Abstract
Nonisothermal crystallization kinetics of ethylene–acrylic acid copolymer (EAA) and thermoplastic vulcanizate (TPV) based on EAA/chloroprene rubber (CR) were extensively studied using differential scanning calorimetry. Several methods, including the Avrami, Ozawa, and Mo equations, were carried out to analyze the process of nonisothermal crystallization kinetics of EAA and EAA/CR TPV. The results showed that the Avrami analysis modified by Jeziorny and a method developed by Mo could describe the nonisothermal crystallizations of pure EAA and the EAA/CR TPV very well. However, the Ozawa analysis did not give an adequate description. The crystallization processes of pure EAA and the EAA/CR TPV were accelerated by increasing the cooling rates. Moreover, the initial crystallization temperature and the crystallization termination temperature of EAA/CR TPV were higher than those of pure EAA at the same cooling rate, thus showing the nucleating function of CR in the beginning. While the crystallization half time of EAA/CR TPV was apparently longer than that of pure EAA, meaning that the more CR could cause the steric effect and retard the crystallization process of the TPV during the late stages of crystallizing. Although the CR phase of EAA/CR TPV could provide more nucleation sites, the presence of more CR phase must impose a much more significant confinement effect on the crystal growth of EAA. It was believed that this confinement effect overweighed the nucleation effect, thereby slowing down the overall crystallization rate.
Keywords
Introduction
Thermoplastic vulcanizates (TPVs), in which cross-linked rubber particles dispersed in continuous thermoplastics matrix, are a special category of high-performance thermoplastic elastomers prepared via dynamic vulcanization.1–4 Because TPVs have good processability of thermoplastics and excellent resilience of traditional vulcanized rubbers, in recent years, TPVs are currently drawing more and more attention and have been widely used in building, automotive, and wearable devices.5–10 Meanwhile, dynamic vulcanization is a method to produce new TPVs, which was firstly discovered by Gessler 11 in 1962 and then further studied by Fischer, 12 Coran 13 and Abdou-Sabet. 14
Ethylene–acrylic acid copolymer (EAA) is a technologically important semicrystalline thermoplastic; shows excellent adhesion, easy processing, good mechanical property and thermal stability; and has been considered as an ideal plastic for preparing TPVs with outstanding physical and chemical performance.15–18 Chloroprene rubber (CR), a kind of polar rubber, with its exceptional physical and chemical properties, is categorized to be suitable for applications of ozone resistance, flame retardancy, and good adhesion of sealing and gaskets.19–21 However, it is barely reported for the EAA/CR TPV until now.
The crystallization kinetics of both polymer and TPVs are particularly important for the analysis and design of processing operations, which would be able to crystallize not only when cooled from the melt but also when heated from the amorphous state.22–27 The former is so-called “melt crystallization” and the latter one is “cold crystallization.”
In this article, the EAA/CR TPV was prepared by dynamic vulcanization where the CR phase had played a role in impacting the nonisothermal crystallization of the EAA/CR TPV significantly. The nonisothermal crystallization kinetics of pure EAA and the EAA/CR TPV were further investigated systematically; meanwhile, it was expected that the research reported herein was of great help for a better understanding of the structure and properties relationship of semicrystalline polymer and TPVs.
Experimental
Materials
EAA, grade N300 (melting temperature (
Preparation of dynamically vulcanized EAA/CR blends
Commercially available EAA and CR, as mentioned above, were manufactured for the TPVs. The concentrations for cross-linking CR system are expressed in parts per hundred CR by weight (phr). The metallic oxides-containing accelerating system recipe consisted of the following ingredients: 100 phr CR, 5.0 phr ZnO, 4.0 phr MgO, 0.5 phr SA, and 2.0 phr antioxidant D.
The dynamically vulcanized EAA/CR TPVs were produced via a two-step mixing process. Firstly, the preblend CR including the cross-linking ingredients was compounded in a two-roll mill (X(S) K-160, Shanghai Qun Yi Rubber Machinery Co., Ltd, China) at room temperature. After 3 min of mixing time, the preblend was removed from the mixer. Secondly, the requisite quantities of EAA were charged into another two-roll mill (SY-6215-AL, Shi Yan precision instruments Co., Ltd, China) and allowed to melt. Then the preblend was also charged into the two-roll mill and continued at 165°C for another 8 min to allow dynamic vulcanization. The CR phase was gradually broken down into dispersed particles by initially large CR entities under the local stress that evolved during melt mixing, the phase morphology was stable then. Finally, the compound was removed from the mixer and then passed through a cold two-roll mill in the molten state to obtain sheet samples about 2 mm thick. All test specimens were used for testing after 24 h.
