Abstract
In this study, treated eggplant peel was used as an adsorbent to remove Pb2+ from aqueous solution. For this purpose batch adsorption experiments were performed for investigating the effect of contact time, pH, adsorbent dose, solute concentrations, and temperature. In order to assess adsorbent’s physical and chemical properties, Fourier transform infrared spectroscopy, scanning electron microscopy, and energy dispersive X-ray spectroscopy were used. The results showed that the adsorption parameters for reaching maximum removal were found to be contact time of 110 min, adsorbent dose of 0.01 g/ml, initial lead(II) concentration of 70 ppm, pH of 4, and temperature of 25°C. Moreover, for the experiments carried out at pH > 4 the removal occurred by means of significant precipitation as well as adsorption. Furthermore, these results indicated that the adsorption followed pseudo-second-order kinetics model implying that during the adsorption process strong bond between lead(II) and chemical functional groups of adsorbent surface took place. The process was described by Langmuir model (R2 = 0.99; maximum adsorption capacity 88.33 mg/g). Also thermodynamics of adsorption was studied at various temperatures and the thermodynamic parameters including equilibrium constant (K), standard enthalpy change, standard entropy change, and standard free energy changes were obtained from experimental data.
Introduction
Lead(II) can usually be found at very low concentrations in the earth’s outer surface, but the common occurrence of this heavy metal in the environment is the result of human activities. Over the centuries the unique properties of lead(II) have resulted in its widespread use in many different applications. These properties are categorized as its high resistance to corrosion, low melting point, and low conductivity. Large quantities of lead(II), both as the dioxide and the metal, are used in storage batteries, cable covering, plumbing, and ammunition. This heavy metal has been of concern for its hostile effects on human body other than cancer, its harmful neurodevelopmental effects on the embryo and young infants. Recently, International Agency for Research on Cancer has classified inorganic lead(II) compounds in group 2A (probably carcinogenic to humans) (Humans IWGotEoCRt and Cancer IAfRo, 2006).
The high concentration of lead(II) is commonly found in urban runoff; thus, sewage discharge and industrial effluents cause damage to the environment and exposure to it can adversely affect human health. Therefore, the danger of lead(II) leaching into the water and runoff has made researchers develop treatment methods to remove this notorious pollutant from wastewater. Currently, the removal of concentrated heavy metal is performed by hydroxide precipitation (the most common method as it is relatively simple to operate) (McNeill and Edwards, 1997), carbonate precipitation (the process of using sodium or calcium carbonate for soluble lead(II) reduction) (Dando and Glasson, 1989), sulfide precipitation (pH and oxidation–reduction potential must be carefully controlled to avoid the risk of producing toxic levels of hydrogen sulfide gas) (Robinson and Sum, 1980), lime precipitation (Esalah et al., 2000), adsorption by means of activated carbon (Toles and Marshall, 2002), membrane processes (Canet et al., 2002), ion exchange (Dabrowski et al., 2004), and electrocoagulation (Escobar et al., 2006), which are energy intensive and expensive due to the need for advanced equipment, skilled operators, and expensive materials. The large capital expense and energy cost have created incentive for extensive researches in this area of waste water treatment, and bioadsorption has been considered as an attractive alternative in the case of dilute wastes, practical for small- and medium-scale industries (Aksu, 2005; Periasamy and Namasivayam, 1994). Efforts to develop new metal sorbents have led to the idea of using various agricultural products, by-products, and waste fruit for the removal of heavy metal from aqueous solutions.
Friedman and Waiss (1972) and Randall et al. (1978) have investigated the efficiency of a wide variety of cellulose-based agricultural wastes and by-products as low-cost sorbents for heavy metals removal by comparing different organic waste materials, capable of removing pollutants. Moreover, they suggested that mercury uptake is roughly proportional to protein content and polyphenolic materials. Reddad et al. (2002) investigated the adsorption kinetics and equilibrium of several divalent metal ions using polysaccharide as an adsorbent. They showed that more lead(II) ions were adsorbed than other ions and the fast metal uptakes were related to the large availability of the carbonyl groups of the polysaccharide. Taty-Costodes et al. (2003) studied adsorption of heavy metal ions, (Pb(II) and Cd(II)), onto sawdust and investigated the main parameters on the sorption of lead(II) by
In this research, treated eggplant peel (TEP) (as an available, natural and inexpensive adsorbent) was used as adsorbent to remove lead(II) from aqueous solutions in a batch process. The prepared adsorbents were characterized by scanning electron microscopy (SEM), EDX, and Fourier transform infrared spectroscopy (FTIR), and the adsorption capacity for lead(II) was measured at different conditions. Thus, the influence of pH on adsorption and ion precipitation was studied. Moreover, other adsorption parameters including temperature, adsorbents dose, initial concentration of lead(II), stirring speed, and adsorbent size were optimized and reported. Finally, to fully understand the adsorption process and compare the results of this study with the findings of other researches the kinetic, isotherm, and thermodynamic studies were performed. The comparison of lead(II) removal by means of precipitation and adsorption methods has not been discussed widely in the previous literature that focused on the implementation of low-cost bioadsorbent, and therefore the aim of this research is to study the effects of precipitation and adsorption at the same time in order to find the optimum pH for lead(II) adsorption from aqueous solution and obtaining kinetics and thermodynamics parameters at optimum condition where minimum precipitation occurres.
Experimental
Preparation and analysis of adsorbent
Iran is one of the significant producer and consumer of vegetables in the Middle East and the eggplants are usually supplied from the suburb of Isfahan in Iran. Adsorbent was provided by chopping the eggplant peel including 20% eggplant’s white, spongy interior, and 80% smooth purple skin into 5 mm × 5 mm pieces (in order to measure the quantity of the spongy interior, the interior side of a sample was separated and then its weight was measured). In order to remove dust particles on the surface of prepared adsorbent, they were washed several times with double-distilled water. Then the waste eggplant peel was immersed in an HCl solution (0.01 M) to remove other impurities for 24 h. After HCl treatment, the waste eggplant peel was washed with distilled water and then immersed in 1 M NaOH solution for 48 h (Min et al., 2004). Next, they were rinsed again with distilled water and heated to 60°C for 30 h within a drying oven. The dried adsorbents were milled using grinding machine and sieved with standard mesh size into sizes of 100–1200 µm.
The adsorbent was characterized to assess its various physical and chemical properties and provide a better interpretation of the mechanism involved during the adsorption process. The surface area measurements of the adsorbents were obtained using BET method (Horiba SA-9600) with nitrogen gas at 298 K. The surface structure and morphological changes of adsorbent before and after lead(II) adsorption were analyzed by SEM (Philips XI30, the Netherlands). Furthermore, FTIR (Bruker Tensor 27, Germany) was performed on the adsorbent to provide information on the chemical functional groups. Finally, energy dispersive X-ray spectroscopy (EDX, Seron AIS 2300) was used for the surface elemental analysis of adsorbent before and after lead(II) sorption up to 10 keV. Atomic absorption (Buck Scientific products 210, USA) was implemented to find the amount of the Pb2+ concentration within water before and after adsorption process.
Preparation of aqueous solution
In order to prepare lead(II) solutions with different concentrations, lead(II) nitrate (Pb(NO3)2) salt powder (Merck, analytical grade) was dissolved in deionized water to obtain 2000 ppm stock solution. Then, the stock solution was further diluted to obtain the required concentration of lead(II) solutions for experimentations. In order to adjust pH, 0.1 M sodium hydroxide (NaOH) and 0.1 M hydrochloric acid (HCl) were used. Lead(II) concentrations in the aqueous solutions were analyzed before and after adsorption using atomic adsorption spectrometer by using standard solutions (5, 10, 15, 20, 30 ppm) prepared by appropriate dilution with double-distilled water. Moreover, high precision 0.001 g scale (Kern-EWJ) was applied to measure the weight of samples with great accuracy. Moreover, combined incubator shaker (Stuart, SI500) was used to control temperature and shaking speed. Deposition and centrifugation methods were used to separate the adsorbent from the solution after adsorption.
Batch adsorption experiments
Batch experimental process was used to investigate the effect of different parameters. To start experiment, a series of 100 ml glass flasks containing 50 ml of aqueous lead(II) ion solutions with different concentrations and specific amount of adsorbents were agitated in a mechanical shaker by continuous mixing. For the optimization of the pH, solutions containing 70 ppm lead(II), adsorbent with dosage of 0.2 g/l at various pH ranging from 1 to 10 were shaken with a constant stirring speed of 250 r/min at room temperature for 110 min. Then, the experiments of batch adsorption were carried out at different contact times, with TEP dosage of 10 and 4.0 g/l and 70 ppm Pb2+ at pH where maximum adsorption was achieved. The adsorption capacity was calculated from the initial (

