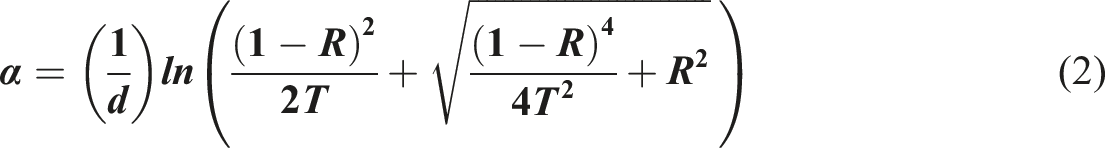Abstract
Nanocomposite films composed of polyvinyl butyral (PVB) and Bi2WO6 were produced through solution casting. The goal of this investigation was to examine the effects of different Bi2WO6 concentrations (0%, 2%, and 4% wt.) on the linear/non-linear optical and optoelectrical properties, as well as the structure and dispersion of films of PVB/Bi2WO6 nanocomposite. The direct band gap Eg1 value falls from 5.1 eV to 3.83 eV with the progressive increase in Bi2WO6 content from 0% to 4% wt., while indirect band gap Eg2 decreased from 4.1 eV to 2.89 eV. Conversely, the PVB + 4% Bi2WO6 nanocomposite increased Urbach’s energy (EU) from 1.00 eV for pure PVB to 1.97 eV. Moreover, our research has documented the impact of different concentrations of Bi2WO6 on a range of optical properties, including the refractive index (n), extinction coefficient (k), and other pertinent parameters. Utilizing the real and imaginary components of the dielectric constants εr and εi, an investigation was carried out into the dielectrics’ behavior and the optoelectrical parameters’ calculation. Furthermore, investigations were performed on the linear optical susceptibility, the non-linear refractive index, and the third-order non-linear optical susceptibility concerning the concentrations of Bi2WO6. In addition, the results indicated that varying Bi2WO6 concentrations substantially affect the oscillator strength, average oscillator wavelength, and optical conductivity. The nanocomposite films of PVB/Bi2WO6 concentrations exhibited favorable associations between their optoelectrical and non-linear/linear optical parameters, rendering them viable candidates for implementation in flexible electronic devices and radiation shielding.
Introduction
Extensive research has been conducted on polyvinyl butyral (PVB) material, which is widely employed owing to its exceptional properties. PVB is valued for its durability, electromagnetic protection, optical clarity, and ease of adhesion to diverse surface characteristics, making it suitable for various applications such as film sheets, paint coatings, and adhesives in automotive, construction, and electronics.1–4 In a recent publication, Lee et al., 5 described the fabrication procedure for a composite consisting of PVB and Ag nanowires. This material possesses remarkable qualities, including exceptional flexibility, mechanical strength, and surface uniformity. The principal aim in developing this system was to improve the effectiveness of light extraction from OLEDs (organic light-emitting diodes). By optimizing the inherent properties of the materials, this novel combination of PVB and silver nanowires intends to accelerate improvements in the functionality and versatility of OLEDs. The emphasis on mechanical robustness, surface smoothness, and flexibility highlights a holistic strategy for augmenting the overall effectiveness of flexible OLED technology.
As a result of their exceptional characteristics, polymer composites have become prominent topics of study in materials science. Composites consist of at least two phases that are physically and chemically identified through a distinct interface. The vast majority of commercially available composites, consisting of a matrix of polymeric substance or resin solution, incorporate various reinforcement materials, such as fibers or common ground minerals. Due to their remarkable amalgamation of characteristics—including strength, deformation resistance, electrical conductivity, and thermal conductivity—these composites are exceedingly favorable for various applications. Because of technological advancements, thermoplastic and thermosetting polymers have been incorporated into the composition of advanced composite materials with extraordinary properties that find application in various industries. Composites possess many unattainable properties with “simple” materials; this attribute renders them extraordinarily valuable in industrial applications and constitutes one of their primary advantages over conventional materials. 6
Furthermore, they are highly prospective candidates for a wide range of purposes, such as healthcare equipment, electronic equipment, detectors, and solar energy generation, because of their ability to alter electrical, optical, and mechanical characteristics. 7 Given the nascent stage of advancement in optical material applications, the development of novel materials possessing improved properties is of the utmost importance. 6 The primary aim of assessing organoelectronic materials is to improve their non-linear and linear optical characteristics, including the refractive index (n), optical band gap (Eg), and third-order non-linear susceptibility.8–10 A more comprehensive understanding of the transition characteristics between the LUMO and the HOMO can be attained through the examination of n and absorption edge. 11 By altering the characteristics, dimensions, and arrangement of the additives within a polymer matrix, it is possible to optimize these parameters. 8 The interrelationships between composites’ organic and inorganic constituents impact their electrochemical properties. Whether polymer chains diffuse into the host structure is a concern is contingent on the chain length and pore dimensions. Nanocomposites demonstrate considerable promise as viable materials for integration into optoelectronic systems. 12
CaIn2O4, 13 Bi4Ti3O12, 14 Bi2MoO6, 15 NaTaO3, 16 and Bi2WO6 17 are some of the substances that have been investigated. Intriguing non-linear dielectric susceptibility, catalytic activity, and piezoelectric properties are bismuth tungstate (Bi2WO6) oxide characteristics that have prompted extensive research. Outstanding in both physical and chemical properties, Bi2WO6 is a member of a significant class of semiconductor oxides exhibiting ferroelectric, 18 piezoelectric, 19 pyroelectric, 20 photoluminescence, 21 non-linear dielectric induction, 22 and photocatalysis23,24 properties among others. Moreover, Bi2WO6 possesses an optimal band gap and exhibits remarkable visible light absorption properties. 25 In their study, Wang et al. 26 utilized a hydrothermal method to produce spherical Bi2WO6 NPs with a band gap of 2.93 eV and an estimated mean particle size of 85 nm.
This study employed the co-precipitation method to produce Bi2WO6 nanoparticles for the first time. These nanoparticles were combined with PVB to form a PVB/Bi2WO6 nanocomposite film. The present investigation outlined PVB/Bi2WO6 nanocomposite film synthesis via solution casting. Then, an investigation was conducted into the structural, dispersion, linear/non-linear optical, and optoelectrical properties of PVB/Bi2WO6 nanocomposite films at variable concentrations of Bi2WO6 (0, 2, and 4% wt.). In order to characterize the PVB/Bi2WO6 nanocomposite films, XRD, SEM, EDX, FT-IR, and UV-VIS-NIR analytical methods were performed. Due to their remarkable properties, the proposed PVB/Bi2WO6 films are exceptionally well-suited for a wide range of purposes in cut-off lasers, organic electronics, and optoelectronics.
Experimental
Materials and methods
One of the constituents utilized in this investigation is PVB, a purified substance manufactured by Sigma-Aldrich, USA. The synthesis of Bi2WO6 was performed by co-precipitation. Bi2WO6 was synthesized by dissolving Bi(NO3)3·5H2O (Sigma–Aldrich) in 0.1 L of diluted HNO3 (10%, v/v) at a temperature of 25°C. Then, while vigorously agitating, the solution was added gradually (5 mL min−1) to 0.1 L of an aqueous solution containing Na2WO4. 2H2O (Aldrich, 99.9%). For gradual water evaporation, the solution was agitated and maintained in a water immersion at 70°C until a white solid, utilized as a precursor, formed. The precursor was decomposed in air using thermal treatments at 200°C to produce Bi2WO6 oxide, as illustrated in Figure 1. Schematic diagram for Bi2WO6 and PVB/Bi2WO6 nanocomposite film synthesis.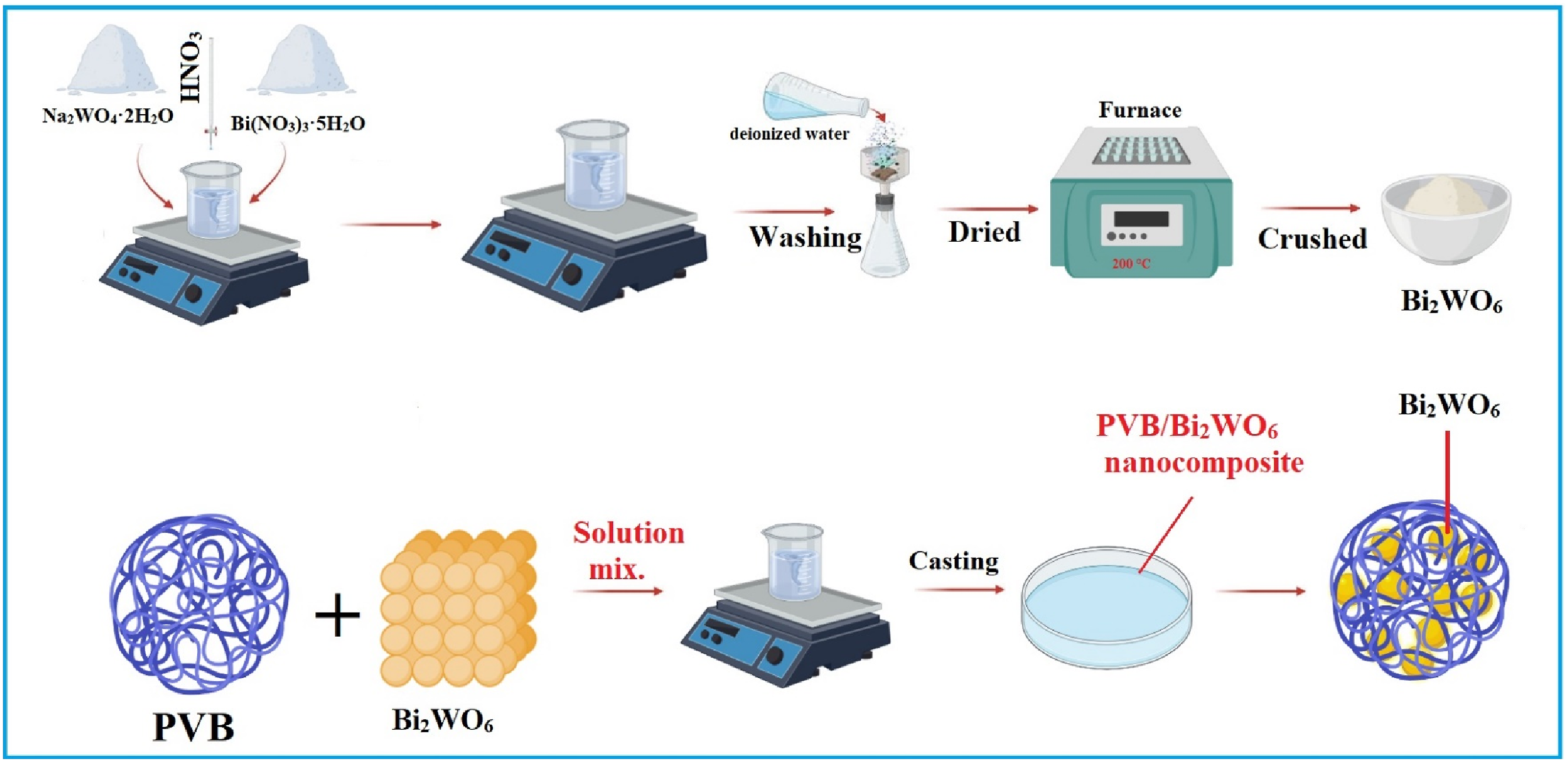
The requisite weight of PVB was dissolved in alcohol to produce a 4% pure PVB polymer solution to fabricate the polymeric films. To ensure solution homogeneity, the solution was magnetically agitated for 30 min at ambient temperature. The prepared PVB solution was then agitated with an ultrasound sonicator for 2 hours while Bi2WO6 powder was dispersed at varying concentrations (0%, 2%, and 4% wt.). Finally, the resultant solutions were applied onto glass substrates via solution casting. In an ambient-temperature environment, the coated layers were dried for 48 h.27,28
Characterization
The PVB/Bi2WO6 nanocomposite films were analyzed using various analytical instruments. The XRD technique was implemented to evaluate the material’s structural characteristics. A Shimadzu 6000 diffractometer was employed and set up in the Bragg–Brentano configuration. The different concentrations of PVB/Bi2WO6 nanocomposite were assessed using an FTIR spectrometer (Nicolet iS10, USA). The measurements were conducted over a spectrum of wavenumbers, specifically from 4000 cm−1 to 400 cm−1. The morphology of the Bi2WO6 samples was examined using a JEOL JTEM-1230 model scanning electron microscope (SEM) equipped with 220 kV. EDX analysis was employed to generate mapping images and elemental composition illustrations. A UV-Visible-NIR spectrophotometer (JASCO V-570) was employed to analyze the optical properties of the films of PVB/Bi2WO6 nanocomposite within 190–1100 nm.
