Abstract
Recently, many agricultural waste were left for open burning or as landfill in many agricultural land, for example, coconut shells. Besides, expanded polystyrene (EPS) foam is one of the most used plastic products which mostly ends up in landfilling as many of its products were made for one-time use only. In order to reduce these wastes, this project focuses on the preparation of a new material that is coconut shell powder (CSP)-filled EPS wood plastic composite (WPC). This research investigates the effects of CSP content and alkaline treatment with sodium hydroxide (NaOH) on mechanical and water absorption properties of EPS/CSP WPCs. The mechanical properties, such as tensile strength, Young’s modulus and flexural modulus, increased as the CSP content increased. However, elongation at break and flexural strength decreased with CSP content. Moreover, water absorption of EPS/CSP WPCs showed an increasing trend with CSP content. After alkali treatment, the mechanical properties of EPS/CSP WPCs were enhanced, but the water absorption properties of treated EPS/CSP WPCs were reduced due to the reduction of the –OH groups of CSP. Fourier transform infrared analysis was carried out to measure the functional groups of untreated and treated CSP. The fracture surface of CSP, untreated and treated EPS/CSP WPCs were examined by scanning electron microscopy.
Introduction
Nowadays, the annual plastic production is more than 300 million metric tons in the globe. Plastics provide multiple benefits as they are lightweight, cost-effective and durable as compared to many other material types. 1 However, about 50% of plastics made are designed to be disposed within a year, for example, food packaging, packaging tray plastic bags and disposal tableware. 2 Plastic waste has become a serious problem for a long time. Furthermore, plastic waste can be disposed of in a few ways such as incineration and landfilling. Nevertheless, these disposal methods are not effective and efficient for plastic disposal because these methods will cause environmental pollution. 2
Expanded polystyrene (EPS) foam is a lightweight and portable material that consists of about 98% of air with 2% of plastic. 3 It is often used as construction’s heat insulator and packaging material. In addition, EPS waste is increasing in the globe because EPS products are widely used. 4 Accordingly, more than 3 million metric tons of EPS foam were used and annually the number of EPS waste increases about 6%. 5 In addition, the EPS foam has a short service life and it mostly ends up in either incineration or landfill. 4,6 Recycling rate of EPS foam is very low and expensive. Thus, most EPS foam wastes are sent to landfill and they contribute to environmental pollution, as they are non-biodegradable plastics. Both incineration and landfilling of plastic wastes would cause serious consequences to the environment because these processes would emit toxic gases and cause air pollution. 7,8 EPS foam can be recycled through a mechanical recycling method and then creating a new product by converting the EPS foam to “scrap” polymer. However, this method is more expensive as compared to producing the virgin plastic. 9 Recently, a new method of recycling EPS foam by dissolving it into a solvent to retrieve its styrene monomer and reusing it as a chemical resource or polymer matrix was introduced. 10
Agricultural waste is a decayed waste. Agricultural biomass causes environmental menace due to leachate and methane emission. Open burning of agricultural waste generates carbon dioxide which is harmful to the environment and also human health. 11 Agricultural waste is broadly available, virtually free and renewable. It can be converted into ethanol, steam, charcoal and heat. However, agricultural wastes are under-utilized. Many were either left to decay or open-burned. 12 Malaysia is located on fertile farmland with high productivity in the farm industry. However, Malaysia’s annual agricultural wastes are over 2 million tons as Malaysia has a high productivity in agricultural products. 11 In general, fine wood is used as a filler to produce wood plastic composites (WPCs). Currently, there are many types of natural filler made from agricultural wastes, such as kenaf fibre, empty fruit bunch fibre, rice husk, wood flour and coconut shell that can replace fine wood to produce WPCs.
Coconut (Cocos nucifera) shells are one of the agricultural wastes in Malaysia. 11 Solid waste from coconut shells causes huge trouble to the world’s pollution due to the massive amount of waste produced. There are 3.18 million tons of coconut shell waste produced annually, which contributes to 60% of world’s waste volume. 13 Coconut shell is a natural lignocellulosic fibre, which has great potential to act as reinforcement in WPCs. Coconut shells can be used as fillers in developing a new WPCs as it has high modulus and strength. The WPCs strength will increase when the coconut shell powder (CSP) content increases. 14 WPCs have good properties and are cheaper than neat plastics. 15 However, the mixture of plastics and fibres possesses a huge problem, which is poor in interfacial adhesion between matrix and filler and low moisture resistance. This issue will decrease the mechanical properties of WPCs. Alternatively, a natural filler can be modified by chemical treatment to enhance the filler–matrix adhesion and hence improve WPCs properties. 16 The most common chemical treatment on natural fillers is alkaline treatment using sodium hydroxide (NaOH). Alkaline treatment of filler increases tensile strength, interfacial shear strength, Young’s modulus, impact strength, flexural strength and moisture resistance of WPCs. 17
Presently, the research is focused on the preparation of WPCs from EPS and CSP. Besides, this research also involves the minimization of large EPS volume by adding chemical solvent to dissolve the EPS and produce styrene monomers. After dissolving the EPS, CSP is added into the styrene monomers to create new EPS/CSP WPCs. In addition, the effect of EPS content and NaOH treatment on the mechanical and water absorption properties of EPS/CSP is investigated.
Methodology
Materials
EPS and CSP are the raw materials used in this research. EPS was used as polymer matrix and CPS was used as a natural filler to produce WPCs. EPS waste was collected from an electronic company (One Living Sdn. Bhd., Malaysia) and CSP was purchased from Sekimdi Industries Sdn. Bhd., Malaysia. The CSP had a particle size of 176 µm. The acetone and NaOH were purchased from Evergreen engineering & resources, Malaysia. Acetone was used to dissolve EPS and form styrene monomer, whereas NaOH was used for alkaline treatment on CSP.
Methodology
Alkaline treatment of CSP
The chemical modification of CSP was done with NaOH treatment method. This alkali treatment method is similar to the method used by Sareena et al. 18 Firstly, the CSP was soaked with 1 mol of NaOH solution for 1 h. Then the mixture was washed and rinsed with distilled water until the pH value of CSP was 7. After that, the CSP was filtered out and oven dried at 60°C for 24 h.
Preparation of EPS/CPS WPCs
The EPS/CSP WPCs were produced through casting and pressing method similar to the technique by Akinyemi et al. 19 First, the EPS was dissolved in acetone. Then, CSP was mixed with EPS solution using a mechanical stirrer at a speed of 250 r/min for 3 min. The homogeneous mixture of EPS and CSP was poured into a metal plate and the mixture was pressed to the desired dimension. The mould was placed at room temperature to dry for 7 days. The formulations of untreated and treated EPS/CSP WPCs are shown in Table 1.
Formulations of untreated and treated EPS/WPCs.
