Abstract
Polyvinyl alcohol (PVA)-based hybrid biocomposite films were developed using coconut shell powder (CSP, 5–25 wt.%) as an organic filler and silver nanoparticles (AgNPs, 1–5 mM) as an inorganic filler. The films were fabricated by molding a homogeneous PVA/CSP/AgNPs solution in Petri dishes, and their mechanical and thermal properties were evaluated according to ASTM standards. The synthesis process and the contributions of CSP and AgNPs for enhanced mechanical and thermal stability were analyzed using the Taguchi-based Grey Relational Analysis (GRA) technique. Tensile strength, Young’s modulus, elongation at break, and maximum degradation temperature were assessed using an L25 orthogonal array. Grey regression analysis identified the optimal film composition as 10% CSP and 1 mM AgNPs, which achieved a tensile strength of 38.1 MPa, Young’s modulus of 92.3 MPa, an elongation at break of 18.1%, and a maximum degradation temperature of 315°C. ANOVA indicated that AgNPs and CSP contributed 50.46% and 41.58%, respectively, to overall performance. FTIR, XRD, TGA, and SEM analyses revealed enhanced matrix-filler compatibility, increased crystallinity (23.4%), uniform dispersion of fillers, and improved thermal stability in the optimal film. UV transmittance and water absorption were reduced by 45% and 56.7%, respectively, compared to pure PVA films. Soil burial tests indicated slower degradation of the optimal film due to reduced moisture absorption. The results highlight the suitability of PVA/10%CSP/1 mM AgNPs films for packaging applications, owing to their superior mechanical, thermal, water barrier, UV barrier, and biodegradability properties. The Taguchi-based GRA technique effectively optimized filler combinations for improved performance.
Keywords
Introduction
In modern centuries, the use of polymeric materials has significantly augmented our daily lives. Commonly used polymers like Polyvinyl Chloride, Polyethylene, and Polypropylene are derived from petrochemical products. 1 Globally, around 359 million tonnes of these synthetic polymers are used annually, with over 60% being utilized for packaging purposes. Due to concerns about non-biodegradability and the depletion of petrochemical resources, researchers are exploring alternative solutions for plastic usage, especially in packaging. One potential alternative is biodegradable films made using agronomic leftovers (fillers) and polymers. 2 Bio-films offer advantages such as biodegradability, ease of processing, and lower costs. Various bio-materials like Agar, starch, alginate, soy protein, whey protein, gelatine, cellulose, Chitosan, Polyvinyl alcohol, Polylactic acid, Polyhydroxy alkanoates, and Polypropylene carbonate are currently used as matrices in bio-films. 3 Among these, polyvinyl alcohol (PVA) is gaining consideration owing to its splendid properties, including high hydrophilicity, water solubility, chemical steadiness, biodegradability, hypoallergenic, and ease of film formation. 4 These properties, along with their attractive mechanical and chemical characteristics, make PVA a suitable choice as a matrix material in this work.
Nowadays, scientists and researchers have explored various natural fillers and incorporated them into polymer matrices in the form of short fibers and particulate fillers to enhance the functional properties of thin films, particularly mechanical properties. Rathinavel et al. (2024) 5 utilized pomegranate peel powder in a PVA matrix, and films loaded with 15% filler exhibited a 35% increase in tensile strength. Mishra et. al (2023) 6 analysed the effect of 2.5%,5%, 10% NaOH and H2SO4-treated Moringa Oleifera seed filler on the mechanical , water absorption, and biodegradable properties of PVA biofilms. The 5% acid-treated PVA/MOSF biofilms showed a 12% increase in tensile strength and a 43% increase in tensile modulus compared to pure PVA films. Rathinavel et. al (2024) 7 reported that PVA/PP/AgNPs hybrid films exhibited the high thermal stability (330°C) and maximum tensile strength (25.7 MPa) at 5 mM concentration of AgNPs. Yaqoob et al. (2023) 8 synthesized the silver nanoparticles and grapes peel extracts infused PVA composites. The silver nanoparticles improved the antioxidant property, thermal and mechanical stability of the composite blends. Nanthakumar et al (2020) 9 described that tensile and water absorption properties of PLA based biofims enhanced gradually with the incorporation of sugarcane leaves fibre. Wang et al. (2024) 10 indicated that starch PVA-based films enhanced with varying lignin content, showing improved mechanical, barrier properties due to strong polymer interactions and lignin’s functional groups. Cheng et al. (2021) 11 developed polylactic acid composite films incorporating silver nanoparticles (AgNPs) reduced using mango peel extract to extend the shelf life of strawberries. Gopalakrishnan et al. (2023) 12 utilized the extracts from pomegranate and kinwoo peels to synthesize AgNPs for the modification of cellulose based films.
The Coconut shell filler (Cocos nucifera) has gained consideration due to its extensive availability. As a lignocellulose filler, coconut shell is an agricultural by-product found in abundance across the Asian Continent. It ranks as the fourth most important crop by acreage, following oil palm, rubber, and paddy. Initially, coconut shells had diminutive to no fiscal value, and their dumping was both pricey and environmentally problematic. 13 Lignocellulose fillers, such as coconut shells, offer several advantages over mineral fillers like calcium carbonate, kaolin, mica, and talc. These benefits include cost-effectiveness, renewability, a high strength-to-weight ratio, innocuous, low density, reduced scuff to machinery, and environmental friendliness. 14 The main constituents of coconut shell powder, a by-product of processing coconuts, are lignocellulosic substances such as cellulose, lignin, and hemicellulose. It also comprises ash, of which the main elements are alumina, silicon dioxide, and iron oxide. Despite these advantages, the use of coconut shell powder (CSP) as a filler in biofilm fabrication technology remains underexplored. However, the literature suggests that incorporating coconut shell powder into biomaterials could add value and help reduce environmental pollution. 2 Metal nanoparticles are integrated to enrich the antimicrobial properties of the material. 15 In certain studies, incorporating nanoparticles, particularly silver nanoparticles, has been shown to improve the mechanical properties of the films produced. 16 Silver nanoparticles were specifically selected in this context due to their thermal stability and their effective antimicrobial properties. 17 The addition of silver nanoparticles (AgNPs) in PVA-based biofilms greatly improves the functional properties of the composite by endowing excellent antimicrobial properties, which are essential for use in food packaging, biomedical devices, and wound dressings. Moreover, AgNPs incorporation can enhance the thermal stability, UV resistance, and mechanical strength of the biofilm because of their nanoscale reinforcing effect and good interfacial interaction with the PVA matrix.
Optimization is a crucial technique used to identify the key design factors that can yield optimal results for specific characteristics. 18 Progressive optimization techniques are employed to optimize multi-response characteristics. Currently, various researchers apply multiple optimization methods for engineering and design problems, including Grey Relational Analysis (GRA),19,20 the principal component method, 21 and genetic algorithms, 22 among others. Taguchi-based GRA is a valuable optimization technique for making decisions involving multi-response characteristics, particularly in conditions of uncertain, incomplete, or inadequate data. It describes situations where insufficient data is termed as black and sufficient data as white. One of the core advantages of this technique is its ability to achieve quantitative and qualitative correlations among numerous variables with limited information, by analysing the interrelationships between multiple responses. The responses are consolidated into a single value using the Grey Relational Grade (GRG), which is then treated as a single response in the GRA technique, allowing for the measurement of the degree of correlation between sequences. In recent times, many researchers have employed GRA to optimize input factors with multiple characteristics through GRG. 23 This approach is particularly valuable for addressing complex project challenges, such as determining the optimal composition of materials in the synthesis of composite materials.
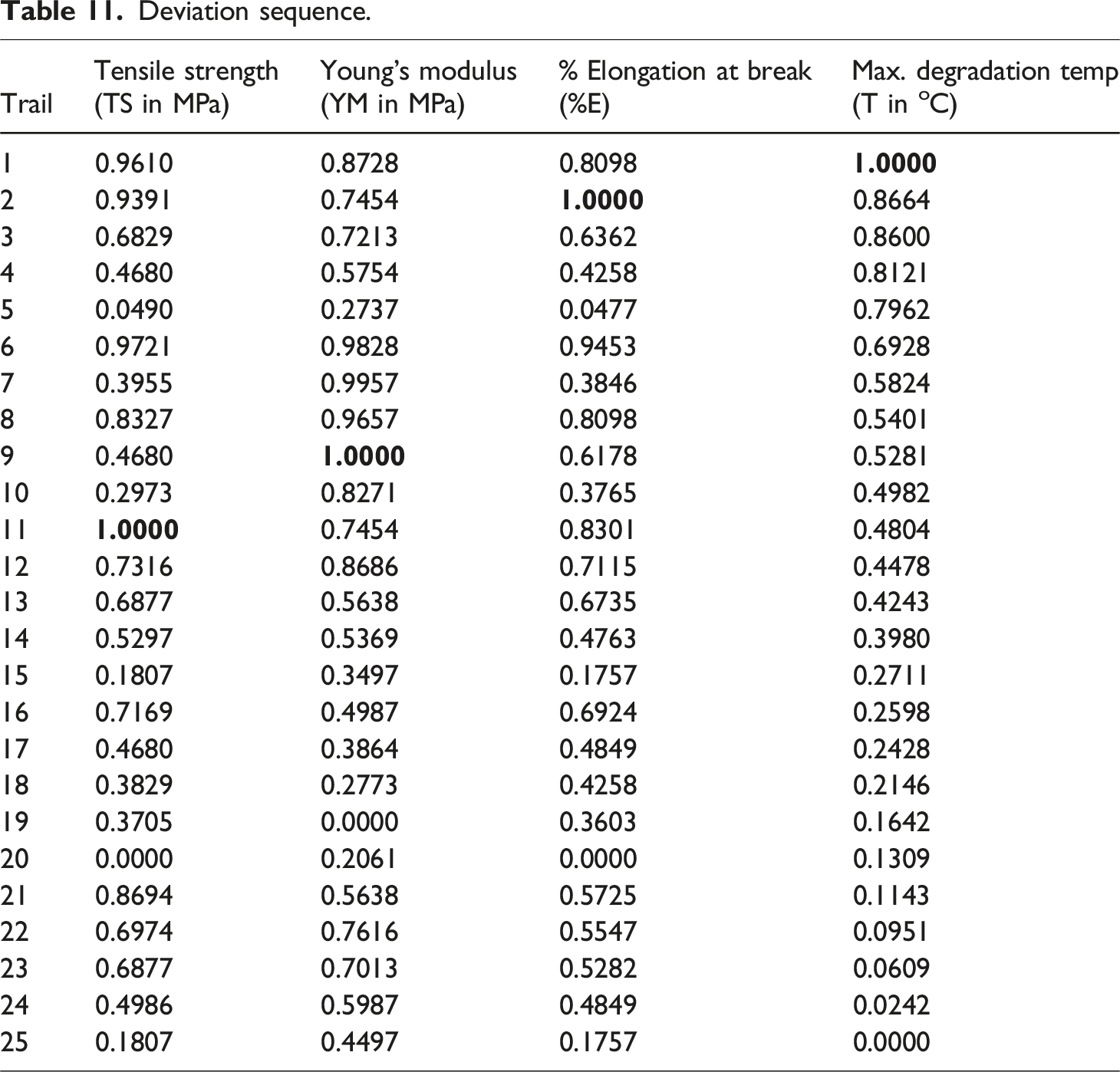
This study intends to fabricate a hybrid bio-composite film using a coconut shell powder (CSP) and polyvinyl alcohol (PVA) matrix, with changeable concentrations of aqueous AgNO3 solution (1 mM, 2 mM, 3 mM, 4 mM, and 5 mM). Further, the Taguchi-based Grey Relational Analysis (GRA) is employed to optimize the various factors involved in the preparation of the biofilm samples. Characterization studies for the optimized data set include surface morphology (FESEM), optical clarity, Fourier-transform infrared (FTIR) spectroscopy, X-ray diffraction (XRD), thermogravimetric analysis (TGA), SEM, water absorption, UV transmittance, tensile testing, and biodegradability testing were done. The Methodology and experimentation of this work are shown in Figure 1. Methodology and experimentation.
