Abstract
Thermoplastic vulcanizate (TPV) made from the ethylene-vinyl acetate copolymer (EVA)/nitrile rubber (NBR) blend was fabricated using the dynamic vulcanization method. Differential scanning calorimetry (DSC) was utilized to analyze the nonisothermal crystallization kinetics of both pure EVA and EVA/NBR TPV. The nonisothermal crystallization mechanism was evaluated using the Avrami method modified by Jeziorny, the Ozawa method and the Mo method. The results demonstrate that the nonisothermal crystallization of both pure EVA and EVA/NBR TPV can be adequately described by the Avrami method modified by Jeziorny and the Mo method, while the Ozawa method is not applicable. By elevating the cooling rate, there was a reduction in the crystallization temperature and t1/2 for both pure EVA and EVA/NBR TPV, while the degree of crystallization increased. At the same cooling rate, the crystallization temperature of the EVA/NBR TPV was found to be higher than that of the pure EVA, suggesting that the presence of NBR facilitated the nucleation of the EVA matrix within the EVA/NBR TPV. Moreover, a steric hindrance effect existed in the NBR phase of EVA/NBR TPV at the same cooling rate, resulting in a larger
Keywords
Introduction
Thermoplastic elastomer (TPE) has a unique combination of properties, including elastomeric behavior at room temperature and the ability to be plasticized at the elevated temperature.1–4 Typically, TPE consist of two components: a soft segment with rubber-like elasticity and a hard segment with crosslinking points. 5 The soft and hard segments form a unique two-phase system that is thermodynamically incompatible, resulting in a material with the mechanical elasticity of traditional rubber pre-blend and the reusability of thermoplastics. 6 A specific TPE is thermoplastic vulcanizate (TPV), which is prepared by mixing rubber with thermoplastic resin under dynamic vulcanization. Usually, the process results in the formation of a dual-phase microstructure, wherein the resin phase is acted as the continuous phase while the rubber phase is dispersed throughout in the continuous phase. TPV is a highly versatile material, exhibiting plasticity at high temperature and high elasticity at room temperature. Moreover, TPV has the advantage of being cost-effectiveness, simple processing and equipment requirements, as well as the ability to adjust the resulting product’s properties across a wide range.7–9
Ethylene-vinyl acetate copolymer (EVA) is a versatile thermoplastic resin composed of polar vinyl acetate unit and non-polar ethylene unit. Compared with the most thermoplastic resins, EVA has several distinct advantages, firstly, it exhibits the good biocompatibility, which makes it widely used in medical devices, pharmaceutical packaging and other fields related to human health; secondly, it has the excellent oil resistance, making it a popular choice in the manufacturing of automotive and industrial equipment, as well as in the construction industry; additionally, EVA has the low-temperature flexibility, high light transmission and excellent melt processing properties, which make it a widely used material in the manufacture of footwear, packaging materials, solar cells and other fields. The high polarity of EVA also makes it particularly suitable for certain specific applications, such as the manufacture of solar cell back sheets and adhesives.10–12 Nitrile rubber (NBR) is a polar, unsaturated rubber consisting of butadiene unit and acrylonitrile unit. NBR is renowned for its excellent oil resistance, as well as outstanding properties such as aging resistance, wear resistance and low gas permeability. Therefore, the applications of NBR are diverse, for example, NBR is commonly used for manufacturing fuel system linings and oil seals in the automotive and aerospace industries due to its excellent oil resistance; NBR is frequently used to produce sealing gaskets for oil and natural gas pipelines in the petroleum industry; NBR’s excellent adhesion properties make it an ideal choice for producing printing rollers in the printing industry.13,14 The blending of EVA with NBR results in the TPV that exhibits both the plasticity of EVA and the high elasticity of NBR. Therefore, it is highly meaningful to combine the advantages of EVA and NBR together to prepare the EVA/NBR TPV with excellent property. Until now, there is no report on the nonisothermal crystallization behavior of EVA/NBR TPV, in spite of the widespread use and popularity of both EVA and NBR. The research in nonisothermal crystallization could potentially provide valuable insight into the behavior of EVA/NBR TPV, enabling the development of new applications and improved product performance.
