Abstract
The acrylic acid was used as chemical modifier to improve the properties of coconut shell powder (CSP) filled polylactic acid (PLA) biocomposites. The effects of filler content and acrylic acid on tensile properties, thermal properties, and morphology of PLA/CSP biocomposites were investigated. It was found that the addition of CSP to PLA biocomposites have decreased the tensile strength and elongation at break, however the modulus of elasticity increased. The treated PLA/CSP biocomposites with acrylic acid have higher tensile strength and modulus of elasticity but lower elongation at break. Surface treatment using acrylic acid has enhanced filler–matrix interaction. The thermal stability of biocomposites increased with increasing CSP content. The treated biocomposites has better thermal stability than untreated biocomposites. The dispersion and interfacial adhesion between the CSP and PLA polymer were important factor to improve the thermal stability of treated biocomposites. The better interfacial interaction between CSP and PLA matrix was confirmed through scanning electron microscope study.
Introduction
For the past few decades, researchers have shown increasingly remarked interest in biocomposite materials as those materials are eco-friendly, biodegradable, and renewable. In the past century, biocomposites were produced by incorporating natural filler into conventional polymer. Current polymer composites use materials derived from petroleum that are nonrenewable and nondegradable. Most polymer composites are difficult to recycle or incur substantial cost for disposal. 1 The issue faced by the environment of conventional polymer increased the interest in production and use of polymer from renewable resources can solve this problem.
Polylactic acid (PLA) is synthesized from renewable resource, which was polymerized from monomers derived from agricultural resources such as corn, sugar cane, and sugar beet. 2 PLA was able to solve the polymer waste management issue, since it can be degraded in soil and enzymatic environment. 1 Formerly, PLA composites improved its properties by blending with low-cost, renewable, and fully biodegradable natural filler, such as wood flour, starch, lignocellulosic fibers/fillers, and natural cellulose fibers. 1,2 There was many advantages in PLA compared to the conventional polymer, however it was not widely used because of its high cost, high brittleness, and poor moisture barrier properties. 3 –6
Coconut shell is one of the lignocellulosic filler, which is an agricultural coproduct found abundantly in Malaysia, since coconut (
In this study, we aim to investigate the effect of filler loading of CSP and acrylic acid (AA) on mechanical properties, thermal properties, and morphology of PLA/CSP bicomposites.
Experimental
Materials
PLA was supplied by TT Biotechnologies Sdn. Bhd., Penang. The coconut shell was obtained from market, Perlis, and cleaned manually. The coconut shell was cleaned, crushed, and grinded into powder. The coconut shell powder (CSP) was dried at 80°C for 24 h. The average particle size of the CSP was 38 μm, which was measured using Malvern Particle Size Analyzer Instrument (Italy). The acrylic acid (grade 01730) and ethanol were supplied by Fluka (Penamg). Table 1 shows the formulation of untreated and treated PLA/CSP biocomposites.
Formulation of untreated and treated PLA/CSP biocomposites.
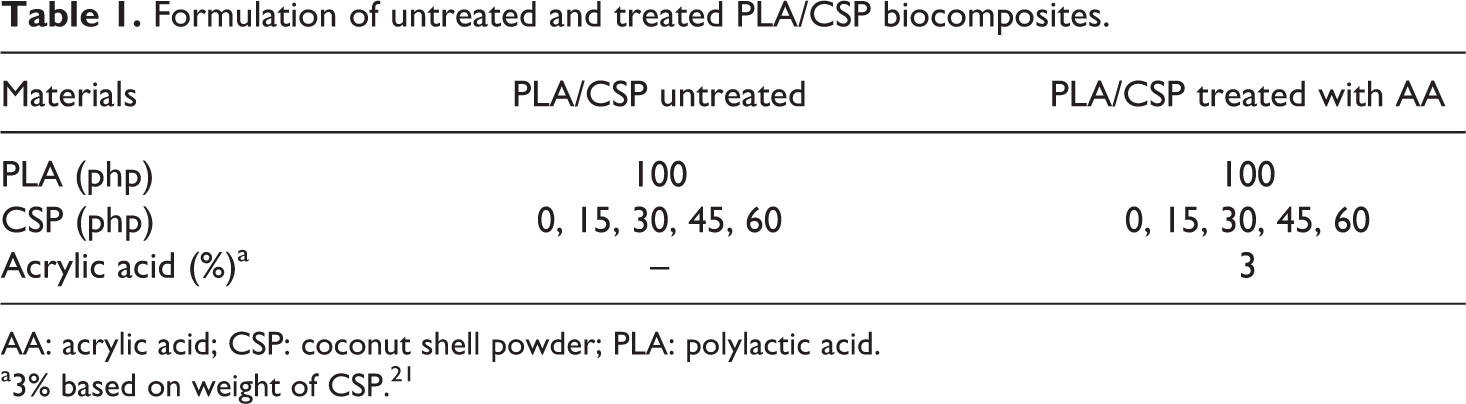
AA: acrylic acid; CSP: coconut shell powder; PLA: polylactic acid.
