Abstract
Herein, cadmium sulfide (CdS) nanospheres were synthesized using the hydrothermal method and then inserted into a polyvinyl butyral (PVB) matrix at different concentrations (0%, 2%, and 4% wt.) using the solution casting method. This work examines the effects of varying percentages of CdS spheres on the structural characteristics, mechanical properties, surface/volume energy loss functions, dielectric constants, and linear/non-linear optical properties of PVB nanocomposite films. The findings confirmed the successful synthesis of sphere-like CdS with a cubic structural phase. The stress-strain curves of PVB and its composites with nano CdS spheres were examined. The PVB has a direct/indirect energy gap of 5.1 eV (4.1 eV). With a CdS doping level of 2% and 4%, the values correspondingly decreased to 4.76 eV (3.36 eV) and 4.49 eV (2.45 eV). The EU values for PVB, PVB-2 wt.% CdS, and PVB-4 wt.% CdS nanocomposite are 15.3, 11.2, and 14.1 meV, respectively. CdS nanospheres decreased the volume and surface energy losses (VELF & SELF) of PVB films. The research demonstrates that the mechanical properties and optical parameters of PVB/CdS nanocomposite films positively correlate with the concentration of CdS nanospheres. These attributes make them well-suited for incorporation into flexible electronic devices.
Keywords
Introduction
Recently, considerable attention has been directed towards polymer nanocomposites composed of semiconductor compounds owing to their adjustable electronic, physical, and chemical characteristics. Polymer nanocomposites’ stability and optical properties were improved by incorporating nanoparticles (NPs) and modifying surfaces via passivation of defect states and dangling bonds. 1 The polymer chains’ linker properties also facilitate the forming of an extended framework for the NPs within the polymer matrix.2,3 Polyvinyl butyral (PVB), an abbreviation for Polyvinyl Butyral, is a polymer substance known for its exceptional mechanical qualities and remarkable optical transparency. 4 It is mainly used as an interlayer material for laminated glass in the construction and automotive industries. In laminated glass production, two glass panes are typically joined using a transparent polymer interlayer. 5 Hashim et al. demonstrated using PVB thin film as a sensing electrode. 6 Further, Miao Han et al. 7 documented the development of a near-infrared blocking window by utilizing antimony tin oxide (ATO)-CsxWO3/PVB nano-lamination.
Researchers have recently developed numerous polymer nanocomposites for non-linear optical (NLO) applications. The optical transparency of the matrix polymer and the dimensions and concentration of the inorganic nanoparticles govern the NLO properties of the polymer nanocomposites. The NLO characteristics of polymer nanocomposites can be improved due to the surface functionalities of the inorganic NPs.8,9 Among NPs, cadmium sulfide (CdS), classified as a semiconductor, has a direct band gap material characteristic of a bulk band gap value of approximately 2.42 eV. It is used in various sectors, including photoconductive cells, optical switches, transducers, optical limiting, and non-linear optical devices.10,11 The research on polymer nanocomposites loaded with CdS NPs reveals an improvement in the optical characteristics of the materials. Boranna et al. studied the non-linear optical characteristics of poly(styrene-co-acrylonitrile)/CdS nanocomposites with varying weight percentages of CdS nanoparticles. 9
Herein, this study aims to fabricate and investigate the linear and non-linear optical characteristics of PVB/CdS nanocomposite films. CdS with nanospheres shapes were synthesized via the hydrothermal method. Then, different percentages (0%, 2%, and 4%) wt. were incorporated into the PVB matrix through solution casting. Finally, we aimed to explore the effect of CdS content on structural, linear/non-linear optical properties of PVB/CdS nanocomposite films.
Materials and experimental procedure
Materials
Cadmium sulfate (CdSO4, 99%) and carbon disulfide (CS2, 98) were purchased from PubChem. Cetyl trimethyl ammonium bromide (CTAB, 98%) was acquired from a Merck, India. Cadmium sulfide nanoparticles were synthesized using deionized water. Polyvinyl butyral and Ethyl alcohol (Ethanol, 99.5%) were purchased from Sigma-Aldrich.
Synthesis of CdS spheres
To synthesize the CdS sphere, 100 mL of deionized water was used to dissolve 0.1 M CdSO4, 0.1 M CS2, and 4 mM CTAB. Afterward, 10 mL of isopropanol was included in the solution combination. A 250 mL Teflon-lined autoclave was used to hold the solution mentioned above while it was subjected to continuous oven heating at 160°C for 15 h. The CdS powder was then centrifuged and washed with deionized water. The result was a dark orange crystalline product.
Synthesis of the PVB/CdS nanocomposite films
The PVB/CdS films were synthesized using PVB (4%) polymer solution, prepared by dissolving the necessary weight of PVB in ethanol. The solution was stirred using a magnetic mixer at room temperature for 30 min to ensure uniformity. The PVB solution was mixed with CdS spheres at varied concentrations (0%, 2%, and 4% wt.) and then subjected to 2 hours of sonication. The last step was to cast the solution in Petri-dish and left in a dark, room-temperature setting to dry for 48 h, as presented in Figure 1.12,13 Schematic diagram for CdS spheres and PVB/CdS nanocomposite synthesis. PVB: polyvinyl butyral; CdS: cadmium sulfide.
Characterizations
The crystal structure of PVB/CdS nanocomposites was analyzed using Shimadzu 6000 X-ray diffraction (XRD) equipment with an 8°/min scanning rate. Fourier Transform Infrared spectroscopy (FTIR) spectra were acquired using a Nicolet iS10 instrument. The elemental composition and surface morphology of a nanocomposite film (JEOL JTEM-1230) model were assessed using scanning electron microscopy (SEM) and energy dispersive X-ray spectroscopy (EDX). The mechanical properties of PVB/CdS nanocomposites were determined using the tensile testing equipment Qchida computerized testing machine from Dongguan Haida Equipment Co. Ltd in China. The optical characterization was conducted using a UV-vis-NIR spectrophotometer (Jasco, V-570).
