Abstract
Achieving considerable shape-memory effects is a challenge for typical sea-island structured polymer blends. In this article, we successfully fabricated a novel heat-triggered shape-memory polymer (HSMP) based on ethylene–vinyl acetate copolymer/nitrile–butadiene rubber (EVA/NBR) thermoplastic vulcanizates (TPVs) via dynamic vulcanization. The influence of deformation temperature (
Keywords
Introduction
Shape-memory polymers (SMPs) are a promising class of smart materials for which externally programmed shape(s) can be temporarily fixed and later recovered on demand. Because of the unique ability to sense and respond to external stimuli, such as heat, light, electricity, magnetic field, and chemicals, 1 –6 SMPs obtain great opportunities to realize their vast potential in a number of scientific and technological fields, such as the intelligent packaging, high-performance textile, self-repairing plastic components, aerospace, and smart medical devices. 7 –9
Heat-triggered SMPs (HSMPs) are the most concerned and attractive SMPs because of their feasible shape-memory and the broad applications. The shape-memory process of the HSMPs can be perfectly realized by regulating the temperature. To get the charming shape-memoryre behavior, HSMPs are usually designed to contain at least two essential functional components: (1) the switching phase with the responsibility to maintain the temporary shape and (2) the reversible phase that is easily deformed to provide driving force for recovery. Conventionally, the reversible phase can be chemical or physical cross-linking structures, whereas the switching phase can be either a crystal or an amorphous structure. When the HSMPs are heated to above the switch temperature (
As far as we know, the preparation of HSMPs can be classified into two main categories: chemical copolymer synthesizing and polymer blending. The copolymer usually includes a hard domain providing permanent memory shape and a soft domain acting as a switching domain, for example, polyurethane. 14 –16 Although the architecture of the copolymer can be designed to offer the required shape-memory effect (SME), the relatively expensive synthesis process limited their broad utilization more or less.
Compared with the complicated chemical synthesis, polymer blending offers a more simple and effective method to fabricate HSMPs because of the ease of changing components to tune the microstructure and properties.
17,18
Polymer blends based on high-density polyethylene/poly(ethylene terephthalate),
19
poly(lactic acid)/poly(vinyl acetate),
20
sodium bisulfite/poly(ε-caprolactone) (PCL),
21
and PCL/poly(
In this article, we presented a novel HSMP derived from the ethylene–vinyl acetate copolymer (EVA)/nitrile–butadiene rubber (NBR) TPVs, which has never been reported to our knowledge. The influence of the EVA/NBR weight ratio, the deformation condition, and the recovery condition on the shape-memory behavior of EVA/NBR TPV was investigated. The dynamic vulcanization has been proved to be an effective method for preparing an SMP. Considering that the EVA is biocompatible and has been widely used in many biomedical engineering applications, 27 such as in drug delivery devices 28 and shape-memory applications, 29,30 the successful preparation of EVA/NBR HSMP provides a promising future for EVA-based HSMP in intelligent medical devices.
Experimental
Materials
EVA copolymer, grade 7470 M (
Preparation of EVA/NNR TPVs
Commercially available NBR and EVA, as above, were used for the TPVs. The concentrations for cross-linking the NBR system are expressed in parts per hundred NBR rubber by weight (phr). The composition for NBR rubber system using sulfur as the cross-linking agent is shown by weight (phr), as follows: 100 phr NBR, 1.0 phr sulfur, 1.5 phr CZ, 1.2 phr TS, 5.0 phr ZnO, 1.5 phr stearic acid, and 1.0 phr antioxidant RD.
The EVA/NBR TPVs were produced via a two-step mixing process. Firstly, the preblends containing NBR and the additives were compounded in a two-roll mill at room temperature; then, the preblends were removed from the mixer after 3 min. Secondly, the TPV compounds were prepared by melt-mixing the NBR preblends with EVA resin using a Brabender PLE 331 plasticorder (Brabender GmbH, Germany). The mixer temperature was kept at 165°C with a constant rotor (camtype) speed of 80 r min−1. The EVA/NBR weight ratio was varied from 50/50 to 90/10. The requisite quantity of EVA resin was charged into the mixer and allowed to melt. The NBR-based preblend was added after 3 min. The mixing was continued for another 8 min to allow the dynamic vulcanization. Finally, the compound was removed from the mixer and passed through a cold two-roll mill in the molten state to obtain a sheet. Then, the sheet was compression-molded under a pressure of 15 MPa at 165°C for 8 min to a thickness of about 2 mm, followed by cold compression for 8 min at room temperature. Dumbbell-like test specimens were die-cut from the compression-molded sheet and used for testing after 24 h. For brevity, TPVs were coded according to the EVA/NBR weight ratios, and the details of the TPV compositions are listed in Table 1.
Various TPVs composition.
