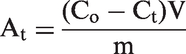Abstract
The aim of this study was to evaluate the adsorption behavior of doxorubicin on MCM-41 silica, which was synthesized by templated sol–gel method, as a function of contact time, pH of solution, and drug equilibrium concentration. Experimental kinetic curves of adsorption were compared with theoretical models of Lagergren and Ho-McKay for pseudo-first- and pseudo-second-order processes. Equilibrium adsorption of doxorubicin was analyzed by Langmuir, Freundlich, Redlich–Peterson, and Brunauer–Emmet–Teller isotherm models. These results can be the basis for direct modification of MCM-41-type silicas sorption activity to drugs.
Keywords
Introduction
Chemotherapy is a type of treatment aimed to weaken and kill cancer cells with the involvement of cytotoxic drugs. There are many types of chemotherapy agents which due to the circulation in the blood can destroy original cancer sites and any cancer cells spreaded anywhere in the body (Skeel and Khleif, 2011). Doxorubicin (Dox) belongs to the most widely used anticancer chemotherapy drugs. It is applied for medication of breast, head, liver, lung, stomach, pancreas, and other cancers (Perry, 1996). However, in addition to a number of toxic side effects typical for the majority of anticancer drugs, the long-term treatment with Dox is associated with risk of myelosuppression (Bally et al., 1990) and dose-related cardiotoxicity (Sumeet and Swati, 2014).
In order to minimize undesired damages of healthy cells without reducing therapeutic action of Dox, drug carrier systems are widely used (Aznar et al., 2011; Bernardos et al., 2010; Chen et al., 2013; Gao et al., 2011; Gu et al., 2012; Hu et al., 2013; Knezevic et al., 2011, 2013; Lee et al., 2010, 2011; Maeda et al., 2003; Meng et al., 2010; Mishra et al., 2014; Nishiyama and Kataoka, 2003; Prokopowicz and Przyjazny, 2007; Prokopowicz et al., 2016; Safari and Zarnegar, 2014; Singh et al., 2011; Tan et al., 2011; Wang et al., 2011; Yang et al., 2008; Yokoyama, 2011; Yuan et al., 2011; Zhang et al., 2011). Among them, ordered mesoporous silicas of MCM-41 type are the most promising solid supports for the formation of biologically active systems (Aznar et al., 2011; Bernardos et al., 2010; Gu et al., 2012; Hu et al., 2013; Knezevic et al., 2011, 2013; Lee et al., 2010, 2011; Meng et al., 2010; Prokopowicz et al., 2016; Yuan et al., 2011). Besides chemical stability and biocompatibility, the potential importance of MCM-41 silica materials as drug carriers is caused by their high surface area, large pore volume, uniform and tunable pores of molecular size, and controllable participation in chemical reactions with a wide range of modifiers. In order to prevent undesirable release of Dox molecules at the first contact of carrier with aqueous solution and provide their liberation only in specific time and location, stimuli-responsive structures were constructed on MCM-41 surface. In particular, effective Dox release systems, which respond to the enzyme presence (Bernardos et al., 2010), light initiation (Knezevic et al., 2011), pH change (Bernardos et al., 2010; Gu et al., 2012; Hu et al., 2013; Knezevic et al., 2011; Lee et al., 2010, 2011; Meng et al., 2010; Yuan et al., 2011), and temperature variation (Aznar et al., 2011; Hu et al., 2013), were reported. At the same time, because of the low loading efficiency of initial MCM-41 (up to 2.6 wt.%) (Kim et al., 2013), many attempts were made to enhance affinity between Dox molecules and silica surface by lining mesopore walls with functional groups (Kardys et al., 2013; Kim et al., 2013; Knezevic et al., 2011; Ma et al., 2014) or immobilization of polymer moieties on outer surface of silica particles (Kim et al., 2013).