Crystallization kinetic parameters are crucial for understanding the microstructure and properties of crystalline polymer, as well as optimizing manufacturing process and improving product quality.15–17 The crystallization behavior of polymer can be categorized into two major classes: isothermal and nonisothermal crystallization.18,19 Isothermal crystallization occurs when the polymer sample is kept at a constant temperature and its crystallization behavior is controlled by temperature. Conversely, nonisothermal crystallization occurs when the polymer specimen undergoes the different cooling rate. Nonisothermal crystallization methods are widely used in industrial production since nonisothermal conditions are more representative of actual production process. Temperature is the most significant factor influencing the crystallization process of polymer. The crystallization process generally consists of two sections: nucleation and growth. During the nucleation stage, the polymer molecules are arranged around the crystalline core to form microcrystals, which is the beginning of the crystallization process; in the growth phase, the microcrystals continue to expand to form the complete crystals. The kinetic parameters of the nucleation and the growth process determine the crystallization behavior and microstructure formation of the polymer. 20 Differential scanning calorimetry (DSC) is a widely employed method for studying the crystallization behavior. By analyzing the crystallization temperature and enthalpy of crystallization of a polymer sample, DSC experiments can simulate the relative or absolute crystallinity of the crystalline polymer, enabling control of the polymer processing. DSC can also be used to research the crystallization behavior of complex polymer, such as TPE. Usually, TPV is typically composed of rubber and thermoplastic resin and the crystallization behavior is also influenced by the rubber. Therefore, an in-depth understanding of the crystallization process is required to optimize TPV production process. Currently, there is extensive research on the kinetics of EVA. Pang et al. 21 conducted studies on the isothermal kinetics of EVA, demonstrating pure EVA resin exhibits the stable and rapid isothermal crystallization behavior at approximately 74°C. However, most processing methods and applications are conducted under nonisothermal conditions, research on nonisothermal crystallization kinetics holds the more practical value than that of isothermal kinetics. Jin et al. 22 conducted research on the nonisothermal kinetics of EVA, elucidating that pure EVA exhibits a homogeneous nucleation mechanism and a three-dimensional spherulitic growth mechanism. There is a considerable amount of literature discussing EVA/NBR TPV both domestically and internationally, including studies conducted by Wang et al.23,24 on the heat-triggered shape memory of EVA/NBR TPV and Li et al. 25 on the influence of chlorinated polyethylene compatibilization on the mechanical performance and morphology of EVA/NBR TPV. However, the level of attention devoted to the nonisothermal crystallization kinetics of the EVA/NBR TPV remains limited. The investigation of nonisothermal crystallization kinetics in EVA/NBR TPV holds significant potential in facilitating the precise control and optimization of the production process.
In this research, the dynamic vulcanization method was employed to prepare the EVA/NBR TPV. The nonisothermal crystallization kinetic behaviors of pure EVA and EVA/NBR TPV were tested by DSC, and in-depth studies were conducted using Avrami, Ozawa and Mo models to systematically examine the effects of the cooling rate and the crystallization mechanism of NBR relative to EVA. This research is important for understanding the crystallization mechanism of pure EVA and EVA/NBR TPV and provides a reference for the crystallization process.
Experimental
Materials
Ethylene-vinyl acetate copolymer (EVA), grade 7470M, vinyl acetate mass fraction 26%, a product of Taiwan Plastics Industry Co Ltd (Taiwan, China). Nitrile rubber (NBR), grade N41, combined with 29% mass fraction of acrylonitrile, a product of PetroChina Lanzhou Petrochemical Co Ltd (Lanzhou, China).
Bis(1-(tert-butylperoxy)-1-methylethyl)-benzene (BIPB), triallyl isocyanurate (TAIC), N-Phenyl-2-naphthylamine (antioxidant D) and other agents are all commonly used industrial grade commercially available products.
Specimen preparation
The peroxide promotion system is utilized in the NBR crosslinking formulation, which consists of the following constituents: 100 phr (per hundred rubber weight, the same as below) NBR, 3.0 TAIC, 1.3 BIPB and 1.0 Antioxidant D.
In order to prepare the EVA/NBR TPV, firstly, the NBR and compounding agent were mixed in an open double roll mill (X[S] K-160, Qun Yi Rubber Machinery Co Ltd, Shanghai, China) at room temperature to obtain NBR pre-blend. EVA particles were then melted and plasticized on an open double roll mill (SY-6215-AL1, Shi Yan Precision Instrument Co, Ltd, Dongguan, China) at 165°C and NBR pre-blend was put into dynamically vulcanize for 8 min to obtain the EVA/NBR compound. The rotor speed of the mixer was maintained at 43 rpm, while the rubber to the plastic ratio of the EVA/NBR TPV was fixed at 70/30. The cooled EVA/NBR compound was placed in a mold, preheated for 6 min on a plate vulcanizing machine (50 T, Qun Yi Rubber Machinery Co Ltd, Shanghai, China) at 165°C, exhausted for 3 times, hot pressed for 8 min and cold pressed for 8 min at a pressure of 10 MPa to obtain EVA/NBR TPV specimen.
Characterization
The nonisothermal crystallization kinetics of pure EVA and EVA/NBR TPV were researched by DSC. The specimen mass was about 5 mg and the testing was conducted under a nitrogen atmosphere. The instrument was set to ramp up from 20°C to 120°C at a rate of 20 °C/min and held at 120°C for 5 min. The cooling temperature range was 120°C–20°C, followed by cooling to 20°C at rates of 5, 10, 15 and 20 °C/min, respectively.
