Abstract
In this paper, transparent polyvinyl butyral/antimony oxide (PVB/Sb2O3) nanocomposites with good UV shielding effectiveness are prepared. Solution casting was used to produce PVB/Sb2O3 nanocomposites with different Sb2O3 percentages. XRD and FTIR confirmed the interaction between PVB and Sb2O3. SEM and EDS mapping revealed that Sb2O3 nanoparticles with an irregular polygonal structure have a Sb/O ratio of ∼0.44. The integration of Sb2O3 resulted in a measurable alteration in PVB’s linear and nonlinear optical characteristics. For example, the reduction in transmittance of PVB was detected upon increasing the nanofiller content. Increasing the Sb2O3 ratio resulted in a reduction of the energy band gap of PVB, attributed to changes in localized energy states and defect levels within the host PVB’s band gap. Increasing the amount of Sb2O3 causes a rise in the refractive index, dispersion energy, static refractive index, and dielectric constant of nanocomposites. The results show that Sb2O3 can alter the optical properties of PVB, enabling it to be utilized in various applications, including UV protection, optical cables, anti-reflective films, and optoelectronic devices.
Introduction
Polymer nanocomposites are essential in the discovery of innovative products utilized in various technological applications.1–4 The polymer nanocomposites predominantly comprise a polymeric medium infused with nanofillers of low aspect ratios. Nanofillers, derived from both organic and inorganic materials, have numerous applications in various fields, including energy storage, radiation shielding, optoelectronics, photocatalysis, quantum nanoelectronics, and biomedical engineering.5–14
Among diverse polymeric materials, polyvinyl butyral (PVB) is regarded as a compelling candidate for the fabrication of polymer nanocomposites across various applications owing to its flexible molecular structure, low glass-transition temperature, superior binding strength, high optical clarity, exceptional mechanical strength, favorable biocompatibility, hydrophilic characteristics, and significant adhesive properties with glass and metal.5,15 PVB was used to increase the stability of copper and nickel-based electromagnetic shielding fabrics. 16 A composite made from PVB and mica flakes was used as an effective barrier coating to protect organic solar cells protected against photobleaching made of poly (3-hexylthiophene). 17
Polymer composites may benefit from the addition of metal oxide nanoparticles due to these particles’ remarkable optical and antibacterial properties, as well as their enhanced mechanical strength, chemical resistance, and thermal stability. Their tiny stature and large surface area give them unique characteristics. 18
Antimony oxide (Sb2O3) is classified as a heavy metal oxide and has a high refractive index, excellent photothermal behavior, and transparency to infrared light. Additionally, Sb2O3 contributes to the semiconductor industry. 19 Furthermore, Sb2O3 has a large and direct band gap in the near-ultraviolet region, which is beneficial for high-efficiency flame-retardant and UV shielding applications. 20
PVDF-CTFE fibrous membrane was modified with Sb2O3 for lithium-ion batteries. A small amount of Sb2O3 enhanced the mechanical, flame retardancy, and thermal stability of PVDF-CTFE. 21
An improvement in tensile strength, resistance of water vapor permeability, and UV absorbance of glycerol-plasticized starch by Sb2O3 nanoparticles prepared via carboxymethyl cellulose sodium as stabilizing agent was reported. 20
The solution casting technique has several advantages, particularly the capacity to control film thickness and its ease of use at the laboratory level. This technique faces numerous challenges that restrict large-scale production, such as excessive organic solvent usage, prolonged manufacturing duration, and the nonhomogeneous distribution of fillers within the polymer matrix. 22
In this work, the impact of Sb2O3 on the structural and linear/nonlinear optical features of PVB was studied for the first time. The PVB/Sb2O3 nanocomposites were synthesized by the solution casting method. Different PVB/Sb2O3 nanocomposites were prepared by incorporating different small ratios of Sb2O3 (1, 2, and 3 wt%) into the PVB host polymer. The samples’ characterization was performed via XRD, FTIR, SEM, EDX, and UV-Vis spectroscopy. The investigation will focus on the behavior of structural, linear, and nonlinear optical characteristics, including the optical energy gap, optical susceptibility, dispersion energy, linear refractive index, and oscillator strength of various nanocomposites.
Experimental
Perpetration of PVB/Sb2O3 nanocomposites
Antimony (III) oxide (Sb2O3) powder and PVB were purchased from Sigma-Aldrich. The PVB/Sb2O3 nanocomposites were fabricated by dissolving 0.2 g of PVB in 5 mL of alcohol. Magnetic agitation was used to stir the solution at the surrounding temperature for 20 min to ensure homogeneity. The PVB solution was doped with Sb2O3 at variable concentrations (0, 1, 2, and 3) wt%. The solution casting procedure adhered the resultant solutions to glass substrates. The PVB/Sb2O3 nanocomposites were cured for 2 days at ambient temperature. The thickness of the nanocomposites was measured using a digital thickness Gauge (approximately 700 ± 20 μm).
Characterization techniques
The XRD pattern of PVB/Sb2O3 was measured using the D8 Bruker with the X-ray generator using the Cu-Kα, λ= 1.54 Å. The FTIR spectrometer, a Bruker Alpha, was successfully used to acquire FTIR spectra. SEM and EDX (Zeiss) were used to determine surface morphology and elemental composition. The UV–Visible spectrophotometer (operating from 190 to 2500 nm) was used to measure the spectra of absorbance and reflection.
Results and discussion
SEM and EDS of Sb2O3 nanoparticles
The morphology and composition of pristine Sb2O3 nanoparticles used in this work were surveyed by SEM and EDS. As revealed in SEM images (see Figure 1(a)), Sb2O3 exhibits an irregular polygon structure with different sizes. SEM image at high and low magnification (a), SEM and its corresponding EDS mapping (b), EDS profile of Sb2O3 nanoparticles (c).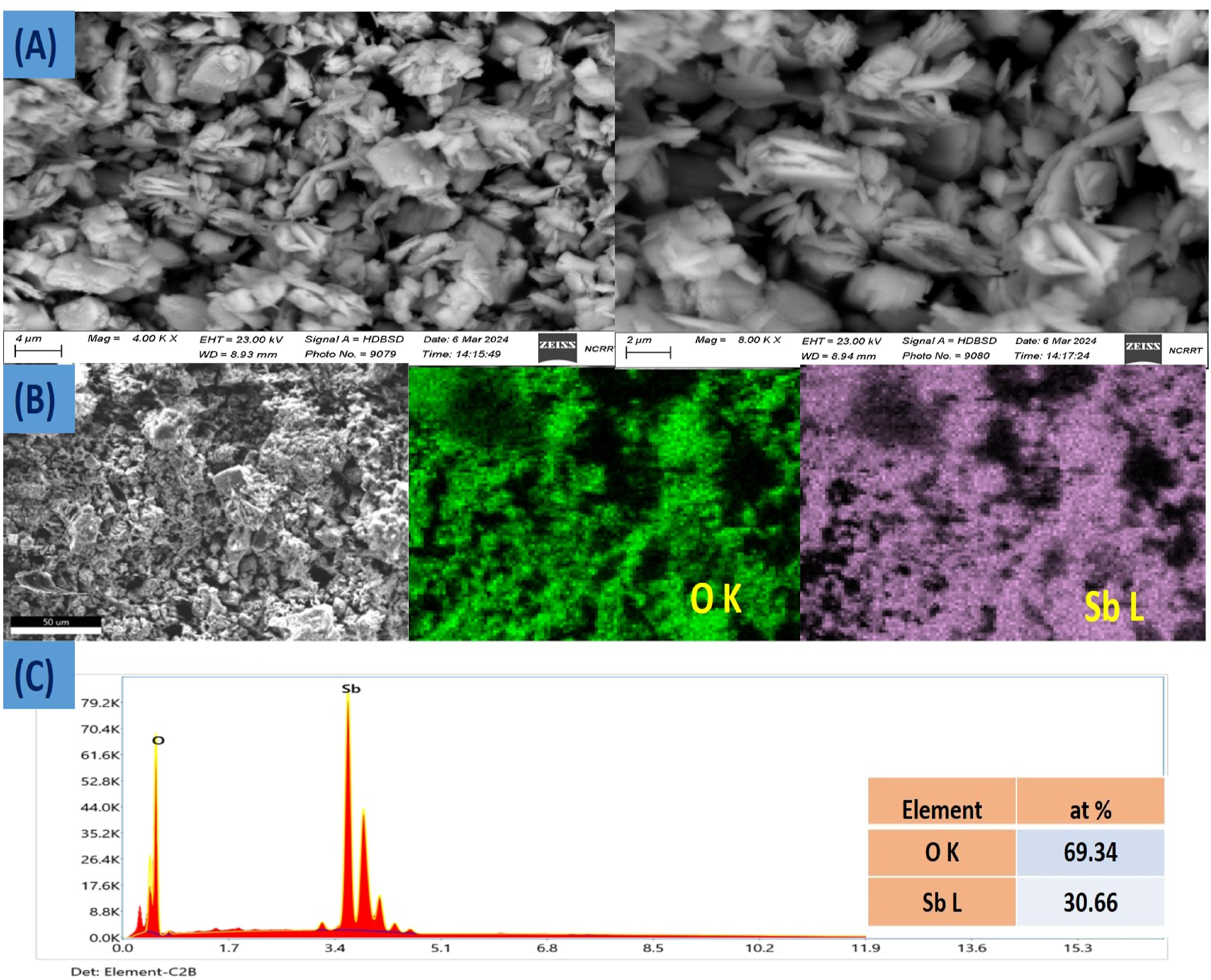
Figure 1(b) shows the low magnification SEM picture and EDS elemental mapping of the specimen. Sb2O3 contains O and Sb, which are evenly distributed across the surface of the product.