Results and discussion
Structural studies
Bi2WO6 powder’s elemental composition and EDX spectrum are illustrated in Figure 2(a). Bismuth (Bi), tungsten (W), and oxygen (O), in that order, constitute the Bi2WO6 powder’s chemical composition. A Bi2WO6 sample is considered extremely pure and contains no extraneous elements. The SEM micrographs of the particle sample of Bi2WO6 are visually represented in Figure 2(b), where Bi2WO6 particles display a uniform plate-like morphology. (a) EDX, (b) SEM, (c) XRD, and (d) FTIR of Bi2WO6 powder.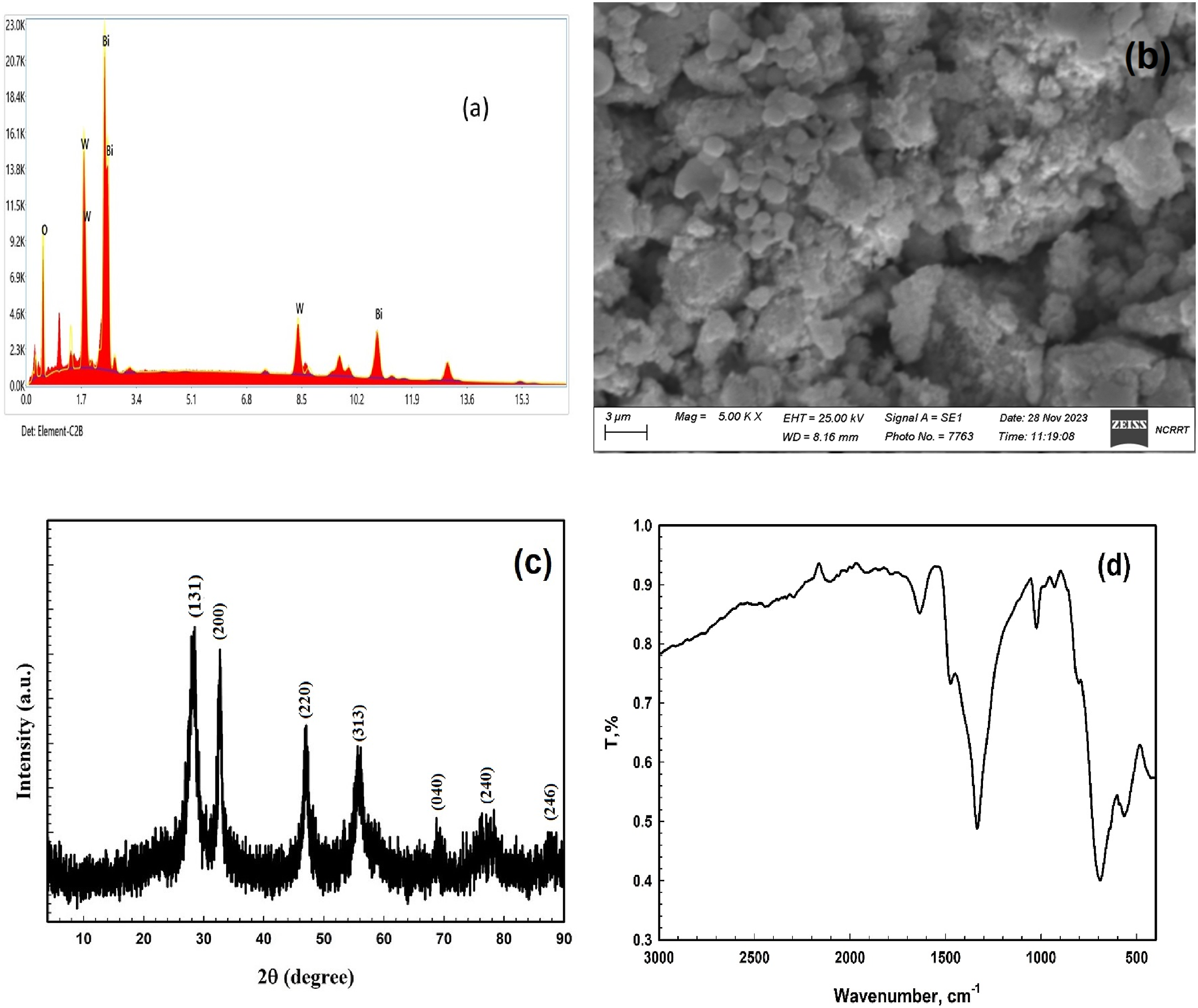
Figure 2(c) depicts the XRD pattern of Bi2WO6 particles. The diffraction peaks with conspicuous characteristic values at 2θ = 28.14°, 32.66°, 46.97°, 55.74°, 68.84°, 78.29°, and 87.51° corresponded to the crystal planes (131), (200), (220), (313), (040), (240), and (246) of the orthorhombic phase of Bi2WO6 NPs, respectively (JCPDS no: 39–0256).29–32
Utilizing the well-known Scherrer equation, the crystallite dimension D of Bi2WO6 powder was calculated:
33
0.15406 nm represents the Cu Kα radiation (λ) wavelength utilized, β represents the full width at half maximum of the diffraction peak, whereas θ corresponds to the Bragg angle. The D value of Bi2WO6 material was determined to be 6.43 nm. There are numerous possible explanations for the variation between the particle diameters determined by XRD and SEM such as nature of analysis. Moreover, variations in the sample preparation and calculation methodologies employed for estimating the particle size via XRD and SEM may manifest differing particle diameters. The size of Bi2WO6 in PVB/Bi2WO6 nanocomposites significantly influences their optical and optoelectrical characteristics. Comprehending and managing these effects that are influenced by size is crucial for enhancing the efficiency of nanocomposites in diverse applications such as photocatalytic degradation, solar cells, and optoelectronics.34–36
The functional groups of Bi2WO6 NPs were revealed by plotting the FT-IR spectrum. Figure 2(d) illustrates that the Bi2WO6 sample exhibits conspicuous absorption bands spanning the wavelength range of 400–800 cm−1. These bands have been attributed to the bridging stretching for (W–O–W), (Bi-O), and (W–O) stretching modes. Furthermore, it was observed that the band at 1333 cm−1 corresponded to the coupling of O–H, whereas the band at 1636 cm−1 was correlated with the undulating resonance of C–H.37,38
The XRD patterns of films composed of PVB/Bi2WO6 nanocomposites are illustrated in Figure 3(a). Significantly, a broad diffraction peak from 2θ of 5° to 25° indicates the development of PVB’s amorphous structure.39,40 Moreover, as the Bi2WO6 was introduced, the conspicuous diffraction peaks of PVB underwent a displacement to a reduced angle and enlargement due to the interaction between the two substances. Furthermore, the contribution of Bi2WO6 leads to a conspicuous amplification in both the peak intensity and sharpness of Bi2WO6. (a) XRD patterns and (b) FTIR spectra of PVB/Bi2WO6 nanocomposite films.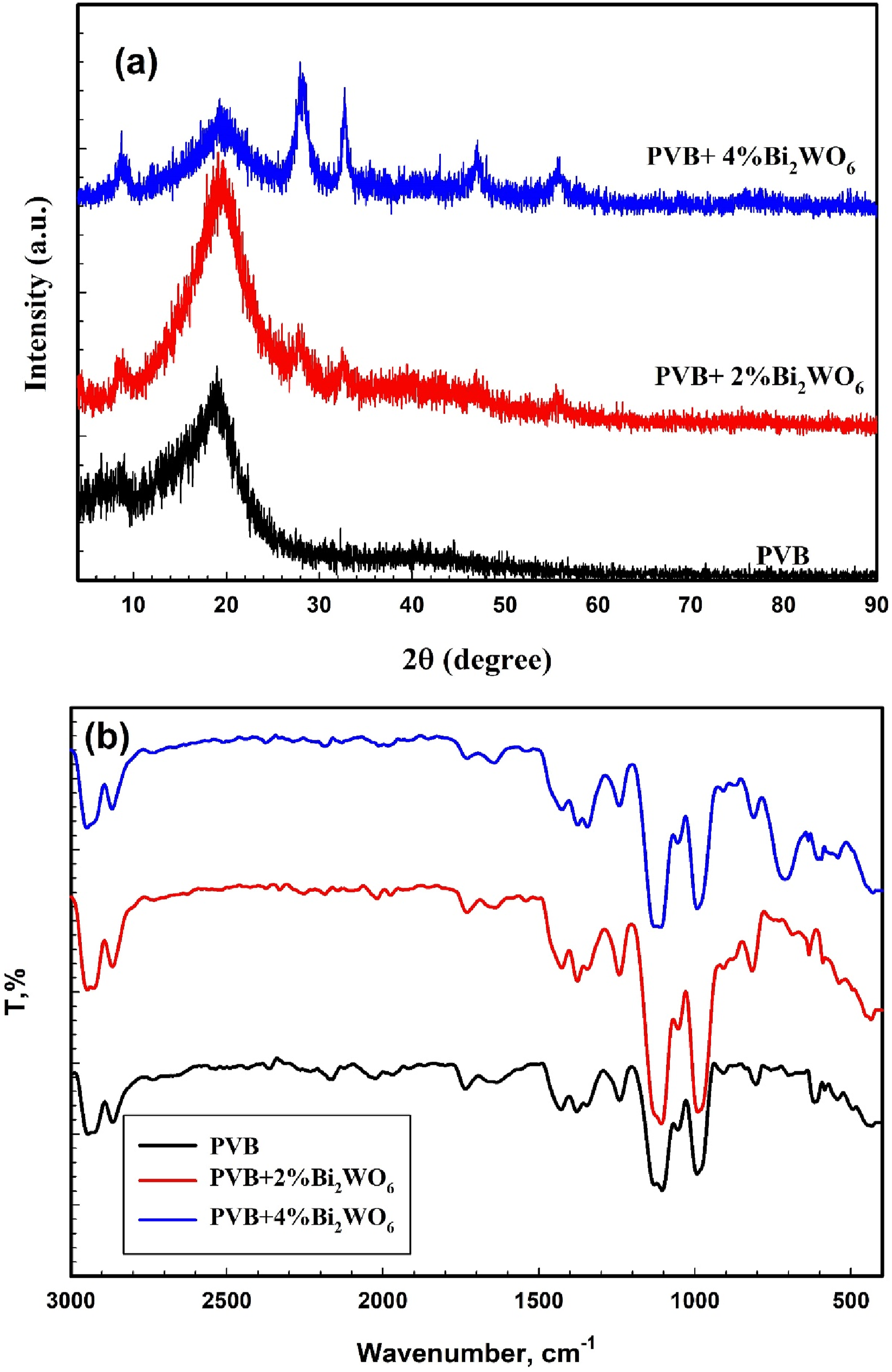
FTIR spectra of pure PVB films and films containing Bi2WO6 are illustrated in Figure 3(b). The induced modifications were evaluated by analyzing the band intensity variations corresponding to each functional group. An examination of the FT-IR spectra validated the existence of a butyral ring (C-O-C) within the pure PVB. O-H stretching is correlated with the band identified at 3476 cm−1. Aliphatic C-H stretching is indicated by the bands detected at 2865 and 2945 cm−1. The C = O carbonyl group is encoded by the band detected at 1736 cm−1. The inclinations of CH2 and CH3, respectively, are indicated at 1429 and 1378 cm−1. 1105 cm−1 is the FTIR band that has been ascribed to C-O-C stretching. In addition, a band at 993 cm−1 is detected; this band is indicative of the C-O bond undergoing elongation.41,42
Linear optical characteristics of films of PVB/Bi2WO6 nanocomposite
Transmission’s (T) spectral characteristics
Examining the optical properties of materials and ascertaining their optical constants are critical steps in the assembly of optoelectronic equipment. The advancement of optoelectronic devices is heavily reliant on comprehending materials’ electrical and band structures. Primarily concerned with semiconductors, optoelectronics is founded upon the quantum mechanical consequences of light on electronic substances. In determining the precision and efficiency of these devices, the optical and electronic characteristics of the active substances are critical. Sustaining materials with favorable dielectric and optical features is imperative for advancing optoelectronic devices. For the development of optoelectronic devices, comprehension of the optical properties of substances is therefore crucial.