Results and discussion
The exothermic crystallization curves of pure EVA and EVA/NBR TPV at various cooling rates (5, 10, 15 and 20 °C/min) were illustrated in Figure 1. The results showed that all curves were single peaks, the increase of cooling rate led to the increase of curve width. The appearance of crystalline peaks was independent of the heating rate. Nonisothermal crystallization DSC curves of specimens at different cooling rates. (a) pure EVA; (b) EVA/NBR TPV (weigh ratio = 70/30).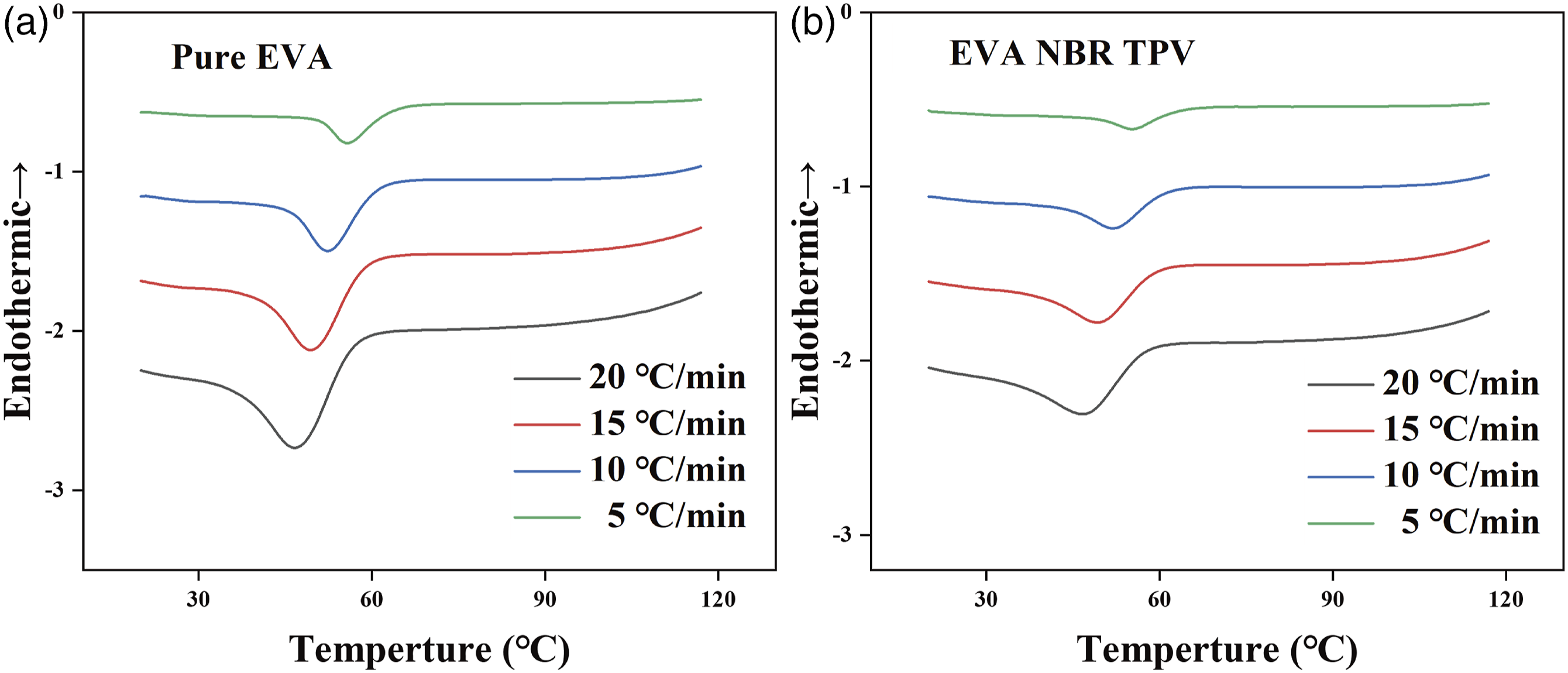
Crystallization kinetic parameters of nonisothermal crystallization of pure EVA and the EVA/NBR TPV.
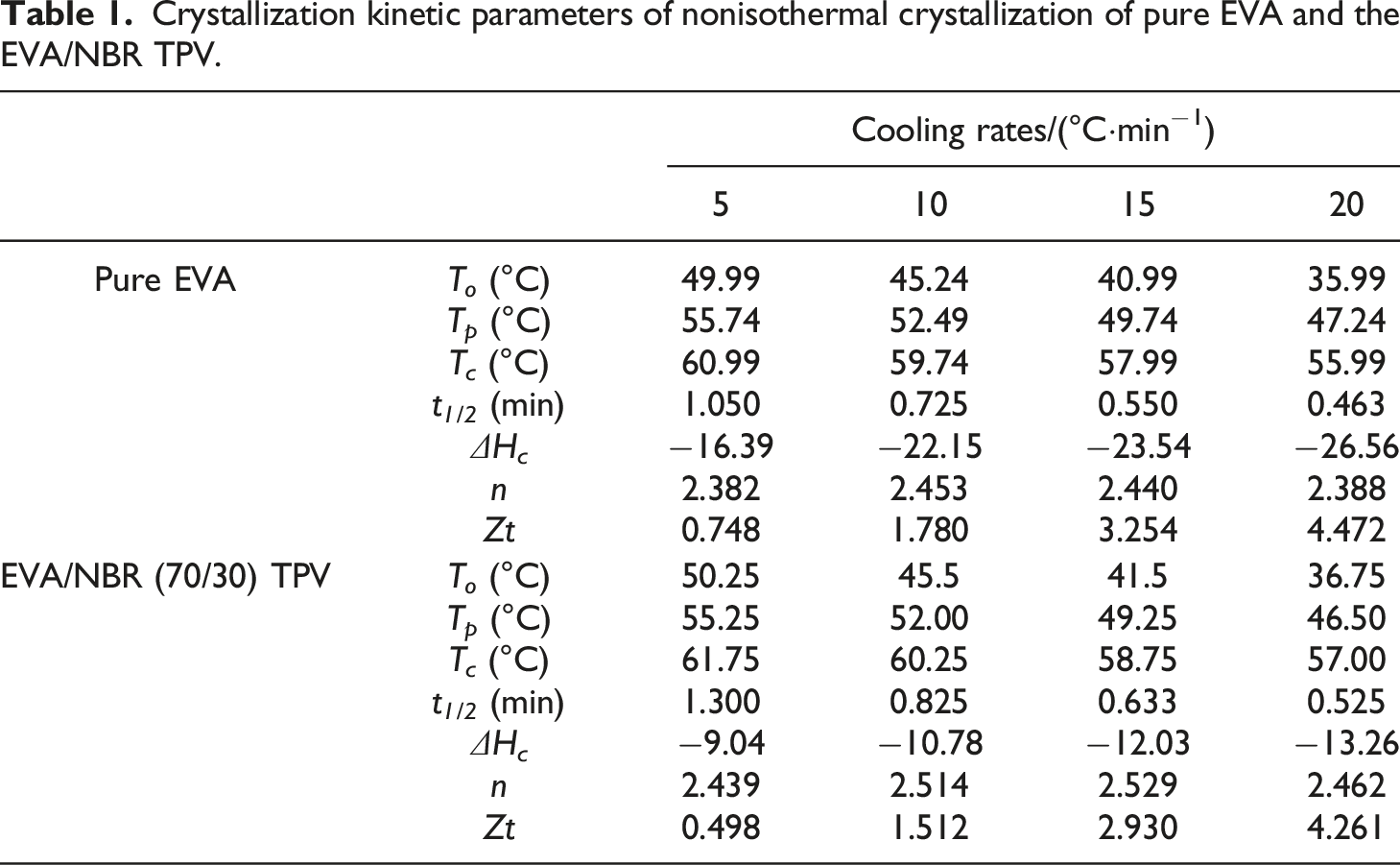
Since the presence of NBR phase in EVA/NBR TPV has an impact on the crystallization behavior of EVA, its relative crystallinity needs to be calculated to further investigate its crystallization kinetic behavior. The Jeziorny method enables the calculation of the relative crystallinity (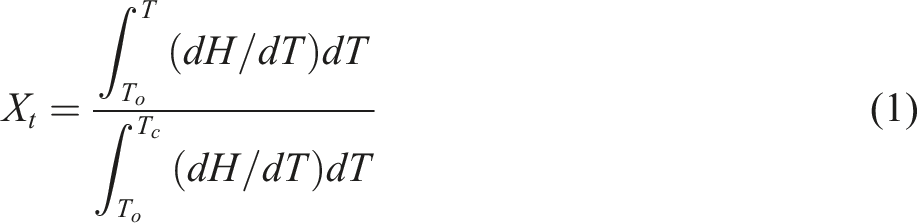