A better understanding of the interaction mechanism of Dox with MCM-41 will provide opportunity to realize the most effective strategy for silica surface modification depending on desired dosage and release kinetics of drug. Adsorption of Dox was studied by Prokopowicz and coworkers on mesoporous xerogels with non-uniform pores (Prokopowicz and Przyjazny, 2007) as well as on mesoporous silica microparticles with ordered structure of uniform pores (Prokopowicz et al., 2016). It was found that interaction of positively charged doxorubicin hydrochloride with negatively charged silica centers (pH 6.6) leads to the drug adsorption on the surface of larger pores and its subsequent diffusion into the smaller ones. Moreover, it was proved that Dox tends to self-aggregation on silica surface.
In the present work, adsorption of antitumor antibiotic doxorubicin on MCM-41 silica was studied as a function of contact time, pH of solution, and drug equilibrium concentration. Analysis of experimental results was carried out by the use of well-known kinetic and equilibrium adsorption models.
Experimental
Materials
Tetraethyl orthosilicate (TEOS) (from Merck, purity ≥ 99%), cetyltrimethylammonium bromide (CTMAB) (from Merck, purity ≥ 97%), doxorubicin hydrochloride (from Sigma-Aldrich, purity ≥ 98%), phosphoric acid, sodium hydroxide, disubstituted sodium phosphate, and monosubstituted potassium phosphate (all from Reakhim, pure analytical), and standard volumetric solution of hydrochloric acid (from RIAP, pure analytical) were used without additional purification.
Methods
Low-temperature nitrogen adsorption–desorption isotherm of silica material was registered at T = 77 K in the region of relative pressures from 0.06 to 0.99 in increment of 0.015 with a Sorptometer Kelvin-1042. Before analysis, the silica was degassed at 423 K under vacuum for 20 h. Specific surface area (SBET) was calculated by Brunauer–Emmet–Teller (BET) equation. The pore diameters (D) and their distributions (dV/dD) were calculated by density functional theory (DFT) method. Pore volume was estimated at relative pressure of 0.99.
X-ray diffraction spectrum was registered by use of a diffractometer DRON-4-02 with monochromatic CuKα emission (λ = 1.54178 Å) and nickel filter.
The transmission electron microscopy (TEM) images of mesoporous silicas were registered on a JEM-100CXII electron microscope at 200 kV. Silica samples were deposited onto carbon-coated copper grids as holders.
Infrared (IR) spectra were recorded at room temperature using a Thermo Nicollet NEXUS FT-IR spectrophotometer in frequency range from 4000 to 1200 cm−1 with a resolution of 2 cm−1 and 32 scans for each run.
Absorption spectra of Dox buffer solutions were recorded in the 400–700 nm spectral range with a Specord M-40. Quarts cells with 2 and 20 mm path lengths were used.
Ultraviolet-visible (UV-vis) reflectance spectrum of Dox adsorbed on mesoporous silica was registered in the 400–700 nm spectral range with a Specord M-40 using BaSO4 as reference material.
pH of buffer solutions were measured by an Ionometer I-160.
The distribution diagrams of protolytic forms of Dox and silanol groups of silica surface were generated for the parameter set chosen in each simulation by the CURTIPOT program version 3.5.4.
Synthesis of MCM-41 silica
MCM-41 silica material was synthesized by base-catalyzed sol–gel condensation of TEOS in the presence of quaternary ammonium salt as structure directing agent. Condensation procedure was realized in water–ethanol–ammonia solution with CTMAB as template in accordance with previously described technique (Roik and Belyakova, 2013, 2014). The precipitated white product was kept in the reaction mixture with stirring for 2 h and then transferred to a polypropylene bottle and hydrothermally treated at 373 K for 24 h. Thereafter synthesized silica was filtered, washed with 100 ml of deionized water, and dried in air at 373 K for 3 h.