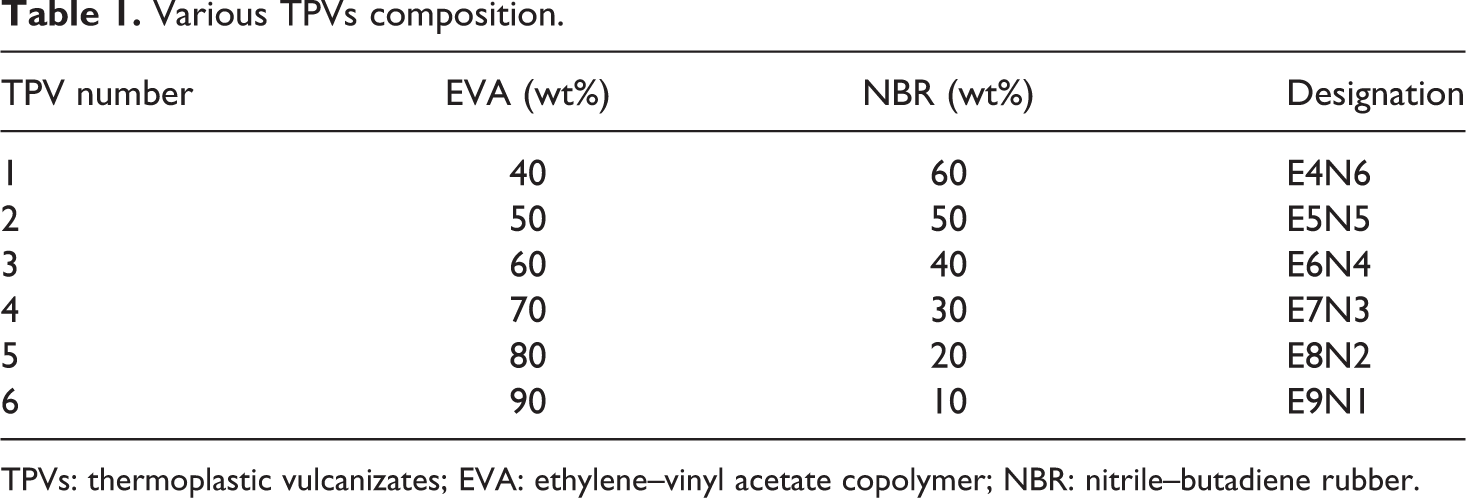
TPVs: thermoplastic vulcanizates; EVA: ethylene–vinyl acetate copolymer; NBR: nitrile–butadiene rubber.
Characterizations
Dynamic mechanical analysis
Dynamic mechanical analyses (DMAs) of the EVA/NBR TPVs were conducted with a dynamic mechanical thermal analysis system (DMTS EPLEXOR, 500 N, NETZSCH GABO Instruments GmbH, Germany). Temperature sweep of the samples was carried out in tension mode over a temperature range of −100°C to +90°C at a heating rate of 3°C min−1. The samples were scanned at a frequency of 1 Hz and a strain level of 1%, and the storage modulus (
Microscopy analysis
Morphological study was carried out using field-emission scanning electron microscopy (FE-SEM; JSM-6700F, Japan Electron Optics Laboratory Co. Ltd, Japan). For the etched specimens, the EVA phase was extracted by immersing the TPVs into toluene for 120 min at 60°C. Then, the specimens were dried at room temperature. The etched surfaces and the fracture surfaces of the specimens were sputtered with thin layers of gold and imaged using FE-SEM.
SME measurement
SME measurement was similar to that in Zhu and colleagues’ report
31
: (a) Two distance lines of 2 cm were marked at the center of a dumbbell specimen (
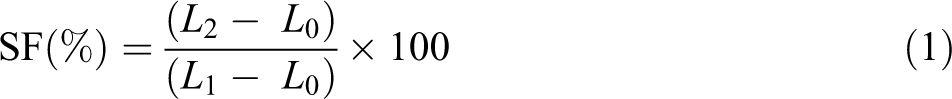
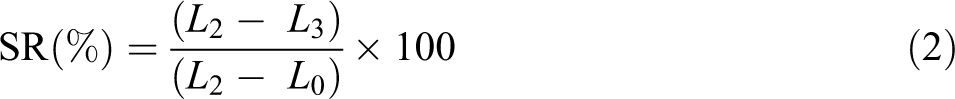
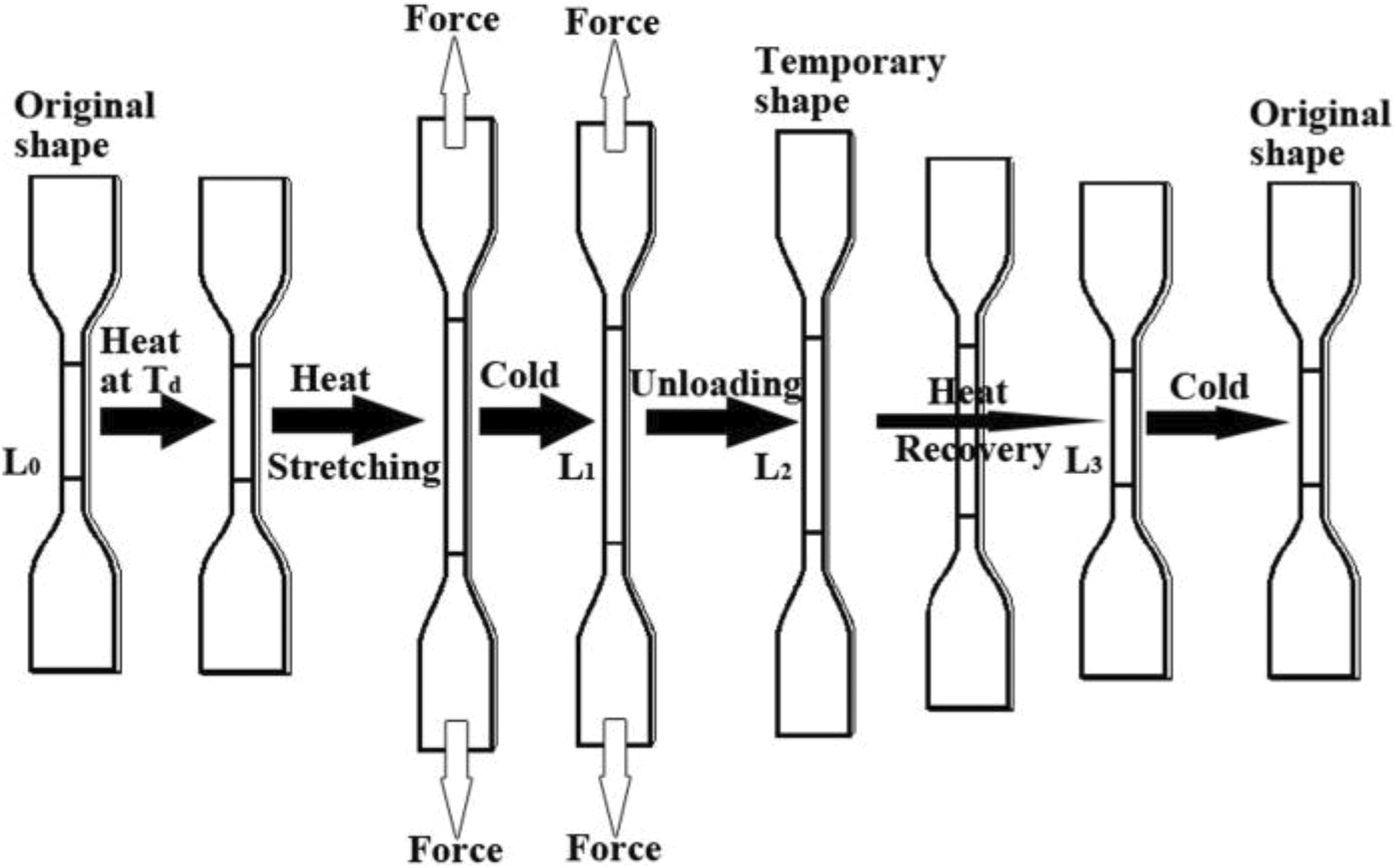
Schematic description of SME measurement.