Results
Structural studies
An investigation was conducted on the crystallinity and structure of grown CdS using X-ray diffraction. The crystal XRD pattern of CdS is illustrated in Figure 2(a). In comparison to the data in JCPDS file no. 42-1411, three diffraction peaks at approximately 2θ ⁓ 26.73°, 44.47°, and 52.2° correspond to three crystal planes (111), (220), and (311), respectively, of the cubic phase of CdS.14,15 This suggests that CdS nanoparticles have been effectively synthesized. The broadening of XRD peaks in CdS nanoparticles might be attributed to their small size. The average crystallite size of CdS nanoparticles was estimated by utilizing the well-known Scherer equation
16
(a) XRD pattern, (b) FTIR spectra, (c) SEM image, and (d) particle size distribution histogram of CdS spheres. XRD: X-ray diffraction; SEM: scanning electron microscopy; FTIR: Fourier Transform Infrared spectroscopy; CdS: cadmium sulfide.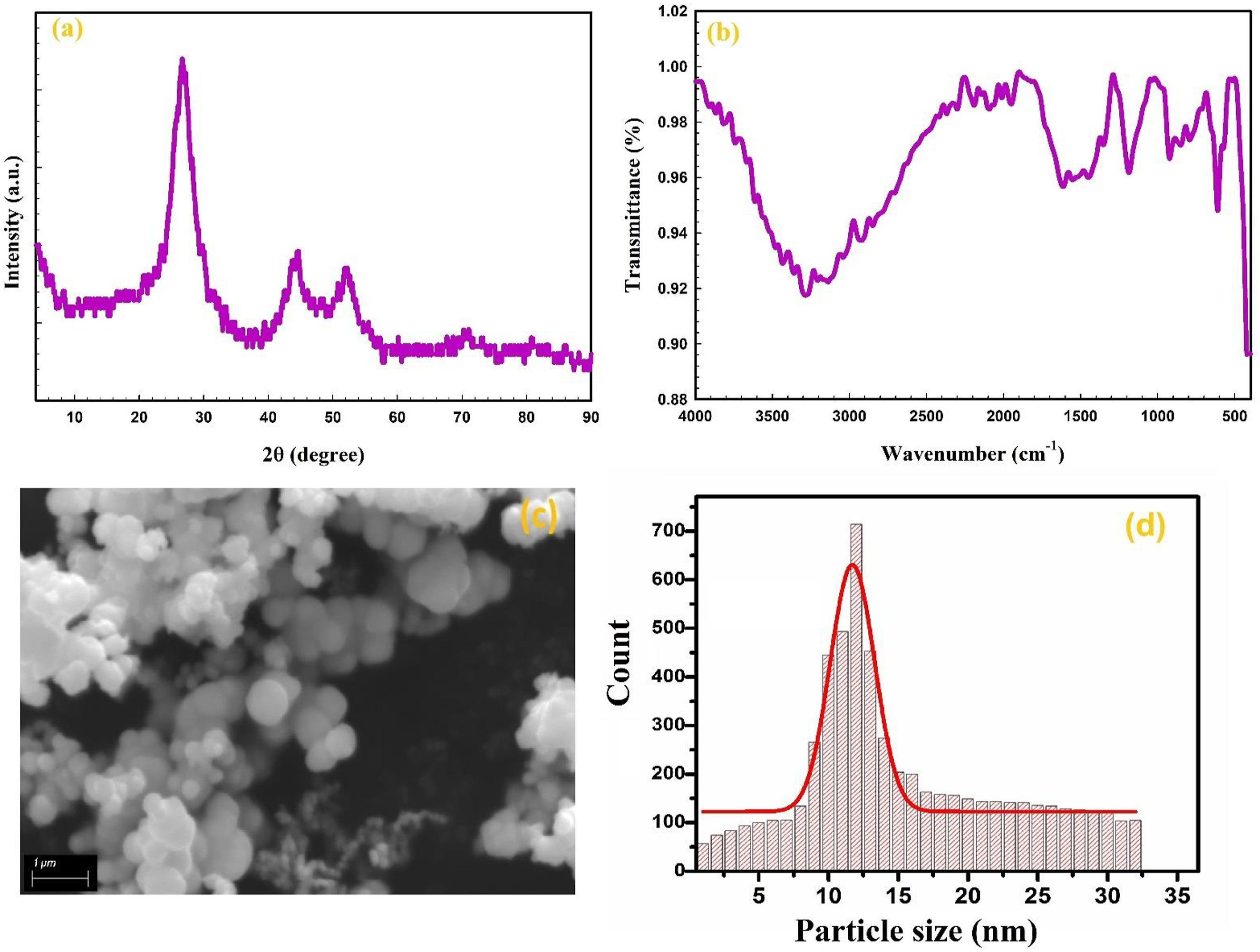
The symbol β denotes the full width at half maximum (FWHM) of the observed peak in radians, whereas the value of the wavelength (λ) of Cu Kα radiation is 0.15406 nm. The average crystallite size of CdS was found to have a D value of 3.6 nm.
Utilizing the following equations, microstrain (εs), dislocation density (δ), stacking fault (SF), and the number of crystallites per unit surface area NC of CdS nanospheres are calculated
17
The values of εs, δ, SF, and NC of CdS nanospheres are 1.06 × 10−2, 7.72 × 10−2 nm−2, 6.24 × 10−3, and 7.14 × 10−3 nm−2, respectively.
Figure 2(b) displays the FTIR spectra of CdS. The band noticed at 610 cm−1 corresponds to the symmetric stretching vibration of Cd-S, whereas the band located at 922 cm−1 is associated with the bending mode of carbonate. The band at 1186 cm−1 corresponds to the S–O stretching, which arises from the interaction between atmosphere oxygen and sulfur in the CdS. The prominent peak at 3286 cm−1 corresponds to water molecules’ O–H stretching vibration.15,18 The SEM image depicted the surface morphology of CdS, as illustrated in Figure 2(c). SEM image demonstrates that CdS consists of sphere-shaped particles with irregular sizes. The particle size distribution histogram performed using ImageJ software (Figure 2(d)) revealed that the smallest average particle size for CdS spheres was 12.5 nm, with the narrowest size distribution.
19
Furthermore, Figure 3 displays visual representations of elemental mapping images of CdS spheres. The images showed a homogeneous distribution of all elements (Cd and S) over the whole sample, with no presence of any additional element. Elemental mapping images of cadmium sulfide nanospheres.
XRD enables us to understand the material’s crystal structure, crystallite orientation, and crystalline sizes. XRD patterns of PVB containing 0, 2, and 4 wt. percent of CdS nanospheres are shown in Figure 4(a). Notably, a broad diffraction peak from 2θ of 5° to 25° shows the amorphous structure of PVB developing.20,21 When the CdS contents in the PVB matrix grow, the diffraction peaks corresponding to CdS spheres rise while the peak of PVB decreases, indicating that the PVB/CdS nanocomposites have been successfully created. (a) XRD patterns and (b) FTIR spectra of PVB/CdS nanocomposites. XRD: X-ray diffraction; FTIR: Fourier Transform Infrared spectroscopy; PVB: polyvinyl butyral; CdS: cadmium sulfide.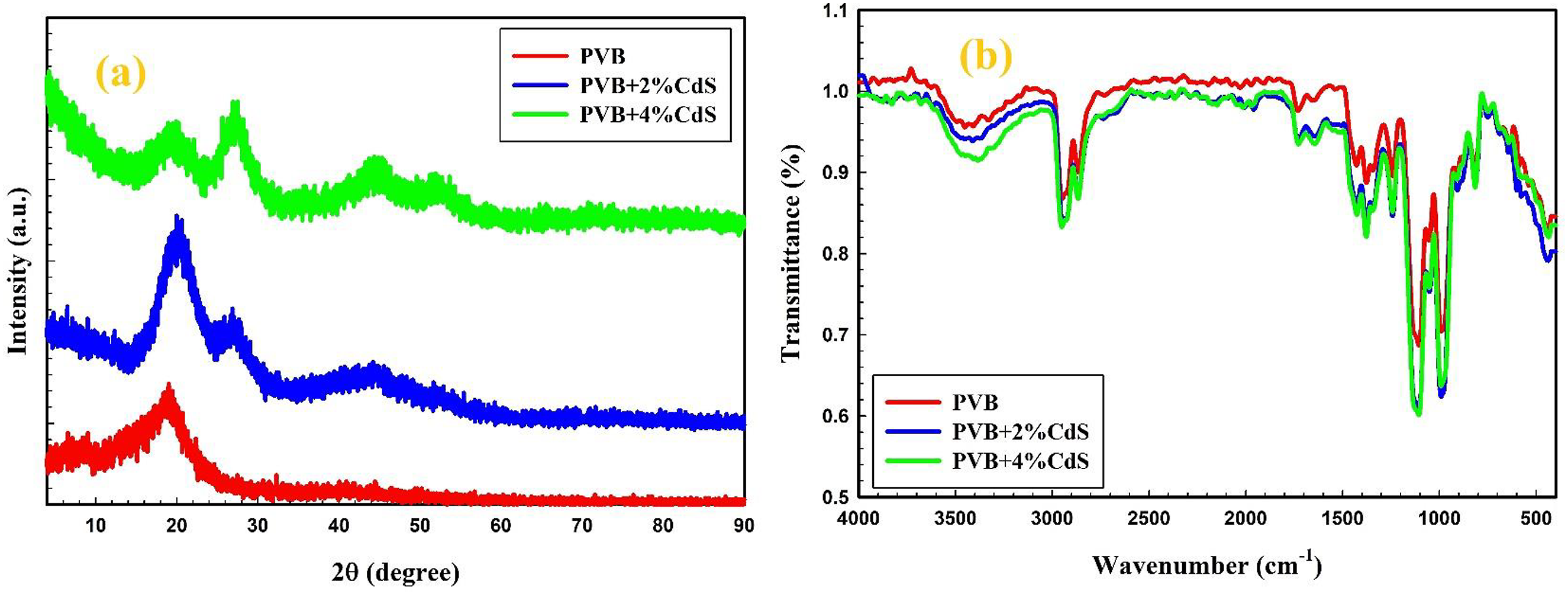