The template was removed from mesoporous channels of MCM-41 by multiple chemical extraction in acid-ethanol medium at 298 K for 24 h. The resulting mesoporous silica was filtered, washed with deionized water until the absence of halogen ions in sparge water (negative probe with silver nitrate), and dried in the air at 373 K for 5 h.
Kinetics of Dox adsorption on MCM-41 silica
The effect of contact time on Dox adsorption by MCM-41 silica was studied in phosphate buffer solutions with pH 5.0 and pH 7.0 at 293 K. The 0.01 g batches of silica material pretreated at 393 K for 2 h were placed into 5 ml of Dox phosphate buffer solutions at a concentration of 0.138 mmol/l. The suspensions were shaken certain time intervals, filtered through syringe filter with a 0.2-µm pore size hydrophilic polyvinylidene fluoride membrane. The Dox adsorption on the surface of MCM-41 silica was determined from the values of optical density of the absorption band at λ = 483 nm using the equation
Equilibrium adsorption of Dox on MCM-41 silica
Adsorption of Dox on silica surface was realized from phosphate buffer solutions with pH 5.0 and pH 7.0 using the multibatch technique under static conditions at 293 K. Briefly, 0.01 g batches of silica material pretreated at 393 K for 2 h were placed into 5 ml of Dox phosphate buffer solutions with concentration of drug ranging from 0.138 to 0.689 mmol/l. Then, the suspensions were shaken up to equilibrium attainment and filtered. The solutions were analyzed by use of spectrophotometer, and values of Dox adsorption on the silica surface were determined by optical density of absorption band with maximum at 483 nm.
Results and discussion
Characterization of MCM-41 structure
The pore structure and morphology of synthesized silica were characterized with low-temperature nitrogen adsorption–desorption, X-ray diffraction, and TEM.
The isotherm of nitrogen adsorption–desorption as well as the pore size distribution is shown in Figure 1. A linear increase in nitrogen adsorption observed at low relative pressures (<0.3) can be attributed to the monolayer formation on mesoporous walls. Specific surface area of MCM-41 evaluated from this linear region by BET method equals 995 m2/g. Inflection step localized in the isotherm at relative pressures beyond 0.3 is observed due to the capillary condensation of nitrogen inside the mesopores with narrow size distribution. Indeed, distinct maximum with high intensity centered on 3.66 nm is registered on the pore distribution curve calculated by DFT method. It is possible that the observed weak peak at 5.09 nm is caused by partial destruction of pore walls during the sol–gel synthesis in water–ethanol–ammonia medium.
Nitrogen adsorption–desorption isotherm (a) and pore size distribution (b) for MCM-41.
The pore ordering of silica material was confirmed by X-ray diffraction analysis. Three well-resolved peaks on the diffractogram of silica can be indexed to (100), (110), and (200) reflections of hexagonally arranged porous structure characteristic for MCM-41 (Figure 2).
X-ray diffraction spectrum of MCM-41 after template extraction.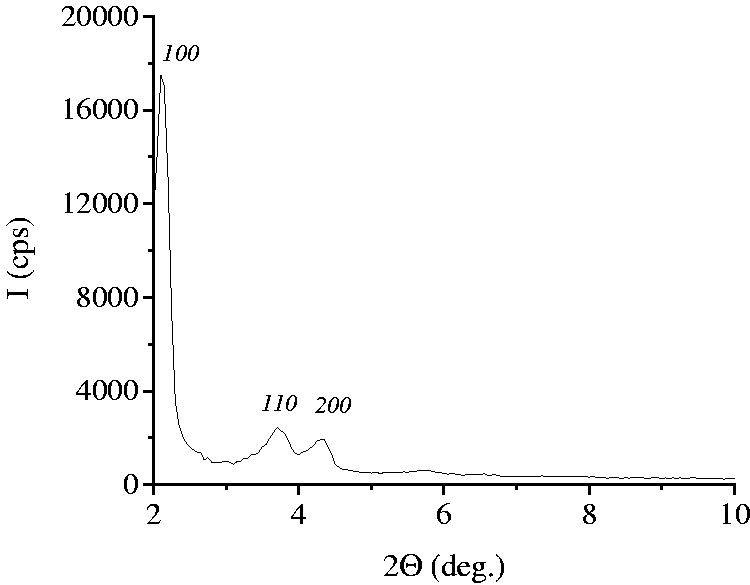
The TEM image of synthesized silica reveals the rod-like morphology of the particles (Figure 3). The pore channels are hexagonally packed and parallel to the horizontal particle axis. Open porous structure of MCM-41 provides accessibility of internal surface for adsorbate molecules.