Figure 2 illustrates the relative crystallinity versus temperature curves for pure EVA and EVA/NBR TPV at cooling rates of 5, 10, 15 and 20 °C/min respectively. The curves in Figures 2(a) and (b) exhibited a typical inverse S-shaped pattern, characterized by rapid initial crystallization in the early stage, followed by slower secondary crystallization in the later stage. The crystallinity curve shifted towards low temperature as the cooling rate increased. This was related to crystal growth and nucleation, and if the cooling rate was relatively fast, nucleation and crystal growth could only be completed at relatively low temperature; furthermore, when the crystallization temperature was the same, an increase in the cooling rate resulted in a decrease in the relative crystallinity of the specimen. This phenomenon can be explained by the fact that with increasing cooling rate, the rate of molecular chain exclusion into the crystal lattice cannot match the cooling rate, resulting in restricted crystallization.
28
Development of relative crystallinity versus temperature during nonisothermal crystallization at four cooling rates. (a) pure EVA; (b) EVA/NBR TPV (weigh ratio = 70/30).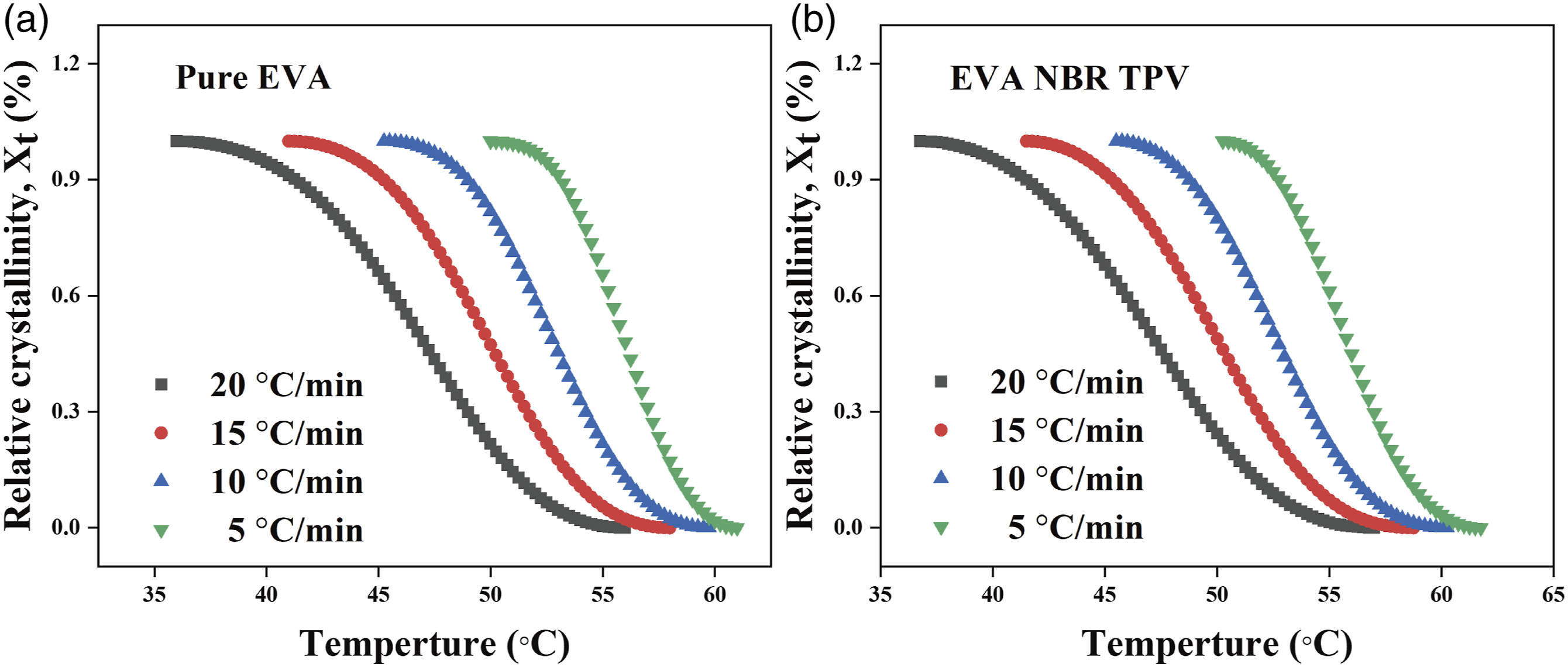
The nonisothermal crystallization process can be described by an equation that expresses the relationship between crystallization temperature (
Figure 3 displays the relative crystallinity versus crystallization time curves for pure EVA and EVA/NBR TPV at various cooling rates. The curves, depicted in Figures 3(a) and (b), exhibit a typical S-shaped trend with increasing time. Moreover, the results indicated that the slope of the curve becomes larger and the crystallization time becomes shorter with the increasing cooling rate. The crystallization time of EVA/NBR TPV was shorter than that of pure