Lead(II) removal was studied at different pH, constant times, temperatures, adsorbent dosages, initial concentrations of lead(II), stirring speeds, and adsorbent particle sizes.
In this study, kinetic models including pseudo-first, pseudo-second-order, and the intraparticle diffusion were used to find best fitted model for experimental data obtained at a condition with adsorbent dosage of 4 and 10 g/l, pH 4, initial lead(II) concentration of 70 ppm, contact time of 110 min, and temperature of 25°C (Sekar et al., 2004). Also in order to find the best isotherm, equilibrium data at the condition with adsorbent dosage of 10 g/l; pH 4; contact time of 110 min; and temperature of 25, 40, and 55°C were obtained and fitted to the Langmuir, Freundlich, and Temkin isotherms (Dehghani et al., 2016). Finally to observe the effect of rising temperature on the adsorption of lead(II) onto TEP, thermodynamic parameters incorporating enthalpy (ΔH0), Gibbs free energy change (ΔG0) of the adsorption, and entropy change (ΔS0) were estimated. The relative standard deviations of the analytical data were measured to be less than 4%.
Results and discussion
Surface characterizations
The SEM was applied in order to study the adsorbent’s surface, the morphology of the TEP samples, and characterize the pore sizes. Electron micrographs of the bioadsorbent before and after lead(II) adsorption are shown in Figures 1 and 2. The SEM images revealed that the surface morphologies of bioadsorbent before and after lead(II) had some obvious changes. As shown in Figure 1, the pore structure of bioadsorbent was inhomogeneous and the surface of bioadsorbent before lead(II) adsorption was rough. It can be concluded from Figure 2 that a layer of materials was covered on the surface of adsorbent which can be attributed to the adsorption of lead(II) onto bioadsorbent (Pavan et al., 2008). However, there are some obvious particles at the surface of adsorbent after adsorption process; consequently, the possible explanation was the precipitation of heavy metal on the surface of bioadsorbent (Ding et al., 2016). The surface area of the TEP was measured by BET method. The SBET of TEP was 965 m2/g showing good agreement with the result of SEM indicating high porous structure of adsorbent.

SEM images of eggplant peel before adsorption of lead(II).

SEM images of eggplant peel after adsorption of lead(II).
In order to investigate the effect of treatment process and study the chemical functional groups responsible in adsorption, FTIR analysis of adsorbent before and after treatment was applied. The FTIR spectra presented in Figure 3(a) and (b) were obtained from eggplant peel before and after alkali treatment, respectively. In Figure 3(b) the presence of broad adsorption peak in the range of 3110–3346 cm−1 and the peak at 1012 cm−1 that are representative of the hydroxyl bonds vibrating in stretching and bending motions can substantiate the existence of the hydroxyl groups on the surface of the adsorbent after treatment. The broad absorbance band (3110–3346 cm−1) is attributed to alcohol, except that it overlapped to some degree with the C–H region. Moreover, the presence of intensive absorption peaks at 1737 and 1631 cm−1 corresponding to carboxylic acids and carboxylate functional groups indicates the presence of C = O groups on the surface of eggplant peel (Davoodi et al., 2016). Therefore, the FTIR spectra clearly show the presence of the chemical functional groups, i.e. hydroxyl (Darvanjooghi and Esfahany, 2016; Darvanjooghi et al., 2016) and carboxyl groups (Guibaud et al., 2003; Gupta et al., 2011). The eggplant peels are composed of cellulose, lignin, and hemicelluloses. As it was showed in previous researches (Brodeur et al., 2011), during acid treatment large amount of lignin is removed (Jahanbaani et al., 2016). The region between 1500 and 1800 cm−1 is the carbonyl and double-bond region and it can be concluded that hydroxyl ions from NaOH (used for the alkali treatment) converted the ester in the eggplant peels (1740 cm−1) to carboxylate (1631 cm−1) which can bind lead(II). In other words, the cellulose from eggplant can be chemically modified by reacting it with sodium hydroxide in order to convert ester group to carboxylate and alcohol. The relative change in the spectral intensities of the carboxyl group in the eggplant peels treated with NaOH can be evaluated by calculating the absorption band ratios of 1631 cm−1 (carboxylate) and 1737 cm−1 (ester). The 1631/1737 ratio for treated (10.5) and untreated (27.3) eggplant peels indicated that the amount of ester in samples was decreased. Therefore, the FTIR spectra clearly show the presence of the chemical functional groups, i.e. hydroxyl and carboxyl group. Indeed, the eggplant peels are composed of cellulose, lignin, and hemicelluloses; and consequently the treatment of waste eggplant peels by NaOH shows that during the alkali treatment the molecular structure of cellulose would change to cellulose acetate (C6H10O5) (Min et al., 2004) containing more oxygen atoms (Da Silva Meireles et al., 2010).

