Abstract
This study introduces the synthesis of graphite-carbon nitrite (g-C3N4) using thermal treatment of urea. The prepared powder is treated to generate nanoparticles. The structure and grain size of the g-C3N4 have been obtained via X-ray diffraction (XRD) technique and transmission electron microscope. The graphite-carbon nitride on Polyvinyl Chloride (PVC/g-C3N4) nanocomposite films were produced by mixing different concentrations (0, 3, 5, 7, 10 and 20 wt%) of nanoparticles with Polyvinyl Chloride (PVC) solutions. The structure and elemental composition of the prepared nanocomposites films PVC/g-C3N4 were studied by using XRD, scanning electron microscope (SEM), (EDX), infrared spectroscopy (IR) and electron spin resonance (ESR). Also, spectral distribution of the optical absorbance for the PVC/g-C3N4 nanocomposites films was studied in the wavelength range from 190 to 1100 nm. Thermal stability of the investigated nanocomposites films was investigated by thermal gravimetric analysis technique (TGA). The effect of various concentrations of the g-C3N4 and different doses of gamma-rays on the polymeric matrix and electrical properties of PVC film also has been investigated. The g-C3N4 additives on PVC influenced the crystal structure and optical properties of the nanocomposites films. The nanocomposite film with 10% g-C3N4 achieved higher values of the refractive index and optical conductivity compared with the other films. With different doses of gamma-ray irradiation, the crystal structure and optical properties of PVC/g-C3N4 nanocomposite films were changed in accordance to the cross-linking process within the PVC polymeric chains where a structural rearrangement can be occurred as a result of gamma irradiation. The elemental composition and morphological study of PVC/g-C3N4 nanocomposite films have been checked by the scanning electron microscopy which indicated the homogeneous dispersion of g-C3N4 nanoparticles within PVC network. Finally, electrical properties of PVC/g-C3N4 nanocomposite films with different ratios and distinct gamma doses also have been studied.
Keywords
Introduction
Carbon nitride with several allotropes can be considered as the oldest synthetically polymeric materials. 1 Graphitic carbon nitride (g-C3N4) as an emerging, and non-toxic organic semiconductor material is the most stable state which can be used active photocatalyst for water splitting, CO2 reduction, and degradation of harmful organic compounds.2–5
In recent years, attention has been paid to an increase polymer such as graphite carbon nitride (g-C3N4), which can The decomposition of the organic pollutants and the water split into the H2 product below Visible light irradiation as a metal-free photocatalyst At a low cost, power with an appropriate band-gap energy of 2.7 eV, and outstanding thermal and chemical stability.6,7 After the first report of a metal complex/C3N4 hybrid for CO2 reduction, several groups have presented similar reports using cobalt-based metal complexes as reduction cocatalysts.8,9 In these systems, structural properties of g-C3N4 such as specific surface area and porosity have a strong impact on activity, because they strongly affect the efficiency of electron/hole utilization to the surface chemical reactions.10,11 Polymer composites are widely used in industrial applications due to their ability to combine the desired properties of various polymeric species. In recent years, a great deal of interest has been focused on the optical and electrical properties of polymers in optical device technology due to their unique polarization, interference, reflection, and antireflection properties.12–17 In particular, polyvinyl chloride (PVC) belongs to the most frequently used materials in the creation of nanocomposites because of its beneficial properties, such as its comparatively low cost, extensively developed processing, the possibilities of modification of its mechanical properties, and its high environmental resistance.18–20 The production and application of this polymer are constantly growing worldwide. The restrictions of PVC applications are mostly related to the relatively high glass transition temperature of this polymer, resulting from strong polar interactions in the PVC molecules as a consequence of the presence of chlorine atoms. 21 It is well known that the glass transition temperature depends on the molecular structure and molecular weight of the polymer. Moreover, its thermal history depends on the measurement method and the rate of heating or cooling, as well as on many other factors, including the use of various processing aids, such as plasticizers and nanofillers.22,23 However, the most frequently used modifiers of PVC glass transition temperature are specific plasticizers, in the form of small molecules, which are introduced into a polymer macromolecular structure and usually lead to a reduction in the Tg value. 24 Despite a large number of sources25–27 that report on the impact of nanoadditives on the properties of PVC nanocomposites, only some of them concern the influence of nanofillers on the thermal properties of this polymer in its glassy state. The aim of this study is to investigate and analyze the influence of various nanofillers on the processing and physical properties related to polymer vitrification, which is of interest to both industry and science.
Experimental techniques
Materials
Polyvinyle chloride (PVC) was obtained by Sabic Company, Saudi Arabia. It was in the form of a powder (average size of 100 mm, powder fraction of 90–120 mm, density = 1.37 g.cm−3) was utilized as a polymeric matrix for preparation of composites.
Tetrahydrofuran (THF) was obtained from Qualikems fine chemicals Pvt. Ltd New Delhi, India. Urea (99%, M.W 60.06, Melting point 131–134°C) was provided by El-Nasr Pharmaceutical Chemicals Company, Egypt. Nitric Acid (55%, M.W63.01) was supplied from Elsalam for chemical industries, Egypt.