EVA at the same cooling rate and relative crystallinity, where the NBR phase promoted the crystallization of the EVA matrix. A larger Development of relative crystallinity versus time during nonisothermal crystallization at four cooling rates. (a) pure EVA; (b) EVA/NBR TPV (weigh ratio = 70/30).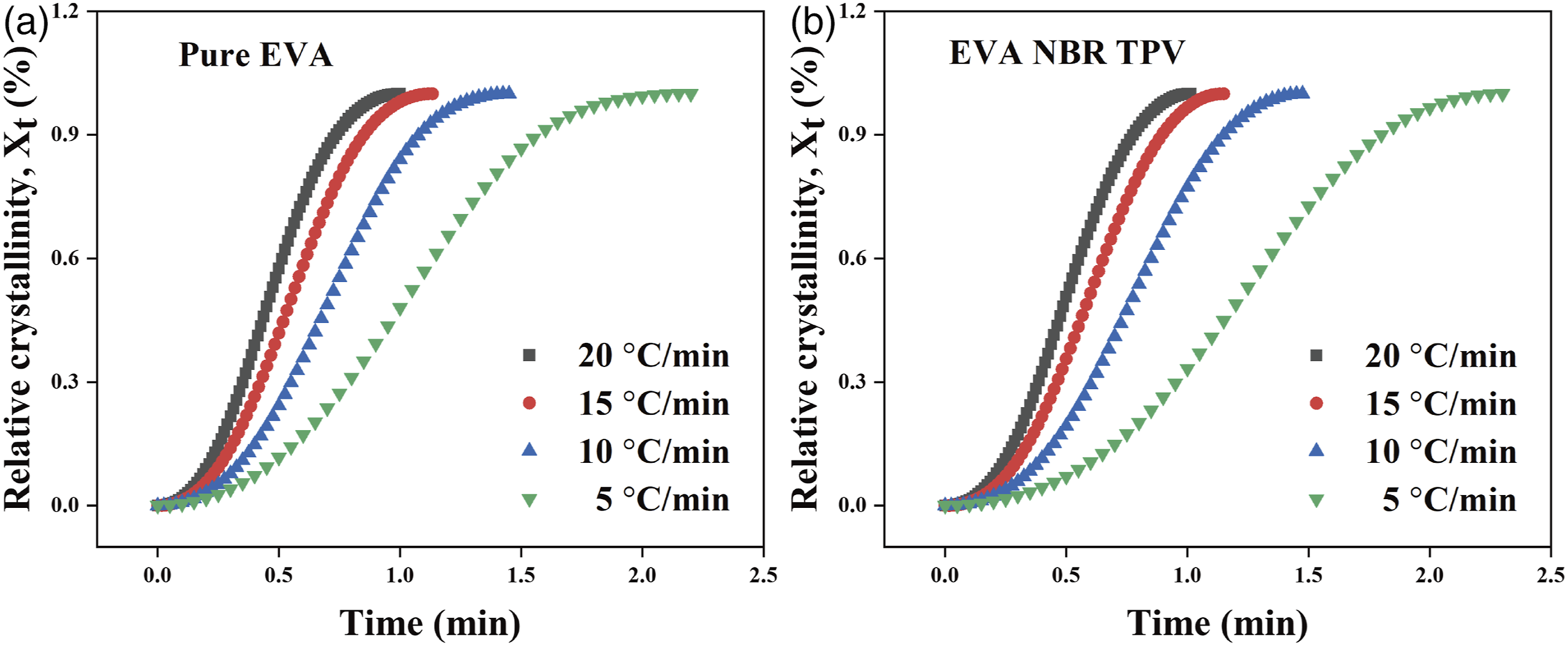
Avrami equation is generally used to analyze isothermal crystallization process, while most of the actual production takes place under nonisothermal conditions, therefore, the Avrami equation is corrected by Jeziorny method to study the nonisothermal crystallization kinetics, and the equation is expressed as follows29,30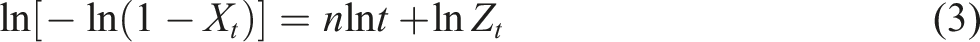

Figure 4 illustrates the ln[-ln(1- The Avrami plots of ln[-ln(1-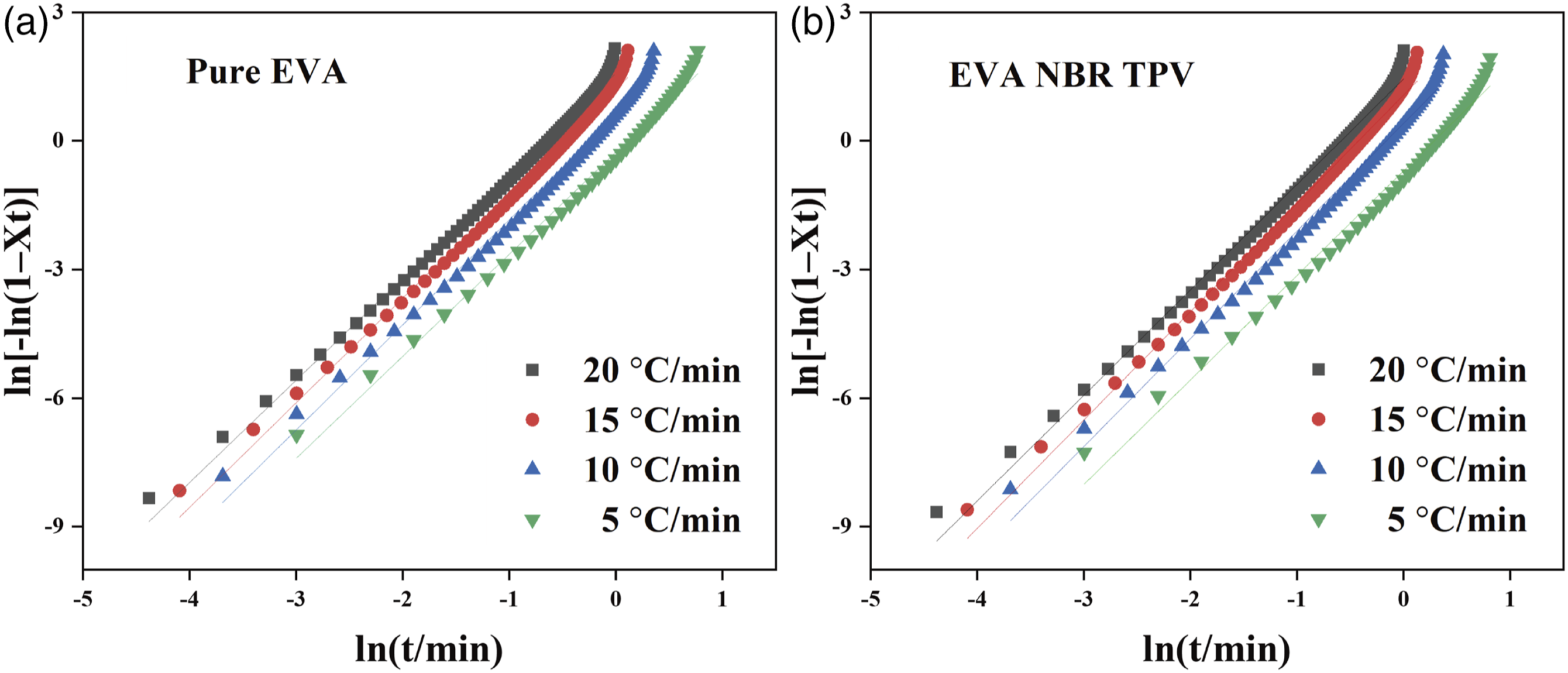
Ozawa et al.33,34 proposed that the nonisothermal crystallization process can be decomposed into a sequence of infinitesimal isothermal