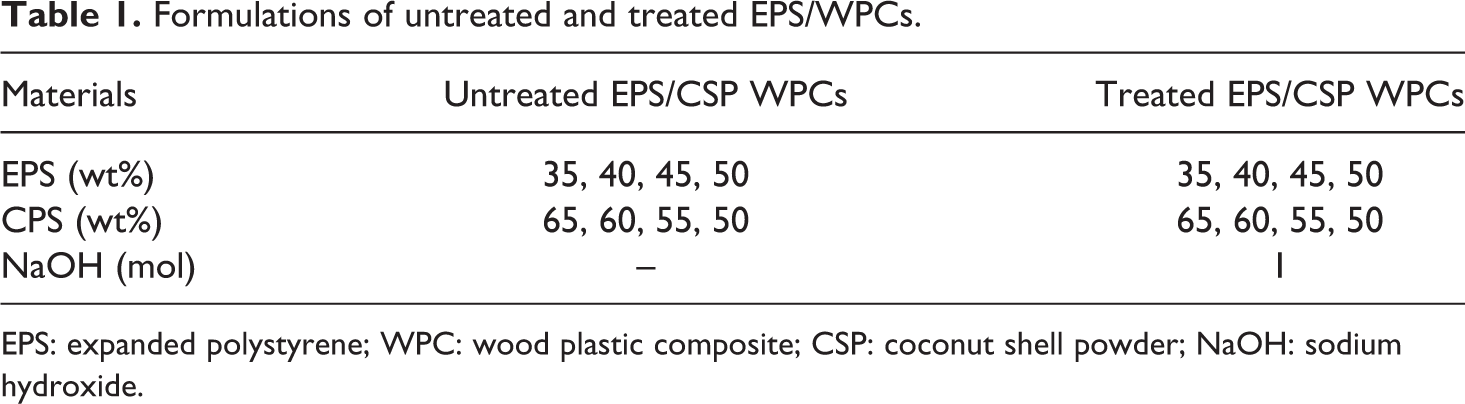
EPS: expanded polystyrene; WPC: wood plastic composite; CSP: coconut shell powder; NaOH: sodium hydroxide.
Testing and characterization
Fourier transform infrared analysis
Fourier transform infrared (FTIR) spectrometer (Perkin Elmer Spectrometer 1000 FT-IR, Waltham, MA, USA) was used to determine the functional groups of treated and untreated CSP with NaOH. A range of 600–4000 cm−1 was scanned and resolution used was 4 cm−1. The 18 scans in the wavelength range were recorded.
Tensile and flexural test
Tensile testing machine (Model VEW 2302, VICTOR Material Testing Equipment, Ho Chi Minh, Vietnam) was used for both flexural and tensile tests. The tensile was used to measure tensile strength, modulus of elasticity and elongation at break of ESP/CSP WPCs, according to ASTM D638. Whereas, the flexural test was used to measure flexural strength and modulus of EPS/CSP WPCs, according to ASTM D790. The dimension of tensile specimens was according to ASTM D638 (120 × 15 × 3 mm3), whereas the dimension of flexural specimens was according to ASTM D790 (100 × 15 × 3 mm3). A cross-head speed of 15 mm/min was used and the testing was performed at room temperature. Fifteen specimens were tested for both tensile and flexural tests to get an accurate average value.
Water absorption test
Water absorption test was carried out according to ASTM D570. For water absorption test, the dimension of specimens were 30 × 15 × 3 mm3. Three specimens from each formulation were tested to get an average value. The thickness and weight of the specimens were measured before being soaked into distilled water and then oven dried at 60°C for 24 h. Then the dried weight of specimens were measured, after which, the specimens were placed in a container filled with distilled water. The container was placed at room temperature. After 25 h, the specimens were taken out from the containers. The water on the surface of specimens were wiped off prior to weighting. Next, the specimens were placed back into the containers and left to soak in the distilled water. This process was repeated by 50, 200, 400 and 900 h. The water absorption of WPCs can be determined using equation (1).
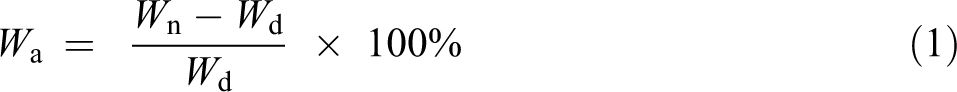
where Wd is the dried weight of specimen and Wn is the weight after soaking of specimen
Scanning electron microscopy
The effect of alkali treatment on the fracture microstructure of WPCs were examined using a scanning electron microscopy (SEM, model JOEL, JSM-6460LA). The fracture surface of each specimen was sputter-coated with a thin layer of palladium for conductive purpose. An acceleration voltage of 5 keV was used to observe the fracture surface of the specimens.
Statistical analysis
Microsoft Excel was used to analyse statistical data from tensile and flexural test of EPS/CSP WPCs. Besides that, analysis of variance (ANOVA) was used to check the statistically significant differences between the untreated and treated WPCs properties. The significance level used was p ≤ 0.05.
Results and discussion
FTIR analysis
Characteristics of treated and untreated CSP with NaOH were analysed by FTIR analysis. The FTIR results for both treated and untreated are exhibited in Figure 1. The peaks at 3387.7 cm−1 (treated) and 3404.9 cm−1 (untreated) show hydroxyl (–OH) groups of CSP. The decreasing of peak intensity at 3387.7 cm−1 (treated) indicated the reduction of the –OH groups in CSP. The peak at 1711.4 cm−1 (untreated) represented the non-cellulosic fibres such as pectin, lignin and hemicellulose of CSP, which were removed after alkaline treatment as the peak intensity reduced. The reduction of peak intensity at 1643.3 cm−1 indicated the reduction of lignin content in CSP. Besides that, the peak at 1242.9 cm−1 (untreated) and 1234.9 cm−1 (treated) represented the hemicellulose content in CSP. The peak intensity of 1234.9 cm−1 in the treated CSP was reduced due to the reduction of hemicellulose content in CSP. Furthermore, peak intensity at 1045.9 cm−1 was reduced, which indicated the removal of the –OH groups from cellulose. Table 2 lists the functional groups in untreated and treated CSP. Similar results were reported by Leonard et al. (2002), who studied the effect of alkali treatment on different natural fibre such as hemp, sisal, jute and kapok. 20 Table 2 summarizes the different functional group of untreated and treated CSP.