Results and discussion
Dynamic mechanical analysis
DMA was often used to study the miscibility in polymer blends. The results of DMA afforded the information about the behavior of the blends and microstructure. Tan
The maximum tan
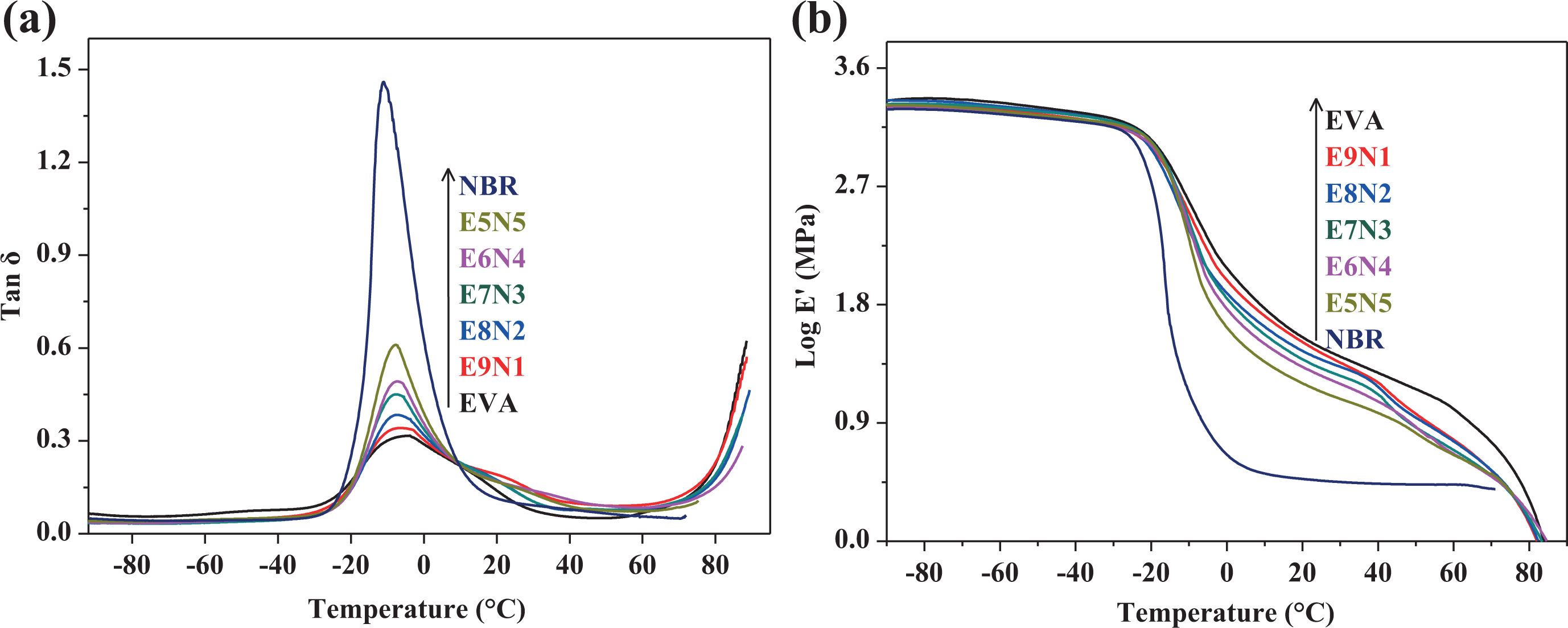
DMA results of (a) tan
The temperature dependence of the storage modulus of EVA resin, NBR vulcanizate, and the series EVA/NBR TPVs is shown in Figure 2(b). From this figure, it is clear that there was a sharp decrease in the area of −30–10°C with the increasing temperature, which corresponded to the glass transition area of NBR vulcanizate. One distinguished phenomenon was clear that the rubbery plateau region was gradually increased with the increasing NBR content in the TPVs. We could also find that the storage modulus value of the TPVs containing higher EVA content was always higher than that of the TPVs containing lower EVA content in the temperature range of approximately 45–50°C. This was due to the higher modulus of pristine EVA than that of pristine NBR, indicating that the higher EVA content helped to enhance the SF ability of the TPV at a temporary shape.
The DMA results of EVA resin, NBR vulcanizate, and series EVA/NBR TPVs at 0°C are summarized in Table 2. EVA showed the highest
DMA results of EVA resin, NBR vulcanizate, and series EVA/NBR TPVs at 0°C.