FTIR spectrum of eggplant peel (a) before pretreatment and (b) after pretreatment.
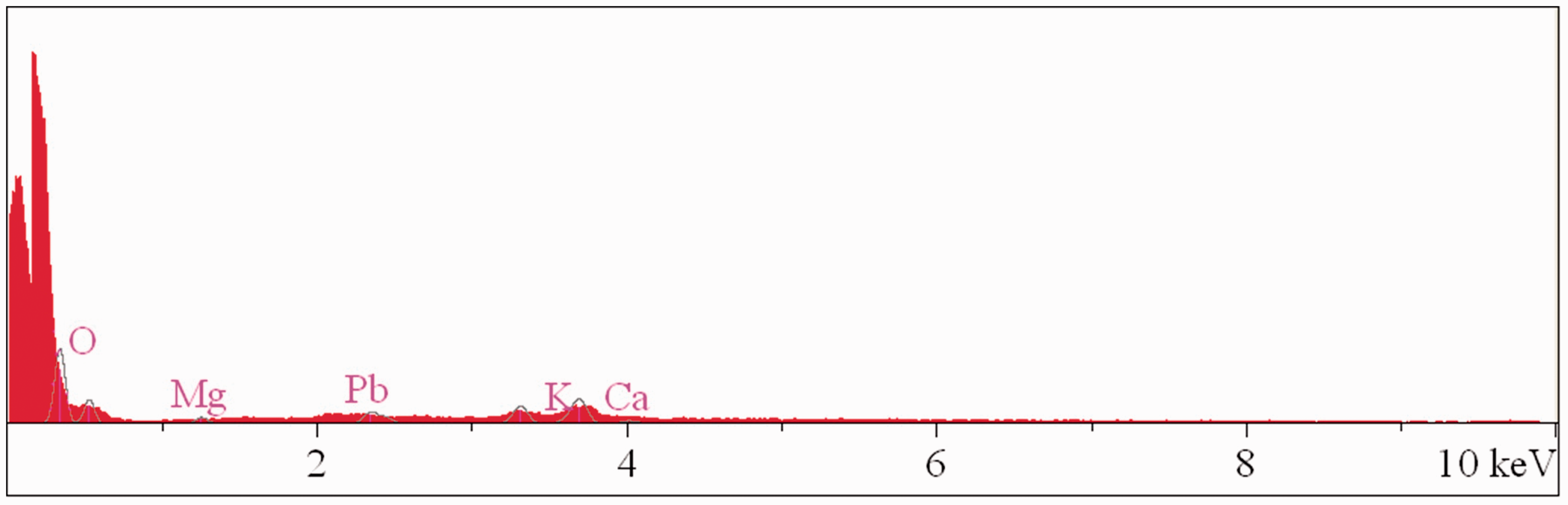
EDX taken from TEP before adsorption of lead(II).
In order to determine the chemical composition at the surface of the adsorbent before and after adsorption, EDX analysis was used (Baruah et al., 2012; Tokcaer and Yetis, 2006). Figures 5 and 6 show the elements composition (%) of Mg, Pb, K, Ca, and O on the adsorbent surface before and after adsorption, respectively. The EDX analysis indicates that the lead(II) content increases by 20% in lead(II)-loaded adsorbent and declares that TEP adsorbed lead(II) by its active groups. First, the significant increase in normalized atomic concentration of Pb(II) onto surface after adsorption led to decrease in the concentration of oxygen. Second, it can be concluded that a top layer of adsorbed lead(II) on the surface of adsorbent covers the oxygen atoms resulted in the detection of oxygen atoms with low concentration (Table 1). Moreover, the results in Table 1 show that the concentration of Calcium increased slightly on the surface of adsorbent which can be related to the small hardness in the aqueous solution during the removal process (about 0.09 wt%), as well as the potential adsorption of Ca2+ ions by using adsorbent surface; however, it is expressed that the deionized water with no high impurity was used for solutions preparation. In addition, this result might be attributed to the uncertainty of EDX test, small amount of impurities (in case of Ca2+ ions) within the deionized water, and any impurities on the surface of laboratory glasswares that could be transferred on the surface of bioadsorbent. However as the main conclusion from the results of this analysis, the significant changes in oxygen and lead(II) concentrations were observed and highlighted by using EDX test.

EDX taken from TEP after adsorption of lead(II).
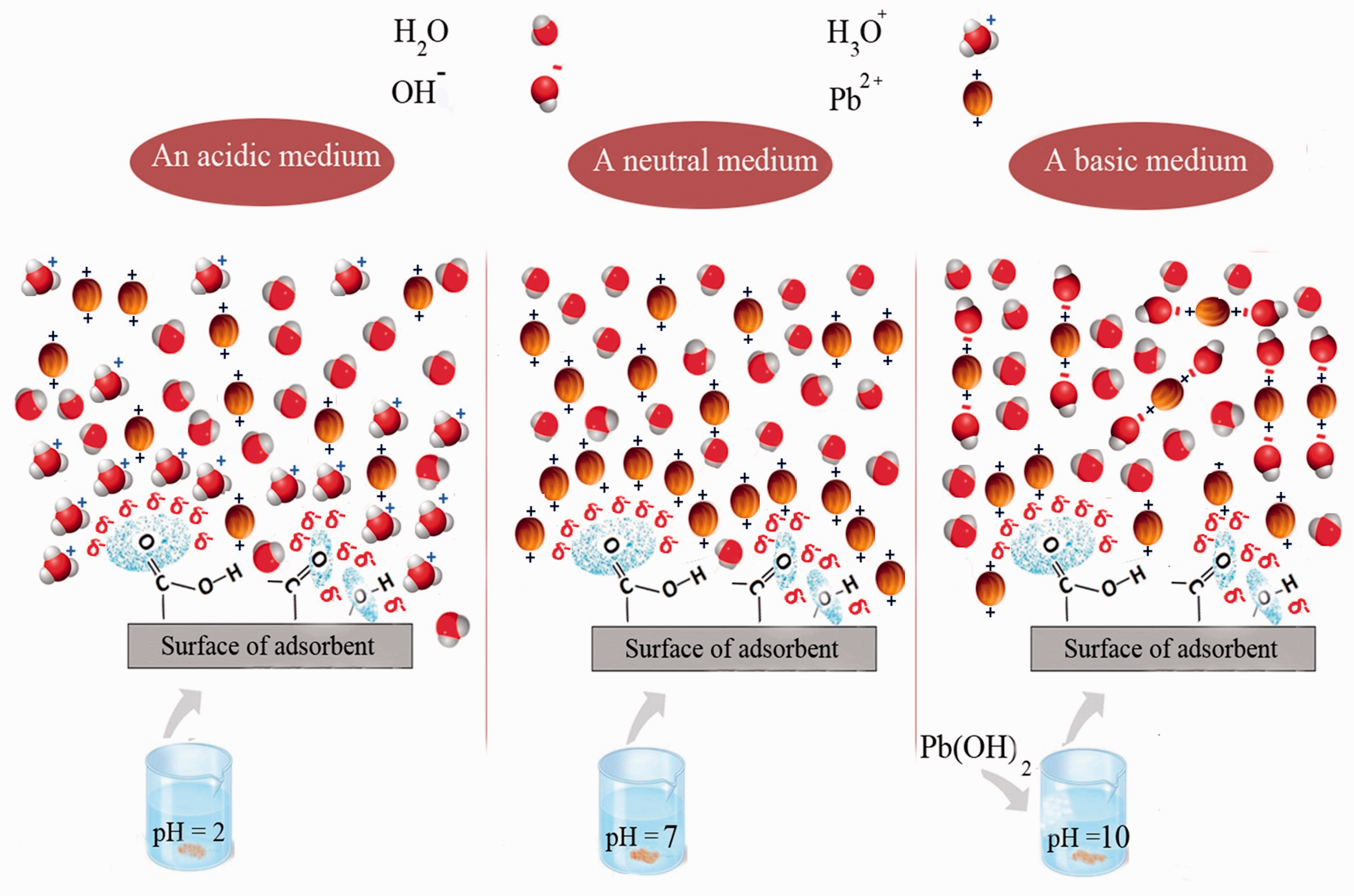
Schematic illustration for functional groups on adsorbent surface and effect of pH on lead(II) adsorption.
Elements present on the surface of TEP before and after adsorption identified by EDX.