Materials and methods
The PVA (99% hydrolyzed, Molecular weight (Mw) = 1, 25,000) is acquired from Sigma Aldrich Chemicals Private Limited, Bengaluru, India. The coconut shells were collected from a local coconut tree garden in Virudhunagar, Tamil Nadu, India. The distilled water and silver nitrate of AR grade were purchased from Nano Wings Private Ltd, Telangana, India.
Preparation of PVA/CSP/AgNPs biofilms
After being gathered from the neighbourhood garden, the coconut shells were cleaned with water to remove any contaminants. After being cleaned, the coconut shells were hammered into tiny pieces and allowed to dry for a couple of days in the sun. A planetary ball mill was used to smash the dry coconut shell fragments for around twenty-four hours. Finally, a standard test sieve ISS: 460-1962 measuring 75 mics (microns was used to filter the pulverized coconut shell powder. The coconut shell powder employed in this study has a particle size of 75 μm. The PVA granules were heated and magnetically stirred with purified water for 3 h (90°C) to create a pure PVA solution. Various proportions of coconut shell powder (5%, 10%, 15%, 20%, and 25%) were added to the pure PVA polymer solution and stirred continuously for 3 hours. Furthermore, varying amounts of AgNPs solution (1 mM to 5 mM) were mixed with the prepared homogeneous PVA/CSP polymer solution and stirred continuously at high speed (1800 r/min) for approximately 36 h.
24
For the process of solution casting, an equal volume of the film-forming solution (PVA/CSP/AgNPs) was cast into levelled Petri dishes of the same size with flat surfaces. The dishes were then positioned on a vibration-free surface to facilitate uniform spreading of the solution, and drying occurred under controlled ambient conditions to prevent gradients or bubbles from forming. The film surface was protected with aluminium foil to prevent the accumulation of dust on it. The solution-cast hybrid composite biofilms were peeled off the Petri plate and desiccated for further processing. Film thickness was measured at several random locations with a digital micrometre, and the average thickness was used. The images of fabricated films is shown in Figure 2. Fabricated PVA/CSP/AgNPs film samples.
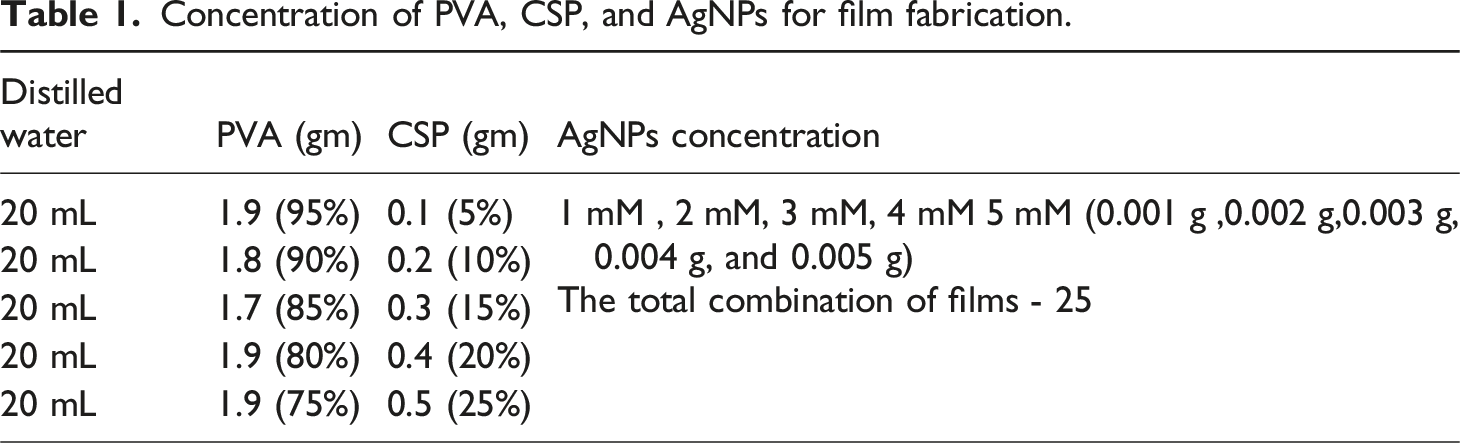
Concentration of PVA, CSP, and AgNPs for film fabrication.
Adhesion mechanism between matrix and fillers
The Figure 3 demonstrates the adhesion mechanism between polyvinyl alcohol (PVA), coconut shell powder (CSP) and Silver nanoparticles (AgNPs). n PVA/Coconut shell powder/AgNPs bio-composite films, the adhesion between the matrix (PVA) and the fillers (CSP and AgNPs) is controlled by a combination of intermolecular and interfacial forces. PVA, having plenty of hydroxyl (–OH) groups along its polymer chain, establishes strong hydrogen bonds with the hydroxyl groups available in the cellulose, hemicellulose, and lignin fractions of the coconut shell powder, thereby increasing matrix–filler compatibility. Covalent bonding in some instances can happen by crosslinking reactions, particularly when the CSP is chemically modified (e.g., esterification). Physical adhesion also occurs due to van der Waals forces. AgNPs, positively charged ions (Ag+), can bind with both PVA and CSP through coordination bonding with lone pair electrons of oxygen atoms on hydroxyl or carboxyl groups. These interactions are further reinforced by hydrogen bonding when AgNPs surfaces are functionalized. Overall, all the mechanisms are responsible for the stable interface, effective stress transfer, and improved dispersion of fillers in the polymer matrix, leading to the improved mechanical, thermal, and antimicrobial properties of the composite. Adhesion mechanism between matrix and fillers.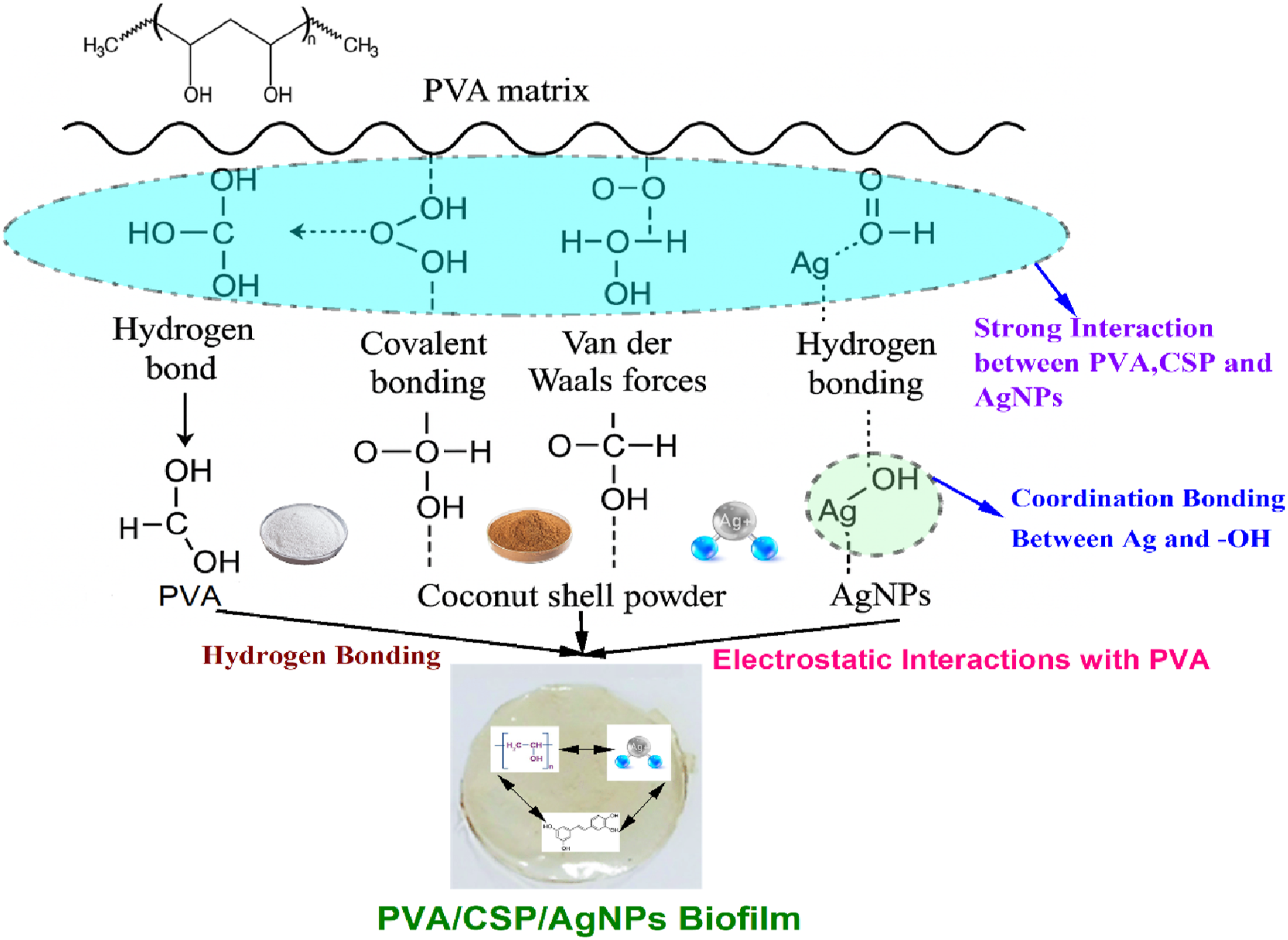
Description of Taguchi-based grey relational analysis
Taguchi-based Grey Relational Analysis (TGRA) is a critical methodology for optimizing multiple responses by analyzing various process parameters. The step-by-step procedure of TGRA is represented in Figure 4. Through TGRA, researchers can identify the optimal parameter combinations that enhance process characteristics. TGRA procedure.
This technique is instrumental in determining the most influential factors affecting the responses by employing systematic optimization processes. TGRA is grounded in the grey system theory fundamentals, which integrates qualitative and quantitative analyses. The core principle of the Grey Relational Grade (GRG) is the transformation of multiple objectives into a single objective, allowing GRG to serve as a unified response measure within Taguchi’s investigational framework.
Experimental design
Factors and their levels.
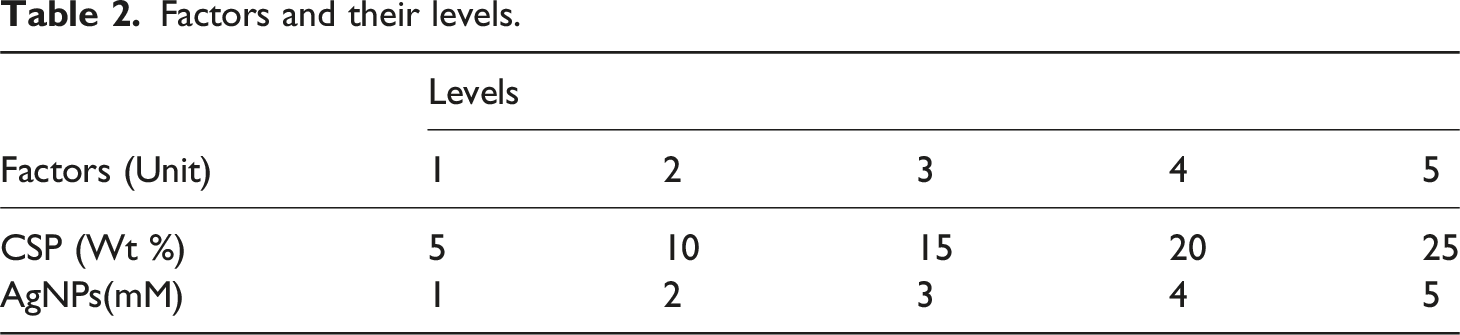
L25 orthogonal array design matrix for formulating biofilm samples.

Testing and characterization techniques
The tensile properties of fabricated hybrid biofilm samples were carried out by a universal testing machine (CMT 6503, Shenzhen SANS Test Machine Co. Ltd, China). The Pure PVA and various combinations of PVA/CSP/AgNPs biofilms are resized to 100 × 10 mm2 samples. In the tests, the initial interval of the fixtures was 30 mm, and the stretch speed was 1 mm/s. The tensile properties of the PVA/CSP/AgNPs biofilms were measured at a strain rate of 50 mm/min. The TGA analysis was performed by using TGA Q50, TA Instruments. The film samples are heated at a rate of 10oC/min up to 600oC in a nitrogen environment.