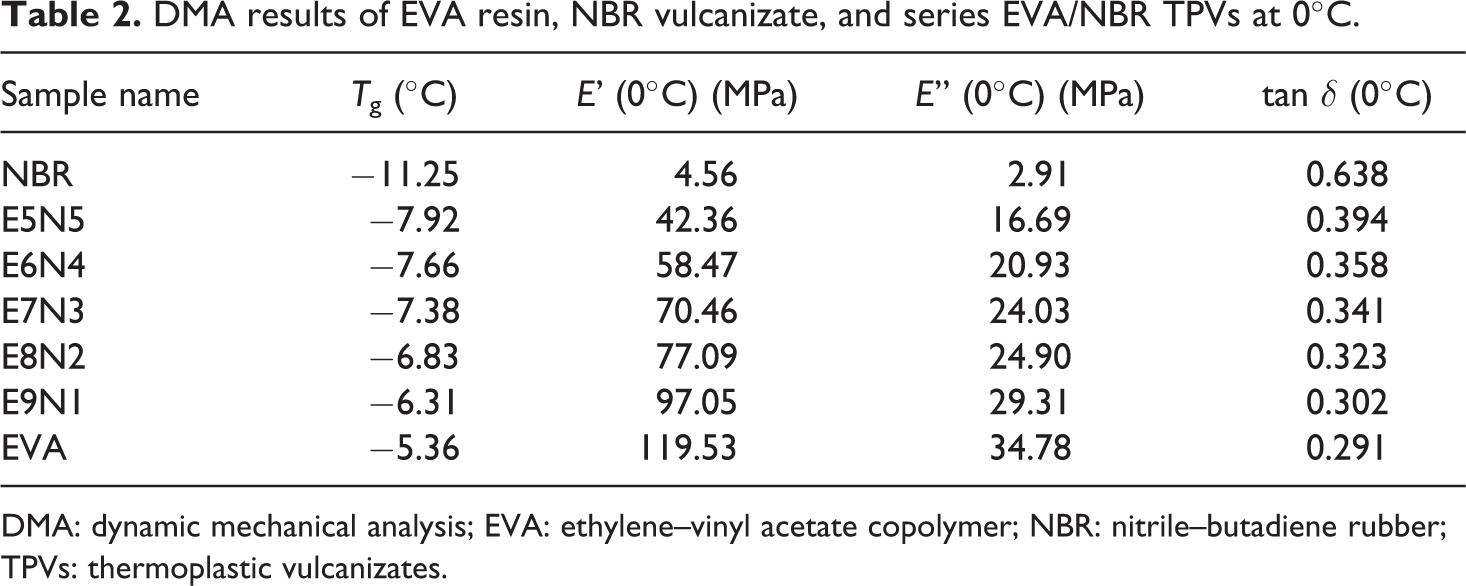
DMA: dynamic mechanical analysis; EVA: ethylene–vinyl acetate copolymer; NBR: nitrile–butadiene rubber; TPVs: thermoplastic vulcanizates.
Shape-memory process
Figure 3 shows a typical heat-triggered shape-memory behavior in a tensile model for an E8N2 specimen while both

Shape-memory behavior in a tensile model for E8N2 while both
Figure 4 shows the entire shape-memory process visually for an E8N2 specimen while both

Shape-memory behavior for an E8N2 specimen while both
Shape-memory effects
To determine an appropriate
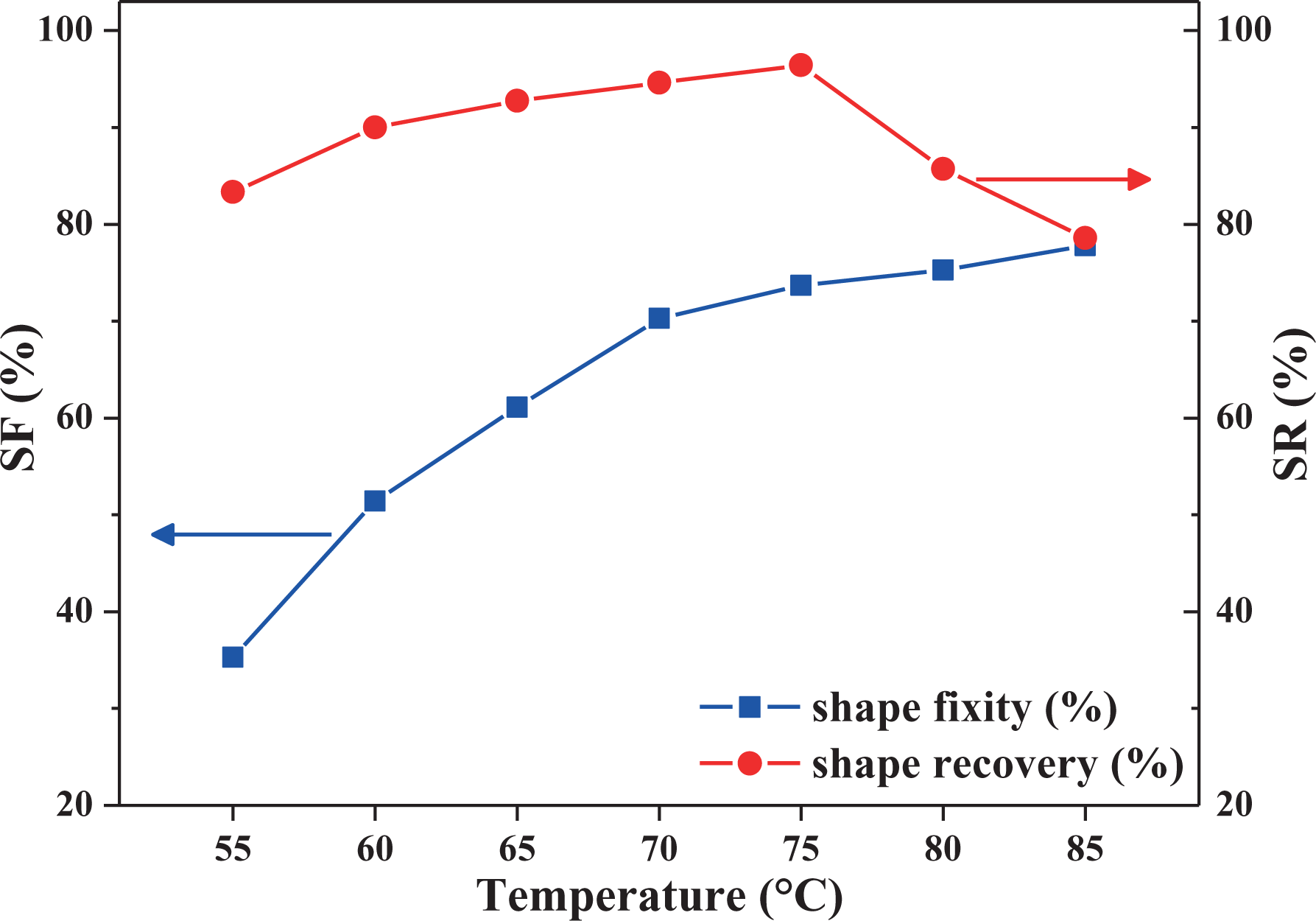
SF and SR ratios for E6N4 obtained at various
To provide an overall understanding of the SME behavior, Figure 6 shows the SF and SR ratios of TPVs with various EVA/NBR weight ratios, while both the
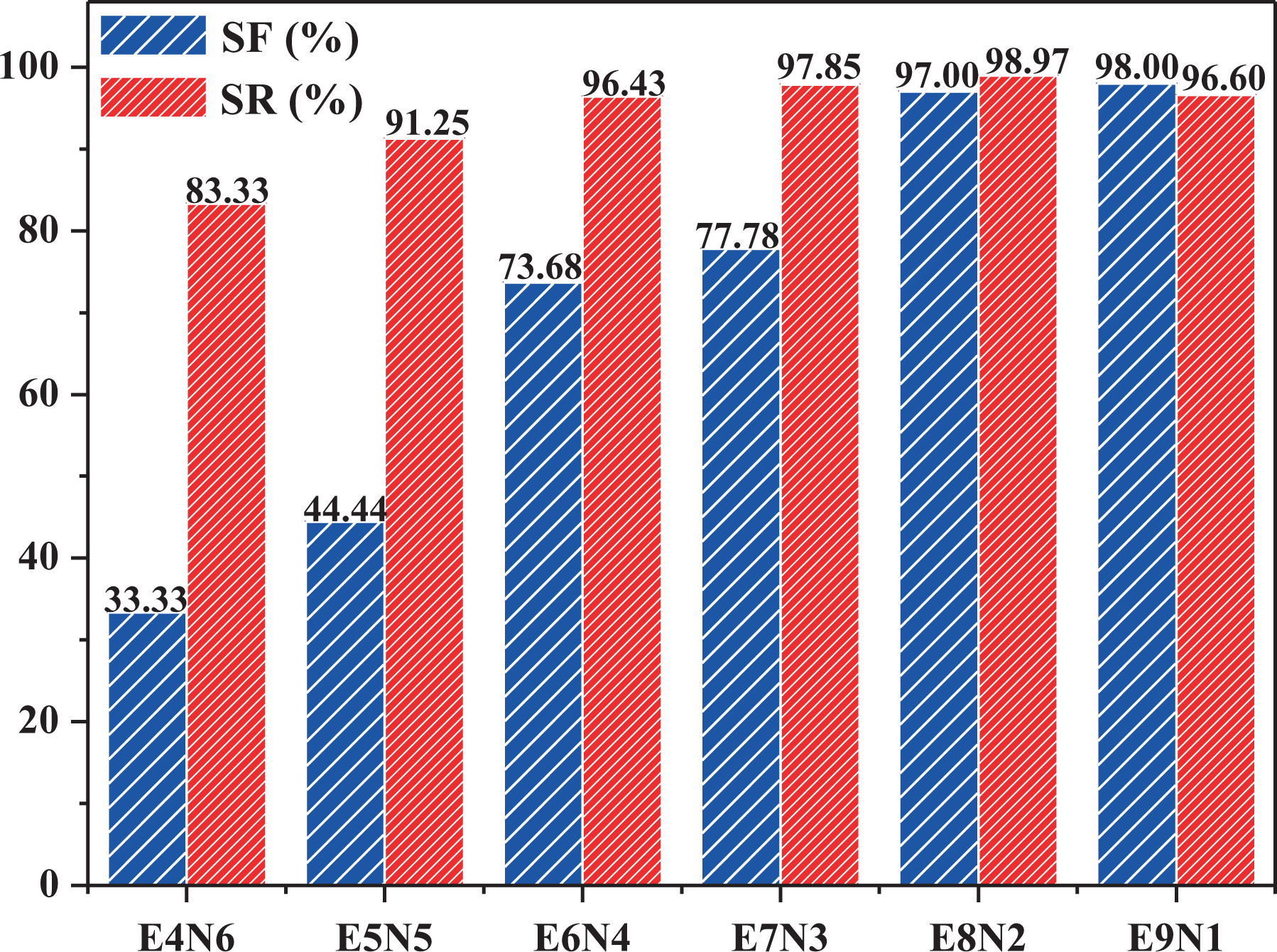
SF and SR ratios of TPVs with various EVA/NBR weight ratios while both
Figure 7 shows the SF and SR ratios of E8N2 specimens obtained at various