FTIR spectroscopy is typically employed to analyze alterations in polymer nanocomposite’s chemical structure and chemical bonds. The FTIR spectra of PVB filled with 0, 2, and 4 wt.% CdS spheres are illustrated in Figure 4(b). Several processes, including physical and chemical interactions, influence the molecular interactions between PVB and CdS. PVB has hydroxyl (-OH) groups that may combine with sulfur atoms in CdS to generate hydrogen bonds. Additionally, any charged areas on the CdS surface interact with the polar PVB regions (hydroxyl groups). Similar mechanisms were observed in Refs. 22–24 FTIR spectra of pure PVB demonstrated that the absorption peak at 3451 cm−1 was attributed to the OH stretching modes of PVB. In PVB/CdS nanocomposites, this band has been repositioned to higher frequencies, and its broadening and transmission values have increased. This illustrates the interaction between CdS molecules and the functional groups in the PVB structure. 25 The 2947 cm−1 and 2867 cm−1 bands represented the symmetric and asymmetric stretching vibrations of the CH2 and CH3 groups. The absorption bands identified C = O groups observed at 1730 cm−1, 1376 cm−1, and 1242 cm−1. At 1730 cm−1, this band was altered to higher frequencies for PVB/CdS nanocomposites. The interaction between CdS and the C = O group of PVB molecules is thus a crucial factor in this process. Due to the C–O–C groups in cyclic ether, the bands observed at 1105 cm−1, 989 cm−1, 816 cm−1, 634 cm−1, and 442 cm−1 were formed. 26 Increasing the concentration of CdS nanospheres in PVB/CdS nanocomposites results in a dispersion of the band position from 442 cm−1 to 436 cm−1. Thus, there is substantial interaction among the constituents of PVB/CdS nanocomposites. A similar behavior was observed between ZnO NPs and the PVA matrix by Abd-Elnaiem et al. 17
Mechanical properties
The structural alterations of PVB/CdS nanocomposites due to CdS sphere intrusion can be evaluated through mechanical examination. The stress-strain curves of PVB and its composites with nano CdS spheres were examined and illustrated in Figure 5. Pure PVB has a tensile strength of 34.7 MPa and an elongation at a break of 7.97%. When the CdS spheres percentage is increased to 4% wt., the tensile strength of PVB/CdS nanocomposites increases notably to 144.9 MPa. In addition, the PVB/CdS nanocomposite doped with a CdS sphere ratio of 2% wt. has elongation at a break of 10.28% and then diminished by increasing the CdS spheres ratio to 4% wt. with elongation at a break of 8.06%, resulting in the PVB/CdS nanocomposites becoming essentially brittle and breaking with a significantly lower strain. The stiffness of PVB/CdS nanocomposites is increased compared to undoped PVB by increasing the content of CdS spheres. This is because CdS is significantly stiffer than PVB and has a lower deformation capacity, which reduces the PVB matrix strain. So, the PVB/CdS nanocomposites flexibility is reduced as the concentration of CdS reaches an optimal level of 2% wt.27–30 Similar results were reported for the study of the effect of fillers on the mechanical properties of polymer nanocomposites. Fouad et al.
31
showed that the PMMA/5 wt.% Al2O3 composite had more incredible compressive characteristics than the undoped-PMMA, with an ultimate compressive strength of 54.75 MPa and a yield strength of 45.6 MPa, which was enhanced by approximately 98% and 117%, respectively. Furthermore, Rhalmi et al.
32
found that raising Young’s modulus when the Mg-Al layered double hydroxide loading reached 5 wt.% improved the mechanical characteristics of the starch nanocomposites. Shokrieh et al.
33
reported that adding graphene to polypropylene significantly increased mechanical parameters at low graphene contents, with a rise of 21% in yield strength and 34% in Young’s modulus at 0.5 wt.% graphene loading. Stress–strain curves of PVB and its nanocomposites with nano CdS spheres. PVB: polyvinyl butyral; CdS: cadmium sulfide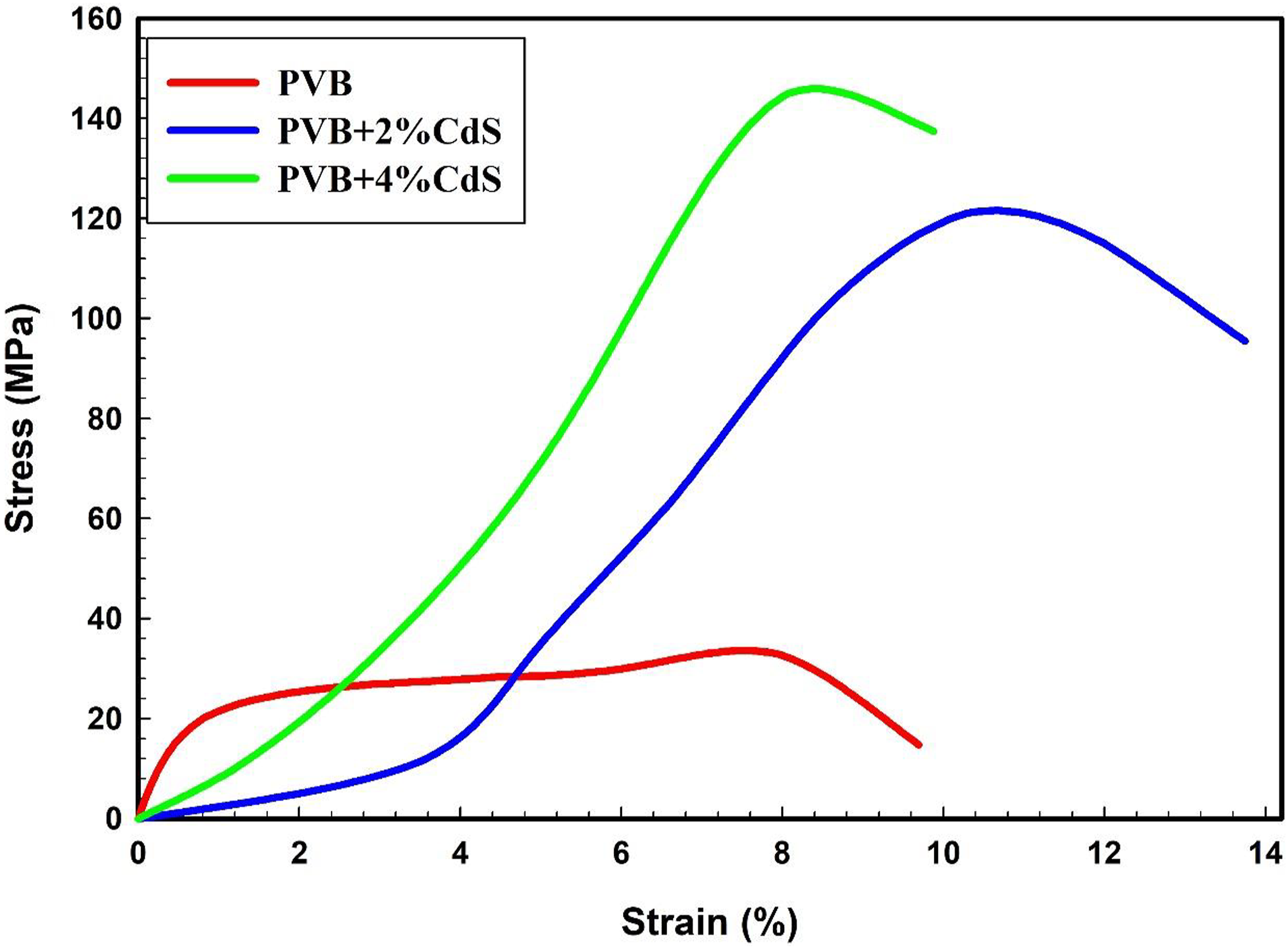
Optical properties
Absorbance and transmittance spectra
Figure 6(a) and (b) displays the absorbance and transmittance of several PVB/CdS nanocomposites throughout a broad spectrum of wavelengths, ranging from ultraviolet to near-infrared. As shown in Figure 6(a), the absorption peak at 280 nm is present in all of the films. These peaks are driven by electron migration from (C = C) unsaturated bonds, namely π → π* transitions.
