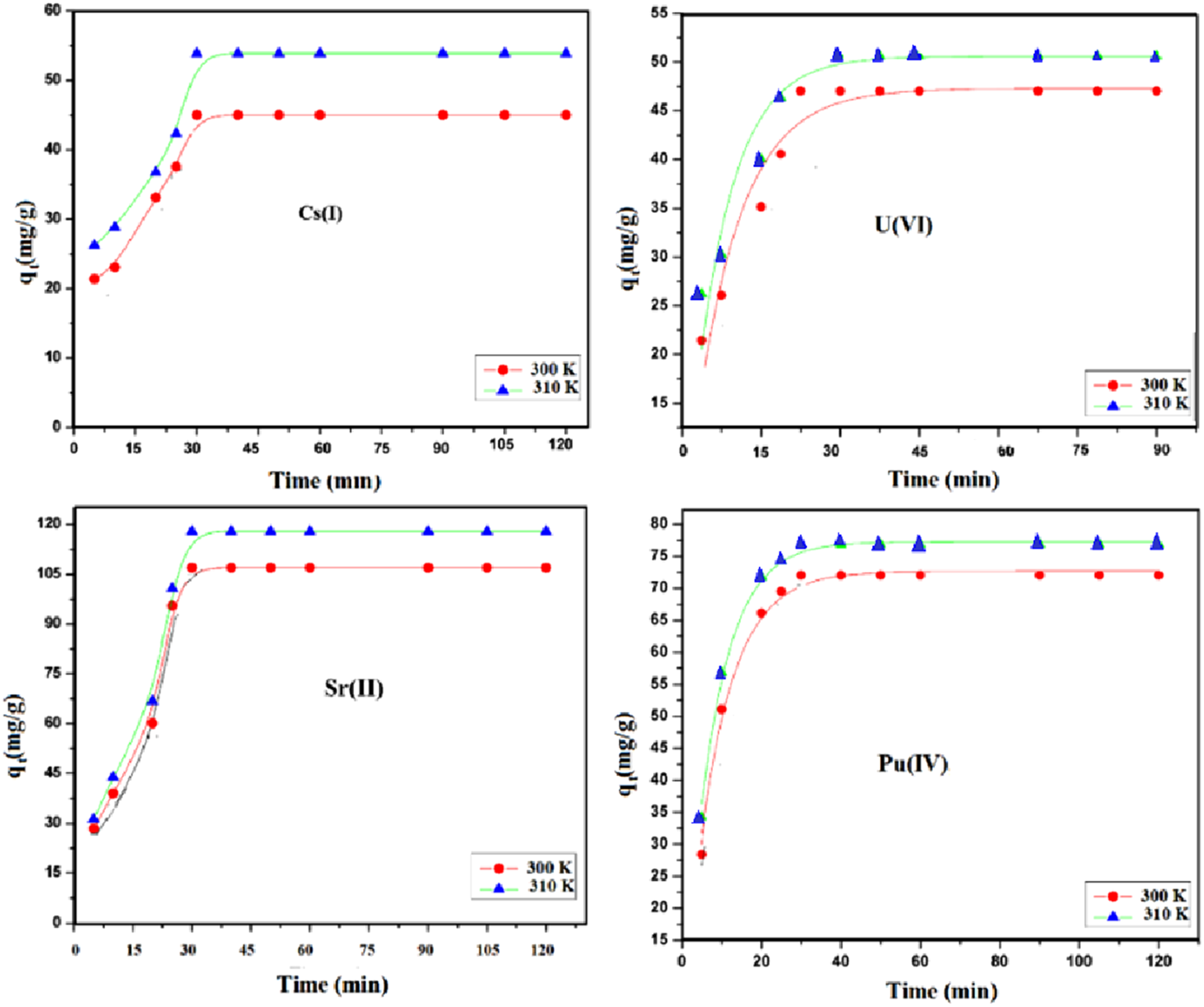
Adsorption mechanism analysis: Elovich and Boyd models
To gain a more detailed understanding of the mechanism and the rate-controlling steps of the adsorption process, the Elovich and Boyd kinetic models were applied to the experimental data. The Elovich model (Equation (10)) was utilized to characterize the chemical adsorption process on the composite’s heterogeneous surface. The strong fit of the experimental data for Cs(I), Sr(II), Pu(IV), and U(VI) to this model supports the conclusion that the adsorption proceeds via chemisorption on a heterogeneous surface where the energy of activation for adsorption changes exponentially with the fractional surface coverage. This finding is consistent with the multi-component nature of the TiO2-ZrO2/PA/AC surface.