The transmittance, denoted as T (λ), and reflectance (R) of the PVB/Bi2WO6 nanocomposite films is illustrated in Figure 4(a) and (b) for a spectrum of wavelengths spanning from 190 to 1100 nm. The PVB/Bi2WO6 nanocomposite films also exhibited a band at approximately 275 nm. The bands that have been observed are indicative of the electronic transitions of the host polymer n→π*.43,44 The modifications in transmittance T (λ) that occurred due to the augmentation of Bi2WO6 in the PVB matrix are evident from the data depicted in Figure 4(a). The PVB/Bi2WO6 nanocomposite films’ (a) transmittance and (b) reflectance.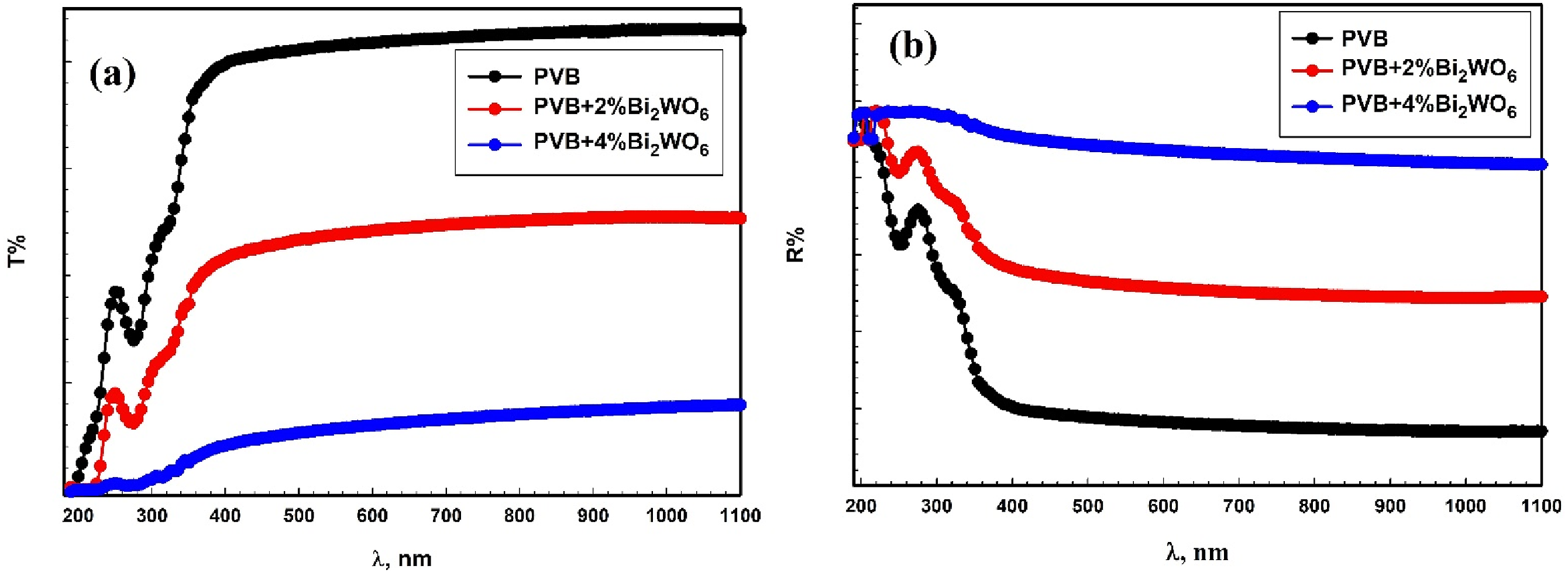
Furthermore, an increase in the contents of Bi2WO6 in PVB leads to a redshift of transmittance toward longer wavelengths. A decline in the optical bandgap is consistent with the observed shift in transmittance. The prospective applications of the PVB/Bi2WO6 nanocomposite films in optical devices are indicated by the broad spectrum of shifts in transmittance and cut-off edges observed.44–46
Promoting consistent distribution of Bi2WO6 throughout the PVB matrix is essential for improving the characteristics of the PVB/Bi2WO6 nanocomposite films. The porosity levels can impact the density of the PVB/Bi2WO6 nanocomposite films, which in turn affects optical qualities like transmittance. 47 The decrease in the ability of PVB/Bi2WO6 nanocomposite films to transmit light makes them well-suited for use as materials that block ultraviolet (UV) radiation. These films are beneficial for protecting sensitive components like electronic devices. By reducing transmittance, these films enhance contrast and reduce glare.48,49 Figure S1 shows the pictures of all PVB/Bi2WO6 nanocomposite films under normal light and UV lamp.
Furthermore, these nanocomposites’ reflectance value (R%) consistently decreases with the incorporation of Bi2WO6 nanoparticles into PVB. Light scattering is a function of both the film’s surface roughness of PVB/Bi2WO6 nanocomposite and the interfacial barriers between it and the air, both of which tend to rise as porosity develops. It is critical to optimize particle size and refractive index compatibility in order to enhance the optical characteristics of PVB/Bi2WO6 nanocomposite films with high refractive index.50–52
Urbach tail and optical energy gap
When considering solid materials for instruments that utilize optoelectronics and the development of innovative solar cells, evaluating the energy gap, Eg, is a critical parameter. By employing measured reflectance (R) and transmittance (T) values in the 2500–190 nm range for nanocomposite films of PVB/Bi2WO6, an approximation of the absorption coefficient (α) is feasible. Employing the subsequent equation, one can execute this evaluation:
53
Based on absorbance A(λ) and transmittance T(λ) measurements, the linear and non-linear optical properties of the nanocomposite films of PVB/Bi2WO6 were determined. Tauc’s relationship was employed to evaluate the indirect and direct optical bandgaps (Eg) of the PVB/Bi2WO6 nanocomposite films.
54
As previously stated, the equation incorporates three variables: the optical absorption, denoted as α; the energy of the incident photon, denoted as hυ; and a constant, designated as B. The t parameter indicates the type of electronic transition observed in the nanocomposite layers of PVB and Bi2WO6. Acceptable indirect transitions are denoted by a value of 2 for t, whereas allowed direct transitions are designated by 1/2 for t. To transfer momentum to the crystal lattice, the electron must pass through an intermediate state to emanate a photon through an indirect gap. Conversely, an electron can undergo transition directly to the conduction band in a direct gap through the absorption of a photon. The occurrence of associated transitions at distinct energies can give rise to direct and indirect band gaps in a given material.
Figure 5 calculated the direct band gap (Eg1) and the indirect band gap (Eg2) of PVB/Bi2WO6 nanocomposite films by extrapolating the line segment until the zero y-axis absorption. The results of the present investigation regarding Eg1 and Eg2 are presented in Table 1. These results indicated a noticeable pattern in the characteristics of Eg. More precisely, it is noted that the Eg1 value falls from 5.1 eV to 3.83 eV with the progressive increase in Bi2WO6 content from 0% to 4%wt, while Eg2 decreased from 4.1 eV to 2.89 eV. The magnitude of this impact is substantial enough to induce localized states at the band boundaries of the HOMO and LUMO, thereby impeding the availability of energy transitions.55,56 Accordingly, the energy gap is substantially reduced in the present case due to the enhanced conjugation between PVB’s unsaturated bonds and Bi2WO6 NPs.57–59 Relation between (a) The optical parameters of PVB/Bi2WO6 nanocomposite films.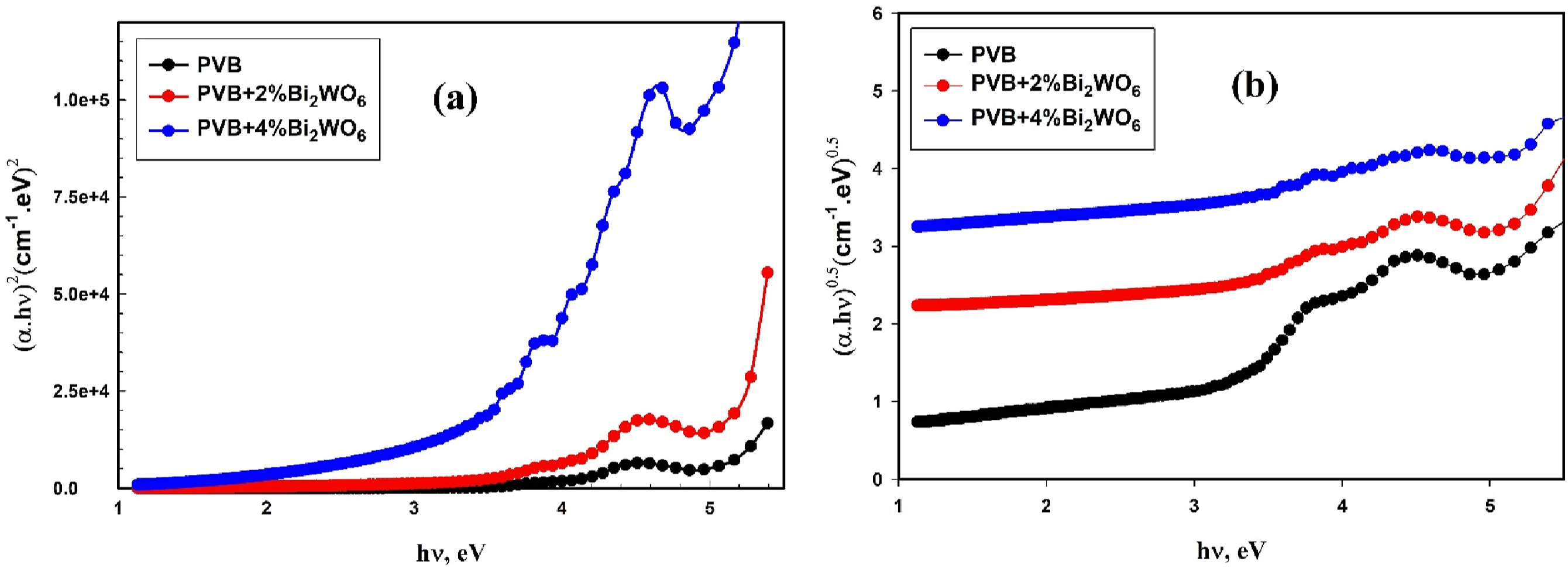
The absorption coefficient curve displays an exponential segment called the Urbach tail close to the optical band edge. Urbach’s equation may be applied to the PVB/Bi2WO6 nanocomposite films, as indicated by their exponential dependence on photon energy (hυ).