crystallization steps, extending the application of the Avrami equation to nonisothermal crystallization conditions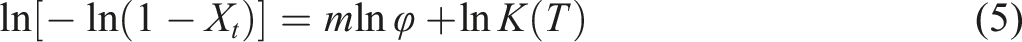
The relative crystallinity is represented by
Figure 5 shows the plot of ln[−ln(1− The Ozawa plots of ln[−ln(1−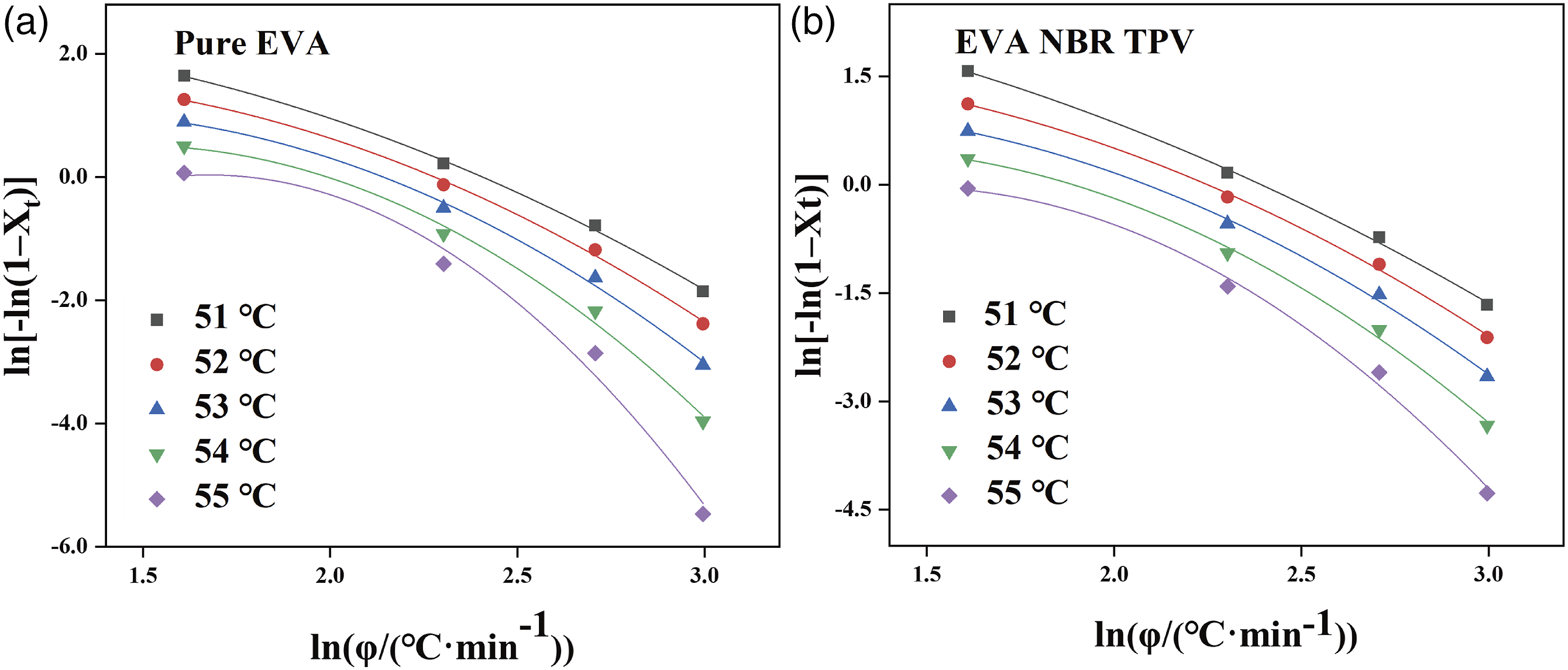
The Mo Zhishen method, which integrates with the Avrami method and the Ozawa method, describes the relationship between cooling rate (
Figure 6 displays the ln The Mo plots of the ln The Mo kinetic parameters of nonisothermal crystallized pure EVA and EVA/NBR TPV determined via the DSC method.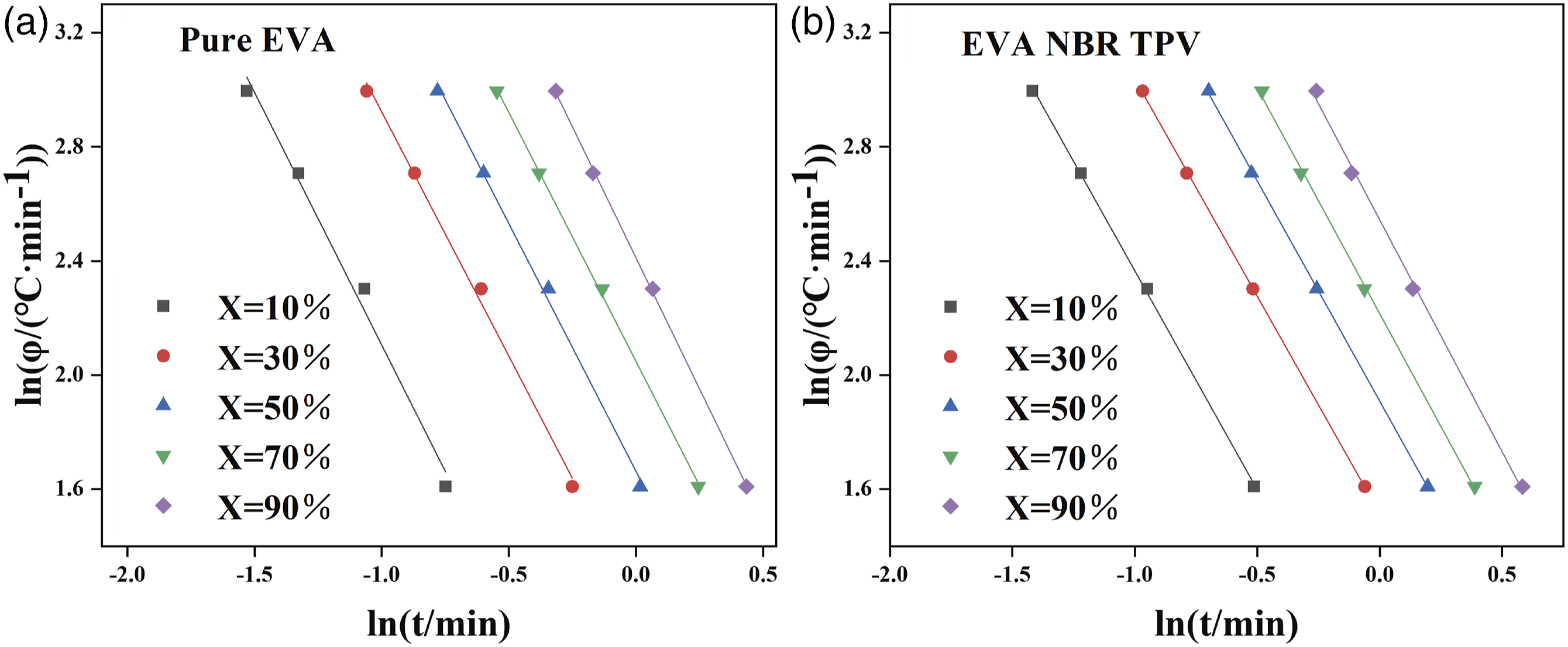
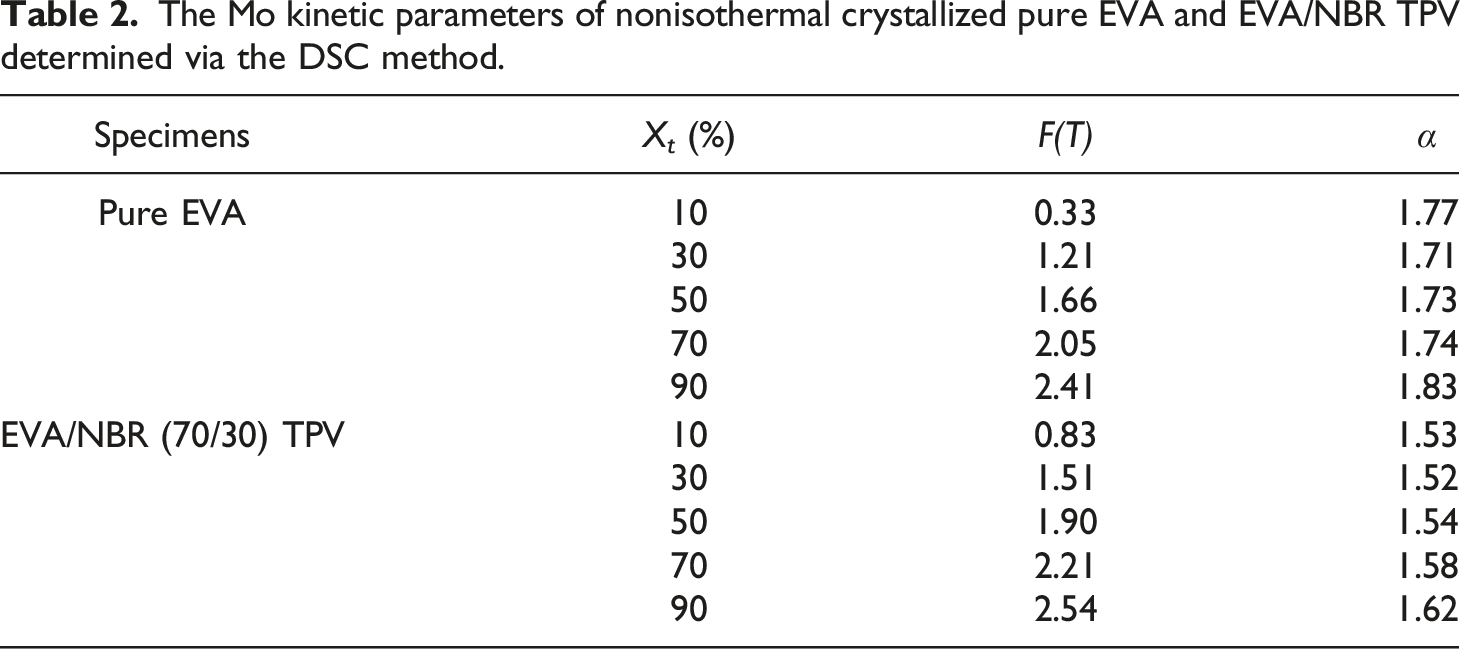
In this research, both the Jeziorny method and the Mo method were successful in describing the nonisothermal crystallization behavior of the EVA/NBR TPV, while the Ozawa method failed to describe the nonisothermal crystallization behavior. This result is similar to the findings of our previous research,28,37 the research further proved the identical perspective.
Conclusion
In this research, the nonisothermal crystallization kinetics of pure EVA and EVA/NBR TPV was employed the Avrami method, the Ozawa method and Mo method. The results revealed that the rate of crystallization in both pure EVA and EVA/NBR TPV was affected by the cooling rate, and the faster cooling rate would lead to the faster crystallization rate. The higher crystallization temperature of EVA/NBR TPV compared with pure EVA proves that the NBR phase plays the role of nucleating agent. Both Avrami analysis modified by Jeziorny and the Mo method were capable of accurately describing the crystallization kinetics of pure EVA and EVA/NBR TPV. Specifically, the Avrami method was employed to determine the key parameters of
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shandong Provincial Natural Science Foundation, China (ZR2021ME028).