Figure 1(c) provides evidence of a typical Sb2O3 EDS pattern. The Sb and O peaks are readily apparent. 23 Quantitative EDS analysis also shows that antimony and oxygen have atomic composition values of around 30.66% and 69.3%, respectively. This indicates that the atomic ratio of Sb to O is 0.44.
XRD
XRD patterns of pure PVB and Sb2O3 are presented in Figure 2. The XRD of pure Sb2O3 show peaks at 19.02°, 25.17°, 28.11°, 32.65°, 33.57°, 36.2°, 43.94°, 46.79°, 50.3°, 54.5°, 58.6°, 60.57°, and 66.5° which corresponded to (1 1 0), (1 1 1), (1 2 1), (0 0 2), (0 1 2), (2 0 0), (0 4 2), (2 4 0), (1 6 1), (1 7 0), (2 4 2), (0 7 2), and (2 7 1) planes which well consistent with orthorhombic Sb2O3, the peaks were well consistent with JCPDS card No. 11-0689.
24
Pristine PVB exhibits a large, broad, and intense diffraction peak at 2θ = 19.42o with an interplanar spacing of 0.46 nm. It is attributable to a polymer’s amorphous phase.
25
Upon inclusion of Sb2O3 into the PVB, both diffraction peaks of PVB and Sb2O3 were observed. As well as, the peak intensity of PVB is decreased upon increasing Sb2O3 concentration. This behavior indicates that the interaction of filler with PVB leads to an increase in the crystallinity of nanocomposites.
7
XRD of Sb2O3 and different PVB-Sb2O3 nanocomposites.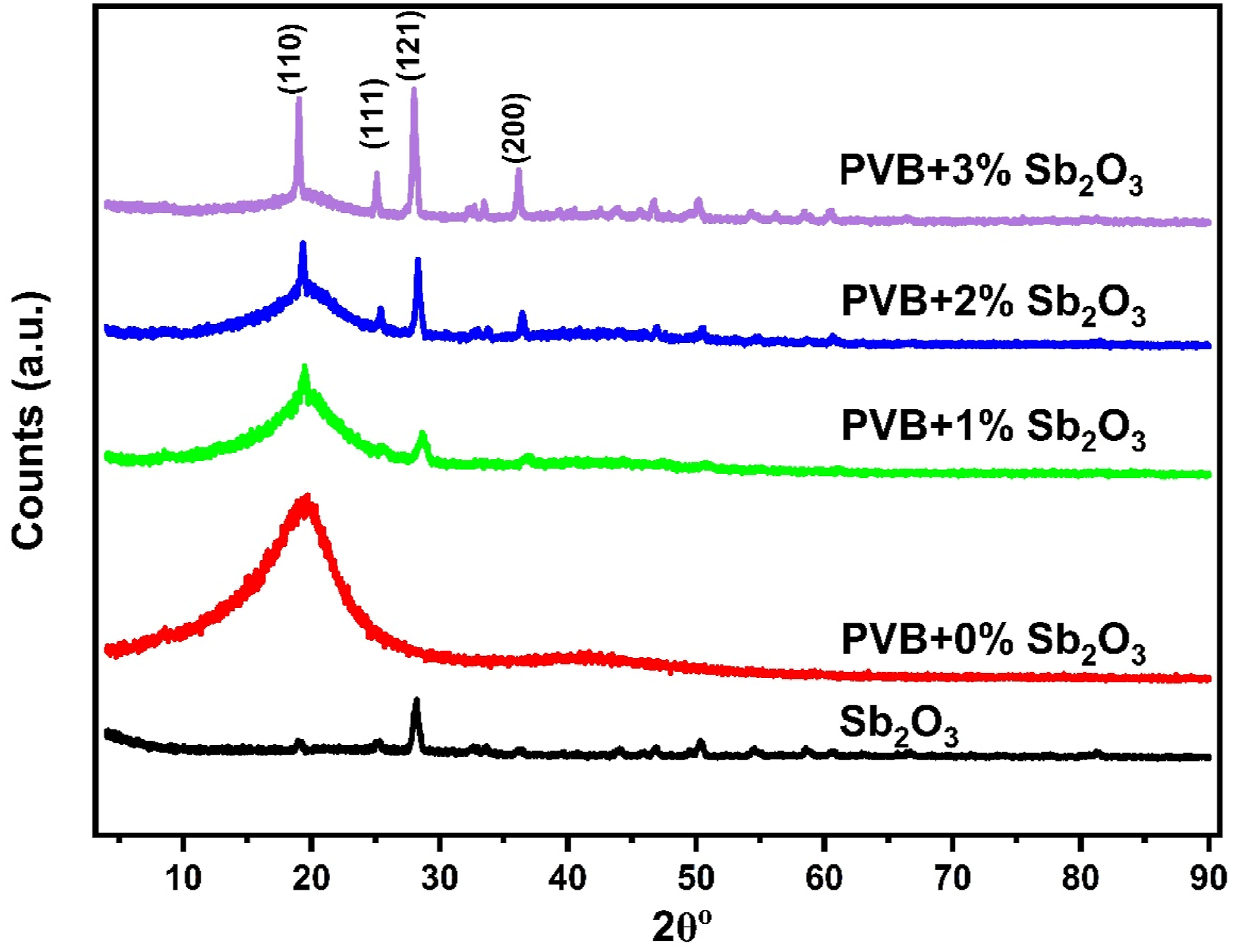
FTIR
FTIR of pure PVB and PVB/Sb2O3 nanocomposite is presented in Figure 3. The pure PVB film shows a small peak at 3399 cm−1 assigned to hydroxyl groups. Additionally, the 2885–2938 cm−1 are attributed to saturated CH, CH2, and CH3 stretching vibrations. The peaks at 1425 cm−1, 1373 cm−1 and 1345 cm−1 are assigned to C–H bending vibrations. The peak at 1133 cm−1 is assumed to be due to C–O–C–O–C stretching vibrations of cyclic acetal groups. The small peak at 1729 cm−1 pertains to C−O stretching vibration of the acetate group. The peaks at 984 cm−1 and 1242 cm−1 are attributed to C–O–C stretching vibration of acetate group.26,27 Upon incorporation of Sb2O3 into PVB, the intensity and broadening of hydroxyl group and CH and CH2 groups are observed. Furthermore, the increasing intensity of the transmission band in region 1000–15,000 was observed. This indicates there is a strong interaction between PVB and Sb2O3.
28
There is a possibility that hydrogen bonds were formed between the PVB chains and the oxygen atoms in the surface of the Sb2O3 nanoparticles.
29
FTIR of PVB and different PVB-Sb2O3 nanocomposites.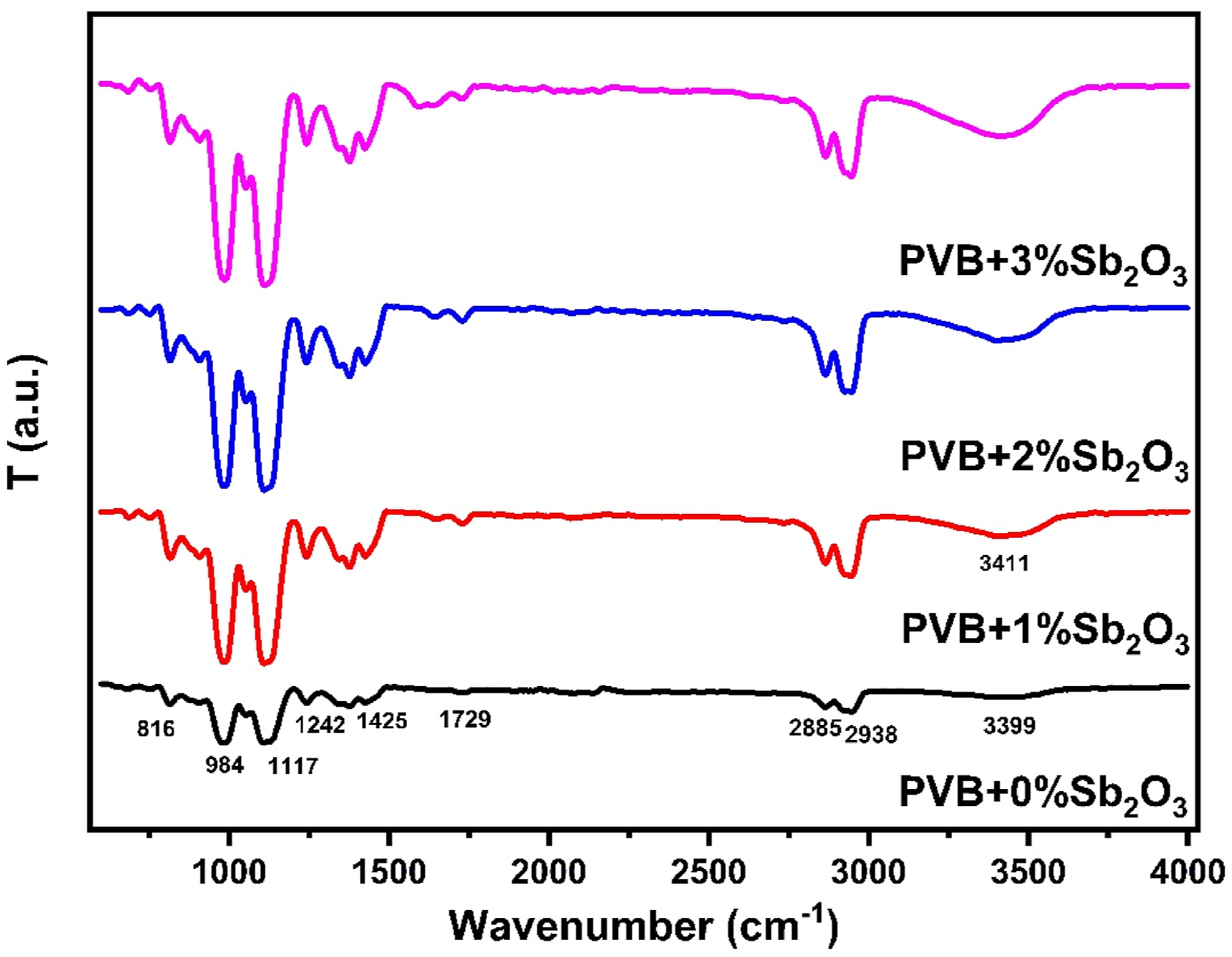
Optical properties
Figure 4(a) elucidates the variation of absorbance (A) versus wavelength (λ) for PVB/Sb2O3 nanocomposites. The incorporation of Sb2O3 NPs into polyvinyl butyral (PVB) films enhances their absorbance properties. The previous spectral line exhibits a redshift accompanied by an additional broadening effect. Furthermore, a distinct redshift in the cut-off edges is observed, ranging from 242 nm (for the PVB) to 255 nm (for the PVB/3 wt%Sb2O3) nanocomposite. Clearly, the transmittance (T) spectra of the PVB/Sb2O3 nanocomposites declined as the Sb2O3 NPs concentration increased. For instance, the T quantity decreased from 88% (PVB) to 21% (PVB/3 wt%Sb2O3 nanocomposite), as illustrated in Figure 4(b). The reduction in T and termination of edges redshift suggests the contraction of optical bandgap of the PVB/Sb2O3 nanocomposites as a consequence of a rise in the Sb2O3 NPs concentration. The evaluation of optical transmittance demonstrates the capacity to modulate the transmittance of the PVB/Sb2O3 nanocomposites through the incorporation of Sb2O3 NPs nanostructures. This novel result advocates for the utilization of PVB/Sb2O3 nanocomposites in applications involving UV-spectrum filters.