The grey regression analysis and ANOVA analysis were used to find the optimal composition of organic filler CPS and inorganic filler AgNPs in PVA films through tensile and thermal properties. The optimal biofilm samples were further characterized by FTIR, XRD, and SEM analysis to verify the chemical functional groups, physical structure, and surface morphology respectively.
The FTIR spectra of film samples were recorded using a Perkin Elmer RXI FTIR Spectrometer in the 4000–500 cm−1 range with a 4 cm−1 resolution. X-ray diffractograms of pure PVA and PVA/10%CSP/1 mM AgNPs films were obtained using a Bruker D8 X-ray Diffractometer. The system, configured with a generator setting of 40 kV and 30 mA, aimed to provide an acceptable diffraction pattern, and scanned samples within the 2θ range of 5° to 80°. The crystalline size of films was calculated by Scherrer’s -Equation 1:
Morphological observation was accomplished using scanning electron microscopy (JEOL, JSM-5410LV). The optimal film samples were further tested for UV-Vis analysis, water absorption, and soil burial properties to evaluate their functional characteristics. The UV- Vis transmittance measurements of the optimal film were carried out using a UV-visible spectrometer (PerkinElmer, Lambda 1050). The water absorption and soil burial tests were performed according to ASTM D570 and ASTM D5988. ASTM D570 is a standardized procedure for assessing the water absorption of biofilms, which entails immersing a film in water for a specified duration (4 weeks) and subsequently measuring the weight gain as a percentage of the initial weight. The test should be conducted using distilled water, with the water temperature maintained at 23°C. The standard film size consists of circular samples with a diameter of 50 mm. The specimen size for the ASTM D5988 soil burial test of pure PVA and PVA/CSP/AgNPs biofilms is roughly 2.5 cm × 2.5 cm. Then, using aerobic, non-contaminated soil that has a moisture level of 50–60% of its water-holding capacity, the biofilm specimens are buried 2.5–5 cm below the surface. To support microbial activity, the test is conducted in mud containers that maintain a constant temperature and moisture level, typically between 25°C and 30°C. The pH level of soil is 6 to 8, and the humidity of soil is 60% must be maintained for the entire test. Weight loss and visual changes are used to measure the biodegradation of biofilms.
Result and discussion
Single-objective optimization
Output responses.
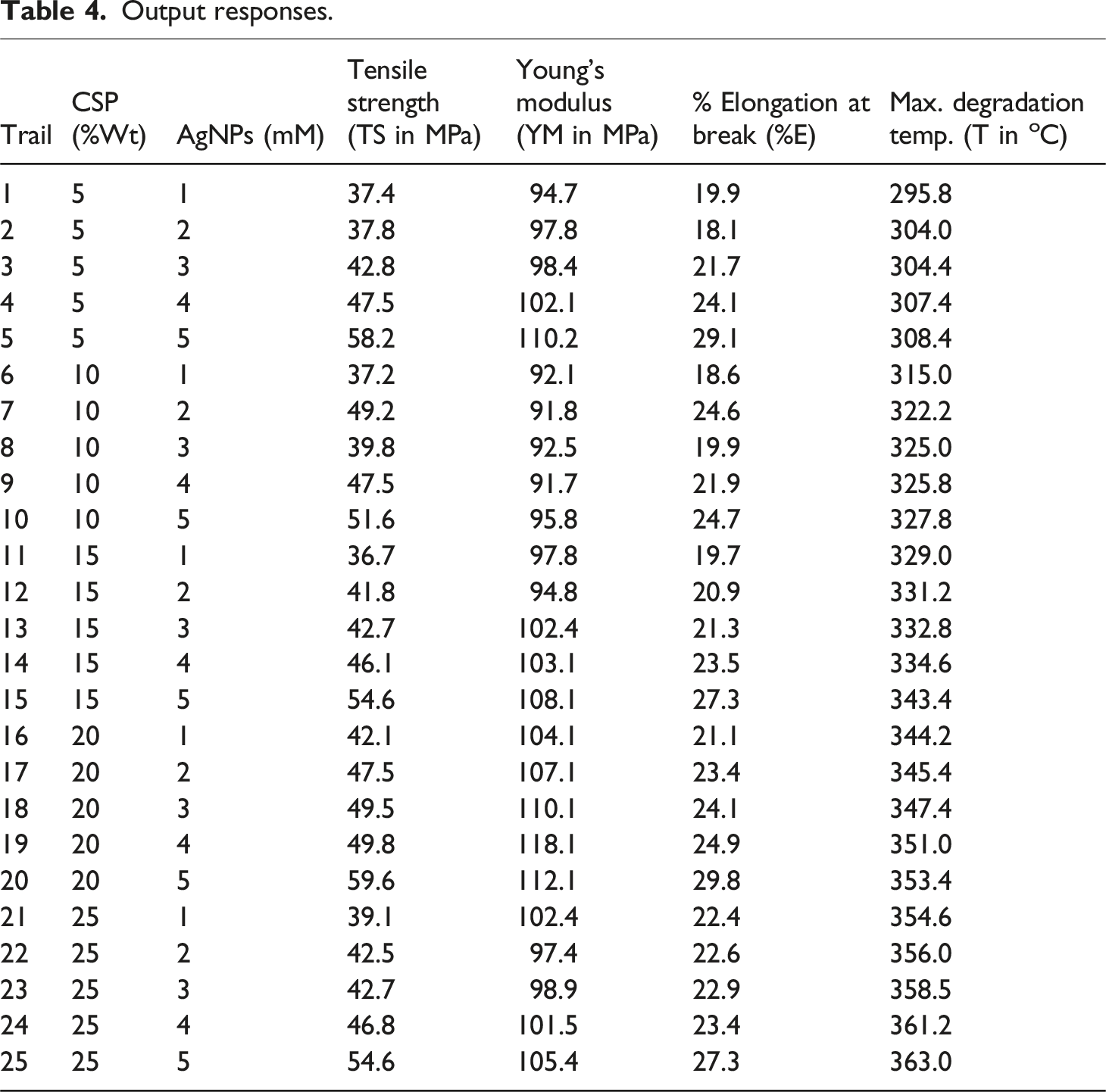
S/N ratio analysis
S/N ratio.
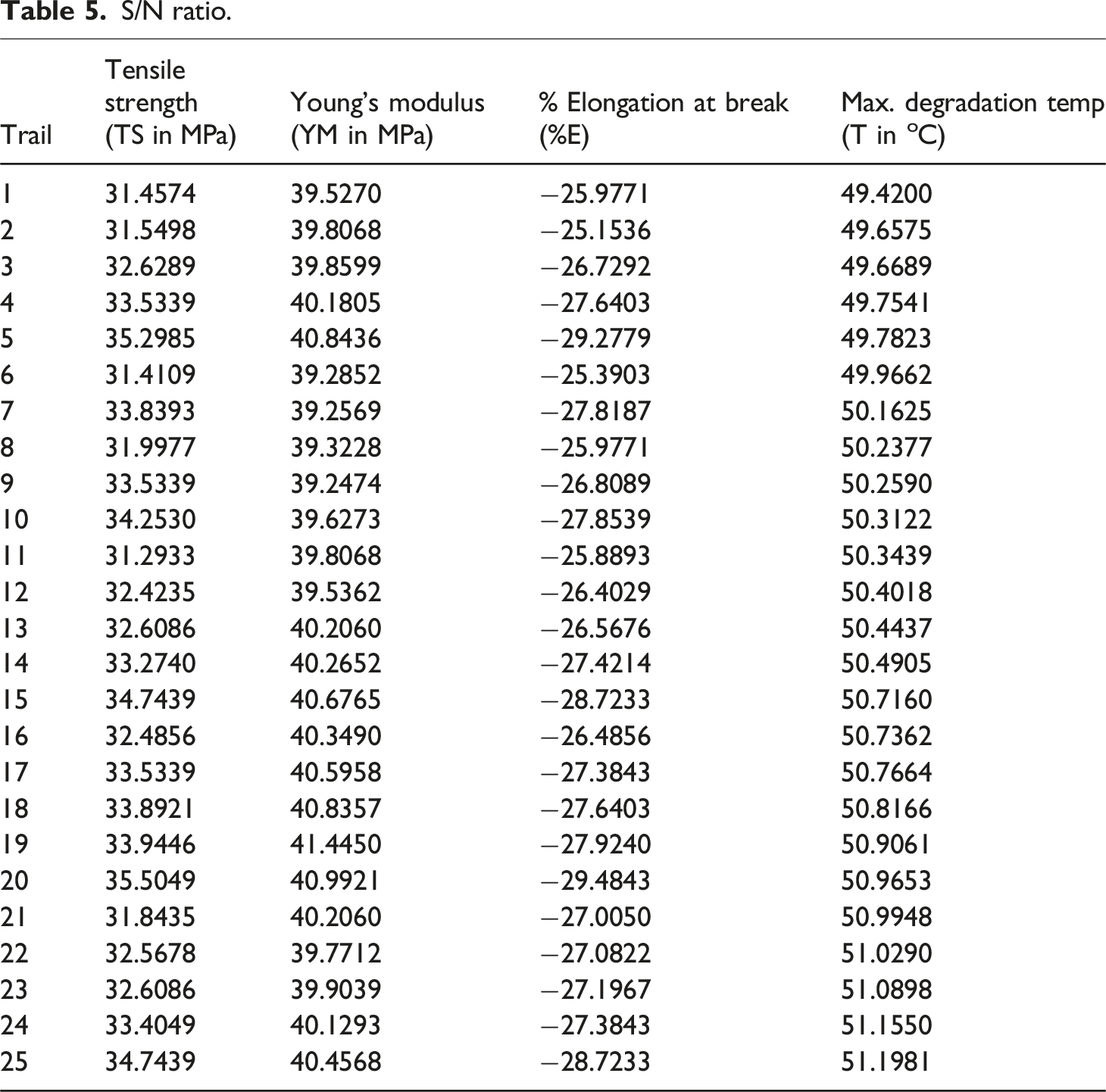
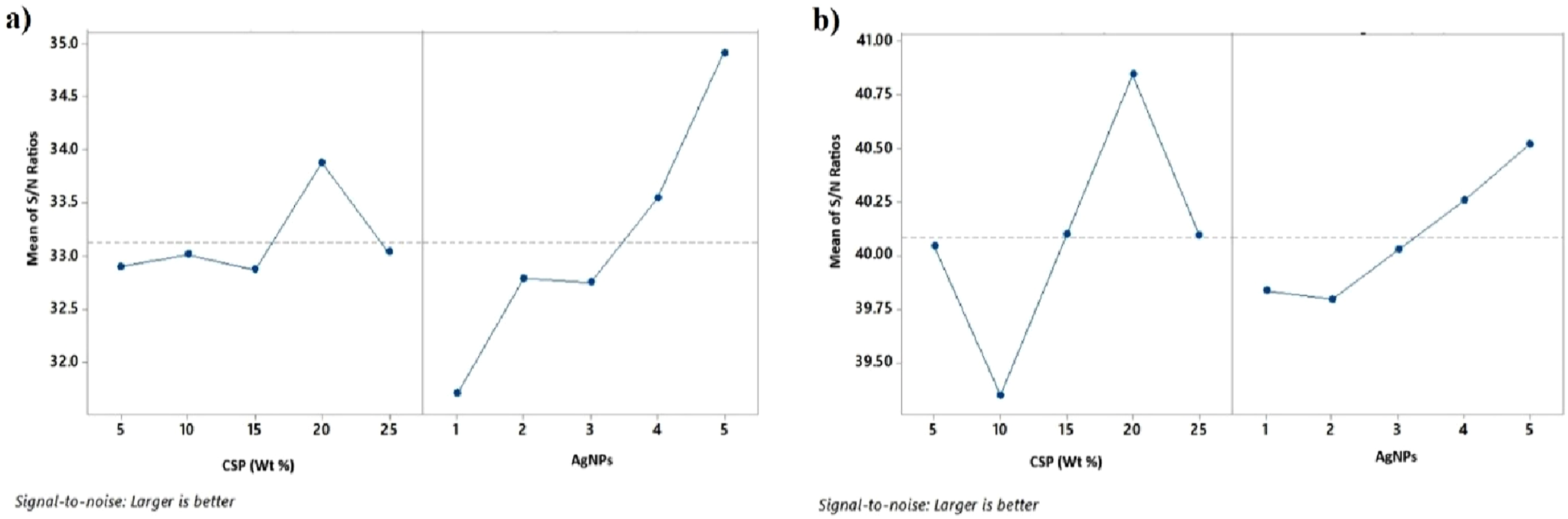
(a) Main effect plot for TS, (b) main effect plot for YM.