By focusing on the Urbach energy (EU), the current study examined the defects produced in PVB/Bi2WO6 nanocomposite films due to the increase in Bi2WO6 content in PVB:
60

Figure 6 illustrates this dependence by plotting ln α against the photon energy hυ for the PVB/Bi2WO6 nanocomposite films under consideration. Computed and enumerated values of Urbach’s energy (EU) are presented in Table 1. The data indicates that the value of EU for PVB + x% Bi2WO6 nanocomposite rises from 1.00 eV for pure PVB to 1.97 eV for PVB + 4%Bi2WO6 nanocomposite. This increase in value may be ascribed to an upsurge in the traps present in the PVB/Bi2WO6 nanocomposite layers, resulting in reduced efficient energy transitions and a consequent decline in Eg.61,62 ln (α) versus photon energy for PVB/Bi2WO6 nanocomposite films.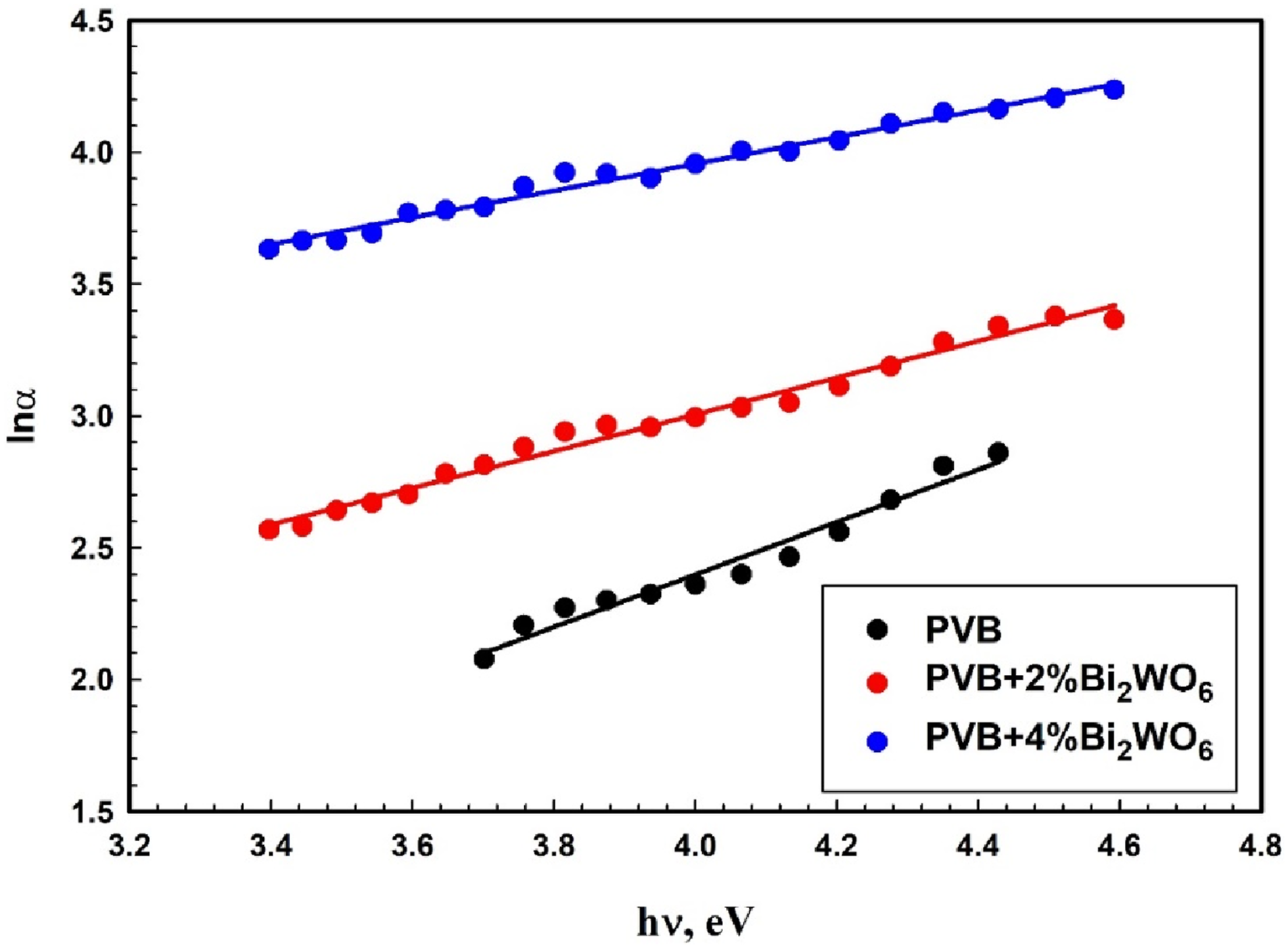
Refractive index and extinction coefficient
A fundamental parameter in optical physics, the refractive index (n) quantifies the electronic polarization of ions within a given substance. The velocity at which light propagates through a medium other than a vacuum is a critical parameter in optics. In the opposite direction, the value of n influences the energy, wavelength, polarization state, and direction of propagation of a light ray. It is considered an indicator of the light-bending capability of an optical medium; n is a dimensionless number. It has significantly advanced the field of communication via optical fibers and spectral dispersion instruments. 63
We obtained k and n by referring to the sources mentioned in ref:53,64–68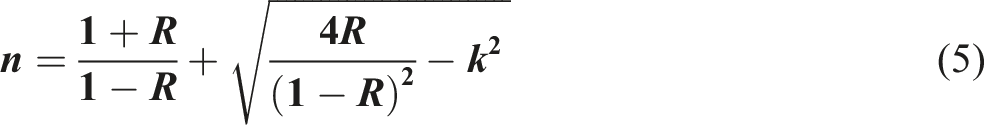

The variation in n for all PVB/Bi2WO6 nanocomposite samples across various sets of λ is depicted in Figure 7(a). Internal modifications to the structure and interatomic forces were responsible for the progressive increase in the value of n with λ, and the concentration of Bi2WO6 in PVB was reflected in the dispersion relation.39,69 A significant decrease in interatomic space is indicated by a high n value, which signifies the film’s dense nature. The reflectivity of PVB is enhanced by the scattering effect of the Bi2WO6 particles inserted within it. Light-emitting diodes, waveguides, and anti-reflective coatings are a few examples of cutting-edge advanced optoelectronic devices that may employ PVB/Bi2WO6 nanocomposites with high n values.
69
(a) Refraction index (n); and (b) k concerning wavelength (λ) for PVB/Bi2WO6 nanocomposite films.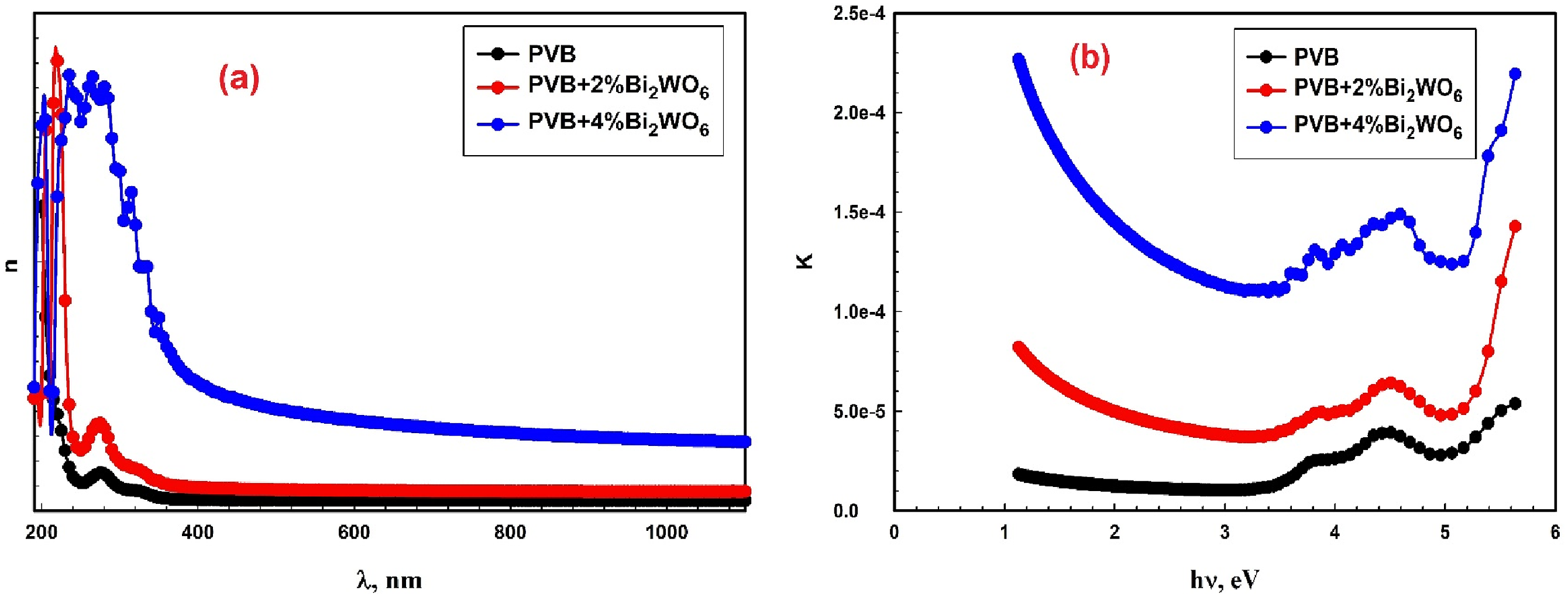
The improvement in the k value of PVB/Bi2WO6 nanocomposite films, as depicted in Figure 7(b), may be attributed to a decrease in the Eg of the PVB/Bi2WO6 nanocomposite films once the Bi2WO6 content is transformed into PVB.
