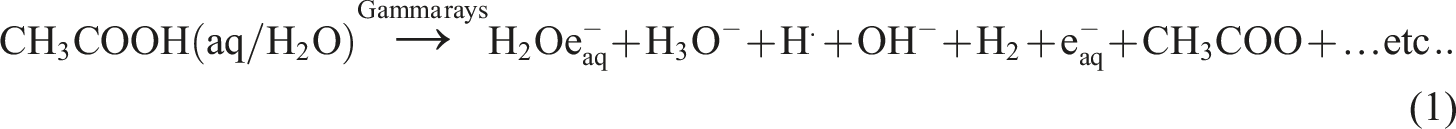Abstract
Poly(N-vinyl pyrrolidone/chitosan/Ag nanoparticles) Poly (NVP/CS/AgNPs) nanocomposite was prepared by using gamma radiation. Silver nitrate was reduced to AgNPs by using gamma radiation to create highly stable, and evenly dispersed AgNPs within the nanocomposite network. Fourier-transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), transmission electron microscopy (TEM), and scanning electron microscopy with energy dispersive X-ray spectroscopy (SEM-EDX) analysis are used to identify the characteristics of the prepared nanocomposite. The newly biocompatible Poly (NVP/CS/AgNPs) nanocomposite prepared by the gamma radiation is used as a model for an anticancer drug. Regarding its application on cancer cells, the results of the outcome demonstrate that the Poly (NVP/CS/AgNPs) nanocomposite significantly decreased the HepG2 cell viability from 100% to 1.5% after 72 hours using the concentration of 30 µg/ml, and the IC50 recorded 10.024 µg/ml at 72 hours of treatment. Moreover, the treatment with the Poly (NVP/CS/AgNPs) nanocomposite increases the cytotoxicity against cancer cells at different concentrations 30 µg/ml and 3.75 µg/ml at different time intervals of 24, 48, and 72 hours. The percent change of MDA, and H2O2 increased in treated cells, and was recorded at 127%, and 69%, respectively, but the CAT activity decreased by 47% in comparison with untreated cells. Relative gene expression was performed in the control, and treated group. The gene expression revealed that the Bcl-2 gene expression as the anti-apoptotic gene was downregulated in the treated group. In contrast, the apoptotic gene BAX was upregulated, and reached 7383-fold in the group treated. The Bax/Bcl2 ratio reached 625.19 which is an excellent indicator of apoptosis. Using western blot, the protein expression increased for Caspse-3 and P53 recorded at 203% and 109%, respectively. In conclusion, Poly (NVP/CS/AgNPs) nanocomposite has a potent effect on killing cancer cells.
Introduction
Polysaccharides are widely distributed in the environment and they are important in different fields since they possess unique characteristics than those of synthetic polymers. Among these kinds of polysaccharides, Chitosan (CS) is the most important chitin derivative. Chitosan has a wide range of applications. Some of the potential applications of this biopolymer are in the areas of medicine, water treatment, drug delivery, membranes, adhesives, hydrogels, biosensors, food packaging, and antioxidants. 1
N-vinylpyrrolidone (NVP) has been widely investigated for applications in many fields, as these are known to exhibit good biocompatibility due to their hydrophilic nature, and low cytotoxicity. 2
The cytotoxic effect of anticancer agents from natural sources or more safe active ingredients rather than chemotherapeutic agents is the main target for scientists in the medical and tumor biology era to avoid the side effects of chemotherapy on physiological agents in vivo in addition to their harmful effect on normal cells. Many natural materials such as EGCG 3 and simvastatin 4 were studied and approved for anticancer properties, but for more efficiency and targeting to specific cancer site, the nanotechnology was used to achieve more potency as an anticancer agent.
Researchers have made extensive efforts to fabricate the physical and chemical properties of drug delivery systems to particularly regulate their permeability, biodegradability, surface functionality, biocompatibility, environmental response, and biorecognition sites to synthesize and obtain intelligent drug delivery systems . 5
Nanocomposites containing metal nanoparticles have gained considerable interest and represent a drug delivery system that is claimed as intelligent drug delivery.6,7 Recent advances in nanoscience, and nanotechnology radically changed the way of diagnosis, treatment, and prevention of various diseases in all aspects of human life. Silver nanoparticles (AgNPs) are one of the most vital and fascinating nanomaterials among several metallic nanoparticles that are involved in biomedical applications.8,9
AgNPs play an important role in nanoscience and nanotechnology, particularly in nanomedicine. Due to their outstanding biocompatibility and antibacterial activities, silver nanoparticles (AgNPs) have a wide range of biomedical uses. AgNPs have a major part in the antitumor impact due to their synergistic and cytotoxic effects on cell survival. 10
AgNPs assist in the collection, and delivery of medications to cancer cells, and they prevent the metabolism of cancer and tumor growth. Through reactive oxygen species (ROS), AgNPs can cause cell death in both in vitro and in vivo environments. 11 Nanoparticles (NPs) have recently been used successfully to deliver therapeutic agents in the diagnosis of chronic diseases as well as to reduce bacterial infections. 12 Specialized AgNPs are appealing antibiotics due to their antimicrobial properties, and distinct mode of action. 13 Green synthesis of metallic AgNPs is a cost-effective, simple, and environmentally friendly method. Silver nanoparticles (AgNPs) have been proposed as treatment agents to overcome the problem of drug resistance caused by antibiotic abuse due to their diverse applications in the medical field. 14
Cytotoxicity of nanoparticles is defined as the extent to which the interaction of nanoparticles with cells disrupts cellular structures, and/or processes essential for cell survival and proliferation. Cytotoxicity assays are a quick and simple way to perform initial acute toxicity assessments. 15 The Poly (N-vinylpyrrolidone/chitosan) system is chosen for this target of application in pharmaceuticals. 16 The Nano-chitosan has a radioprotective efficacy against radiation. 17
This study demonstrates the preparation of Poly (NVP/CS/AgNPs) nanocomposite hydrogel to create a nano-drug system has anticancer activity by green synthesis method using gamma radiation preparation technique, 18 that there is no initiator, catalyst or any other chemicals were added.
Materials and Methods
N-vinylpyrrolidone (NVP) with a purity of 99% was obtained from Sigma-Aldrich Company (Germany), chitosan (CS) (C6H11NO4)n low molecular weight (Mt = 100,000-300.000 was purchased from Acros, New Jersey, Sigma Aldrich. Silver nitrate (AgNO3) with a minimum assay of 99% was obtained from Merck Specialities Private Limited Mumbai-India. All other chemicals and solvents are reagent grade and were used without being purified further.
Preparation of Poly (NVP/CS/AgNPs) nanocomposite
The synthesis of Poly (NVP/CS/AgNPs) nanocomposite was carried out using the method described by Dara et. al. 2020 19 and by using gamma radiation preparation technique with irradiation dose 0.66 kGy/h. Chitosan of 1% concentration was first dissolved in 1% acetic acid till complete dissolution, and then (NVP/CS) in composition of (NVP/CS) (0.5 mL/0.5 mL) was mixed for 1 hour before adding different concentrations of AgNO3 (10, 20, 30, 40, and 50 mmol/ml) then irradiated with gamma radiation at dose of 10 kGy (gamma radiation). The radiation reduction of silver nitrate (AgNO3) salt occur at this dose (10 kGy) within the Poly (NVP/CS), and the Poly (NVP/CS/AgNPs) nanocomposite hydrogel was successfully prepared. The yellow color of the (NVP/CS) mixture turned brown, and the prepared Poly (NVP/CS/AgNPs) nanocomposite hydrogel dried till constant weight. It was found that the only stand free nanocomposite hydrogel sample was that composed of (NVP/CS) (0.5 mL/0.5 mL), and containing 50 mmol/ml of AgNO3 which was selected for application, and characterization in this study.
In the synthesis of the Poly (NVP/CS/AgNPs) nanocomposite the solvent was 1% acetic acid solution [CH3COOH(aq/H2O)] which composed of acetic acid 1 mL diluted with 99 mL bi-distilled water which when exposed to Gamma rays created solvated electrons H2Oe-aq and hydrated electrons e-aq and other primary radicals and molecules20,21 as shown in equation (1):
The solvated electrons H2Oe−aq and hydrogen H. atoms are strong reducing agents so that in the following step they easily reduce silver ions down to the zero-valent state as shown in equations (2) and (3):
Also in the acetic water solution ,
21
the hydrated electrons e-aq can also reduce the Ag+ ions and the neutral atom Ag0 is formed as shown in equation (4):
Finally the produced mixed AgNPs were incorporated within the Poly(NVP/CS) to form Poly(NVP/CS/AgNPs) nanocomposite as follows:
Characterization of the Poly (NVP/CS/AgNPs) nanocomposite
Fourier Transform Infrared
The FTIR analysis of the Poly (NVP/CS/AgNPs) nanocomposite was measured by using Fourier transform infrared (Thermo-scientific-iS50-FT-IR, Nicolet USA) spectroscopy in a spectral range of 4000 to 400 cm−1.