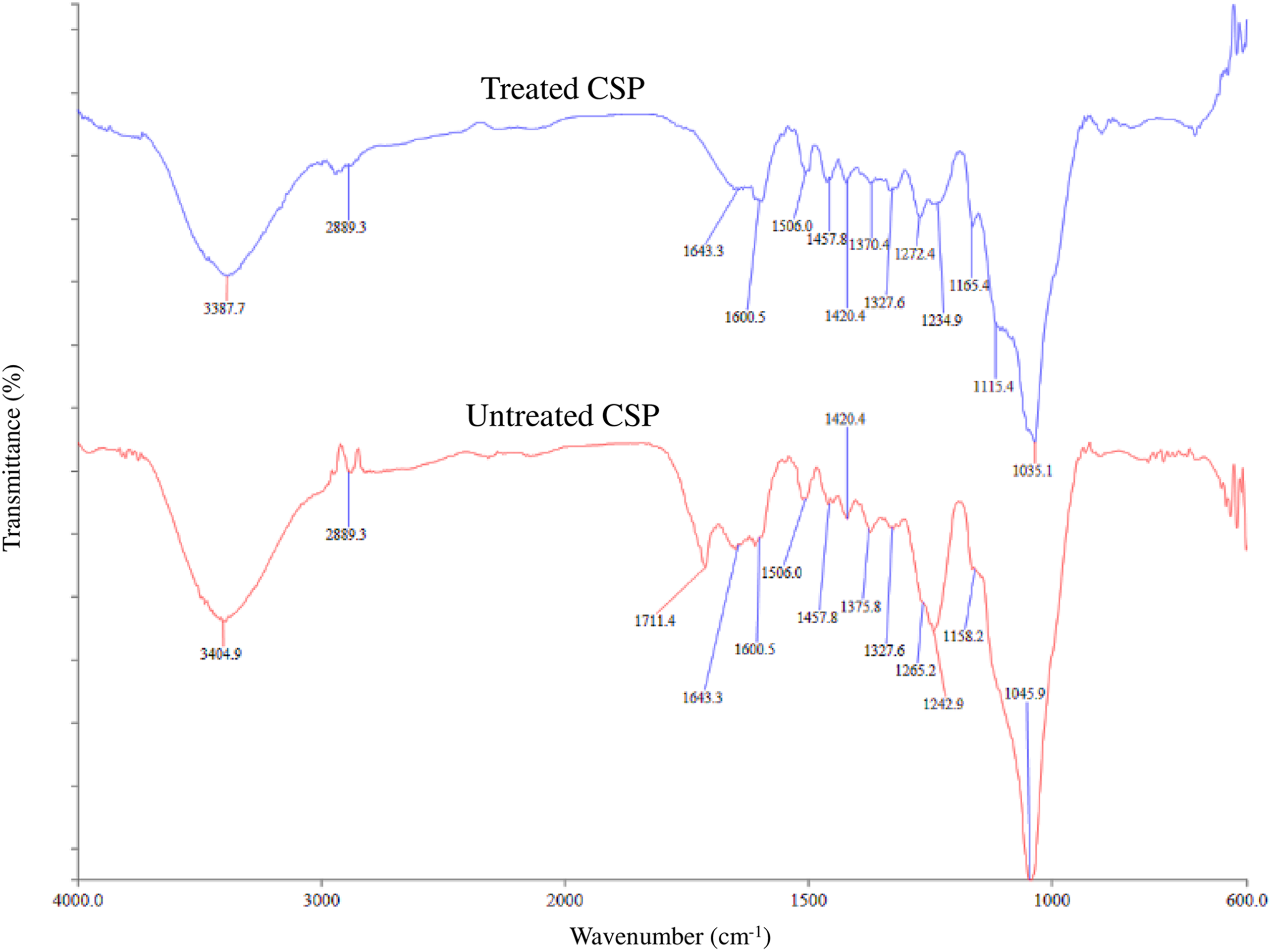
FTIR analysis of treated and untreated CSP by NaOH. FTIR: Fourier transform infrared; CSP: coconut shell powder; NaOH: sodium hydroxide.
Functional groups in untreated and treated CSP.
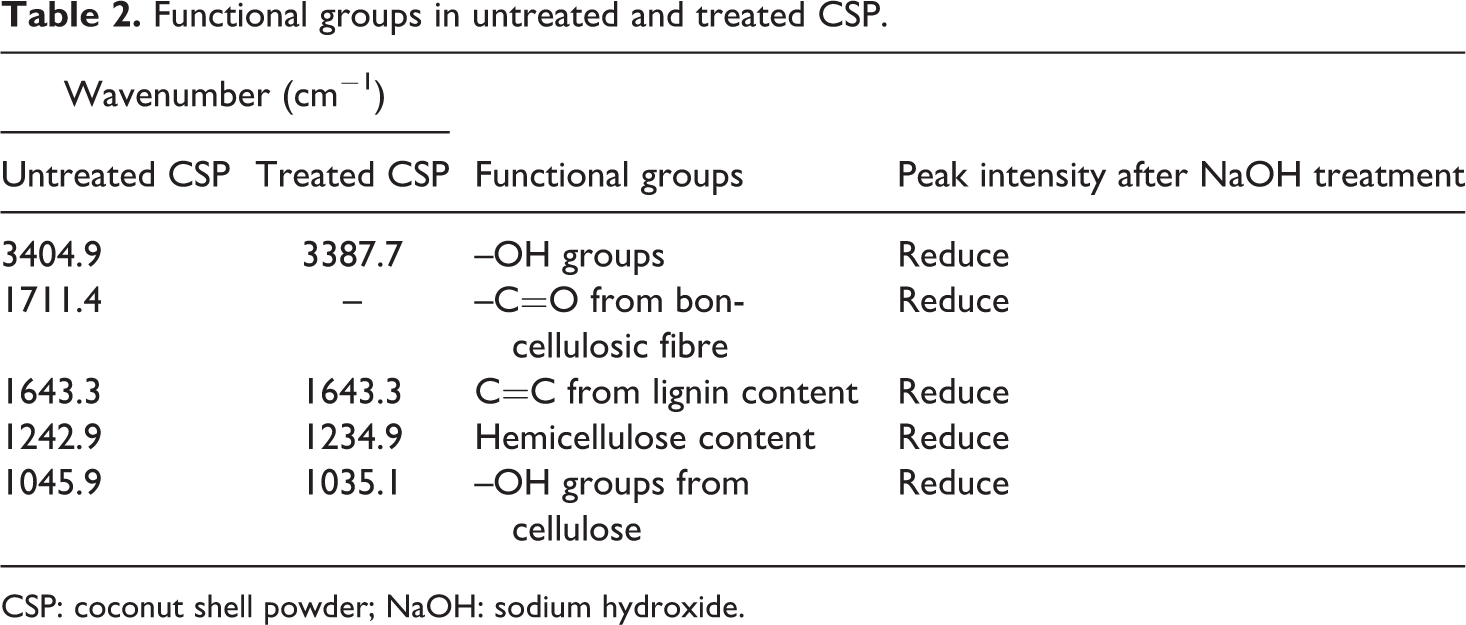
CSP: coconut shell powder; NaOH: sodium hydroxide.
Tensile properties
Figure 2 represents the tensile strength of both untreated and treated EPS/CSP WPCs. The result shows an increasing trend from 50 to 65 wt% of CSP content for both untreated and treated EPS/CSP WPCs. This was due to the incorporation of high lignocellulose CSP filler into polymer matrix. The high lignin and cellulose content in CSP enhanced the tensile strength of WPCs. According to Kim et al., 21 lignin plays an important role in plant cell wall because it works as a stiffening agent in cellulose molecules. A similar trend was claimed by Husseinsyah and Mostapha. 22 However, the treated EPS/CSP WPCs possessed better tensile strength than untreated EPS/CSP WPCs. The tensile strength of treated WPC increased (p > 0.05) about 25–30% as compared to untreated WPCs. These changes happened because of the compatibility between EPS and CSP. EPS is a hydrophobic material while CSP is a hydrophilic material, both materials are incompatible with each other due to poor wetting between hydrophobic and hydrophilic materials, resulting in the reduction of the interfacial bonding between EPS and CSP. However, after alkaline treatment, the CSP possessed a lower –OH concentration and a rougher surface as compared to untreated CSP. The rough surface of treated CSP created a better interlocking mechanism with EPS which allows a better interfacial bonding and surface wetting with EPS and in return increases the tensile strength of EPS/CSP WPCs. In general, the alkaline treatment would exhibit two effects on the fibre such as: (i) it increases surface roughness and thus increasing the mechanical interlocking between fibre and matrix; and (ii) it increases the amount of cellulose exposed on the fibre surface and hence increasing the number of possible reaction sites. 23 Wong and Chan also mentioned that the rough surface of filler can create a better interlocking mechanism with the polymer matrix, in order to improve filler–matrix interaction. 24 Similar results were obtained by Thaker et al. 25 In their research, they investigated the effects of alkaline treatment on the tensile strength of WPCs. They discovered that the tensile strength of the treated WPCs was higher than the untreated WPCs due to the alkaline treatment.
