Abstract
This comprehensive review explores polyvinyl alcohol (PVA) and its nanocomposite films with a focus on their optical and electronic properties for optoelectronic applications. The study begins by examining the fundamental characteristics and applicability of PVA as a versatile polymer matrix, followed by an in-depth analysis of PVA films and their tunable features. The synthesis methodologies for PVA/nanocomposite films are critically evaluated, highlighting recent advancements in fabrication techniques. A significant portion of the review is dedicated to investigating the optical properties of these materials through various theoretical frameworks, including dielectric function analysis, microscopic and classical models, harmonic oscillator models, and nonlinear optical behaviors. The review thoroughly examines interband transitions and employs analytical approaches such as Tauc’s method and the Wemple-DiDomenico technique to elucidate band gap characteristics. The optical and electronic properties of various PVA/nanocomposite systems are systematically compared, establishing structure-property relationships crucial for device engineering. Finally, the review concludes by exploring prospects of PVA-based advanced materials in emerging optoelectronic applications, identifying research gaps and promising directions for further investigation. This work provides valuable insights for researchers and industry professionals seeking to develop next-generation optoelectronic devices utilizing PVA-based nanocomposite materials.
Introduction
Polymeric substances show remarkable promise in the field of sustainable energy collection. Unlike conventional energy supplies for instance coal and oil-derived fuels, solar power offers the constant, renewable, as long as long-lasting supply that could meet the growing global energy needs. Devices that harness sunlight utilize the photoelectric phenomenon to efficiently convert solar radiation into usable electric power. 1 In the realm of optoelectronics, polymer-based mixtures serve as both passive and active optical components. Their unique light-related properties make them suitable for various uses, including high-refractive-index films, field-effect transistors, solar cells, light-emitting diodes, optical waveguide materials, and light-sensitive compounds.2,3 These mixtures are heterogeneous materials consisting of two distinct substances that retain their individual identities while interacting through a specific boundary known as the composite interface. The optical and electrical traits of polymeric materials could be adjusted by introducing additional substances. 4 Choosing the right additives is crucial, as different substances uniquely affect the host material. 5 Ongoing research is essential to enhance our understanding and develop future technologies for polymer mixtures. By adjusting the makeup and properties of additives at the interface boundary, a broad span of opportunities for novel and multifunctional materials becomes available.
The current worldwide energy usages are approximately sixteen terawatts which are assumed to increase fourfold through the 50th period of the century. The urgent need to address global climate change and carbon emissions has driven the search for sustainable alternatives to fossil fuels through solar energy utilization. Consequently, the recent two decades have seen increased momentum in developing photovoltaic technologies. In this area, hybrid solar panels that incorporate both inorganic and organic elements have been suggested as a potential replacement for traditional organic solar plates. This innovative method combines the cost-effective production of organic photovoltaic (OPV) cells with the inherent advantages of natural energy harvesting. These solar energy systems typically employ a combination of various inorganic plus organic semiconductors for the front active layer of the devices. 6
Semiconductor compounds with a direct optical band gap (OBG) are often utilized in solar panels as well as optoelectronic devices. This structure is preferred because electrons could directly move to the conduction band upon receiving photon energy. In contrast, indirect OBG semiconductors involve phonons, which result in heat dispersion inside the matter and lower device reliability. Transitions between bands are more likely for wide optical band gap semiconductors than transitions within bands. Throughout the transition between the bands known as the inter-band transitions that occurs between the deep and shallow-levels transition from the valence band to the conduction band can occur. 7 The movement of charge in molecular approaches exhibits a subject of great interest in materials science due to its close relationship with magnetic transformations, transportation through photonics, dielectrics, and structural characteristics. The transport of the charge complexes demonstrates remarkable optical, electrical, and photoelectric properties, which are critical for various electro-physical as well as the optical trends. 15 Additional information related to the mechanism of the absorptivity of light is essential headed for gain a more profound insight into the optical energy band structure in nanocomposites of polymers. 8 This information is crucial for evaluating the spectral properties for polymeric nanocomposites designed for definite purposes. 9
Shows the physical properties of PVA.

Various PVA based composites were prepared and achieved an excellent optical property. Ahmed Hashim et al prepared PVA/SiO2/BaTiO3 Nanostructures Films. When the SiO2/BaTiO3 NPs content reached 3.9 weight percent for the forbidden transition, the indirect energy gap of PVA decreased from 4.8 eV to 3.8 eV for the allowed transition and from 4.4 eV to 3.2 eV. 15 Also, PVA/Fe2O3/SiC Hybrid Nanosystem was prepared for Optics and Nanoelectronics applications. It’s found that when the amount of Fe2O3/SiC nanostructures reached 6 weight percent, the energy band gap (Eg) for PVA decreased from 5 eV to 3 eV. 16 PVA-Li-Si3N4 were found to have an effective composite in optoelectronics field. 17 In other work, SiC and CuO are added to PVA, the energy gap decreases from 7.184 to 2.604 eV and from 7.184 to 4.229 eV, respectively. These worldwide findings have significant electronic applications. 18 And also, when 6 weight percent TiC/SiC NPs were added, the absorbance of pure PVA increased by 98%, and the optical conductivity increased by 99%. 19 In testing the effect of SiC/CoFe2O4 NPs addition to PVA, it is observed that, at 6.3 weight percent of SiC/CoFe2O4 NPs, the PVA energy gap dropped from 5.1 to 2.3 eV for the permitted transition and from 4.7 to 0.6 eV for the banned transition. 20 PVA-CoO-SiC, 21 PVA/PEO/CuO, 22 PVA/PEG/Si, 23 and many other composites have prepared and investigated a great effect in optoelectronics applications.
The applicability usage of PVA-doped films in cutting-edge photonics are as diverse as they are impressive. From highly efficient holographic plates that push the boundaries of real-time holography 24 to biocompatible micro-lasers that hint at a future of targeted medical interventions, 25 PVA continues to surprise and delight. It plays a crucial role in white laser emission systems, 26 breathes life into LEDs and electroluminescent devices, and forms the backbone of linear film polarizers that have become ubiquitous in modern displays. 27 Even in the realm of UV-VIS laser cut-off filters, PVA demonstrates its remarkable versatility. 28
In this review, the characteristics and the compatibility of using PVA were investigated. The typical preparation techniques were also covered. The optical parameters that play a vital role in investigating and calculating the different optical characteristics were reported to make this review a reference for the researchers who want to use the full equations and relations for PVA polymeric materials and other optical materials to shed light on their optoelectronic applications. Also, the role of dopants from various inorganic metals, nanoparticles, and ions implementations, and the other polymers’ effects on the PVA host matrix’s structural, spectroscopic, and optical properties are reported gently in this review. Also, the ability to use these different composite films as optoelectronic and other optical devices is covered in many ways in the review.
Fundamental characteristics of PVA
The genesis of PVA is a testament to chemical ingenuity. Synthesized through the saponification of poly (vinyl ester) utilizing NaOH (Figure 1)
13
or the hydroxylation of polyvinyl acetate, this process meticulously strips away acetate groups from PVAc molecules. The result is a water-soluble marvel that stubbornly resists dissolution in most organic solvents.
13
In its commercial form, PVA typically boasts a degree of polymerization ranging from 500 to 5000, corresponding to molecular weights that span from 20,000 to an impressive 200,000.
29
Synthesis of PVA. Reproduced with permission.
30
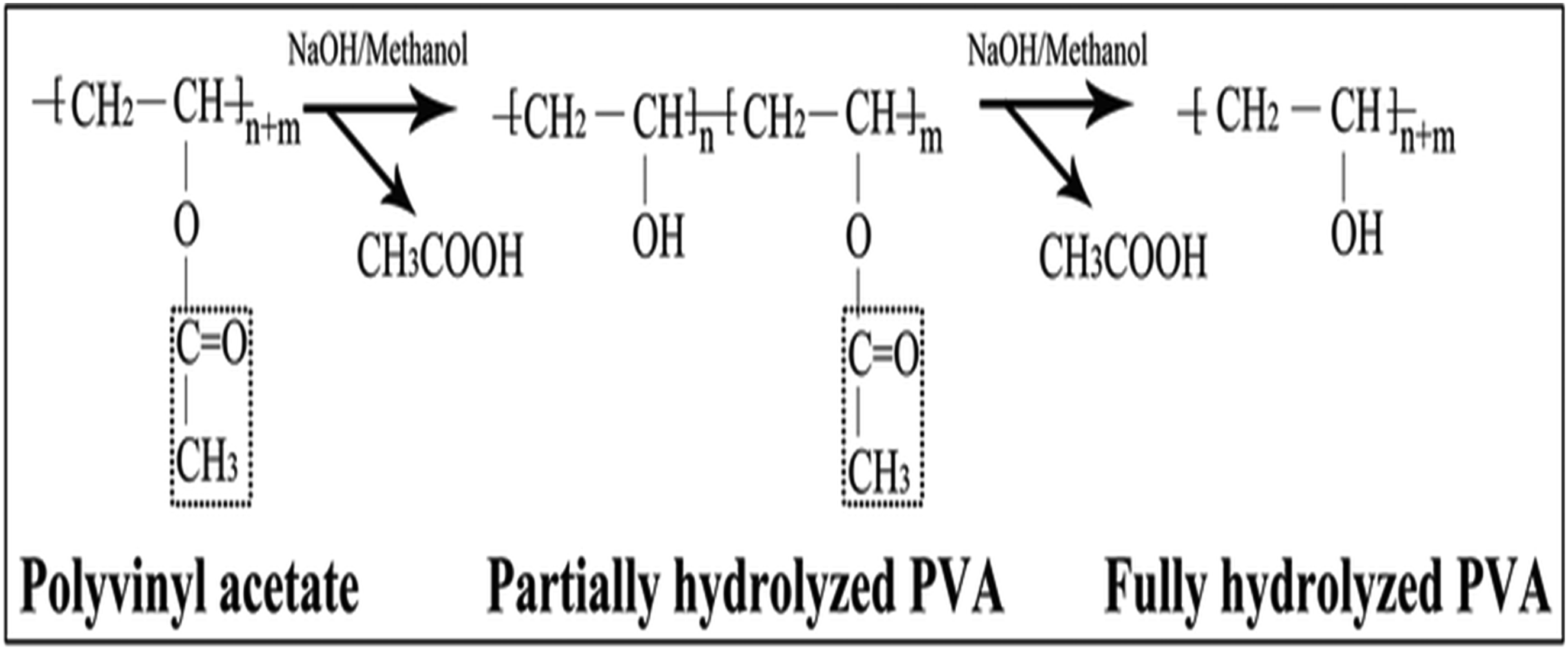
Delving deeper into its physical properties, PVA exhibits a density range of 1.19 to 1.31 gm/cm3, with a melting point that reaches a scorching 230°C. However, it’s not without its vulnerabilities – PVA rapidly succumbs to decomposition when temperatures soar above 200°C, falling prey to pyrolysis in these extreme conditions. 31 The fundamental characteristics of PVA are a complex interplay of factors, including its degree of polymerization, the intricate spreading of hydroxyl groups along its backbone, its stereoregularity, and the degree of crystallinity it achieves. 32 This intricate balance of properties and synthesis techniques has positioned PVA at the forefront of polymer science, continuing to inspire new applications and push the boundaries of what’s possible in material engineering.
Polyvinyl alcohol (PVA) exhibits a unique crystallization ability despite its atactic nature, primarily due to the compact size of its OH-groups. This characteristic, coupled with its excellent optical transparency, 33 makes PVA an exceptional material for various applications. In the visible spectrum, PVA boasts a refractive index ranging from 1.48 to 1.50, 34 with transparency levels soaring from approximately 40% at 200 nm to nearly 100% beyond 500 nm. 35 This optical profile renders PVA invaluable in optics and photonics, enhancing light output while minimizing reflection losses.
The applicability of PVA
The versatility of PVA extends across numerous industries. It serves as a crucial component in adhesives, emulsifiers, and protective films, while also acting as a precursor for other resins and polymeric nanoparticles. Its role in stabilizing Water-in-Oil-in-Water emulsions is particularly noteworthy. 36 PVA’s biocompatibility, biodegradability, and non-toxicity, combined with its remarkable water retention capabilities, position it as a leading synthetic polymer hydrogel. Its applications span wound healing, drug delivery systems, synthetic organs, as well as contact lenses. 37
In the medical field, PVA’s tensile strength, in comparison with the human articular cartilage, along with its hydrophilicity and chemical stability, make it an ideal candidate for various biomedical applications. 38 Its ability to absorb toxic products and facilitate the breakdown of necrotic tissue has led to its use in medical fibers and films. 39 Furthermore, PVA has shown promise in gene therapy applications, particularly in coating superparamagnetic iron oxide nanoparticles (SPIONs). PVA’s environmental impact is relatively benign, with solutions restraining up to 5% PVA showing no toxicity to aquatic life. Its carbon-carbon backbone resists hydrolysis, 40 necessitating oxidation for biodegradation. This process can occur under both aerobic and anaerobic conditions, involving microorganisms and enzymes. 41
The polymer’s unique structure, featuring numerous functional groups, enables it to form hydrogen bonds readily, facilitating polymer blending. 42 Its hydrophilic nature as well as profusion of reactive functional groups make it amenable to cross-linking with various dopants. 43 Beyond its traditional uses in nutrition, medical applications, as well as cosmetics, PVA has found applications in excellent thin film depositions, optical devices, as well as photographic film production. 44 PVA is renowned due to it has high dielectric strength, 45 thermos-chemical stabilities, as well as its ability to exhibit dopant-dependent physical properties. 46 These characteristics make it a versatile substance representing a varied scale of technological purposes.
PVA films: properties and synthesis
PVA films and their tunable features
The synthesis and tailoring of PVA films present both opportunities and challenges. While PVA excels in film formation, processability, and mechanical strength, its high-water absorption can be problematic for certain applications. Strategies to mitigate this include protective coatings, such as triacetyl cellulose (TAC) films used in LCD polarization layers, and modifications to reduce intrinsic hydrophilicity through thermal crosslinking and additives. 47
The polymer’s thermal degradation begins just above 200°C, coinciding with its melting point. Interestingly, PVA crystallizes so rapidly from the melt that achieving 100% amorphous materials is virtually impossible. Studies have shown that PVA consists primarily of crystalline and mobile amorphous phases, with no confirmation of a rigid amorphous phase. 48 The semicrystalline nature of PVA allows for a gradual transition between crystalline and amorphous regions, with individual macromolecules potentially spanning both. 49 Untreated PVA films typically exhibit a crystallization degree of 30–40%, which can be increased to about 60% through annealing (up to 150°C). However, dopants like lithium chloride or chitosan can significantly reduce crystallinity. 50
Controlling the crystallinity of PVA is crucial for tailoring its properties. Methods include the film/gel technique, polymer blending with plasticizers, 50 and dopant insertion. For instance, adding conductive ions like Ni2+51 or Fe3+52 can dramatically decrease crystallinity. The presence of acetate groups also influences PVA’s crystallization behavior upon heat treatment. 53 Alternative approaches to modifying PVA’s properties include repeated freeze-thaw cycles, gamma ray irradiation, 54 and the use of derivative polymers like acrylamide-modified PVA. 55 PVA’s tendency to form gels at room temperature due to crystallite formation 56 can be exploited through freezing/thawing methods to enhance crystallite stability. Recent advancements in PVA gel preparation have focused on eco-friendly methods. One such technique involves hot-pressing swollen PVA, followed by a controlled gelation and drying process, resulting in gels with enhanced crystallinity and mechanical properties. 57
Blending PVA with other polymers offers another avenue for property enhancement. Combinations with poly(vinylpyrrolidone) (PVP), 58 polyethylene glycol (PEG), 59 and poly (glucosyloxyethyl methacrylate) (poly (GEMA)) 60 have shown improvements in thermal stability, mechanical properties, and gas transport characteristics. Additionally, reinforcing PVA with carbon nanotubes 61 has yielded promising results in enhancing its overall performance.
Synthesis methodologies for PVA films
Commercially available PVAs offer remarkable versatility, allowing for tailored adjustments to meet specific needs. These polymers are described through a spectrum of polymerization and hydrolysis depths, varied molecular weights (reflected in various solution viscosities), as well as varying degrees of acetate group removal from the polyvinyl acetate precursor. A development of doped PVA films for optoelectronic applications necessitates a holistic approach, as alterations at any phase could substantially impact the final yield physicochemical properties. 13
The preparation of PVA films typically follows a multi-step process. Initially, the PVA powder, through specific molecular weight as well as hydrolysis/polymerization degrees, is dissolved in a solvent. Common solvents include mono-distilled H2O, deuterium oxide, water-methanol mixtures, acetonitrile, dimethyl sulfoxide (DMSO), dimethylformamide (DMF)/water blends, or chloroform. This dissolution process often involves ultrasonication or heating at 60–90°C for 1–3 h, with duration as well as circumstances varying based on the polymer’s characteristics. A solution then undergoes doping, followed by additional heating to eliminate air bubbles. According to the technique of the dopant and the blend processing with other substances, additional steps may be incorporated. 62 The obtained yield from solution/gel stands and placed onto betray dishes using various techniques such as cast method, dip coated technique, spin coating process, or substrate immersion, with controlled thickness. The final treatment involves drying through heat or vacuum processes, and potentially irradiation. Thick films often develop a crystalline skin at the air-sample interface due to rapid solvent evaporation. 63 Film properties can be enhanced through simple methods like gel ultrasonication. High-power ultrasonic treatment (>300 W for up to 10 min) can increase tensile strength by nearly 30% and reduce water vapor permeability by 11%, albeit with a decrease in opacity. 64 Some applications utilize multilayered structures, with each layer possessing distinct thickness or physicochemical properties. 65 PVA can also be fabricated into microspheres ranging from 10 to 250 µm in size, with wall thicknesses of 1 to 5 µm. These can be produced using droplet techniques or self-assembly of dispersed microdroplets, finding applications in microsphere bio-lasers. 25
Organometallic PVA films sometimes serve as intermediaries in creating structures for optoelectronics. When cured, these films can decompose to form highly dispersed nanoscale metal oxide phases, imparting high refractive indices (up to 1.9) to the final coating. 66
PVA films also play a crucial role in nanoparticle and nanowire synthesis. Acting as a capping agent, PVA regulates development and the final crystallite size in aqueous-based synthesis methods. For instance, binary nanoparticles (NiO and Cr2O3) can be synthesized using PVA as a medium in a thermal treatment method. 67 Similarly, ternary metal oxide nanocomposites have been created using zinc and iron nitrates or manganese sulphate. 68
This versatile approach to PVA film preparation and modification opens up a wide array of possibilities for tailoring material properties to suit specific optoelectronic applications, demonstrating the polymer’s continued relevance in advanced materials science.
Theoretical frameworks for optical analysis
It is known that light interacts with matter and forms the basis of optical properties, offering valuable insights into a material’s internal structure, bonding nature, and potential applications. This analysis begins by examining how incident light interacts with a substance - specifically, how much is reflected, transmitted, and absorbed. 69 When light transitions between media (e.g., from air to a solid), some may pass through while the residual part suffers from the absorption or either reflection at the interface
Equation (1) illustrates this relationship:
Dividing equation (1) by 
Here, A represents absorbance (
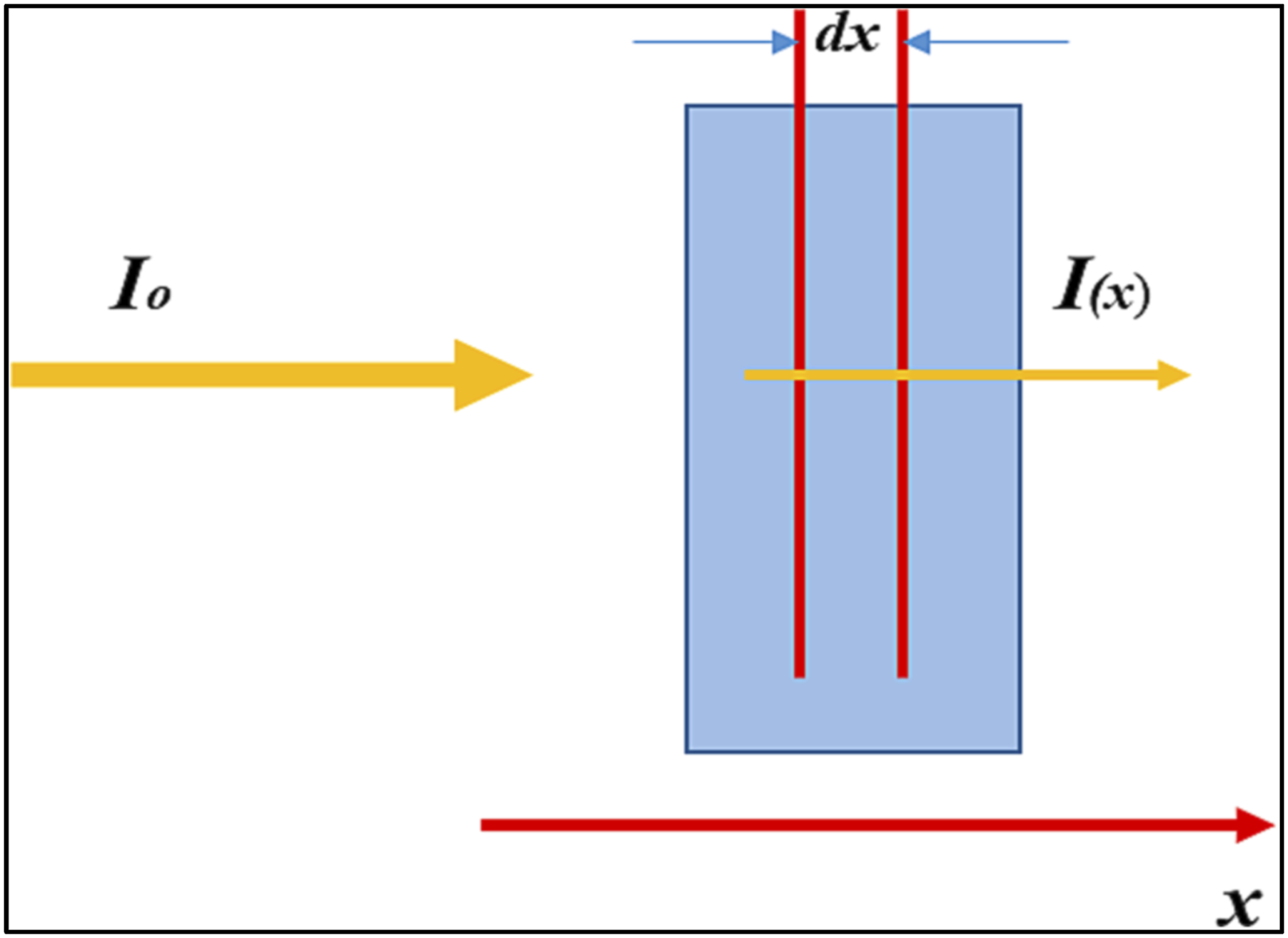
The absorption coefficient quantifies the reduction in the radiation energy flux per unit distance as a wave propagates through a material. It depends on the absorbing substance’s nature and the incident wave’s wavelength. All materials exhibit some degree of absorption, which varies based on their ability to guide electromagnetic waves. General absorption occurs when a medium absorbs all wavelengths relatively equally, while selective absorption or “window” absorption happens within specific spectral ranges. It is known that the medium can transvers the plane wave by reducing both the intensity and the amplitude of it along the propagation path. This phenomenon, illustrated in Figure 2, is mathematically described by the Beer-Lambert law. This law asserts through investigates the quantity of the absorbed light as a function of the thickness of the material. that the amount of light absorbed by a material depends on and is a function of its thickness. For a homogeneous material of thickness dx, Beer-Lambert established a quantitative relationship between the instantaneous light intensity I and the decrease in monochromatic light intensity. Mathematically
70
: Schematic diagram for the interaction between the light with intensity (I0) that decreases when passing through a slab or sample with thickness (x), where the final intensity of the light is (I). (Reproduce with permission
73
).
The absorption coefficient also allows for the calculation of optical density (OD) and skin depth (δ):