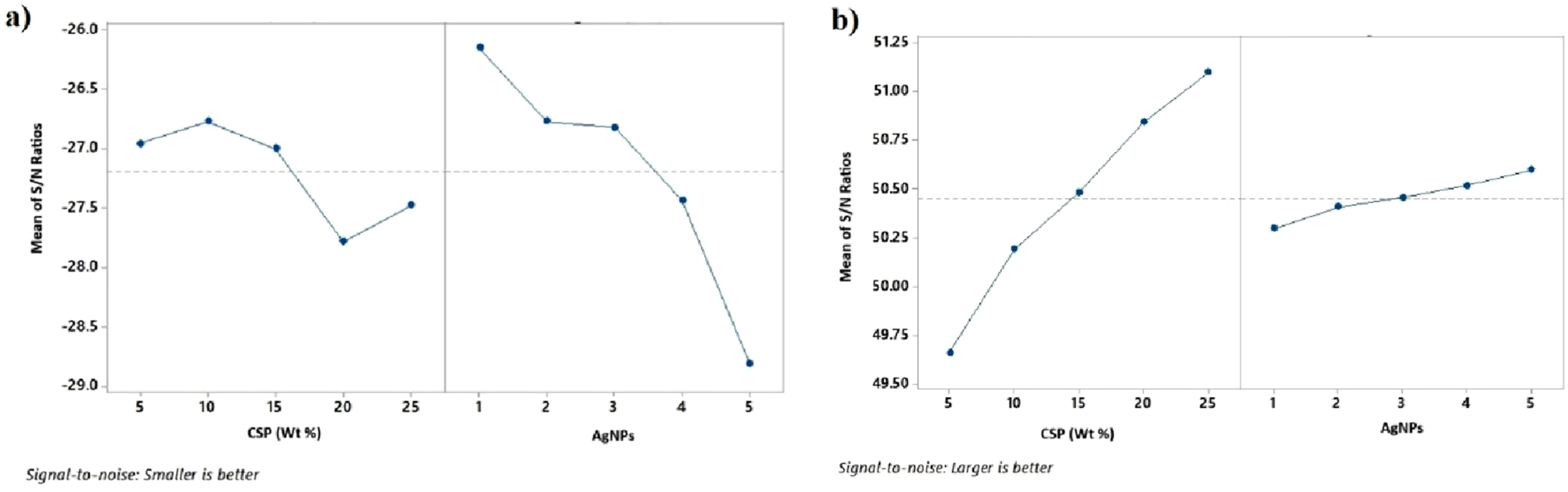
(a) Main effect plot for %E, (b) main effect plot for T.
ANOVA analysis
ANOVA for TS.
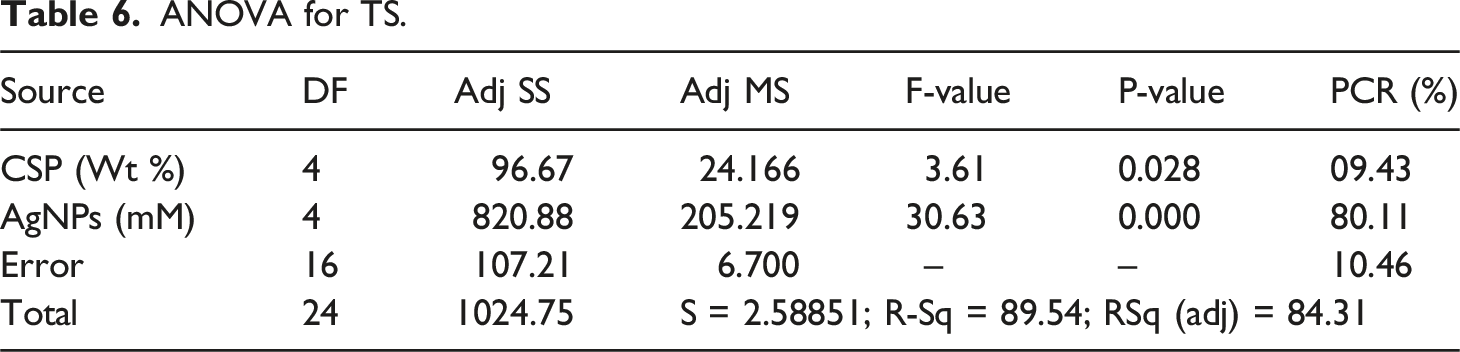
ANOVA for YM.
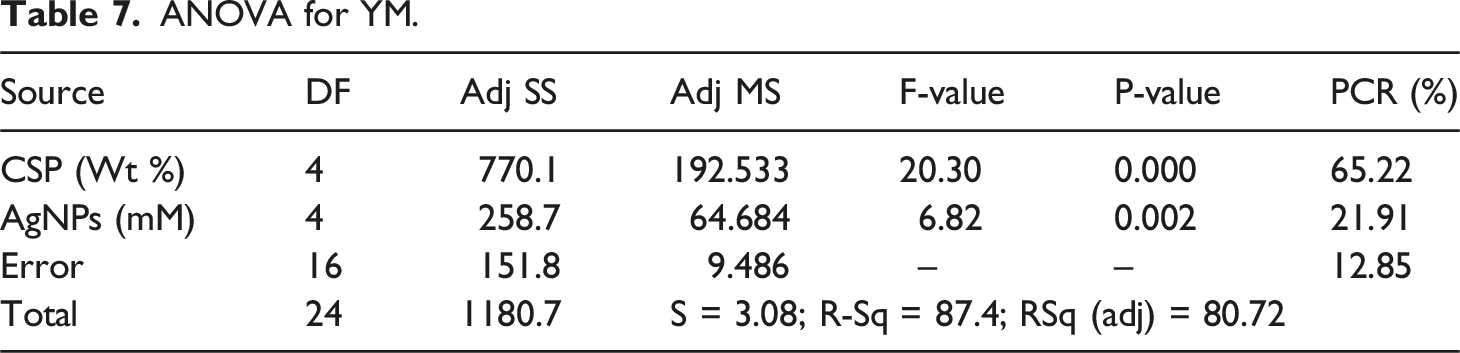
ANOVA for %E.
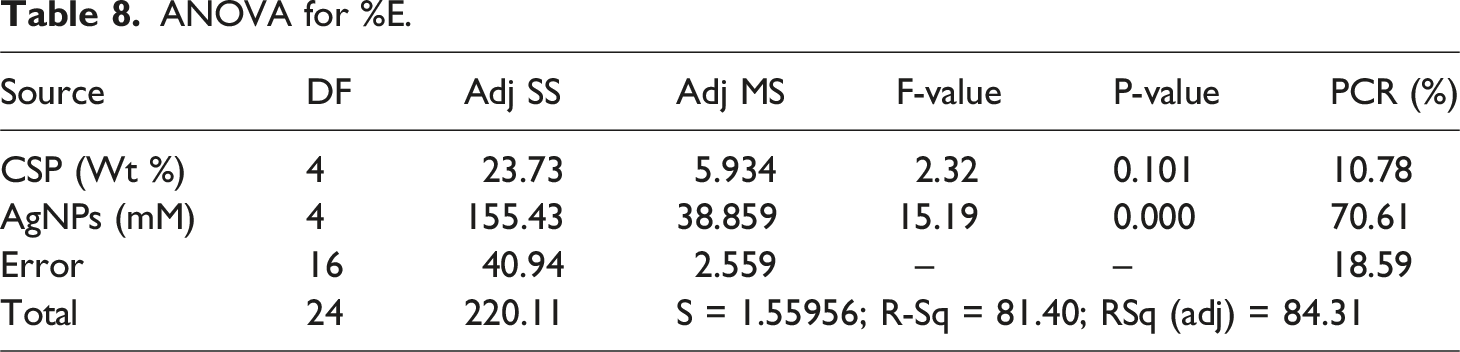
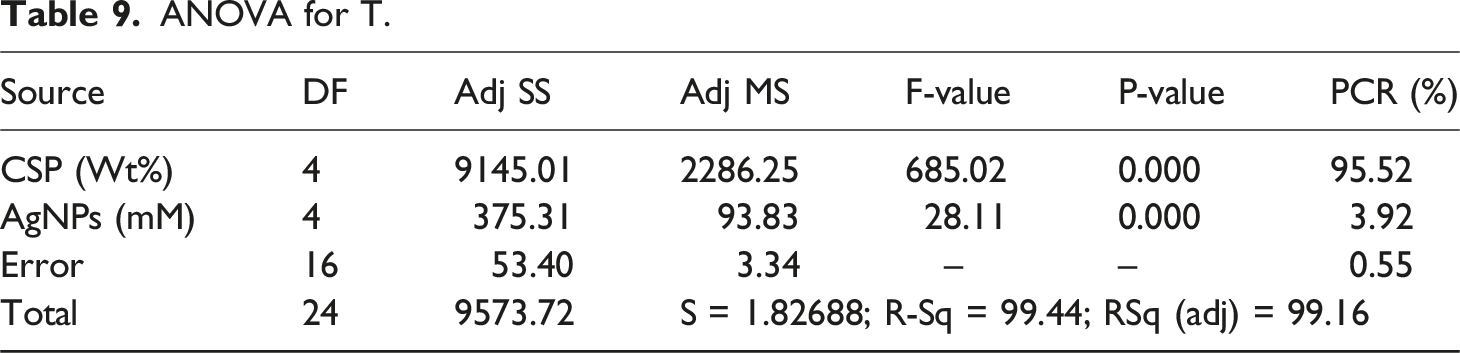
ANOVA for T.
Multi-objective optimization using TGRA
Normalization
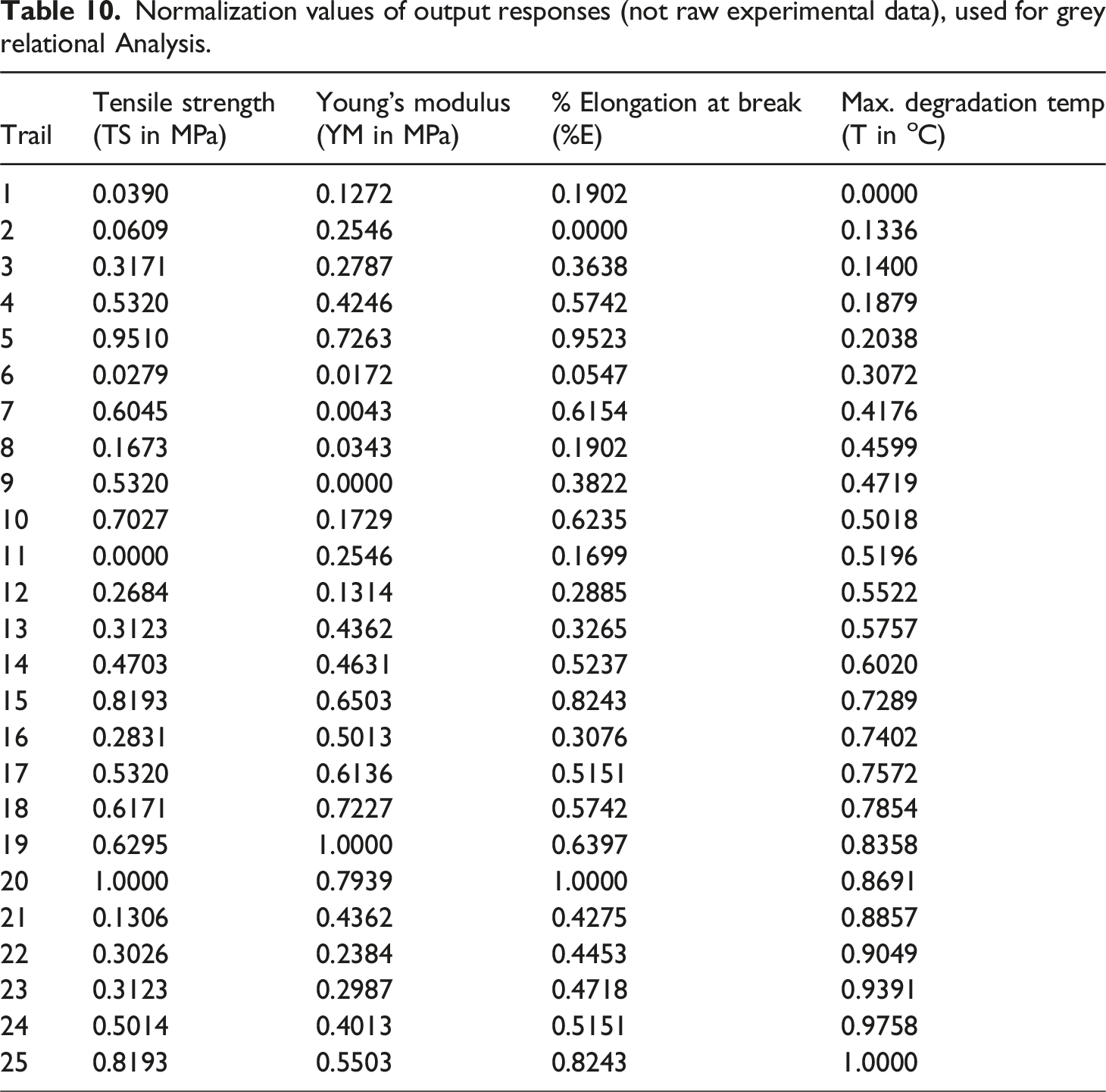
Normalization values of output responses (not raw experimental data), used for grey relational Analysis.