UV-Visible Spectrometer
The Poly (NVP/CS/AgNPs) nanocomposite solution was characterized by a light absorption pattern that was kinetically monitored using (Shimadzu (UV2500), Japan), for analysis.
Field Emission Scanning Electron Microscope (FESEM) and EDX Analysis
The surface of the prepared Poly (NVP/CS/AgNPs) nanocomposite was examined using a field emission scanning electron microscope (FESEM) equipped with a field emission Gun (TESCAN, VEGA3) and linked to an energy-dispersive X-ray analyzer (EDX, unit) with an excitation source of 20 kV for energy-dispersive X-ray evaluation (EDX) and mapping. The elemental composition of the Poly (NVP/CS/AgNPs) nanocomposite was determined by using an EDX analysis.
Transmission Electron Microscopy (TEM)
The TEM method was used to analyze the Poly (NVP/CS/AgNPs) nanocomposite. The Ultra High-Resolution transmission electron microscope (JEOL- 2100, Tokyo, Japan) has a 200 kV voltage has been used. To make TEM grids, a drop of the particle solution was placed on a carbon-coated copper grid and dried under light.
Cytotoxicity Assay
Stock solutions of the tested compound were prepared in 1% DMSO in ddH2O and diluted further to working solutions with DMEM to evaluate the cytotoxic concentration on HepG2 cells. The cytotoxic activity was evaluated in the HepG2 (Hepatocellular carcinoma) cell line using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method with minor modifications . 22 In brief, the cells were seeded in 96-well plates (100 µl/well at a density of 3 × 105 cells/ml) and incubated at 37°C in 5% CO2 for 24 h. After 24 h, cells were treated in triplicate with various concentrations of the tested compounds. The supernatant was removed after 72 h, and the cell monolayers were then washed three times with sterile 1x PBS before being filled with the MTT solution (20 µl of a 5 mg/ml stock solution) and incubated at 37°C for 4 hours. The media was then aspirated. The produced formazan crystals were dissolved in 200 µl of isopropanol that had been acidified (0.04 M HCl in 100% isopropanol = 0.073 mL HCL in 50 mL isopropanol) in each well. Using a multi-well plate reader (BMGLABTECH®FLUOstar Omega, Germany), the absorbance of formazan solutions was measured at a maximum wavelength of 540 nm with 620 nm serving as the reference wavelength. The calculation below was used to calculate the proportion of cytotoxicity compared to the untreated cells. Calculating the concentration that displayed 50% cytotoxicity was done using a plot of % cytotoxicity against sample concentration (TC50).23,24
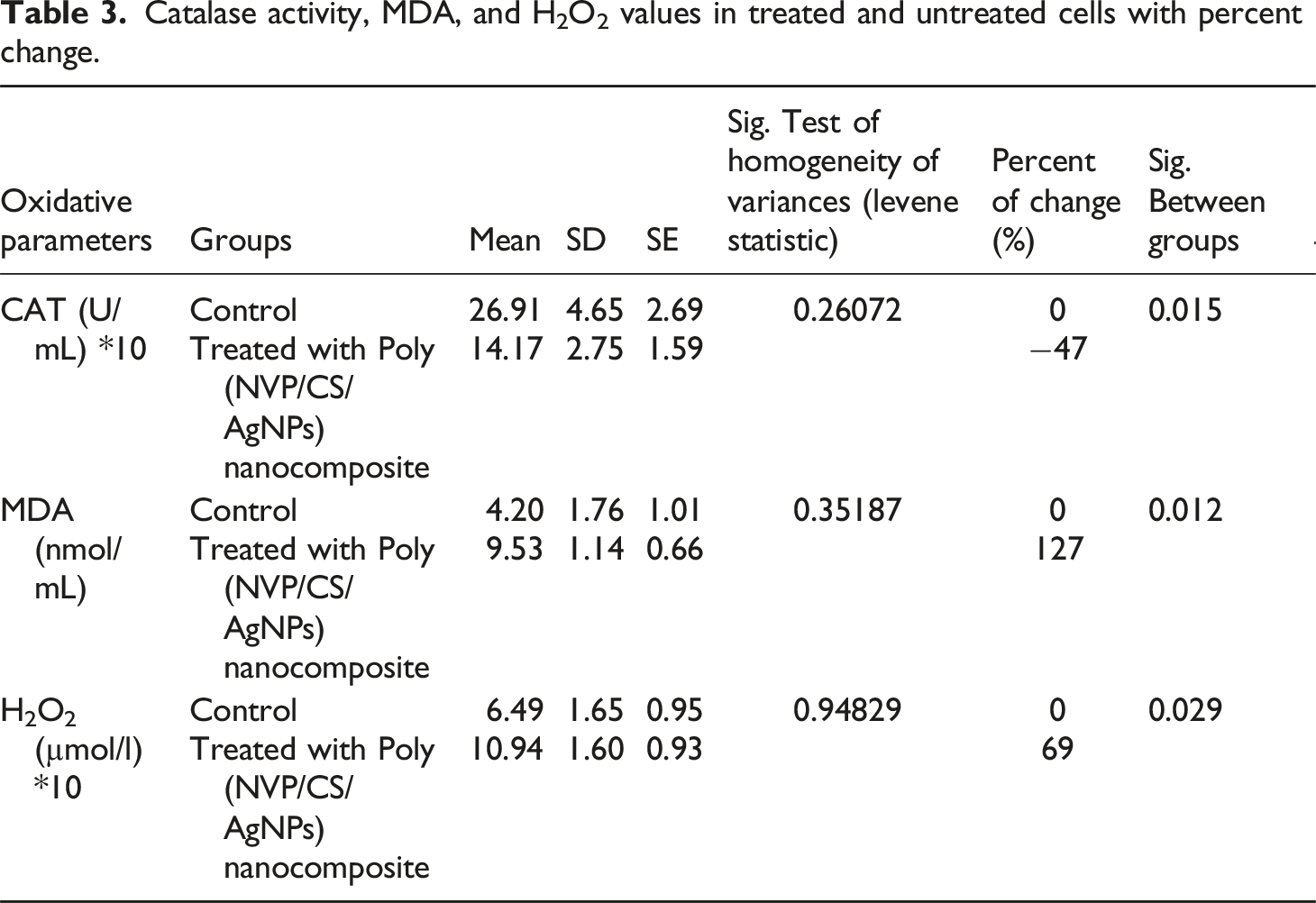
Oxidative Stress and Antioxidant Enzyme Activity
After removing the media, add 50 µl/well of lysis buffer and incubate them for 10 minutes at room temperature, then the cell lysate was gathered from different groups after adding 2 mL/well of cold phosphate buffer, and centrifuged at 3000 rpm for 10 min. And then the supernatants were utilized to quantify hydrogen Peroxide (H2O2), MDA and Catalase (CAT). The levels of MDA were measured using the endpoint calorimetric assay kit (Malonaldehyde (MDA) colorimetric assay kit, catalog No: E-BC-K025-S, Elabscience Biotechnology, USA, https://file.elabscience.com/Manual/biochemical_kits/E-BC-K025-S.pdf. Catalase was measured using calorimetric commercial kits Catalase (CAT) Activity colorimetric kit, cat no: E-BC-K031-S, Elabscience Biotechnology, USA, https://file.elabscience.com/Manual/biochemical_kits/E-BC-K031-S-Elabscience.pdf. Hydrogen Peroxide (H2O2) was measured according to the manufacturer’s instructions (Biodiagnostic Company, Giza, Egypt).
Gene Expression
Total RNA was extracted from the sample using a miRNeasy Serum/Plasma Advanced Kit (Qiagen, Hilden, Germany), as directed by the manufacturer. To summarize, RNA was extracted from each sample to yield 60 µL of RNA solution. The quality of the RNA was determined using a Nanodrop Spectrophotometer (A260/280 ratio). The purified RNA was immediately used to synthesize cDNA. 25
The Rever Aid RT Kit (ThermoFisher Scientific, Waltham, USA) was used to reverse transcribe 5 µL of RNA according to the manufacturer’s procedure by using random dt18 and hexamer primers. Real-time PCR Amplification on the Bio-Rad platform was used to measure mRNA expression using the primer sets of Nawah-Scientific. As a control, the housekeeping B-actin was employed. In a final volume of 20 µl, the cDNA was mixed with RT2 SYBR Green qPCR Mastermix.