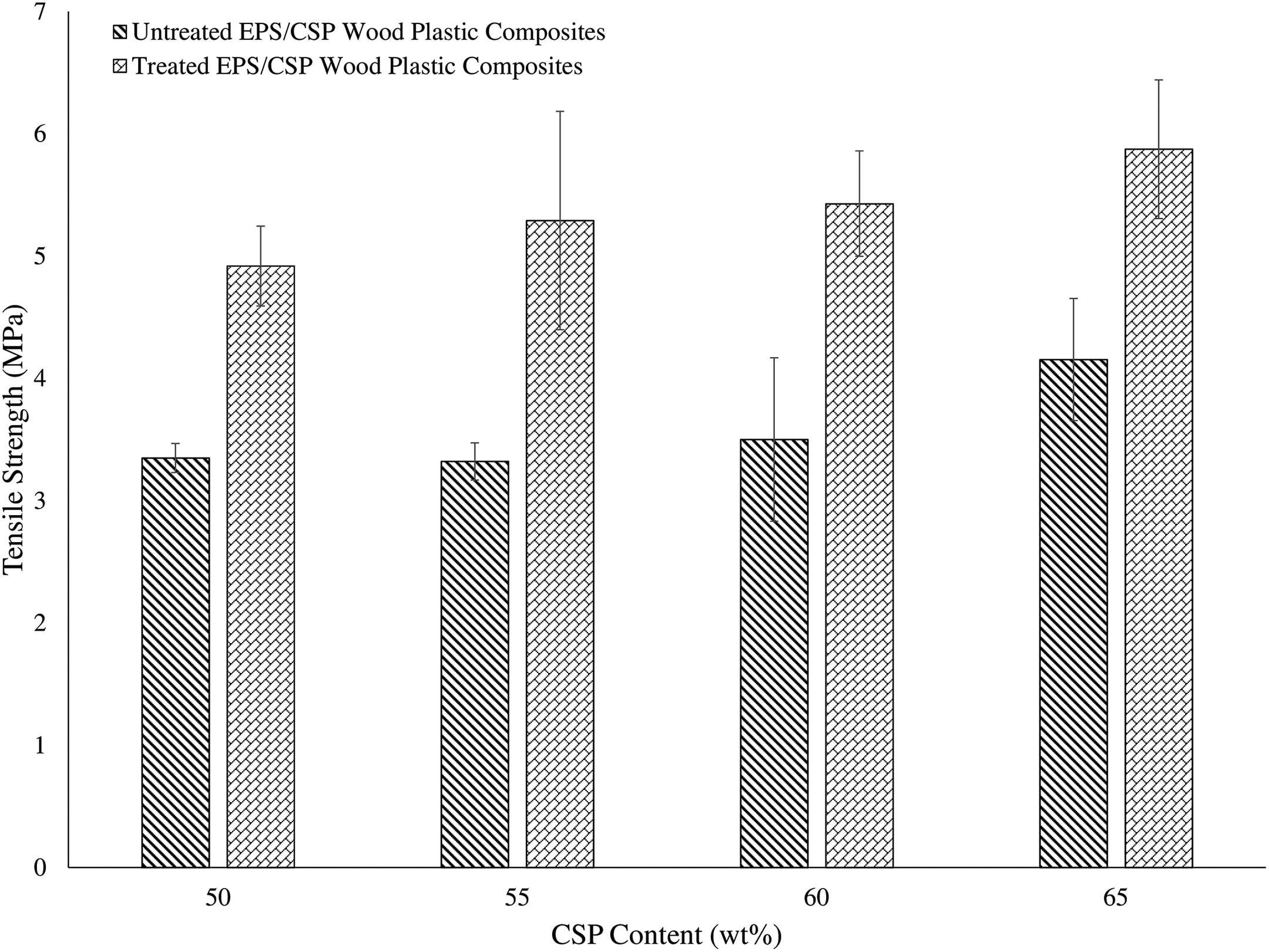
Tensile strength of untreated and treated EPS/CSP WPCs. EPS: expanded polystyrene; CSP: coconut shell powder; WPC: wood plastic composite.
The interfacial bonding differences between untreated and treated EPS/CSP WPCs can be determined using a simple model developed by Pukánsky.
26
Major factors which influenced the tensile strength were considered in this model, including: n represents the specimen dimension changes during deformation and when tensile strength raises due to strain hardening and
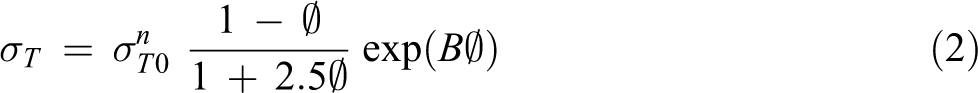

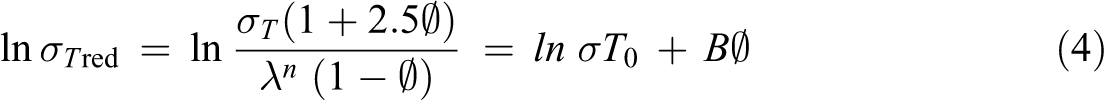
where
The reduced tensile strength against filler fraction graph was plotted and the graph is shown in Figure 3. There is a difference in the gradient line of both untreated and treated WPCs. The untreated WPCs have a B factor of 5.143 and treated WPCs have 6.039. B factor represents the effects of interfacial bonding in the WPCs. The B factor of the treated WPCs was higher than the untreated WPCs. This certified that the treated WPCs possessed better filler–matrix interfacial bonding than untreated WPCs.