34
Doping PVB films with CdS were shown to increase their absorbance. Additionally, as the CdS ratio increases, a red shift toward longer wavelengths occurs. The increase in absorbance can be attributed to the fact that CdS spheres modify the internal structure of the host polymer and reduce the strength of hydrogen bonds between polymer chains, thereby enhancing the amorphous phase of the polymer.
35
Further, the localized electronic states in the Highest Occupied Molecular Orbital-Lowest Unoccupied Molecular Orbital (HOMO-LUMO) gap may be produced through CdS spheres. These localized electronic states strongly influence the optical and electrical properties, which serve as recombination and entrapment centers.
36
The addition of CdS spheres to the PVB matrix could result in a red shift in the absorption spectrum due to the quantum confinement effect and altered energy band structure. Consequently, the low-energy transitions cause the observed shift in the optical absorbance.37,38 (a) Absorbance and (b) transmittance of PVB/CdS nanocomposites. PVB: polyvinyl butyral; CdS: cadmium sulfide.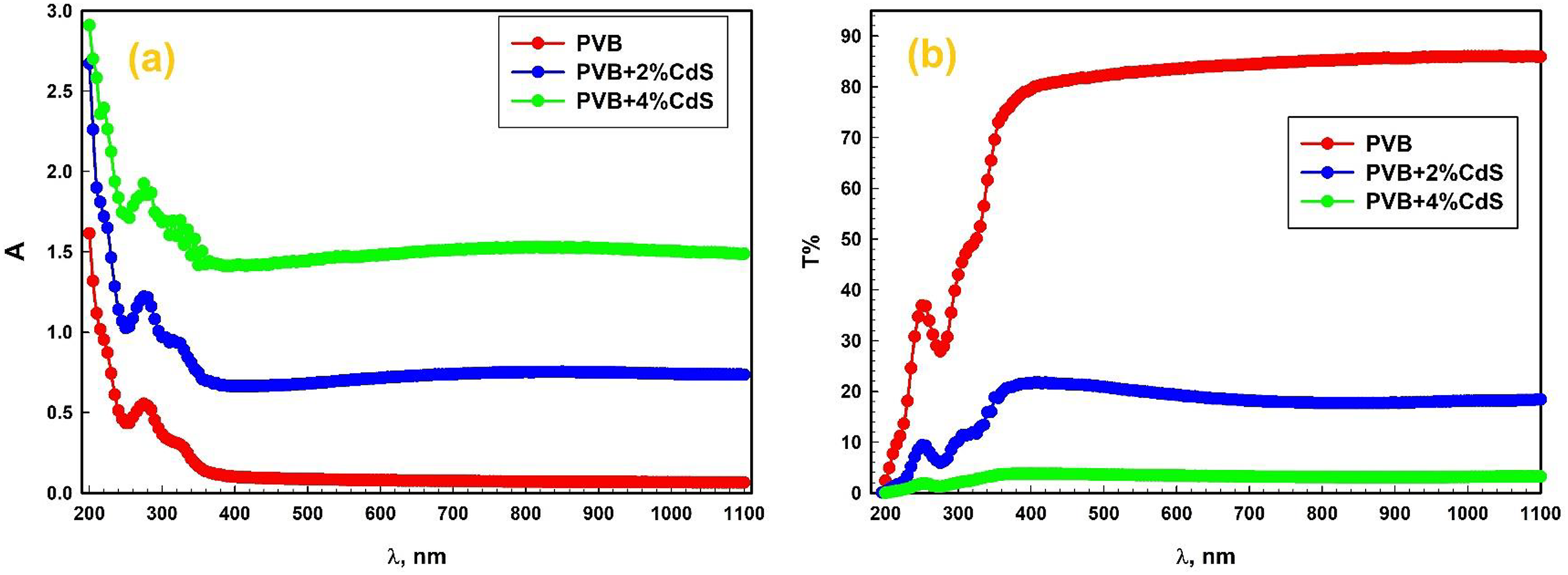
Figure 6(b) shows that the transmittance changes in two stages based on the wavelength of the incident light: (1) T increases noticeably as the incident wavelength increases in the ultraviolet area, which is between 200 and 255 nm in wavelength. (2) T grows little with wavelength for wavelengths more than 255 nm and then increases to reach a stable value for the remaining portion of the studied wavelength. The PVB exhibited a significant transmittance at wavelengths exceeding 385 nm, reaching 87.6% before a sharp decrease when CdS was added.
Optical band gap and Urbach energy
Below, we can observe the relationship between the incident light intensity (Io) and the transmitted light intensity (I)
39
The sample thickness is denoted as t and measured in cm, whereas the absorption coefficient is represented as α and measured in cm−1.
Consequently, the following relationship provides for the direct calculation of the absorption coefficient (α)
Figure 7(a) displays the spectral absorption coefficients (α) of PVB/CdS nanocomposites. As the proportion of CdS nanospheres grows, the value of α (λ) rises. The absorption properties of materials are strongly influenced by their electronic band structures, which influence their optical characteristics.40–45 (a) The absorption coefficient (α) versus hυ, (b) (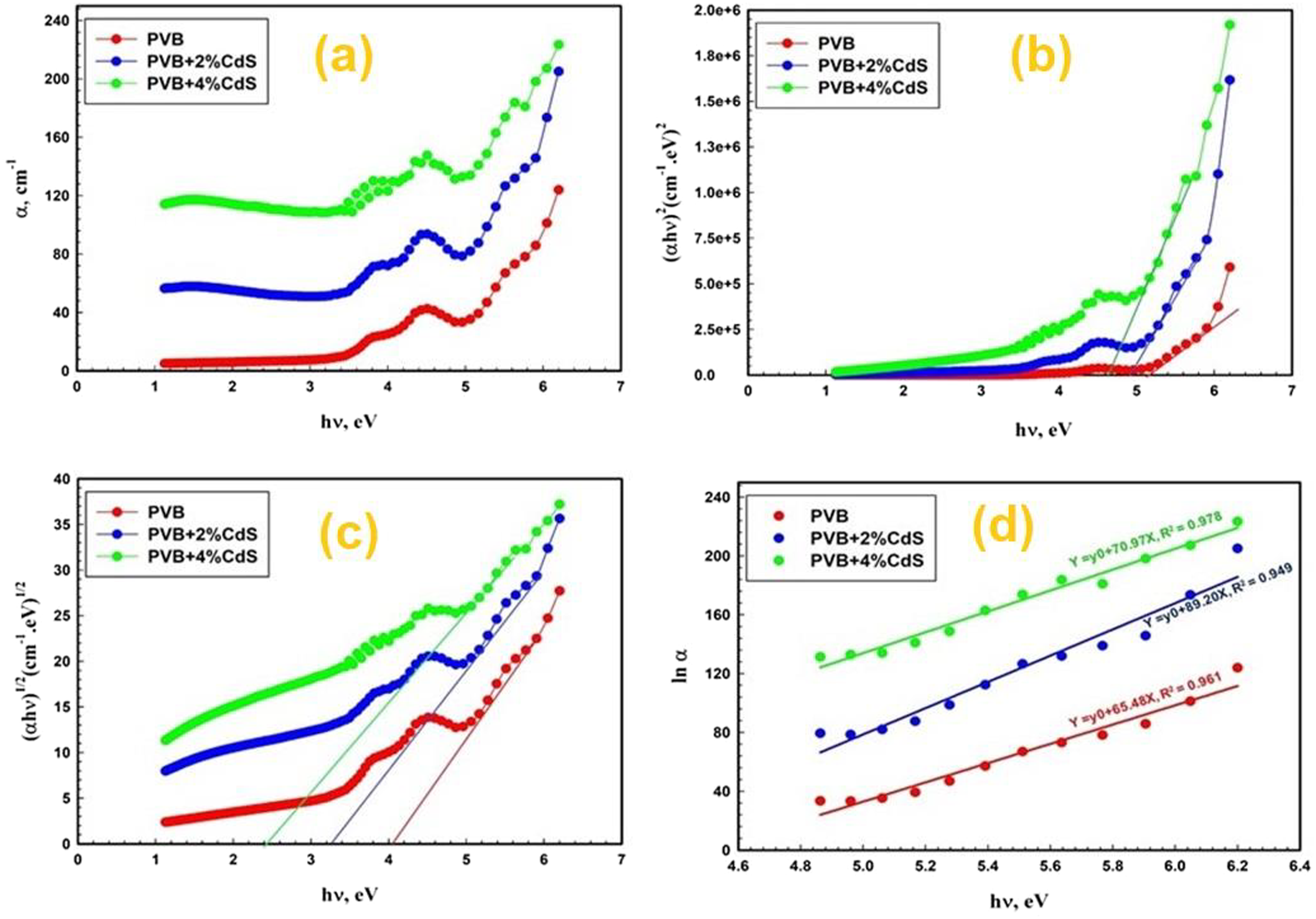
The conventional relation was used to estimate optical direct (Eg1d) and indirect (Eg2ind) energy transitions for nanostructured films of undoped PVB and CdS-doped PVB46–48
In this context, the photon frequency is represented by the symbol υ, the Planck constant is indicated as h, the proportionality constant is represented by A, and the index of the electronic transition is designated as m. The value of m is 2 or 3 for direct energy bandgaps, whereas the values of m are 1/2 or 3/2 for indirect optical bandgaps.