Characterization
Differential scanning calorimetry
Nonisothermal crystallization kinetics was carried out using a DSC Q20 (TA Instruments, New Castle, Delaware, USA) differential scanning calorimeter calibrated the temperature with indium. All differential scanning calorimetry (DSC) analyses were performed under nitrogen atmosphere; sample weights were between 5 mg and 8 mg.
Nonisothermal crystallization process
The nonisothermal crystallization was performed as follows: The samples were firstly heated from 40°C to 140°C (above
Results and discussion
Nonisothermal crystallization behavior of pure EAA and EAA/CR TPV
The nonisothermal crystallization exotherms of the pure EAA and EAA/CR TPV at five cooling rates of 5, 10, 15, 20, and 25°C min−1 are shown in Figure 1. As shown in Figure 1(a) and (b), the crystallization process of all samples demonstrated a single peak. Some kinetic parameters could be derived from the DSC curves, including the initial crystallization temperature (
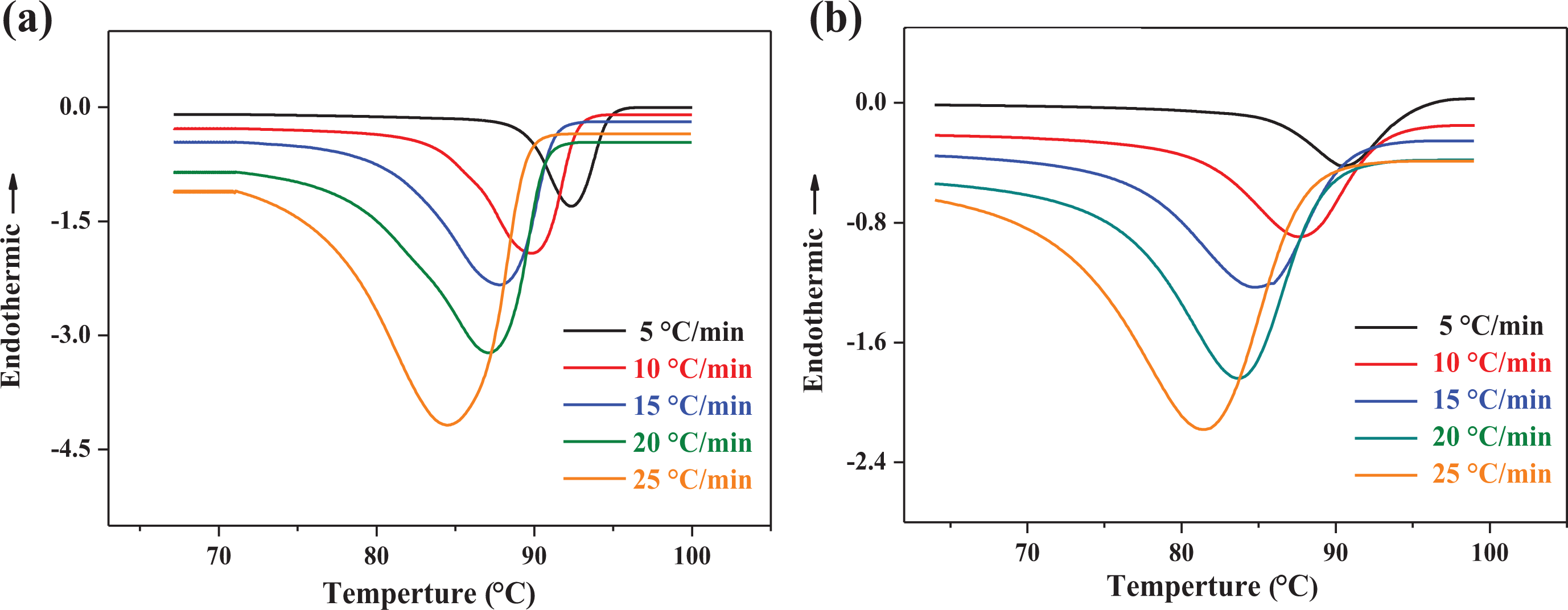
Nonisothermal crystallization DSC curves of samples at different cooling rates. (a) pure EAA, (b) EAA/CR TPV (EAA/CR = 60/40).
Kinetic parameters of nonisothermal crystallization of pure EAA and the EAA/CR TPV.