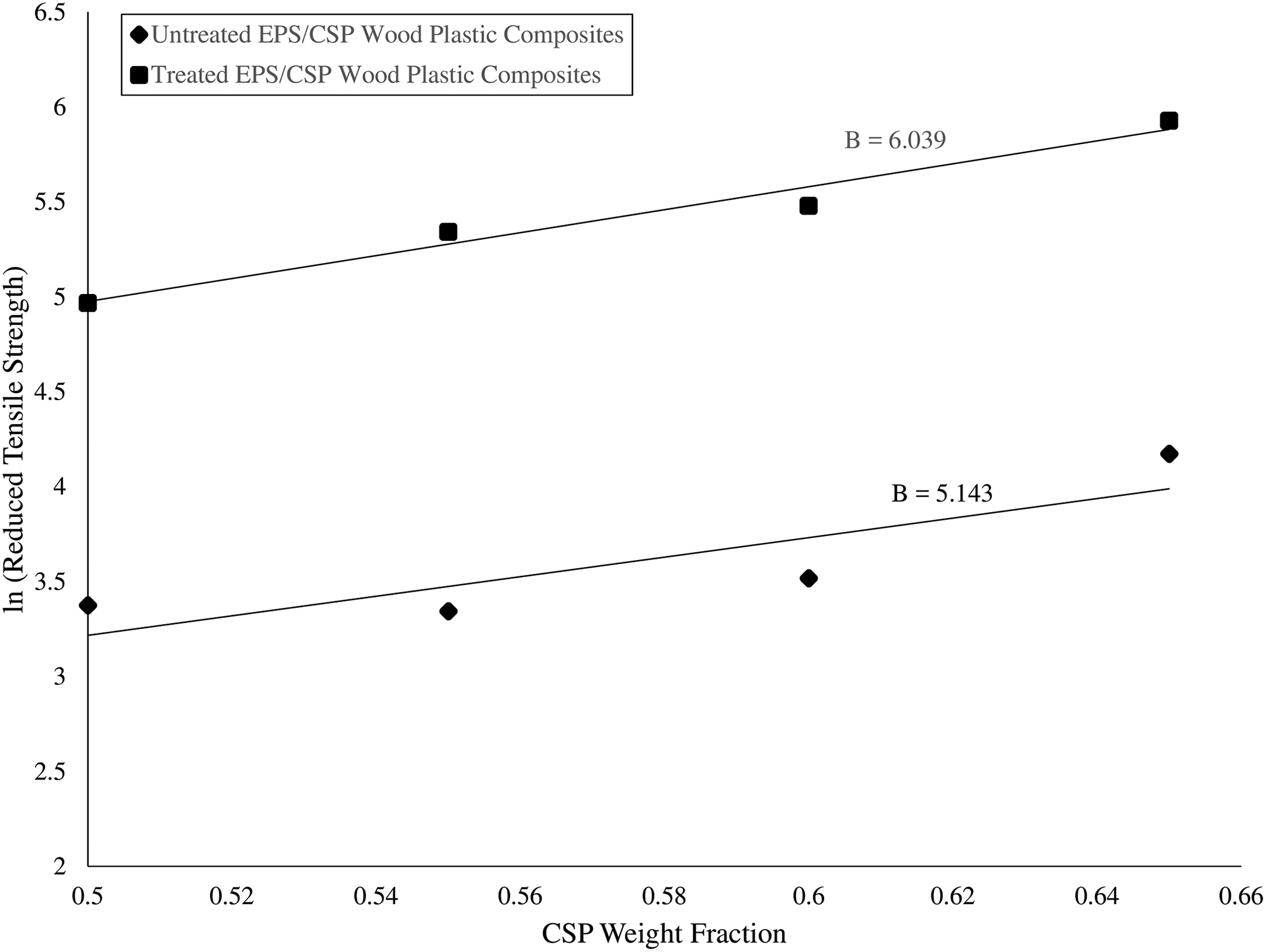
ln (reduced tensile strength) versus CSP weight fraction graph. CSP: coconut shell powder.
Young’s modulus of EPS/CSP WPCs increased as the filler content increases as shown in Figure 4. This act is due to the presence of the mechanical bonding between filler and matrix. The presence of these bonding in the WPCs led to restrict chain mobility of EPS and hence reduced the flexibility of EPS, as well as increased the rigidity of the WPCs. The Young’s modulus of WPCs can also be affected by the high stiffness of filler used in WPCs. Stiffness of filler plays an important role in WPCs 27 because the higher the filler content in WPCs the higher the stiffness of WPCs. Thus, high stiffness of CSP would increase the stiffness of WPCs, resulting in the increase in Young’s modulus of WPCs. On the other hand, treated EPS/CSP WPCs showed an improvement (p < 0.05) in Young’s modulus as compared to untreated EPS/CSP WPCs due to the enhancement of filler–matrix adhesion by the mechanical interlocking mechanism.
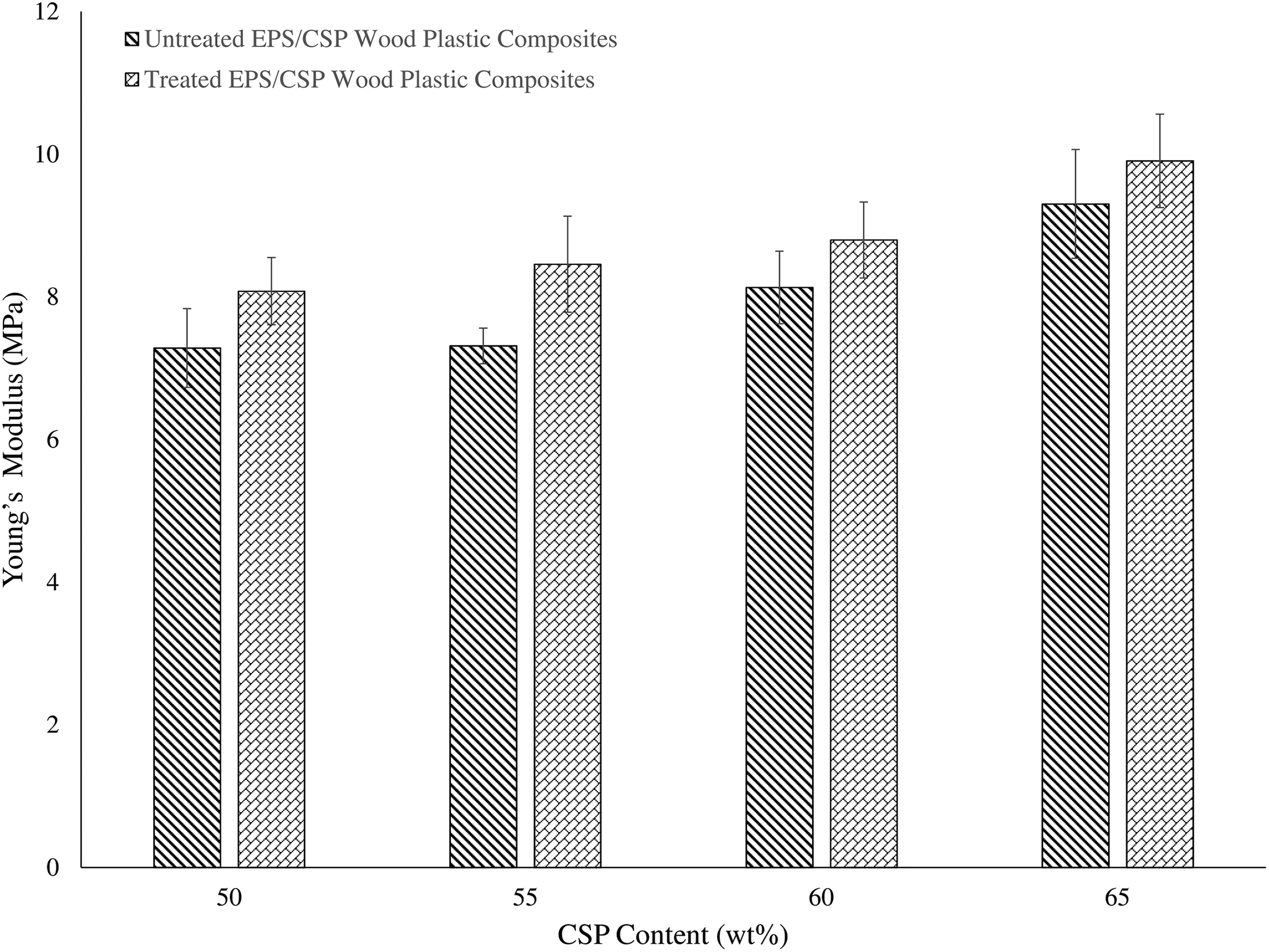
Young’s modulus of untreated and treated EPS/CSP WPCs. EPS: expanded polystyrene; CSP: coconut shell powder; WPC: wood plastic composite.