EDX: energy dispersive X-ray spectroscopy; TEP: treated eggplant peel.
According to the result of FTIR and the results of previous researches (Jahanbaani et al., 2016; Pérez et al., 2002; Yang et al., 2007), the main components of bioadsorbents include lignin, hemicellulose, and cellulose structure and these molecules are majorly made of carbon atoms. Thus, in order to detect the element available on the surface of adsorbent before and after adsorption and see the change in their surficial compositions, it is wise to eliminate carbon atoms content at the surface of TEP and present the element content based on carbon-free composition.
Effect of pH on adsorption
The pH of aqueous solution is considered as one of the most important adsorption variable that affects the adsorption behavior and solubility of the metal ions as it determines total surface charge of the adsorbent (Dursun et al., 2003). The pH dependence of metal ions adsorption is largely related to competition between adsorption of hydronium and lead(II) ions on the same sites. In an acidic medium, the lower amount of lead(II) adsorption might be concluded due to the competition between lead(II) ions and H+ that lead to bind sites for the adsorption process (hydroxyl (–OH), carbonyl (–C = O), and carboxyl (–COOH) groups). The initial pH of solution affects both available surface sites of adsorbent and metal ions chemistry in water (Dursun et al., 2003; Faust and Aly, 2013). The oxygen at the surface of adsorbent has an available pair of electrons that can form bonds with lead(II) cation and H+ due to unoccupied atomic orbital. When functional groups are protonized at low pH, it results in passivation of the adsorption sites and suppressing the lead(II) adsorption process (Figure 6). Because the electrical attraction of lead(II) is not as stronger as hydronium in binding with the active group, at lower pH the surface contains a large number of hydronium, thus, lead(II) cations need to compete for active sites and the amount of adsorption decreases. However with the increase of pH the amount of hydronium ions decreases and more available oxygen atom at the surface of adsorbent would be achievable for lead(II) ions. Figure 7 shows the percent removal of Pb2+ at different pH (ranging from 2 to 10), which indicates lead(II) removal after pH adjustment resulting in ions precipitation as well as adsorption by means of TEP. The samples with pH 2 and initial lead(II) concentration of 70 ppm were prepared and after reaching to specific pH (from 3 to 10), the concentration of lead (II) ions before and after adsorption was determined using atomic absorption mass spectrometry. The removal of lead(II) ions before adsorption was attributed to precipitation that was calculated by comparing the concentration of samples after pH adjustment (before adsorption) with those measured at pH 2. The result of Figure 7 indicates that with the increase in pH the amount of pollutants eliminated by means of precipitation increases and for those that pH were set on 6 about 20% of ions were eliminated by this phenomenon. However with the increase in pH up to 6 more lead(II) ions are removed by means of adsorption. Furthermore, the results of this figure confirmed that the effect of pH on lead(II) removal due to precipitation (more than 5% of removal) at pH more than 4 (Dursun, 2006).

Removal of lead(II) at various initial pH (T, 25°C; adsorbent dosage, 10 g/l; initial lead(II) concentrations, 70 ppm).
These results showed that with the increase in pH from 1 to 6 the removal of Pb2+ increased and by increasing the pH from 7 to 10, a slight decrease in lead(II) adsorption was observed. In order to understand the mechanism of pH effects on adsorption and get a better comprehending, the competition between hydronium and lead(II) to be adsorbed onto active sites, the pH of solutions was measured after adsorption at various fixed initial pHs (Figure 8). These results show that with the decrease in initial pH the value of final pH increases which is indicating the number of hydronium decreases due to the adsorption by means of active sites.

Final pH of solution after lead(II) adsorption at various initial pHs (adsorbent dose of 10 g/l).
It is apparent that with the increase of pH from 7.0 to 10.0 the adsorbent surfaces become more negatively charged and electrostatic attraction from free active groups enhances the adsorption process; however, the reduction in the amount of lead(II) adsorption was observed. In order to explain the reason, the literature attributed this phenomenon to the fact that Pb(OH)2 is formed at pH more than 7, thus, the optimum pH value for adsorption is where precipitation does not occur significantly (Atieh et al., 2011; Balouch et al., 2013; Faust and Aly, 2013; Molazadeh et al., 2015). The results indicated that the maximum uptake of Pb2+ was obtained at pH 6 (similar to the results of other researches (Ertaş and Öztürk, 2013; Gupta et al., 2011; Kanjilal et al., 2015)) and Figure 8 also shows that the final pH is 6 for adsorption at condition with initial pH 6, which is indicating that no hydronium uptake has occurred during adsorption. All the following experiments on the removal of lead(II) from aqueous solution should have been carried out maintaining the solutions at pH 4 because 20% of lead(II) removal has occurred by precipitation at pH 6 and experimental data could not have been fitted by equations developed for adsorption. In other words, precipitation and adsorption are completely different process and while investigating adsorption process, it is expected that all the removal of the lead(II) ions have occurred by adsorption not precipitation. By considering the fact that the removal obtained through precipitation can lead to unreliable results, it is wise to carry out experiments at pH 4 (lead(II) precipitation can be neglected) instead of pH 6. Finally, batch adsorption experiments were performed as a function of other parameters including contact time, adsorbent dose, solute concentrations, and temperature to identify their optimized values.
Effect of contact time
The effect of contact time was studied by keeping pH, stirring speed, temperature constant for an initial lead(II) concentration of 70 ppm and adsorbent dosage of 4 and 10 g/l. The amount of lead(II) adsorbed onto TEP, as shown in Figure 9, rapidly increased with contact time until equilibrium condition was reached. For the first 20 min, the adsorption rate rose rapidly then the adsorption proceeded at a slower rate and finally it attains maximum at 110 min. Rapid increase in the adsorption during the beginning 15 min can be attributed to contacts and collision between lead(II) ions and adsorbent surface and the fact that at the beginning a large number of vacant sites are available for adsorption. Furthermore, these observations showed that the dosage of adsorbent had no effect on the required time for equilibrium; these results concerning contact time are consistent with the results of the other studies (Atieh et al., 2011; Balouch et al., 2013; Faust and Aly, 2013; Molazadeh et al., 2015).