30
The reflectance (R) measurements (Figure 4(a)) suggest that the behavior of the PVB/Sb2O3 nanocomposites is depends upon the quantity of Sb2O3 NPs. The packaging density of the PVB/Sb2O3 nanocomposites can be altered by doping, which then affects their reflectance properties and, as a result, R values.
31
(a) The variation in A, (b) T, and (c) R spectra versus λ for PVB/Sb2O3 nanocomposites.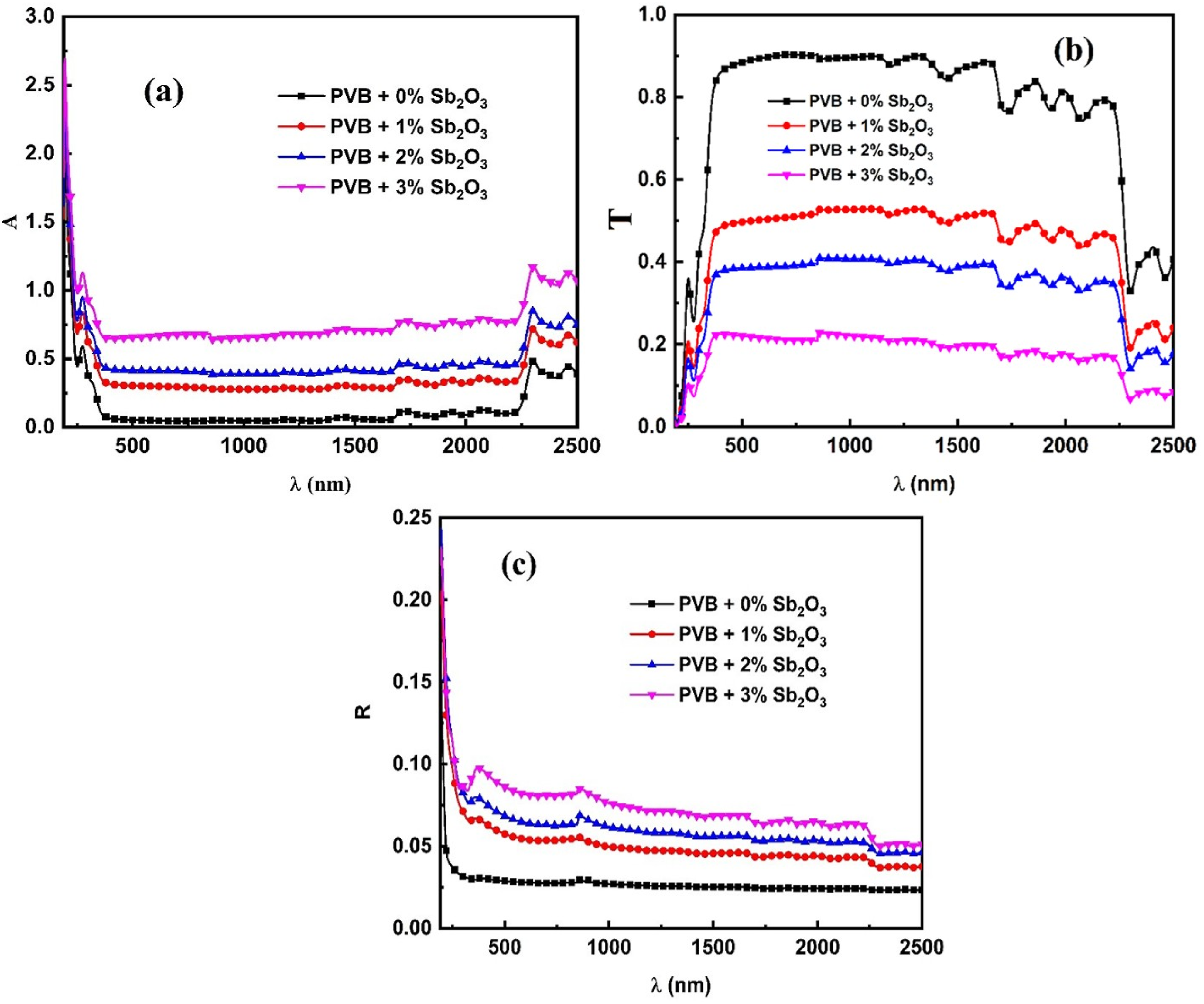
The optical absorption coefficient (α cm−1) versus the energy of incident photons (hv) for PVB/Sb2O3 nanocomposites was determined using next equation
32
and is depicted in Figure 5. Absorption coefficient (α cm−1) versus hv for PVB/Sb2O3 nanocomposites.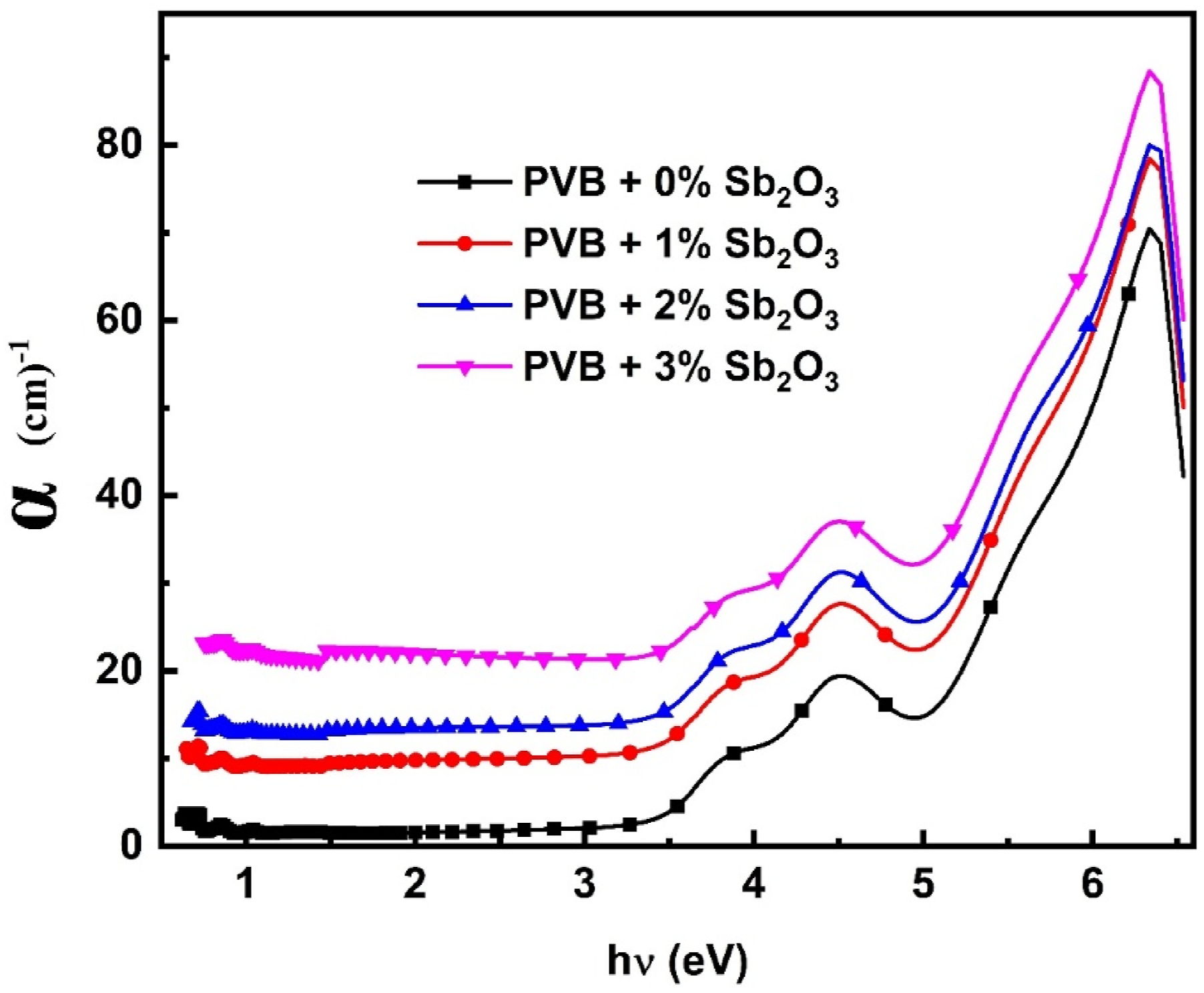
The thickness of the film is represented by the variable (d).