The elongation at break of both untreated and treated EPS/CSP WPCs is exhibited in Figure 5. The result shows that the elongation at break of untreated and treated EPS/CSP WPCs decreases as the CSP content increases. In general, the amount of polymer matrix used in WPCs determines the deformability of the WPCs because polymer matrix would undergo plastic deformation when the load is applied. The higher the polymer matrix content is used in WPCs, the better the plastic deformation of the WPCs that fracture at higher elongation as compared to the low polymer matrix content of WPCs. However, if more filler content is added into the WPCs instead of polymer matrix, the elongation at break decreased as seen in Figure 5. Besides that, the addition of filler in WPCs restricts the chain mobility of polymer. There is presence of gaps within the polymer chains but with the increase of filler content in the WPCs, these gaps are filled with the filler which in turn reduces the mobility of the WPCs. A similar finding was reported by Salmah et al. 28 In contrast, treated EPS/CSP WPCs showed a higher elongation at break than untreated WPCs. The elongation at break of treated WPCs was significantly increased (p < 0.5) as compared to untreated WPCs. This is due to better filler dispersion in treated WPCs as compared to untreated WPCs. According to Kutz, 29 well dispersion of filler in the composites would enhance and promote elongation at break of composites. Besides that, the fine filler was least deleterious to the elongation at break of polymer and thereby helps to maintain good elongation at break.
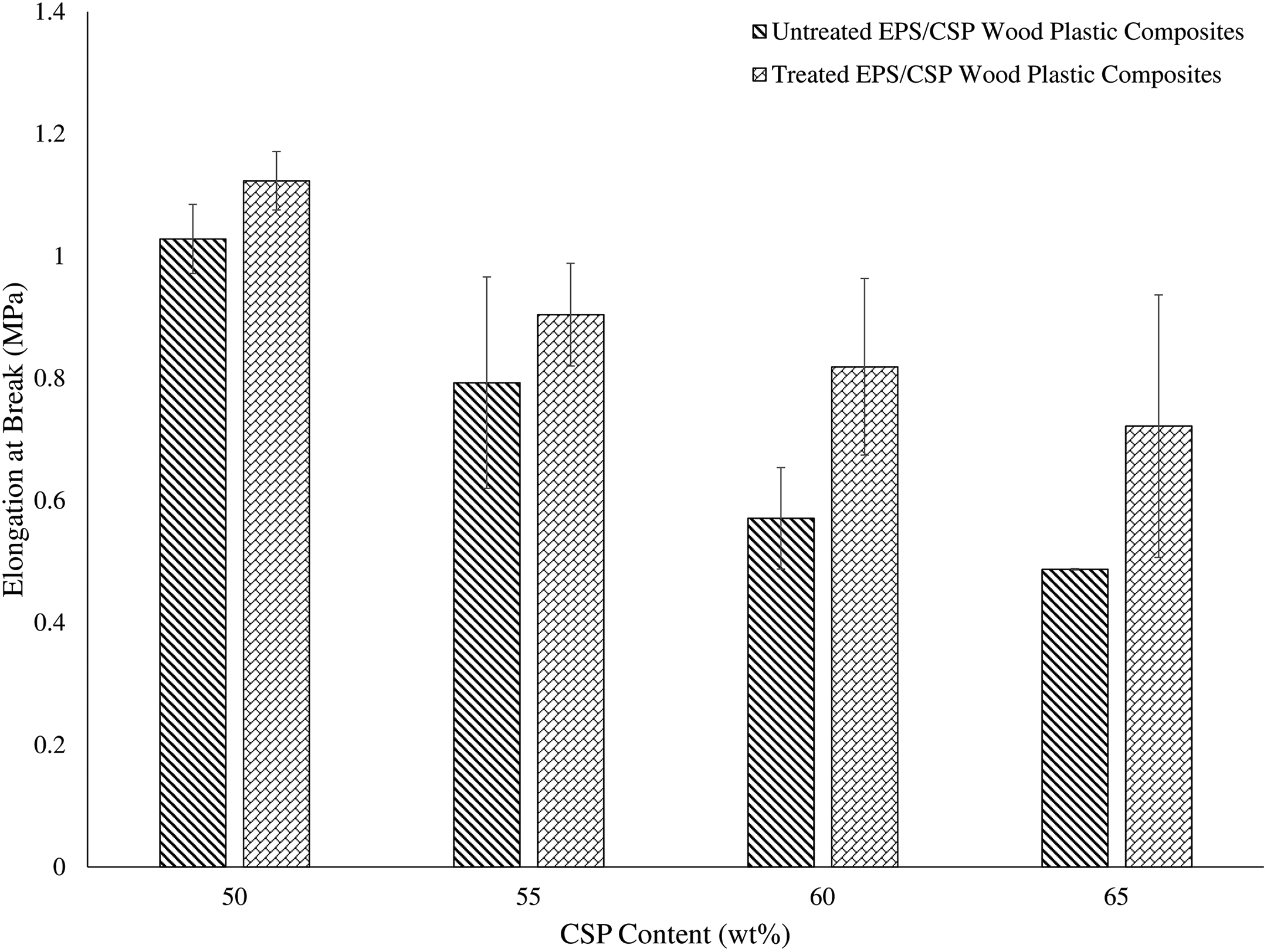
Elongation at break of untreated and treated EPS/CSP WPCs. EPS: expanded polystyrene; CSP: coconut shell powder; WPC: wood plastic composite.
Flexural properties
Figure 6 represents the flexural strength of both untreated and alkaline-treated EPS/CSP WPCs. The graph showed an increasing trend in WPCs as the filler content increased. On the other hand, treated EPS/CSP WPCs by NaOH showed a better flexural strength than the untreated EPS/CSP WPCs due to the removal of non-cellulosic content from the filler which makes the surface rougher and provides a better interfacial bonding with EPS. These results were in line with the results in tensile strength.