Real-time PCR for expression of GAPDH, BAX, and BCL-2 genes was performed using Bio-Rad CFX OPUS 96 Real-time PCR Platform using protocol 94.0°C for 3:00 min, 94.0°C for 0:15 sec, 60.0°C for 0:30 min for 39 cycles using SYBR Green dye. The relative gene expression was calculated by Double Delta Ct” Method for Relative Quantification RQ = 2−ΔΔCt.
Western Plot
The cell pellet was collected and then Lysis by lysis buffer, bicinchoninic acid (BCA) for the colorimetric detection, and quantitation of total protein and protein quantification was done by The Thermo Scientific™ Pierce™ BCA Protein Assay Kit. The samples were prepared with loading buffer, then the Protein Electrophoresis (SDS-PAGE) and Transfer to the Membrane and finally detected the Targeted Protein(s) Using Enhanced Chemiluminescence (ECL) and ChemiDoc Imaging.
The primary antibodies against B-Actin, Caspase-3, and P53 and HRP-conjugated secondary antibodies.

Results and Discussion
Fourier Transform Infrared (FTIR)
An FTIR spectrum of chitosan is shown in Figure 1(a). The characteristic bands at 3431 cm−1 correspond to a hydroxyl group, 2919 cm−1 correspond to the CH2 stretching vibration of the pyranose ring, and 1648 cm−1 correspond to C = O in the amide group. The peaks appeared at 1418 cm−1 and 1252 cm−1 corresponding to the NH2 group and CH3 in the amide group respectively, the peaks appeared at the range 1156–903 cm−1 which correspond to C-O-C in glycosidic linkage are represented. FTIR analysis of (a) pure chitosan, (b) Poly (NVP/CS) copolymer, and (c) Poly (NVP/CS/AgNPs) nanocomposite.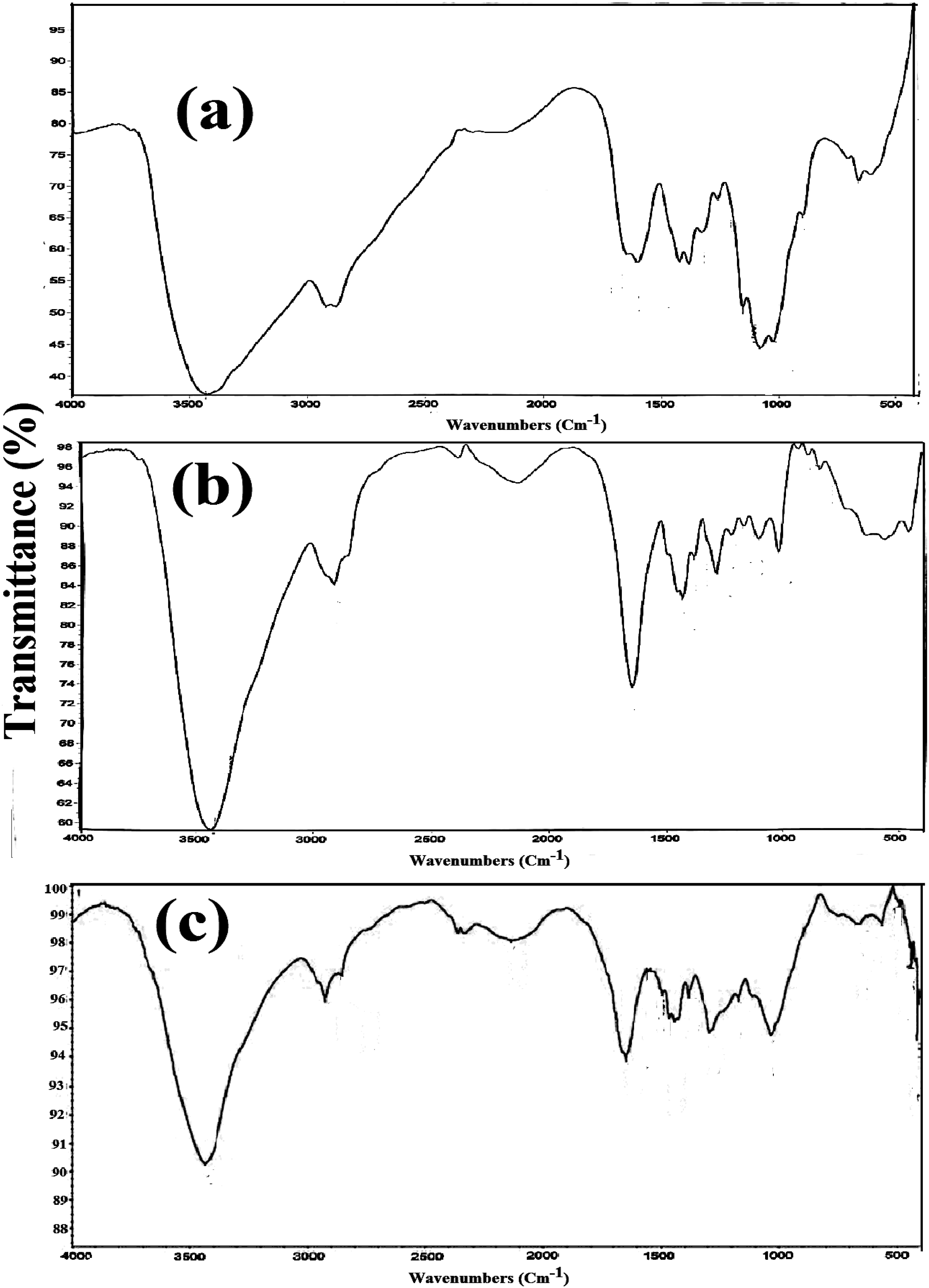
FTIR spectrum of Poly (NVP/CS) copolymer is shown in Figure 1(b). The characteristic peak that appeared at 3428 cm−1 corresponds to the overlap between hydroxyl groups of chitosan and amine groups of NVP. The peak appeared at 2922 cm−1 which corresponds to the CH2 stretching vibration of the pyranose ring. The peak appeared at 1645 cm−1 corresponding to the C = O group. The peaks appeared at 1436 cm−1 and 1224 cm-1 corresponding to the NH2 group and CH3 groups respectively, the peaks appeared at the range 1103–847 cm−1 which corresponds to C-O-C in glycosidic linkage.
In the case of FTIR analysis of the Poly (NVP/CS/AgNPs) nanocomposite the characteristic peaks are similar to that of the Poly (NVP/CS) copolymer but shifting of the position of these peaks due to the presence of AgNPs as shown in Figure 1(c). The occurrence of a new absorption peak at 2,922 cm−1 ascribed to the –C–H stretching band further indicates that the NVP monomer are successfully polymerized and crosslinked in the presence of CS and AgNPs by breaking the C = C bond. The interaction between AgNPs and chemically functional groups inside the Poly (NVP/CS) networks, including formation of a coordinate bond (forming a ligand complex) and/or an electrostatic action. Consequently, this study features a new production technology for metal nanoparticles manipulated by multicomponent means of a non-covalent polymeric complex 26 that are a candidate of nano-drug system has anticancer activity. Also, the gamma-ray radiation preparation technique causes structural rearrangements in the nanocomposite chains upon the embedding of AgNPs nanoparticles. 27 Therefore, the FTIR analysis confirms the successful preparation of the Poly (NVP/CS/AgNPs) nanocomposite.
UV-Visible Spectrometer
The Poly (NVP/CS/AgNPs) nanocomposite solution was characterized by a UV-visible spectrometer. Visual observation revealed that the hue of the solution changed to a yellowish brown color, of the Poly (NVP/CS/AgNPs) samples, confirming the synthesis of AgNPs within the Poly (NVP/CS/AgNPs) nanocomposite matrix.
28
The activation of AgNPs’ surface plasmon resonance (SPR) is what causes the yellowish-brown color to become more intense.
29
Kinetic monitoring of the solution was done between 300 and 700 nm. The size and form of the solution might be examined using UV-vis spectroscopy.
30
In the UV-vis spectrum of AgNPs, the distinctive absorbance peak was found at 424 nm, suggesting the presence of metal AgNPs. Additionally, the broadening of the peak demonstrated the polydispersity of the AgNPs. The findings demonstrate the interaction of amino groups with metal surfaces, which served as stabilizing capping sites for AgNPs.