Influence of contact time on the removal of Pb2+ with different adsorbent dosage (T, 25°C; stirring speed, 250 r/min; pH, 4; initial lead(II) concentrations, 70 ppm and temperature of 25°C).
Effect of stirring speed
The experiments were done with variable stirring speeds 50, 120, 250, 300, and 375 r/min; 10 g/l adsorbent dosage; optimized pH 4; contact time 110 min at room temperature to investigate the effect of the agitation of the sorbent/sorbate system on adsorption. Results are shown in Figure 10 and presenting that the lead(II) removal steadily increased with stirring speed up to 250 r/min, after which it decreased with increasing stirring speed insignificantly. Maximum of 75% lead(II) removal was achieved at 250 r/min and it may be explained to development in proper contact between ions and the binding sites which enhanced effective transfer of adsorbate ions onto the adsorbent sites (Baruah et al., 2012; Tokcaer and Yetis, 2006).

Removal of lead(II) using TEP at various stirring speeds (T, 25°C; adsorbent dosage, 10 g/l; pH, 4; initial lead(II) concentrations, 70 ppm).
Effect of the adsorbent particle size
Various particle sizes of adsorbents in the range 100–1100 µm were used to carry out experiments to investigate the effect of particle sizes at an initial lead(II) concentration of 70 ppm for an adsorbent dosage of 10 g/l and pH 4 (Figure 11). These studies revealed that by increasing the particle size the removal rate decreased and the particle size of 100 µm would be suitable for adsorption. The relatively higher adsorption rate for those where smaller particle size used is attributed to the increase in surface area. In other words, the reduction in particle size provided an increased surface area leading to larger amount of lead(II) ion adsorbed from the solution (Baruah et al., 2012; Tokcaer and Yetis, 2006).

Removal of lead(II) using TEP with different adsorbent particle size (T, 25°C; adsorbent dosage, 10 g/l; pH, 4; initial concentrations, 70 ppm).
Effect of temperature
The result obtained for the bioadsorbent upon varying temperature is illustrated in Figure 12, where the lead(II) removal (%) decreased with increasing temperature. Despite some of the sorbents reported in other literature (Gueu et al., 2007; Yao et al., 2014), higher level of lead(II) adsorption obtained in this study at lower temperature substantiated that lead(II) adsorption was exothermic and caused a decrease in the surface energy of the adsorbent. The decrease in the sorption capacity with increasing temperature may also be explained to the increasing tendency of lead(II) to escape from the adsorbent surface as the temperature of the solution rises (Ertaş and Öztürk, 2013; Kanjilal et al., 2015; Namdeti and Pulipati, 2014; Thajeel, 2013).
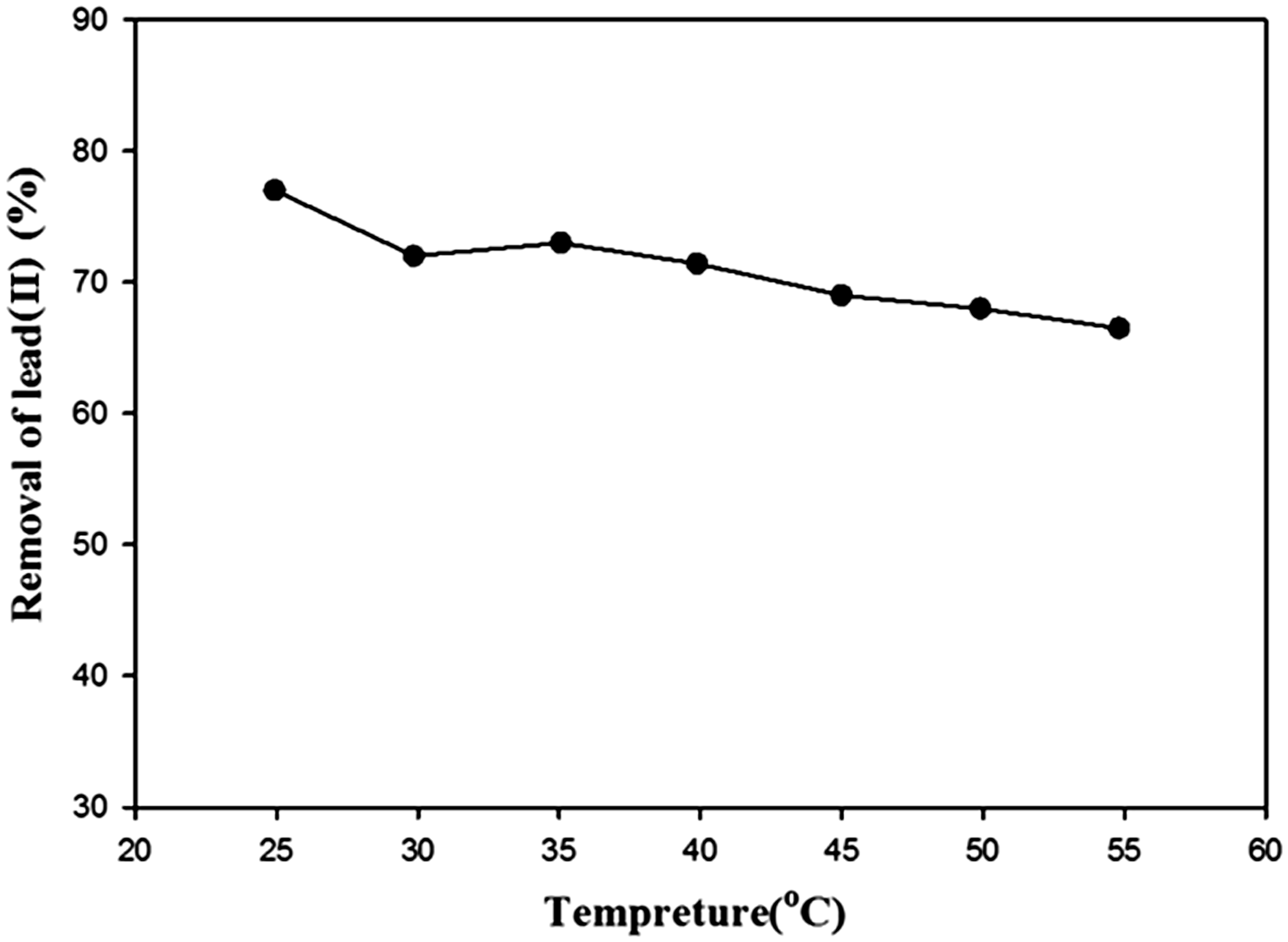
Removal of lead(II) using TEP at various temperatures (stirring speed, 250 r/min; contact time, 110 min; adsorbent dosage, 10 g/l; pH, 4; initial concentrations, 70 ppm).
Effect of adsorbent dosage
The effects of adsorbent dosage on removal of lead(II) and the rate of lead(II) uptake at equilibrium were studied with different adsorbent dosages from 2 to 20 g/l. Figure 13 shows the dependence of adsorption to adsorbent dosage. The other parameters were contact time 110 min, pH 4, and initial concentration 70 ppm. The study results revealed that the adsorption capacity decreased when the dosage increased due to the fact that the increase in adsorbent surface and the availability of more active sites made it hard for all active site to get saturated, which reduced the adsorption capacity (Figure 14). Lead(II) removal increased due to the increase in the accessibility to more active sites, when the quantity of adsorbent in the solution was too much and the adsorbent grains ended up clustering and the aggregates formed, although lead(II) removal remained almost constant with increase in adsorbent dosage above 10 g/l (Baruah et al., 2012; Tokcaer and Yetis, 2006).