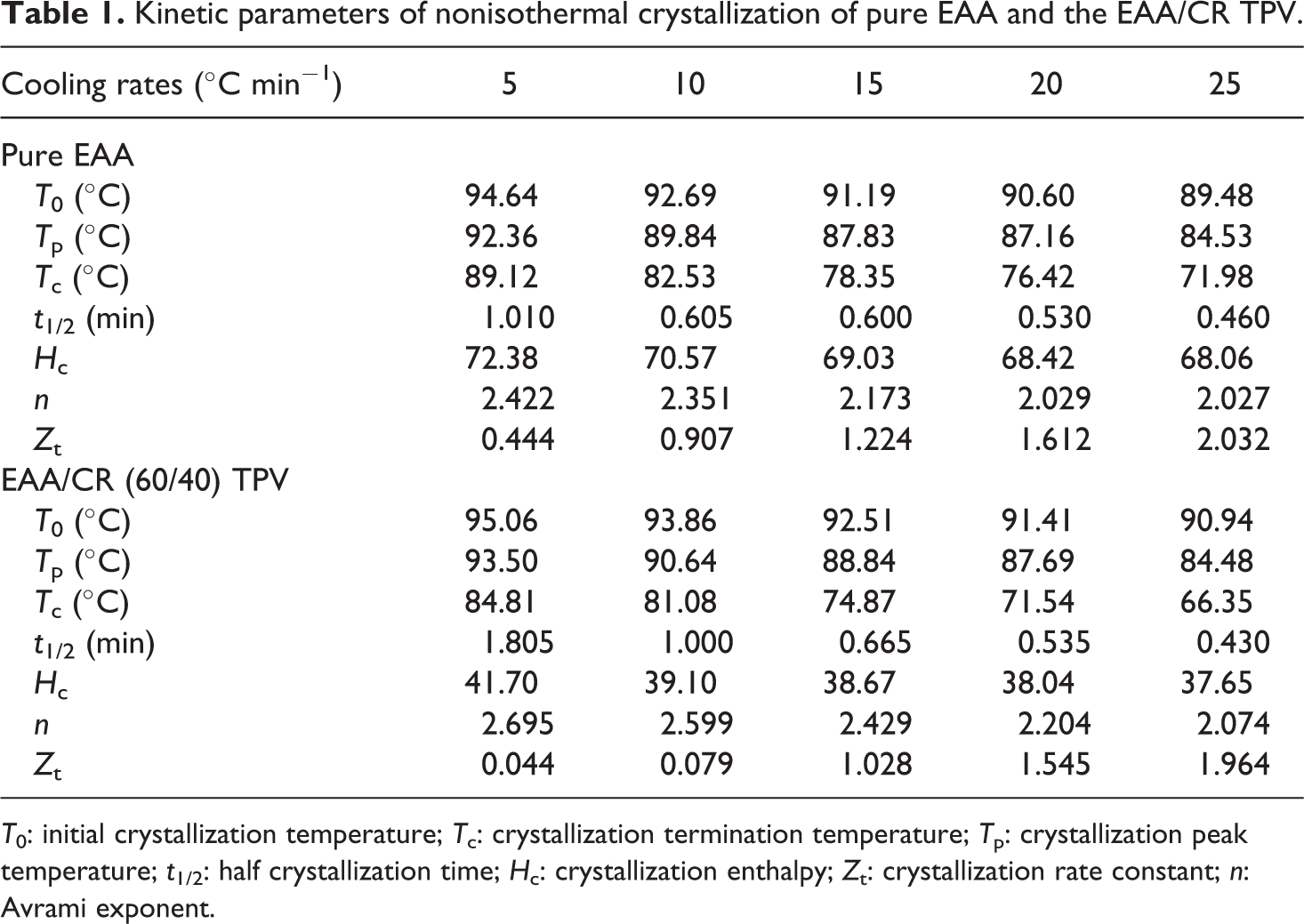
Effect of cooling rate on crystallization temperature
During the nonisothermal crystallization process, the relative crystallinity as a function of temperature could be expressed as:
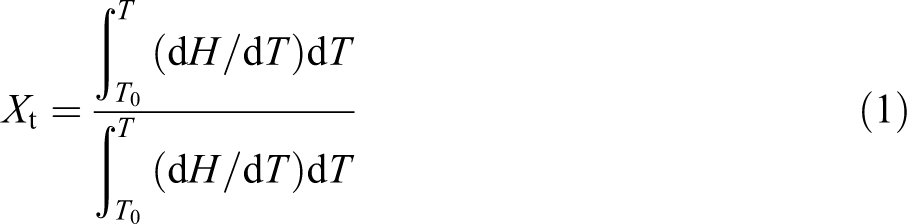
where
Figure 2 shows the development of relative crystallinity as a function of temperature for the pure EAA and EAA/CR TPV at various cooling rates. As can be seen from Figure 2(a) and (b), the development of relative crystallinity displayed the familiar sigmoidal shape with the increasing