Dielectric and dispersion constants
By implementing the single oscillator model, the Wemple and DiDomenico formula might be used to calculate the dispersion energy (Ed) and oscillator energy (Eo) of the films of PVB/Bi2WO6 nanocomposite.
70
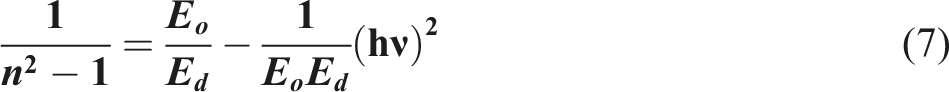
The definition of Eo is associated with the mean excitation energy necessary to induce electronic transitions. On the contrary, Ed is strongly associated with the intensity of interband transitions in the optical spectrum, as evidenced by numerous characteristics intrinsic to the ordered structured material in conjunction with the effective oscillator energy. Eo is a measurable parameter that characterizes the material’s reaction to external stimuli by representing the energy necessary for electronic transitions. Conversely, Ed provides a more intricate aspect to the comprehension of interband transitions by revealing how the structural properties of a material affect its optical characteristics; thus, it enhances the understanding of electronic behaviors in various materials.
64
As shown in Figure 8 and presented in Table 2, the Eo and Ed of films of PVB/Bi2WO6 nanocomposite were measured. Ed, which varies with the Bi2WO6 content, may be estimated by examining the slope of the lines that fit the data on a plot of 1/(n2-1) versus (hυ)2 and the intersection. (n2-1)−1 against (hν)2 graphs of PVB/Bi2WO6 nanocomposite films. Values of dielectric constants and dispersion parameters for films of PVB/Bi2WO6 nanocomposite.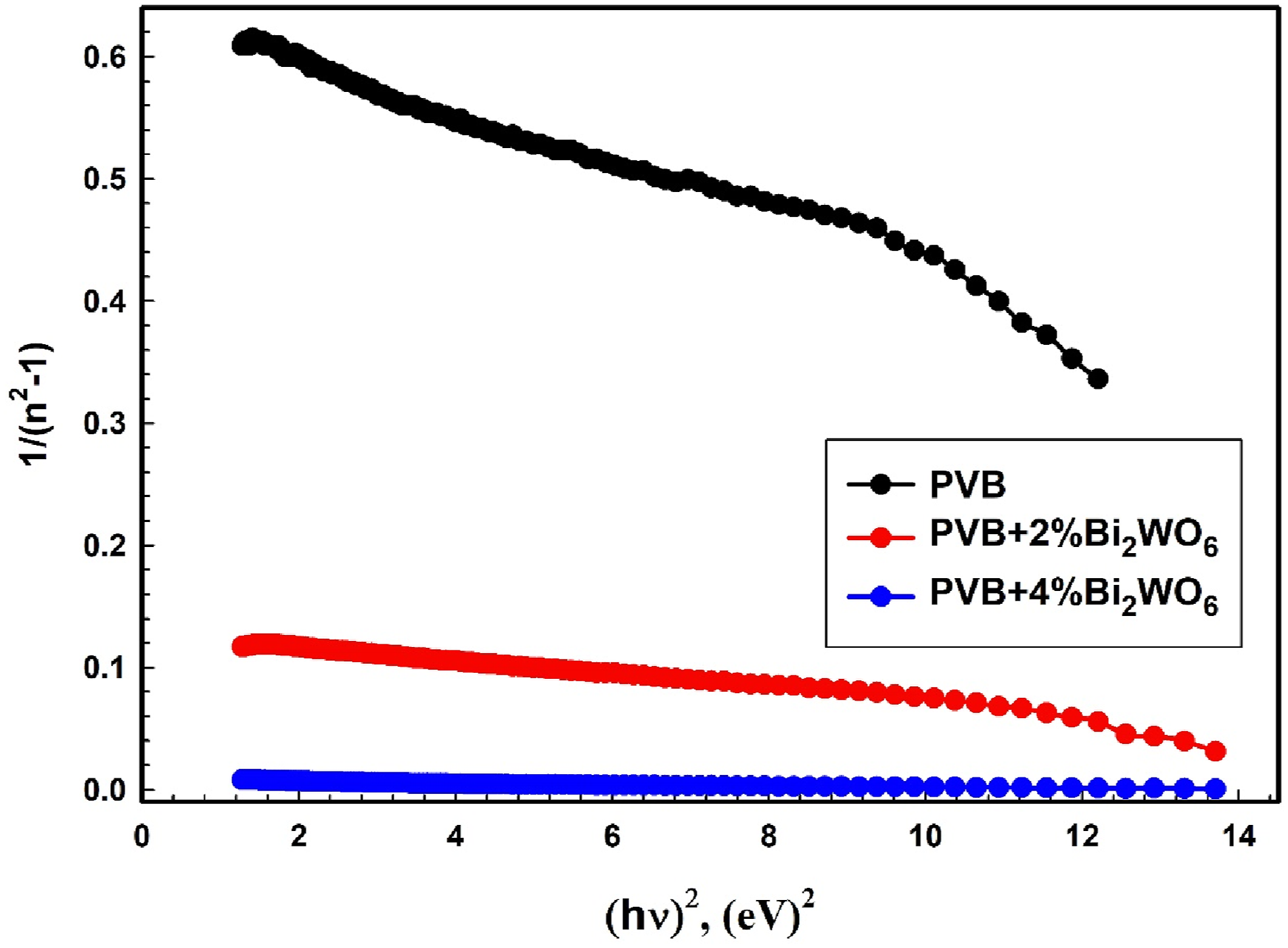
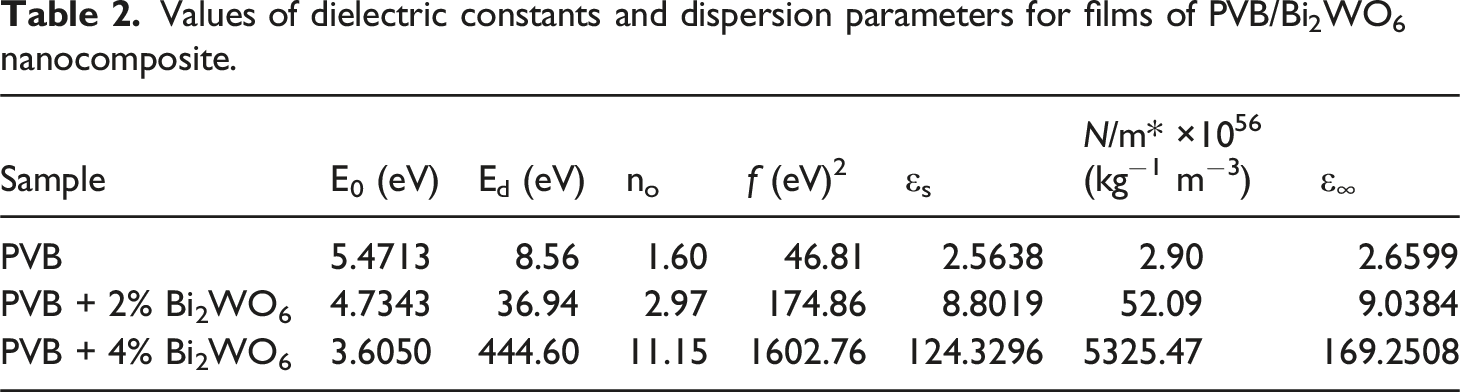
The subsequent equation was utilized to determine the formula for computing the static refractive index no, which is associated with hυ = zero:
64

By employing the no, it is possible to determine the static dielectric constant (εs = no2). 45 Furthermore, Table 2 presents the values of Eo and Ed, and the definition of optical oscillator strengths (f) of optical transitions. These strengths are calculated using the amount of photon energy the electron absorbs as it passes from the initial to final states. The relationship between this photon absorption and E0 and Ed can be expressed as f = EoEd. 64 An increase in the concentration of Bi2WO6 incorporated into the PVB matrix results in a noticeable decrease in oscillator energy and a simultaneous increase in dispersion energy. This phenomenon highlights the complex correlation between the concentration of Bi2WO6 and the unique energetic parameters attributed to the composite material. An increase in the concentration of Bi2WO6 results in a decrease in the oscillator energy, which suggests that the material has a reduced energy demand for electronic transitions. Concurrently, the rise in dispersion energy indicates that the ordered structure of PVB/Bi2WO6 nanocomposite significantly impacts the intensity of interband transitions in the optical spectrum. The complex relationship between fluctuations in concentration and energetic properties highlights the subtle influence of Bi2WO6 content on the electronic behaviors and optical characteristics of the PVB matrix. This generally signifies that the Bi2WO6 NPs modify the electrical configuration of PVB, resulting in the dispersion of defects throughout the PVB matrix.
Determining the relationship between electron transitions across bands in materials and their underlying structural properties is a critical function of the dielectric constant. The dielectric constant, an essential parameter in material science, provides vital insight into how the intrinsic properties of a material’s structure impact the electronic transitions occurring within its bands. An in-depth examination of the dielectric constant provides significant knowledge regarding the complex correlation between a substance’s electronic characteristics and its fundamental structural attributes. Utilizing this analytical methodology facilitates a thorough comprehension of how fluctuations in the dielectric constant indicate the intricate interaction between the electronic configuration and the intrinsic structure of the material. As a result, it becomes possible to predict and characterize the optical and electrical characteristics of a wide range of substances.
71
The material’s polarizability is attributable to the dielectric constant, which influences the non-linear optical properties of substances.
72
It is possible to determine the dielectric constants by utilizing the absolute magnitudes of the n and k. The derivation of the real and imaginary components of the dielectric constant, denoted as εr and εi, respectively, is possible through the utilization of the subsequent equations.65,66

As illustrated in Figure 9(a) and (b), the current investigation presents the dielectric constant values (εr & ε
i
) of PVB/Bi2WO6 nanocomposite films with the photon energy hυ. The εr and the εi exhibited an upward trend with the addition of Bi2WO6.