a3% based on weight of CSP. 21
Filler treatment
The AA was dissolved into ethanol (3% (v/v)). The CSP was added slowly into the solution and stirred for 1 h for completed esterification. The CSP was filtered and dried in oven at 80°C for 24 h.
Preparation of biocomposites
The PLA/CSP biocomposite was prepared using Brabender Plastograph mixer Model EC PLUS at temperature 180°C and a rotor speed of 50 r/min. Firstly, PLA was transferred into mixing chamber for 1 min until it completely melts. After 1 min, CSP was added and mixing continuously for 7 min. The total mixing time was 8 min. The biocomposites was compressed into tensile bar using compression moulding machine model GT 7014A (Gotech, Taiwan). Tensile bar was compared to American Society for Testing and Materials (ASTM) D638 tensile bar type IV with 1 mm thickness. The compression procedure involved preheating at 180°C for 1 min followed by compression for 1 min at the similar temperature and subsequent cooling under pressure for 5 min. The formulation of PLA/CSP biocomposites with different filler loading was shown in Table 1.
Tensile testing
The tensile testing was carried out according to ASTM D638 using Instron Machine model 5569. The across head speed of 50 mm/min was used and the tests were performed at 25 ± 3°C. Tensile properties for the five identical samples of different compositions were measured and the average values were reported.
Thermal analysis
The thermal analysis was carried out by thermogravimetric analysis Diamond, Perkin-Elmer (Malaysia). Samples of 6 ± 2 mg were scanned from 30 to 600°C at a heating rate of 20°C/min under nitrogen atmosphere. The nitrogen flow rate was 50 ml/min.
Morphology analysis
Scanning electron microscope (SEM) model JEOL JSM-6460LA (Japan) was used to examine the dispersion of filler in PLA matrix and bonding between filler and matrix. The fracture ends of the specimen were mounted on an aluminum stub and sputter coated with a thin layer of palladium electrostatic charging during examination.
Results and discussion
Tensile properties
Figure 1 illustrated the effect of CSP content on tensile strength of untreated and treated PLA/CSP biocomposites. The tensile strength of untreated PLA/CSP biocomposites decreased with the increase in the filler content. This occurs because of the weak interfacial adhesion, low wettability, and poor dispersion between hydrophilic CSP and hydrophobic PLA matrix. Nevertheless, the treatment of CSP with AA in PLA biocomposites has increased the tensile strength of PLA/CSP biocomposites. This behavior attributed to presence of strong interfacial adhesion and better dispersion between CSP and PLA matrix. AA reacts through esterification with hydroxyl group of CSP to form hydrogen bond. The improvement in tensile property of treated biocomposites is an indication of improved adhesion at the interface between CSP and PLA after chemical treatment. The schematic reaction between CSP and AA is shown in Figure 2.