Determination of deviation sequence
Establishment of the sequence of grey relational coefficient
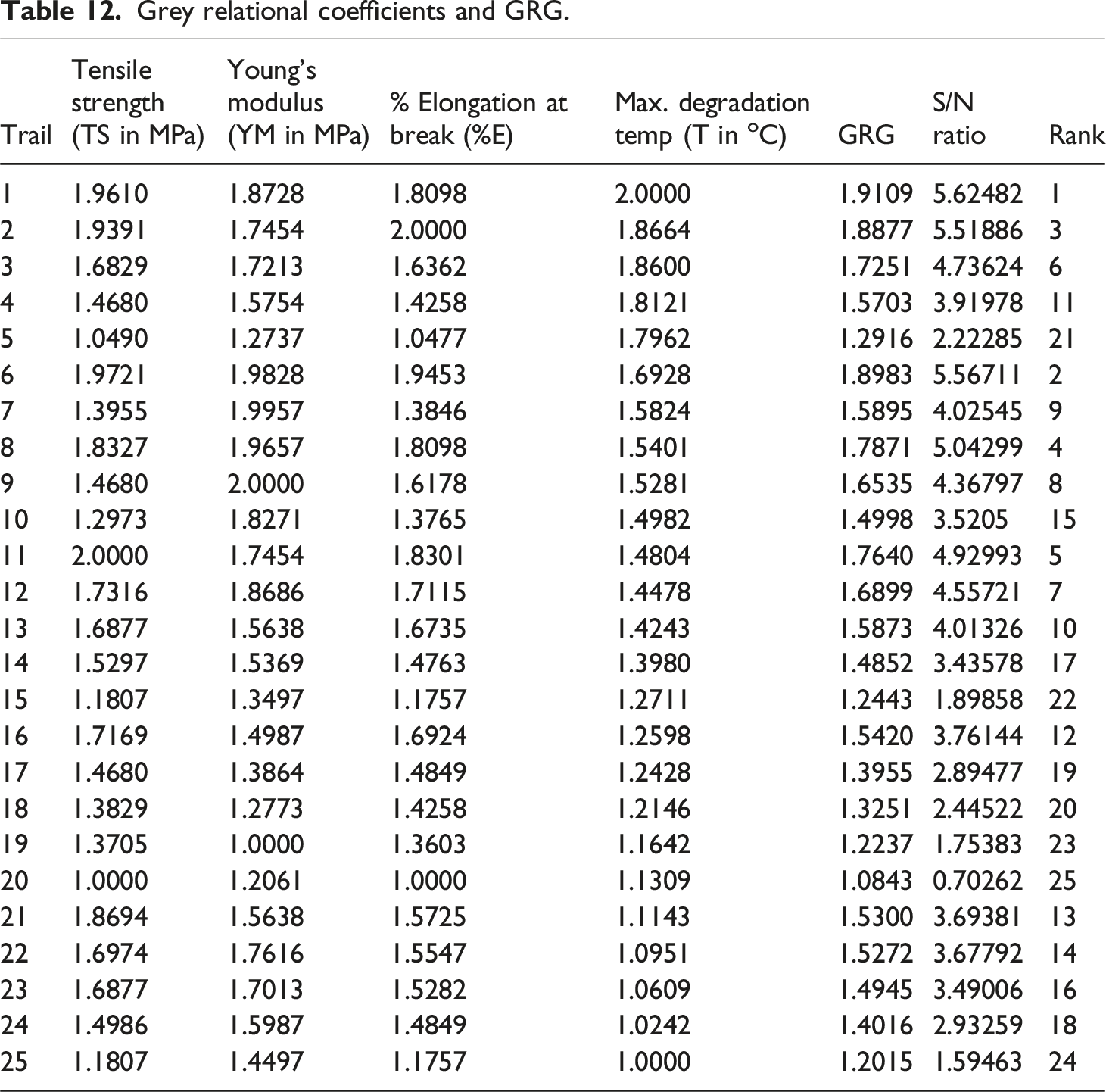
Grey relational coefficients and GRG.
Determination and order of GRG
The Grey Relational Coefficient (GRC) is evaluated to assess the relationship between the reference sequence and the normalized values. A higher GRC indicates that the observed trials are closer to the ideal sequence. The GRC was calculated using formula (7) mentioned in Figure 4, with each experimental trial detailed in Table 12. The grey relational grade (GRG) was evaluated by computing the average GRC using equation (8), which was then used to identify the optimal processing factors.
Conversion from a multi-objective to a single-objective
In the final phase, GRG simplifies multi-objective optimization into a single-objective problem. Following the Taguchi method, the Multi-Objective Evaluation Analysis (MEA) for GRG is used to determine the optimal settings for the design variables. To achieve better performance across multiple characteristics, the “larger-the-better” signal-to-noise (S/N) ratio quality characteristic is applied. The response graphs for GRG, presented in Figure 6, is based on the S/N ratio calculations for each design variable level listed in Table 11. The results, shown in Figure 7, indicate that the optimal settings for the responses to achieve multiple performance goals are CSP of 10% and AgNPs concentration of 1 mM. Response graph.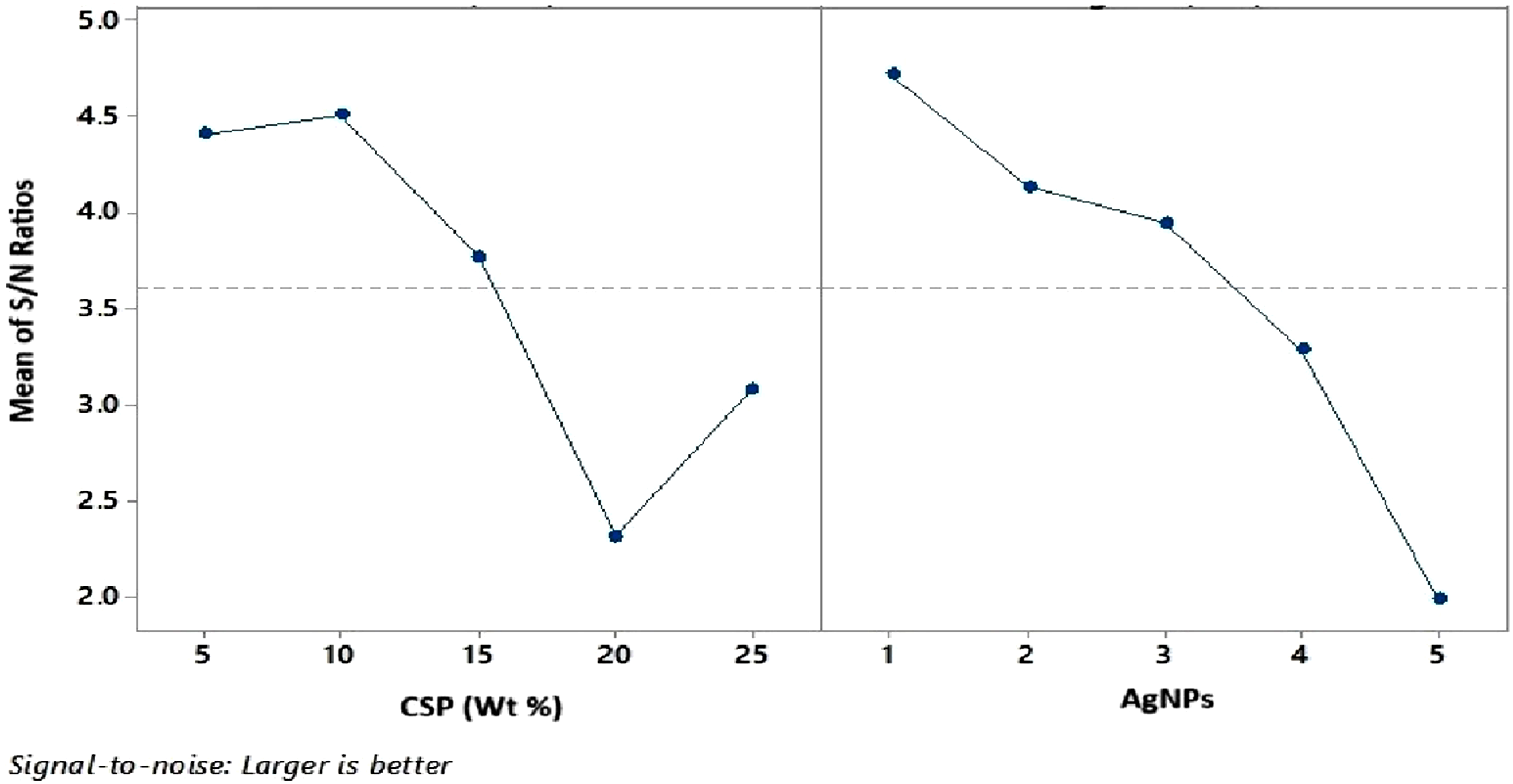
ANOVA analysis
ANOVA for CRG.
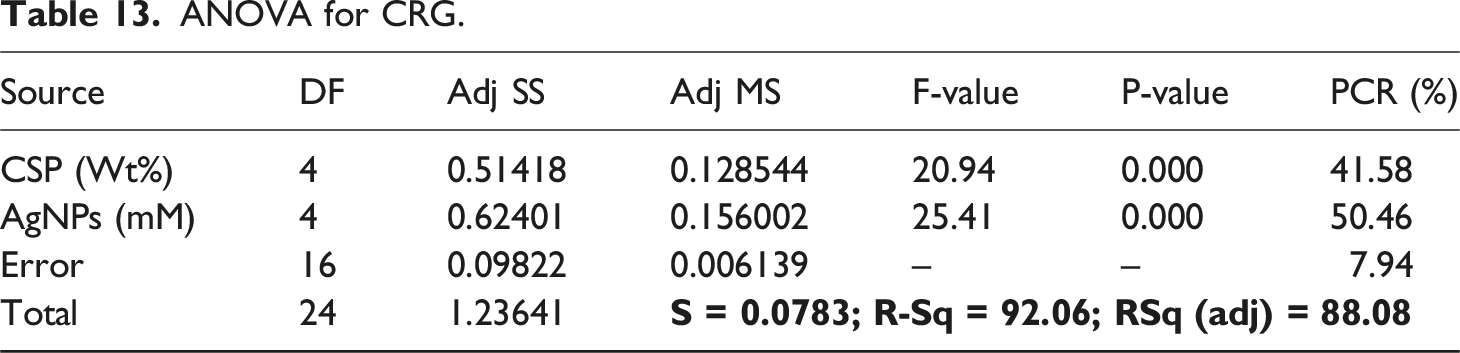
The Percentage Contribution Ratio (PCR) is calculated as: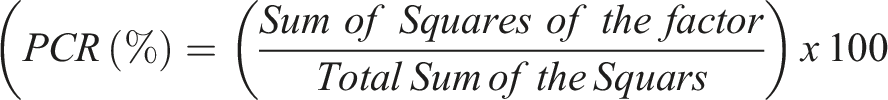
So, for AgNPs: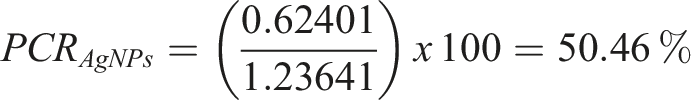
And for CSP: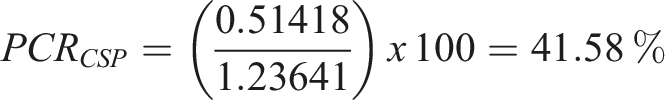
Verification of optimization and confirmation test
Confirmation test results.