of temperature, manifesting fast primary crystallization during the early stages and slow secondary crystallization in the later stage. Moreover, the crystallization curve shifted to lower temperature with the increase in cooling rates, signifying rapid crystallization rates. However, because of shorter crystallization time at a faster cooling rate, the values of
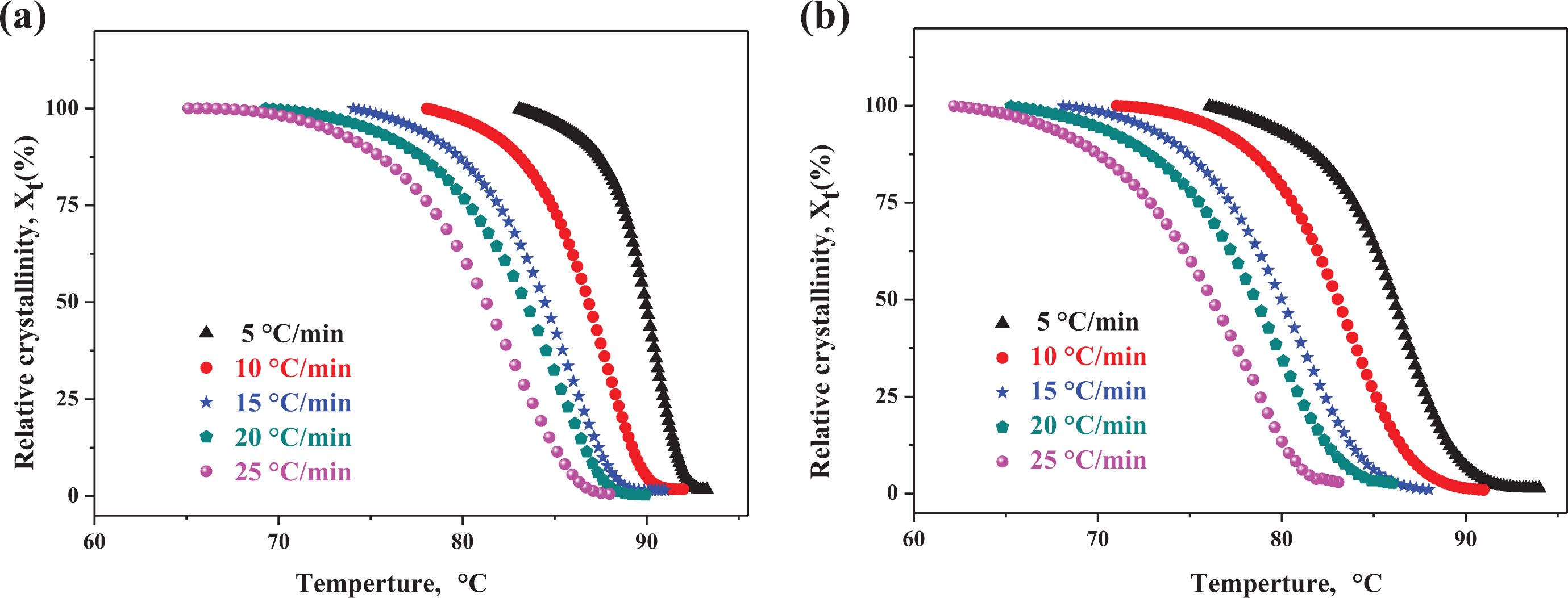
Development of relative crystallinity versus temperature during nonisothermal crystallization: (a) pure EAA and (b) EAA/CR TPV (EAA/CR = 60/40).