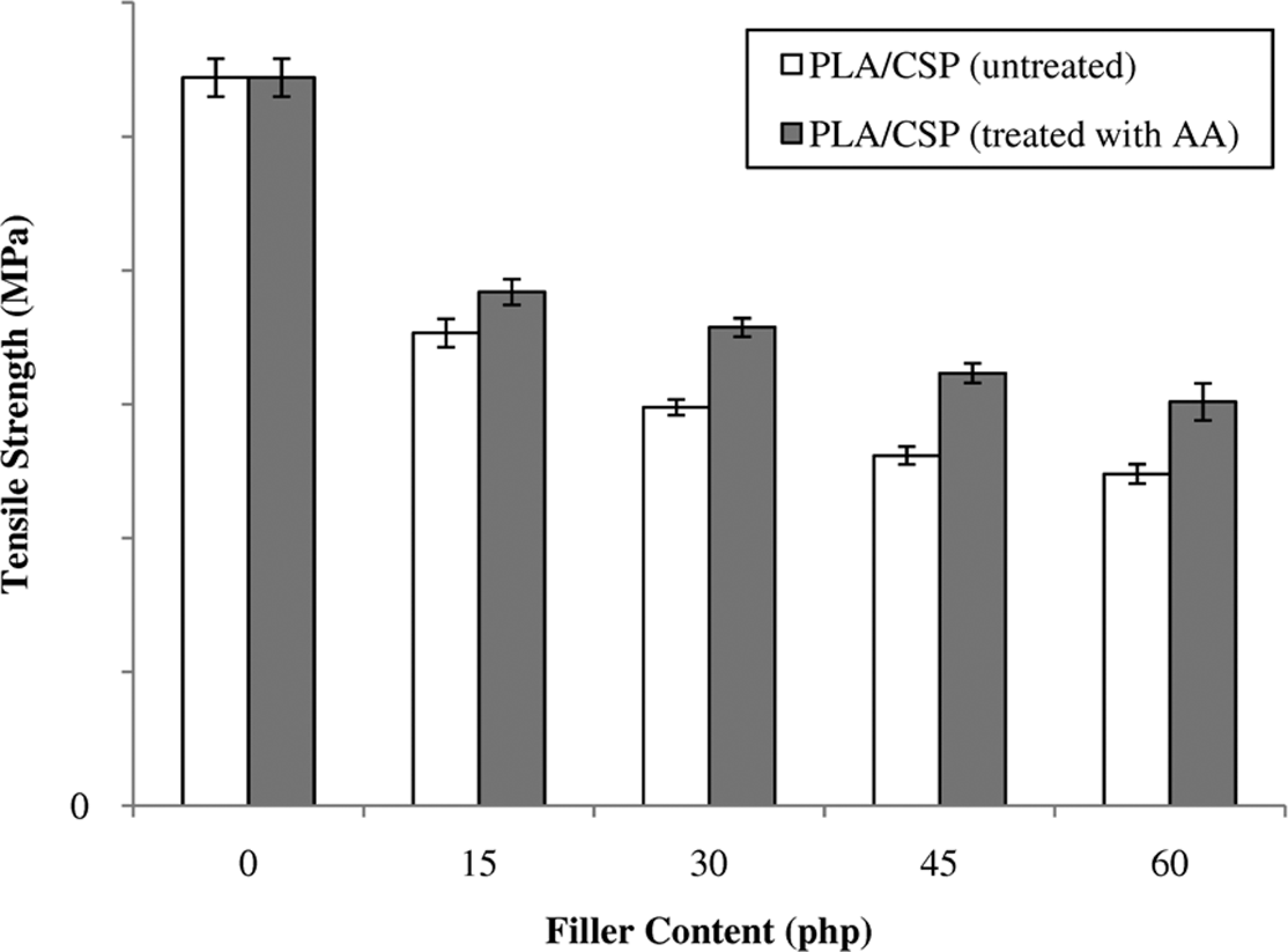
The effect of filler content on the tensile strength of untreated and treated PLA/CSP biocomposites. CSP: coconut shell powder; PLA: polylactic acid.