FTIR analysis
Figure 8, demonstrates the FTIR spectra of pure PVA, coconut shell powder, and PVA/10%CSP/1 mM AgNPs film (optimal) sample. The neat PVA exhibits a characteristic peak at 3310 cm−1, concomitant with hydroxyl (-OH) bonds. The strong absorption peak absorbed at 2970 cm−1 corresponds to C-H stretching. The concentration peaks at 1430 cm−1 and 1285 cm−1 were indorsed to the CH2 bending motion of the methylene and the C-O stretching of aromatic ester, respectively. The characteristic peak of pure PVA at 880 cm−1 corresponded to the C = C bending of an alkene. The FTIR peaks of coconut shell powder show broad characteristics peaks between 3500 cm−1 – 3200 cm−1, attributed to –OH stretching of hydroxyl bonds and the absorption at 2860 cm−1 corresponds to C-H vibrations.
28
The peaks at 1670 cm−1 and 1014 cm−1 observed in CSP were ascribed to the C = C stretching of alkene and CO-O-CO stretching, respectively. The FTIR peaks of PVA/10% CSP/1 mM AgNPs biofilms and their significance is shown in Table 15. FTIR spectra of PVA, CSP, and PVA/10%CSP/1 mM AgNPs film (optimal) sample. FTIR spectra and implications of PVA/10%CSP/1 mM AgNPs film (optimal) sample.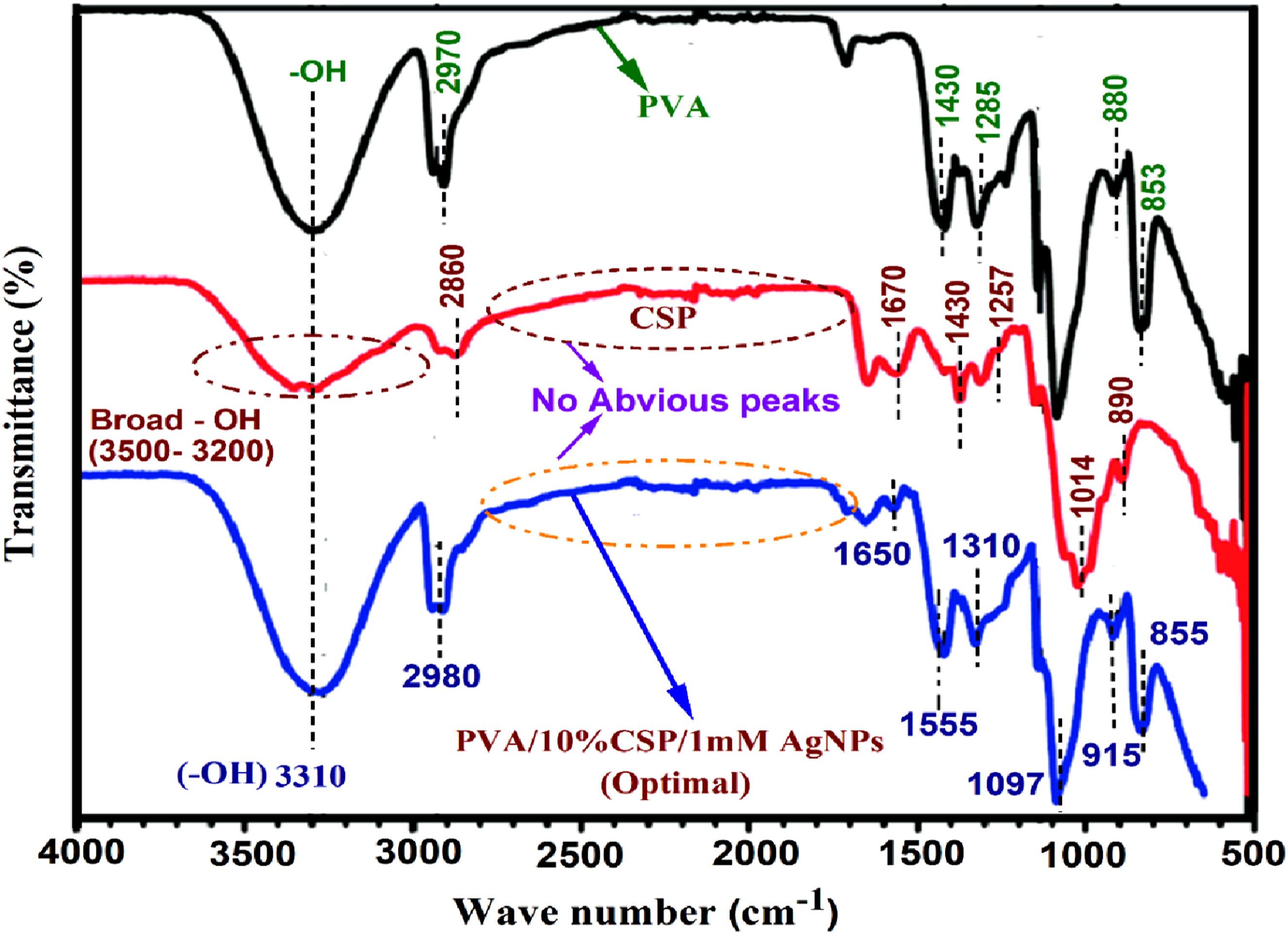
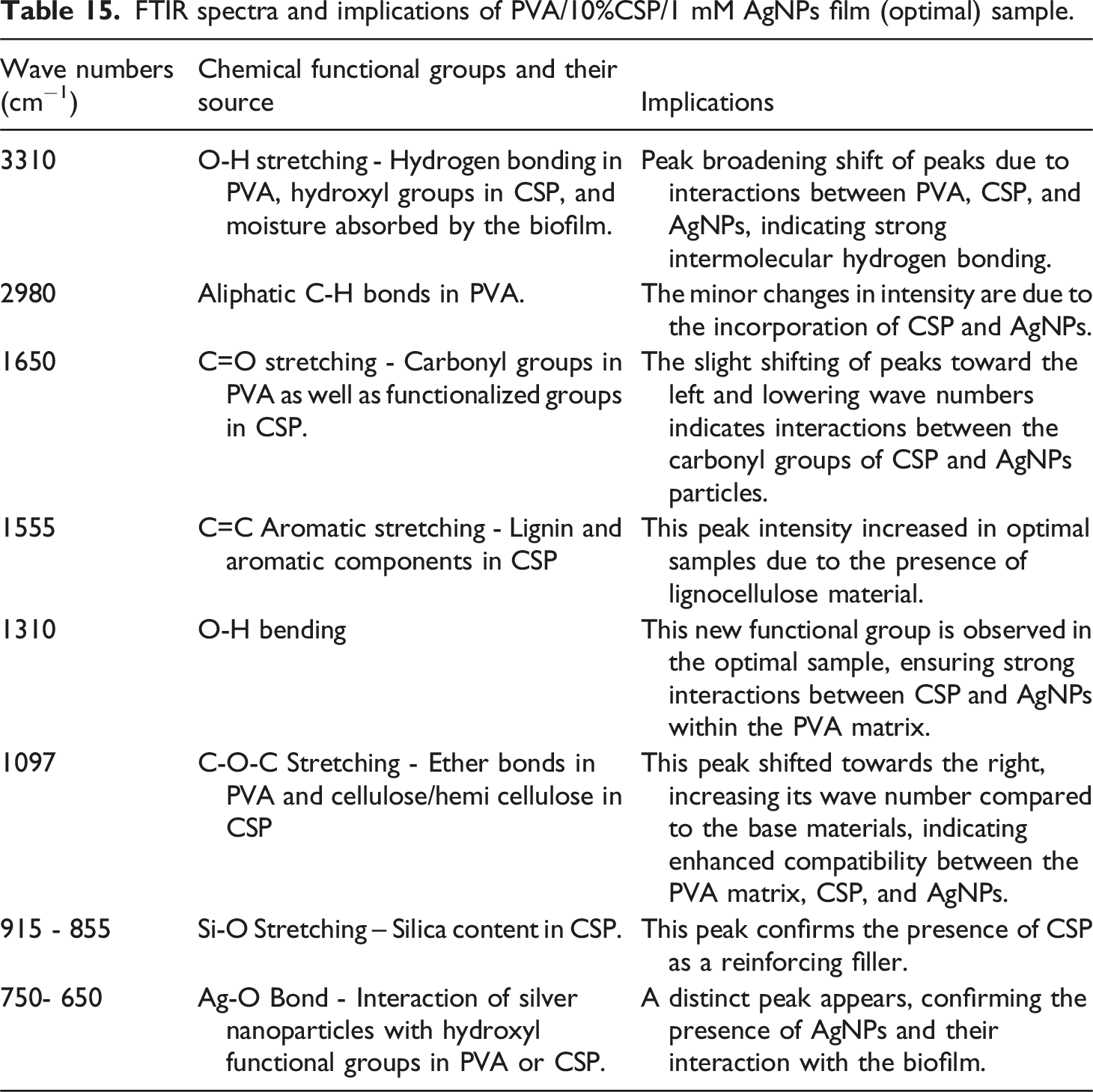
The FTIR analysis of the optimal film sample is summarized in Table 15. It shows robust hydrogen attachment between PVA, CSP, and AgNPs, causing shifts in the O-H and C=O regions. Peaks from lignin, cellulose, and hemicellulose confirm the presence of CSP, while new Ag-O peaks and changes in PVA and CSP peaks confirm the incorporation of AgNPs.