Preparation of g-C3N4
Graphite carbon nitride (g-C3N4) was synthesized by heat treatment of urea (15 g) in a crucible under ambient pressure in air. After drying at 80°C, the urea was placed in a covered crucible and heated in an oven at 500–550°C (heating rate 20°C/min) for 3 h to complete the reaction to obtain g-C3N4 which is yellow color. To remove any residual alkaline species (eg ammonia) that have been adsorbed onto the sample surface the yellow colored product (g-C3N4) is washed with nitric acid (0.1 mol/L) and distilled water, then dried at 80°C for 24 hours. To obtain nanoparticles of g-C3N4, ethanol is added to g-C3N4 and ultrasonic processor for 60 minutes, as shown in Figure 1. Synthesis of g-C3N4 nanoparticles by thermal treatment of urea.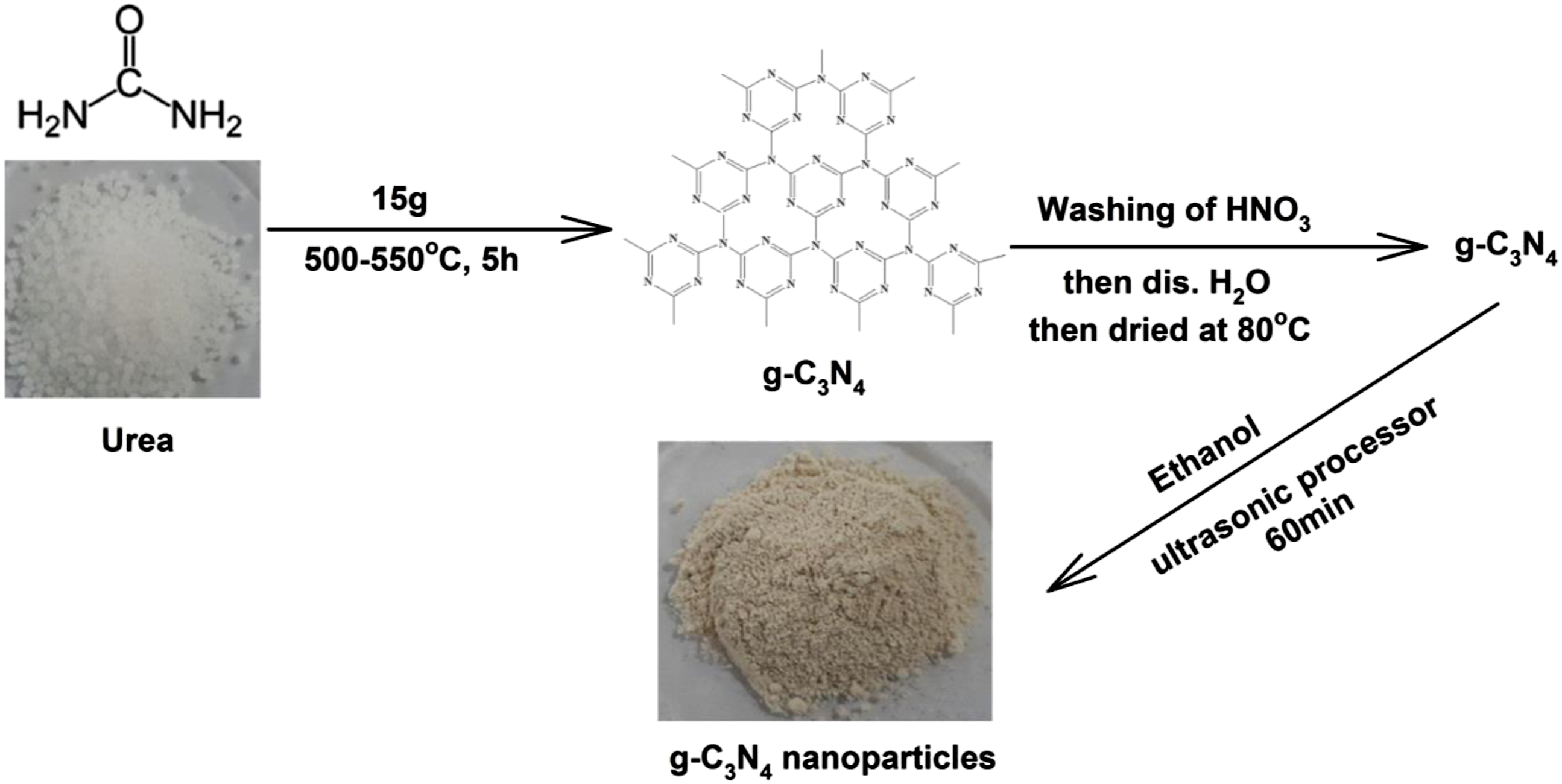
Preparation of PVC/g-C3N4 nanocomposites
PVC solution (10%) was prepared by PVC dissolved in THF at 60°C for 30 min and then mixed with g-C3N4 nanoparticles. This solution was then sonicated for 30 min to obtain PVC/g-C3N4 nanocomposites solution. PVC/g-C3N4 nanocomposites were prepared with different concentrations of g-C3N4 nanoparticles (0, 3, 5, 7, and 10 wt%). According to the findings, the optimum nanocomposite is nanocomposite (PVC/10%g-C3N4), was exposed to gamma irradiation for a dose range of 5–20 kGy, and also te optimum irradiation dose is 20 kGy. The g-C3N4 nanoparticles were mixed with PVC using an ultrasonic processor. A number of samples with a thickness of 1.3 mm were prepared by pouring into Petri dishes. Square sheets of prepared samples were irradiated in the presence of air at different doses (5–20 kGy), using a cobalt-60 gamma radiation source manufactured in Egypt Mega, Gamma 1, supplied by Canadian Atomic Energy at a dose rate of 1.1 kGy/hr, at the National Center for Radiation Research and Technology Cairo, Egypt.
Characterization techniques
The surface morphology, elemental composition and average grain size of PVC and PVC/g-C3N4 nanocompositefilms have been evaluated by using a scanning electron microscope (SEM). Model ZEISS- EVO 15- UK. The X-ray diffraction (XRD) patterns of PVC and PVC/g-C3N4 nanocomposite films have been obtained by using a Shimadzu XD-DI Diffractometer operated at 40 kV and 30 mA. Thermogravimetric analysis (TGA) of PVC and PVC/g-C3N4 nanocomposite films was performed using Shimadzu-50 TGA in Nitrogen atmosphere. The infrared absorption spectra of the nanocomposite films within the wavenumber range of 4000–400 cm−1 have been recorded at the ambient conditions by a Bruker’s VERTEX 70 FTIR Spectrometer. A double beam Shimadzu UV-VIS spectrophotometer was used to measure the spectral distribution of the absorbance within the wavelength range 200–1100 nm. The influence of gamma-irradiation with different doses (5–20 kGy) on the structural and optical properties of the PVC and PVC/g-C3N4 nanocomposite films also has been studied.
Results and discussion
X-ray diffraction
XRD is used to observe different types of polymers phases (crystalline, semi-crystalline and amorphous). To investigate the microstructure evolution of PVC, g-C3N4 and PVC/10%g-C3N4 before and after 20 kGy, XRD analysis was performed. Figure 2 show the XRD patterns of pure PVC, g-C3N4 and PVC/10%g-C3N4 before and after 20 kGy. The XRD pattern of the PVC film showed broad peaks for values of 2θ around 23.3° and 41°, confirming the structure of the amorphous nature of PVC.