Variation of optical parameters of PVB/CdS nanocomposite films.
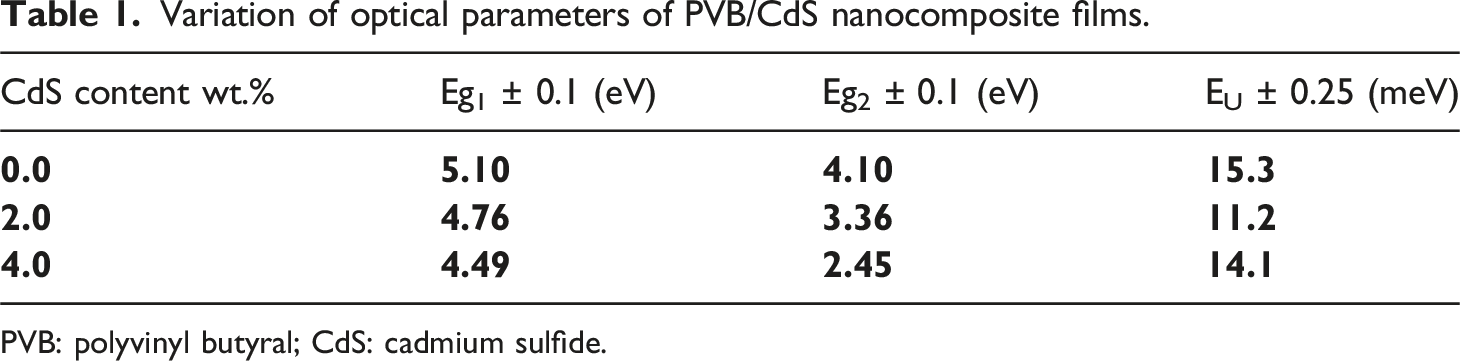
PVB: polyvinyl butyral; CdS: cadmium sulfide.
Figure 7(c) illustrates the relationship between the energy of incoming photons (hυ) and the square root of the absorption edge (αhυ) for the CdS/PVB nanostructured materials with varying doping ratios of CdS. The relationship between the indirect optical bandgaps and the CdS doping percentages was inverse. The CdS-doped PVB nanocomposite films exhibited indirect energy bandgaps (
The optical energy bandgaps of PVB/CdS nanofilms exhibit an inverse correlation with the weight concentrations of CdS nanospheres owing to the formation of intermediate energy levels when the dopants are increased. When the CdS nanosphere dopants are incorporated, the decrease in energy bandgap may be attributed to the creation of intermediate energy levels, which facilitate the movement of electrons between the conduction and valence bands. This suggests that the relative proportions of impurities influence the electronic transitions. 39
The width of localized states (EU) was established by evaluating the slope of the fitted lines, which is related to the Urbach formula
52

The value of Eu of PVB/CdS nanocomposites is determined by the reciprocal of the slopes of the linear part, which is located below the optical band gap. From Figure 7(d), the EU values are 15.3, 11.2, and 14.1 meV for PVB, PVB-2 wt.% CdS, and PVB-4 wt.% CdS nanocomposite, respectively (Table 1). The variations in value may be attributed to an increase in the number of traps in the PVB/CdS nanocomposite, which leads to a change in the number of efficient energy transitions and a resulting change in Eg.53,54
Refractive index and extinction coefficient
The assessment of the refractive index (n) and, consequently, the extension coefficient (k) of optical material may be critical, given its extensive relevance to the electronic polarization of ions and the optical field contained within materials, and thus to the development of a wide range of optical devices. Additionally, k illustrates the discrimination of materials concerning the light of a specific wavelength and demonstrates the variations in absorption that occur as an electromagnetic wave passes the optical material. 17
Both k and n for all CdS-doped PVB nanocomposite films can computed utilizing the subsequent equation.55–57
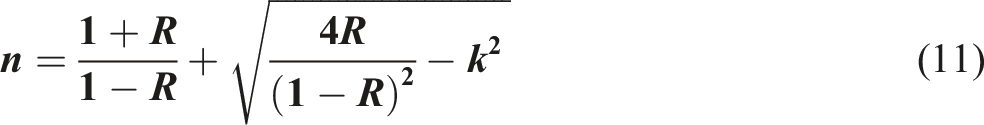
Figure 8(a) and (b) illustrate the relationship between the extension coefficient, the refractive index, and wavelength for PVB/CdS nanocomposite. Both n and k exhibit a substantial decrease as the wavelength increases within the range of 200–250 nm, followed by a slight rise within the range of 250–300 nm. However, the primary distinctions in their behavior are that n remains constant while k increases as the wavelength increases between 400 and 1100 nm. Furthermore, with increased CdS ratios on the PVB matrix, both n and k were increased. With no CdS nanosphere embedding into PVB, the k value is 1.97 × 10−4, but with 4% CdS nanosphere, it rises to 3.55 × 10−4 at 200 nm. Since CdS nanospheres are incorporated with PVB, their absorption of light at this wavelength is significantly affected. When we shift our focus to longer wavelengths, we often find that the k values go higher for any concentration of CdS nanosphere. The value of k tends to rise, suggesting a more significant light extinction as CdS nanosphere concentration increases, which is consistent with the tendency. These findings indicate that the PVB nanocomposite’s light absorption capacity is enhanced when the concentration of the CdS nanosphere increases, particularly in the UV-visible spectrum. The variation of the extinction coefficient with different concentrations of CdS nanosphere is substantial in applications where the material’s optical characteristics are vital. For instance, these extinction coefficient fluctuations impact efficiency and sensitivity in optoelectronic devices or sensors. Furthermore, with increased CdS ratios in the PVB matrix, both n and k were increased.
58
The anomalous dispersion behavior of the refractive index below the absorption edge is also noticeable. This phenomenon may be ascribed to electron polarization due to the light and CdS nanosphere resonance interaction. Moreover, the refractive indices rise as the concentration of the CdS nanosphere increases due to the augmentation of dislocations and the level of structural disorder.