45
(a) Real part and (b) imaginary part of the dielectric constant for PVB/Bi2WO6 nanocomposite films.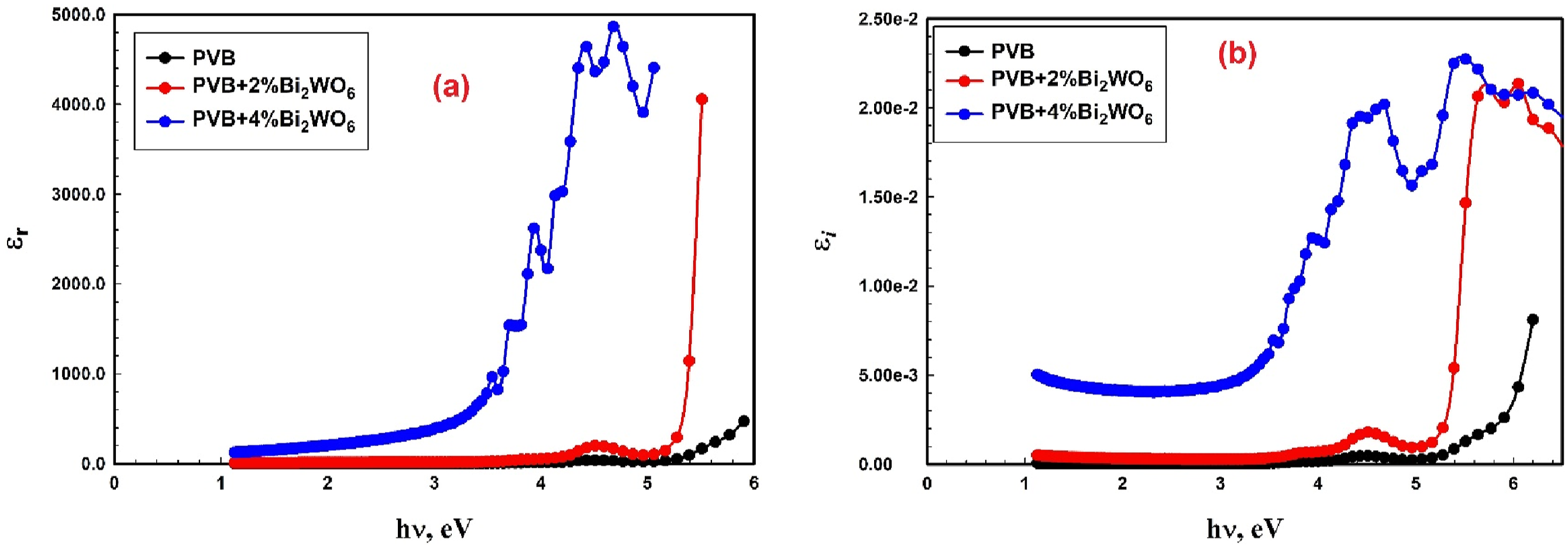
In order to determine the high-frequency dielectric constant ε∞, we must calculate the real portion of the dielectric constant εr using the subsequent equation:73,74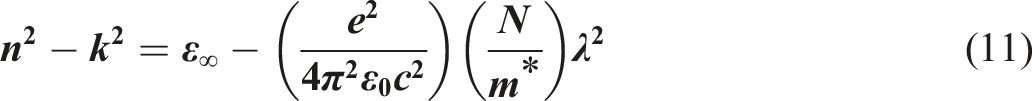
The formula mentioned above incorporates several physical parameters, including the effective mass of the speed of light in vacuum (c), the charge carriers (in kilograms) (m*), the charge of the electron (e), the concentration of free charge-carriers (N), and the permittivity of the free space (ε0).
The high-frequency dielectric constant (ε∞) and the ratio of free carrier ion concentration to effective mass (N/m*) can be determined by calculating the intercept and slope of the linear portion in the εr versus λ2 plots, as illustrated in Figure 10. The corresponding values can be found in Table 2. Bi2WO6 is observed to have a substantial effect on both ε∞ and N/m*. The relation between εr and λ2 for PVB/Bi2WO6 nanocomposite films.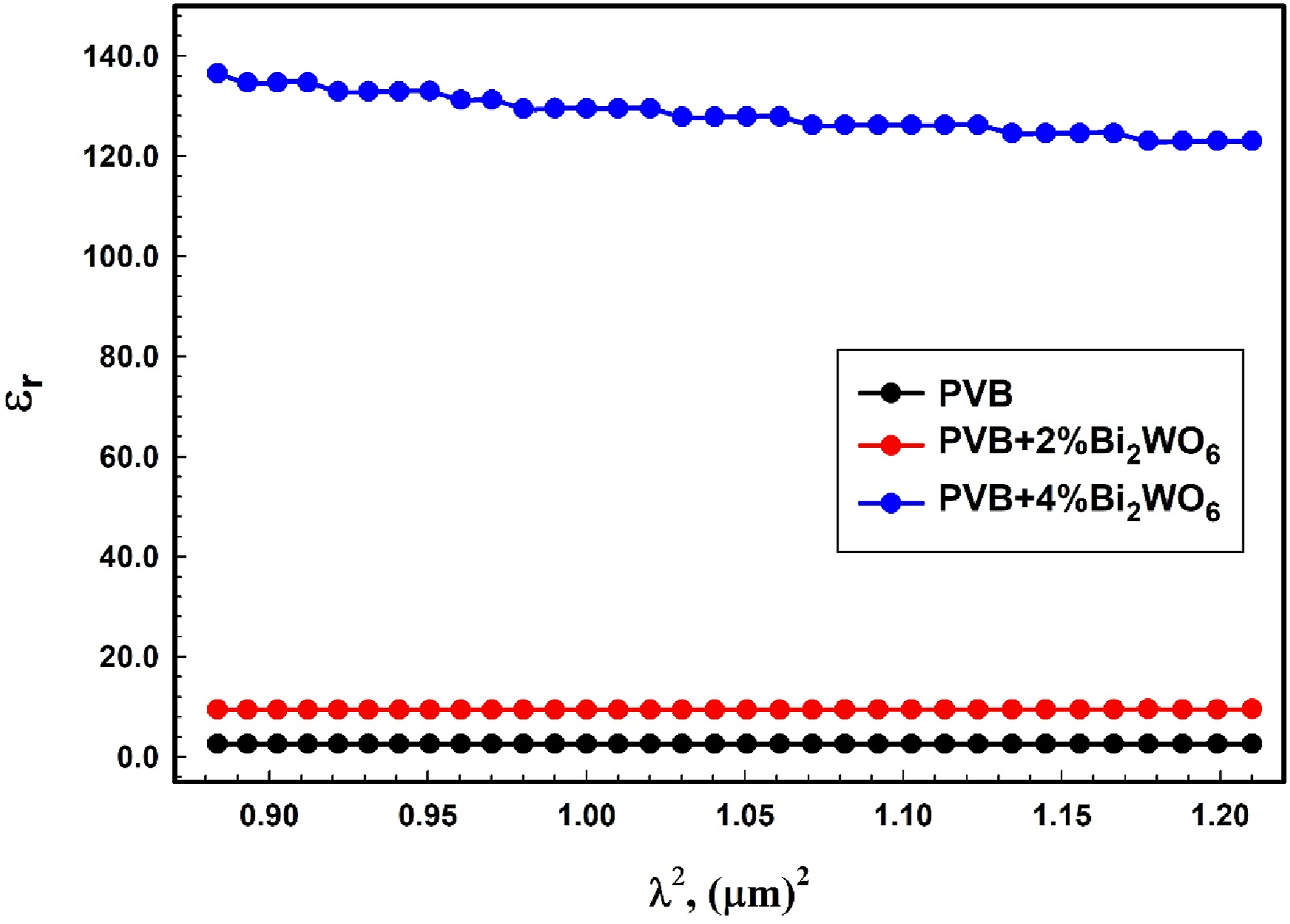
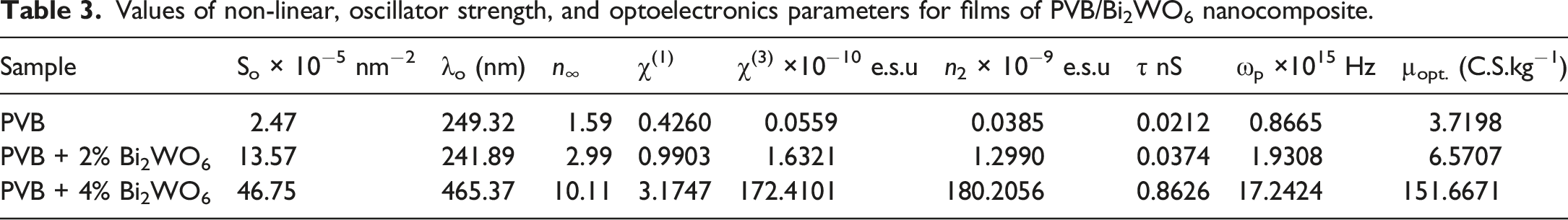
In spectroscopy, the oscillator strength is a dimensionless parameter that signifies the likelihood of absorption or emission of electromagnetic radiation during transitions between energy levels of atoms or molecules. By applying the single oscillator model to PVB/Bi2WO6 nanocomposite films, we can calculate n at an elongated wavelength (n∞), average oscillator wavelength (λ0), and oscillator strength (S0). The linear regression of (n2-1)−1 against λ−2 can accomplish this, as depicted in Figure 11.
64
The relation between (n2- 1)−1 and λ−2 for PVB/Bi2WO6 nanocomposite films.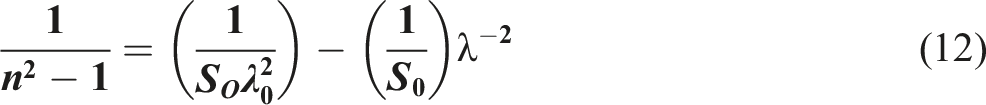

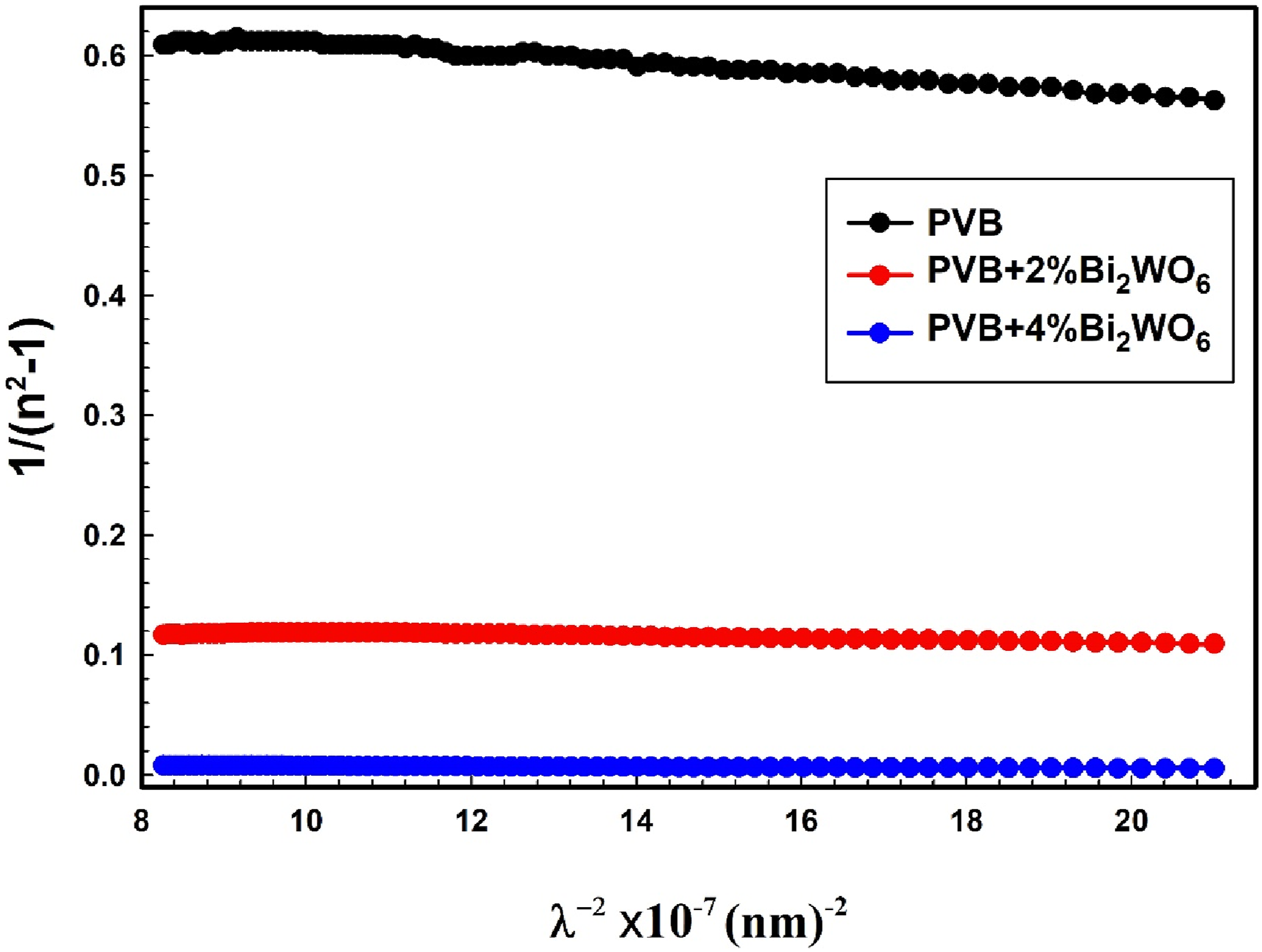
Values of non-linear, oscillator strength, and optoelectronics parameters for films of PVB/Bi2WO6 nanocomposite.