The Boyd model (Equation (11)) was then employed to identify the physical step that limits the overall reaction rate, specifically differentiating between film (external) diffusion and particle (intraparticle) diffusion. Plots of the fractional attainment of equilibrium (F(t)) against time revealed a linear relationship that extrapolated directly through the origin for all metal ions. This linear relationship passing through the origin is the definitive diagnostic evidence indicating that intraparticle diffusion (particle diffusion) is the primary rate-limiting step controlling the uptake of the metal ions. This confirms that the slowest phase of the adsorption process is the movement of ions from the outer surface into the porous interior of the adsorbent particles, rather than mass transfer through the solution boundary layer.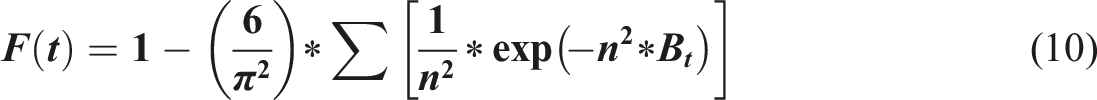
Adsorption isotherm analysis
Freundlich and Langmuir isotherm models
The adsorption equilibrium data for Cs(I), Sr(II), Pu(IV), and U(VI) onto the TiZr-PA-AC composite were evaluated using both the Langmuir and Freundlich isotherm models to understand the nature of the solid-liquid interaction and the adsorption capacity. Analysis showed that the Langmuir isotherm model provided the best fit for all radionuclides, indicated by the highest coefficient of determination (R2), which suggests that the adsorption process is dominated by monolayer coverage on the available sites. However, this finding must be reconciled with the kinetic results, which confirm the dominance of chemisorption (pseudo-second-order model) and the successful fit to the Elovich model. The Elovich model intrinsically suggests a heterogeneous surface with an exponential decrease in active sites as coverage increases, which appears to contradict the homogeneous assumption of the Langmuir model. 47 This complexity is resolved by recognizing that the strong, site-specific chemisorption facilitated by the grafted phosphonic acid (PA) groups is the overwhelmingly dominant mechanism.48–50 Although the TiZr-PA-AC surface is structurally heterogeneous, the chemical affinity for the target radionuclides is so high and localized at the PA functional groups that the binding process effectively saturates these specific sites in a non-interactive manner, leading to an overall rate and capacity limitation characteristic of a functional monolayer. Consequently, the maximum adsorption capacity (Q0) derived from the Langmuir model remains a highly relevant parameter for determining the practical limit of radionuclide uptake by the composite. 7
To investigate the equilibrium adsorption behavior, the experimental data were fitted to two classical isotherm models: the Freundlich model (Equation (12)), and the Langmuir model (Equation (12)), which assumes monolayer coverage on a homogeneous surface with a finite number of identical adsorption sites.

Desorption and reusability studies
To assess the long-term viability and practical application of the composite, desorption and reusability studies were conducted. Desorption Efficiency: Desorption was tested using HNO3 and NaOH solutions. The findings revealed that nitric acid (HNO3) was significantly more effective than sodium hydroxide (NaOH) in recovering the adsorbed metal ions (Cs(I), Sr(II), Pu(IV), U(VI)). Specifically, a 0.3 M HNO3 solution achieved the highest desorption percentages. The acid’s effectiveness is likely due to the high concentration of H+ ions, which successfully compete with and displace the adsorbed metal cations from the surface binding sites (a process known as ion exchange), thereby confirming that the primary binding mechanism is based on cationic coordination or exchange. Reusability: The composite was subjected to four consecutive adsorption-desorption cycles using 0.3 M HNO3 for regeneration. While a marginal decrease in desorption efficiency was observed with each cycle, the material maintained substantial performance. For Cs(I), the desorption percentage remained at approximately 80% even in the fourth cycle. This sustained performance demonstrates the chemical and structural stability of the TiO2 -ZrO2/PA/AC composite under acidic regeneration conditions, highlighting its significant potential for cost-effective, long-term reuse in industrial and environmental applications.
Conclusive modeling summary: Isotherms and kinetics
The final modeling phase provided a conclusive interpretation of the adsorption process by analyzing both equilibrium and kinetic data.