31
The synthesis of the AgNPs was described by using UV spectroscopy. Visual inspection demonstrated that the solution’s color changed to a yellowish-brown tone, indicating that AgNPs had indeed been synthesized. The intensification of the yellowish-brown color is due to the activation of the surface plasmon resonance (SPR) of AgNPs. Between 300 and 700 nm, the solution’s kinetics were monitored. UV-vis spectroscopy could be used to analyze the size and shape of the solution. The existence of metal AgNPs was suggested by the appearance of a characteristic absorbance peak in the UV-vis spectra of AgNPs at 424 nm. Additionally, the broadening of the peak showed the particles’ polydispersity as shown in Figure 2 UV-Visible spectra of the Poly (NVP/CS/AgNPs) nanocomposite.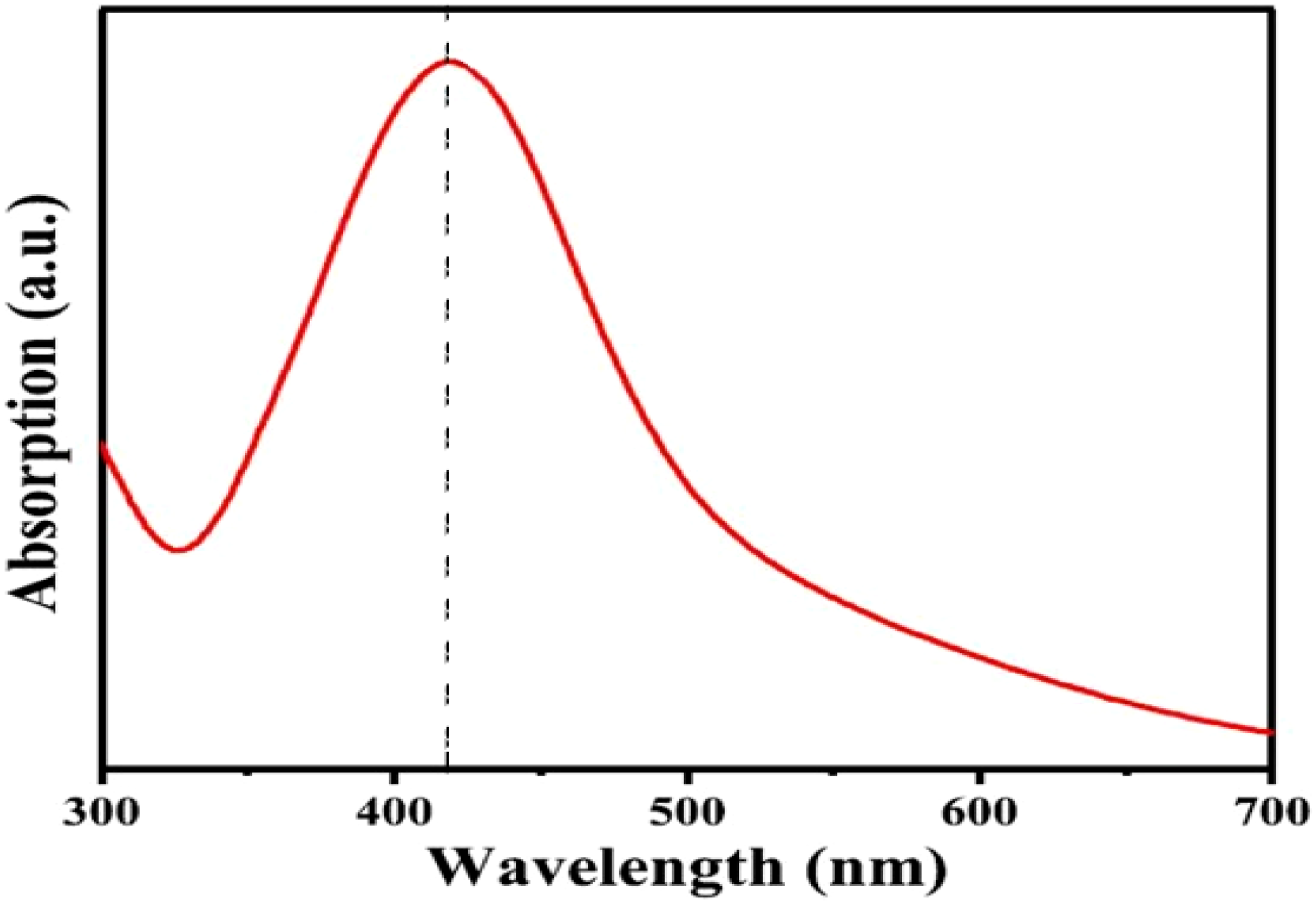
Field Emission Scanning Electron Microscope (FESEM)
FESEM image show the nature of the AgNPs distributed within the Poly (NVP/CS/AgNPs) nanocomposite surface as shown in Figure 3 (a, and b). The AgNPs were spherical particles in shape within surface of the Poly (NVP/CS/AgNPs) nanocomposite, and there was no evidence of aggregation. The AgNPs completely covered nanocomposite surface and has average particle size of 70 nm. FESEM of the Poly (NVP/CS/AgNPs) nanocomposite.
Transmission Electron Microscopy (TEM)
TEM pictures indicate that the Poly (NVP/CS/AgNPs) nanocomposite containing AgNPs which are nearly spherical, polydisperse, and with average particle size of 56 nm. TEM micrographs also demonstrate uniform dispersion of Ag nanostructures within the Poly (NVP/CS/AgNPs) surface as shown in Figure 4. TEM of the Poly (NVP/CS/AgNPs) nanocomposite.