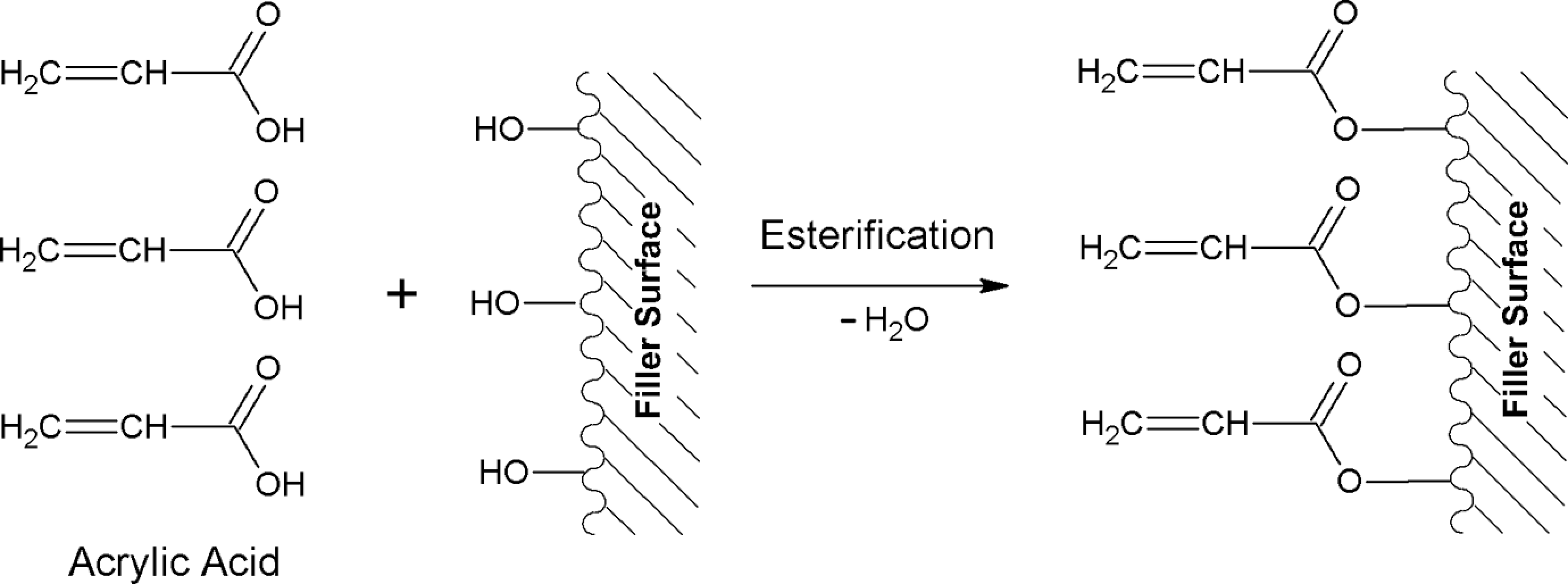
Schematic reactions between coconut shell powder and acrylic acid.
Figure 3 shows the effect of CSP content during elongation at break of untreated and treated PLA/CSP biocomposites. It can be seen that the elongation at break of untreated biocomposites was a dramatic drop at higher CSP content. The brittleness and stiffness of PLA/CSP biocomposites increased with increasing CSP content and reduced the polymer chain mobility. However, the treated PLA/CSP biocomposites with AA exhibit lower elongation at break compared to untreated PLA/CSP biocomposites. The filler surface treatment with AA enhanced the rigidity of biocomposites and decreased the ductility of composites.
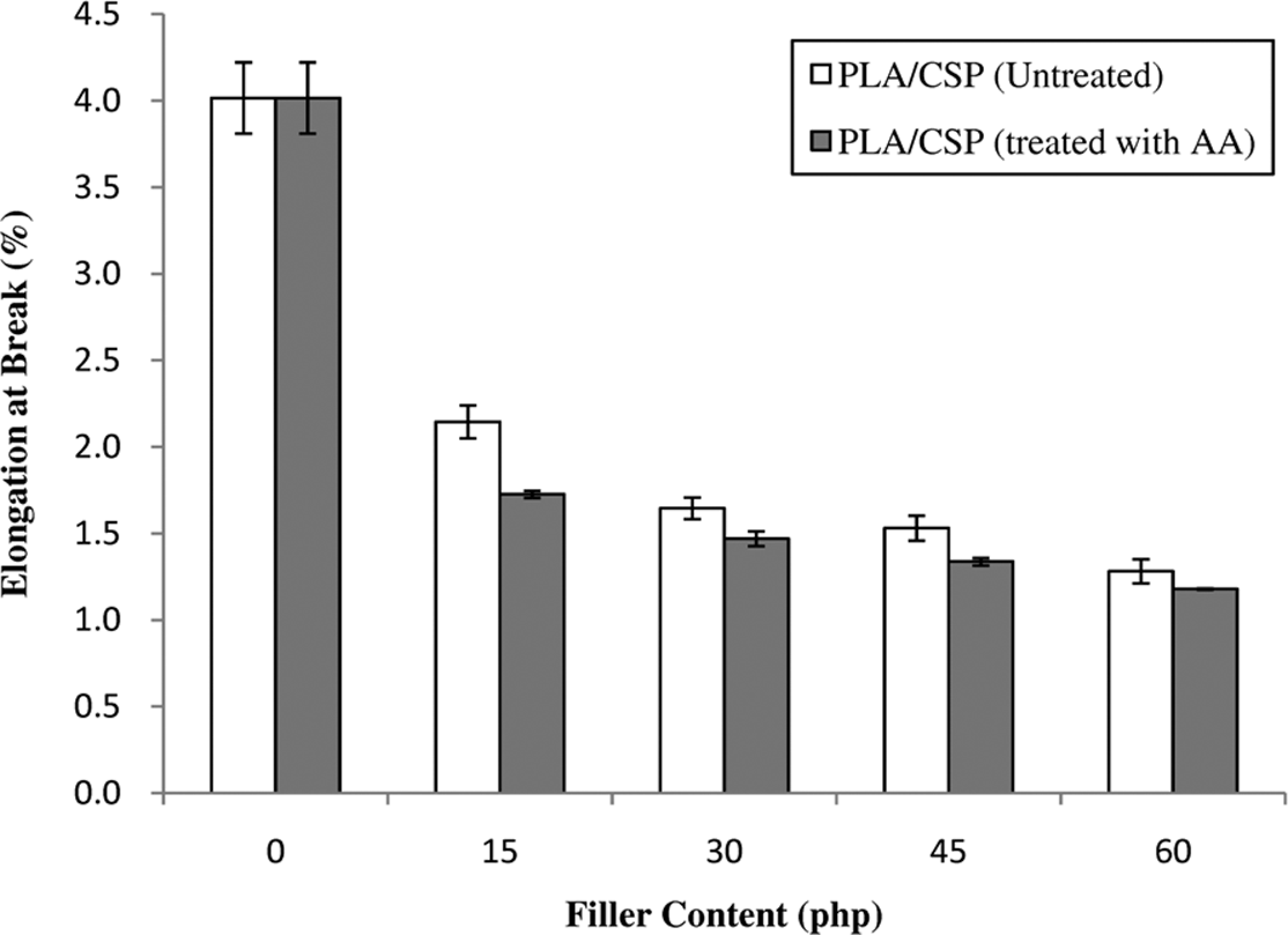