XRD analysis
Figure 9 displays the XRD patterns of PVA, CSP, and PVA/10%CSP/1 mM AgNPs film (optimal) sample. When PVA is in its pure form, it has a semi-crystalline structure with crystalline and amorphous domains. The XRD patterns of the pure PVA, which usually contain a sharp diffraction peak at approximately 19.21° (2θ), belonging to the (101) crystal plane of the crystalline phase, along with a broad hump between approximately 24° and 30° (2θ),
29
corresponding to the amorphous region, are depicted in Figure 8. Disordered molecular chains can give rise to multiple glass transition events, including a broad hump, indicating the amorphous character of PVA. Coconut shell powder is mostly amorphous material including lignin, hemicellulose, and amorphous cellulose, which is shown in an XRD pattern that is observed as a broad hump. XRD patterns of PVA, CSP, and PVA/10%CSP/1 mM AgNPs film (optimal) sample.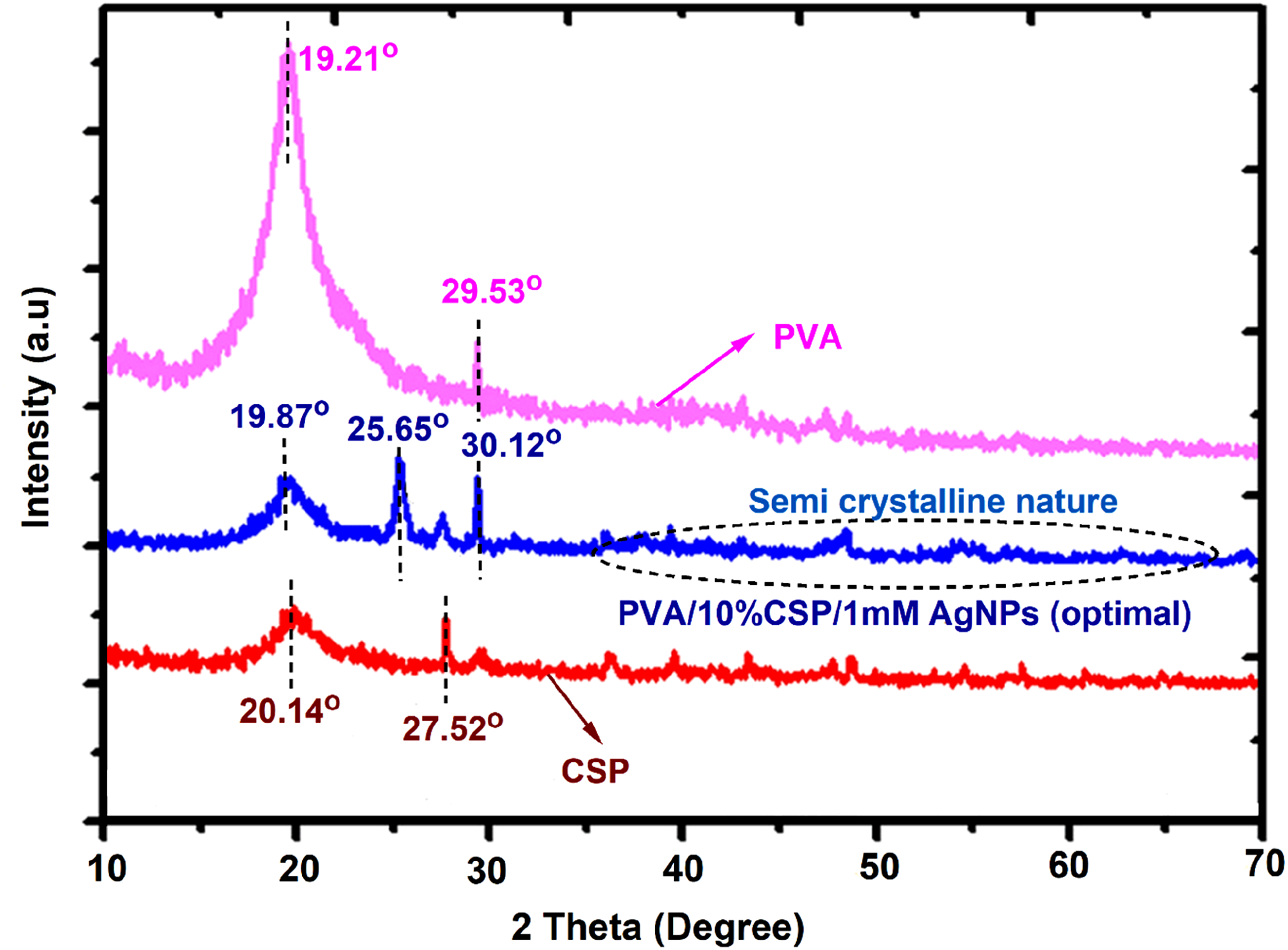
Crystalline peaks are observed by 20.14o and 27.52o ° (2θ) in a smaller area, which corresponds to the crystalline section of cellulose in the coconut husk powder. 30 The respective crystallinity of coconut shell powder and pure PVA is 32.9% and 14.3%. In addition to this CSPs were also integrated into the PVA matrix, resulting in an improvement in the crystallinity of PVA. The XRD peak variations on the addition of 1 mM AgNPs particles as shown: The characteristic sharp peak of native PVA around 19.21° (2θ), corresponding to (101) planes, is observed and is enhanced, indicating partial disruption of the crystallinity of PVA due to CSP and AgNPs addition. The optimal sample shows the crystalline peak at 19.87o due to filler interactions. The shift of PVA’s crystalline peak reflects strong hydrogen bonding and molecular interactions between PVA, CSP, and AgNPs, disrupting the regular arrangement of the PVA chain. A broad peak at 25.65o attributed to the cellulose interactions of CSP was slightly broadened and shifted due to interactions with AgNPs. Furthermore, the new sharp peaks around 30.12° (2θ) and possibly at 41°, 48°, and 58° (2θ) correspond to the (111), (200), (220), and (311) planes of silver nanoparticles, confirming their presence and successful integration. 31 The sharp peaks associated with AgNPs indicate their crystalline nature and successful dispersion in the biofilm matrix.
The crystalline size of pure PVA and Coconut shell powder is 13.5 nm and 27.5 nm. The optimal film sample (PVA/10%CSP/1 mM AgNPs) shows a crystalline size of 32.8 nm. The increase in crystallite size within the PVA/10%CSP/1 mM AgNPs hybrid composite results from the nucleation effect of AgNPs, which promotes crystal development in the PVA/CSP matrix. Silver nanoparticles serve as heterogeneous nucleation sites that facilitate the alignment and ordering of polymer chains, as well as the organization of CSP particles during film making. The significant interfacial contacts between AgNPs and the polymer matrix augment structural stiffness, reducing defects and improving crystallinity.
The overall pattern shows the integration of CSP and AgNPs into the PVA matrix, creating a hybrid material with modified crystalline and amorphous characteristics. These interactions enhance the mechanical strength, thermal stability, and functional activity of the fabricated biofilms.
Thermal analysis
The TGA and DTG images of PVA, CSP, and PVA/10%CSP/1 mM AgNPs film (optimal) samples are shown in Figure 10(a) and (b) respectively. From Figure 10(a) the TGA plot of pure PVA shows three stages of thermal decay and it is initiated with a temperature range of 60oC to 180oC. The next stage of ruin starts at 250oC and ends at 370oC. The % weight loss of PVA films is 10% and 65% during the first and second stages of degradation. The third stage of weight loss occurs between 370oC–550oC. The absorbed moisture, non-cellulosic elements, and foreign matter are degraded in successive stages of thermal degradation of pure PVA. The same weight loss pattern is also exhibited for coconut shell powder. The maximum weight loss of CSP occurred between 270oCand 380oC owing to the deprivation of cellulose, hemicellulose, and lignin components. The Pure PVA and CSP possess 12% and 38% residue char respectively. The addition of CSP and AgNPs to the PVA matrix does not significantly influence the thermal behavior of the optimal film sample, which also undergoes three distinct stages of thermal degradation.
17
The thermal stability of the optimal film sample increased, with weight losses during the successive stages of the thermal degradation process recorded as 6.1% (moisture and volatile components), 64.4% (polymer constituents), and 13.2% (charred materials and additives), respectively. The presence of AgNPs contributes to improved thermal stability by restricting polymer chain agility and enhancing the thermal resistance of the composite. A residual mass may remain at the end of the analysis, indicating the presence of thermally stable inorganic components, such as silver nanoparticles.
32
(a) TGA plots, (b) DTG plots of PVA, CSP, and PVA/10%CSP/1 mM AgNPs (optimal) film sample.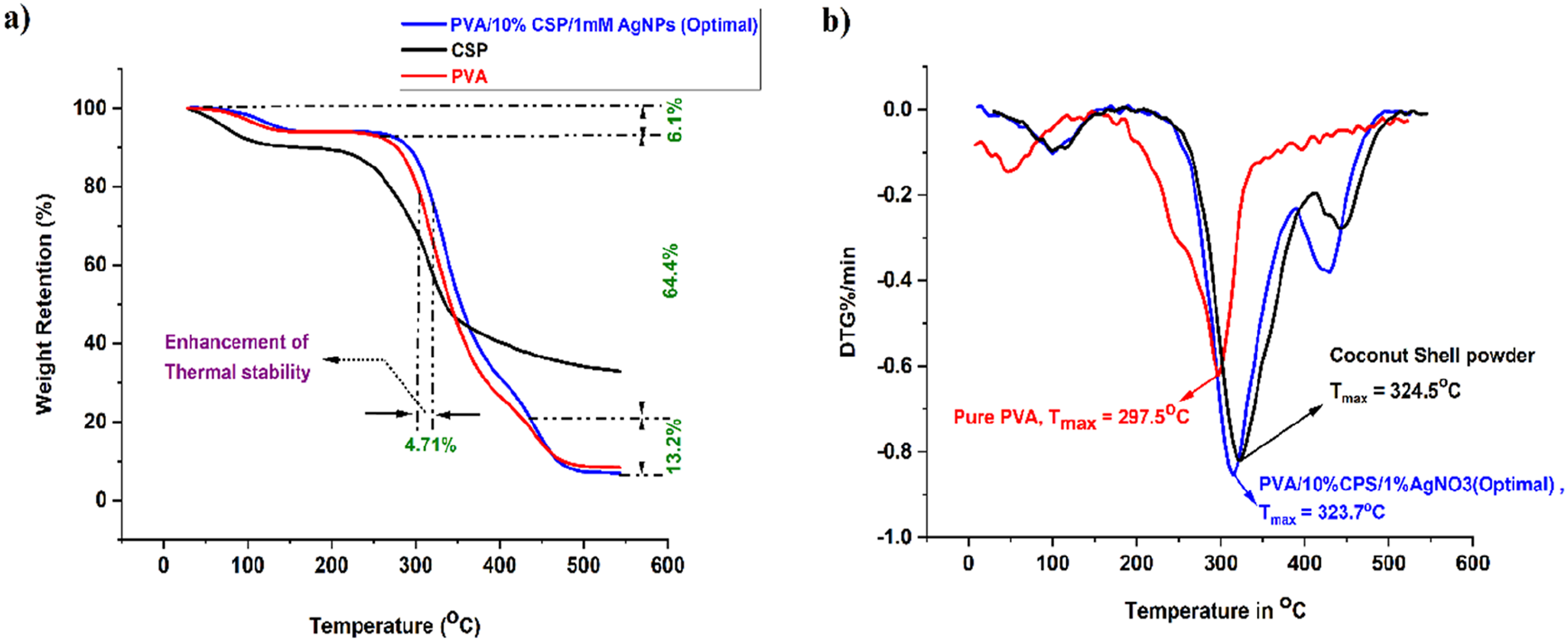
The DTG plots of base materials and optimal film sample are shown in Figure 10(b). The maximum degradation temperature for the Pure PVA and PVA/10%CSP/1 mM AgNPs (optimal) film sample is 297.5oC and 315.6oC respectively. The Tmax for coconut shell powder is 324.5oC. The DTG curves of the optimal sample ensure the shifting of degradation temperatures slightly higher (297oC to 315.6oC) as well as reducing the peak intensity of weight loss, signifying enhanced thermal stability. DTG curves confirm that the incorporation of 10% CSP and 1 mM AgNPs improves the thermal stability of the PVA biofilms, making them suitable for applications requiring controlled thermal resistance.