Removal of lead(II) using TEP as a function of adsorbent dosage (stirring speed, 250 r/min; contact time, 110 min; pH, 4; initial concentrations, 70 ppm).

The sorption capacity of lead(II) as a function of adsorbent dosage (stirring speed, 250 r/min; contact time, 110 min; pH, 4; initial concentrations, 70 ppm).
Effect of initial Pb(II) concentration
The plot of removal efficiency versus initial concentrations of lead(II) shows that the change in concentration of the initial ions has effect on removal of this pollutant. It is likely that a given mass of adsorbent has a finite number of active sites, and that as lead(II) concentration increases, these sites become saturated. There is a concentration that produced the maximum adsorption for certain amount of adsorbent mass and adding more lead(II) ions cannot increase adsorption because no more adsorption sites are available. Another reason could have been a decrease in the proportion of covalent interactions and an increase in the proportion of electrostatic interactions at sites with a lower affinity for these ions as the initial concentrations increased. From Figure 15, it is clear that the removal of lead(II) increased with decreasing of initial concentration and maximum uptake efficiency was attained for an initial concentration of 70 ppm (Ertaş and Öztürk, 2013; Kanjilal et al., 2015; Namdeti and Pulipati, 2014; Thajeel, 2013).
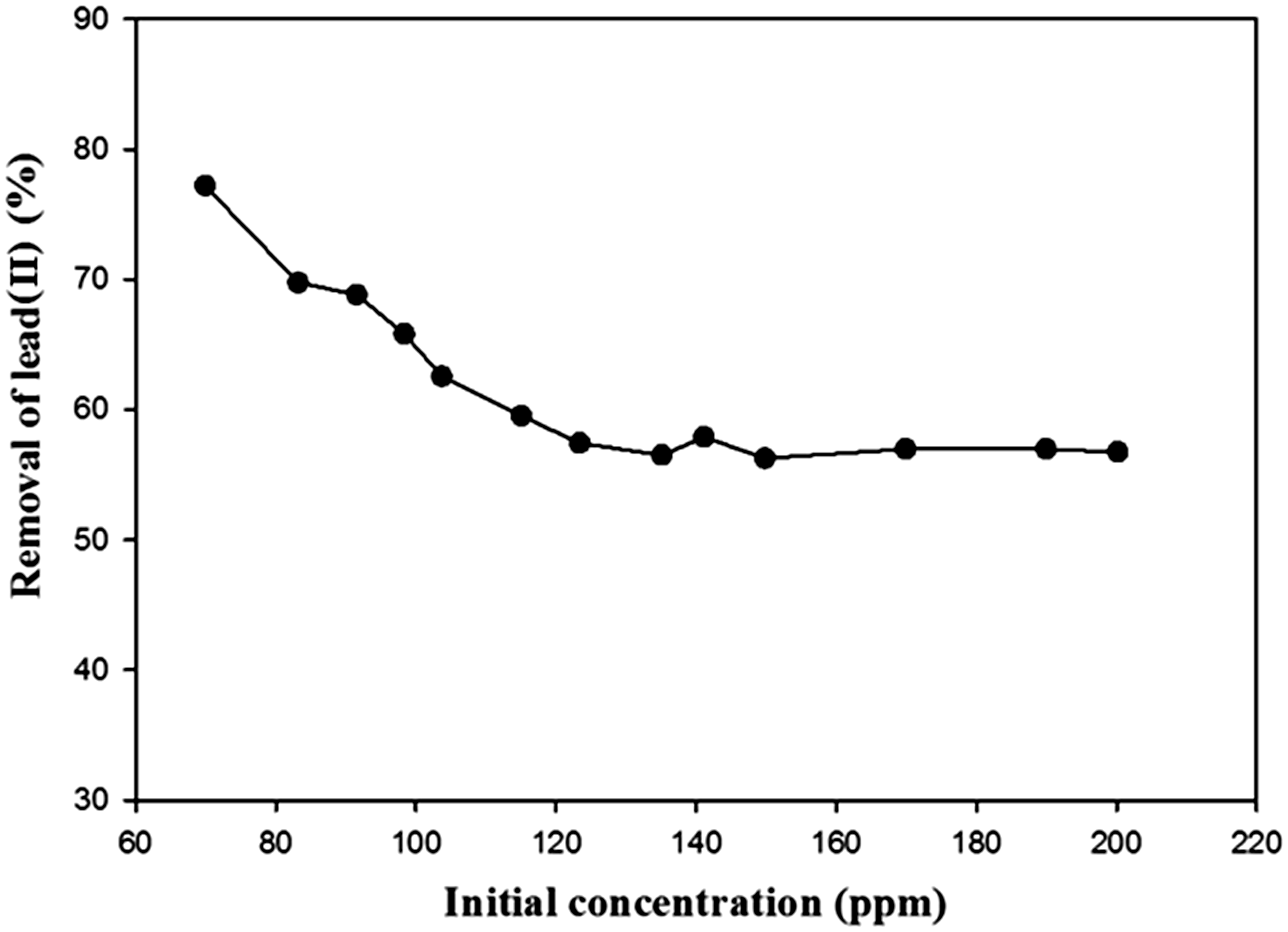
Removal of lead(II) using TEP as a function of initial ions concentration (stirring speed, 250 r/min; contact time, 110 min; pH, 4; adsorbent dosage, 10 g/l).
Adsorption isotherms
Another important method for describing a sorbent is through evaluation of equilibrium adsorption isotherms. Equilibrium adsorption isotherm indicates how lead(II) ions are partitioned between liquid phases and the adsorbent at equilibrium condition as a function of lead(II) concentration in aqueous solution. When adsorbent comes into contact with ions within the solution, the concentration of lead(II) ions on the adsorbent surface will increase until a dynamic equilibrium is reached. A wide variety of equilibrium isotherm models have been formulated to drive the adsorptions parameter for heavy metal ions on bioadsorbents. In this research the equilibrium sorption data of lead(II) on TEP were fitted to three different isotherm models including Langmuir, Freundlich, and Temkin isotherm, and values of the isotherm parameters are shown in Table 2 at three different temperatures (22, 40, and 55°C). All solutions contained a fixed mass of adsorbent under optimum adsorption condition (adsorbent dosage of 10 g/l, pH 4, contact time of 110 min) (Ertaş and Öztürk, 2013; Kanjilal et al., 2015; Namdeti and Pulipati, 2014; Thajeel, 2013).
Parameter values calculated using Langmuir, Freundlich, and Temkin adsorption models.