28
The XRD diffraction pattern of the g-C3N4 nanoparticles a pattern of the sample of g-C3N4 is characterized by diffraction peaks at angles 14.1°, and 33.1° is an indication of the formation of g-C3N4, which are assigned to an in-planar repeating motif and the stacking of the conjugated aromatic system, respectively.
29
Notably, the crystal state is the dominating crystalline phase for pure g-C3N4. The introduction of g-C3N4 nanoparticles in the PVC resulted in similar peaks, and the presence of the g-C3N4 nanoparticles was confirmed by the peak readings. The new peaks located at 14.7°, 24°, 33.8° and 43.1° are associated with the g-C3N4 nanoparticles for PVC/10%g-C3N4 before irradiation and also the new peaks located at 14.7°, 24°, 33.8° and 43.1° for PVC/10%g-C3N4 at 20 kGy. Furthermore, the XRD pattern of the PVC film which exhibits abroad hump indicating the amorphous nature of the PVC film. While the XRD patterns of PVC/10%g-C3N4 nanocomposites before and after 20 kGy show diffraction peaks at 14.7°, 24°, 33.8°, 43.1° 14.7°, 24°, 33.8° and 43.1° corresponding to (100), (110), (200), (002), (102) and (210) planes of g-C3N4.26,28 Also, as we note from Figure 2 the peaks intensity increases with the g-C3N4 ratios which indicate the improvement of the crystal structure of the PVC/g-C3N4 nanocomposite films. As shown from Figure 2 the intensity of the diffracted XRD peaks for PVC/10%g-C3N4 nanocomposites film increase with the irradiation dose (20 kGy) which indicates the improvement of the crystal structure of the nanocomposites film. XRD pattern of PVC, and g-C3N4 and PVC/10%g-C3N4 before and after 20 kGy.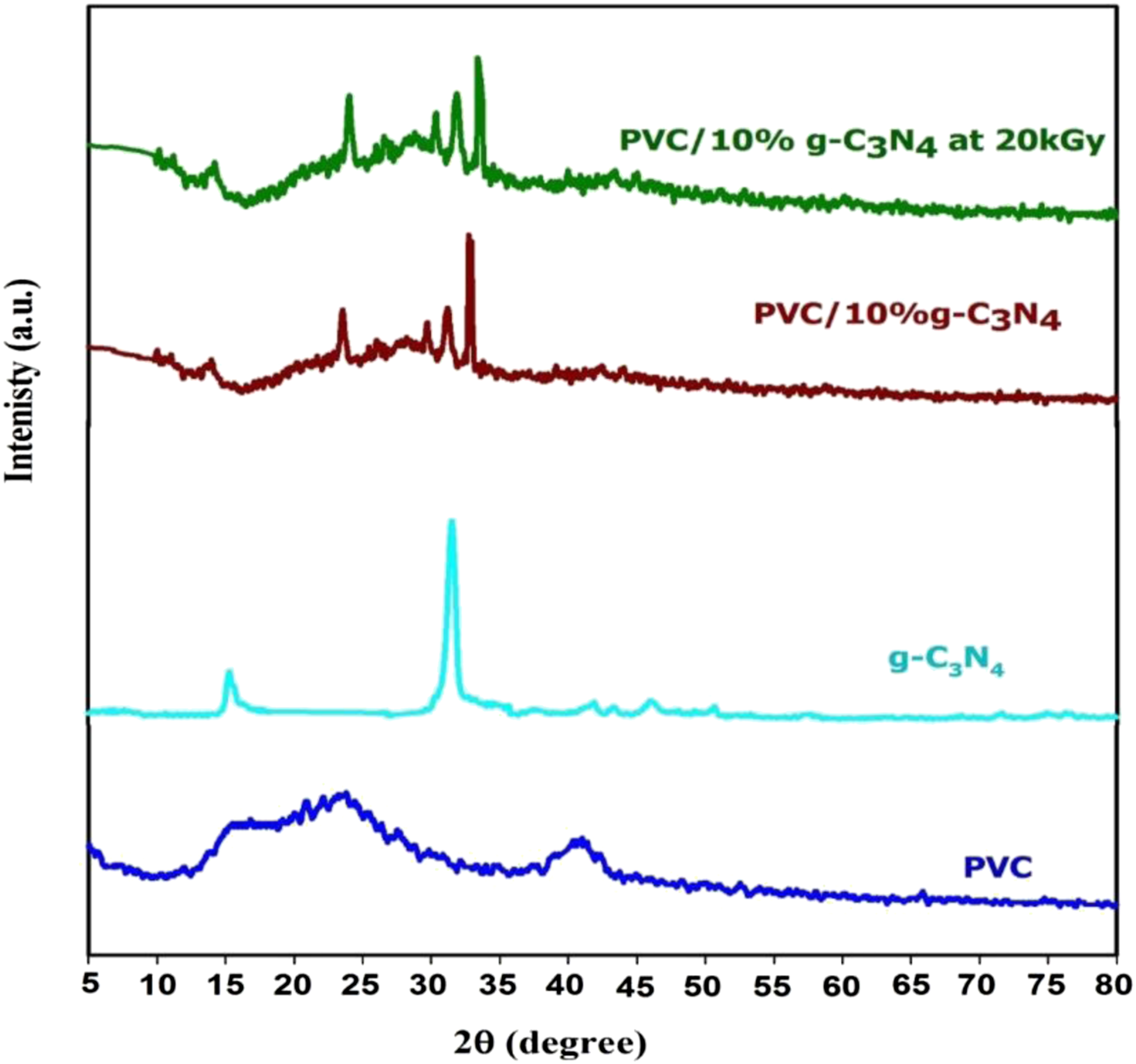
FTIR spectroscopic analysis
The infrared spectra of pure PVC and PVC/g-C3N4 nanocomposite films were measured at room temperature to investigate the interaction between g-C3N4 nanoparticles and PVC matrix. Figure 3 shows FT-IR spectra of PVC, g-C3N4 and PVC/10%g-C3N4 before and after 20 kGy. FTIR transmittance spectra of pure PVC, g-C3N4 nanoparticles and PVC/10%g-C3N4 nanocomposite before after irradiation at 20 kGy.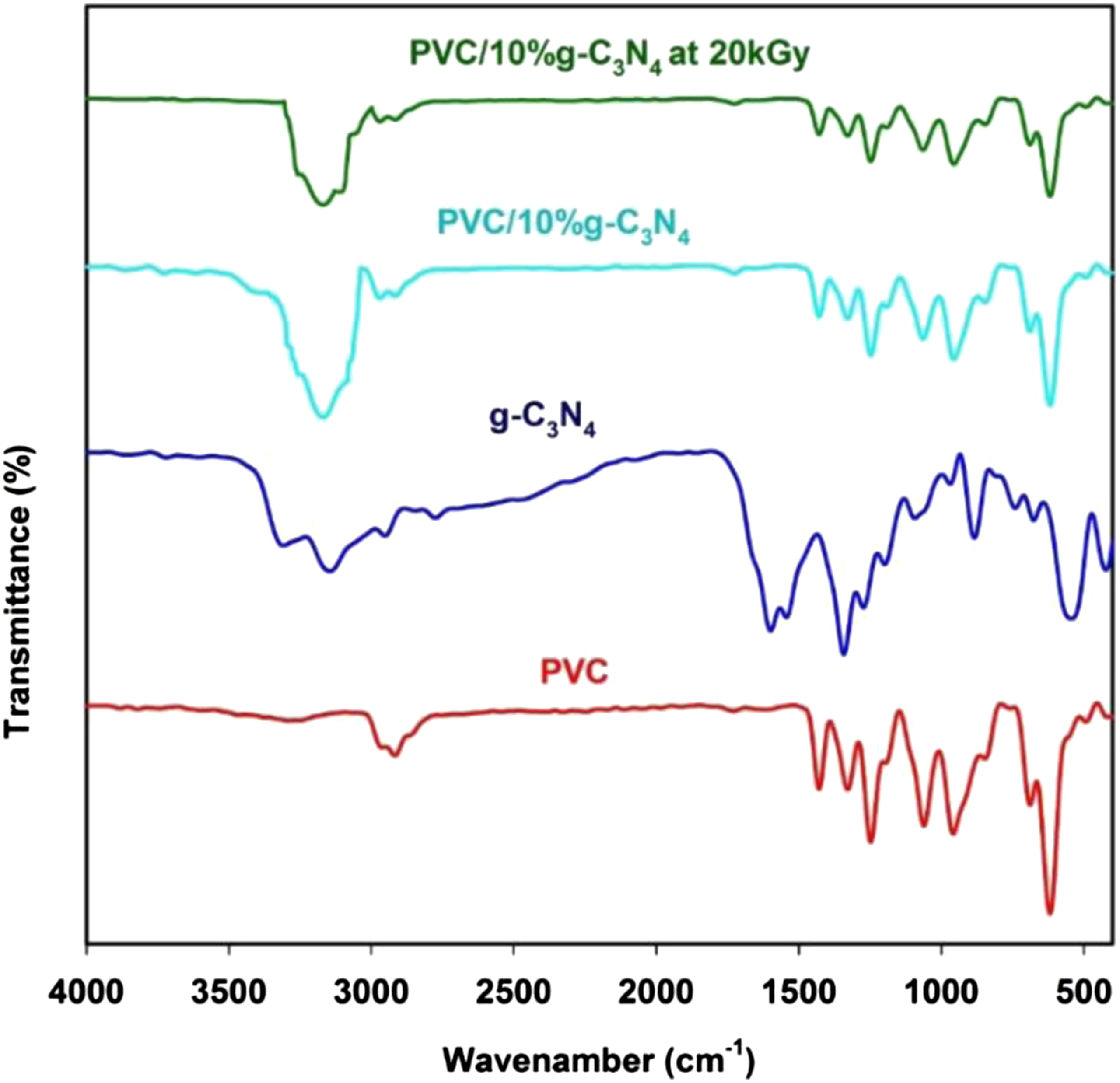