Energy-Dispersive X-Ray (EDX)
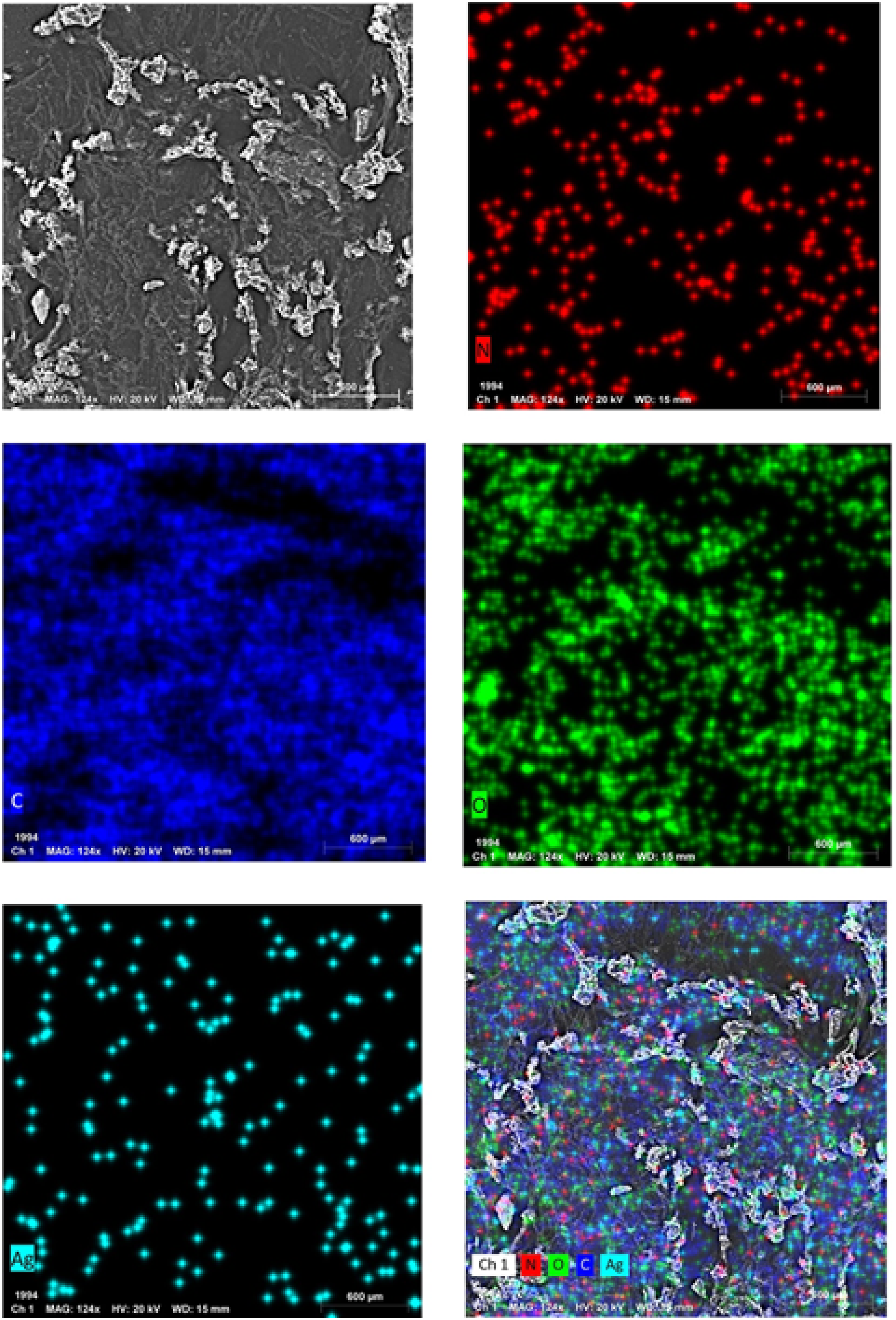
The elemental composition of the Poly (NVP/CS/AgNPs) nanocomposite was determined by using an EDX analysis as shown in Figures 5, and 6. The EDX spectra showed the presence of known peaks of (Ag), (O), (C), and (N), where the presence of an Ag signal suggests the formation of Ag nanostructures. EDX of the poly (NVP/CS/AgNPs). EDX mapping for the elemental composition of Poly (NVP/CS/AgNPs) nanocomposite.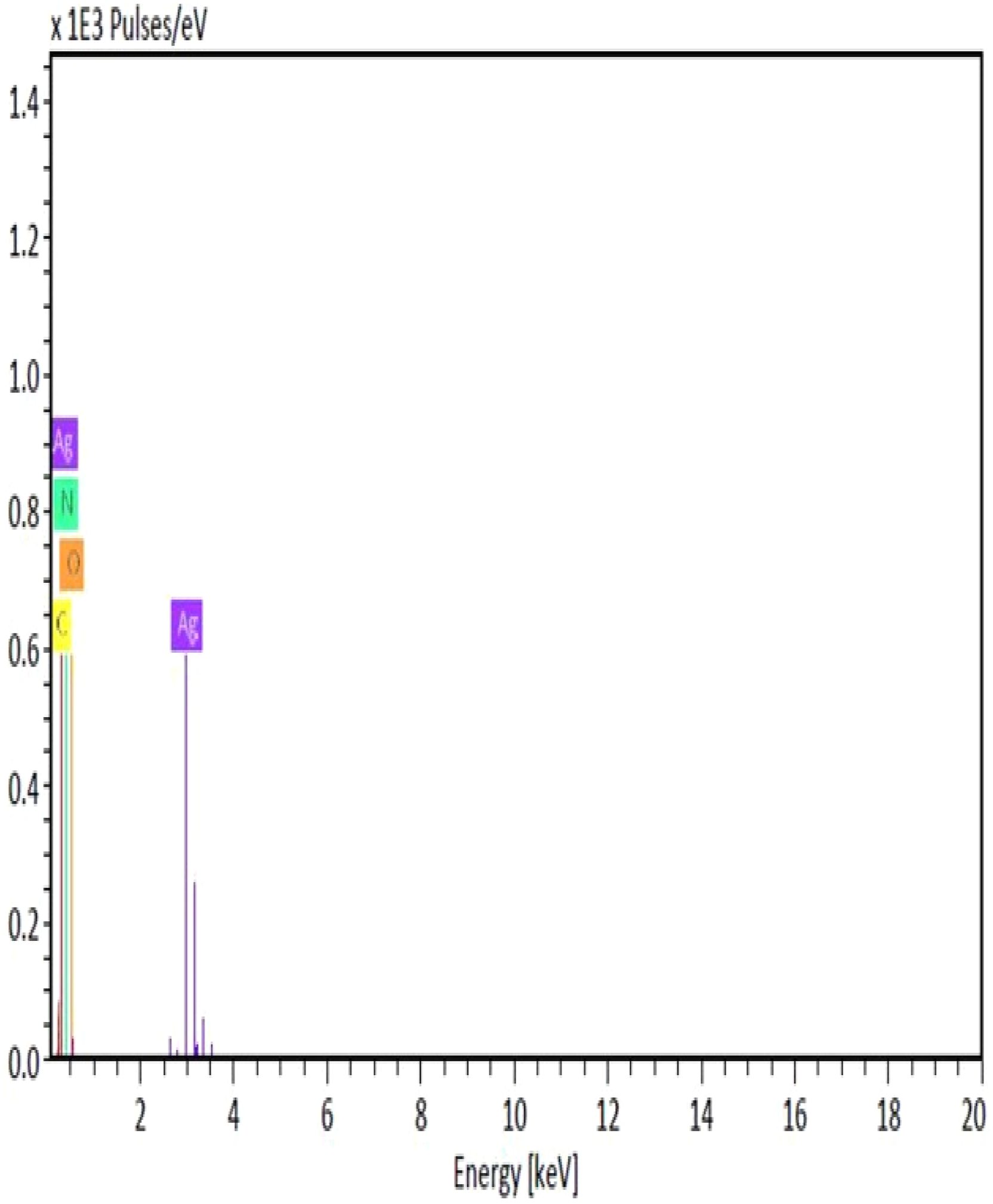
Cytotoxicity MTT/HepG2 Cell Line
The cytotoxicity effect of the Poly (NVP/CS/AgNPs) nanocomposite using MTT assay resulted in the IC50 is 10.024 µg/ml after 72 hours of treatment in vitro on HepG2 cell line as mentioned in Figure 7. At dose, 3*105 µg/ml, the viability recorded 1.53%, and the inhibition rate recorded 98.47%, but at doses 9.4*103, and 4.7*103 µg/ml, the viability recorded 60.9%, and 75%, respectively as mentioned in Table 2. The result of IC50 revealed the efficiency of the current nanocomposite in killing cancer cells where the 10.024 µg/ml killed 50% of cells causing an increasing the rate of apoptosis through decreasing the cell resistance and growth rate in addition to increasing the oxidative stress of cells and decreasing the antioxidant enzyme activity. Shows the IC50 sigmoidal fit for Poly (NVP/CS/AgNPs) nanocomposite. The correlated viability, and inhibition percentages to different concentrations of Poly (NVP/CS/AgNPs) nanocomposite.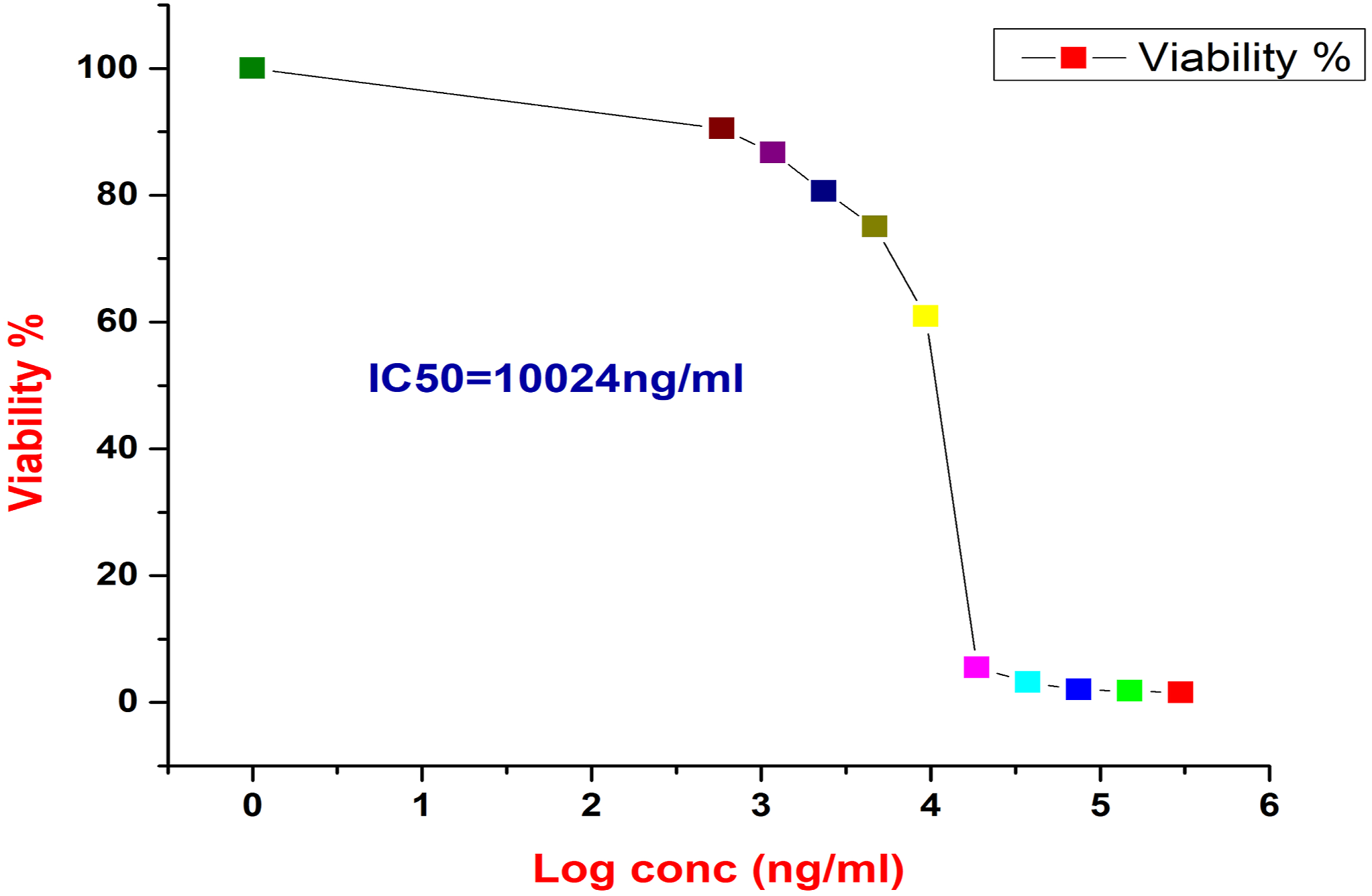