Effect of cooling rate on crystallization time
The relationship between crystallization temperature (

where
Figure 3 shows the plots of relative degree of crystallinity versus crystallization time at various cooling rates for the pure EAA and EAA/CR TPV. It could be seen from Figure 3(a) and (b) that the development of relative crystallinity also displayed the familiar sigmoidal shape with increasing time. Moreover, the plots shifted to more time range with increasing the cooling rates. As shown in Figure 3(b), it could be easily found that the plots of EAA/CR TPV shifted to more time range at the same cooling rate, compared the plots of pure EAA in Figure 3(a). On the one hand, the CR particles could act as a nucleating agent for the EAA matrix under nonisothermal conditions, due to
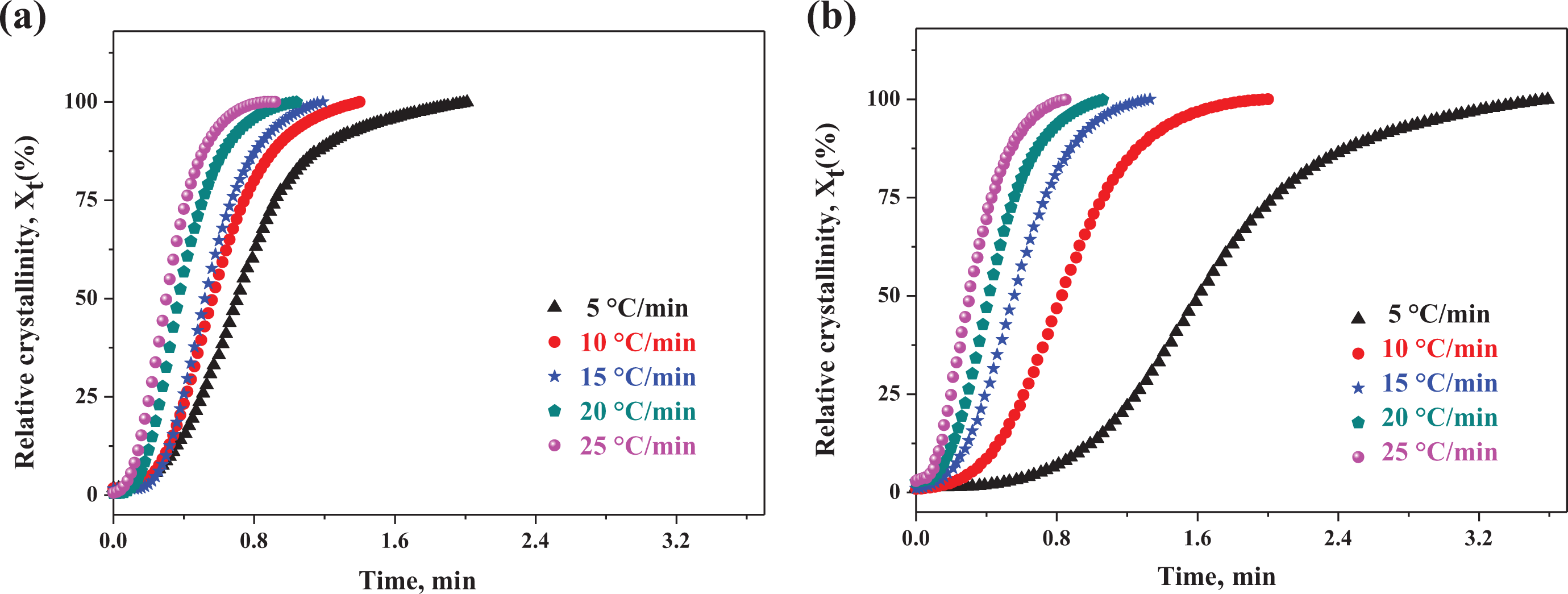
Development of relative crystallinity versus time during nonisothermal crystallization. (a) pure EAA and (b) EAA/CR TPV (EAA/CR = 60/40).