SEM with EDX
The surface morphology of films was used to analyze the matrix-filler interactions and the compatibility of inorganic nanoparticles AgNPs with polymer matrix. Figure 11(b) and 9(c) showed the SEM and EDX images of the optimal (PVA/10%CSP/1 mM AgNPs) film sample. The SEM image (Figure 11(a)) of pure PVA shows that the surface is very smooth and free of any impurities. The SEM images of the optimal film samples, highlighting the improved compatibility of organic and inorganic fillers within the polymer matrix. The AgNPs particles are evenly distributed on the film surface and might act as water and UV barriers while the film is exposed to the functional environment. The EDX image of the optimal film sample (Figure 11(c)) shows the elemental distribution and confirms the existence of AgNPs on the film surface. The highly compatible PVA and CPS, combined with the AgNPs film system, exhibited superior mechanical, thermal, water barrier, and UV barrier properties in the proposed optimal film sample. (a) SEM image of pure PVA film, (b) SEM image of PVA/10%CSP/1 mM AgNPs (optimal) films, (c) EDX plot of PVA/10%CSP/1 mM AgNPs (optimal) film.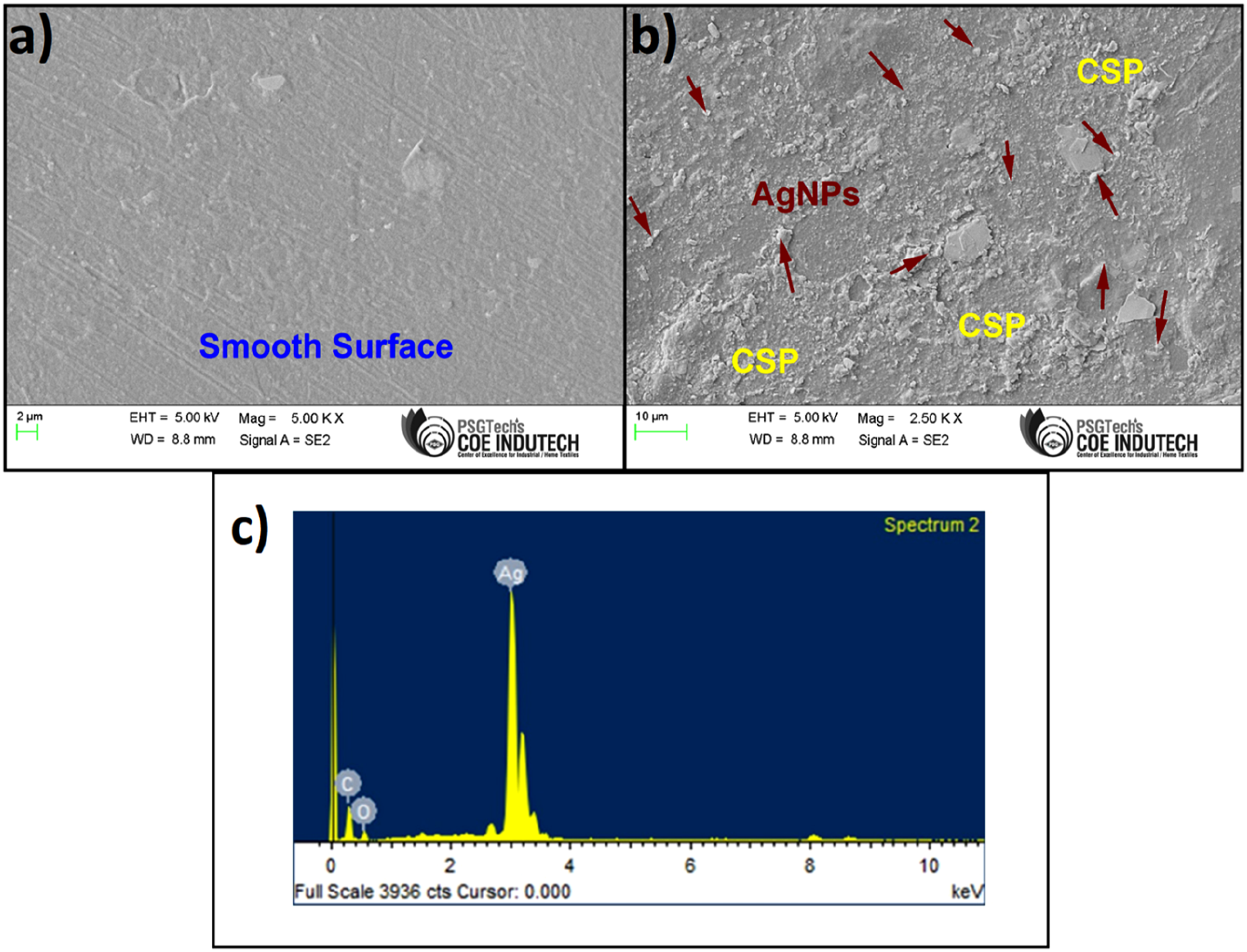
UV-transmittance
Figure 12(a) shows the UV Transmittance of pure PVA film and the optimal film sample. The films are checked with different wavelengths of UV rays. Pure PVA films are UV barrier and more transparent. Since it has little interaction with UV light and is transparent, pure polyvinyl alcohol (PVA) transmits most UV radiation in the range of 200–400 nm. PVA films allow 70% of UV rays to pass through. Coconut shell powder is a natural, lignocellulosic material that contains high amounts of lignin and phenolic compounds which to some extent can absorb and scatter UV rays.
33
CSP incorporation imparted some UV-absorbing properties to the composite film. Surface plasmon resonance (SPR) of silver nanoparticles (AgNPs) is characterized by considerable absorption and scattering in the UV band.
34
AgNPs play a crucial role in enhancing the UV-shielding properties of PVA-based hybrid films and are highly efficient in reducing UV-ray transmission. The inclusion of CSP and AgNPs significantly reduces (45%) UV-ray transmission as compared to pure PVA, especially in the UV-A (320–400 nm) and UV-B (280–320 nm) ranges. The film is appropriate for applications demanding strong UV resistance because of the collective influence of 10% CSP and 1 mM AgNPs, which improves UV protection. (a) UV transmittance, (b) % water absorption of pure PVA and PVA/10%CSP/1 mM AgNPs films.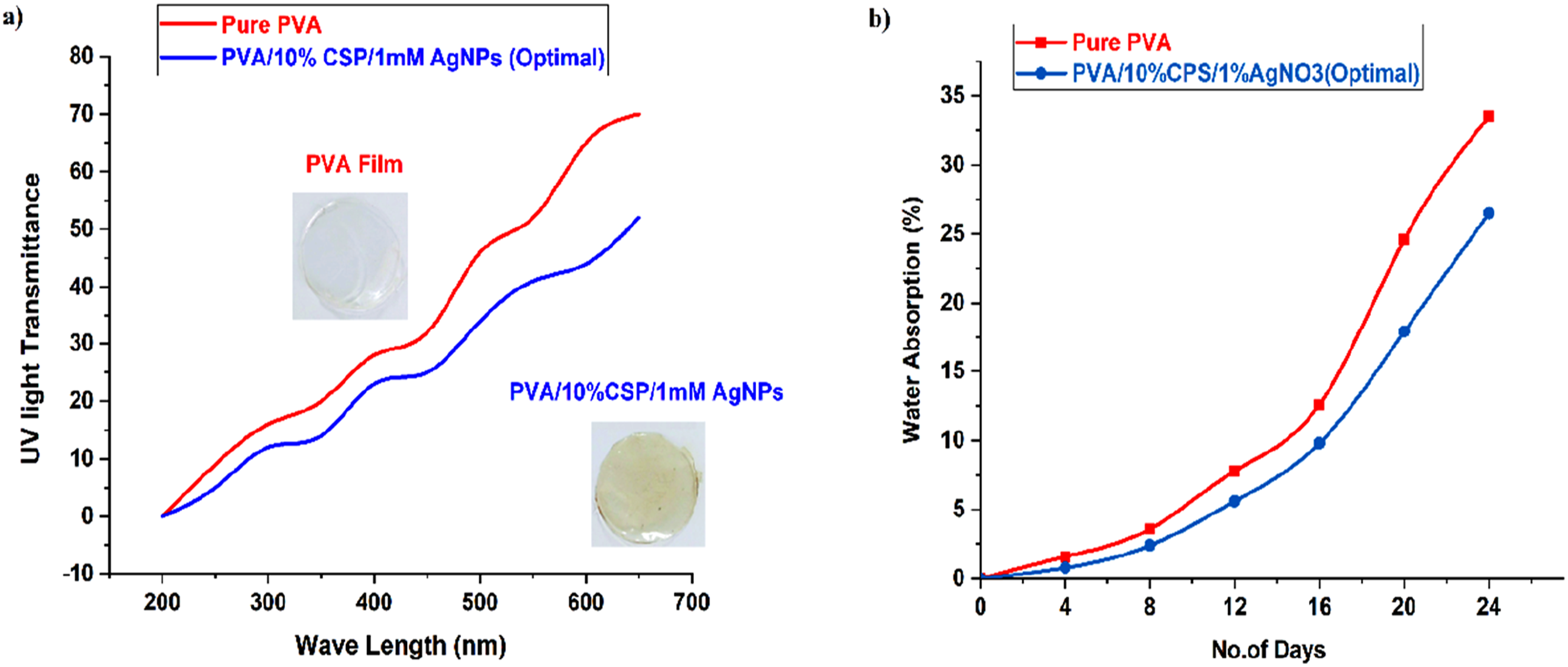
Water absorption
The water absorption of pure PVA film and PVA/10%CSP/1 mM AgNPs (optimal) films is shown in Figure 12(b). The pure PVA film is a water-soluble material and absorbs water molecules. When exposed to moisture or submerged in water, pure PVA absorbs a significant amount of water. The water absorption rate of films was evaluated every 4 days of 24 days test duration. The pure PVA film absorbs 33.5% of water at the end (24 days) of the water absorption test run. The incorporation of coconut shell powder partially reduces the water absorption of films because it contains both hydrophilic cellulosic elements and hydrophobic non-cellulosic components. 35 The presence of lignin in coconut shell powder restricts moisture absorption as it is a hydrophobic component. The inclusion of silver nanoparticles (AgNPs) in the hybrid system acts as an inert filler and restricts the movement of water molecules in the available free-volume polymer matrix. 36 AgNPs also act as a shield against water diffusion into the hybrid composite films. Due to the inclusion of CSP and AgNPs in PVA films, the water absorption is abridged to 14.5%. The inclusion of 1 mM AgNPs enhances this effect by limiting water penetration pathways in the matrix. The PVA/10%CSP/1 mM AgNPs (Optimal) film is better suited for water-resistant applications than pure PVA because it exhibits better dimensional stability and resistance to swelling in water.
Soil burial test
The biodegradation of films in soil is an important functional property to access its usage in food packaging applications. The pure PVA films and PVA/10% CSP/1 mM AgNPs (optimal) films were subjected to a soil burial test, and their weight loss is depicted in the Figure 13. Figure 13, shows that the weight loss of pure PVA film and optimal film sample is 32.5% and 19.5% respectively after 30 days of burial. Pure PVA films degrade relatively quickly in soil, depending on soil composition, moisture content, and microbial activity.
26
Pure PVA films allow the polymer and soil bacteria to interact directly because they do not contain any fillers or reinforcing substances. The inclusion of coconut shell powder also improves biodegradation because it contains cellulosic components. On the other hand, CSP also contains lignin, lignin slows down degradation due to its hydrophobic and recalcitrant nature.
37
The AgNPs possess antimicrobial properties and reduce microbial growth on the film surface. This antimicrobial effect slows down the degradation process in soil by limiting microbial colonization on the surface of the optimal film sample.
38
Overall, because of the combination of antimicrobial properties of AgNPs and the partial biodegradation nature of coconut shell powder, the PVA/10% CSP/1 mM AgNPs composite film generally exhibits a slower rate of degradation. The PVA/CSP/AgNPs hybrid biofilm is appropriate for applications demanding regulated biodegradation in soil because of this balance. % Weight loss of film samples in soil.
Conclusions
The 25 different combinations of PVA/CPS/AgNPs hybrid biocomposite films are fabricated by the solution casting method. The effect of variations of CSP (5–25 wt. %) and AgNPs (1 mM – 5 mM) content in the PVA matrix was analyzed by mechanical and thermal studies. To predict the effective filler combination and optimal film sample, the multi-objective optimization of mechanical and thermal properties of CPS/AgNPs loaded PVA biocomposite was conducted through the Taguchi-based GRA technique. The optimal elements level was identified through GRG term and further confirmation test was conducted. Through the GRA technique it is exhibited that among all various factors, the 1 mM of AgNPs and 10% of CSP provide a significant effect on the mechanical and thermal properties. The optimal sample tensile strength is 38.1 MPa, Young’s modulus is 92.3 MPa, % elongation at brake is 18.1 % and the maximum degradation temperature is 315oC. Additionally, the optimal film is subjected to characterization techniques such as FTIR, XRD, TGA, SEM, UV- transmittance, water absorption, and soil burial test. The FTIR and XRD analysis revealed a good interaction between fillers (10% CSP and 1 mM AgNPs) and the PVA matrix. The surface morphology plots confirmed that better dispersion of CSP and AgNPs in the film surface as well as the surface of the film is free of voids and clusters. The optimal films allow only 45% of the total incident UV rays to pass through their surface. The water absorption of pure PVA film is 33.5% and optimal film (PVA/10%CSP/1 mM AgNPs) is 14.5%. The improvement in UV ray blocking and water absorption behavior of the optimal film sample is achieved through the proper dispersion of AgNPs particles on the film surface. Additionally, the lignin content in coconut shell powder plays a vital role in preventing moisture penetration through the film surface. The optimal films degrade (19.5%) more slowly compared to pure PVA films (32.8%) over a 30-day soil burial period.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by King Mongkut's University of Technology North Bangkok with Contract no. KMUTNB-68-KNOW-16.