In the spectra of the PVC and PVC/GN shown in Figure 3, bands at the wave numbers of 2941 cm−1 and 1435 cm−1 were observed, attributed to the presence of C–H bonds in the PVC macromolecule.30,31 The peak at 1255 cm−1 corresponds to bending vibrations of the C–H originating from CHCl.20,24 The bands of C–C at 1075 cm−1 and a high intensity band assigned to the stretching vibration C–Cl at 690 cm−1. 32
The slightly broader vibration bands at around 3205 cm−1 indicate N−H stretching vibrations on the edges of g-C3N4, suggesting more compact interlayer interaction 33 as shown in Figure 3. The vibration bands at 1000–1690 cm−1 are attributed to the CN stretching vibrations in the CN heterocycles. The strong band at 660 cm−1 is ascribed to the vibration mode of the triazine-based unit. 34
The change in FTIR of PVC due to the changes in structures of the polymeric systems can be used to examine how the incorporation of g-C3N4 nanoparticles influences the chemical properties of the PVC polymer. By comparing the infrared spectrum of PVC with the spectrum of PVC/10%g-C3N4 before and after 20 kGy, it is clear that there is a significant shift in position of bands which can be attributed to interaction between g-C3N4 nanoparticles and host PVC polymer matrix.
Furthermore, the introduction of g-C3N4 nanoparticles in the PVC matrix is accompanied by a decrease in the intensity of peaks characteristic for the polymeric matrix. Also, we note that the peak at 1075 cm−1 became weaker when g-C3N4 nanoparticles were added to the PVC and The new peak appeared at 3197 cm−1. 35
As shown in Figure 3, the exposure of the PVC/10%g-C3N4 nanocomposite at 20 kGy films to gamma ray irradiation decreasing and damping all of the transmittance peaks in comparison to the fresh peaks. This damping can be explained as a result of the cross-linking process of the PVC/10%g-C3N4 nanocomposite chains under the effect of the gamma-ray irradiation.36,37 The FT-IR analysis strongly confirm that all of the main characteristic vibration bands of g-C3N4 nanoparticles were successfully embedded with PVC matric chains.