It is evident that α exhibited a progressive increase with the rise of hv within the visible to near-infrared (NIR) spectrum. Also it showed a significant increase in ultraviolet region. Additionally, it is noted that the absorption edges of the PVB/Sb2O3 nanocomposites shift towards lower hυ as the percentage of Sb2O3 NPs increases. Our findings demonstrate a decrease in optical bandgap values of the PVB/Sb2O3 nanocomposites resulting from doping with Sb2O3 nanoparticles.
The optical bandgap of the PVB/Sb2O3 nanocomposites can additionally be obtained by applying Tauc’s relation
33
:
The symbols for photon frequency (υ), Planck constant (h), proportionality constant (A), and electronic transition index (s) are used.
To ascertain the direct optical band gap (Eg) of the PVB/Sb2O3 nanocomposites, plots of (αhv)2 against hν were generated, as illustrated in Figure 6. The Eg values have been identified by extrapolating the linear sections of the plotted curves to the point where hν equals zero.34–36 Table 1 presents the estimated Eg values. It can be observed that the Eg1 and Eg2 values of the blank PVB are 5.10 eV and 5.28 eV, respectively. On the other hand, the Eg1 and Eg2 of the PVB/Sb2O3 nanocomposites diminish when the Sb2O3 nanostructures are doped to 4.72 eV and 4.88 eV, respectively, for the 3 wt% Sb2O3 doping. Sudarat Kumsaart et al.
37
have synthesized polyethylene glycol with various Nb2O5 and CaBr2⋅xH2O concentrations. They reported that the band gap values of the polymer layers go down from 3.0 eV to 2.06 eV. Plots of (αhv)2 against hν of the PVB/Sb2O3 nanocomposites. The values of Eg and Eu, for PVB/Sb2O3 nanocomposites.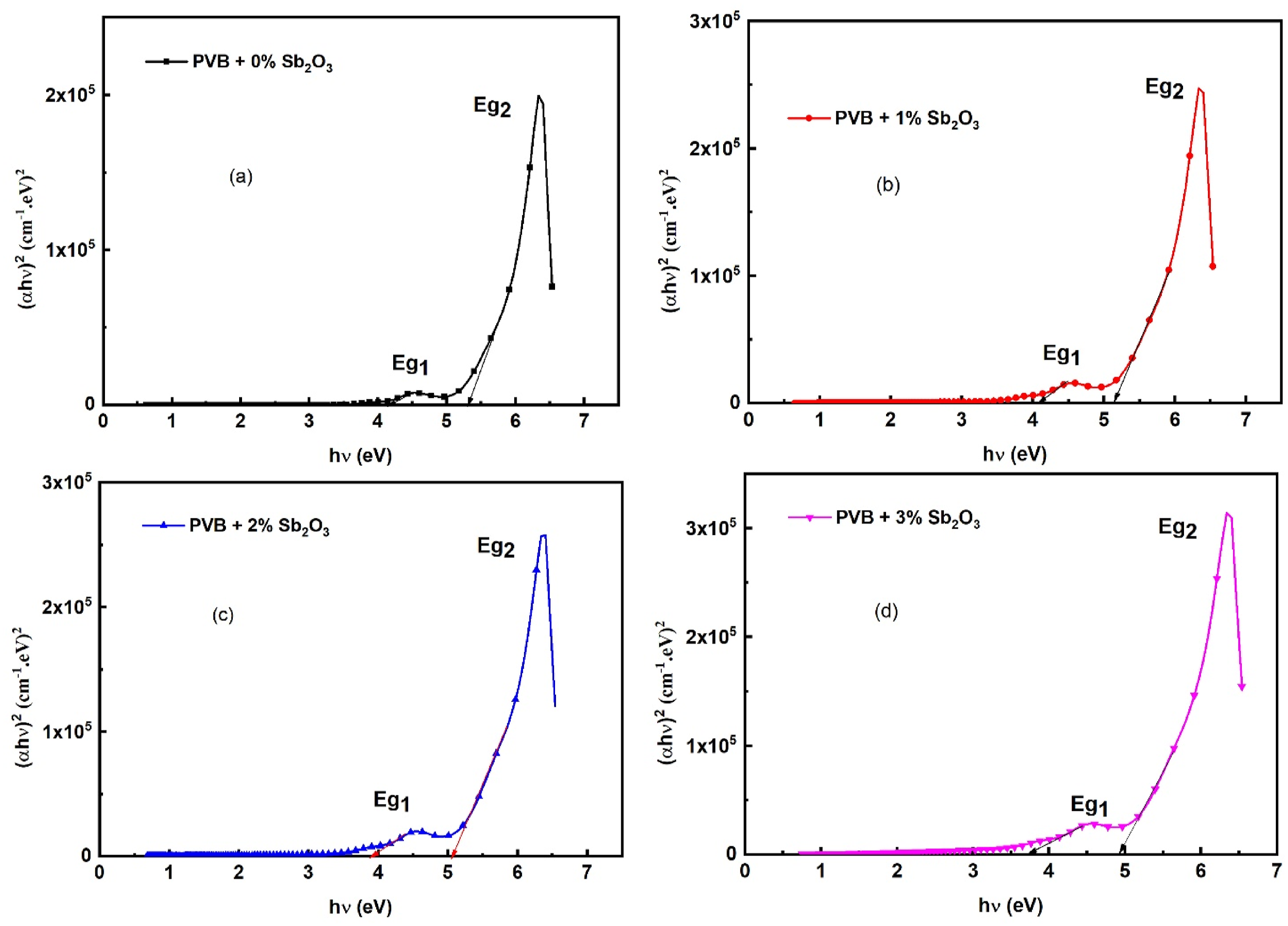
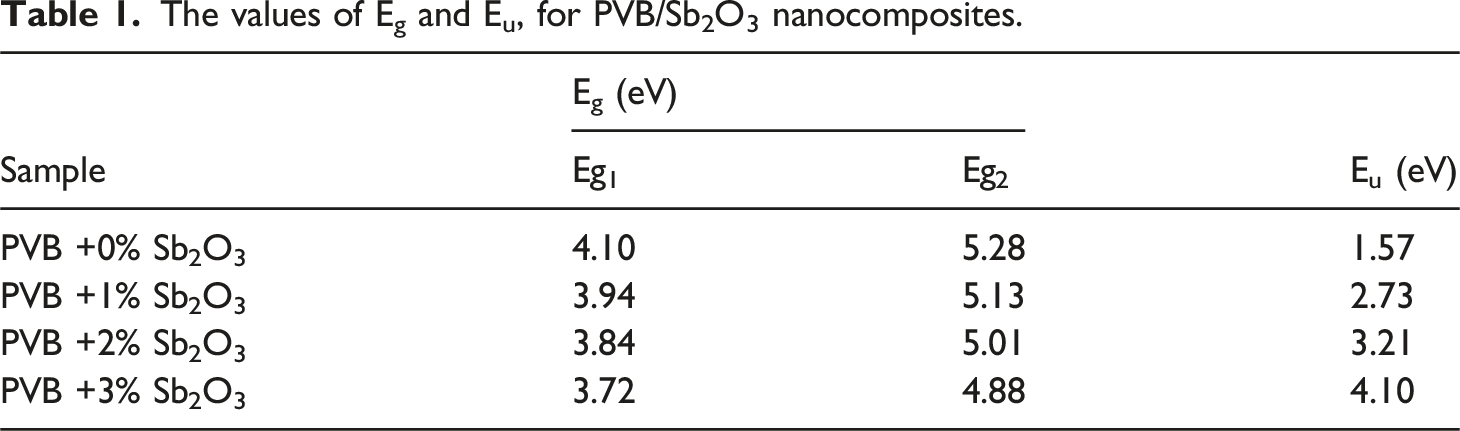
The decrease in Eg of the PVB/Sb2O3 nanocomposites is mostly due to the Sb2O3 NPs, which notably alter the structure of PVB. The adjustments are achieved by modifying the amounts of defects and localized energy states within the bandgap of the PVB. The inclusion of Sb2O3 NPs in the PVB induces significant structural