58
(a) The relationship between the extension coefficient and (b) the refractive index and wavelength for PVB/CdS nanocomposite. PVB: polyvinyl butyral; CdS: cadmium sulfide.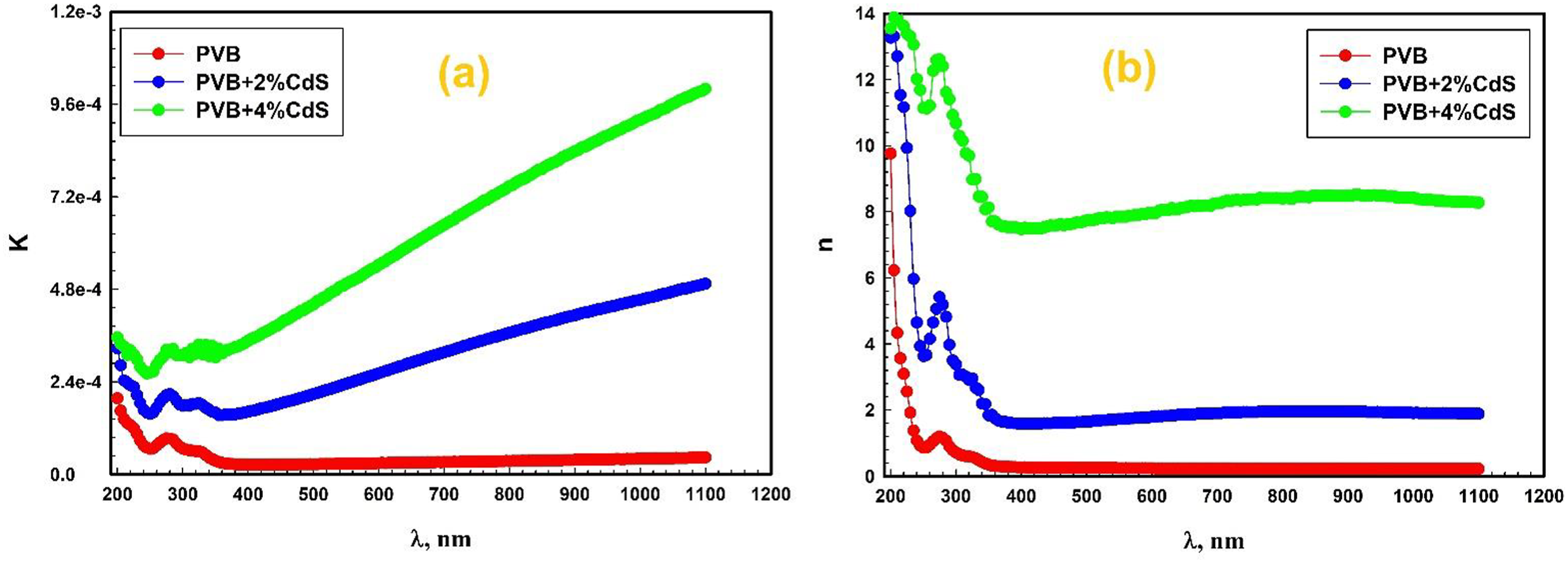
Dielectric constants
One of the most essential characteristics of materials is their complex dielectric constant, which describes how the material conducts electrically and optically when exposed to electric fields. It comprises two constituent parts. The material’s polarizability in response to an electric field is represented by the real dielectric constant, which is also associated with the capacity of the material to store electric field energy. Consequently, the subsequent expression may derive the real part (εr) of PVB/CdS nanocomposite films.59,60
The second part of the complex dielectric constant is the imaginary dielectric constant, quantifying the extent of dielectric losses or energy absorption by the material. It is determined using the next formula
58

The relationship between photon energy (hυ) and the real dielectric constant for both the pure PVB film and the CdS nanosphere-doped PVB film is seen in Figure 9(a). The optical spectroscopy findings show that the real dielectric constant εr of the PVB polymer is substantially increased with the addition of CdS nanosphere. The imaginary dielectric loss of the CdS nanosphere-doped PVB film over the photon energy (hυ) is illustrated in Figure 9(b). Similar behavior was detected for PVC/PE/Cu films.
58
(a) Real dielectric constant (εr) and (b) Imaginary dielectric constant εi, as a function of hυ of the PVB/CdS nanocomposite. PVB: polyvinyl butyral; CdS: cadmium sulfide.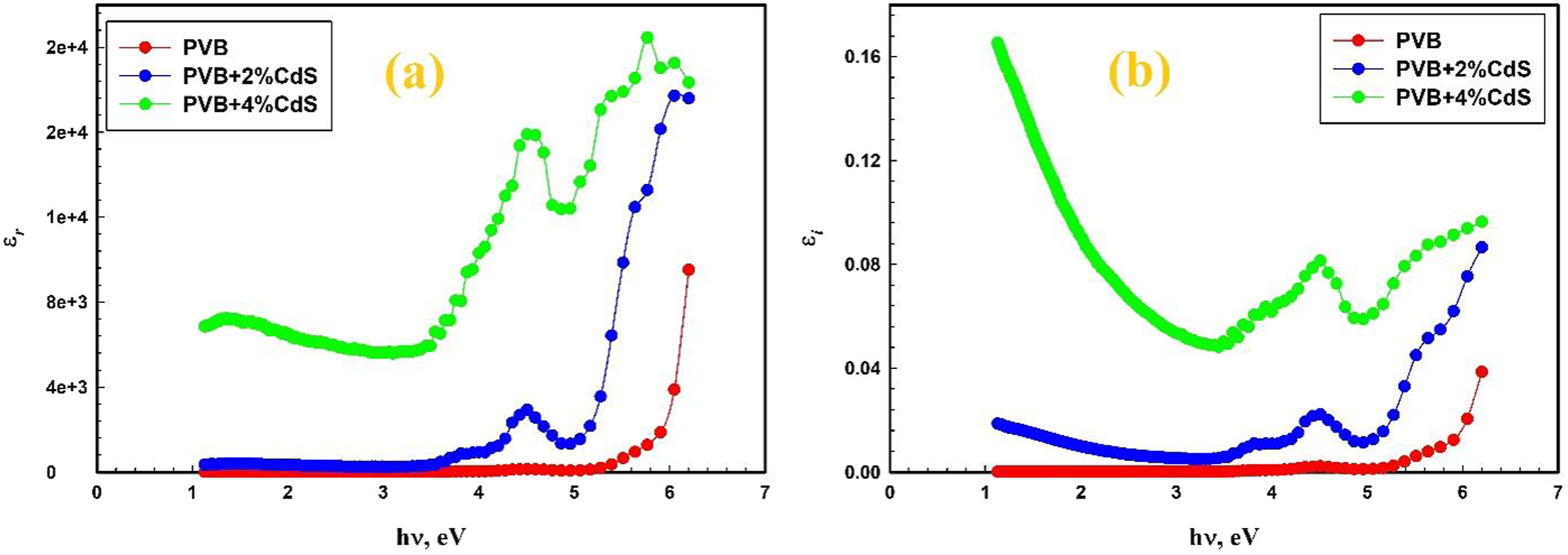
Variations of optical surface resistance and optical conductivity
The surface functions denote the resistance of the PVB/CdS nanocomposite film’s surface, which is generated when photons strike the material and excite electrons. Utilizing the subsequent formulas, the optical surface resistance (Rs) was computed
34

The wavelength dependence of resistance (Rs) for PVB/CdS nanocomposites is depicted in Figure 10(a). The graph illustrates that as λ increased, there was a tendency for Rs to rise. Adding CdS nanospheres to the PVB matrix resulted in a decreasing trend in Rs values. Doping with CdS nanospheres resulted in an alteration of the microstrain in the host PVB, which subsequently impacted Rs. Variation of (a) Rs and (b) 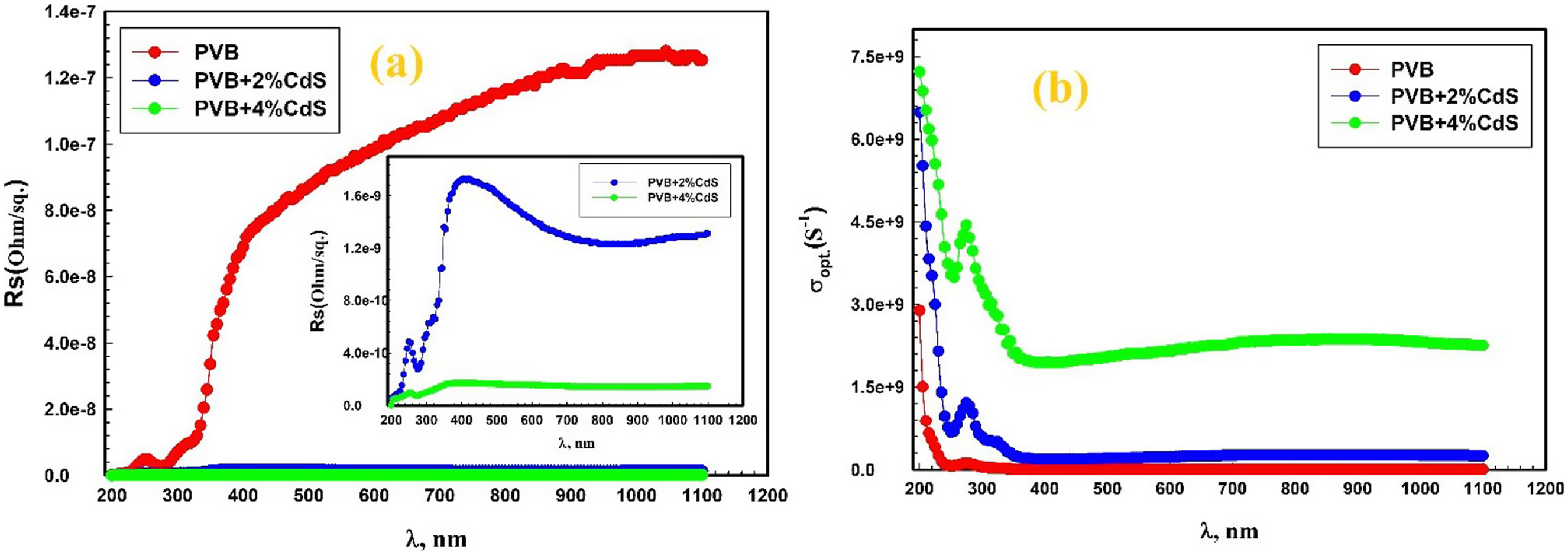
To facilitate the examination of charge transport mechanisms occurring within doped polymers, an optical parameter called optical conductivity (
As illustrated in Figure 10(b), the optical conductivity (
Energy loss functions
The energy dissipation is correlated with the optical characteristics of the substance via the dielectric function. The energy loss rate of electrons as they pass through the material and interact with its surface is referred to as the volume energy loss function (VELF) and surface energy loss function (SELF). The VELF and SELF of CdS nanospheres doped PVB are determined by the real and imaginary components of the dielectric constant, which may be expressed using the following equations assessed
63


Figure 11(a) and (b) demonstrate that the energy loss experienced by the free charge carriers is consistent whether they migrate through the bulk material or the surface of PVB/CdS nanocomposites, especially at lower energy levels. Furthermore, no noticeable bands corresponding to interband transitions of the deposited films were seen at visible photon energy. Additionally, the addition of CdS nanospheres resulted in a reduction in both the volume and surface energy losses of PVB films. Based on the analysis of Figure 11, it is evident that the VELF values exhibit a more significant rise compared to the SELF values in the PVB films doped with CdS nanospheres.