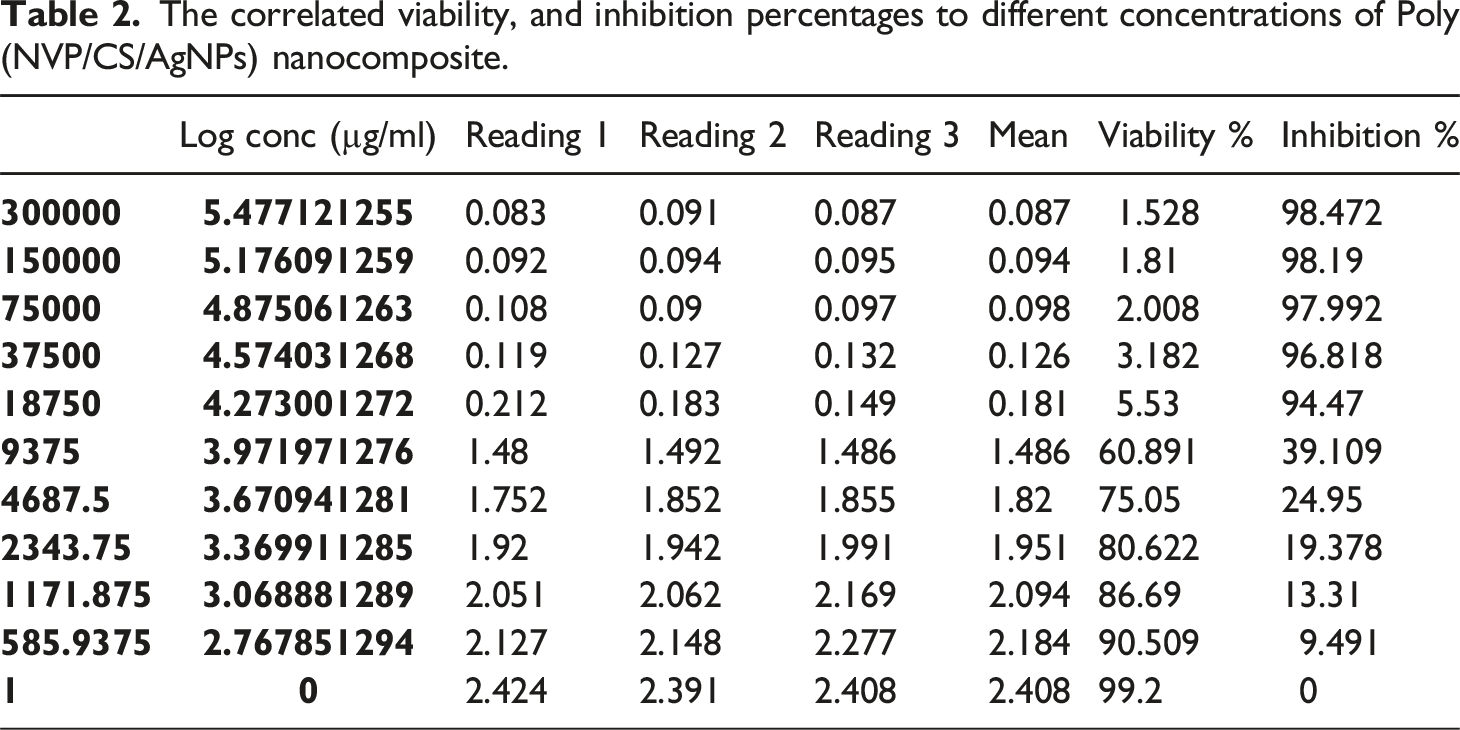
Microscopic imaging of HepG2 cells under different concentrations of Poly(N-vinyl pyrrolidone/chitosan/AgNPs)
The HepG2 cells were propagated in tissue culture plates till reached 99% of viability. HepG2 cells with high proliferation rates and an epithelial-like morphology that perform many differentiated hepatic functions were isolated from a hepatocellular carcinoma of a 15-year-old Caucasian male; a White, male youth with liver cancer according to ATCC (HB-8065) information. The Control cells appear with a highly mitotic rate and with a high viability epithelial shape. Under the treatment, the most of cells were changed to apoptotic shape in addition each drug concentration and time the cell death was increased the cell debris was increased and dead cells were elevated in correlation with drug concentration in addition the ability of division was decreased as shown in the microscopic image in Figure 8 Shows the microscopic images for HepG2 cells at different interval times, and under different concentrations of Poly (NVP/CS/AgNPs) nanocomposite. The percent change of Catalase activity, MDA, and H2O2 values in untreated, and treated HepG2 cells.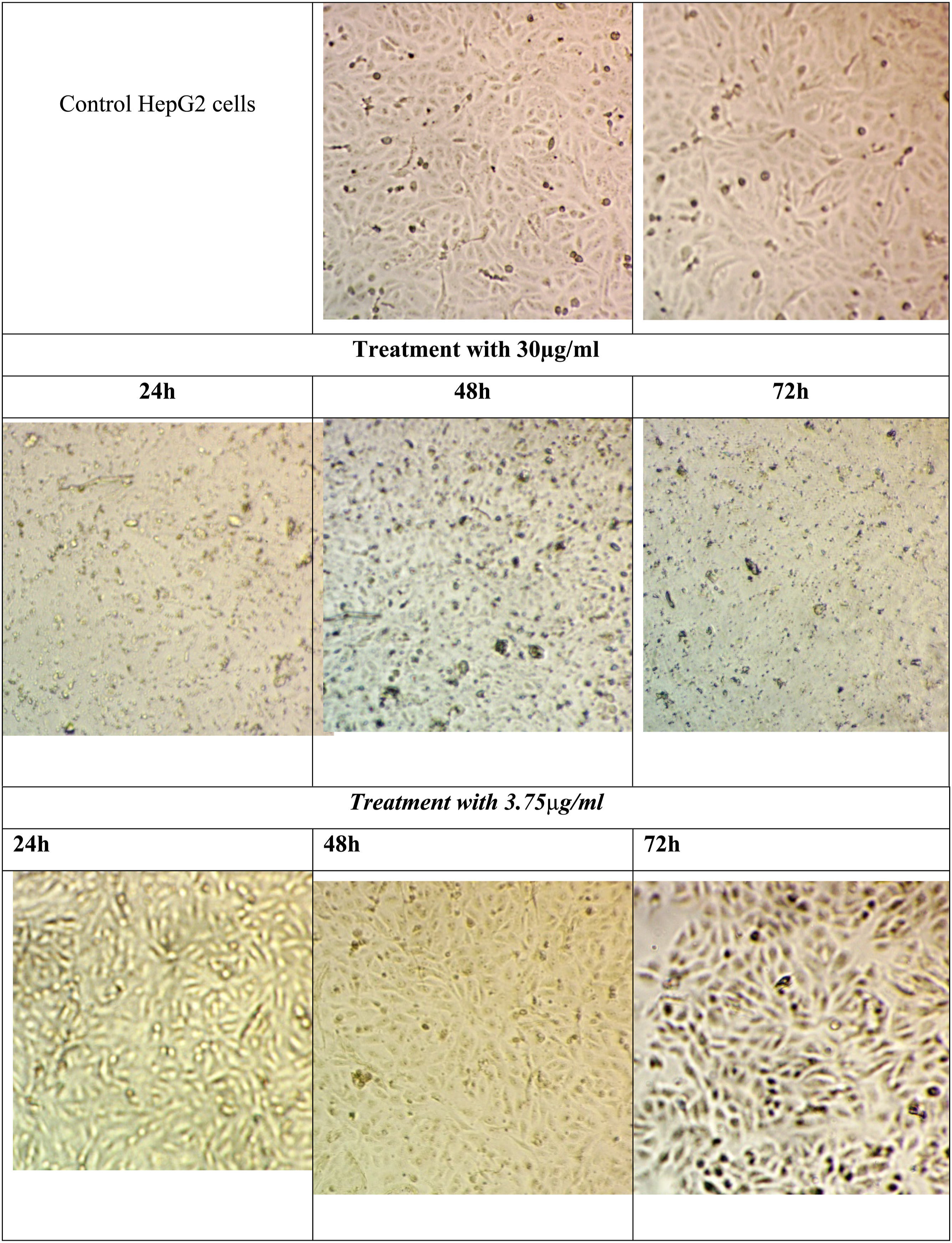
Oxidative Stress and Antioxidant Enzyme Activity
Catalase activity, MDA, and H2O2 values in treated and untreated cells with percent change.