disorder, resulting in a notable alteration of its overall physical characteristics.30,38 The disorder level and defect ratios in PVB and PVB/Sb2O3 nanocomposites may be examined by analyzing the Urbach energy (EU). The EU of the blank PVB and PVB/Sb2O3 nanocomposites is ascertained based on the empirical methodology delineated in formula
39
:
The EU corresponds to the reciprocals of the slopes of the ln α versus hv plots, as seen in Figure 7. The identified EU values are shown in Table 1. Significantly, the EU value of the blank PVB is 1.57 eV. The EU value of the blank PVB increases to 4.1 eV with the addition of Sb2O3 NPs. This data indicates the rise in defect levels and localized states inside the bandgap of the host PVB. Urbach energy assessment of the PVB/Sb2O3 nanocomposites.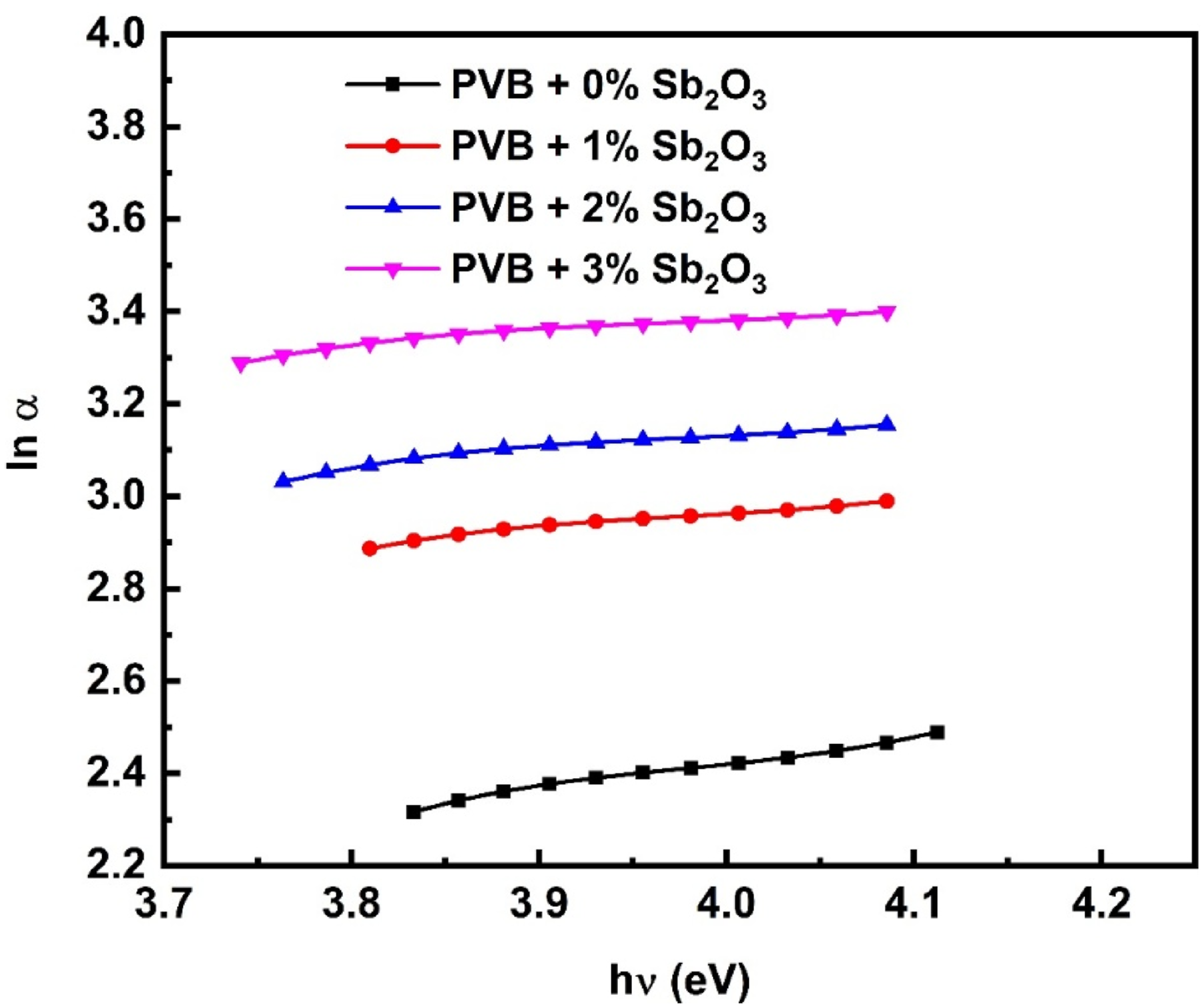
Furthermore, the coefficient of extension (k) ( The correlation between k and hυ for the PVB/Sb2O3 nanocomposites.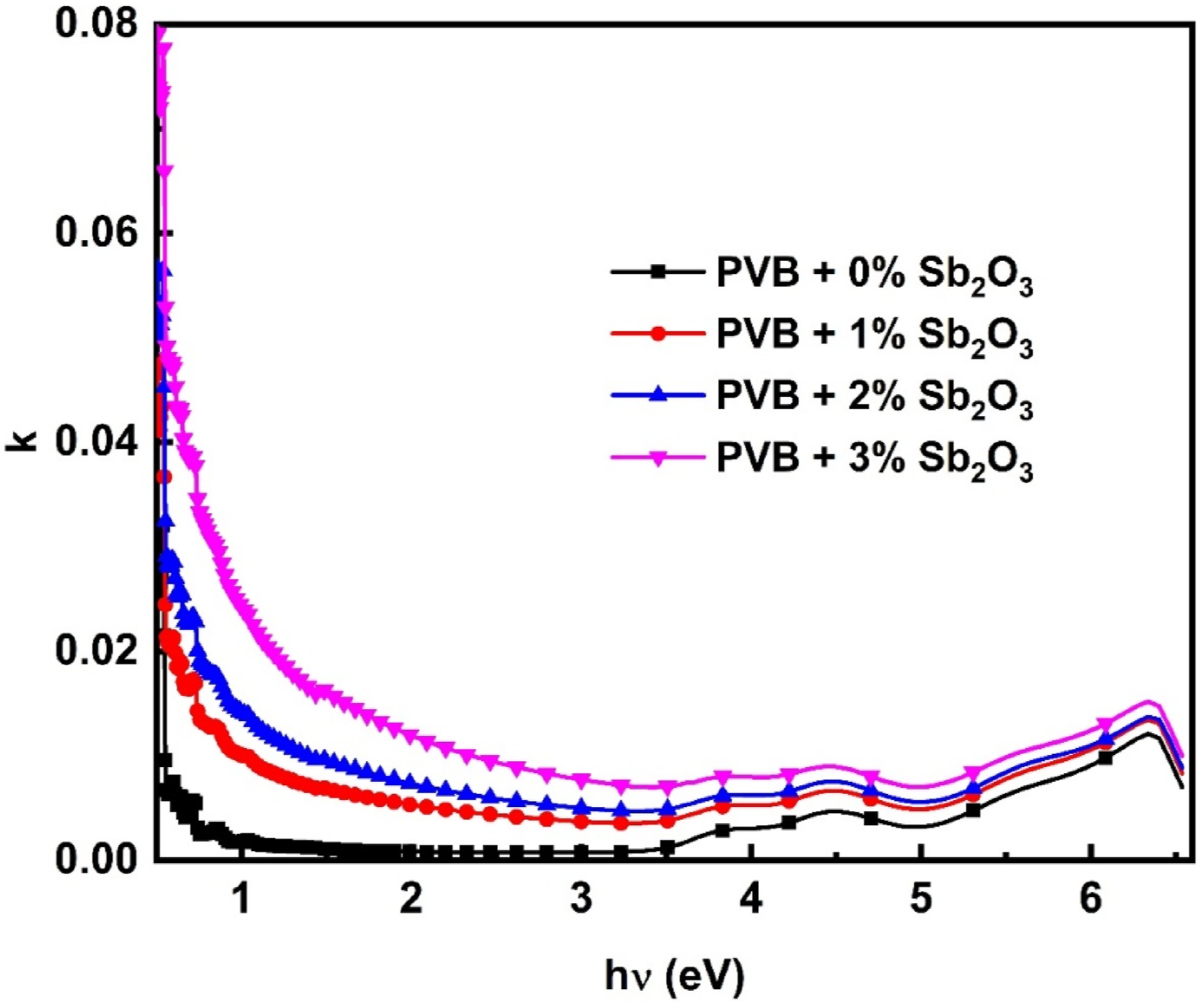
Additionally, the refractive index (n) is a critical element of various optical parameters that must be identified to recommend its suitability for use in optical device technologies. The refractive index investigation provides insight into the behavior of electromagnetic waves as they traverse the optical material.
30
The n values of the PVB/Sb2O3 nanocomposites were determined by utilizing the following expressions41–43: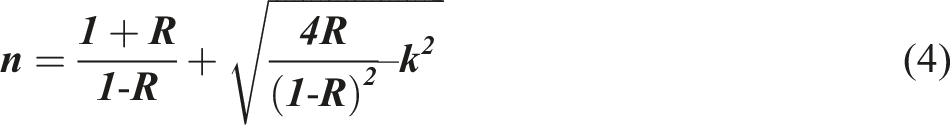
The n curves of pure PVB and PVB/Sb2O3 films are illustrated in Figure 9. The incorporation of Sb2O3 NPs results in an upsurge in the values of the n. As the concentration of Sb2O3 NPs rises the density of packaging improves which can be attributed to modifications to the PVB matrix. Additionally, the quantity of unrestricted carriers and the degree of reflection in the PVB/Sb2O3 nanocomposite films grow as the concentration of Sb2O3 in the PVB/Sb2O3 nanocomposite films increases. Consequently, the refractive index increases as well. The comparable features have been observed in PEO/PVA/MWCNTs/ZnO nanocomposites.