Scanning electron microscope analysis
The morphology of the surface of the PVC and PVC/10% g-C3N4 before and after irradiation was examined by the SEM, at different magnification powers, at room temperature. The SEM images indicated that the surface of the PVC before irradiation was generally smooth (Figure 4(a)). However, the SEM images recorded after irradiation (Figure 4(b)) showed a damaged within the PVC surface and formation of many cracks as a result of photodegradation of polymeric chain. It was evident that there was no PVC porous structure before irradiation but after irradiation the PVC appeared porous. SEM micrographs of (a) pure PVC (b) PVC at 20 kGy (c) PVC/10%g-C3N4 nanocomposite (d) PVC/10%g-C3N4 nanocomposite at 20 kGy.
Figure 4(c) and (d) shows the SEM images for PVC/10% C3N4 nanocomposite before and after irradiation. Figure 4(c), showing the surface of PVC after adding g-C3N4 nanoparticles is a homogeneous disperse of g-C3N4 nanoparticles appearing as large granules on the surface of the PVC. After the irradiation process at 20 kGy, the surface of the PVC was modified and the surface became rough and most of the voids disappeared or became smaller as in Figure 4(d) indicating the linking process within the PVC chains.
Thermal analysis
TGA investigation delivers direct indication around thermal stability and degradation evidence by evaluating weight loss of specimen as a function of temperature. Figure 5 shows the TGA curves of PVC and PVC/10%g-C3N4 nanocomposite before and after irradiation at 20 kGy. TGA curves of PVC and PVC/g-C3N4 nanocomposites films before end after irradiation at 20 kGy.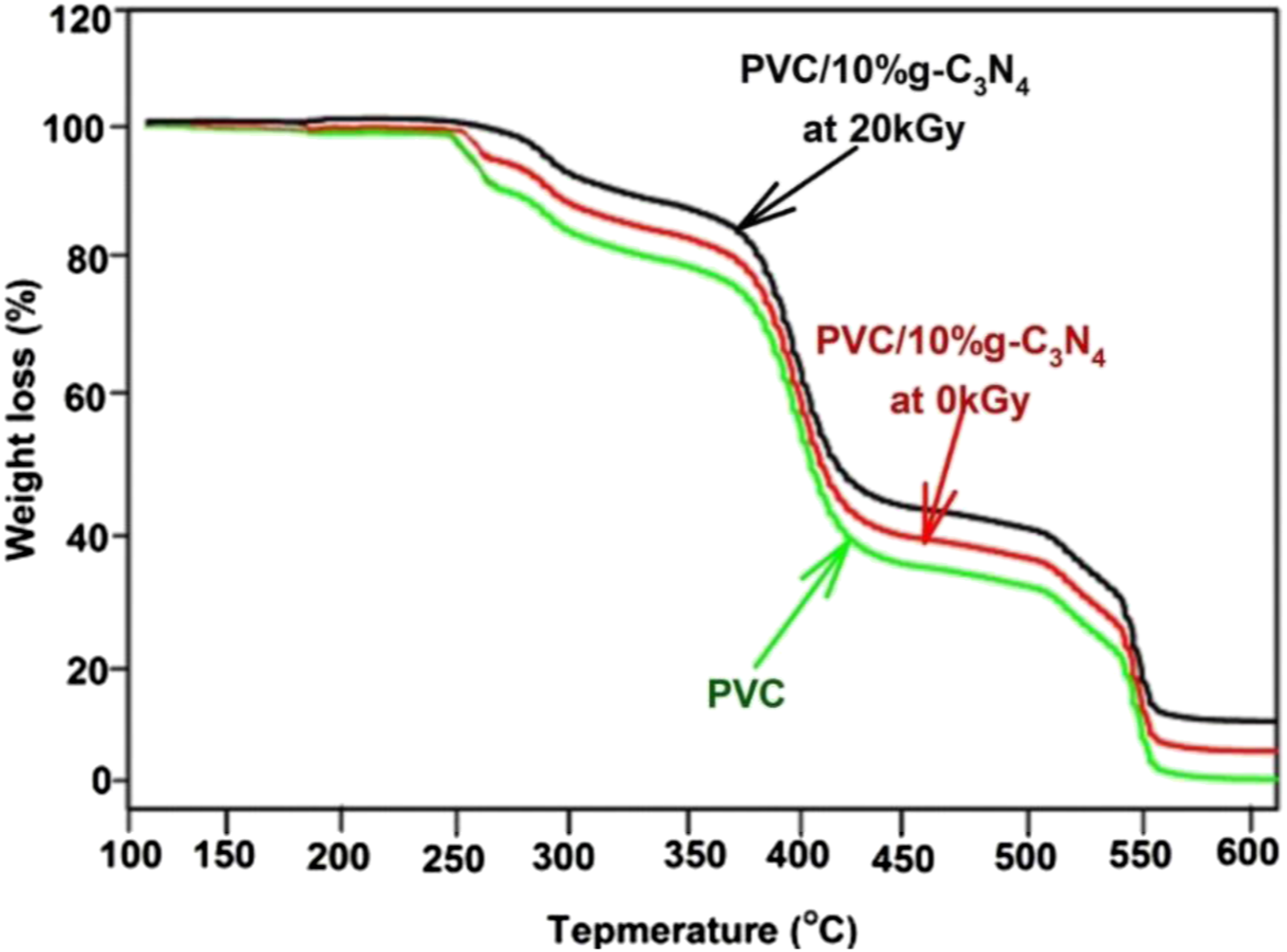
There are three decomposition steps in the temperature ranges from 250°C to 380°C, from 380°C to 430°C and from 380°C to 550°C. The main loss of the weight was in the first temperature range from 250 to 380°C. The pure PVC decomposed in the first stage then the second stage of loss is due to the degradation of the PVC polymer structure in the temperature range from 380°C to 550°C. As we noted also from Figure 5, the residual quantity of weights is larger in the case of PVC/g-C3N4 films than that in pure PVC polymer film according to the inclusion of g-C3N4 NPs in the polymeric matrix. The two decomposition stages are attributed to the removing of side-groups in the low temperature range and according the breakdown of the PVC backbone at the higher temperature range, respectively. 38
The interior of g-C3N4 NPs in the PVC polymeric matrix reduce the dissociation temperature of PVC film. This effect can be explained in accordance to the strong coupling between g-C3N4 NPs and PVC polymeric chains, where the great internal connection between PVC chains within the hydrogen bonding was decreased with the addition of g-C3N4 NPs and hence the thermal stability. 33 According to TGA outputs the incorporation of g-C3N4NPs in PVC matrix and irradiation at 20 kGy obviously changes the thermal stability and degradation of PVC film to lower values by 100°C of temperature.