Gene Expression
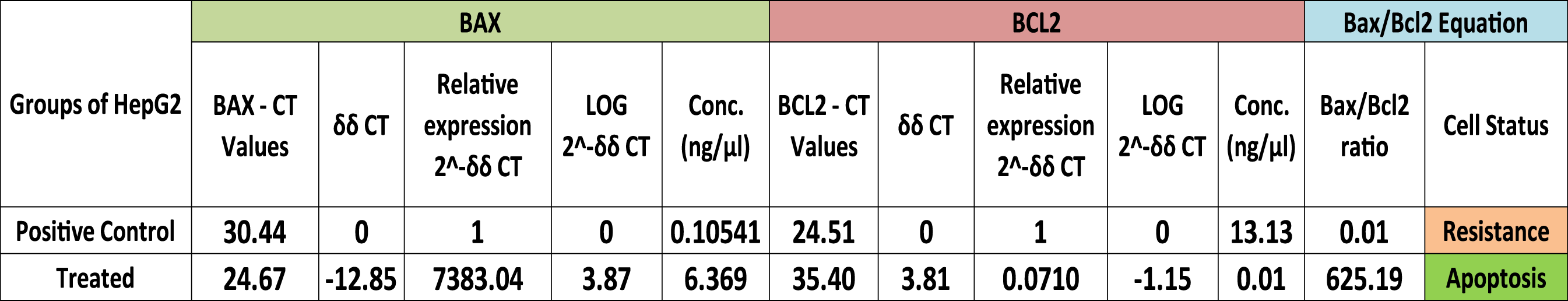
Showing relative gene expression of Bax and Bcl2 which confirms the cell Status under treatment with Poly (NVP/CS/AgNPs) nanocomposite through Bax/Bcl2 Ratio.
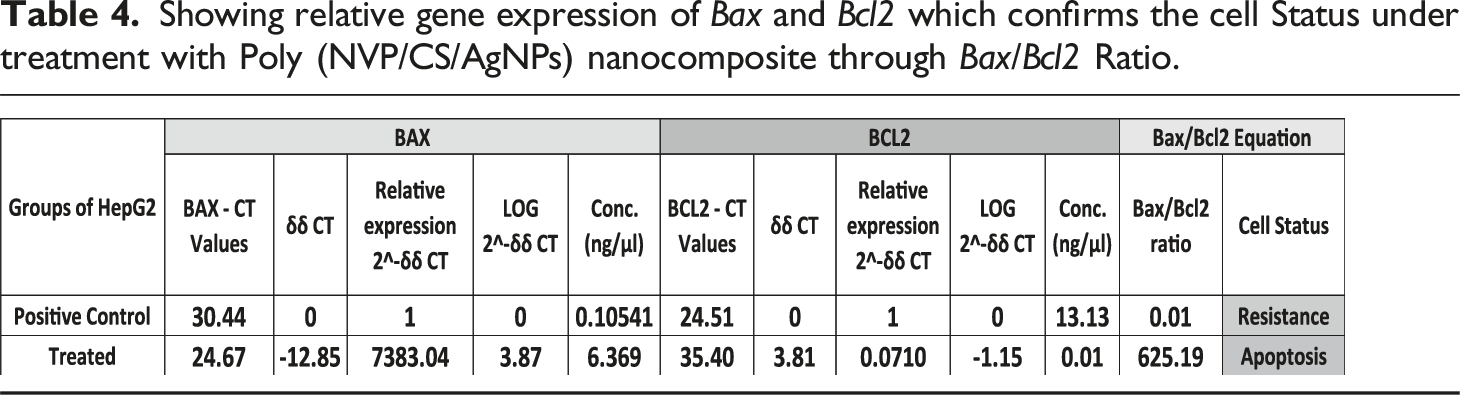
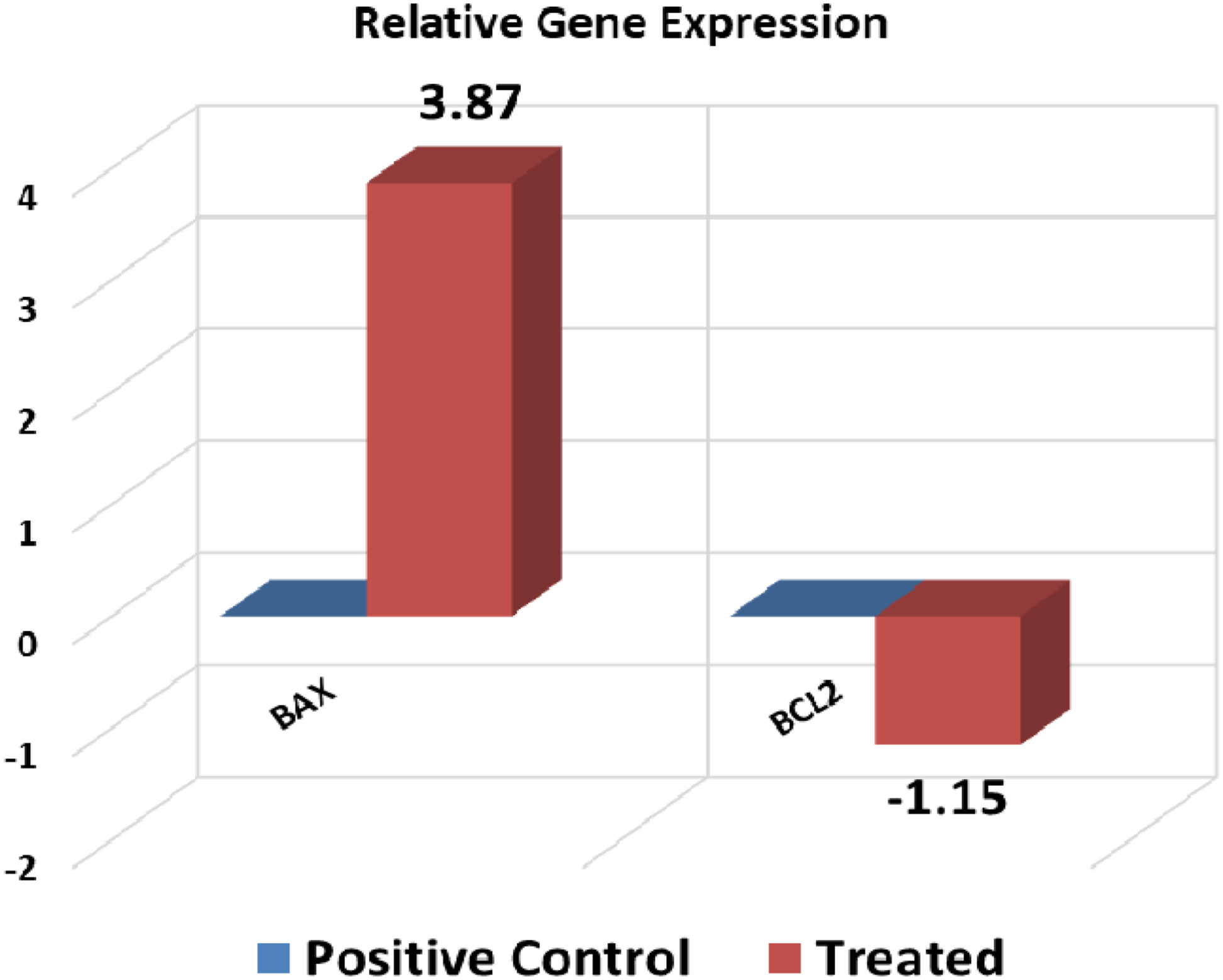
Showing relative gene expression which confirms the cell Status under treatment with Poly (NVP/CS/AgNPs) nanocomposite through Bax/Bcl2 Ratio.