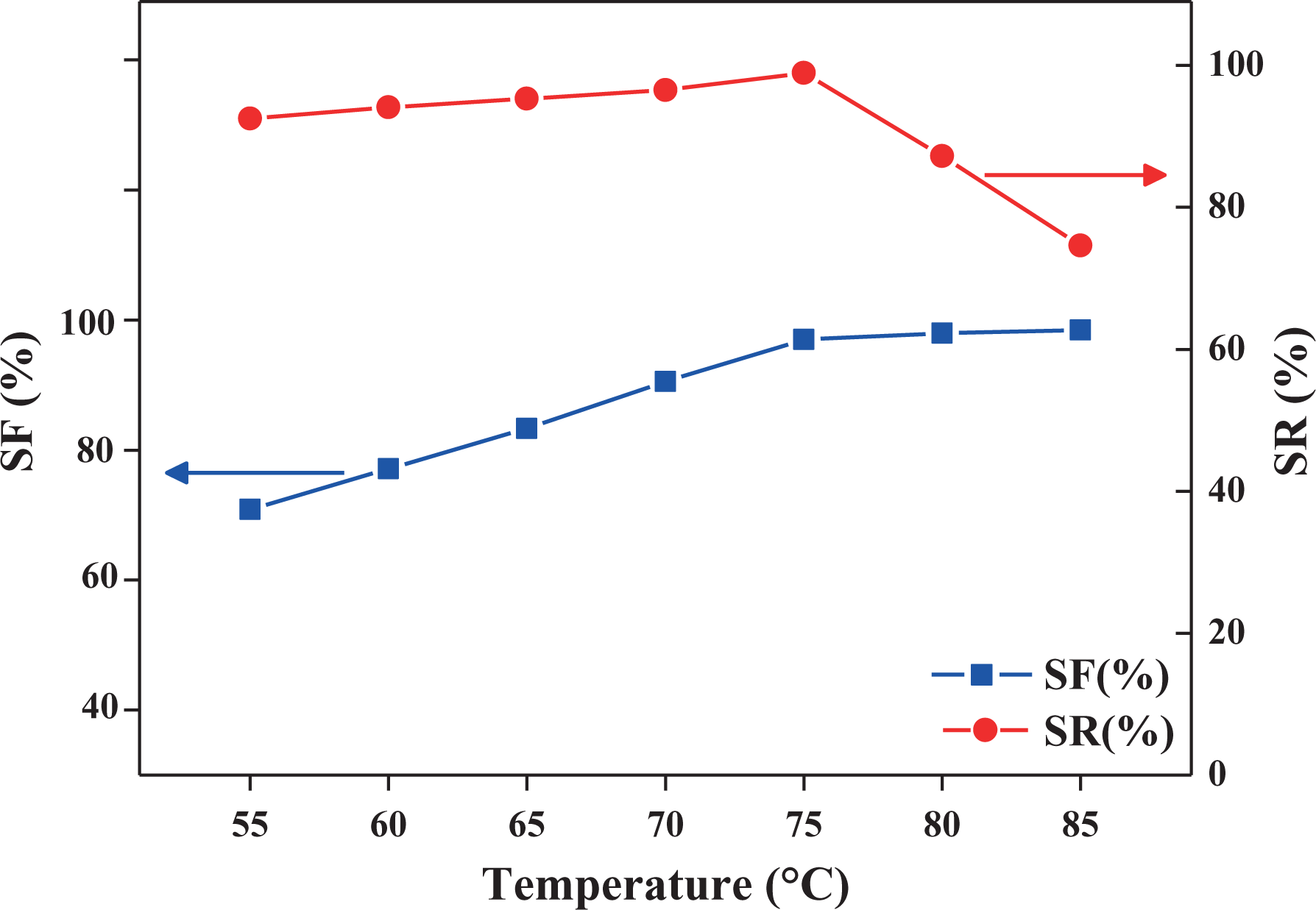
SF and SR ratios for E8N2 obtained at various
Based on the above research, we deduced a schematic structure of the EVA/NBR TPV during deformation to demonstrate the SME mechanism while
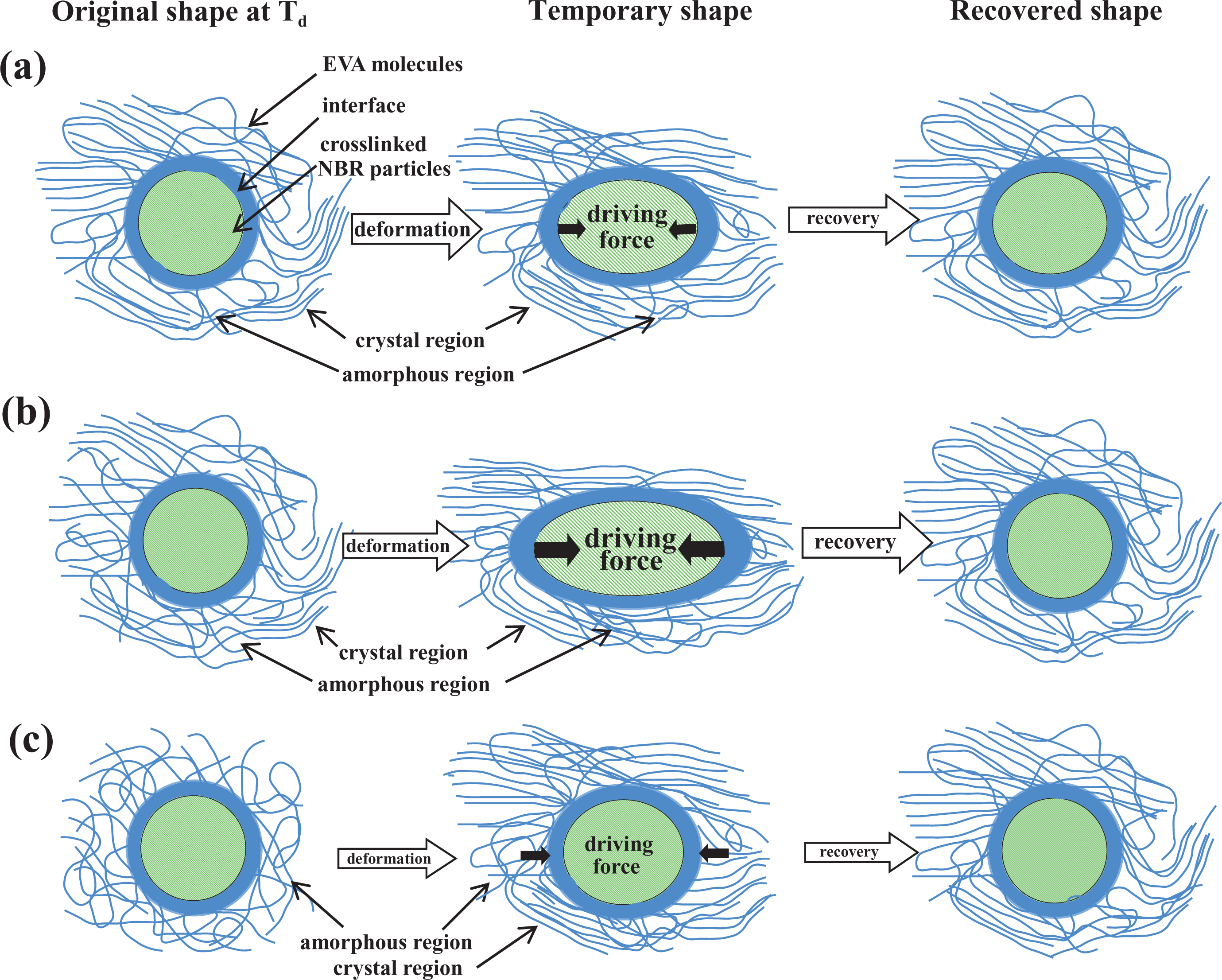
Schematic representation of the detailed structure of an EVA/NBR TPV during deformation while
From Figure 8(a), we could understand that when the
Figure 9 shows the correlation curve between the SR ratio, the time required to reach the maximum SR ratio, and
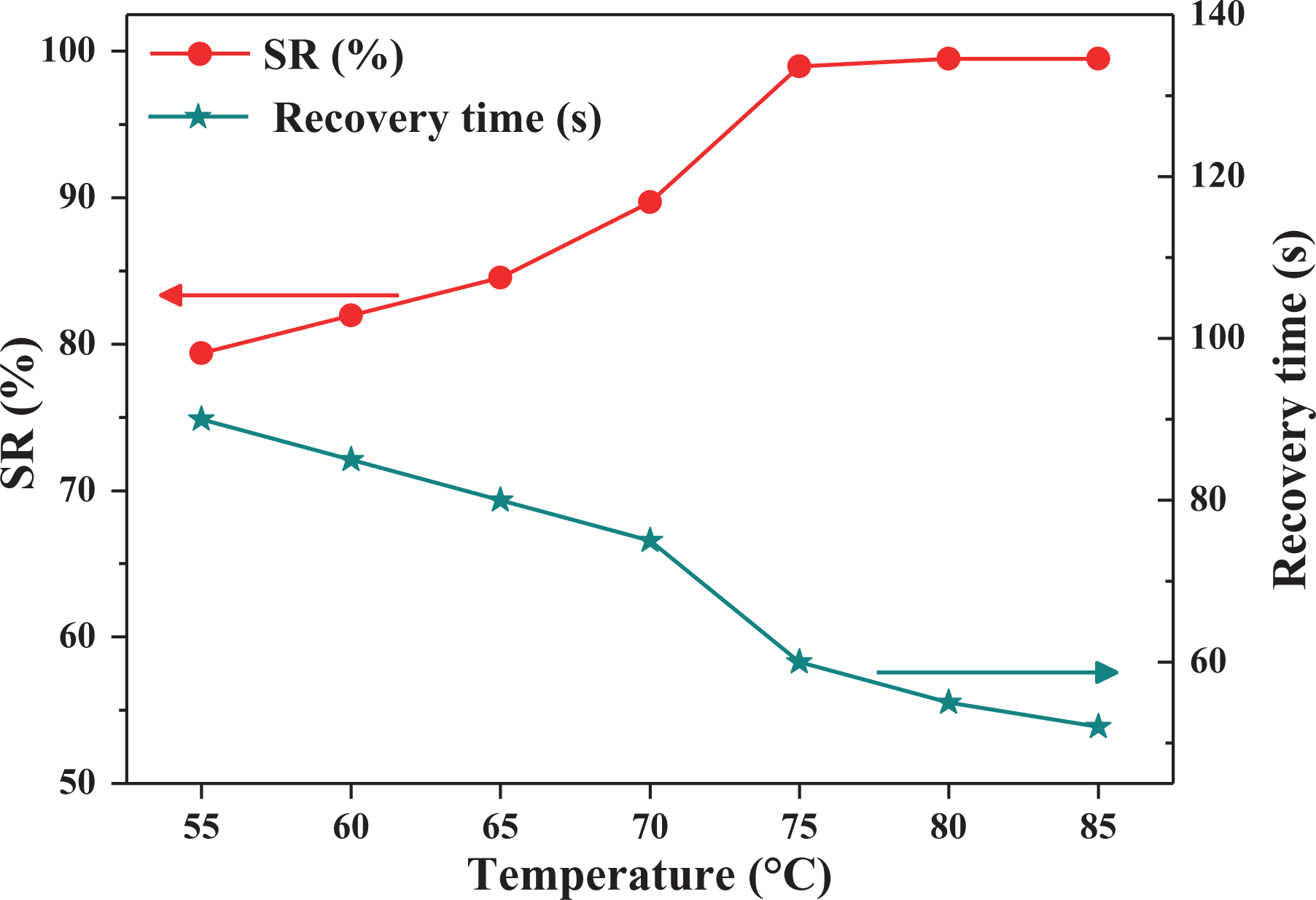
Correlation curve between SR ratios, the time required to reach the maximum SR ratio and
In the present research, to assess the SR behavior of the TPV under the different heating atmosphere, the SR was carried out both in the hot water and in the heating chamber. Figure 10 shows the correlation curve between SR ratios and recovery time for E8N2 in the different recovery environments while both the
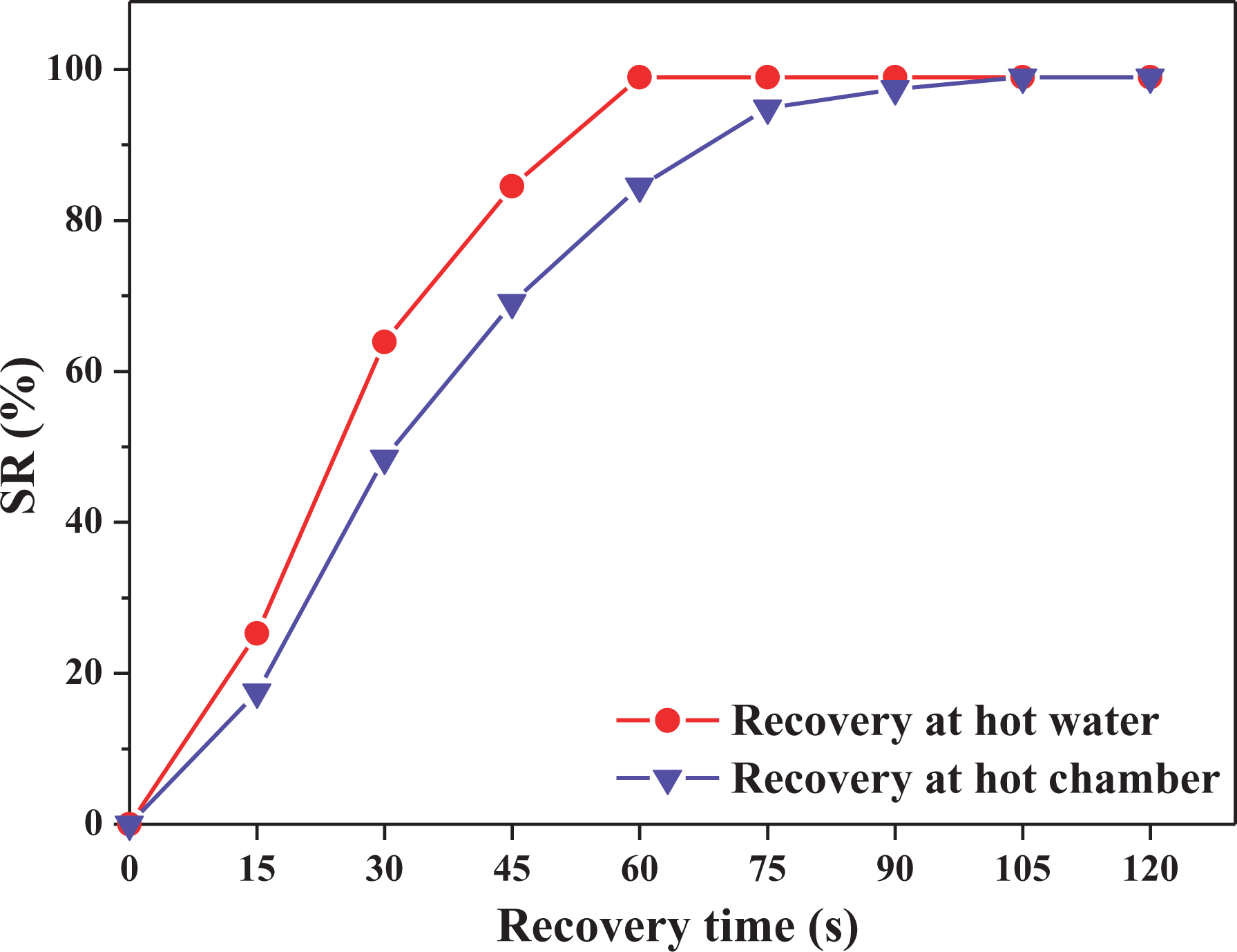