Optical analysis
Figure 6 shows the spectral distribution of absorbance in the wavelength range from 200 to 1100 nm (UV-Vis) of pure PVC and PVC/10%g-C3N4 nanocomposite before and after irradiation at 20 kGy, where there is no optical response for PVC film. In 10% of g-C3N4, the absorbance values of PVC film increase in the UV-Vis range. Also, absorption edge of PVC polymer film has been shifted to longer wavelength with the increase of g-C3N4 in the polymeric network. The red shift in the absorption edge and the changes in the absorbance values can be ascribed to the dispersion and impact of g-C3N4 NPs in the PVC matrix. It is observed from Figure 6 also, the absorbance values increase with the irradiation dose at 20 kGy according to the cross-linking process between the polymeric chains as a result of the gamma irradiation doses. Absorbance of pure PVC and PVC/g-C3N4 nanocomposite fresh films before and after irradiation at 20 kGy.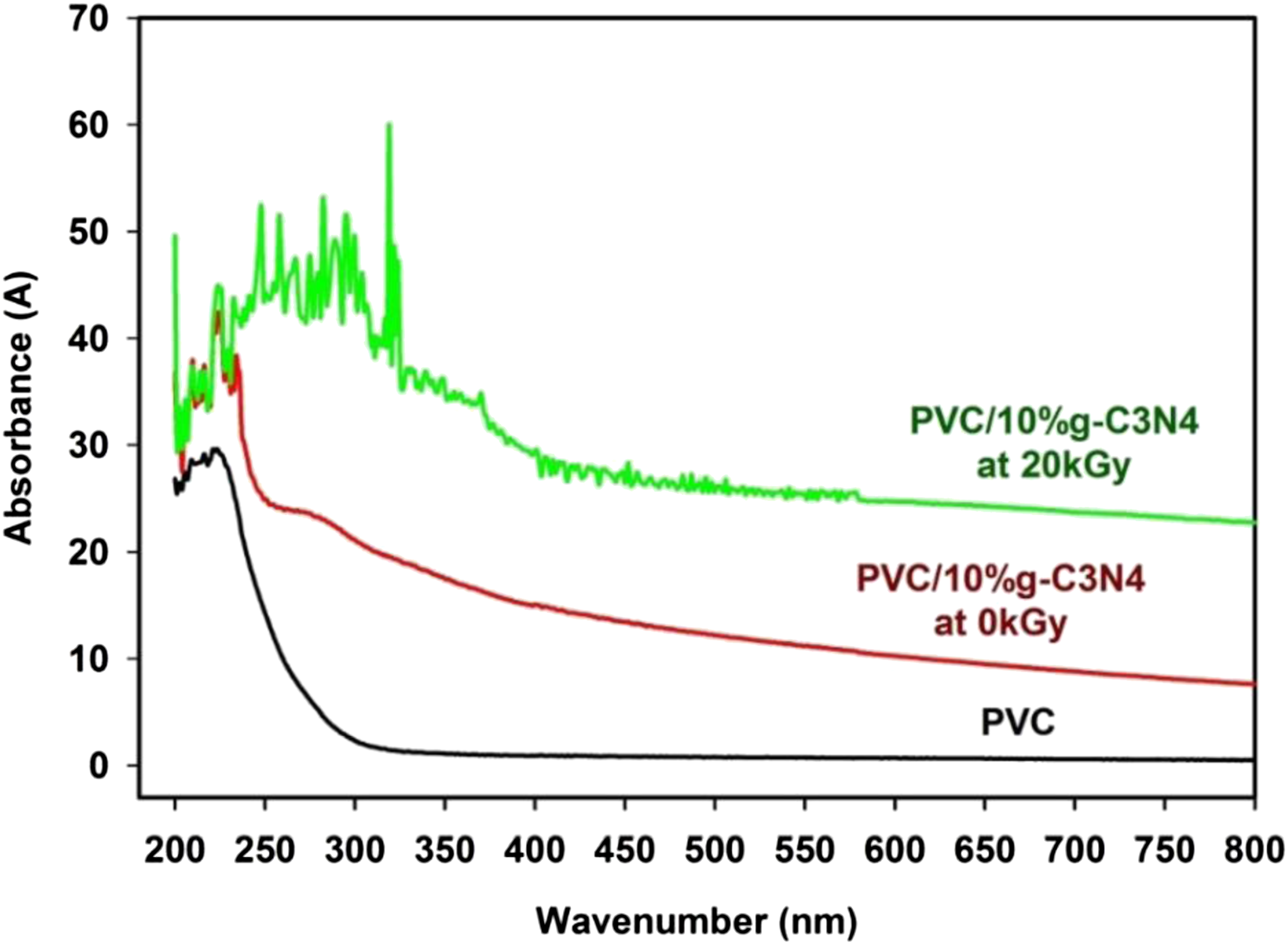
Electron spin resonance analysis
An effective method for the observation of the absorbed microwave radiation by the spins of an electron of an atom or molecule in a magnetic field is the Electron spin resonance (ESR) spectroscopy. Also determined by this technique a valuable information about the lattice site in which a ferromagnetic dopant ions are located. 39