40
The refractive index (n) spectra of the PVB/Sb2O3 nanocomposites.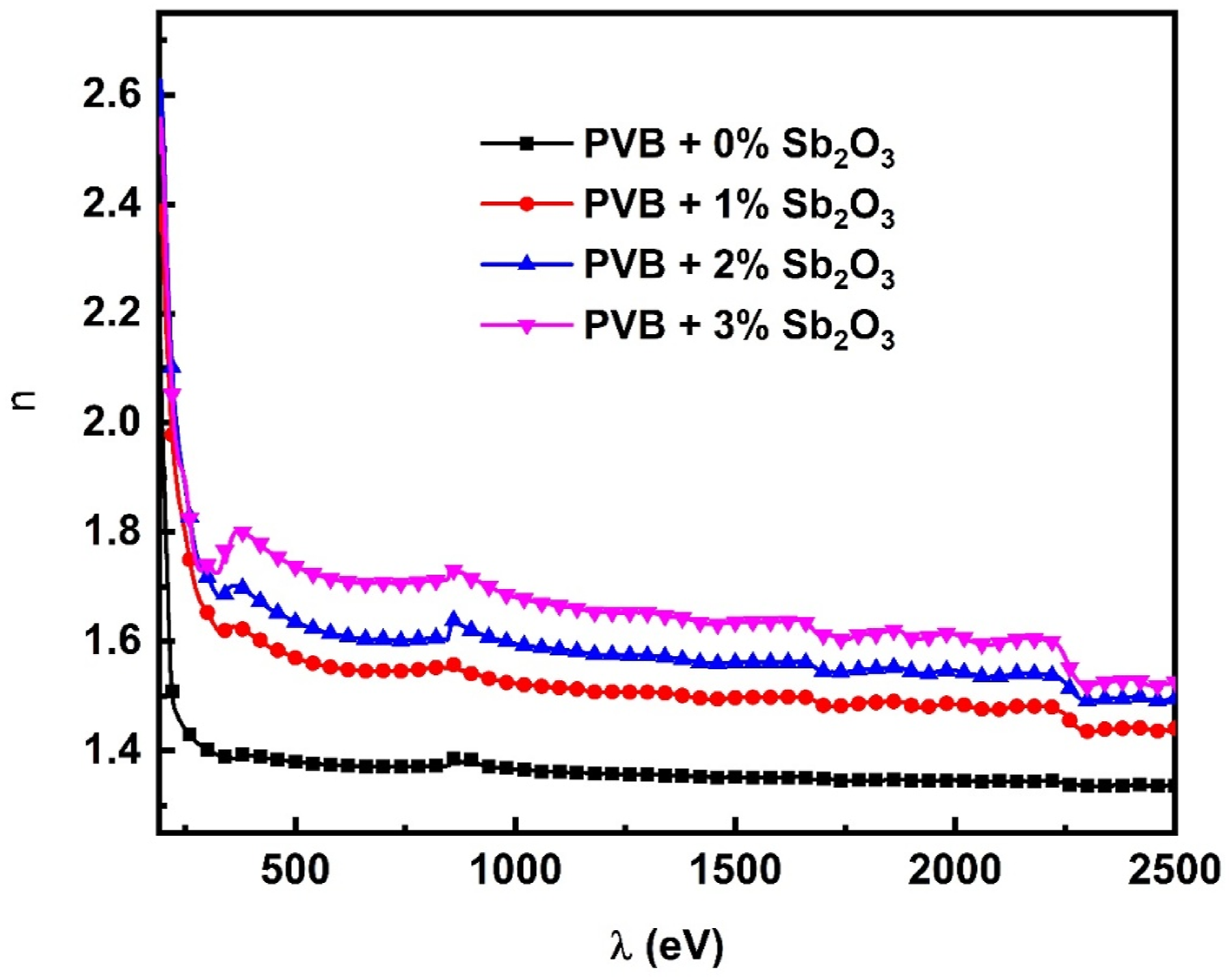
The increased refractive index of PVB/Sb2O3 makes it suitable for enhancing the functionality of applications in optical waveguides, optical fibers, anti-reflective coatings, and optoelectronics.
Additionally, the optical dispersion parameters are among the most critical parameters in the field of applications involving optical electronics. The Wemple–DiDomenico (WDD) single-oscillator model is employed for computing these parameters, which is denoted as
44
:
The static refractive index values (no) of PVB/Sb2O3 nanocomposites at frequency is zero (hv→0)
45
:
The static dielectric constant (εs) may be obtained by using the static refractive index εs= no2.
46
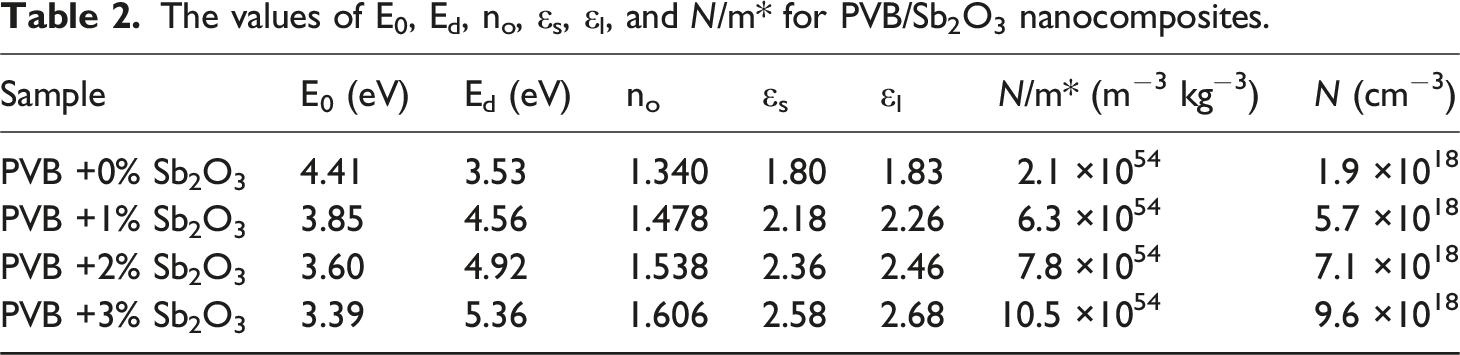
Where Ed represents the dispersion energy and E0 represents the average excitation energy of electronic transitions (oscillator energy). The relationship between (n2 – 1)−1 and (hυ)2 has been shown in Figure 10. This relationship is represented by a linear line. Table 2 displays the calculated values. It is evident from Table 2 that the concentration of Sb2O3 in the PVB matrix leads to an increase in the values of Ed, n0, and εs. The values of Ed increased significantly from 3.53 to 5.36 eV, and no enhanced from 1.340 to 1.606, while εs, rose from 1.80 to 2.58, while the E0 values declined from 4.41 to 3.39 eV for PVB/3 wt%Sb2O3 film. This suggests that the charge transfer between the Sb2O3 and the PVB macromolecules has been rising, alongside the degree of disorder in the PVB structure.