3
Variation of (a) the volume and (b) the surface energy loss functions against photon energy of PVB/CdS nanocomposites. PVB: polyvinyl butyral; CdS: cadmium sulfide.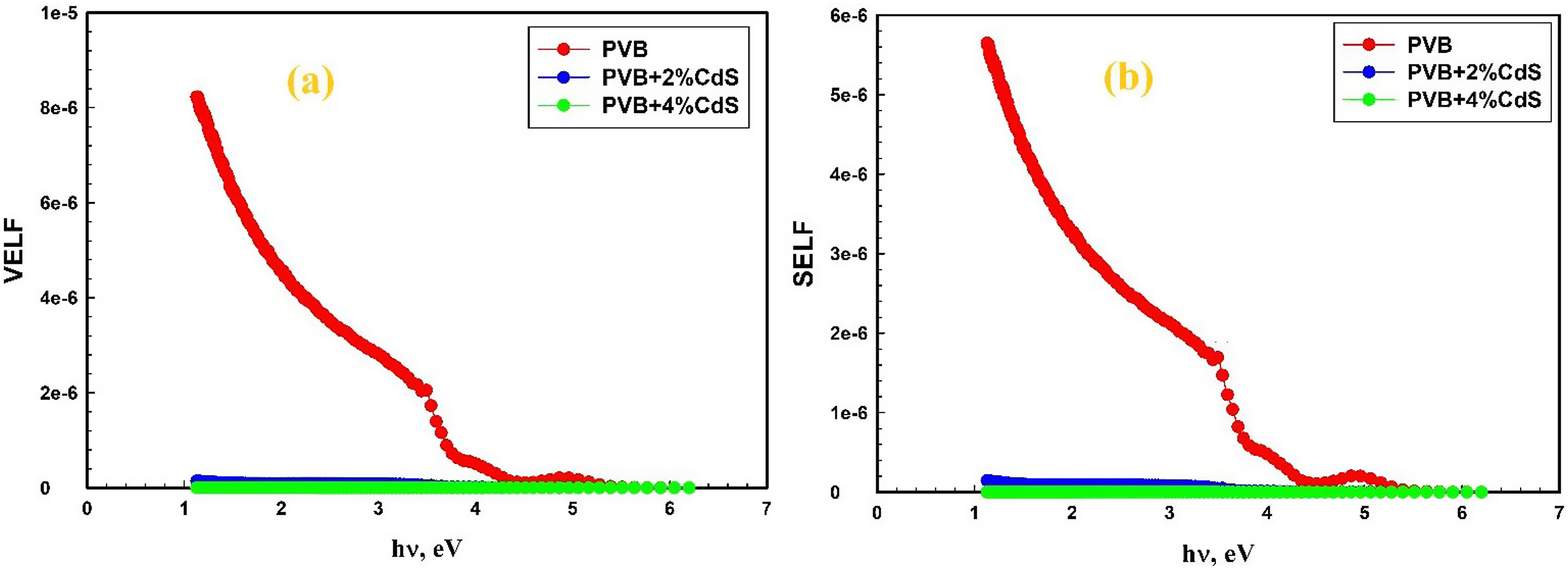
Non-linear optical properties
The linear and non-linear optical susceptibilities are significantly impacted by the electrostatic configuration of molecules within polymer films, which is closely associated with their chemical structure. In addition to optical data storage, optics switching, and the advancement of non-linear optical devices, these parameters are crucial to many applications. Thus, it is critical to comprehend and characterize their linear and non-linear optical susceptibilities to maximize the potential of polymer films in these technological domains. 64
The subsequent equations can potentially be employed to compute the third-order non-linear optical susceptibility (χ(3)) and linear optical susceptibility (χ(1)) of PVB/CdS nanocomposite films
61

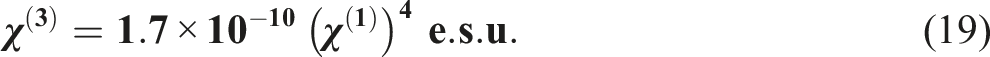
The polarizability of the molecules influences the non-linear refractive index (n2) of a polymer film; consequently, the chemical structure of the polymer molecules can impact the non-linear refractive index (n2). To calculate the non-linear refractive index (n2), the subsequent equation is utilized
65

Figure 12 shows the relations between the variations of the linear optical susceptibility (χ(1)), the third-order non-linear optical susceptibility (χ(3)), and the non-linear refractive index (n2) of PVB/CdS nanocomposite films versus the wavelength (λ). The relations between the variations of (a) the linear optical susceptibility (χ(1)), (b) the third-order non-linear optical susceptibility (χ(3)), and (c) the non-linear refractive index (n2) of PVB/CdS nanocomposite films versus the wavelength (λ). PVB: polyvinyl butyral; CdS: cadmium sulfide.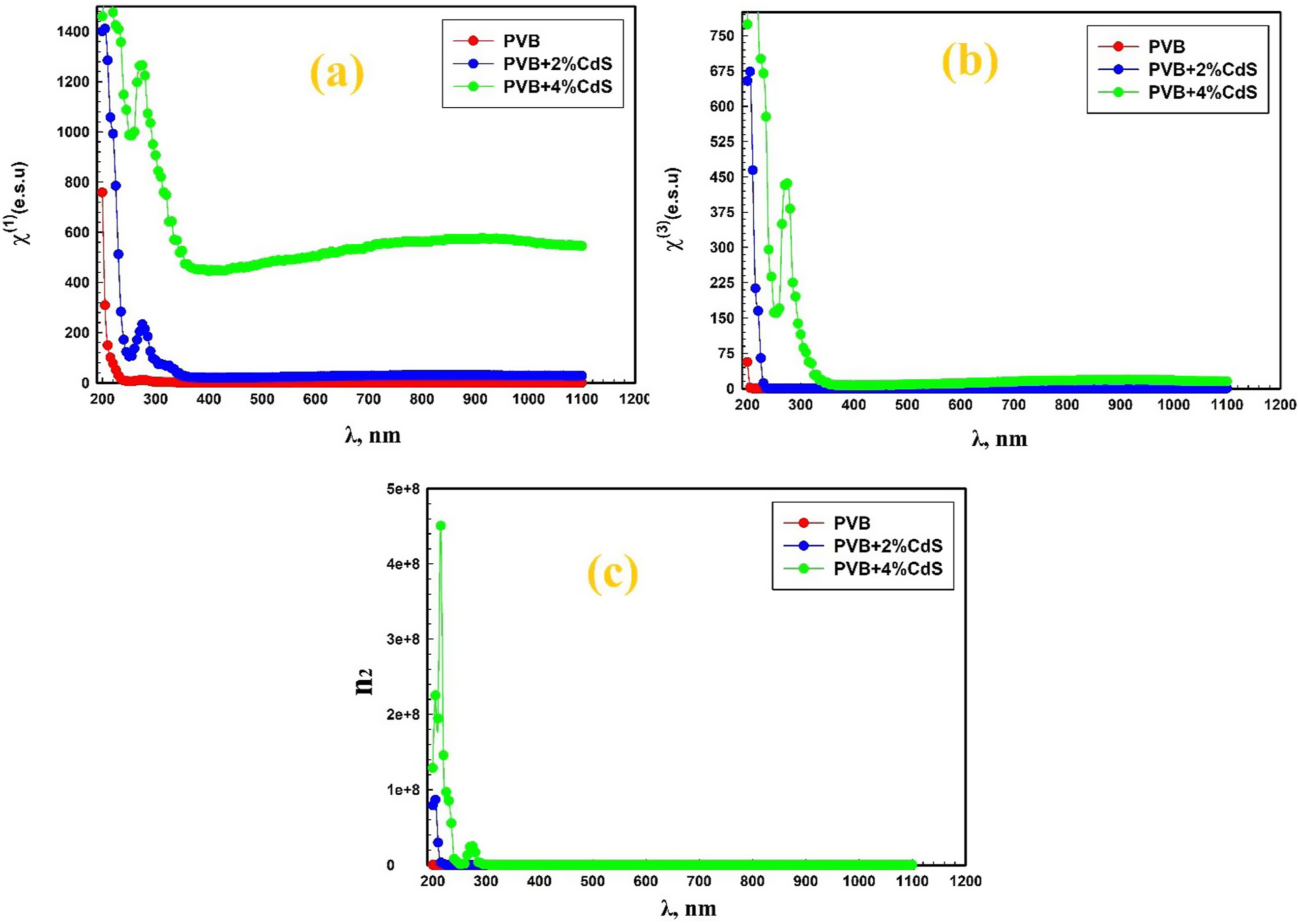
The study reveals that the linear optical susceptibility, non-linear third-order optical susceptibility, and non-linear refractive index of PVB/CdS nanocomposite films tend to increase with the concentration of CdS nanospheres. The improvement can be ascribed to the intense interfacial interactions between the CdS nanospheres and the PVB polymer matrix, resulting in a deformation of the electron cloud around the polymer molecules. The deformation of the polymer enhances its susceptibility to electric fields, which in turn leads to an enhancement of the material’s non-linear optical characteristics. Enhancing the non-linear refractive index of PVB/CdS nanocomposite films has potential for practical use. Enhancing the non-linear refractive index allows for the creation of polymer films with superior optical characteristics, suitable for a wide range of applications. 66 The PVB/CdS nanocomposites demonstrate suitable mechanical properties, high values of the non-linear third-order susceptibility, and non-linear refractive index, indicating their suitability for various non-linear optical and photonic devices.