The ratio of Bax to bcl-2 determines the susceptibility of a cell to apoptosis. 4 The current results assured the previous findings regarding to Bax/Bcl-2 ratio as a good prognostic indicator. The results indicated that if the ratio >1 it can serve as a good prognostic indicator but if it is <1 it means that resistance takes place and it should be changed to the regimen of treatment to get a good prognosis. The cell response in different groups in the current study ensured that the current nanocomposite increased the cell death rate (Bax/Bcl-2 ratio >1). The Bax/Bcl-2 ratio in the control group was equal to 0.01 which shows the highly active growth and HepG2 cell resistance in its normal state of growth but in the case of treatment with Poly (NVP/CS/AgNPs) nanocomposite, it was 625.19.
Western Blot Images
The P53, and caspase-3 protein expression in lysates from the HepG2 cell line under the treatment with Poly (NVP/CS/AgNPs) nanocomposite represents in Figure 11 that the size of each band on the membrane which is specific for each protein, B-Actin (MW 45 KDa), P53 (MW 53 KDa) and Caspase-3 (MW 35 kDa). In Table 5 the protein expression of Caspse-3 increased 203% in treated cells in comparison to untreated cells and the P53 increased 109% in comparison to untreated cells. Western blot analysis of B-Actin, caspase-3, and P53 protein expression in a human hepatocellular cell line (HepG2) including the specific band for each one. Shows the band Density of each gene, and their normalization to B-actin and control group.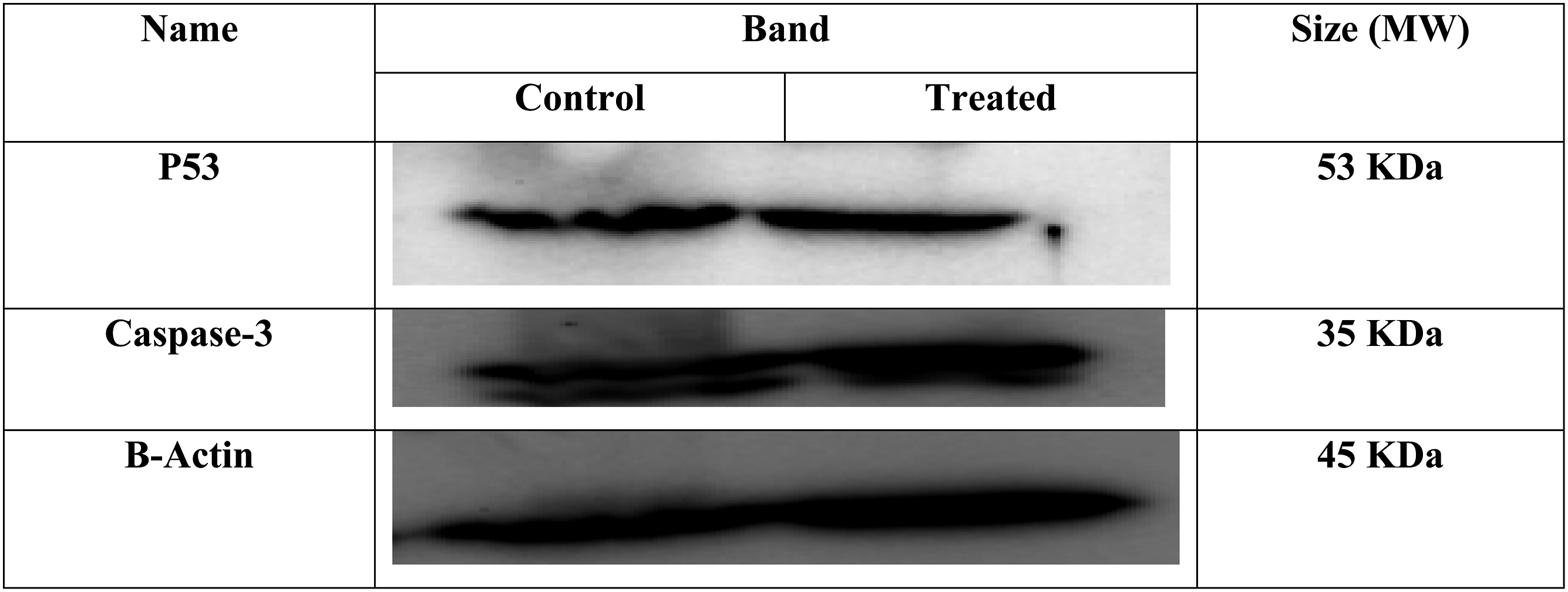
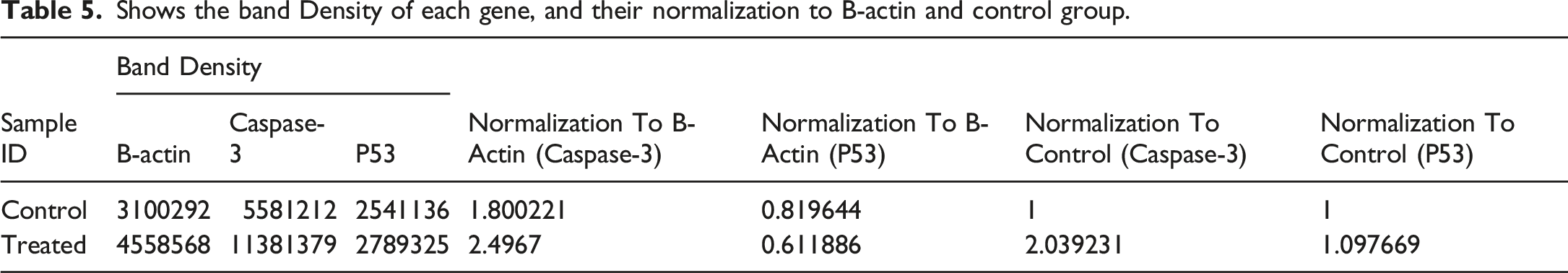
Depending on the gene expression and Protein expression, all of those results confirming the anticancer effect of Poly (NVP/CS/AgNPs) in vitro through the intrinsic pathway of apoptosis where the BCL-2-family pro-apoptotic proteins induced the permeabilization of the mitochondrial outer membrane leading to up-regulated the gene expression of apoptotic gene (BAX and Caspase-3) and down-regulation of the anti-apoptotic gene (Bcl2) even on the level of gene expression or protein
Conclusion
The FTIR, XRD, TEM, DLS, and SEM-EDX analyses confirm the successful preparation of the Poly (NVP/CS/AgNPs) nanocomposite by green radiation technique. The TEM pictures and field emission scanning electron microscope indicate that the Poly (NVP/CS/AgNPs) nanocomposite containing AgNPs are nearly spherical, polydisperse, and with average particle size ranging from 56-70 nm. The analysis of the data described in this study offers a great starting point for the creation of the subsequent generation of anticancer nanomedicine drug delivery systems. The Poly (NVP/CS/AgNPs) nanocomposite has antibacterial and antimicrobial activities . 32 In addition to our study, the Poly (NVP/CS/AgNPs) nanocomposite has an anticancer effect on the HepG2 cell line. The Poly (NVP/CS/AgNPs) nanocomposite significantly decreased the HepG2 cell viability from 100% to 1.5% and the IC50 recorded 10.024 µg/ml after 72 hours using the concentration 30 µg/ml. The percent change of MDA and H2O2 increased in treated cells and was recorded at 127% and 69%, respectively but the CAT activity decreased by 47% in comparison with untreated cells. Relative gene expression was performed in the control and treated group. The gene expression revealed that the Bcl-2 gene expression as the anti-apoptotic gene was downregulated in the treated group. In contrast, the apoptotic gene BAX was upregulated, and reached 7383-fold in the treated group. The Bax/Bcl2 ratio reached 625.19 which is an excellent indicator of apoptosis. Using western blot, the protein expression increased for Caspse-3 and P53 recorded at 203% and 109%, respectively. In conclusion, the Poly (NVP/CS/AgNPs) nanocomposite has potent effect on killing cancer cells.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.