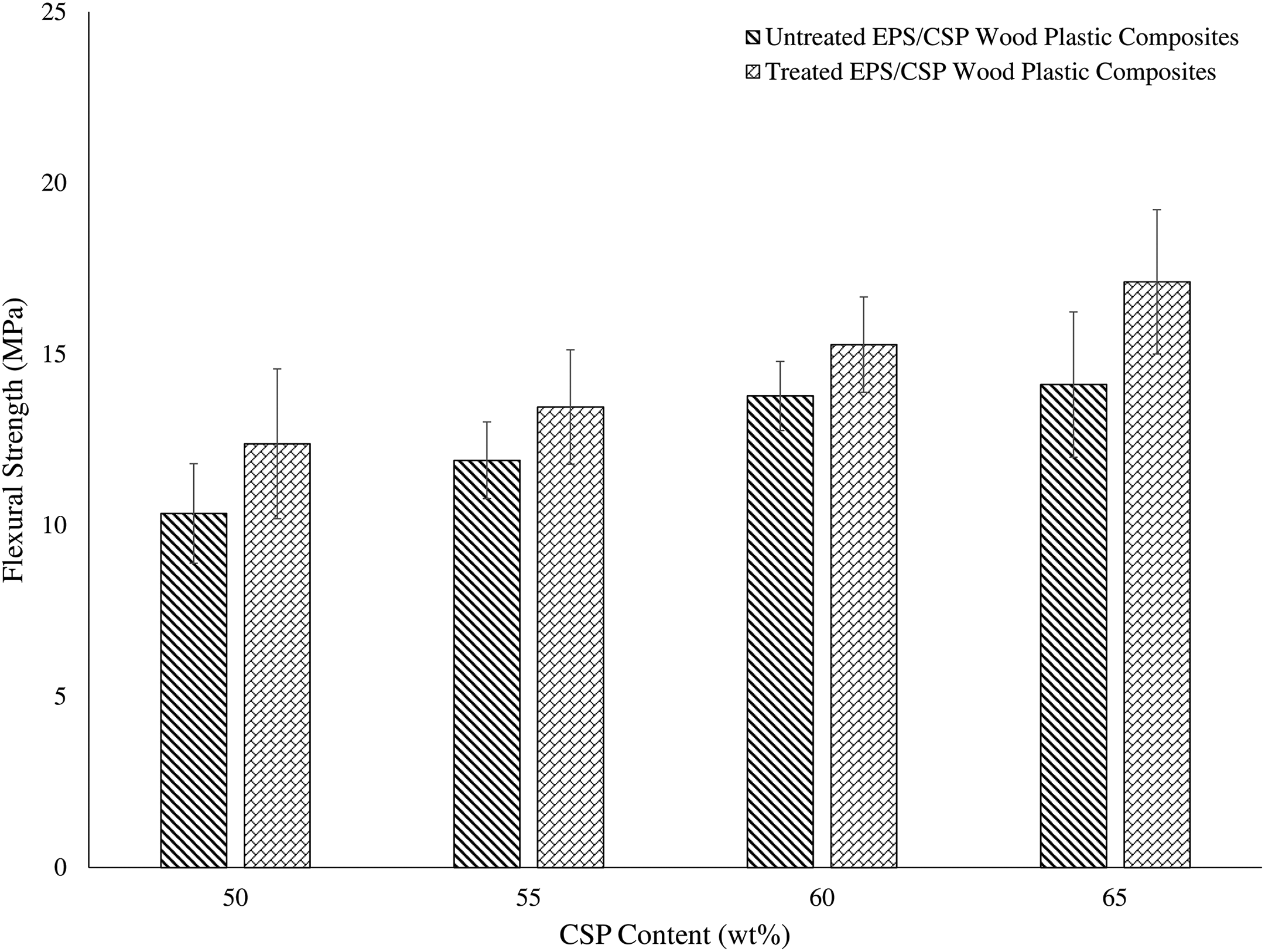
Flexural strength of untreated and treated EPS/CSP WPCs. EPS: expanded polystyrene; CSP: coconut shell powder; WPC: wood plastic composite.
The flexural modulus of EPS/CSP WPCs shows a same trend with flexural strength, as exhibited in Figure 7. The flexural modulus of EPS/CSP WPCs increased with increasing of CSP content. This happened because the CSP filler possesses greater Young’s modulus than polymer matrix. Nevertheless, the treated EPC/CSP WPCs shows higher flexural modulus than untreated WPCs. These results are in line with the Young’s modulus results. A similar trend was claimed by Flores-Hernández (2017), 30 where the author discovered that the flexural modulus of WPCs was directly proportional to filler content.
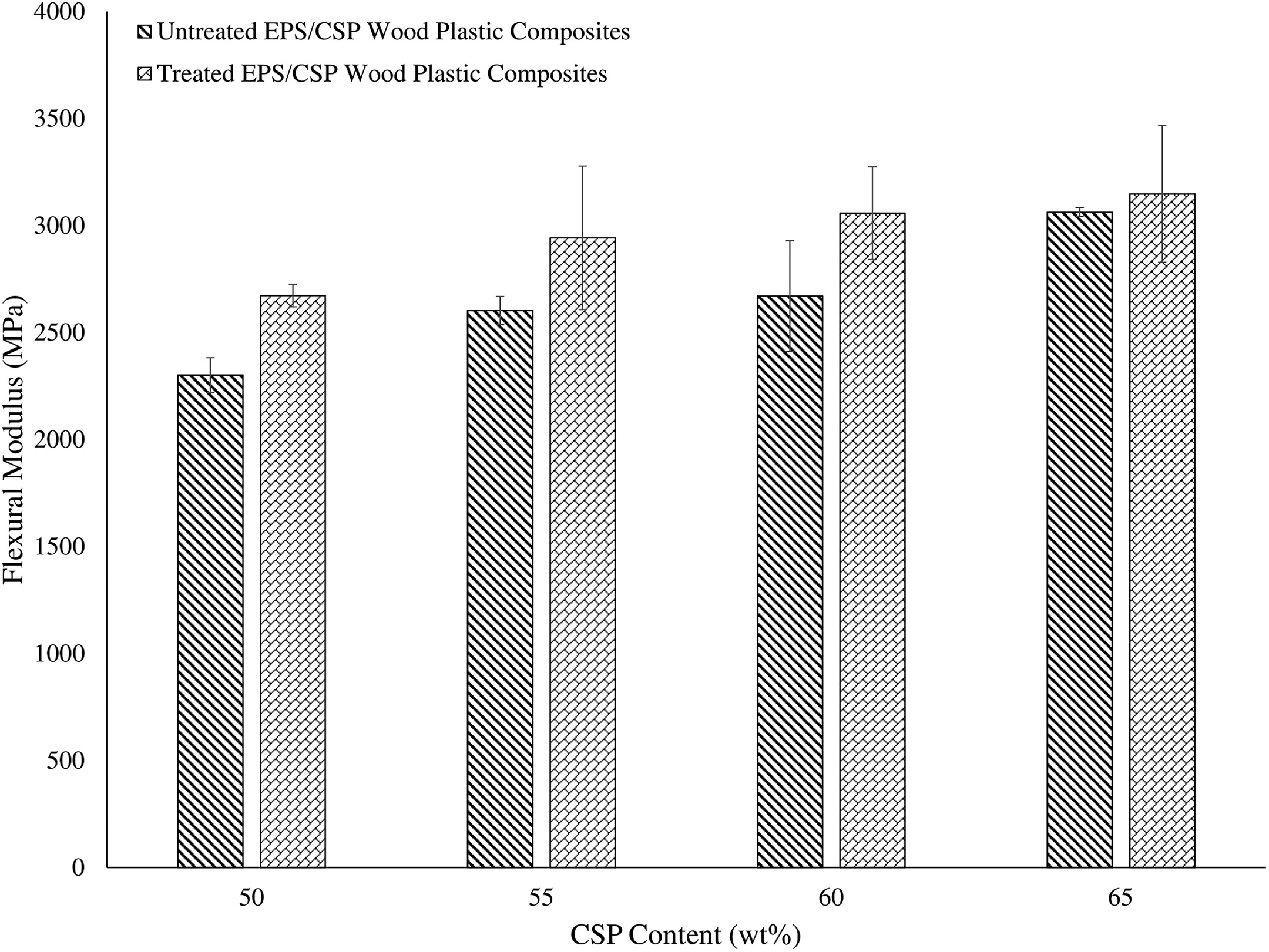
Flexural modulus of untreated and treated EPS/CSP WPCs. EPS: expanded polystyrene; CSP: coconut shell powder; WPC: wood plastic composite.
Scanning electron microscopy
The irregular shape of the coconut shell filler is shown in Figure 8. Figure 9(a) and (b) show SEM micrograph of the tensile fracture of untreated and treated EPS/CSP WPCs, respectively. Based on Figure 9(a), the fillers were well-dispersed in the untreated WPCs, resulting in increasing of tensile strength of WPCs. But, there are few of micro-voids and holes found in SEM micrograph of the untreated EPS/CSP WPCs. However, after NaOH treatment of WPCs, there are no micro-voids and holes in the SEM micrograph, as exhibited in Figure 9(b). This indicated that the alkali treatment improved the filler dispersion and mechanical interfacial interaction between filler and matrix. This also proved that the improvement of tensile and flexural properties of EPS/CSP WPCs was due to NaOH treatment.