TEM image of MCM-41, enhancement 1· × 104 (a) and 2· × 105 (b).
Kinetics of doxorubicin adsorption
Doxorubicin is an anthracycline antitumor antibiotic consisting of chromophoric anthraquinone and sugar moieties (Figure 4). Due to the presence of amino group in Dox structure, its physicochemical properties depend essentially on the pH.
Molecular structure of doxorubicin in protonated form.
As can be seen from Figure 5, non-ionized form of drug is dominant at neutral and basic pH values, whereas protonated one exists in acidic medium. Acidic environment is the most appropriate for adsorption studies due to Dox stability in the pH range from 3.0 to 6.5 (Florey, 1980). However, taking into account the chemical structure of silica surface in this pH region (Figure 5), it can be supposed that non-ionized surface silanol groups cannot contribute into the interaction with fully protonated amino groups of Dox. As a result, we cannot expect high adsorption capacity of silica surface for Dox molecules in acidic medium. Noticeable influence of interactions between protonated or non-ionized daunosamine groups and silica surface on the adsorption process is possible at neutral pH values when nearly 50% of surface silanol groups exist in ionized state (Figure 5). These ionized silanol groups are able to electrostatic interactions with the positively charged amino groups of Dox. Moreover, hydrogen bonds formation between neutral drug molecules and non-ionized silanol groups can take place contributing into the adsorption process.
Distribution diagrams of protolytic forms of Dox (a) and surface silanol groups (b) as a function of pH.
In order to investigate the mechanism of Dox adsorption on MCM-41 silica, the multibatch kinetic experiments were carried out at pH 5.0 and 7.0. As can be seen in Figure 6, adsorption capacity of MCM-41 increases during the first 6 h until equilibrium is reached. The dependences of Dox adsorption on silica surface on the time of contact were analyzed by equations of Lagergren and Ho-McKay for pseudo-first- and pseudo-second-order kinetic models (Table 1), respectively. Plots of lg(Aeq−At) (or t/At) versus t are the linear relationships for both pH of solutions (Figure 6). A relatively high correlation coefficients indicate that adsorption of Dox on silica surface at pH 5.00 and pH 7.00 can be described by pseudo-second-order kinetic model better than by pseudo-first one. Kinetic rate constants and amounts of Dox adsorbed from phosphate buffer solutions at equilibrium were calculated from the slope and intercept of linear plots (Figure 6, Table 1). It was found that despite of the well fitting by pseudo-first-order kinetic model for the whole range of contact time, the values of Aeq evaluated by the Ho-McKay equation agree with the experimental data better than those obtained by Lagergren equation. Obviously, this discrepancy is generated by the necessity to specify the equilibrium sorption capacity in the linear equation of the pseudo-first-order kinetic model. At the same time, fitting of linear form of pseudo-second-order kinetic model does not require assigning the Aeq and provides better agreement of calculated values with the experimental data.
Kinetic curves of Dox adsorption on MCM-41 silica at pH 5.0 (a) and pH 7.0 (b). The insets show the linear plots of pseudo-first- and pseudo-second-order kinetic equations. Kinetic parameters for Dox adsorption on MCM-41 calculated using pseudo-first- and pseudo-second-order kinetic models. Note. Aeq and At: the adsorption capacity at equilibrium and at time t, µmol/g; k1 and k2: the rate constants of pseudo-first- and pseudo-second-order adsorption processes, 1/min and g/µmol·min, respectively.