Non-linear optical properties
The potential applications and adaptability of organic non-linear optical (NLO) materials have attracted considerable interest within the scientific community. The applications mentioned earlier comprise various advanced disciplines, including developing optical communication systems on chips, microlasers, illumination technologies, displays, and biological sensing. Due to their multifarious properties, organic NLO materials contribute to technological advancements in various fields. Scholars are actively investigating and capitalizing on the distinctive characteristics of these substances in order to drive progress in various domains, including but not limited to improving communication technologies and transforming biomedical sensing and imaging methods. The increasing attention towards organic NLO materials highlights their critical significance in influencing modern research and technological advancement. 75 When a system is exposed to a substantial amount of energy, nonlinearity manifests itself and causes a transformation in the system’s intrinsic properties. Polarization, frequency, and the path or phase of light that passes through a material are all components of its optical response, referred to as non-linear optics. 76
PVB/Bi2WO6 nanocomposite films must possess the non-linear refractive index, n
2
, linear optical susceptibility, (χ(1)), and third-order non-linear optical susceptibility, (χ(3)), in order to be utilized for a variety of purposes, including communication. As a result, our investigation was directed to establish correlations between (χ(3)) and interband transitions, and to develop a computational formula that calculates the third-order susceptibility of PVB/Bi2WO6 nanocomposite films. For the mathematical calculation of the non-linear optical parameters of PVB/Bi2WO6 nanocomposite films, the subsequent equations were utilized:64,77,78
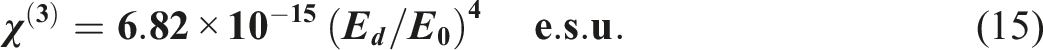

Significantly, the polarization process induces optical nonlinearity in an optical material when it is subjected to an electric field. The findings suggested that as the concentration of Bi2WO6 increased, the values of (χ(1)), (χ(3)), and the n 2 increased, as depicted in Table 3. As the Bi2WO6 content increases, it is observed that χ(1) increased from 0.4260 to 3.17, and χ(3) increased from 0.0559 × 10−10 esu to 172.4 × 10−10 esu. During this time, n2 increased from 0.0385 × 10−9 esu to 180.2 × 10−9 esu. Similarly, Taha 64 investigated the influence of Al2O3 irradiation on the structural, linear, and non-linear optical properties of a PVC composite. Increasing the Al2O3 concentration increases χ(1) from 0.19 to 2.38 and χ(3) from 0.027 × 10−12 esu to 5483.71 × 10−12 esu. When Al2O3 concentration grew from 0 to 6% wt., the value of n 2 increased from 0.0066 × 10−10 esu to 371.81 × 10−10 esu. Additionally, Al-Shawabkeh et al. 79 have reported the effect of ZnO with different concentrations on linear/nonlinear optical properties of PVC/ZnO nanocomposite films.
Opto-electrical properties
The correlations between the imaginary and real components of dielectric constants have been established with the assistance of the Drude model, enabling the calculation of an extensive array of optoelectric factors. The parameters mentioned above are utilized to ascertain critical variables, including optical resistivity (ρopt), relaxation time (τ), and plasma frequency (ωp). Researchers can obtain significant knowledge regarding substances’ electrical and optical properties by employing the Drude model. A comprehensive understanding of materials’ conductive properties and response to external stimuli can be obtained from the calculated parameters, including relaxation time and plasma frequency. By offering a reliable framework for analyzing and predicting the optoelectrical performance of a wide range of substances, the optical mobility and resistivity parameters provide additional insight into the behavior of materials under different optical conditions.
The following represents the connection between the real component of the dielectric constant, εr, the high-frequency dielectric constant, ε∞, and the plasma frequency, ωp:80,81
The slopes of plots between εr and 1/ω2 (see Figure 12) are used to determine ωp values for the analyzed compositions. Table 3 shows the derived ωp values for the tested compositions. Bi2WO6 presence has a substantial influence on ωp variation in PVB composites. The value rises from 0.8665 × 1015 Hz to 17.24 × 1015 Hz with increasing Bi2WO6 concentration. Variation of the real dielectric constant concerning 1/ω2 for PVB/Bi2WO6 nanocomposite films.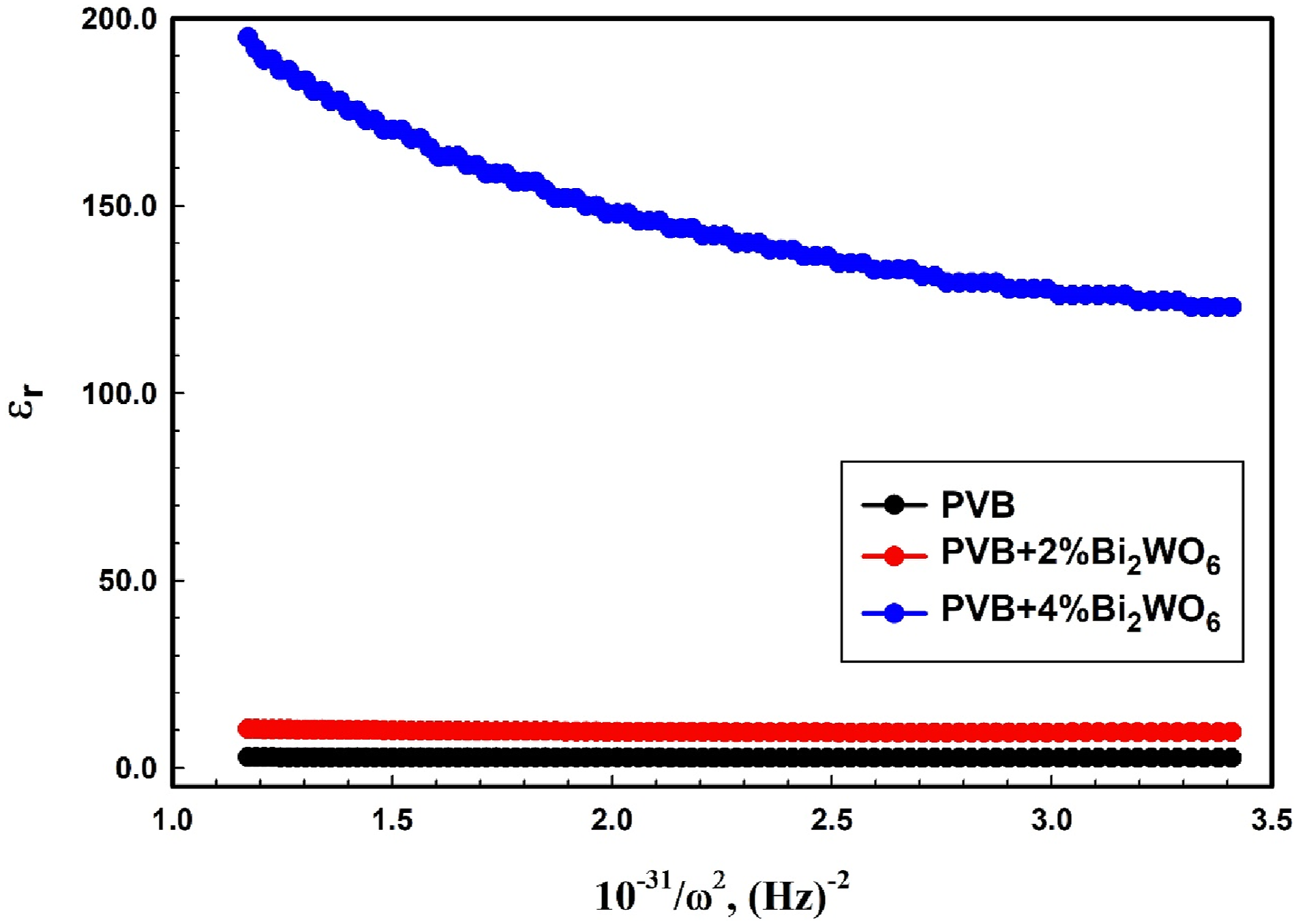
Furthermore, the relaxation time, τ, may be calculated by examining the correlation between the imaginary dielectric constant and wavelength using the given formula:
74
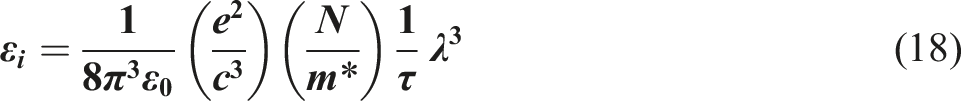