OD, furthermore, named as absorbance, is proportionate to both the optical medium’s thickness and the amount of absorbing material. 71 Skin depth, or penetration depth, represents the energy loss as an electromagnetic wave propagates through a material. 72
The refractive index is a fundamental optical parameter that measures how light speed changes when passing from a vacuum into a material. It essentially describes the degree of light “bending” or refraction upon entering a medium.
74
The basic definition of refractive index, n, is:
For thin films on substrates, optical constants are vital parameters, particularly for measuring the coefficient that represents electro-optic parameter for the polymer’s structures. This review applies a fundamental mathematical approach in order to estimate the film thickness (d) and the additional essential optical parameters like refractive index (n), absorption coefficient (α), and extinction coefficient (k) from transmitted spectrum data. 76 Accurate measurements require sensible treatments of film thickness. Designed for transmission-based measurements, the film thickness should be optimized to maintain transmittance within 3% to 90%. Transmittance below 1% leads to unreliable and inaccurate results. 77
Figure 3 illustrates the principle of obtaining a transmitted spectrum as light propagates through a material. For thin films, the Swanepoel method is particularly effective in calculating both refractive index and film thickness directly from the transmittance spectrum.
78
This approach applies to thin films on transparent substrates, where the substrate maybe significantly has more thickness than the film. In homogeneous films of uniform thickness, interference effects create consecutive maxima and minima in the transmittance spectra, as depicted in Figure 4. Where the transmittance can be estimated from the next relationship: Illustration represents the propagate of the electromagnetic beams of the light throughout the given thin film on the substrate. Reproduce with permission.
76
Schematic diagram represent the interface fringes obtained at the interface related to the thin films and the transparent substrate upon light beam transmission. Reproduce with permission.
79
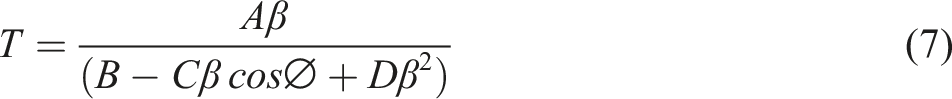
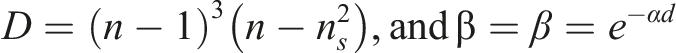
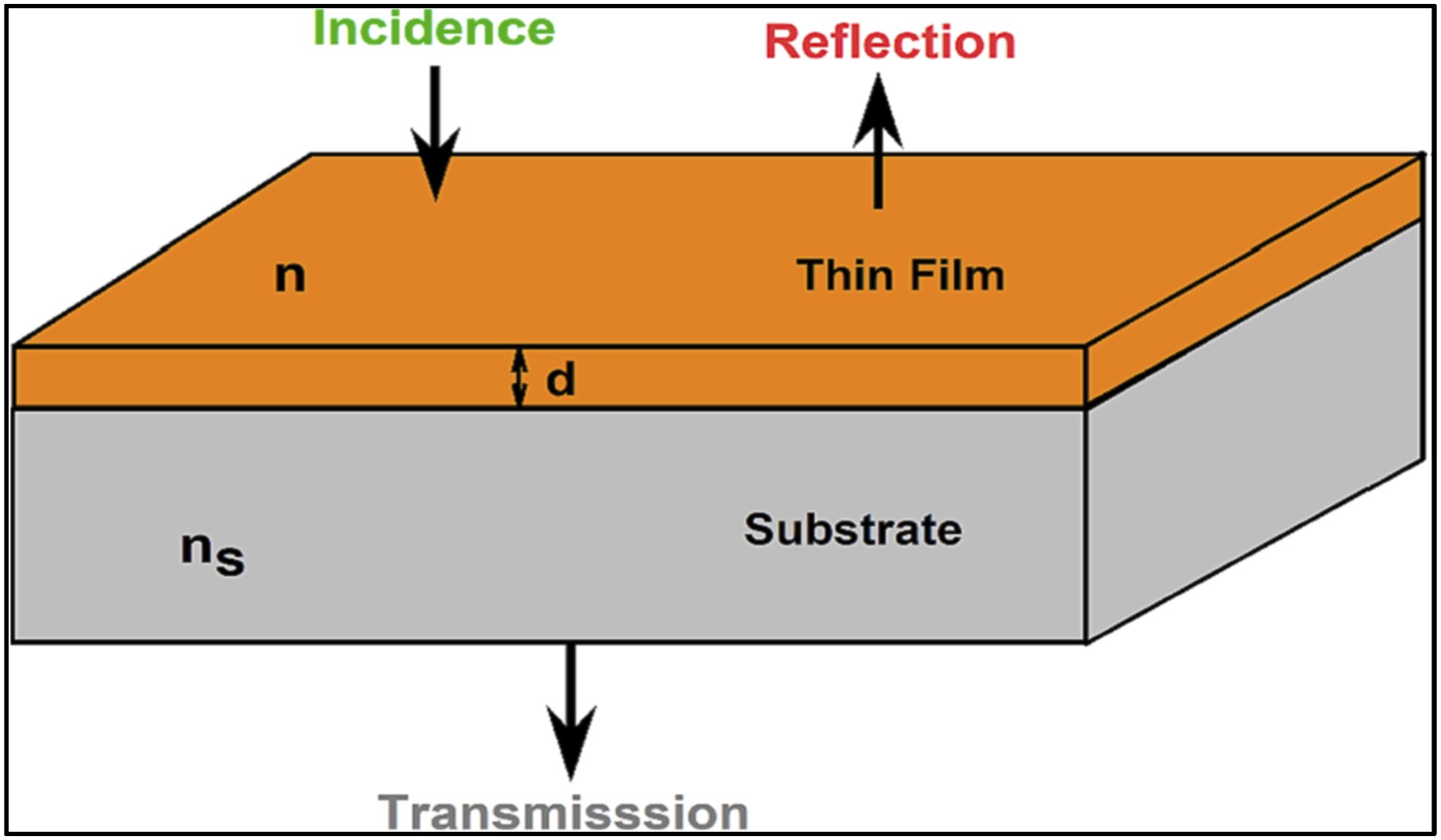
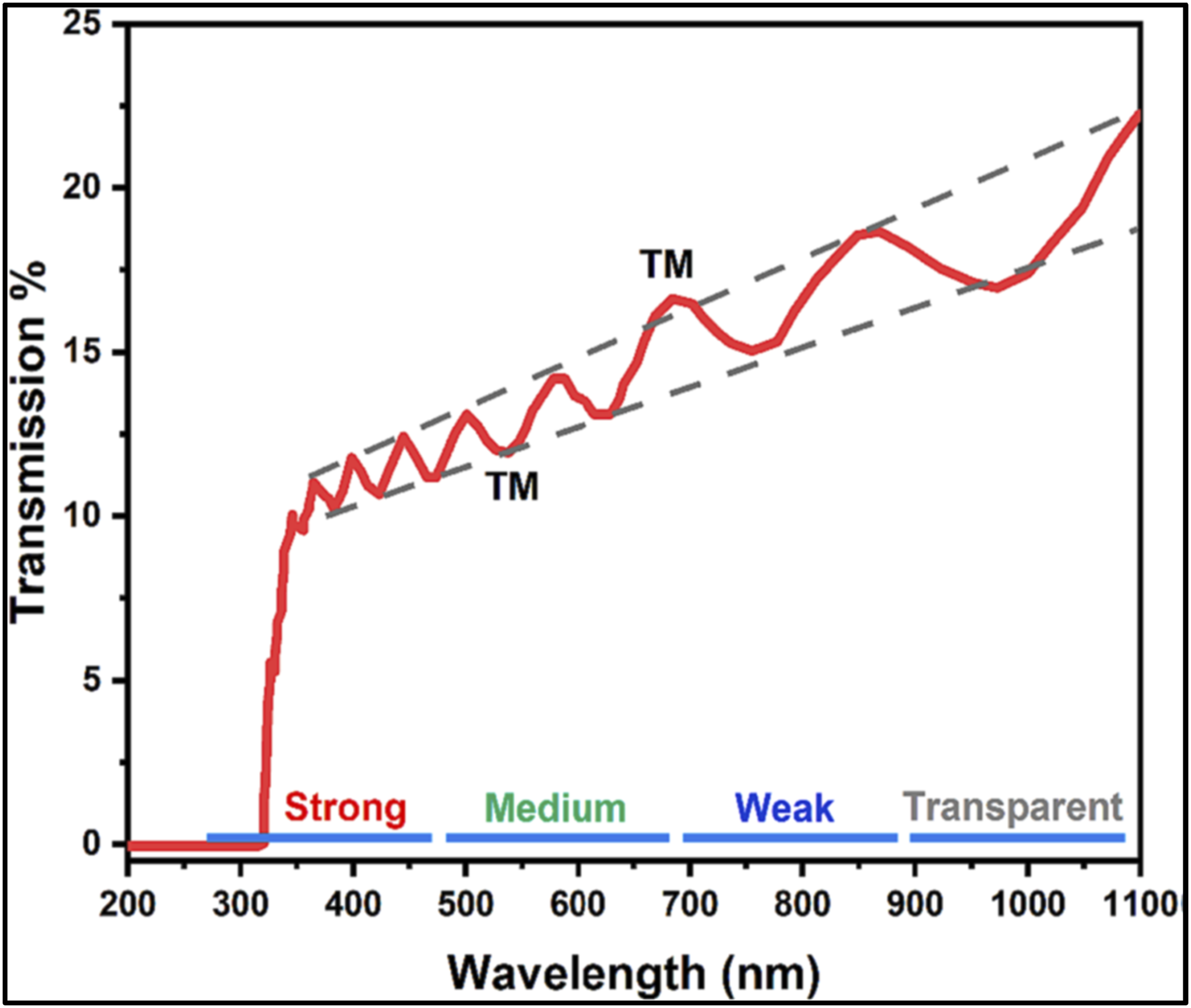
Also, n, d, ns represents the refractive index, thickness for the film, and the refractive index of the transparent substrate.
On the other hand, the obtained maximum (TM) and minimum (Tm) transmission at a fixed wavelength could be expressed as:

To apply this method, one first calculates the maximum and minimum transmittance envelope functions, TM(λ) and Tm(λ). So, the substitution into the equations for A and C into these functions, the refractive index n(λ) could be determined.
79
Also, by the assistance with the previously equations, the extinction coefficient could be determined by the given equations:
Understanding n and k allows for the determination of both real and imaginary components of the complex dielectric constant, ε1 and ε2. In thin film analysis, transmittance spectra can be used to derive n, α, and d, from which other optical parameters could be determined. For freestanding films without substrates, the absorbance spectra are obtained optical constrains.
Dielectric function analysis
In polymer composites, optical properties and electromagnetic wave transmission are mainly described throughout alterations in the optical dielectric constant. This finding portrays the substance’s ability to control electromagnetic waves and redistribute charge within a waveguide structure. 80 The dielectric constant comprises a real part (ε1) and an imaginary part (ε2). A real component governs a sample’s ability on the way to store light energy, while the imaginary parameter represents energy dissipation due to polarization.
Maxwell’s equations relate a media’s refractive index with the relation of it with the dielectric constant, εr:
This implies that both n and εr are complex. The complex relative dielectric constant 



Expanding this relationship yields:

The Kramers-Kronig relations, derived from complex number analysis and causality theory, connect the real and imaginary components of the refractive index
81
: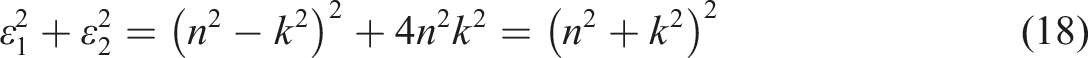


From this, we can derive:



These formulas demonstrate the essential relationship between absorption as well as an imaginary parameter related to the dielectric function and the relation tied the refractive index and the real component of the dielectric function. This generalization, however, is predicated on controlling medium that have a lower absorption coefficient 82
Microscopic models
Throughout optical media, light-matter interactions and optical phenomena take place on a minuscule microscopic scale. Various investigations were employed for generating as well as characterize optical factors. There are two types of models that have been developed to explain optical behavior in solid materials: semiclassical and classical models. 83 :
Classical model
The classical approach treats electromagnetic radiation and the optical medium in accordance with the laws of classical physics. In specifically, a typical methodology is used to quantitatively interpret the issue using a simple dipole oscillator model. This one delivers the basic outline for sympathetic general optical characteristics of a medium and for articulating the major effects in the free electron theory. 84
Harmonic oscillator model
The Drude and Lorentz models clarify the optical properties for different types of substances and are based on the electronic kinetic theorem of tiny electrons. As seen in Figure 5, the model was further improved upon from its first formulations throughout undertaking that a charge (electron) within a medium (atom) act as a harmonic oscillator limited by a nucleus. In proportion to the applied electric field, electrons oscillate when an incident electromagnetic wave passes throughout a medium. This charge oscillation causes charge redistribution, which creates an additional induced electric field from the viewpoint of the dynamic or atomic world. The charges then, with the help of the induced field, return to their equilibrium position. The illustration depicts the conventional conceptualization of a polar molecule’s behavior. Within this model, the constituent atoms, bearing opposite electrical charges, oscillate in a circular pattern around their equilibrium positions. Upon attaining resonance frequency, these vibrational motions generate a fluctuating electric dipole, which consequently emits electromagnetic radiation. Furthermore, the electrostatic forces exerted on the charged atomic components induce a responsive interaction between the molecule and the electric field component of incident light waves. This representation elucidates the fundamental principles underlying molecular spectroscopy and light-matter interactions at the atomic scale.
73
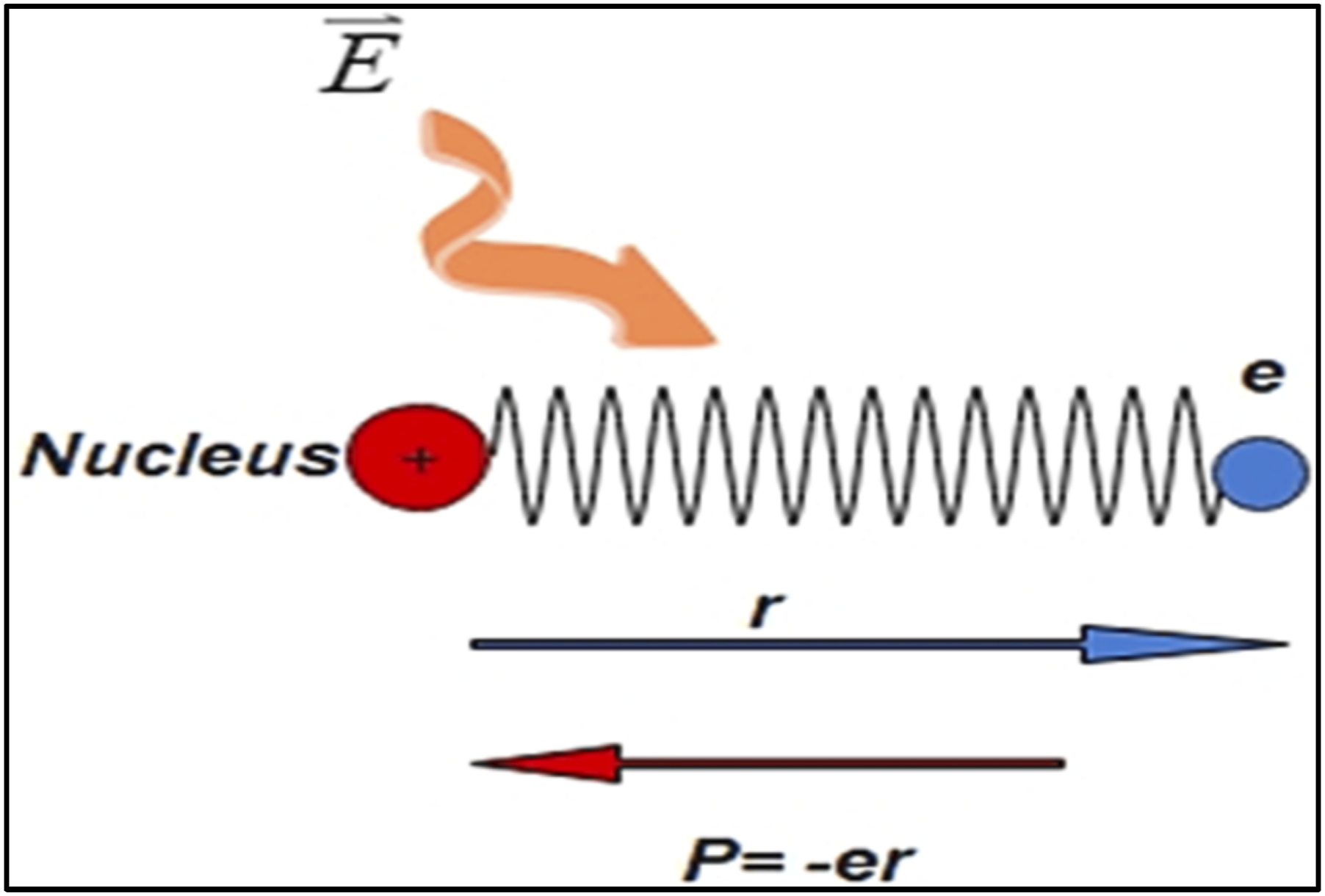
Through this model, the plasma frequency ωp is introduced, indicating the rapidity with which electrons could pursue the oscillation related to the application of the electric field. It varies for several substances that are related to N, which differs in proportion to the material composition. It is known that plasma frequencies obtained for some materials may have high values of N typically exist with the UV spectral region through the following relation:
Equation (25) describes the physical mechanism that is related to the plasma frequency associated with the bulk material ωp. The plasma frequency represents the frequency which the material response due to the electromagnetic waves related to the transition through the dielectric from metallic behavior. The optical features of the media perform akin to those of a metal at frequencies lower than the plasma frequency.
An attenuation occurs for the wave plus the limitation of the propagation throughout the metals owing to the complex nature of the refractive index n. For instance, these media present in the ionosphere, where electromagnetic waves below the plasma frequency ωp are impeded by the plasma. This phenomenon explains the success of Marconi’s pioneering experiment in 1901 toward the transmitting of the radio signal transversely the Atlantic Ocean. Contrary to skeptics who predicted failure due to the signal traveling into space without return, the radio wave was reflected to Earth’s surface by the ionosphere. This unexpected outcome rendered the experiment an extraordinary achievement, catalysing a revolution in wireless communications. Conversely, at frequencies higher than the plasma frequency, the refractive index n becomes real. Consequently, metals are translucent to visible light while remaining transparent to high-frequency radiation such as X-rays and gamma rays.
The phenomena of energy dissipation by high-velocity electrons as they traverse the internal and upper surfaces of a material are expressed through the Volume Energy Loss Function (VELF) and Surface Energy Loss Function (SELF), respectively. These parameters serve as quantitative measures of energy absorption within the substance, attributable to either single-electron transitions or multiple transitions occurring within the solid matrix. The mathematical relations elucidating the dependence of these energy-loss parameters, VELF and SELF, on the real (ε1) and imaginary (ε2) components of the complex dielectric constant are formulated as follows 85 :
These formulations provide a theoretical outline for sympathetic interaction between high-energy electrons and the material’s electronic structure, offering insights into the substance’s optical and dielectric properties. The analysis of VELF and SELF can yield valuable information about the electronic band structure, plasmon excitations, and other fundamental material properties, making them essential tools in the field of solid-state physics and materials science.
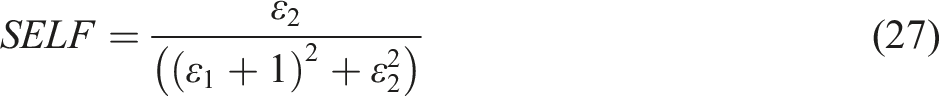
Additionally, according to above equations, the optical resistivity as well as the optical mobility can be estimated by using the following relations
86
:

Nonlinear optics
Nonlinear optics is a field that examines the interaction between matter and high-intensity coherent light, such as lasers, where multiple photons simultaneously engage with the exposed material. This phenomenon contrasts with linear optics, which occurs in low-intensity light-matter interactions involving minimal photon-atom or photon-molecule interactions. 87 The nonlinear optical effect can be conceptualized as the interaction between a material’s atomic oscillators and a high-intensity light beam.
PVA, organic and other polymeric materials show 2nd and 3rd harmonic generation when exposed to high-intensity laser beams, resulting in nonlinear polarization terms. Conversely, under low-intensity electromagnetic fields, polarization maintains a linear relationship with the electric field. 88 Nonlinear optics has found significant applications in various domains, including electro-optical modulators, self-phase modulators, signal processing, harmonic generator, ultrafast switching devices, frequency converting devices, including the pulse broadening, predominantly in case of the polymers substituted or doped with other materials. 89
The mathematical formulation of nonlinear polarization optical susceptibility is derived from the connection between displacement current and electric field intensity
90
:

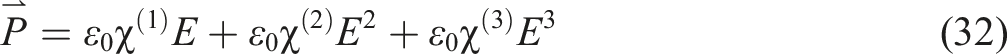
In this equation, χ(1) is the linear optical susceptibility tensor, while χ(2) and χ(3) denote 2nd as well as 3rd-order nonlinear optical susceptibility tensors, respectively.
The optical nonlinearity result typically includes a nonlinear refractive index term
91
:
The relation that tied the linear refractive index and the linear susceptibility according to the expression
91
:
In materials lacking centro-symmetry, such as lithium niobate, the second-order nonlinear term dominates. Conversely, in optical media with centrosymmetric atomic distribution, like certain transparent polymer materials, the second term in equation (32) is negligible, and the third term becomes the primary nonlinear optical polarization. 93
The relationship between χ(1) and χ(3) could be take the expression:


This treatment shed on the light on the nonlinear optical effects which provides a comprehensive overview of the phenomenon, its mathematical foundations, and its practical applications in various fields of optics and materials science.
Band gap analysis methodologies
Interband transitions
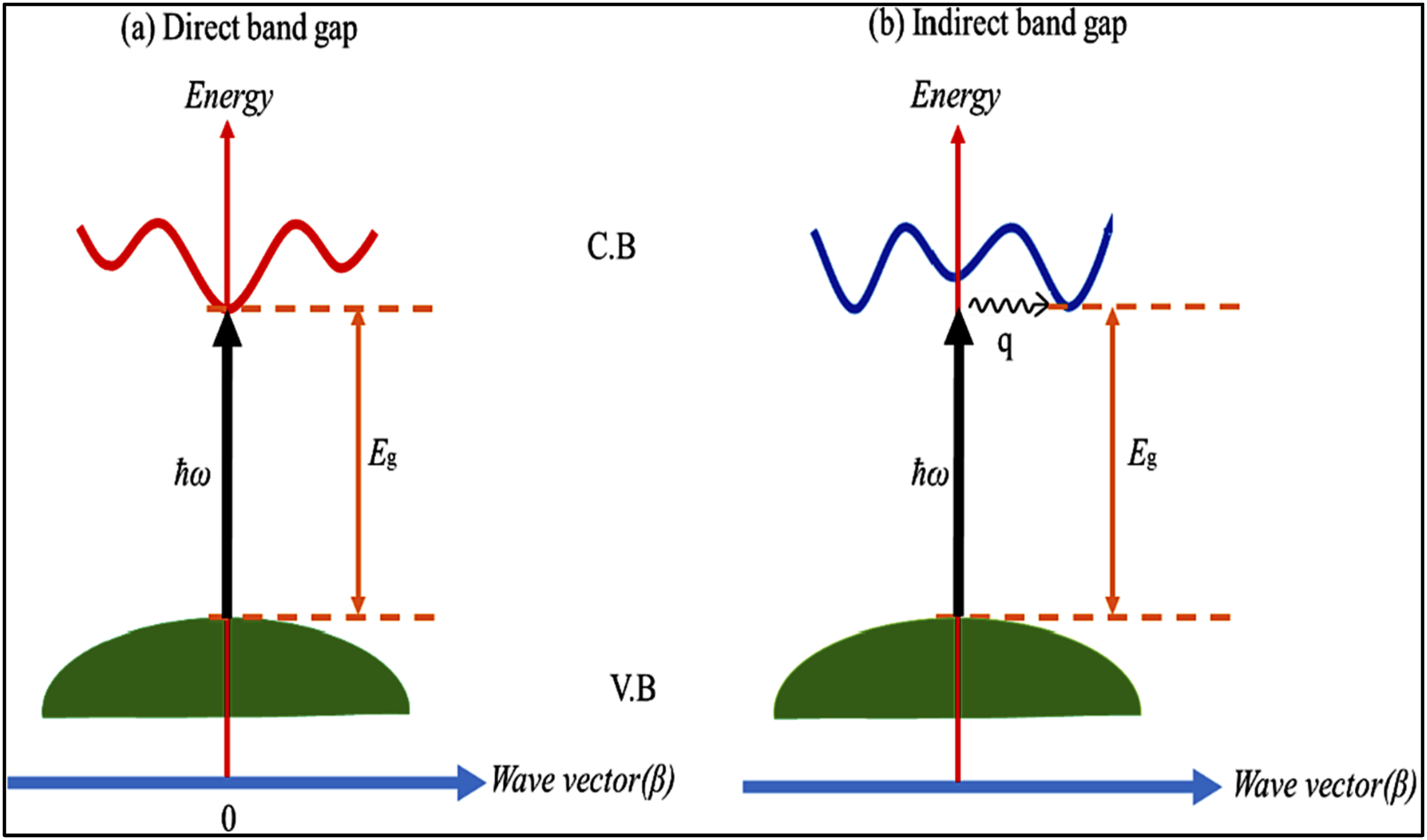
The band gap energy (Eg) in solids represents an energy zone where electrons cannot occupy electronic states. The band gap is known as the free space between the totally filled energy gap level or the valence band and the totally empty or partially empty energy level or the conduction band. This space or the Eg is strongly related to the electron transition between the two bands and associated with the gaining energy that the electrons have. The movements or the transition depends on some parameters like the atomic transition selection rules, Pauli exclusion principle. This Interband or the energy gap is related to distinctive energies for Insulators (∼10 eV), Semiconductors (∼1 eV) where in case of the conductors, an overlapping between the valence and conduction bands were found. 83
Applying energy conservation to Interband transitions: Demonstrates (a) direct and (b) indirect band gap optical transitions in solids. Reproduced with permission.
97

Momentum conservation is crucial in explaining these phenomena:

In indirect transitions, substantial electron wave vector changes require phonon involvement to conserve momentum, unlike direct transitions (See Figure 6(a) and (b)).
97
Indirect absorption, common in semiconductor-like structures, is technologically significant but more complex to analyse than direct transitions.
98
In bulk materials, the high number of atoms results in densely populated energy bands, leading to much higher absorption intensities for allowed transitions compared to dilute media like gases.
99
Basically, the momentum conservation is related to the transitions of the electrons in the materials, for instance, the indirect energy gap transitions is dominant for the non-crystalline semiconductors. On the other hand, the indirect energy gap. Consequently, according to the fundamental principles of physics, in order to assure the momentum conservation due to the electron transitions, the production of phonons is electron transition dependent. The relation ascribed the absorptivity of the materials that related to the absorption coefficient is given by
100
: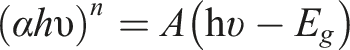
Tauc’s method to band gap analysis
At the atomic level, “basic absorption” states band-to-band transitions managed through identifiable according to the selection rules, characterized through a notable evolution of the basic absorption band. Transitions are classified depending on the substance’s band structure.
79
The photon energy (hυ), a constant associated with the band tailing (A), as well as the optical energy gap (Eg) are key factors. The exponent n defines the nature of optical transitions producing light absorption.
101
Depending on electron transfer mechanisms, n can take values of 1/3, 2, 1/2, or 2/3, related to the indirect forbidden, direct allowed, indirect allowed, and direct forbidden transitions, respectively
102
(equation (41)).
Wemple-DiDomenico (WD) technique
The Wemple-DiDomenico model, based on a single oscillator concept, examines optical dielectric response through refractive index dispersion at low frequencies.
103
This model evaluates crucial optical dispersion parameters such as dispersion energy (Ed), oscillator energy (Eo), and static refractive index (no at hυ = 0), essential for optoelectronic applications. The model relates incident light energy within the absorption edge to the refractive index:
Recent studies have utilized the WD model to assess optical dispersion parameters. For instance, Abdulla et al. 104 examined a PMMA composite doped with silicon powder, reporting Ed = 4.93 eV and Eo = 5.19 eV at 4 wt% Si. Nofal et al. 105 investigated PVA composites with cobalt (II) complexes, finding Ed = 4.93 eV and Eo = 2.15 eV at 60 mL concentration. Aziz and colleagues 106 studied PVA doped with aluminum (III) complexes, noting Ed = 1.34 eV and Eo = 1.86 eV at 60 mL concentration.
These values are typically derived from plotting Schematic diagram for the Variations of (n2 − 1)1/2 as a function of the energy of the photon (hυ)2 for the PVALMC1 and PVALMC2 samples. Reproduced with permission.
86

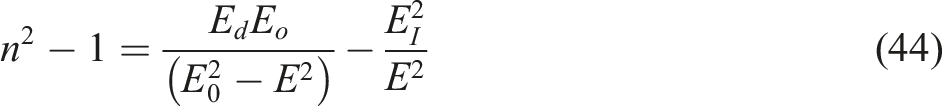
For wavelengths longer than phonon resonance (
Plotting
The WD model also estimates optical spectra moments M-1 and M-3:

In order to estimate M-1 and M-3, one can divide equation (46) on (47),

These relationships allow further characterization of a material’s optical properties.
PVA/nanocomposites films optical and electronic characteristics
Comparative analysis of various nanocomposite systems
The unique structure of polyvinyl alcohol (PVA) and the chemical versatility of various additives allow for facile manipulation of the polymer film’s physical characteristics. Enhancement of film properties can be achieved through the incorporation of diverse dopants or fillers through the host matrix of the polymer, including salts, carbides, nanocomposites, ions, and even other polymeric materials.
The resultant composite characteristics are heavily manipulated through the nature of the modifying agent. An illustrative example is the use of two quaternary ammonium salts with similar compositions as polymer additives: Cloisite15A and Cloisite30B. 108 The former, which experiences slight intercalation by interacting polymer macromolecules throughout melt compounding, influences an increase in glass transition temperature and improved mechanical assets of the final composites. In contrast, Cloisite30B, which undergoes significant intercalation, partially disrupts the bonds network of the hydrogen inside the polymeric host matrix leads to the reduction in both mechanical and thermal characteristics.
Performance can frequently be improved for a variety of solid-state devices, including emissive displays, integrated optical circuits, and optical sensors, by coating the light-emitting or sensing surface with a transparent material that has a high refractive index. By implementing this technique, a progressive shift in refractive index is produced, which enhances the efficiency of light coupling between components with different optical characteristics. By covering the light-emitting or sensing surface of many solid-state devices—such as integrated optical circuits, emissive displays, and optical sensors—with a transparent coating that has a high refractive index, performance can be greatly improved. More efficient light coupling is made possible by this method’s smooth shift from the active circuitry’s high refractive index to the air’s low index.
One method to achieve such a film is by blending PVA with other polymers. For instance, research described in Ref. 13 demonstrates that a biodegradable composite film of PVA and PVP can enhance the values of the refractive index of PVA by up to 20%. Another works had proven the positive effect of using Metal oxide nanoparticles as dopant in PVA/PVP blend matrix which affect the optical and electrical properties of PVA/PVP blend. 109 By combining two polymers—polyvinyl alcohol (PVA) and cashew gum (CG)—with varying amounts of nanochitosan (nCS), biopolymer nanocomposite films were created using the green solution casting technique. The nanofiller loading improves the electrical conductivity and dielectric characteristics, according to the electrical impedance measurements. PVA/CG/7wt% nCS had an AC conductivity that was 1.46 times greater than that of the pure mix. 110 Using in-situ polymerization with water as the eco-friendly solvent, Ayisha Jemshiya Kalladi et al reports the successful synthesis of boehmite (BHM) nanofiller-reinforced ternary mix nanocomposite films made of polyvinyl alcohol (PVA), cashew gum (CG), and polypyrrole (PPy). Analysis of AC conductivity showed a rise in dielectric constant (from 565.28 to 4411.22) and a decrease in activation energy (1.277 × 10−5 eV at 7 weight percent BHM). 111 M.T. Ramesan et al. created a polymer blend composite using poly (vinyl alcohol) (PVA) and cashew tree gum (CTG) along with different amounts of pumice particles. Because the pumice particles inside the polymer chain are more ordered, the mix composite containing 10% sample exhibits better conductivity and dielectric qualities than the pure blend. The agglomeration of filler particles in the polymer matrix caused the composite’s electrical characteristics to diminish when the pumice loading was raised above 10 weight percent. The biodegradation of the composite is slower than that of the pure blend. 112
Alternatively, doping PVA with various substances can also alter its optical properties. Metal ion implantation and plasma treatment are techniques employed to modify the physicochemical and electromechanical properties of materials, enhancing optical aa well as the conduction possessions, particularly in ceramic materials. Designed for inertly polymeric materials, ion beam treatment can generate ultra-thin conducting or reflecting films beneath the polymer surface. This method has found applications in strain gauge development, 113 increasing PVA’s crystallinity and microhardness, 114 producing planar light guides, 115 as well as enhancing photoluminescence. 116
XPS or X-rays photoelectron Spectrometer analysis of ion beam-treated PVA reveals that metallic implants (such as magnesium and zinc) exist in both metallic and oxidized states. The surface morphology modifications, dependent on implantation dose, result in increased free surface energy, crystallinity, and microhardness. 113 It was found that the implementation of lithium and nitrogen ions into the PVA films could demonstrate anisotropy in the refractive index related to the variations throughout the irradiation process exceeded 0.05. The obtained result is attributed to the production of aromatic composites in different sites of the electronic scattering. M-line spectroscopy analysis of a 3–5 μm thick film irradiated with a low implantation dose (<1014 ions/cm2) shows total waveguide loss values below 2 dB/cm at 633 nm wavelength. 115
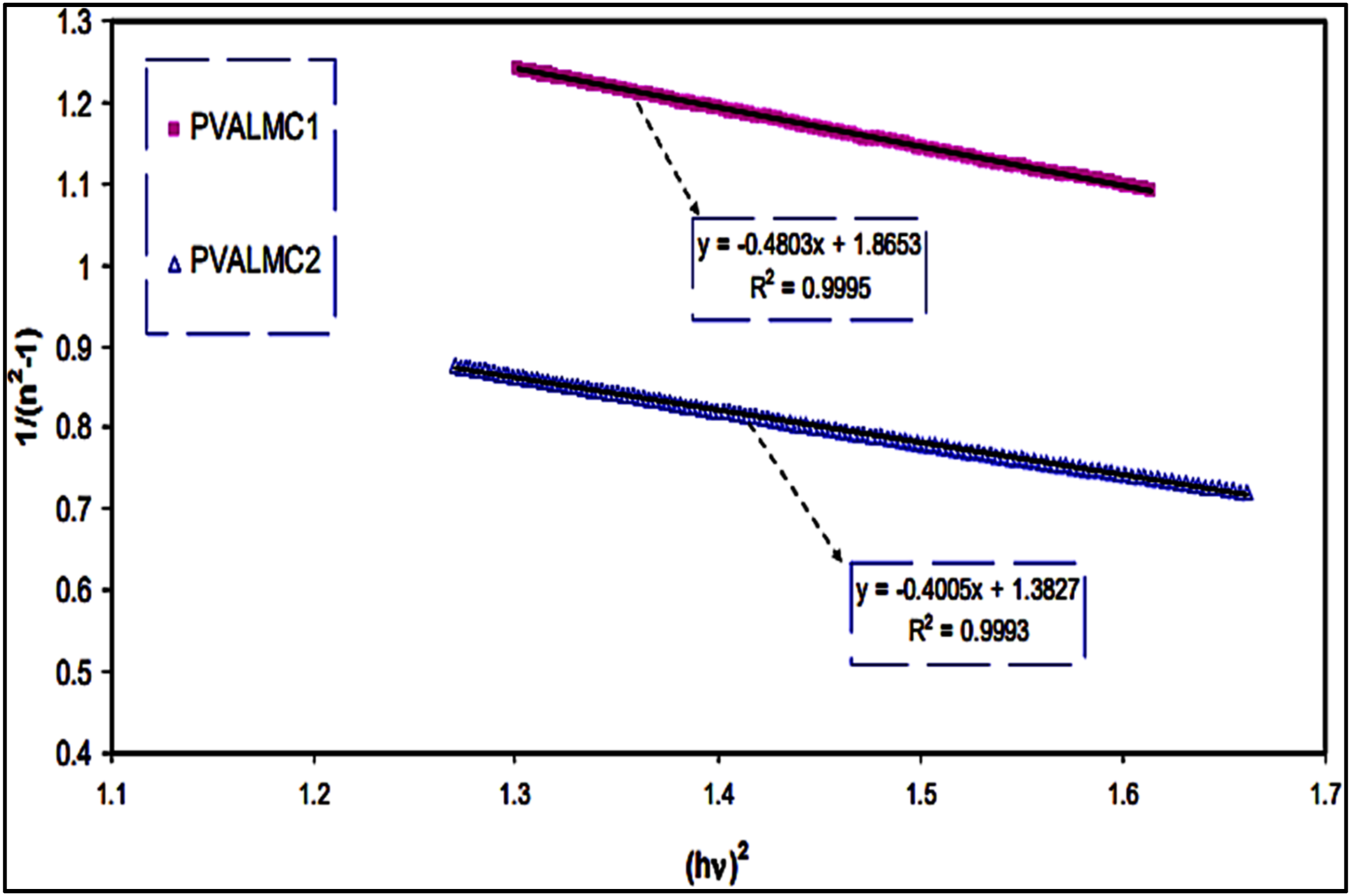
Likely, triple valent nitrogen ions were implemented into (Cel-PVA-Si) which is named cellulose-reinforced polyvinyl alcohol silica composition
116
demonstrates notably modified luminescent, thermal (Figure 8(a)–(c)), and mechanical characteristics, along with a 25% improvement in electrical conductivity (Figure 8(d)). FTIR analysis (Figure 8(e) and (f)) indicates bond breakage in ion-implanted samples. The production of microstructures enhances surface roughness and increases sample stiffness by approximately 50 times compared to unaltered films. The native cellulose’s photoluminescence is greatly improved due to defect sites and dangling bonds. PVA exhibits beneficial compatibility with most inorganic and organic fillers, allowing for the preparation of enhanced composites without the need for coupling agents or interfacial modifiers.
117
This versatility has made PVA-based materials particularly interesting for various applications in optoelectronics and materials science. (a) First DSC heating scan of PVA, cellulose, and pristine and implanted composites, (b) detail of the low temperature range, (c) second DSC heating scan of PVA, cellulose and pristine and implanted composites, (d) nyquist plot of (a) pristine (b) 1e15 (c) 5e15 (d) 1e16, (e) FTIR spectra of pristine and ion implanted Cel-PVA-Si, (f) zoomed image showing the peak shift of Si-O-Si bond (Reproduced with permission
116
).
The dopants with metal inorganic composites into PVA: PVA is compatible with most inorganic as well as organic fillers, therefore coupling agents and interfacial modifiers don’t always need to be included to create improved composites. 117 In earlier times, the huge interest in the optoelectronic data availability and data processing was initiated in the 1990s to use polymers that doped with various transition metals. In vinyl-polymer solutions, metal oxides as well as inorganic salts are the most utilized additions; nevertheless, practically several inorganic metallic compound could serve this purpose. An ultimate allocation of metallic ions into polymeric materials are not have similar size or complexity, even when doping is carried out among metal salt solutions and the dopant metal was thought to be a straightforward chemical combination. For instance, the octahedral complex [FeCl2(H2O)4] + and the tetrahedral [FeCl4] − are the primary species in aqueous solutions of ferric chloride; the latter is more common in solutions with high concentrations of chloride and at high temperatures. 118 Because of the unique intimate semi-crystalline structure of PVA, the obtained PVA film doped with Fe, which is produced from the combination of both polymer and salt solutions, comprises small collections of iron ions.
The distinct performance of the temperature-dependent Mössbauer curves of PVA films doped with FeCl3 provides direct evidence of the accumulation of iron ions onto microscopic clusters of 1–2 nm size (<100 atoms). 92 The presence of the magnetic spectral component (sextet) at 5 K 118 indicates the existence of tiny clusters of Fe3+ ions with a blocking temperature lower than 50 K, even though the Mössbauer spectra of specific molecules of FeCl3 in PVA had better composed of paramagnetic doublets till the temperature reaches to its lowest value at 5 K. It is really common for the metal compound to form clusters, making it challenging to differentiate between polymers substituted with metals and those doped with nanoparticles. 119
The modification of PVA films through doping offers a versatile approach to tailoring their mechanical, crystalline, and optical properties. This technique allows for precise control over various characteristics, making PVA-based polymers are employed for multiple appliances in optoelectronics and materials science. Doping with conductive ions can significantly alter the crystallinity of PVA films. For instance, the additives from the divalent Nickel ions 51 or Fe3+ ions 52 can dramatically reduce crystallinity, with Fe3+ decreasing it from 41.6% to 7.7% at a 2.0% doping level. This reduction in crystallinity is accompanied by a substantial increase in tensile strength, up to 240% in some cases. 52