Langmuir isotherm
The Langmuir adsorption isotherm for removal of Pb2+ in the aqueous phase at a solution pH of 4 is shown in Figure 16(a). Equation (3) shows the linear expression of Langmuir isotherm (Özer and Dursun, 2007)



(a) Langmuir, (b) Freundlich, and (c) Temkin isotherm plot for adsorption onto TEP under optimum adsorption condition (adsorbent dosage =10 g/l, pH of 4, initial lead(II) concentration of 70 ppm, and contact time of 110 min).
RL > 1 unfavorable
RL = 1 linear
0 < RL < 1 favorable
RL = 0 irreversible
In this study the calculated RL value for adsorption of Pb(II) on the TEP adsorbent using equation (4) was found to be ranging from 0.012 to 0.342 for different temperatures. The value of RL for the results of this study showed that TEP is desirable for adsorption of lead(II) from solutions under condition where other parameters were optimized.
Freundlich isotherm
Freundlich adsorption isotherm is considered as the earliest known relationship describing the data for the nonideal and heterogeneous adsorbents. Currently, it is widely applied for organic compounds and highly interactive species on activated carbon and molecular sieves (Namdeti and Pulipati, 2014; Thajeel, 2013). Freundlich adsorption equation can be presented as

And the linear form is as follows
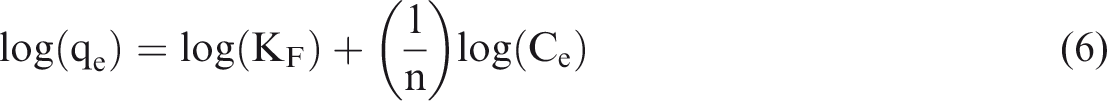
Temkin isotherm
The equilibrium sorption data of lead(II) on TEP were also fitted to Temkin isotherm model and values of the isotherm parameters are shown in Table 2. The equilibrium removal of ions can be mathematically expressed according to the Temkin model by the following equation
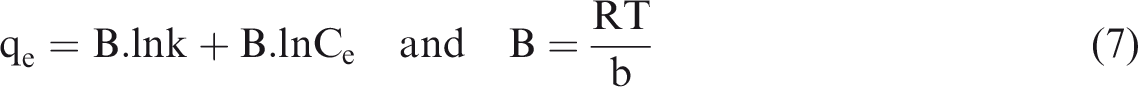
The Langmuir isotherm model assumes that the active sites possess equal affinity for pollutant ions, and a monolayer is formed on sorbent surface adsorption sites; the Freundlich isotherm can be applicable for the adsorption on heterogeneous surface sites and multilayer of the lead(II) is formed at surface sites at saturation condition. Temkin model describes the effects of indirect adsorption interaction and adsorption substances of adsorption isotherms by assuming that the heat of adsorption decreases linearly with increasing coverage and the adsorption is characterized by a uniform distribution of binding energies. The estimated model parameters of Langmuir, Freundlich, and Temkin and their corresponding linear regression correlation coefficient values (R2) at various temperatures are given in Table 2. Comparing the amount of the lead(II) adsorbed by per unit mass of TEP and R2 values showed that the linear fits using these three equations were good for studying the adsorption of lead(II) onto TEP within the used concentration range but the fit with the Langmuir equation was better. Thus, the result showed that monolayer adsorption process took place and active sites possess equal affinity for Pb2+.
Comparison of TEP with other adsorbents
The adsorption capacity of lead(II) onto TEP was compared with adsorbents which have been made of low-cost materials (Table 3). The results indicated that the maximum adsorption capacity (
Comparison of adsorption capacity of TEP for removal of Pb2+ with other adsorbents.

NR: not reported; TEP: treated eggplant peel.
As it is evident in Table 3 the amount of adsorbents for removal of Pb2+ is different and the lower value of adsorbent dosage is not the indication of the higher value of maximum adsorption capacity, for this study the adsorbent dosage was chosen as 10 g/l. Although the value of adsorbent dosage is the major parameter for comparing sorption capability of adsorbent for lead(II) uptake, the other factor that affects the adsorption is maximum adsorbent capacity. For some researches (introduced in Table 3), the value of adsorbent dosage is below 10 g/l while maximum capacity is much lower than 88.33 mg/g. As it can be seen in Table 3 other scholars also used different adsorbents (chestnut shell (Ertaş and Öztürk, 2013), Turkish low rank coal (Arpa et al., 2000),
Thermodynamics of adsorption
The effect of temperature on the sorption of lead(II) onto TEP was studied in the range of 25–55°C at the condition where other parameters were optimized (Raul et al., 2014). The adsorption standard free energy (kJ/mol) and equilibrium constant can be calculated according to

Equilibrium constant at different temperatures and various adsorbent dosages.

The effect of temperature on thermodynamic equilibrium constant is determined for adsorbent dosages of 10 and 4 g/l by (Figure 17)