The spectrum of the ESR for PVC and PVC/10%g-C3N4 nanoparticles before and after irradiation at 20 kGy, powder sample documented at room temperature and focused on the center of the curve in Figure 7. It is clear that in PVC/10%g-C3N4 as a DMS, the dopant atoms have two opportunities. The ESR signal amplitude versus the magnetic field of PVC and PVC/10%g-C3N4 nanoparticles before and after irradiation at 20 kGy.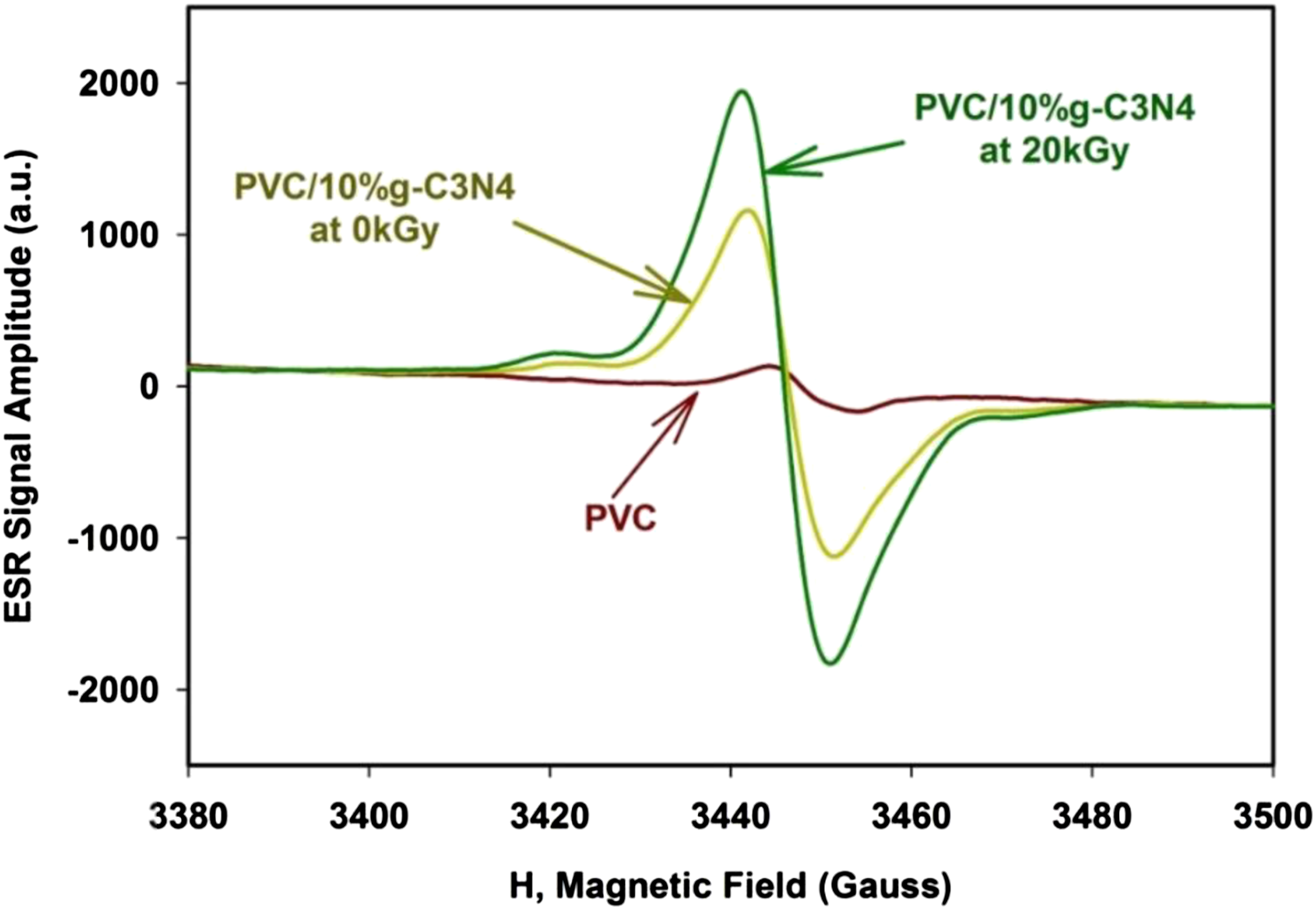
Then the ESR was reinvestigated to the irradiated sample at 20 KGy under the same condition before irradiation. Figure 7 shows the ESR signal amplitude (a.u.) for the prepared sample before and after irradiation. It is clear from Figure 7 the ESR signal increased linearly with the increasing of the irradiation dose (20 kGy).
The increase in the ESR signal appears due to the effects of gamma irradiation which breaks bonds in the g-C3N4 lattice which in turn leads to increasing of the free radicals.
Electrical properties
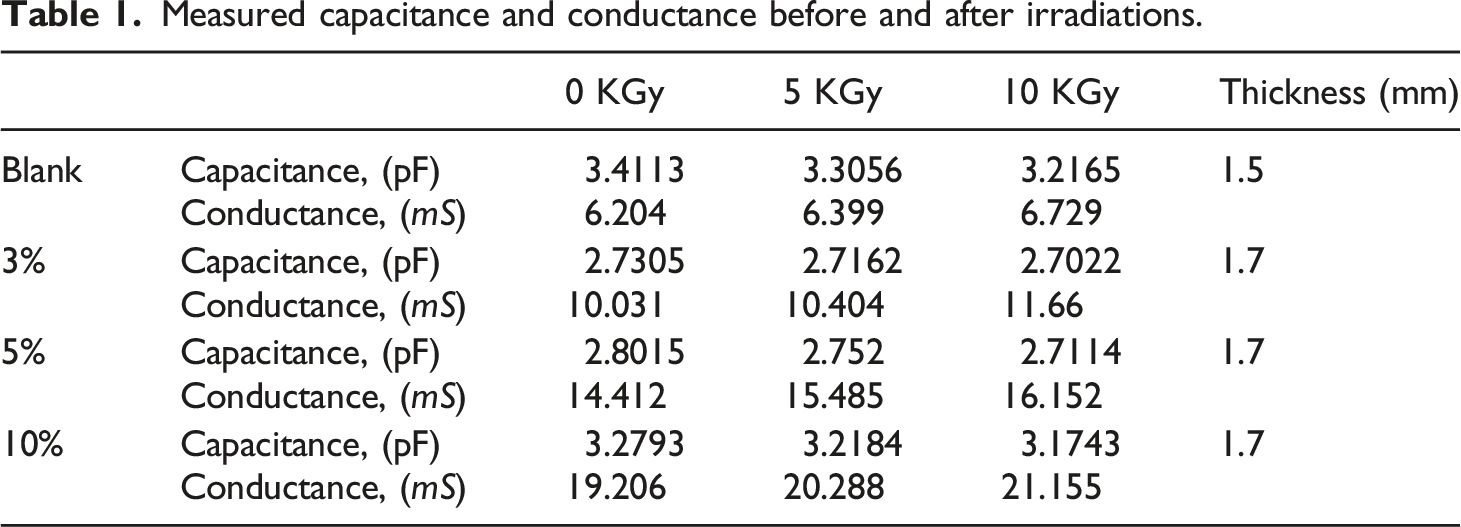
Measured capacitance and conductance before and after irradiations.