Transition metal dopants, such as ferric chloride (Fe:PVA), have been extensively studied for their effects on optical properties, particularly under UV exposure. The UV-induced changes in refractive index and absorption coefficient are attributed to alterations in the metal’s oxidation state and electronic density redistribution. These effects have potential applications in holography and integrated optics. 120
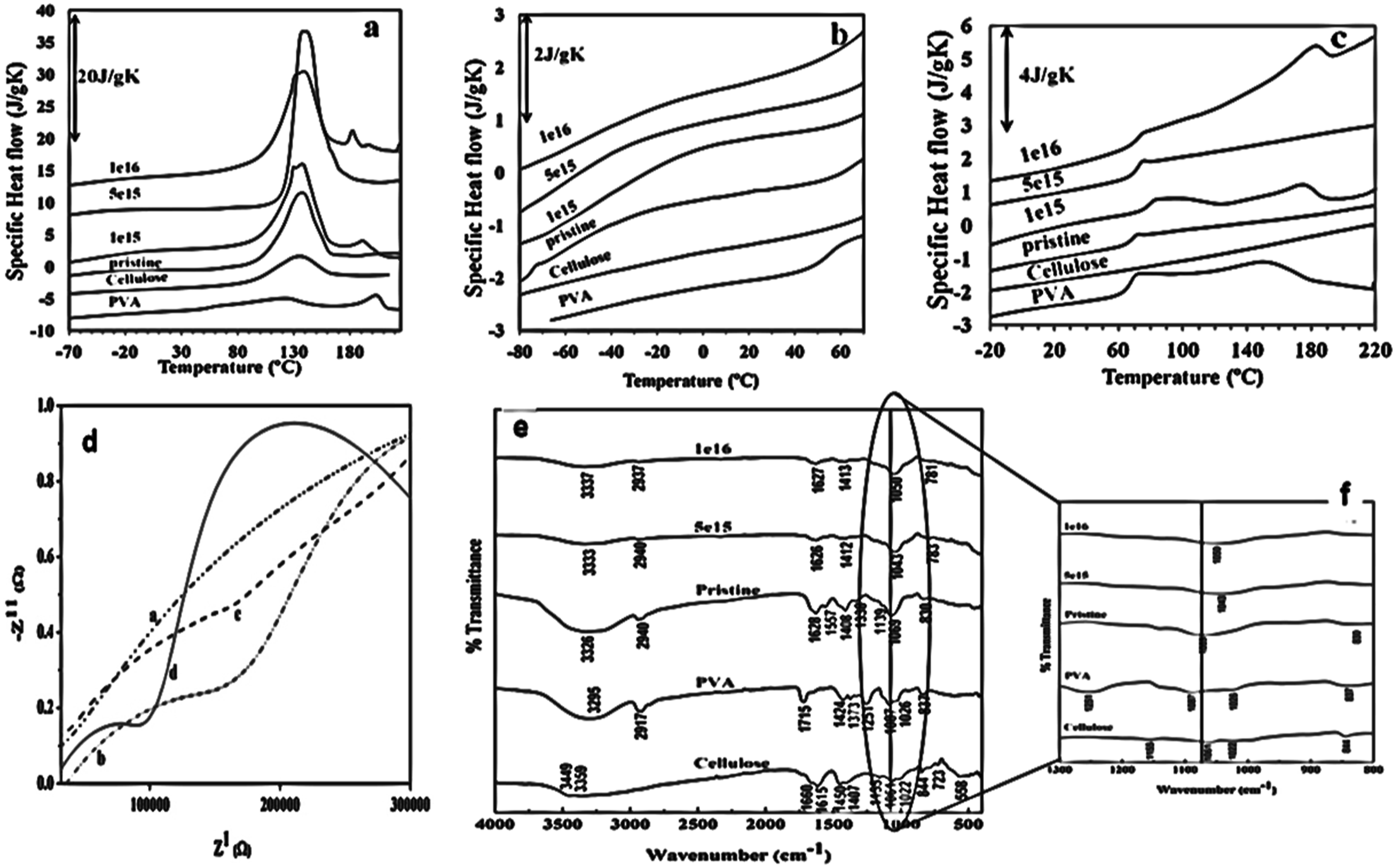
PVA can be implanted with metals, such as cobalt (Co), which can then be ultrasonically combined with the PVA solution to create metal-doped PVA sheets. 121 FTIR, UV- Vis spectra in the 190–900 nm range, scanning electron microscopy, and X-ray diffraction were employed to examine the entire dried films that were produced using the casting procedure. The transmission and absorbance data are employed to compute several optical characteristics, including energy bandgap and dielectric loss. At wavelengths of 632.8 and 533 nm, the optical limiting performance of PVA/Co-metal films were investigated. A strong interaction between the PVA matrix as well as the metal, in this case Co, causes a rise in internal strain and a reduction in particle size, which in turn leads to the expansion of distortion and impurities. This effect of the fillers’ concentration on the polymer material’s crystallinity is established by XRD. It was found that the PVA diffractograms showed the dropping of the peaks intensity, most likely as a result of interaction between the Co-particles and the hydrogen link, while the peaks from the Co-metal grew with the concentration of Cobalt in the polymer.
At low Co-metal percentages, the PVA matrix highlight is the dominations the film of Co/PVA FTIR spectra (Figure 9(a)), and the strength of the functional vibration groups in the matrix decreases, reflecting the strong interactions between the metal and PVA. The transmittance gradually dropped in the UV-Vis spectrum (Figure 9(b) and (c)), accomplishment less than 8% for PVA/10% Co films, as a result of the presence of Co-metal, which causes the particles to aggregate and raises the amount of incident light that is absorbed or reflected. The induced absorption bands, which display an energy-band gap dropped from 5.33 eV to 4.61 eV within the π and π* bonding along with antibonding molecular orbitals, were obtained by adding Co-particles at a percentage of 0.1–10% in the PVA matrix (Figure 9(d)). Overall, the polymer nanocomposite films are fit for multiple applications starting with attenuated laser machines and opto-photonic devices. Where, the play maker for achieving a promising optoelectronic device or solar cell panels is the optical energy gap of the material. (a) Schematic diagram for FTIR curves for (a) PVA film, and PVA/Co-metal composite films with various Co-metal ratios (b) 0.1%, (c) 0.5%, (d) 1%, (e) 5%, (f) 7.5%, (g) 10% (b) optical transmission absorbance spectra for PVA/Co-metal composite films (c) optical absorbance spectra for PVA/Co-metal composite films (d) the absorption coefficient α as a function of photon energy hυ for each as-prepared film (Reproduced with permission.
121
).
Rare-earth metal chlorides, like erbium chloride (ErCl3), when used as dopants, can introduce new optical transitions due to the unique electronic structure of Er3+ ions. 122 PVA: ErCl3 composites exhibit modified energy gaps and refractive indices, making them promising for optoelectronic applications. Other metal chlorides, such as titanium chloride (TiCl3), antimony (Sb), and tin (Sn), have been investigated for their effects on PVA’s optical and electrical properties. 123 This causes a change in the refractive index, which may have real-time holographic applications. Under UV radiation, experiments using 57Fe, 119Sn, and 121Sb Mössbauer spectroscopy reveal that the iron ionic state decreases, tin oxidizes (demonstrating an inverse electron transfer when compared to the case of Fe), and antimony stabilizes in its lower Sb3+ valence state and does not show any charge transfer phenomena.
These dopants can significantly alter the dielectric constant, electrical conductivity, and optical energy gap of PVA films. Multi-element metal doping offers another avenue for property enhancement. Combinations like (Fe + Sn) and (Fe + Sb) show complex interactions affecting the optical absorption and refractive index of PVA films. 13 These effects are attributed to charge transfer phenomena between different metal species under UV irradiation.
Nanocomposite formation with metal nanoparticles is an emerging area of interest. For example, PbS nanoparticles doped with Zn, Mg, and Cd in PVA matrices show promising optical limiting properties. 124 The exploration of multi-metal doping in PVA films has unveiled a rich landscape of material properties and potential applications. For instance, the combination of CrO3 and CuO as dopants in PVA induces significant changes in the polymer film morphology and electronic charge transfer processes. These modifications directly impact the optical characteristics of the films, particularly their transmittance and refractive index. 125
Over the years, MnO2 and its derivatives have attracted numerous notice due to their remarkable structural flexibility and distinctive physicochemical properties. MnO2, a member of the manganese oxide series, has attracted much of studying by scientists because of its capacitance (1375 Fg−1), wide application window (1.0 V), and numerous crystal forms (β-, α-, and γ-types). Using the solution cast process, polyvinyl alcohol (PVA) was combined with concentrations of 5%, 7.5%, and 10% manganese dioxide (MnO2) to create PVA/MnO2 films, as reported by Norah A. M. Alsaif et al. SEM micrographs of the films PVA, PVA/2.5%MnO2, PVA/5%MnO2, and PVA/10%MnO2 are shown in Figure 10(a)–(d). By the addition of MnO2, SEM pictures of the PVA film demonstrated the existence of morphological alterations. In the frequency range of 190–1000 nm, Alsaif et.al, displays the optical reflectance (R) with wavelength for PVA and PVA/MnO2 films. The optical reflectance is affected by wavelength changes. MnO2 application also increased the PVA film’s reflectivity. Then, changes in optical reflectance followed an increase in MnO2 concentrations. The refractive index of PVA and PVA/MnO2 changed with wavelength. When the wavelength was extended, all films displayed an enhancement in their index n. Both the incoming radiation dispersion and the refractive index of the pure PVA film were improved by the addittive of MnO2. The PVA’s dispersion energy was 2.53 eV, compared to 4.86 eV for PVA with 5% MnO2, 5.74 eV for PVA with 7.5% MnO2, and 7.12 eV for PVA with 10% MnO2. Furthermore, for PVA/5%MnO2, PVA/7.5%MnO2, and PVA/10%MnO2, the oscillation energy E0 dropped from 3.79 to 1.94, 1.72, and 0.99 eV, respectively. Also, they show the relation through (n2–1)−1 and (hυ)2 for PVA and PVA/MnO2 films and the other optical remarks.
126
Schematic diagrams for SEM micrographs of PVA/MnO2 wt.% : (a) PVA, (b) PVA/5.0%MnO2, (c) PVA/7.5%MnO2, and (d) PVA/10% MnO2 (Reproduced with permission
126
).
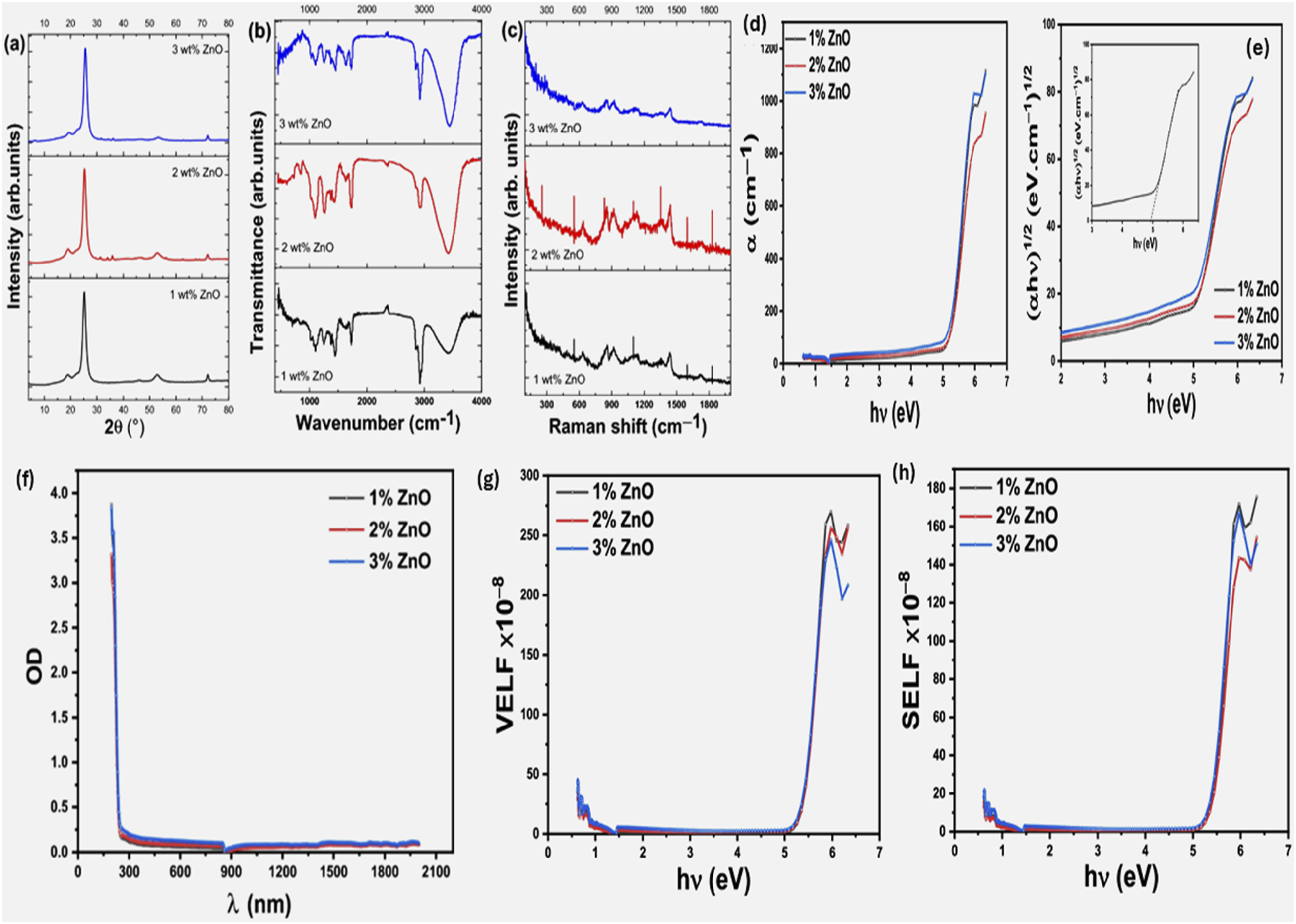
Alaa et al., studied thick films from PVA with different weight percent form ZnO nanoparticles with ratios ranged from 1–3 %. They achieved the wurtize hexagonal ZnO phase which used as a dopant in the PVA host matrix. These data confirmed from the XRD, and the structural parameters were calculated besides the vibrational modes for PVA/ZnO nanocomposite films from Raman spectroscopy as well as the different functional groups and the chemical bonds from FTIR Analysis, See Figure 11(a)–(c).
115
On the other hand, the optical properties and parameters, especially, the linear and nonlinear optical characteristics were investigated. The authors proved that ZnO nanoparticles has slight effects on the structural parameters of PVA but demonstrates noticeable changes on the optical characteristics (linear/nonlinear). Figure 11(d) and (e) show the absorption coefficient and the optical energy gaps for the PVA/ZnO nanocomposite films.
115
The direct and indirect optical energy gaps were 5.1 & 4.93 eV and 4.45 & 4.89 eV for PVA films with 1 wt% and 3 wt% from ZnO nanoparticles concentration. Also, Figure 10(f)–(h) show some of the optical parameters that the authors obtained from the optical properties of PVA/ZnO nanocomposite films for the optical density, VELF, and SELF.
115
Finally, the authors recommended to use their work according to the obtained optical results in various electronic and optoelectronic applications. Schematic diagram for the obtained results form PVA/ZnO with different ZnO weight perecnts (1,2, and 3%): (a) The XRD diffractograms, (b) FTIR obtained results, (c) the Raman spectrometer, (d) the absorption coefficient, (e) the optical energy gaps for the obtained samples (f) the optical density results as a function of the wavelength, (g) & (h) the VELF and SELF as a function of (hυ) for the obtained films (Reproduced with permission.
115
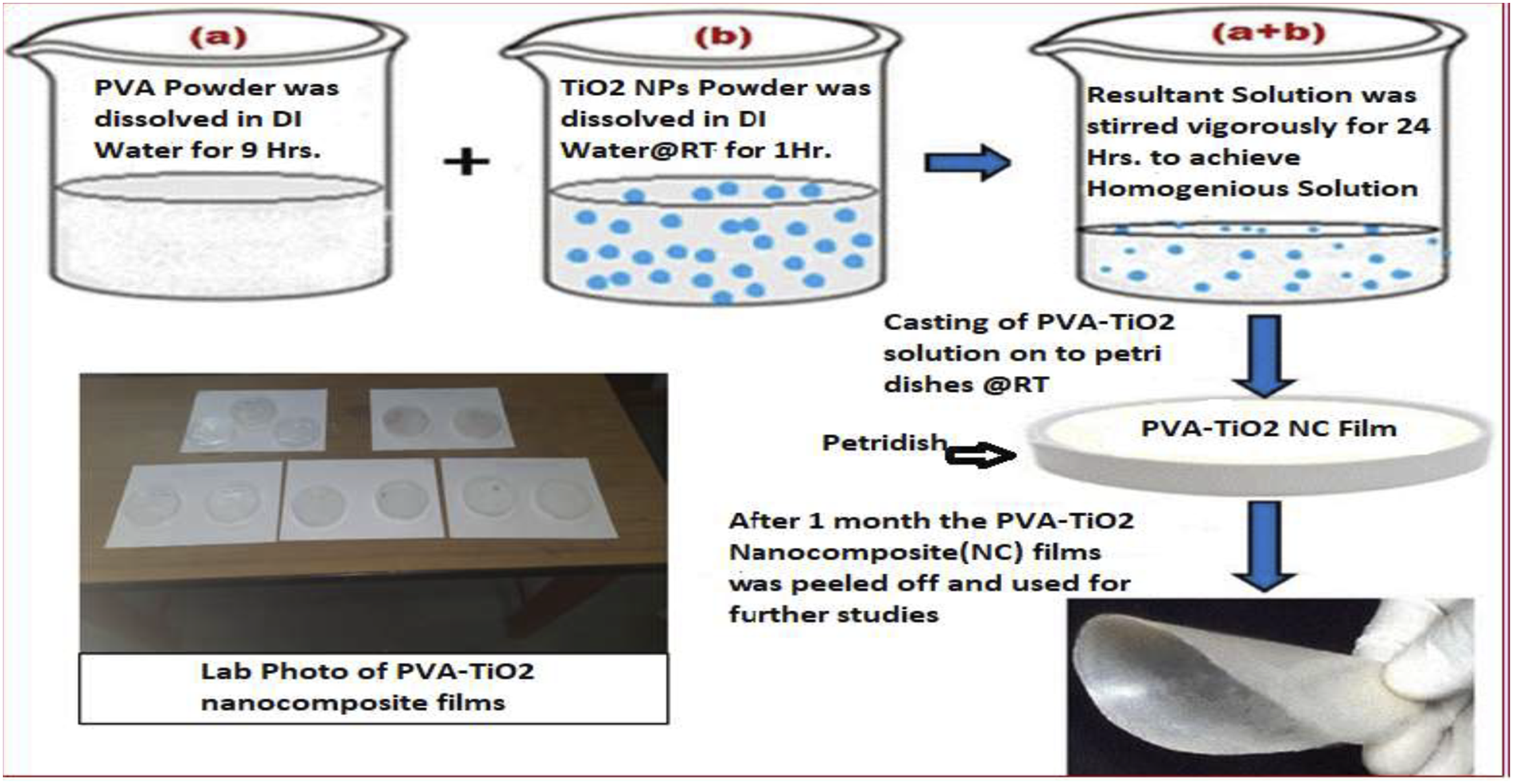
PVA nanocomposites with TiO2 incorporation have improved optical characteristics. TiO2 nanoparticles are well renowned for their superior photocatalytic activity and UV absorption. R Venugopal et al. used a facile casting technique to create the PVA-TiO2 nanocomposites films with various contents of TiO2 nanoparticles Figure (12). The direct bandgap decreased from 6.09 eV to 5.28 eV and the indirect bandgap decreased from 5.53 eV to 4.64 eV as a result of the addition of 8.0 weight percent TiO2 NPs to PVA polymer. The Urbach energy also dropped, going from 0.82 eV to 0.548 eV. The sample containing 8.0 weight percent at 338 K and 393 K, TiO2 demonstrated the maximum DC electrical conductivity, measuring 5.59 × 10−13 Scm−1 and 4.01 × 10−8 Scm−1, respectively.
127
Preparation of PVA-TiO2 nanocomposite films. Reproduced with permission.
127
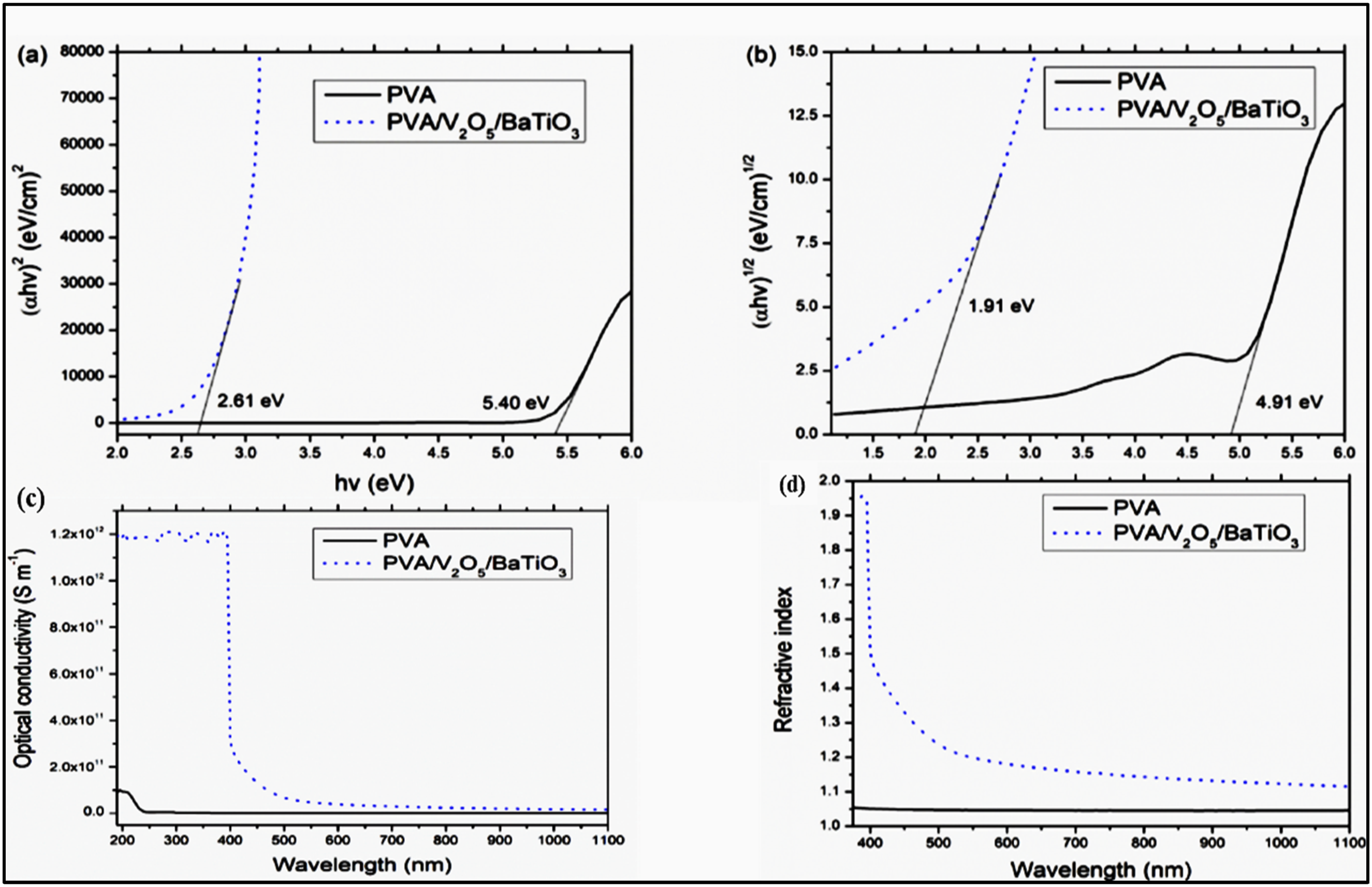