Avrami method
The Avrami equation is usually used to describe the kinetics of isothermal crystallization of polymers; however, it has also been employed to investigate nonisothermal crystallization kinetics,28–31 the Avrami equation was shown as follow:
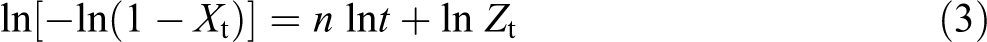
where the

where
The plots of ln[−ln(1 −
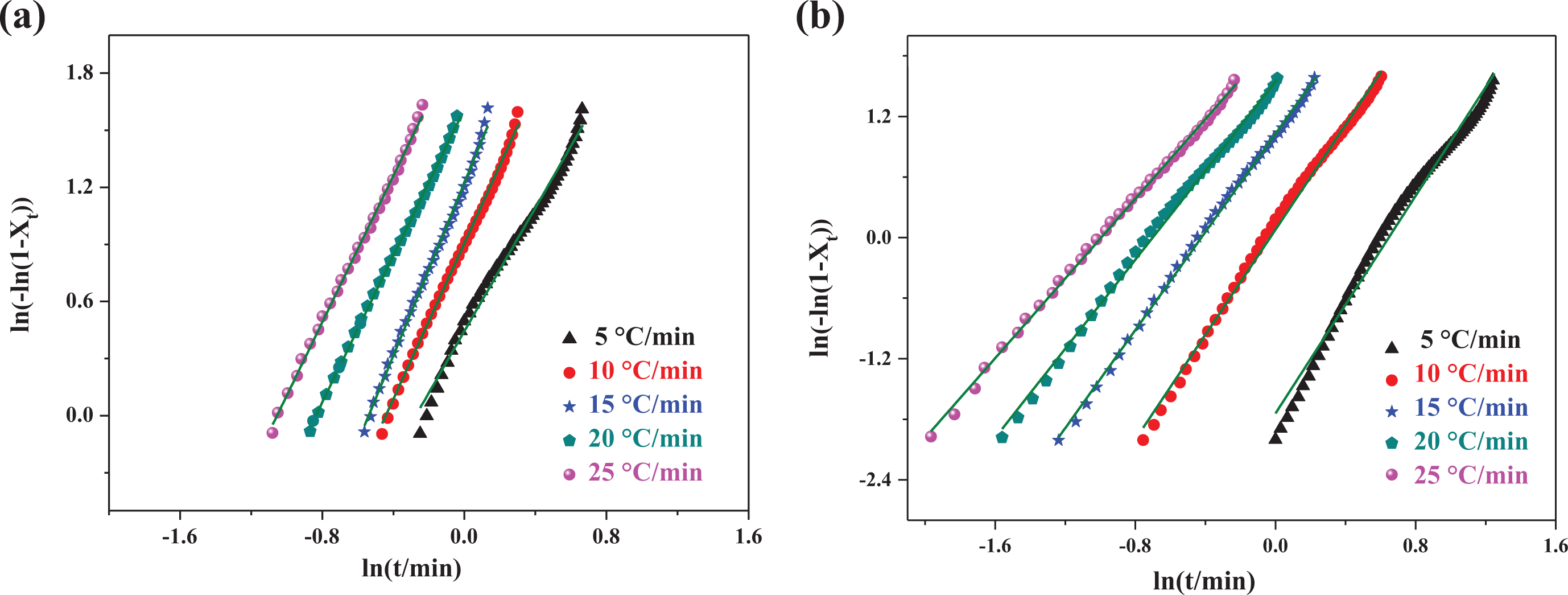
The Avrami plots of ln[−ln(1 −
The Ozawa method
Ozawa 34 extended the Avrami equation to the nonisothermal condition. Assuming that the nonisothermal crystallization process may be composed of infinitely small isothermal crystallization steps, the following equation was derived:
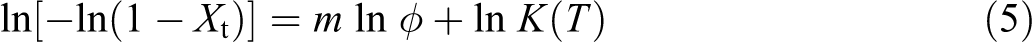
where
The nonlinear dependence of ln[−ln(1 −
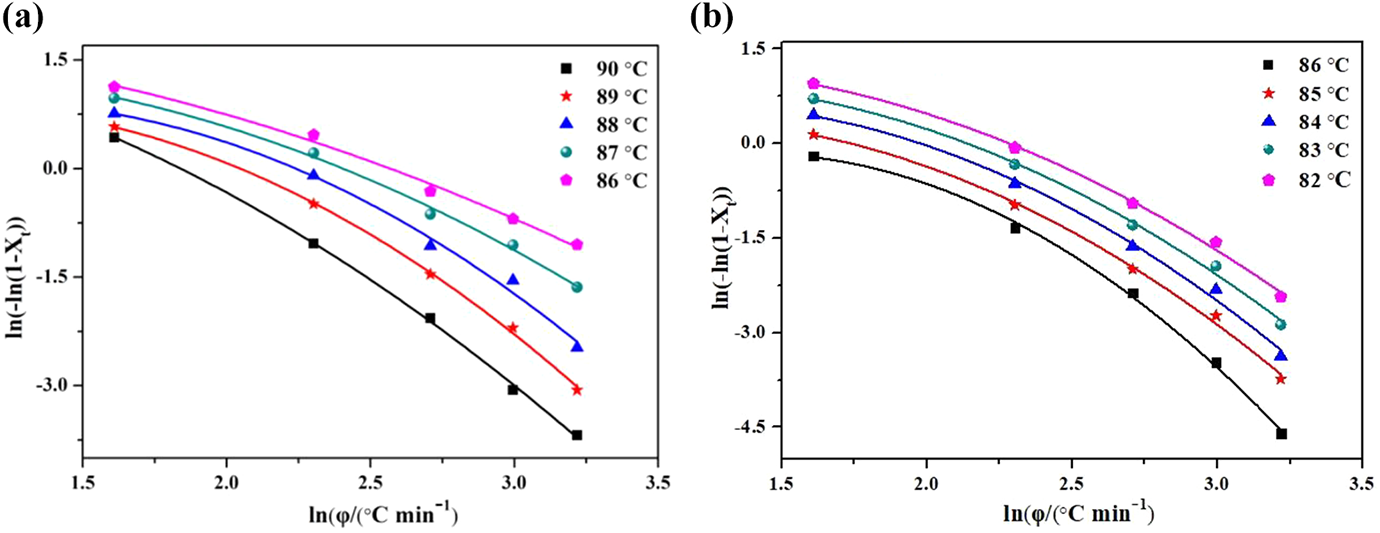
The Ozawa plots of ln[−ln(1 −
The Mo method
A method developed by Mo was also employed to describe nonisothermal crystallization which combines the Avrami equation with the Ozawa equation. Its final form was given as follows 23 :

where the parameter
The plots of the ln
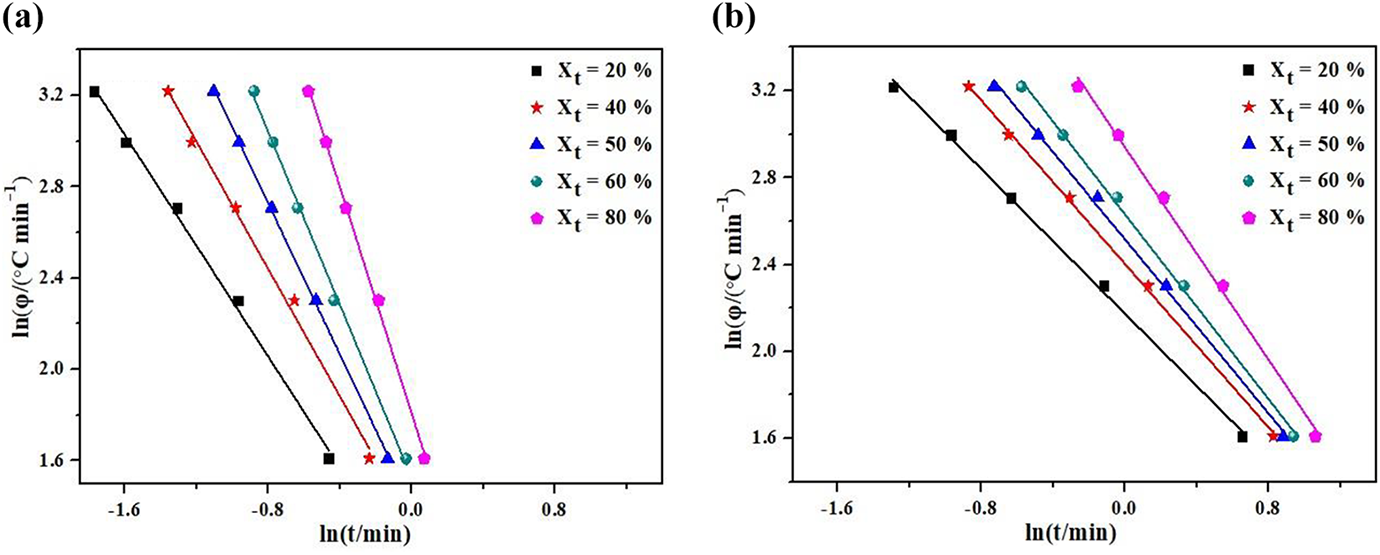
The Mo plots of the ln
The Mo kinetic parameters of nonisothermal crystallized EAA/CR TPV determined using the DSC method.