The effect of filler content on elongation at break of untreated and treated PLA/CSP biocomposites. CSP: coconut shell powder; PLA: polylactic acid.
The effect of CSP content on the modulus of elasticity of PLA/CSP is shown in Figure 4. The modulus of elasticity of untreated biocomposites increased with increasing CSP content. It is known that filler which has higher stiffness than matrix can increase the modulus of biocomposites. Figure 4 indicates the treated PLA/CSP biocomposites have higher modulus elasticity than the untreated biocomposites. This result provides evidence that the stiffness of the PLA/CSP biocomposites increases with the chemical treatment.
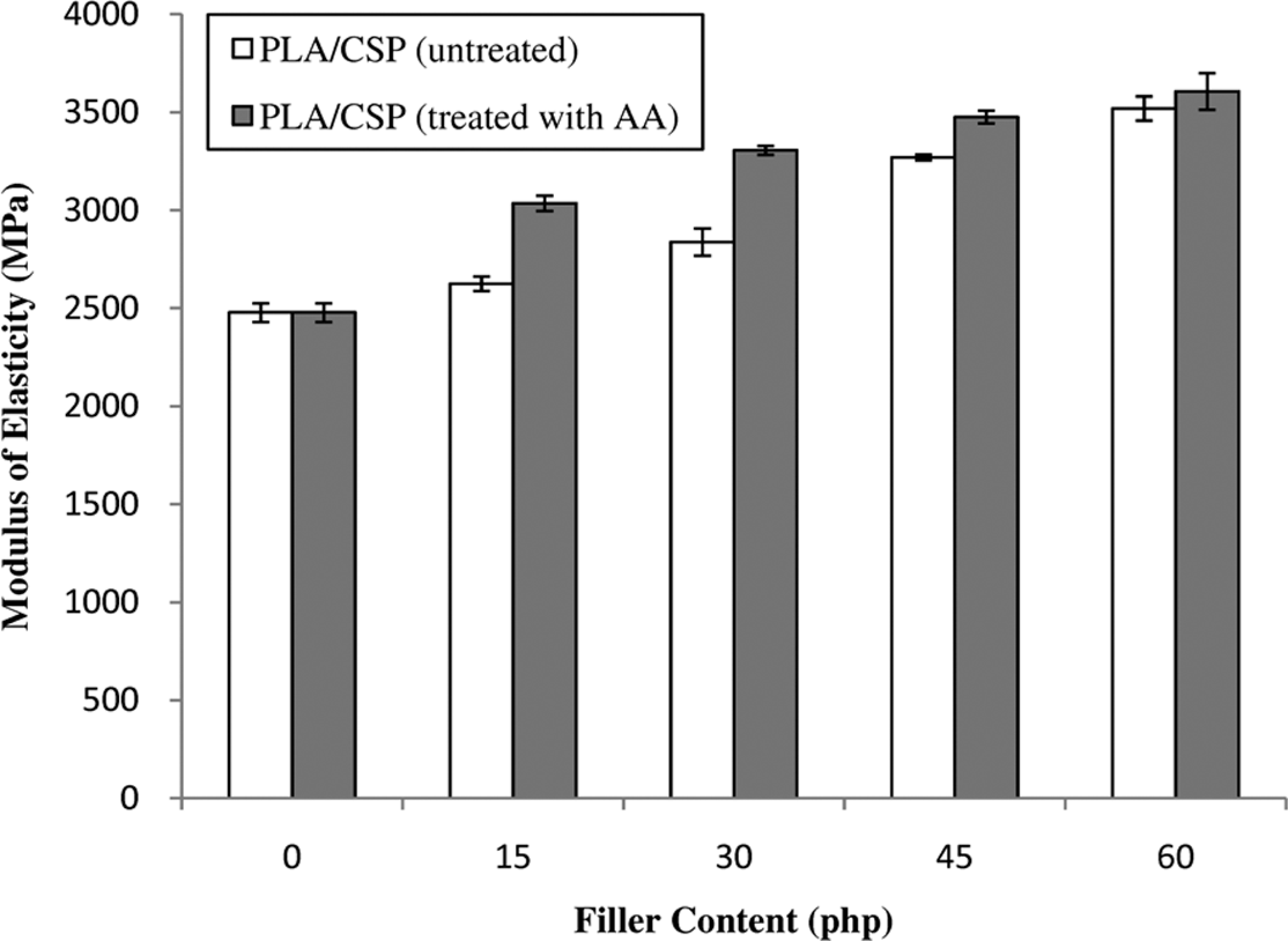
The effect of acrylic acid filler content on modulus of elasticity of untreated and treated PLA/CSP biocomposites. CSP: coconut shell powder; PLA: polylactic acid.
Thermal properties
Thermal degradation curve of untreated and treated PLA/CSP biocomposites with AA is shown in Figure 5. It can be seen from the figure that the degradation temperature of biocomposites reduced with increase in CSP content. Table 2 shows the percentage total weight loss of untreated and treated PLA/CSP biocomposites at different temperatures. Table 2 shows that the total weight loss of PLA/CSP biocomposites decreased with increasing CSP content. At similar filler content, the treated PLA/CSP biocomposites exhibit lower total weight loss than untreated biocomposites. This indicates that the presence of AA increased the thermal stability of PLA/CSP biocomposites.