Correlation curve between SR ratios and recovery time for E8N2 in different recovery environments while both
Morphology and microstructure of the EVA/NBR TPV
The FE-SEM image of the etched surface of the E8N2 specimen is shown in Figure 11. The EVA matrix in the TPV surface was etched in order to provide a better insight into the phase morphology. From Figure 11, it could be seen that the vulcanized NBR rubber domains remained undissolved and adhered to the surface. The cross-linked NBR particles with irregular morphologies were dispersed evenly in the thermoplastic matrix with an average diameter of approximately 2–6 μm, which was benefit to the stress delivery during deformation of TPVs. In the EVA/NBR TPV, the dispersed NBR phase was acted as the reversible phase, while the EVA matrix was acted as the switching phase, which determined the

FE-SEM image of etched surface of E8N2.
To investigate the details of the microstructure upon deformation, the FE-SEM images of the tensile fractured surface and the longitudinal stretching surface of the E8N2 specimen are presented in Figure 12(a) and (b), respectively. It must be pointed out that the tensile speed was 50 mm min−1, which was quite slower than a common testing speed for vulcanizates. The slow tensile rate ensured that the stress transferred effectively through the interface and worked on the rubber particles. As shown in Figure 12(a), the tensile fractured surface of E8N2 consisted of numerous strip-like fibers; however, no rubber particles were observed on the fracture surface due to the firm adhering and coverage of the EVA matrix. Upon elongation, the thermoplastic layers around the NBR particles were subjected to plastic yielding; upon relaxation, they can fix the deformed NBR elastic rubber domains effectively and store the driving force for SR. Figure 12(b) shows the detailed microstructure of the distinct orientation texture on the longitudinal stretching surface. Numerous banding-like morphology microstructure oriented along the tensile direction can be observed clearly, which was related to the deformed EVA phase and the elongated NBR phase. During the stretching process, the plastic deformation of the EVA phase resulted from the stress field caused the deformation of NBR particles where the tensile stress was transmitted through the interface between the EVA matrix and dispersed NBR particles. Undoubtedly, this physical microstructure helps to exhibit the shape-memory behavior for the EVA/NBR TPV.

FE-SEM images of E8N2: (a) tensile-fractured surface; (b) longitudinal stretching surface.
Conclusions
This study presented a novel HSMP derived from the EVA/NBR TPV. The DMA results under temperature sweep mode showed that with the increasing EVA content in TPVs, the tan
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Shandong Provincial Natural Science Foundation, China (ZR2017MEM021) and Upgraded Project of Shandong Province for Guidance Ability of Graduate Tutors (SDYY17044).