47
Variation of (n2−1)−1 versus (hυ)2 for the PVB/Sb2O3 nanocomposites. The values of E0, Ed, no, εs, εl, and N/m* for PVB/Sb2O3 nanocomposites.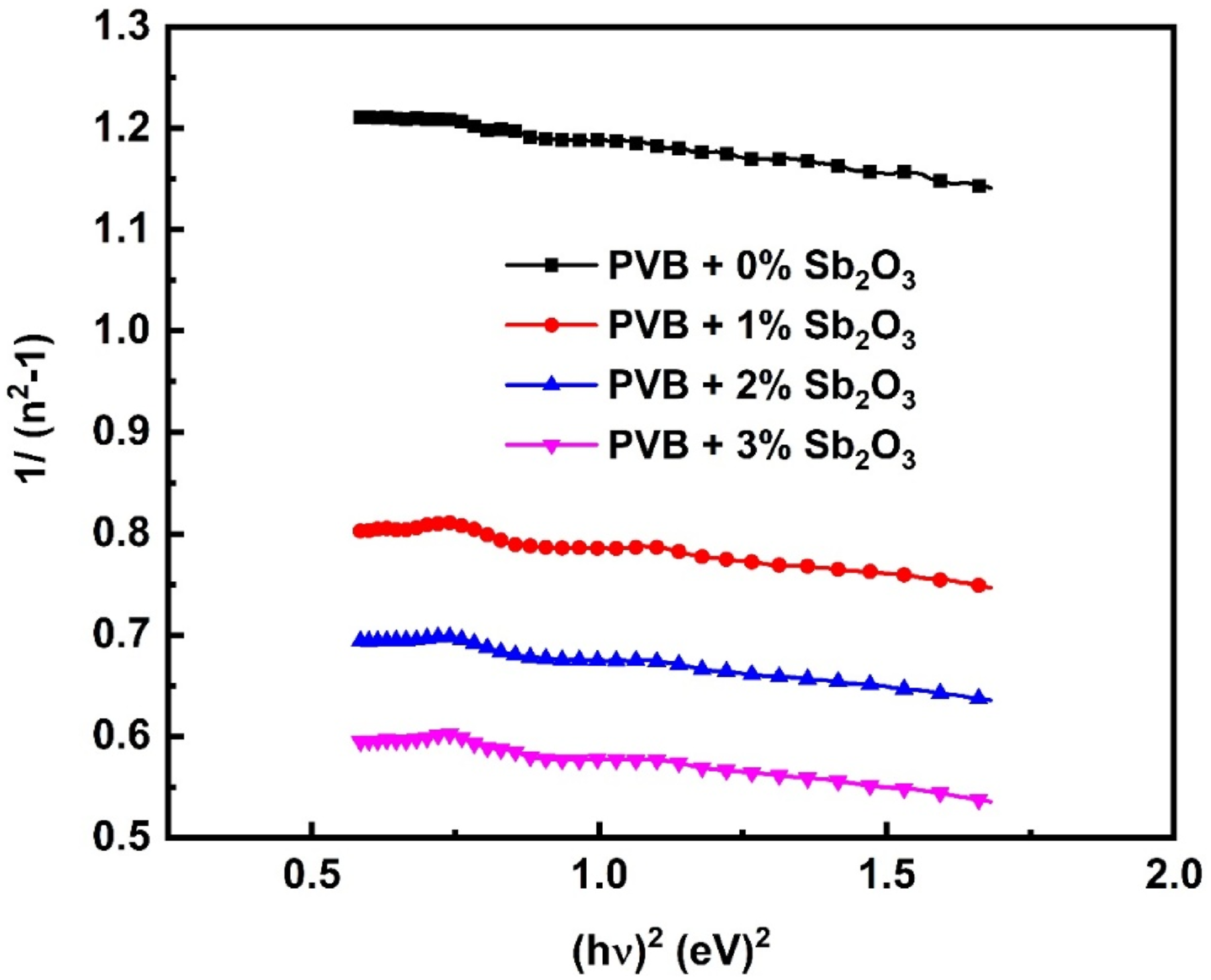
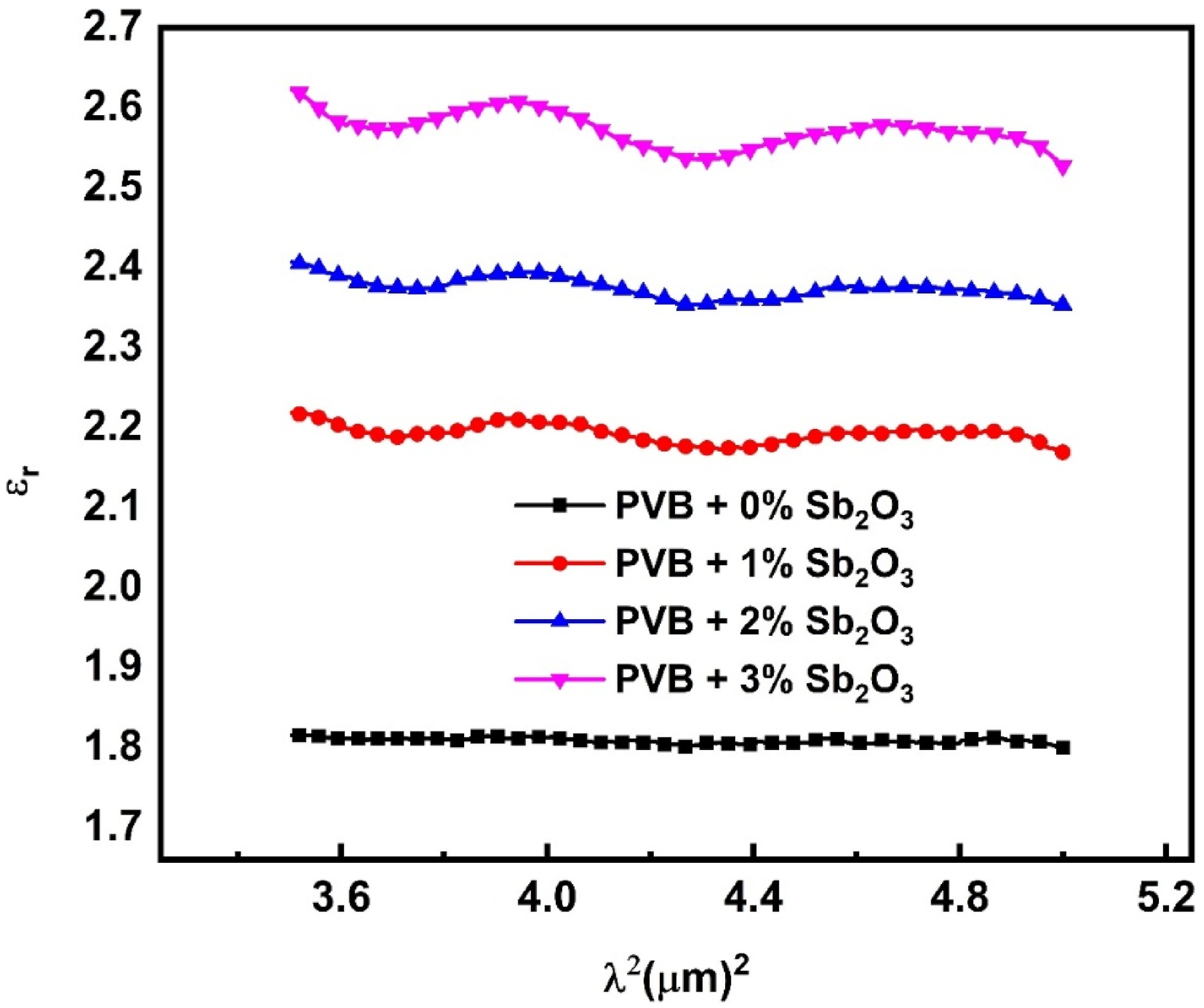
The formula shown below may be used to express the real component of the dielectric constant, denoted by εr
48
: εr versus λ2 for the PVB/Sb2O3 nanocomposites.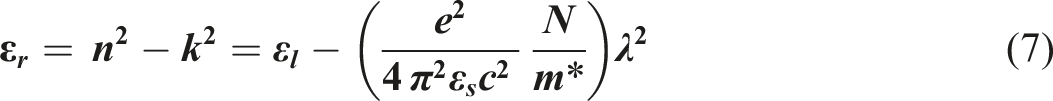
The formula may be used to determine the connection between the imaginary dielectric constant ( The correlation between 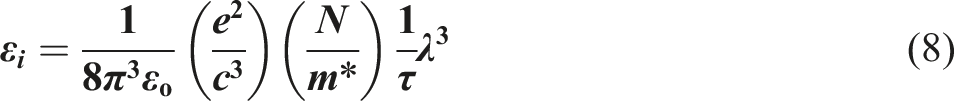
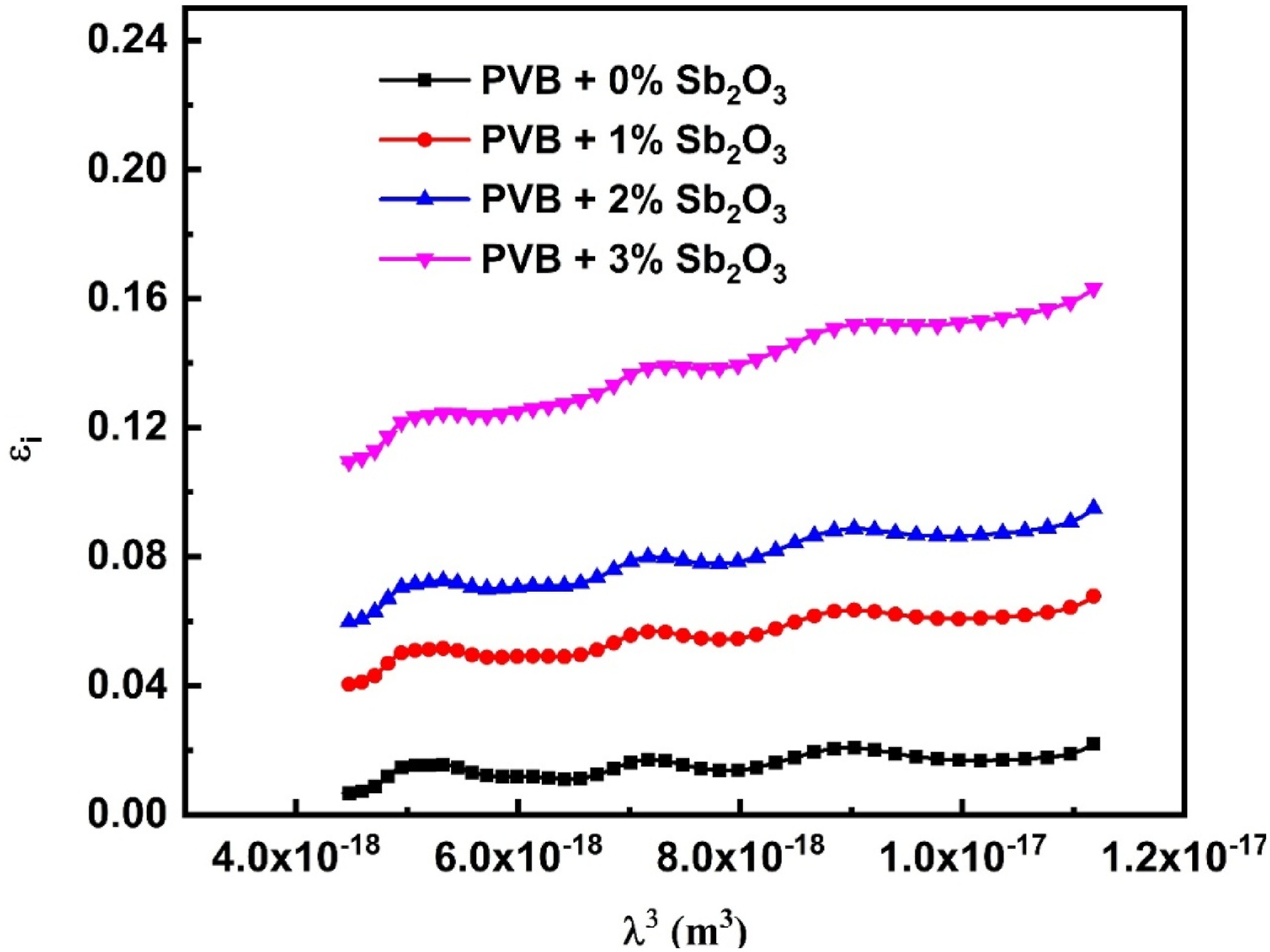
Optoelectrical parameters for PVB/ Sb2O3 nanocomposites.