Using solution casting, Aayesha Shoukat et al. created a flexible film by including 1% of each of the barium titanate (BaTiO3) and vanadium pentoxide (V2O5) powder into the polyvinyl alcohol (PVA) matrix. By adding V2O5/BaTiO3 NPs, the PVA’s directly bandgap was decreased from 5.40 eV to 2.61 eV. The bandgap may have decreased as a result of the NPs from the added filler making a bond with the PVA matrix’s hydroxyl groups Figure 13(a). Moreover, the indirect bandgap decreased from 4.91 eV to 1.91 eV upon the addition of the fillers Figure 13(b). After adding V2O5/BaTiO3 NPs to the PVA matrix, the UV visible examinations revealed an excellent increase in the optical conductivity (2.7 × 109 to 1.2 × 1012 S m−1 at 400 nm) Figure 13(c) and an improvement in the refractive index (1.05–1.95 at 400 nm) Figure 13(d).
128
(a) absorbance against wavelength, (b) α versus hv for pristine PVA and PVA/V2O5/BaTiO3 film (c) optical conductivity of pristine PVA and PVA/V2O5/BaTiO3 films (d) refractive indices versus wavelength. Reproduced with permission.
128
M. T. Ramesan et al. used in situ polymerization to create composites of poly(vinyl cinnamonate) with varying concentrations of NiO nanoparticles (0, 3, 7, 10, 15 weight percent). The DC conductivity of nanocomposites was significantly higher than that of pure PVC, reaching its maximum at 10 weight percent of the composite. After that, the conductivity was shown to decline. 129
Recently, nanomaterials declared garnered extensive attention due to their extensive-ranging purposes and innovative potential. The formation of nanocomposites, however, often faces the challenge of nanofiller agglomeration, a result of their high surface energies. Achieving a uniform dispersion of nanofillers within a polymer matrix is crucial for enhancing the properties of nanocomposites, and PVA frequently plays a key role in this process. A study 124 examining multi-metal polymer doping demonstrated that mixture of Zinc, Magnesium, as well Cadmium could form doped PbS/PVA freestanding nanocomposite films. The preparation process involved creating an initial Pb2+/PVA solution using Pb(NO3)2 and PVA in deionized water. Various concentrations of metal ions, including Zn(NO3)2, MgSO4, and Cd(NO3)2, were then added directly to this solution. The final step involved adding Na2S to form the metal-doped PbS/PVA nanocomposite, which was subsequently cast, dried, and peeled off to produce 80 µm thick freestanding films.
Analysis of the absorbance spectra for PbS/PVA, Zn-PbS/PVA, Mg-PbS/PVA, and Cd-PbS/PVA revealed that the calculated optical band gap (initially 2.44 eV for PbS/PVA) enhanced with higher dopant molar ratios across various obtained materials. This trend indicates that the dopants effectively modulate the electronic structure of the nanocomposites. The nonlinear refraction and all-optical limiting performance of these films are determined through the modification of Z-scan approach, 124 employing a continuous wave (CW) laser at 532 nm wavelength. The results illustrated a modest raise in the limiting threshold, less than a factor of two, between undoped PbS/PVA and Zn-PbS/PVA doped with 0.008 M Zn. Specifically, a limiting threshold of 3 mW was observed, with an optical damage threshold of 21 mW, yielding a high dynamic range of 7. These findings underscore the potential of multi-metal doped PVA nanocomposites in optical limiting applications. The ability to fine-tune optical properties through careful selection and combination of metal dopants opens up new avenues for designing materials with tailored optoelectronic characteristics. Such materials could find applications in areas like optical sensors, photonic devices, and advanced display technologies. Similarly, cerium oxide (CeO2) nanoparticles in PVA films exhibit potential for use in filters and solar cells due to their unique optical absorption characteristics. 130 Metal oxide nanofillers like ZnO and CuO have been studied for their impact on the electrical and dielectric properties of PVA composites. 131 These modifications make the materials suitable for microelectronics and microwave applications. Novel 2D materials like MXenes have also been incorporated into PVA matrices, resulting in composites with interesting optical modulation properties. 131 These materials show promise for applications in broadband and ultrafast optics.
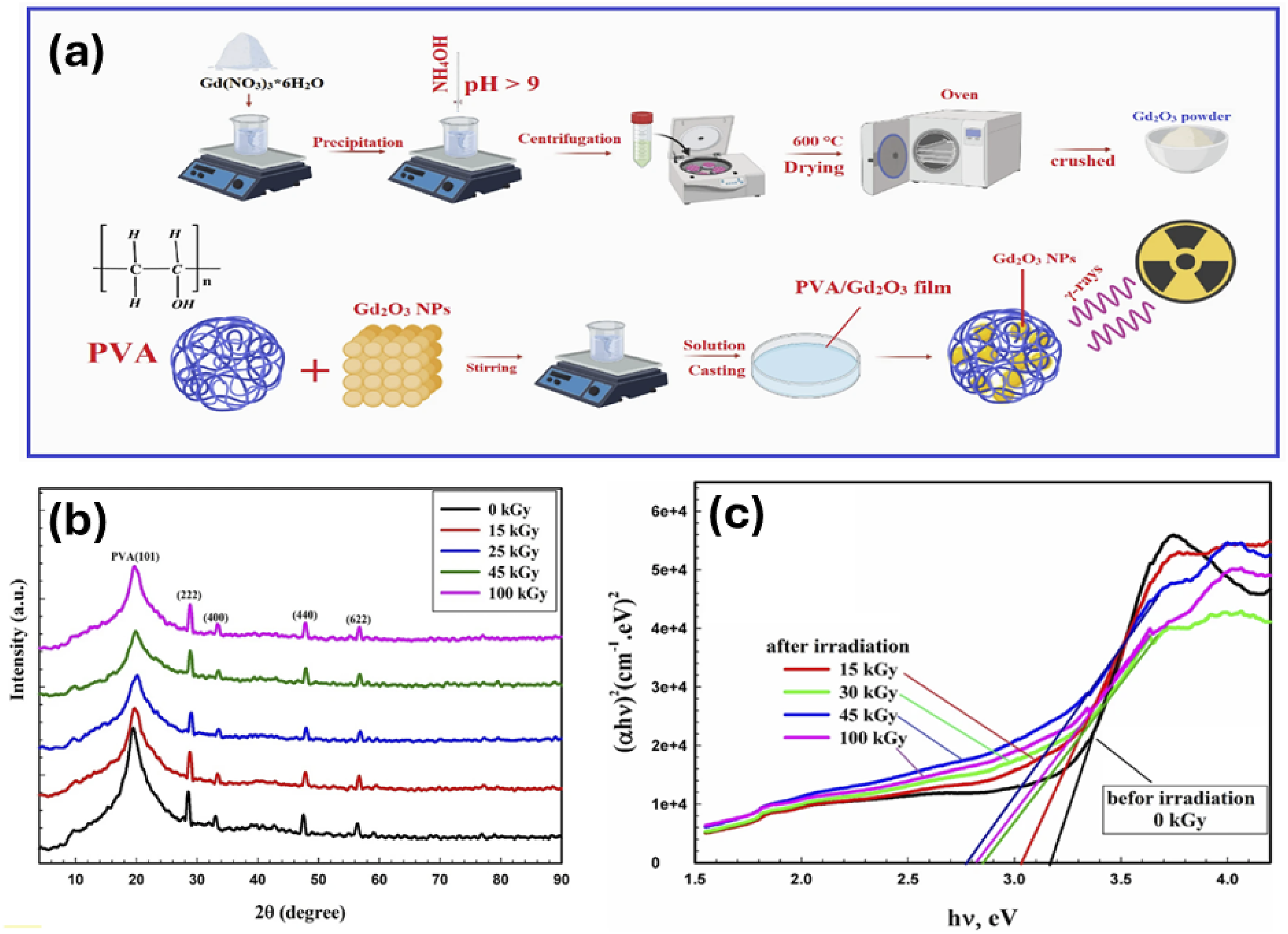
On another subject, Abd El Maksoud et al., explored the influences of gamma rays on the different characteristics of PVA/Gd2O3 nanocomposite films. The samples were achieved the assistance of the solution casting method. Also, the authors investigate the structural, thermal, and optical characteristics for the obtained films after irradiation process at 0, 15, 30, 45 and 100 kGy from gamma rays. Figure 14 exhibits the schematic diagram for the preparation technique, the obtained diffractograms for PVA/Gd2O3 nanocomposite films, and the optical energy gaps for the irradiated films according to the different doses. It was found that the energy gaps reduced with the increase, especially from 15 to 45 kGy while dramatically increase in the Eg value was seen at 100 kGy. Also, the obtained Urbach’s energy from 3.65 eV to 6.9 eV for the un-irradiated and 45 kGy films, respectively. According to the results obtained, it was recommended that the irradiated PVA/Gd2O3 nanocomposite films employ these films for the flexible optoelectronics.
119
The results proved that gamma irradiation plays a significant role on PVA matrix modifications which are seen throughout the structural and optical properties of the PVA/Gd2O3 films. The chosen doses were perfect to modulate the energy gaps of the films which proves the applicability of films for optical applications. Illustrates, (a) the synthesis procedure for PVA/Gd2O3 nanocomposite films through casting techniques, (b) the XRD diffractograms for PVA/Gd2O3 nanocomposite films exposed to different doses, (C) the energy gap values illustration for PVA/Gd2O3 nanocomposite films at different doses. Reproduced by permission.
119
Practical implementations of PVA-based composites in optoelectronic devices
Polyvinyl alcohol (PVA) composites have shown significant promise in various optoelectronic applications due to their unique optical, dielectric, and structural properties. Recent advancements in the development of PVA-based composites doped with materials such as TiO2, WO3, and Co3O4 have demonstrated enhanced performance characteristics suitable for real-world applications.
One notable application is in the fabrication of flexible optoelectronic devices. For instance, PVA/rGO (reduced graphene oxide) composites have been utilized to create flexible films with improved UV-blocking capabilities, making them suitable for UV filters and laser hindering filters. 132
These composites exhibit enhanced transport mobility and electrical conductivity, which are critical for the development of smart optoelectronic devices.
Additionally, PVA/MnO2 (manganese dioxide) composites have been produced through cost-effective methods, showing tunable optical properties that can be leveraged in multifunctional optoelectronic fields 126
The reduction in optical bandgap and improved dielectric properties of these composites make them ideal for applications such as eco-friendly packaging, fuel cell membranes, and photonic devices. Moreover, hybrid nanostructures of titanium carbide (TiC) and silicon carbide (SiC) doped PVA have been fabricated, demonstrating promising optical, structural, and dielectric properties for various optoelectronic applications. 12
These nanocomposites offer potential for use in advanced optoelectronic devices due to their flexibility and enhanced performance metrics.
In summary, the practical implementations of PVA-based composites in optoelectronic devices are vast and varied. The advancements in their optical and dielectric properties, coupled with their flexibility and environmental friendliness, position them as valuable materials for future technological applications.
Some contributions to engineering
Electrical properties
PVA/Cadmium sulphide (CdS) nanocomposite films containing 0–3% CdS nanofiller have been produced. The effective interaction between PVA and CdS enhanced dielectric properties, as evidenced by uniform distribution of the nanofiller within PVA, making these nanocomposite films suitable candidates for electrical charge storage devices. 133
Researchers prepared and studied graphene oxide (GO)/PVA/H3PO4/H2O gel electrolyte. Due to its excellent electrochemical and mechanical properties, this material represents an ideal choice for flexible energy storage devices and wearable electronic applications. 134
Nanocomposite films based on PVA and reduced graphene oxide (RGO) were prepared. The dielectric properties of PVA/RGO nanocomposite films were investigated in the frequency range of 100–25 Hz, showing increases in dielectric permittivity and alternating current conductivity as RGO content increased. 135
Nano-silver particles (nAgs) were used to prepare PVA/nAg composite films, and their electrical conductivity was investigated. Different shapes and sizes of nAg were obtained by altering the concentration of sodium borohydride solution. Depending on the shape and size of nAg, colloidal solutions displayed different colours. High-resolution transmission electron microscopy confirmed the spherical and triangular shapes of yellow and blue nAg respectively. The blue nAgs, due to their uniform distribution in PVA, demonstrated better charge carrier generation and enhanced surface plasmon resonance, exhibiting superior electrical conductivity compared to yellow nAgs. 136
PVA/CaAl2ZnO5 nanocomposites were prepared through solvent intercalation. Studies of the optical, cyclic voltammogram, and electrical properties indicate that these nanocomposites are promising materials for energy storage and electrical applications. 137
A functional thin film of PVA/strontium titanate (PVA/SrTiO3) hybrid nanocomposite was placed between silver (Ag) electrodes for random access memory applications. Flexible resistive switching demonstrated excellent bipolar resistive switching behavior when subjected to both electrical and medical endurance tests. 138