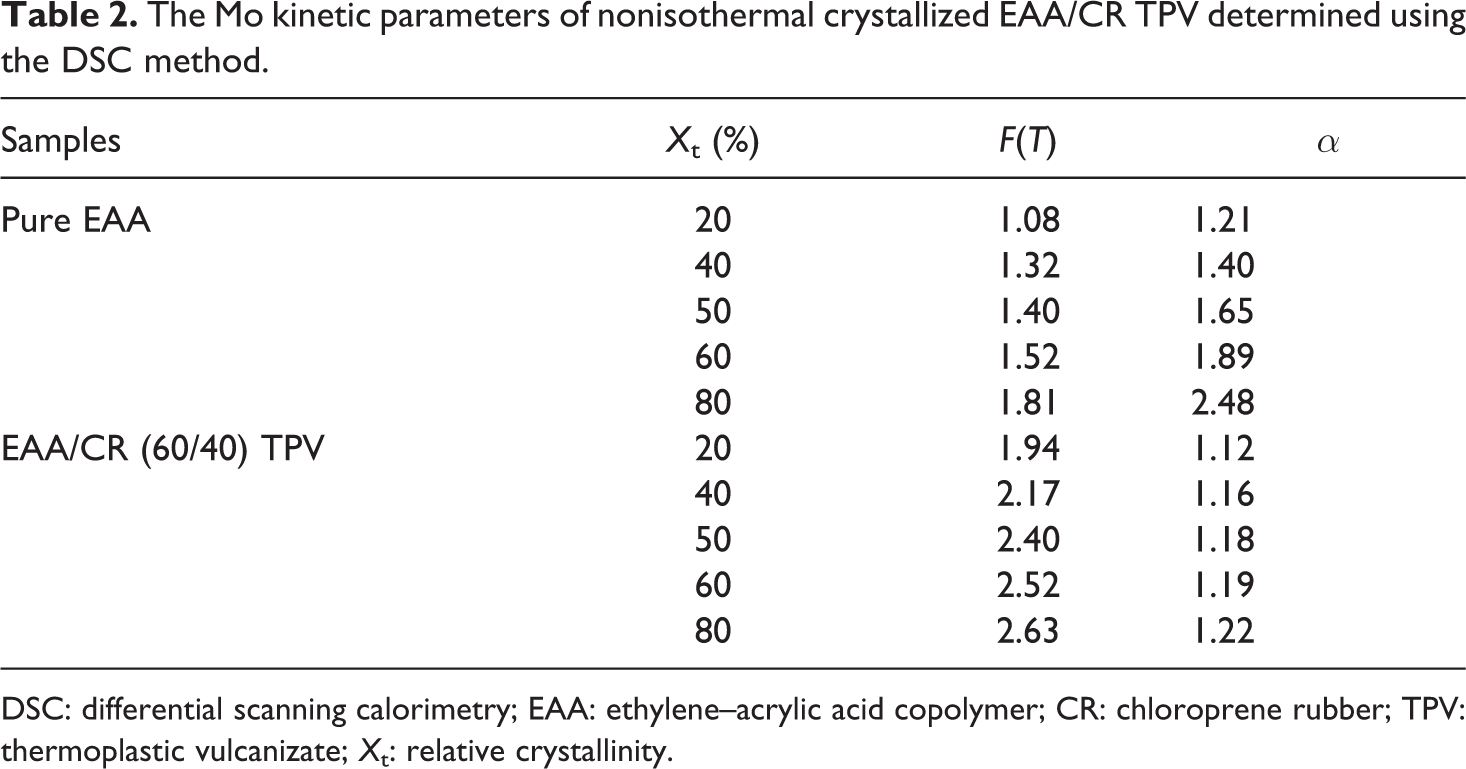
DSC: differential scanning calorimetry; EAA: ethylene–acrylic acid copolymer; CR: chloroprene rubber; TPV: thermoplastic vulcanizate;
Conclusion
In this work, according to three various kinetic models, namely, the Avrami, the Ozawa and the Mo method, nonisothermal crystallization kinetics of pure EAA and the EAA/CR TPV were investigated in detail. The Avrami equation modified by Jeziorny’s method and the Mo method were successful for describing the nonisothermal crystallization process of the pure EAA and the EAA/CR TPV. Meanwhile, the Ozawa equation failed to provide an adequate description of nonisothermal crystallization. Both cooling rate and CR dosage were the two main factors that influence the nonisothermal cold crystallization behavior of EAA in the EAA/CR TPV. On the one hand, with the increasing of cooling rates, the crystallization exotherm shifts to larger temperature range, and the crystallization process was enhanced for both pure EAA and the EAA/CR TPV. On the other hand, at a given cooling rate, the addition of CR enhanced the nonisothermal cold crystallization of EAA; however, the more CR could cause the steric effect and retard the crystallization process of the TPV; furthermore, in the Avrami method, the parameters
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Shandong Provincial Natural Science Foundation, China (ZR2017MEM021) and Upgraded Project of Shandong Province for Guidance Ability of Graduate Tutors (SDYY17044).