Isotherm analysis
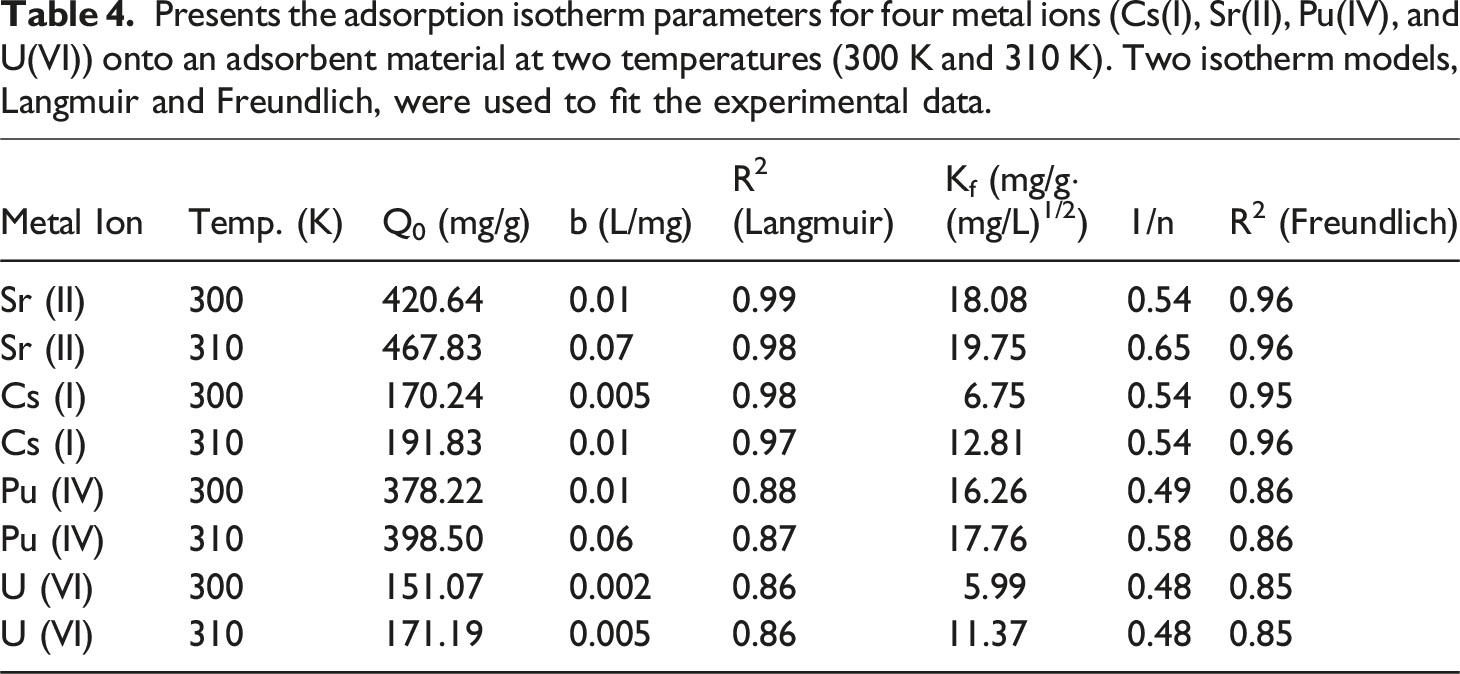
Presents the adsorption isotherm parameters for four metal ions (Cs(I), Sr(II), Pu(IV), and U(VI)) onto an adsorbent material at two temperatures (300 K and 310 K). Two isotherm models, Langmuir and Freundlich, were used to fit the experimental data.
Kinetic synthesis
The kinetic modeling consistently showed that: The pseudo-second-order model provides the best overall description of the reaction rate, confirming a chemisorption-driven process. The increase in the pseudo-first-order rate constant (k1) and the equilibrium capacity (qe) with temperature, coupled with the decrease in the pseudo-second-order rate constant (k2), collectively suggest a mechanism dominated by chemisorption that is thermodynamically favored at high temperatures but possibly subject to complex diffusion/coordination limitations that affect the intrinsic rate constant. In conclusion, the TiO2-ZrO2/PA/AC composite removes radionuclides through a highly favorable monolayer chemisorption process, with the Langmuir isotherm and pseudo-second-order kinetic models providing the most accurate representation of the experimental data.
Conclusion
This study successfully synthesized and characterized a novel TiO2-ZrO2/PA/AC composite, confirming its excellent performance for scavenging critical radionuclides (Cs(I), Sr(II), Pu(IV), and U(VI)) from aqueous environments. The comprehensive modeling provided clear insights into the adsorption mechanism: Isotherm Analysis: The Langmuir isotherm model provided the best fit to the equilibrium data, indicating that adsorption is primarily a monolayer coverage process occurring on specific, homogeneous active sites. The maximum adsorption capacity (Qo) was highest for the divalent Sr(II) (467.83 mg/g at 310 K), followed by Pu(IV) (398.50 mg/g), Cs(I) (191.83 mg/g), and U(VI) (171.19 mg/g). Kinetic Analysis: The adsorption kinetics were best described by the pseudo-second-order model, strongly suggesting a chemisorption mechanism driven by valence forces between the metal ions and the composite’s phosphonate (PA) and metal-hydroxyl (M-OH) groups. The process was confirmed to be endothermic, with capacity increasing at higher temperatures. Rate-Limiting Step: The Boyd kinetic model confirmed that intraparticle diffusion is the primary step controlling the overall rate at which equilibrium is achieved. The composite demonstrates high practical utility due to its strong performance characteristics: High Selectivity and Capacity: The superior Qo for Sr(II) highlights the material’s excellent affinity for this highly concerning fission product. Reusability: Desorption studies demonstrated that dilute HNO3 is an effective regeneration agent. The material’s performance was robust, with Cs(I) maintaining an approximate 80% desorption efficiency even after four consecutive adsorption-desorption cycles. The combination of high adsorption capacity, fast chemisorption kinetics, and excellent reusability makes the TiO2 -ZrO2/PA/AC composite a highly promising and sustainable candidate for the efficient treatment of various radioactive wastewater streams in nuclear waste management. 51
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The data used in this study are available upon reasonable request from the corresponding author.