Compared with our optical results, Vyshakh et al. 67 have investigated the impact of boehmite nanoparticles on the optical characteristics of PMMA nanocomposites for flexible electronic devices. They reported that the gap energy values diminish from 3.08 eV for pure PMMA to 3.0 eV as the boehmite content in the starch increases from 0 to 10 wt.%. Furthermore, Alziyadi et al. 68 showed that the direct band gap value decreases from 5.1 eV to 3.83 eV when the Bi2WO6 percentage in the PVB matrix rises to 4% wt., whereas the indirect band gap drops from 4.1 eV to 2.89 eV. Hiba Kamil Jaafar et al. 69 examined the optical characteristics of PMMA/PEO/SiC/BaTiO3 nanocomposites. They discovered that the direct energy band gap reduced from 3.4 to 1.9 eV, while the indirect band gap reduced from 2.8 to 1.7 eV. According to Al-Shawabkeh et al., 70 the optical energy gap of pure PVC was determined to be 4.0 eV. The study also determined that the optical energy gap of PVC doped with 2.5 wt.% nonannealed ZnO nanoparticles was 3.5 eV, which decreased to 3.03 eV when the concentration of nonannealed ZnO nanoparticles increased to 10 wt.%. Upon investigation, it was found that the optical energy gap of PVC/ZnO increased from 2.89 eV to 3.76 eV when the annealed ZnO nanoparticles were increased from 2.5 wt% to 10 wt.%. This indicates that the crystalline structure of the ZnO nanoparticles was altered due to variations in the annealing temperature. Furthermore, Othmane Rhalmi et al. 32 observed a decrease in the gap energy values from 4.88 eV for undoped starch to 4.66 eV as the Mg-Al layered double hydroxide content in starch rose from 0 to 7 wt.%.
It is clear that our PVB/CdS nanocomposites with low concentrations of CdS spheres have promising optical band gaps and suitable mechanical properties, making them appropriate for flexible electronic devices.
Conclusion
This study incorporated hydrothermally synthesized CdS nanospheres into a PVB matrix through solution casting, forming PVB/CdS nanocomposite films. The characterization techniques employed, including XRD, SEM, EDX, FTIR, and Ultraviolet-visible-near-infrared (UV-Vis-NIR) spectrophotometry, provided valuable insights into the structural and optical properties of the films.
The XRD analysis verified the successful formation of cubic phase PVB/CdS nanocomposite films, while SEM images demonstrated the presence of CdS nanospheres within the film matrix. The FTIR spectra exhibited a shift in band position with increasing CdS concentration, indicating the interaction between CdS and PVB. The UV-Vis-NIR measurements revealed enhancement in the absorbance, a red shift in the absorption edge, and a decrease in the bandgap with increasing CdS concentration.
Moreover, higher CdS concentration in the PVB matrix led to increased refractive index (n) and extinction coefficient (k) values, emphasizing the tunability of the optical properties through CdS incorporation. The improved absorption capacity of the PVB nanocomposite, particularly in the UV-visible spectrum, further supported the potential of CdS nanospheres to enhance the optical performance of the films.
Notably, the increased CdS nanosphere concentration also resulted in enhanced optical susceptibilities, non-linear third-order optical susceptibility (χ(3)), and non-linear refractive index, indicating the suitability of the PVB/CdS nanocomposite films for various non-linear-optical-and-photonic devices.
In summary, we investigate PVB/CdS nanocomposite films and their potential as materials for non-linear optoelectronic applications. The study successfully incorporates CdS nanoparticles into the PVB matrix and observes enhancements in structural, mechanical, and optical properties. This paves the way for further exploration and utilization of these nanocomposites in advanced photonic devices and systems. Our research shows promising results for using PVB/CdS nanocomposite films in optoelectronic applications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
All data generated or analyzed during this study are included in this published article.