Isotherms of doxorubicin adsorption
Adsorption isotherms, reflecting distribution of Dox between the liquid and solid phase at various concentrations of drug in phosphate buffer solutions with pH 5.0 and pH 7.0, are represented in Figures 7 and 8. It can be seen that at pH 5.0 the adsorption grows with the increase of Dox content in the whole range of concentrations (Figure 7(a)), and at pH 7.0 the amount of adsorbed Dox increases rapidly in the initial branch of isotherm and reaches a plateau (0.1 mmol Dox/g silica) and then it again grows (Figure 8(a)).
Isotherms of Dox adsorption on MCM-41 silica at pH 5.0 obtained from multibatch adsorption experiment (a) and calculated with Freundlich (curve 1) and Redlich–Peterson (curve 2) models (b). Isotherms of Dox adsorption on MCM-41 silica at pH 7.0 obtained from multibatch adsorption experiment (a) and calculated with Redlich–Peterson (curve 1) and Brunauer–Emmet–Teller (curve 2) models (b).

Parameters of Dox adsorption on MCM-41 calculated using Langmuir and Freundlich models.

Note. Ceq: the concentration of Dox at equilibrium, mmol/l; KL: the Langmuir constant related to the energy of adsorption, 1/mmol; Am: the monolayer adsorption capacity, mmol/g; KF: the Freundlich constant which corresponds to adsorption capacity, 1/g; n: the Freundlich constant which corresponds to adsorption intensity.
It is known that Langmuir isotherm model is valid for monolayer adsorption on finite number of identical sites. At the same time, Freundlich one describes the adsorption on non-uniformly distributed sites of heterogeneous surface which have a different affinity to the adsorbate molecules. To determine adsorption constants for these two-parameter isotherm models, the linear forms of Langmuir and Freundlich equations were used. The parameters of Dox adsorption on MCM-41 surface at different pH values were calculated from the slope and intercept of linear plots (Table 2). Obtained results reveal that the Dox adsorption at pH 5.0 can be better described by Freundlich adsorption model with higher value of correlation coefficient (R2 = 0.976) compared to that for Langmuir model (R2 = 0.429). In the case of Dox adsorption at pH 7.0, correlation coefficients of Langmuir and Freundlich linear plots are too low to ascribe the occurring adsorption process to one of these two models (Table 2).
Parameters of Dox adsorption on MCM-41 calculated using Redlich–Peterson and Brunauer–Emmet–Teller models.

Note. KR and aR, the Redlich–Peterson isotherm constants, l/g and l/mmol, respectively; B: the exponent, which lies between 0 and 1; KS: the equilibrium constant of adsorbate–adsorbent interactions, l/mmol; KL: the equilibrium constant for surface adsorption–desorption, l/mmol; Am: the monolayer adsorption capacity, mmol/g.
BET model was originally developed for gas multilayer adsorption description. Therefore, direct application of BET theory to liquid–solid phase process leads to erroneous results. In the present paper, corrected form of the BET equation proposed by authors (Ebadi et al., 2009; Gritti and Guiochon, 2003) was used to model the adsorption of Dox on MCM-41 from the phosphate buffer solutions. As in the case of Redlich–Peterson model, adsorption parameters of BET equation were obtained from non-linear regression analysis. The highest value of correlation coefficient indicates that BET model describes adsorption of Dox on MCM-41 surface from phosphate buffer solution with pH 7.0 better than previously discussed theories (Table 3). The monolayer adsorption capacity of MCM-41 calculated using BET adsorption model is in a good agreement with experimental results (Figure 8, Table 3).