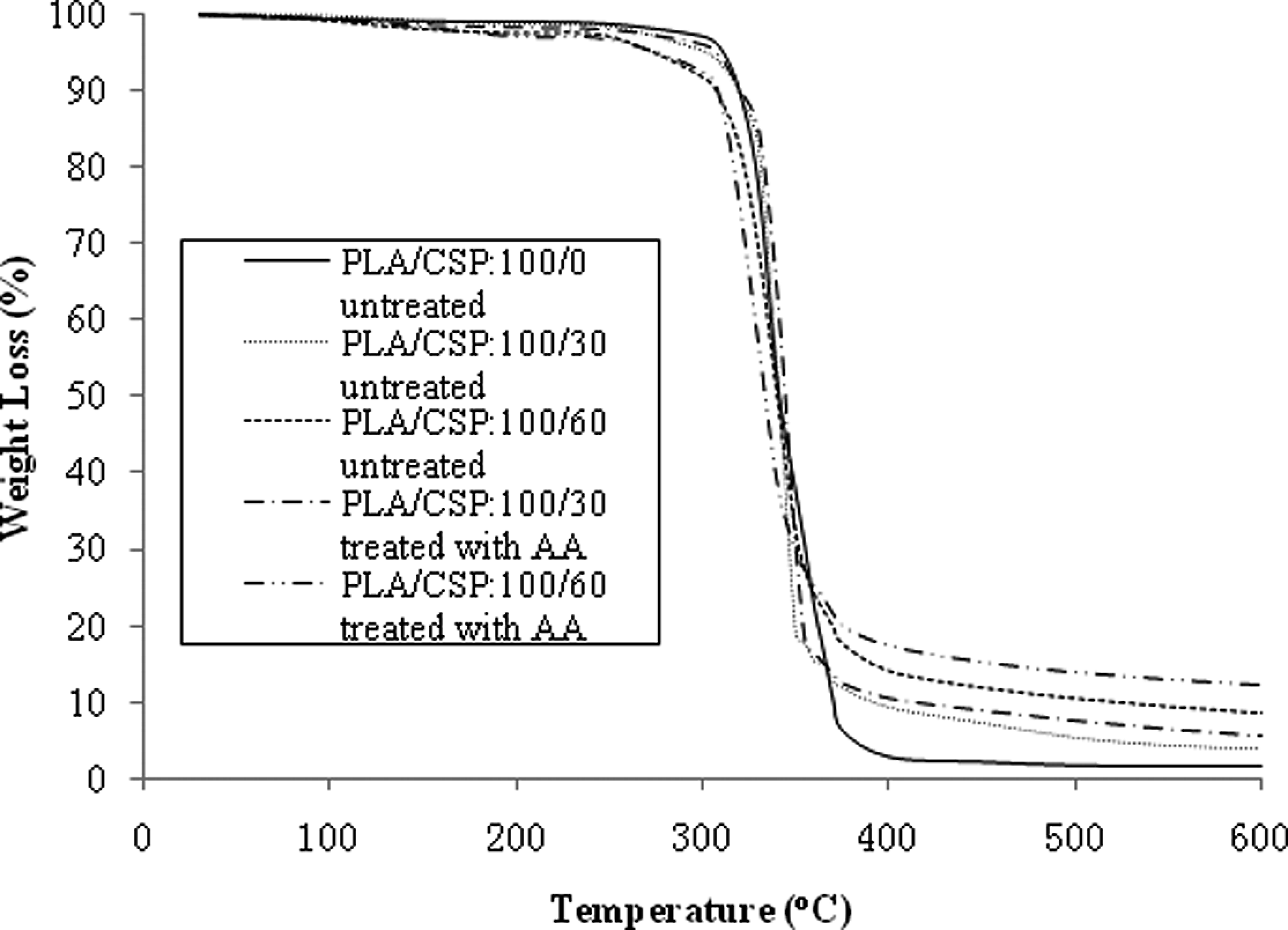
Comparison of thermogravimetric analysis curves of untreated and treated PLA/CSP biocomposites with AA at 0 and 60 php. AA: acrylic acid; CSP: coconut shell powder; PLA: polylactic acid.
Percentage of total weight loss of PLA/CSP biocomposites untreated and treated with AA at different filler loading and range of temperature.
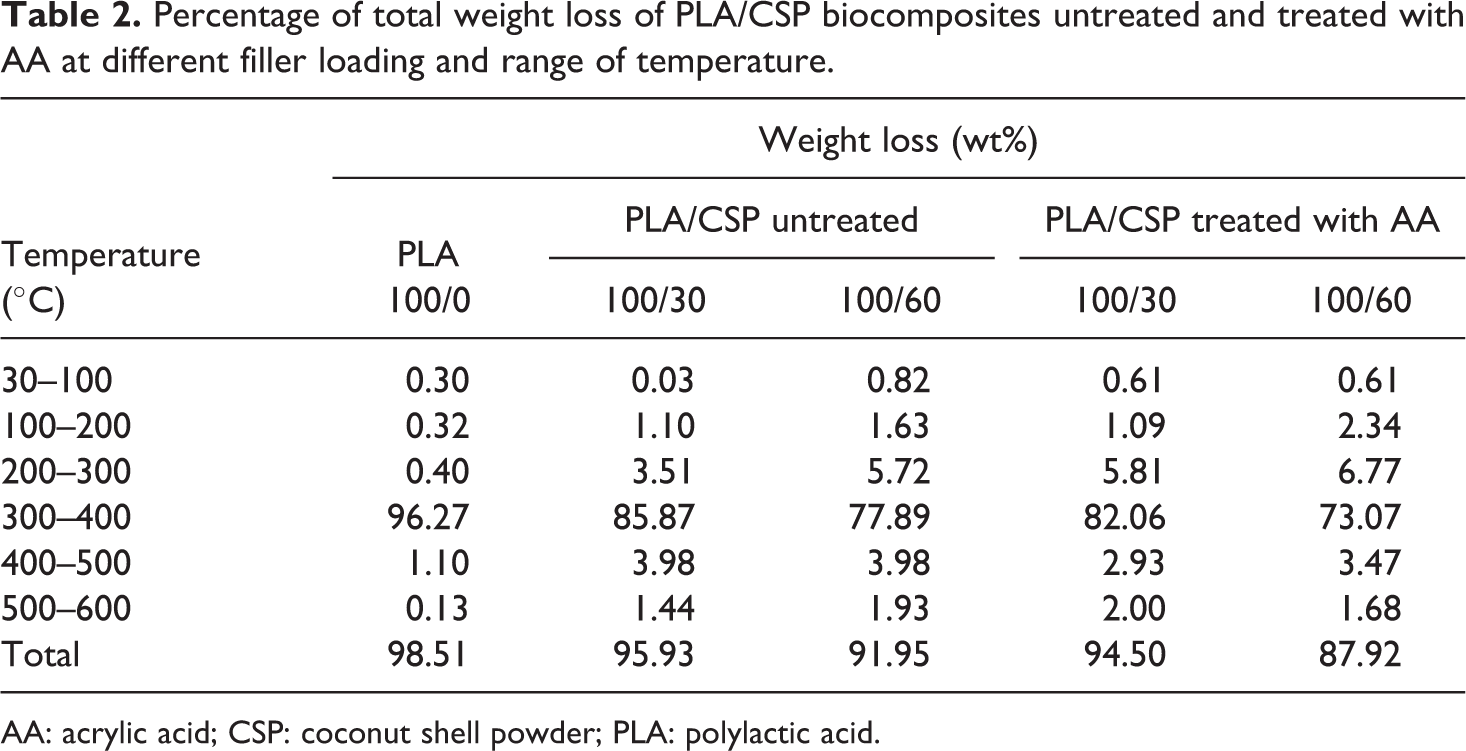
AA: acrylic acid; CSP: coconut shell powder; PLA: polylactic acid.
Morphology study
The micrograph of tensile fracture surface of untreated PLA/CSP biocomposites at 30 and 60 php CSP content are shown in Figures 6 and 7, respectively. Both the micrographs show that the surface exhibits poor dispersion and adhesion of CSP in PLA matrix. The filler was pulled out from the matrix and unwetted CSP in PLA. The agglomeration occurred at higher CSP content (Figure 7). The polar characteristic of CSP is not capable of forming a good filler–matrix interaction with nonpolar PLA biocomposites. The SEM micrograph of treated PLA/CSP in Figures 8 and 9 indicates better wettability and adhesion of CSP in matrix. The filler agglomeration was reduced with AA treatment and improved the interfacial interaction between filler and matrix.

Scanning electron microscope (SEM) micrograph of tensile fracture surface of untreated PLA/CSP biocomposites (30 php CSP) at ×200 magnification. CSP: coconut shell powder; PLA: polylactic acid.

Scanning electron microscope (SEM) micrograph of tensile fracture surface of untreated PLA/CSP biocomposites (60 php CSP) at ×200 magnification. CSP: coconut shell powder; PLA: polylactic acid.

Scanning electron microscope (SEM) micrograph of tensile fracture surface of treated PLA/CSP biocomposites (30 php CSP) at ×200 magnification. CSP: coconut shell powder; PLA: polylactic acid.

Scanning electron microscope (SEM) micrograph of tensile fracture surface of treated PLA/CSP biocomposites (60 php CSP) at ×200 magnification. CSP: coconut shell powder; PLA: polylactic acid.
Conclusion
The incorporation of CSP decreased the tensile strength and elongation at break but increased the modulus of elasticity and thermal stability of PLA/CSP biocomposites. The surface modification of filler using AA improved the tensile strength, modulus of elasticity, and thermal stability of PLA/CSP biocomposites. SEM studies indicate that the interfacial adhesion between CSP and PLA matrix improved in the presence of AA.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