Plot of ln K versus 1/T for the lead(II) adsorption via TEP.
The values of ΔH0 and ΔS0 were estimated from the slope and intercept of the linear plot of 
The average thermodynamic parameters for removal of lead(II) with adsorbent dosage of 10 and 4 g/l, that had the correlation coefficient of linear regression closer to unity, are listed in Table 5.
Thermodynamic parameters for the adsorption of lead(II) onto TEP.
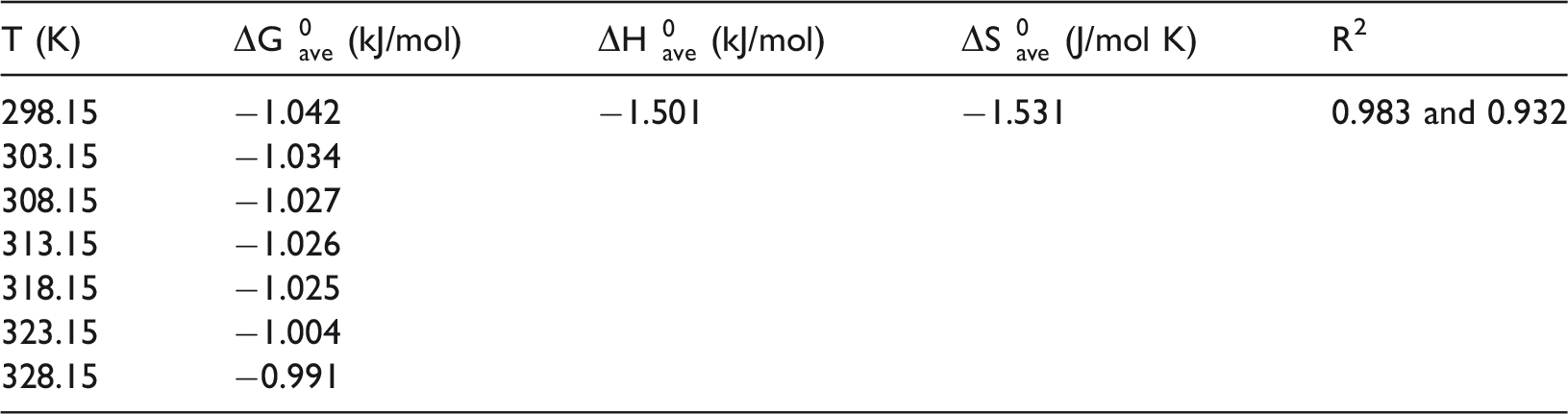
TEP: treated eggplant peel.
The calculated negative values of ΔH0 and ΔG0 revealed that the sorption process was feasible, exothermic, and spontaneous and the magnitude of the Gibbs free energy change increased with the rising temperatures. The increase in the value of ΔG0 with the increase in temperature shows that the reaction is more spontaneous at low temperature. Furthermore, the negative value of entropy change confirmed the decrease in the degree of freedom at the solution–adsorbent interface during the adsorption of lead(II) ions on the active sites of the adsorbent (chemical functional group of cellulose acetate) (Li et al., 2005; Thajeel, 2013). The negative sign of ΔH0 (−1.501 kJ/mol) for this research declared that the interaction between Pb2+ and TEP surface (Uslu and Tanyol, 2006) is exothermic and might be attributed to the decrease in the residual forces on the surface of the adsorbent (Gupta et al., 2004).
Kinetics study
Adsorption kinetics data obtained empirically were modeled using three kinetic models of pseudo-first-order kinetics equation, pseudo-second-order equation, and intraparticular diffusion models (Mouni et al., 2014).
The Lagergren pseudo-first-order kinetics
Lagergren-first-order rate equation has been widely applied and it considers the rate of occupation of active sites to be proportional to the number of unoccupied active sites (Dehghani et al., 2016). As fully described elsewhere, the pseudo-first-order equation of Lagergren is generally expressed as follows


(a) Pseudo-first-order, (b) pseudo-second-order, and (c) intraparticle diffusion plot of lead(II) adsorption onto TEP.
Parameter values calculated using the pseudo-first-order, pseudo-second-order kinetic models, and intraparticle diffusion transfer model.

Pseudo-second-order equation
If the adsorption of the lead ions onto eggplant peel is controlled by chemisorption, the rate of sorption is a second-order mechanism and is written as

The calculated pseudo-second-order parameters and the corresponding linear regression correlation coefficient value (R2) are given in Table 6. The second-order equation has the higher R2 and provides the best correlation of the data. Moreover, the calculated amount of adsorption equilibrium agrees perfectly with experimental
Intraparticle diffusion model
The so-called Weber–Morris (Weber and Morris, 1962) intraparticular mass transfer diffusion model parameters were calculated to determine if film diffusion or intraparticle diffusion is the rate-limiting step. Since the pseudo-second and first-order kinetic models cannot identify the influence of diffusion on sorption, diffusion-based kinetic models are used for calculation of the rate constants of intraparticle diffusion (Tepe et al., 2015)

If the sorption mechanism is via intraparticle diffusion then the plot of
As seen from Figure 18(c), the plots do not show the linear behavior over the whole time range, indicating that more than one process affected the adsorption. For both plots with different value of mass of adsorbent (mg/l), the adsorption of lead(II) on TEP showed a similar pattern. The three steps of process contained an initial burst adsorption that can be explained by boundary layer diffusion and the fact that the diffusion in bulk phase to the exterior surface of sorbent which started over the first period time of 15–20 min was the fastest step; moreover, secondary and tertiary (the slowest one) linear patterns with a slower rate could be explained by diffusion of ions into porous structure and adsorption into porous TEP, respectively. If the intraparticle diffusion was the only rate-limiting step of process, the plot of data passed through the origin; if not, the boundary layer diffusion limited the adsorption to some extent. The reported slope of the linear portion indicated the rate of molecular adsorption (Table 6). The lower slope could be attributed to a slower adsorption process. This implied that the intraparticle diffusion of lead(II) molecules into TEP was the rate-controlling step in the adsorption of lead(II) on TEP, particularly over long contact time periods.
Recovery of Pb2+
In order to recover adsorbed Pb(II) from the adsorbent HCl solution, with concentrations ranging from 0.001 to 0.5 M, was used. In this part experimentations were carried out with lead(II) ions concentration of 70 mg/l (50 ml of solution) and TEP dosage of 10 g/l. Then lead(II) ions were adsorbed by TEP and desorption studies were done in order to recover Pb(II) from metal ions loaded adsorbent. The results indicated that 77% of the adsorbed Pb(II) was desorbed from TEP using 0.2 M HCl and during desorption studies, according to Figure 8, the TEP surface was completely covered by hydronium. Also Figure 19 shows that the regeneration of TEP and release of Pb(II) ions from adsorbent’s surface to the solution is affordable and after removal of Pb2+ ions from adsorbent surface TEP can be used for adsorption process again. According to the results obtained from kinetics study and data fitted with pseudo-second-order kinetic equation, it has been concluded that valence forces form through exchange of electrons between adsorbate and adsorbent during adsorption of lead(II) ions by means of TEP; consequently, these forces are responsible for remaining 30% Pb(II) ions on the surface of adsorbent after desorption by HCl solutions.

Recovery of Pb2+ ions from TEP after adsorption at various HCl concentrations.
Conclusions
In this study, the sorption properties of TEP were studied for lead(II) ions removal. The results showed that the sorption capacity is strongly dependent on pH of solution. Even though it seemed that the maximum uptake of ions was obtained at pH 6, all the following experiments on adsorption of lead(II) from aqueous solution should have been carried out maintaining the solution at pH 4 on the ground that at pH 6, 20% lead(II) removal has occurred by precipitation and experimental data could not have been fitted by equations developed for adsorption. Moreover, the isotherm study confirmed the favorable uptake of lead(II) process according to Langmuir adsorption isotherms (R2=0.999) with maximum adsorption capacity in comparison with other low-cost adsorbents. The kinetic of sorption can be described by pseudo-second-order model and intraparticle diffusion model as a model of transfer; nonlinearity of plot over the time range implies that the three processes affected the adsorption. The calculated thermodynamic parameters also showed the exothermic, feasibility, and spontaneous nature of the lead(II) adsorption process using TEP.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