The inverse gradient of the linear relationship between εi and λ3 (Figure 13) may be used to approximate the relaxation time τ. The relaxation time values for the examined compositions are shown in Table 3. The relaxation time values varied from 0.0212 ns to 0.862 ns with increased Bi2WO6 concentration. The relationship between ε
i
and λ3 for PVB/Bi2WO6 nanocomposite films.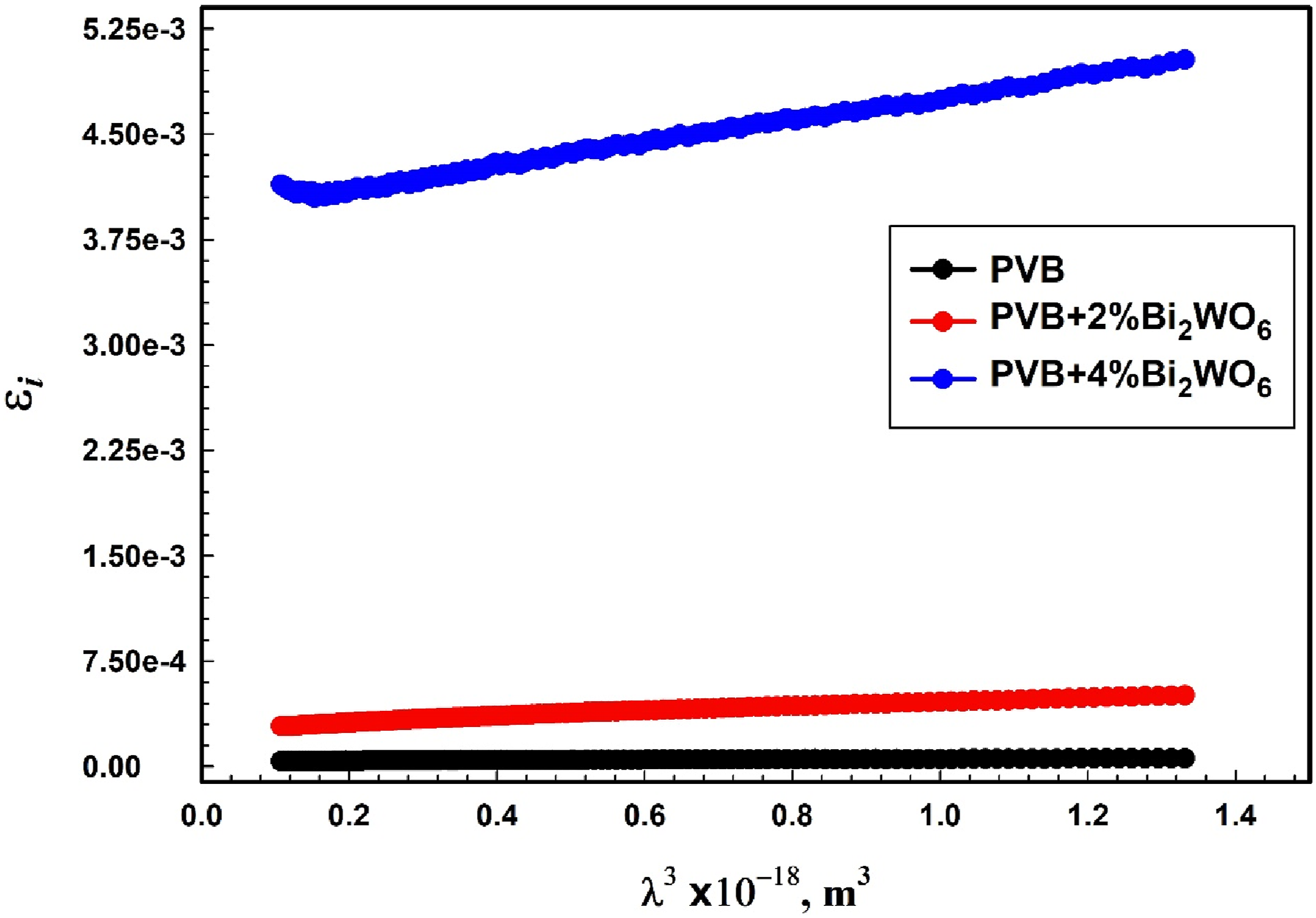
The calculation of optical mobility (µopt) was performed using the relations that include the values of τ:78,82
The PVB + 4% Bi2WO6 sample exhibited a remarkable enhancement in optical mobility (µopt), which increased from 3.7198 C.S/kg for pure PVB film to 151.66 C.S/kg.
Due to the alternating electric field generated by the incoming electromagnetic waves, charge carriers can facilitate optical conductivity. The light response of a substance is primarily determined by its optical conductivity (σopt).
64
Deriving σopt is possible using the following equation:53,64–68
The symbol “c” represents the velocity of light in a vacuum.
The correlation between the photon energy and σopt of the PVB/Bi2WO6 nanocomposite films is illustrated in Figure 14. The optical conductivity exhibited an upward trend as the Bi2WO6 content was modified. The phenomenon arises when the bandgap diminishes, thereby enabling improved conductivity as electrons can more easily transition from the valence band to the conduction band.
83
At a transition potential of 4.5 electron volts, the σopt in our investigation rose from 3.75 × 107 S−1 to 1.059 × 109 S−1 with the addition of 4% wt. Bi2WO6.
43
The optical conductivity (σopt) versus photon energy for PVB/Bi2WO6 nanocomposite films.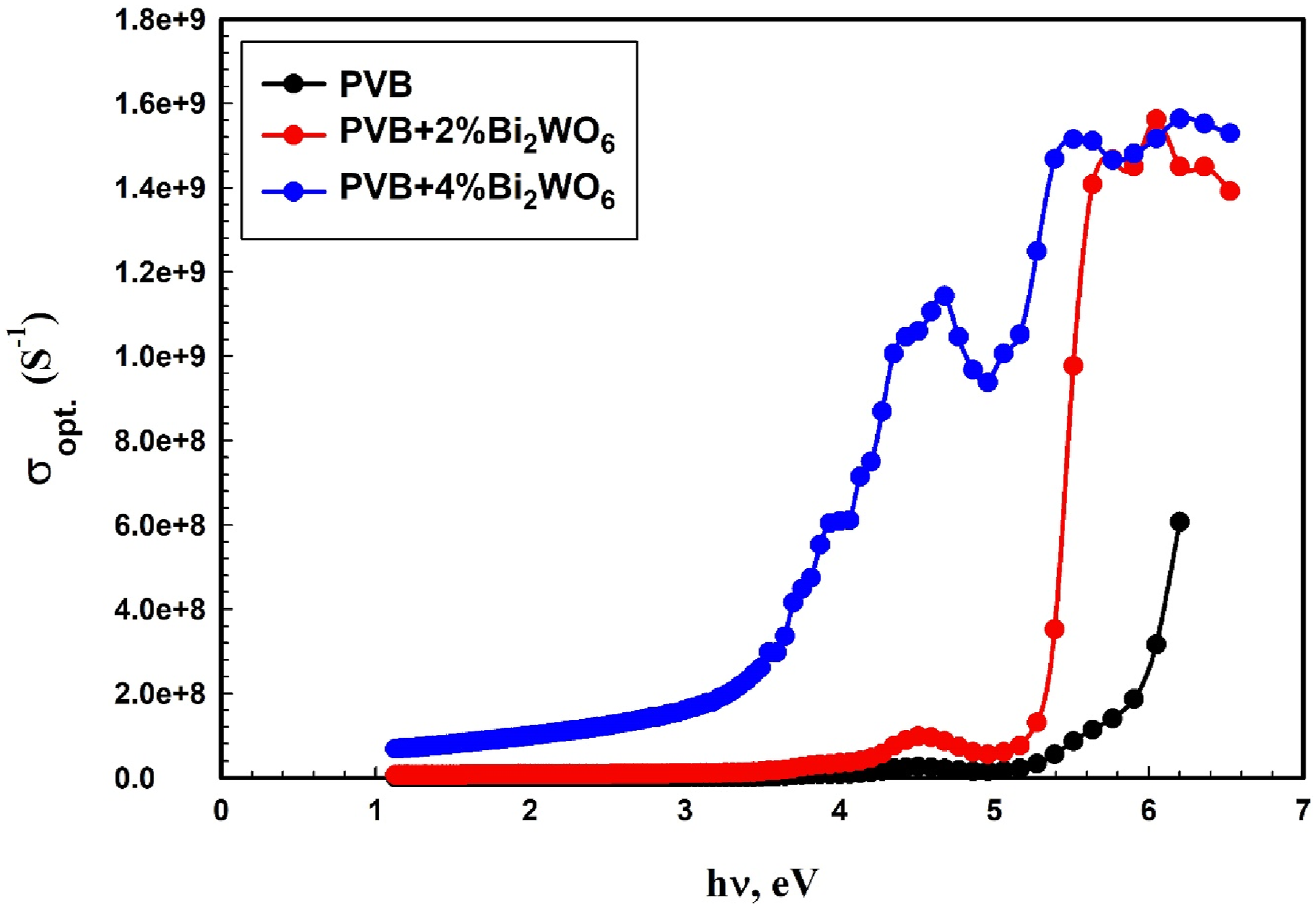
Conclusion
The current research examines the effects of Bi2WO6 nanoparticles (NPs) on PVB/Bi2WO6 nanocomposite films regarding their structural, dispersion, linear/non-linear optical, and optoelectrical characteristics. The information regarding the impact of Bi2WO6 concentration on the degree of crystallinity of the PVB/Bi2WO6 films is obtained through XRD and FT-IR analyses. Dielectric constants and other relevant parameters were calculated using the k and n approximations. In order to determine the compositional optical behavior dependence on the concentration of Bi2WO6, the relaxation time, plasma frequency, and optical mobility were estimated. The Urbach energy increased from 1.00 to 1.97 eV. The oscillator’s energy reduced from 5.47 eV for the pure PVB film to 3.60 eV for the PVB + 4% Bi2WO6 nanocomposite film. Furthermore, the dispersion energy exhibited a range of 8.56 to 444.60 electron volts (eV), while the optical mobility µopt of the enhanced from 3.7198 C.S/kg for pure PVB film to 151.66 C.S/kg for PVB + 4% Bi2WO6 nanocomposite. PVB/Bi2WO6 nanocomposite films have the potential to be utilized in flexible organic electronic devices such as organic light-emitting diodes and solar cells due to their linear/non-linear properties, dispersion parameters, and optical conductivity.
Future prospectives and limitations of the study
Investigating PVB/Bi2WO6 nanocomposite presents several future possibilities and possible constraints worth contemplating. Below are a few observations about both facets: I. A more comprehensive investigation into the impact of various compositions on the physical, mechanical, optical, and electrical properties of PVB/Bi2WO6 nanocomposite can be achieved by examining broader contents of Bi2WO6 % wt. Similar studies have been reported.79,84,85 II. At higher concentrations, achieving a consistent dispersion of Bi2WO6 within the PVB matrix can present an enormous obstacle. The aggregation of Bi2WO6 has the potential to result in performance degradation and non-uniform properties of PVB/Bi2WO6 nanocomposite films. III. Elevated levels of Bi2WO6 have the potential to detrimentally impact the mechanical characteristics of PVB matrix, resulting in a decline in flexibility. Finding a balance between Bi2WO6 concetrations and mechanical performance of PVB/Bi2WO6 nanocomposite films is vital. According to the findings of Zhou et al.,
84
PC/SiO2 nanocomposites containing particle loadings of SiO2 over 4% exhibit a decline in mechanical properties. IV. PVB/Bi2WO6 nanocomposite films include significant amounts of the high atomic number elements Bi and W, making them suitable as flexible materials for shielding against ionizing radiation.39,86
Supplemental Material
Supplemental Material - Structural, linear/non-linear optical, and optoelectrical properties of PVB/Bi2WO6 nanocomposite for industrial applications
Supplemental Material for Structural, linear/non-linear optical, and optoelectrical properties of PVB/Bi2WO6 nanocomposite for industrial applications by Mohammed O Alziyadi, Asma Alkabsh, Basmat Amal M Said and MS Shalaby in Journal of Thermoplastic Composite Materials.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in this published article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.