In order to give the visual assessment of different isotherm models and their ability to correlate with experimental results, the theoretical plots from the most appropriate isotherms have been fitted and compared with the experimental data for Dox adsorption on MCM-41. It can be seen that experimental data of Dox equilibrium adsorption on MCM-41 from phosphate buffer solution with pH 5.0 fit well to isotherm calculated according to Freundlich model (Figure 7), whereas liquid–solid BET model is the most appropriate for description of Dox adsorption on MCM-41 at pH 7.0 (Figure 8).
Taking into account chemical structure of doxorubicin and silica surface, it can be suggested that adsorption is driven mainly by physical interactions between amino groups of Dox macromolecules and silanol groups of silica surface. At pH 7.0 nearly 56% of surface silanol groups exist in ionized state, whereas 93% of Dox daunosamine groups are protonated (Figure 8). Ionized silanol groups are able to electrostatic interactions with the positively charged amino groups of Dox. Moreover, residual unprotonated daunosamine groups contribute into the adsorption process due to the hydrogen bonds formation with silanol groups of MCM-41. Contrary situation is observed at pH 5.0 when non-ionized surface silanol groups lose their ability to interact with fully protonated aminogroups of Dox (Figure 7). As a result, substantial lowering of MCM-41 adsorption capacity is registered at pH decreasing (Figures 7 and 8).
In addition to the direct interactions of adsorbate–adsorbent, cooperative forces between molecules of Dox play an important role in adsorption. It has been reported (Agrawal et al., 2009; Anand et al., 2012; Fulop et al., 2013; Hayakawa et al., 1991; Li et al., 1998; Porumb, 1978; Rizzo et al., 1989) that Dox tends to self-assemble in aqueous solutions forming dimers or higher order oligomers. The extent of agglomeration is highly sensitive to concentration of drug macromolecules, acidity of solution, and presence of additives (Anand et al., 2012; Hayakawa et al., 1991; Porumb, 1978). Self-assembling of Dox molecules leads to the changes in the relative intensities of absorption peaks in the visible region of spectra (Figure 9). It can be seen that in 8 × 10−6 mol/l Dox solution, when drug exists only as monomer (Agrawal et al., 2009; Rizzo et al., 1989), absorption band with λ = 483 nm is less intense in comparison with the 498 nm one (Figure 9(a)). In a more concentrated solution, intensity of absorption bands at 483 and 498 nm becomes the same (Figure 9(b)). Such changes in the spectrum are assigned to the formation of dimers and oligomers of Dox. It can be assumed that stacking of the aglycone moieties of Dox on the silica surface occurs as well as in a solution (Agrawal et al., 2009), when parallel or antiparallel orientation of aromatic chromophores takes place. According to the results obtained by diffuse reflectance UV–vis spectroscopy, immobilization of Dox on silica surface at pH 7.0 leads to the red shift of the absorption band from 483 to 491 nm, whereas the intensity of absorption peaks at 491 and 503 nm becomes the same (Figure 9(c)). This tendency can be attributed to the π−π stacking interactions between adsorbed Dox molecules (Porumb, 1978). So, the formation of Dox polylayers on silica surface at pH 7.0 is due to the hydrophobic and π−π interactions between adsorbed Dox molecules and supplied from solution ones. Phosphate buffer medium may contribute to the adsorption process, as self-association of Dox is facilitated at ionic strength increasing (Hayakawa et al., 1991). Perhaps, phosphate ions promote stacking of the chromophores due to the screening of the positively charged Dox molecules or formation of intermolecular bonds (Porumb, 1978). Taking into consideration the geometry of pore structure of MCM-41, the formation of Dox polylayers mainly on the external surface of silica particles is quite probable (Figure 10). Polylayers organization does not occur at pH decreasing, when content of adsorbed Dox is significantly less than is necessary for the formation of a monolayer (Figure 10).