SEM micrograph of irregular shape of CSP. SEM: scanning electron microscope; CSP: coconut shell powder.

SEM micrograph of the fracture surface of (a) untreated EPS/CSP and (b) treated EPS/CSP WPCs at 60 wt% of CSP content. SEM: scanning electron microscope; EPS: expanded polystyrene; CSP: coconut shell powder; WPC: wood plastic composite.
Water absorption
The water absorption results of untreated and treated EPS/CSP WPCs are shown in Figure 10. The figure shows that the water absorption ability of WPCs increases as the CSP content increases. Accordingly, the water absorbed in polymer composites can be divided into free water and bound water. Free water is the water molecules contained in the free volume of polymer composites such as micro-voids and holes in the polymer composites. Whereas, the bound water is the water molecules that are reacted with the polar groups of the polymer matrix. 31 Based on Figure 10, this is expected as the increase in CSP content indicated that the increase in water absorption ability of WPCs due to the increasing of hydroxyl groups from CSP. The water absorption occurs not only on the CSP surface. The water can be also absorbed in the cell wall, lumen and the gaps between EPS and CSP due to poor interfacial bonding. The presence of hemicelluloses and cellulose in CSP also affects the water absorption as they contain many accessible hydroxyl groups. The water absorption of WPCs will be lower when the matrix content increase as EPS is a hydrophobic material which does not absorb water. Similar findings were claimed by Iulianelli et al. 32 and Kord. 33 Moreover, the treated EPS/CSP WPCs with NaOH has a lower water absorption rate than the untreated EPC/CSP WPCs. This happened because the untreated CPS is a hydrophilic filler which has many hydroxyl groups (–OH) in its structure. These hydroxyl groups caused the poor adhesion between EPS and CSP. This leads to the formation of hydrogen bonding between water molecules and CSP, resulting in high water absorption and low water resistance of untreated WPCs. However, treated EPC/CSP WPCs shows lower water absorption because the hydroxyl group in CSP are reduced after alkaline treatment. As the hydroxyl groups are reduced, the water resistance of WPCs become higher, similar findings were reported by Thaker et al. 25 A similar result was also found by Munirah and Ahmad. 34 They claimed that the chemical treatment on composites possessed higher water resistance than untreated composites. Table 3 summarizes the percentage of water absorption in untreated and treated EPS/CSP WPCs.
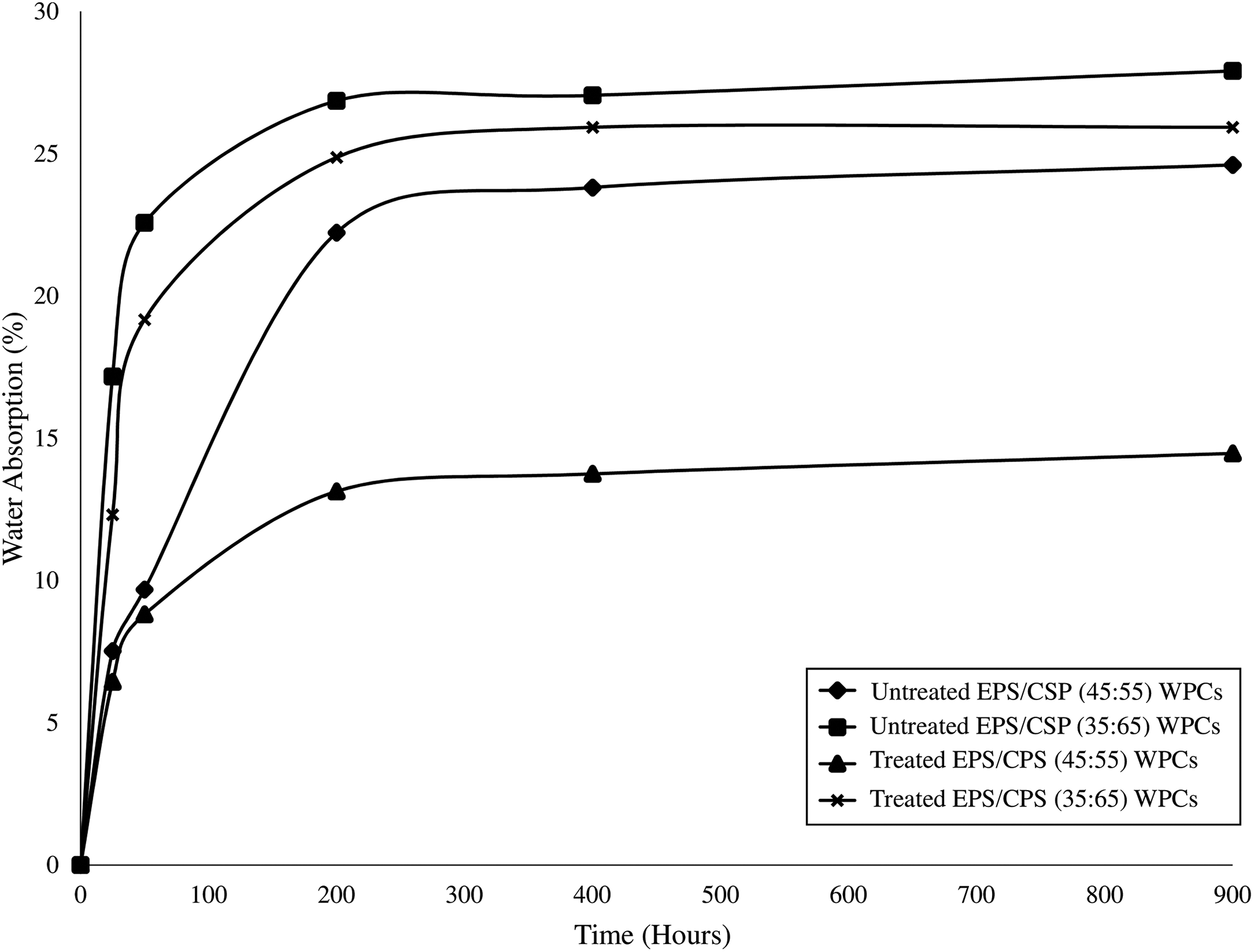
Percentage water absorption of untreated and treated EPS/CSP WPCs. EPS/CSP WPCs. EPS: expanded polystyrene; CSP: coconut shell powder; WPC: wood plastic composite.
Percentage water absorption of untreated and treated EPS/CSP WPCs.

EPS: expanded polystyrene; CSP: coconut shell powder; WPC: wood plastic composite.
Conclusion
In conclusion, the tensile strength, Young’s modulus, flexural modulus and percentage water absorption of EPS/CSP WPCs increased with the CSP content. In contrast, the elongation at break and flexural strength of WPCs decreased with the increasing CSP content. An alkaline treatment was conducted with the use of NaOH to remove lignin, hemicellulose and impurities in order to create a rough surface of CSP filler. The treated EPS/CSP WPCs possessed better results as compared to untreated EPS/CSP WPCs in terms of tensile strength, Young’s modulus, elongation at break, flexural strength and flexural modulus. However, treated WPCs showed a lower water absorption than untreated WPCs.