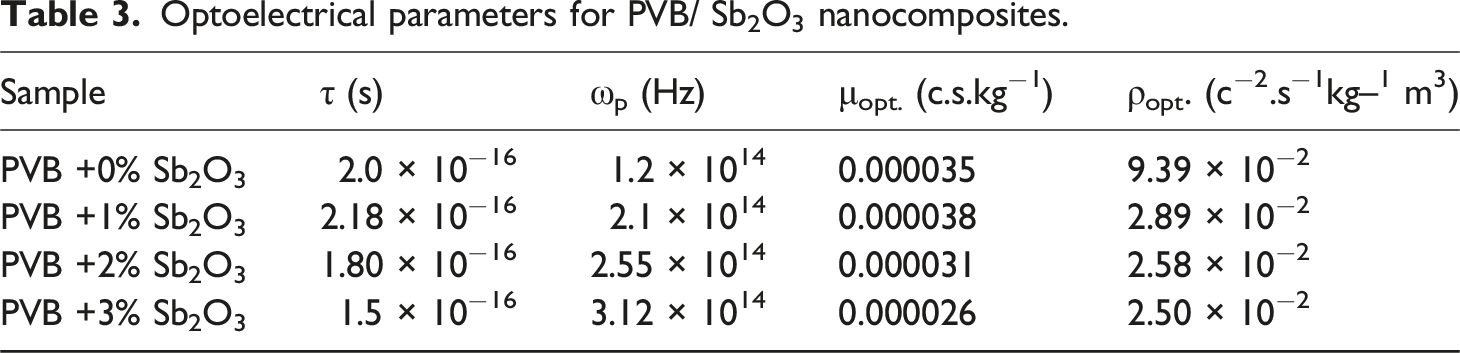
The real component of the dielectric constant (εr) can be used to estimate the oscillation frequency (ωP) for PVB/Sb2O3 nanocomposites with varying Sb2O3 percentages (0, 1, 2, and 3 wt%) employing the formula listed below
50
:
The relationship between εr and ω−2 has been demonstrated in Figure 13, which illustrates the interaction through PVB/Sb2O3 nanocomposites with varying Sb2O3 percentages (0, 1, 2, and 3 wt%). The values of ωp for nanocomposites were obtained by calculating the slope of those straight lines and are shown in Table 3. Increasing the amount of Sb2O3 results in a rise in the refractive index and polarisation, which in turn leads to significant fluctuations in the values of εr. Through the addition of Sb2O3, the change in ωp for PVB/Sb2O3 nanocomposites is influenced by the increase in the concentration of free carriers. As the percentage of Sb2O3 rises, the oscillation frequency value falls within the range of 1.2 × 1014 to 3.12 × 1014 Hz. The correlation amongst εr and ω−2 for PVB/Sb2O3 nanocomposites.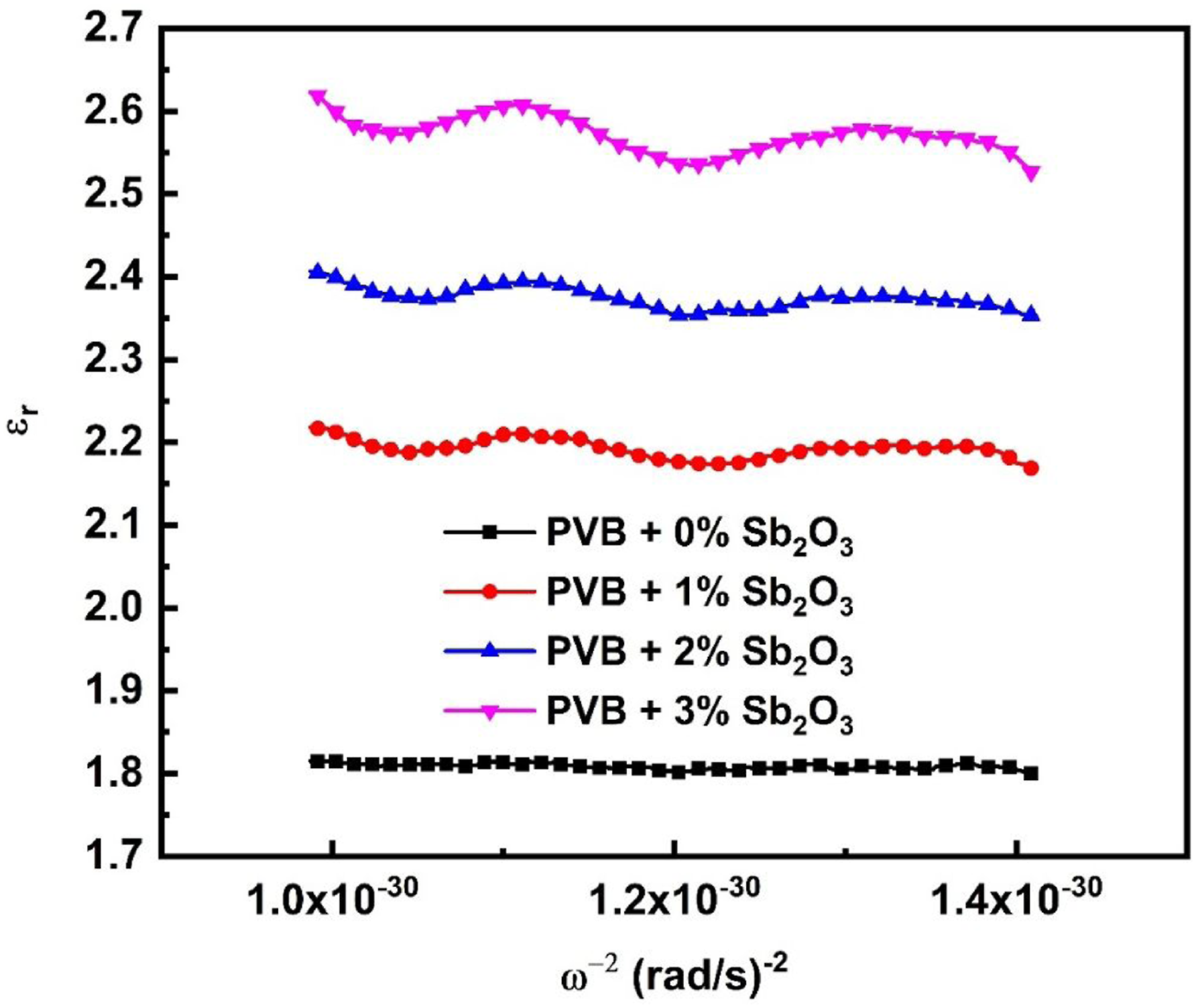
The optical mobility (µopt.), and the optical resistivity (

The optical mobility for PVB/Sb2O3 nanocomposites ranged between 26 × 10−6 c.s.kg−1 and 38 × 10−6 c.s.kg−1. Conversely, the optical resistivity decreased from 9.39 × 10−2 to 2.50 × 10−2 c−2.s−1.kg−1.m3, as seen in Table 3. The phenomenon arises when the bandgap diminishes, hence promoting increased conductivity as electrons may rapidly transition from the valence band to the conduction band. 53 The optoelectrical parameters have been studied in many previous literatures.54,55 Suwannakham et al., 54 reported that the oscillation frequency value falls within the range of 6.67 × 1014 to 8.22 × 1014 Hz, and the optical resistivity ranged between 7.2 × 10−2 to 2.152 c−2.s−1.kg−1.m3 for antimony-doped tin oxide thin films.
Conclusion
XRD revealed that the interaction of Sb2O3 with PVB results in an increase in the crystallinity of the nanocomposites. Furthermore, the FTIR confirmed the interaction between PVB and nanofiller. The transmission spectra of the PVB/Sb2O3 nanocomposites declined upon increasing the Sb2O3 NPs ratios inside the polymeric matrix. The absorption coefficient increased with the rise of photon energy within the visible to near-infrared (NIR) spectrum and exhibited a significant increase in the ultraviolet region. The structural disorder caused by nanofiller decreases the optical band gap and increases Urbach energy inside nanocomposites. The incorporation of Sb2O3 NPs results in an increase in the values of the refractive index and extinction coefficient, which can be attributed to modifications to the PVB matrix. Additionally, the values of Ed., n0, and εs increased with the addition of nanofiller. In contrast, Eo declined, indicating a rise in charge transfer between Sb2O3 and PVB, accompanied by an increase in disorder in the PVB. Lattice dielectric constant εl improved from 1.83 to 2.68 as the concentration of Sb2O3 increased. The N/m* value is increased from 2.1 × 10 54 kg−1 m to 10.5 × 10 54 kg−1 m−1 with increasing Sb2O3 up to 3 wt%. The relaxation time and oscillation frequency values decrease once Sb2O3 is embedded. The outcome confirms that Sb2O3 can modulate the optical features of PVB, making it suitable for various applications, including UV shielding, optical fibers, anti-reflective coatings, and optoelectronic devices.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Ongoing Research Funding program, (ORF-2025-845), King Saud University, Riyadh, Saudi Arabia.
Data Availability Statements
The Authors confirms that data will be available on reasonable request.