The inertness of graphite (Gr) when used as a nanofiller was enhanced through a Diels-Alder reaction with acrylic acid (AA). Analyses of PVA/AA@Gr nanocomposite revealed excellent thermal stability, potentially expanding opportunities for enhancing thermal conductivity in flexible electronics applications with excellent combined properties. 139
Other properties
Folic acid (FA) has been utilized as a biosafe and biodegradable material for surface modification of zinc oxide (ZnO) nanoparticles. The PVA/ZnO/FA nanocomposite film is highly recommended for food packaging and textile industry applications. It demonstrated good resistance against Escherichia coli and Staphylococcus aureus. 140
A PVA-based active food packaging product with nano-silver (nAg) was developed. Ginger rhizome extract (g) was employed for in-situ reduction of silver nitrate (AgNO3) to nAg (gnAg) within the PVA matrix. The presence of gnAg nanofiller in the PVA/gnAg film provided ultraviolet and light barrier properties with antibacterial activity against foodborne pathogens Salmonella Typhimurium and Staphylococcus aureus. Such films show promise for food packaging applications. 141
Study results rank PVA/zinc peroxide (ZnO2) and PVA/carbon dot zinc peroxide (CZnO2) as candidate adsorbent products for wastewater management, desalination, and catalytic applications. 142
A novel fabrication of durable, fluorine-free, and self-healing superhydrophobic cotton textiles using boric acid as a cross-linker for silica nanoparticles and PVA, followed by polydimethylsiloxane modification through grafting, is reported. This simple fabrication technique shows promise for large-scale superhydrophobic textile manufacturing applicable to accidental oil spills in oceans and oily industrial wastewater. 143
The double polyblend PVA/poly (ethylene glycol) (PEG) networks are hydrophilic smart materials exhibiting shape memory phenomena through thermal stimuli-responsiveness. Researchers believe that the concept of three-dimensional/four-dimensional (3D/4D) printing through double-network structures warrants further exploration in additive manufacturing for shape memory hydrogels across various technological fields. 144
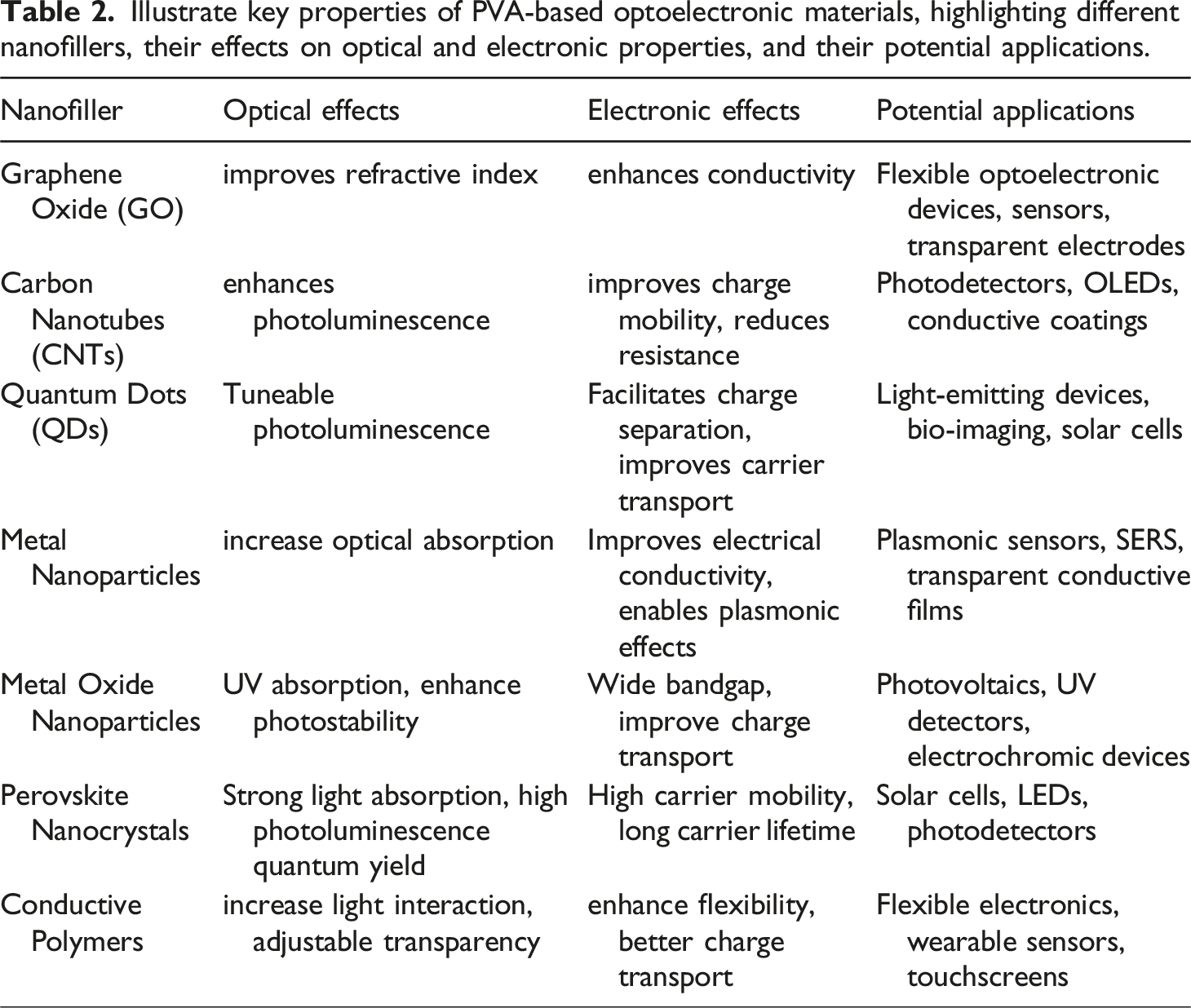
Illustrate key properties of PVA-based optoelectronic materials, highlighting different nanofillers, their effects on optical and electronic properties, and their potential applications.
Key challenges for using PVA
The key challenges and the gaps for the current studies for using such materials to to provide a comprehensive understanding of the field.
One significant challenge is the limited understanding of the long-term stability and durability of PVA-based composites under various environmental conditions. Studies have shown that factors such as humidity, temperature fluctuations, and UV exposure can affect the mechanical and optical properties of these composites. 146 Further research is needed to develop strategies to enhance the stability and longevity of PVA-based materials in real-world applications.
Another gap in current research is the insufficient exploration of the interactions between PVA and various nanoparticles. While the incorporation of nanoparticles like MnO2 and V2O5 has shown promising results, the mechanisms underlying these interactions are not fully understood 8
Detailed studies on the dispersion, compatibility, and bonding of nanoparticles within the PVA matrix are crucial for optimizing the performance of these composites.
Additionally, there is a need for more research on the biocompatibility and toxicity of PVA-based composites, especially for biomedical applications. Although PVA is generally considered biocompatible, the addition of various fillers and nanoparticles may introduce cytotoxicity or immunogenicity concerns. Comprehensive in vitro and in vivo studies are required to ensure the safety of these materials for medical use. 147
Lastly, the scalability and cost-effectiveness of producing PVA-based composites remain significant challenges. Current synthesis methods may not be economically viable for large-scale production, limiting the practical implementation of these materials. 148
Research should focus on developing efficient and cost-effective manufacturing processes to facilitate the commercialization of PVA-based composites.
Future prospects: PVA and advanced materials in optoelectronics
The future of optoelectronics utilizing PVA, and advanced materials presents exciting opportunities for research and development. Several key areas warrant further investigation: 1. Multi-functional nanocomposites: Developing novel PVA-based composites that incorporate multiple types of nanofillers (e.g., metal nanoparticles, quantum dots, and 2D materials) to achieve synergistic effects and multi-functional optoelectronic properties. 2. Tailored bandgap engineering: Exploring new combinations of dopants and nanofillers to precisely control the bandgap of PVA composites, enabling the creation of materials with customized optical and electronic properties for specific applications. 3. Enhanced nonlinear optical materials: Investigating PVA composites with improved nonlinear optical responses for applications in optical limiting, frequency conversion, and all-optical switching devices. 4. Optoelectronic structures: Drawing inspiration from natural light-harvesting systems to design PVA-based materials with hierarchical structures for improved light absorption and energy transfer in photovoltaic and photonic applications. 5. Flexible and wearable optoelectronics: Leveraging PVA’s flexibility to develop stretchable and conformable optoelectronic devices for applications in wearable technology, healthcare monitoring, and human-machine interfaces. 6. Self-healing optoelectronic materials: Exploring the potential of PVA’s reversible hydrogen bonding to create self-healing optoelectronic materials that can recover from mechanical damage and maintain long-term performance. 7. Sustainable manufacturing processes: Developing eco-friendly synthesis methods and exploring the use of bio-based fillers to create more environmentally sustainable PVA composites for optoelectronic applications. 8. Advanced characterization techniques: Utilizing cutting-edge analytical tools, such as in-situ spectroscopy and high-resolution microscopy, to gain deeper insights into the structure-property relationships of PVA-based optoelectronic materials. 9. Computational materials design: Employing machine learning and artificial intelligence algorithms to predict and optimize the properties of PVA composites, accelerating the discovery of new material combinations for optoelectronic applications. 10. Integration with emerging technologies: Exploring the potential of PVA-based materials in emerging fields such as neuromorphic computing, quantum optics, and terahertz technology.
As research progresses in these areas, we anticipate significant advancements in PVA-based optoelectronic materials and devices. These developments are poised to drive innovations in energy harvesting, information display, optical communication, and sensing technologies, ultimately contributing to more efficient, sustainable, and versatile optoelectronic systems.
Conclusion
This review has highlighted the significant role of polyvinyl alcohol (PVA) and advanced materials in shaping the landscape of modern optoelectronics. PVA’s exceptional optical clarity, flexibility, and compatibility with various fillers position it as a versatile matrix for optoelectronic applications. We have explored how the incorporation of metal ions, nanoparticles, and other polymers can dramatically alter PVA’s properties, enabling the creation of materials with tailored optical and electronic characteristics.
The investigation of metal-doped PVA composites, including those with transition metals, rare earth elements, and metal oxides, has revealed complex interactions that significantly influence optical absorption, refractive index, and electrical conductivity. These modifications open new possibilities for applications in holography, integrated optics, and photonic devices.
Nanocomposite formation, particularly with metal and metal oxide nanoparticles, has emerged as a promising approach to enhance PVA’s optoelectronic properties. The incorporation of nanofillers like TiO2, MnO2, and multi-metal doped nanoparticles has demonstrated the potential to fine-tune bandgap energies, improve optical conductivity, and enhance nonlinear optical responses.
Despite these advancements, challenges remain in achieving uniform dispersion of nanofillers, optimizing interfacial interactions, and ensuring long-term stability of the composites. As research progresses, interdisciplinary collaborations combining materials science, physics, and engineering will be crucial in overcoming these hurdles and realizing the full potential of PVA-based materials in next-generation optoelectronic devices.
The versatility of PVA as a host matrix for various dopants and fillers, coupled with emerging nanomaterials and advanced fabrication techniques, positions PVA-based composites at the forefront of optoelectronic material development. These materials hold promise for revolutionizing applications ranging from flexible displays and smart windows to high-efficiency solar cells and advanced optical sensors. Compliance with ethical standards.
Footnotes
Acknowledgment
The authors extend their appreciation to the Dean ship of Scientific Research at the Northern Border University, Arar, KSA, for funding this research work through the project number NBU-FFR-2025-885-03.
Author contributions
Amani Alruwaili: Funding resources, Validation, Software, review & editing. Hadeer Gamal: measurements, Software, Validation, Writing - review & editing. Mohammed Jameel Alawi: Funding sources, measurements, Software, Mustafa Shalaby: Conceptualization, Methodology, Software, Data curation, Project administration, Writing - original draft, Writing - review & editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