Absorption spectra of 8 × 10−6 mol/l Dox solution in water (a), 2.7 × 10−4 mol/l Dox solution in phosphate buffer (b) (quarts cells with 20 mm path length), and diffuse reflectance spectrum of Dox adsorbed on MCM-41 silica (c). Schematic representation of doxorubicin adsorption on MCM-41 silica at pH 5.0 and pH 7.0.

IR spectral studies
Physical adsorption of Dox on MCM-41 surface was confirmed by IR spectroscopy. The IR spectra of Dox, MCM-41 before and after drug adsorption are shown in Figure 11. In the IR spectrum of Dox, the absorption bands at 3000–2500 cm−1 corresponding to the valence vibrations of the C–H bonds in the CH2 and CH3 groups are registered (Figure 11, curve 1). Characteristic absorption band with maximum at 1729 cm−1 is caused by stretching vibrations of the C=O bond in the hydroxyacetyl group of Dox. Whereas absorption bands at 1616 and 1583 cm−1 belong to the stretching vibrations of the C=O bonds incorporated in anthracene ring. It should be noted that the absorption bands attributed to the deformation vibrations of the N–H bonds in the primary amino group of Dox usually occur at 1560–1640 cm−1 (Nakanishi, 1962). However, identification of amino groups in Dox is complicated by the overlapping of their signals with characteristic absorption bands of carbonyl groups. The band at the lower frequencies with maximum at 1414 cm−1 is assigned to the deformation vibrations of the C–H bonds in the CH, CH2, and CH3 groups as well as in-plane deformation vibrations of the O–H bonds in the hydroxyl groups of Dox (Figure 11, curve 1).
IR spectra of Dox (1), MCM-41 before (2), and after (3) Dox adsorption.
In the IR spectrum of MCM-41 after template extraction, the broad band attributed to the stretching vibrations of the O–H bonds in the surface silanols and adsorbed water molecules is appeared at 3650–3800 cm−1 (Figure 11, curve 2). The absorption band with maximum at 1640 cm−1 is attributed to the deformation vibrations of the O–H bond in the surface-adsorbed water, whereas the band at 960 cm−1 is assigned to the stretching vibrations of the Si–OH bonds in the silanol groups of mesoporous silica. Absence of absorption bands belonging to the valence or deformation vibrations of the C–H bonds in the CH2 and CH3 groups of template molecules confirms the complete removal of CTMAB from mesoporous channels of MCM-41.
Adsorption of Dox on silica surface causes appearance in the IR spectrum of MCM-41 absorption bands with maxima at 2929 and 2858 cm−1 belonging to the valence vibrations of the C–H bonds in the CH2 and CH3 groups of drug (Figure 11, curve 3). The absorption band with maximum at 1640 cm−1 attributed to the deformation vibrations of the O–H bond in the surface-adsorbed water gains a complex nature with shoulders in the low- and high-frequency regions. Their appearance is caused by stretching vibrations of the C=O bonds in the carbonyl groups at the 5-, 12-, and 13-keto positions (Figure 4), respectively. Moreover, the absorption bands with maxima at 1478 and 1415 cm−1 belonging to the deformation vibrations of the C–H bonds in the hydrocarbon groups of adsorbate are observed (Figure 11, curve 3). Obtained IR spectra demonstrate the successful adsorption of Dox and its chemical stability on silica surface.
Conclusions
Adsorption of doxorubicin on mesoporous silica nanoparticles of MCM-41 type was studied depending on contact time, pH of phosphate buffer solution, and drug concentration. It was proved that the kinetics of Dox adsorption on silica surface at pH 5.0 and pH 7.0 agrees with the kinetic model of pseudo-second order. It was found that BET model is the most appropriate for prediction of Dox equilibrium adsorption on MCM-41 silica from phosphate buffer solution with pH 7.0, whereas experimental data obtained at pH 5.0 can be described by Freundlich model. Obtained results can be useful in prediction of the most suitable way of silica surface modification depending on desired dosage and release kinetics of Dox.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.