The calculated dielectric constant and conductivity.

Capacitance
Investigating Table 2, the material showing a dielectric constant of 1043.503 for 10% sample which is decreased by irradiation to 1029.567 at 10 KGy. The same response was found to all samples.
The capacitance can measured throw equation (1). With C is the measured capacitance, 
Rearranging equation (1), the dielectric constant can be found as:
Conductance
Revisiting Table 2, the electrical conductivity was 18.5126 (S/m) for the 10% sample before irradiation and increased to 20.08 (S/m) at 10 KGy gamma-ray dose. All other samples have the same response.
The conductance can be calculated using equation (3). With G is the measured conductance in (S), 
Rearranging equation (3), the conductivity can be found as:
Figure 8 shows the calculated dielectric constant of the prepared samples against frequency before and after irradiation, Figure 8(a) before irradiation, Figure 8(b) after 5 KGy gamma-ray irradiation and Figure 8(c) after 10 KGy gamma-ray irradiation. It is clear that the increase of the concentration as well as radiation dose enhances the conductivity of the prepared samples as shown in Figure 8. Dielectric constant of (a) un-irradiated samples (b) irradiated samples at 5 kGy (c) irradiated samples at 10 kGy.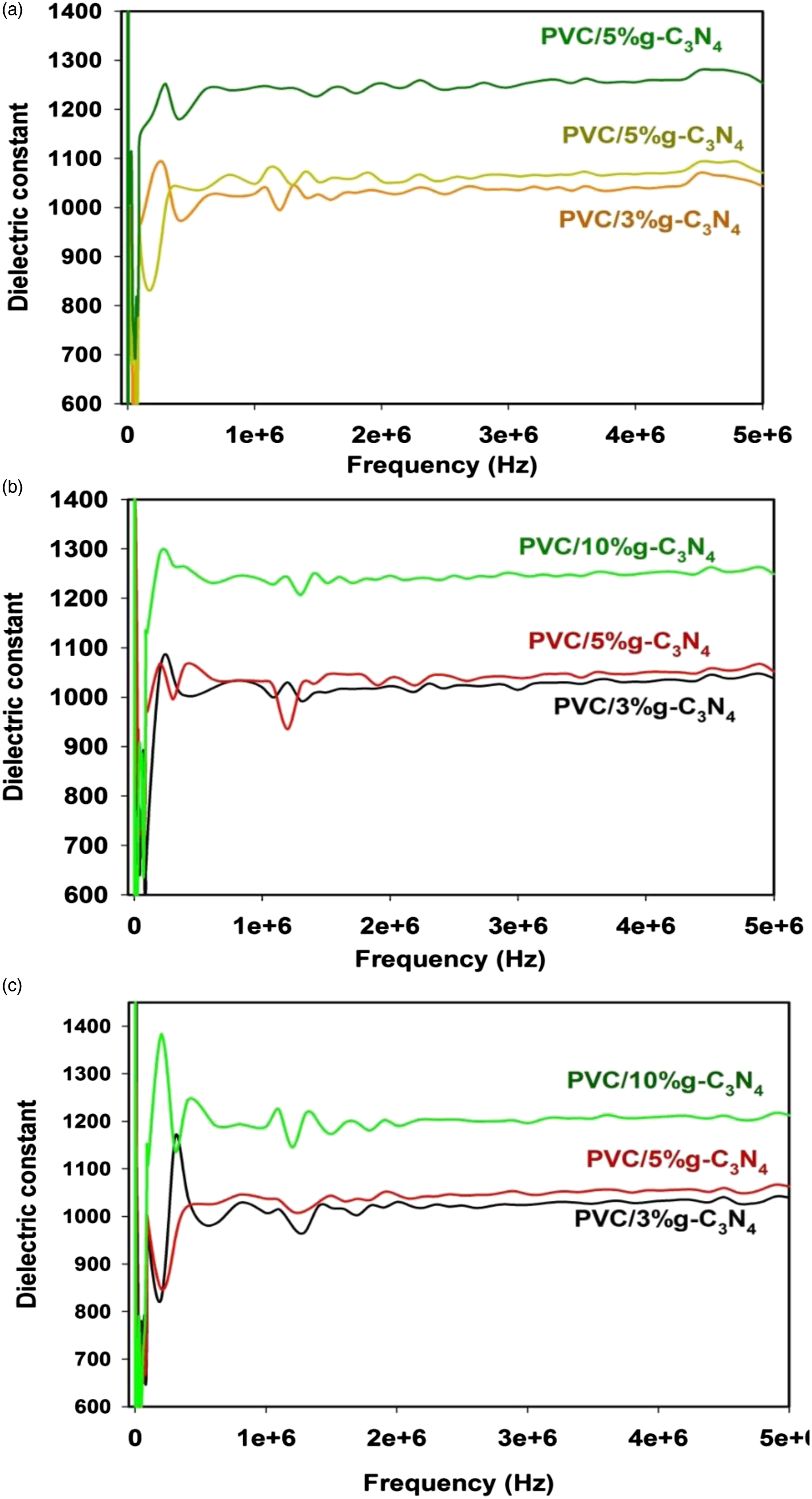
Conclusion
The effect of adding g-C3N4 nanoparticles with different contents and gamma-ray irradiation on the mechanical and electric properties of PVC was investigated. From the obtained data, we can conclude the following: Analysis of XRD spectra showed that the addition of g-C3N4 nanoparticles with different contents and the gamma-ray irradiation affected the lattice parameters of PVC. An analysis of the FTIR results indicated chemical bonding/conjugation between the g-C3N4 nanoparticlesand nylon 66 chains. Subsequent exposure to gamma-ray irradiation causes structural rearrangements in the PVC chains upon the embedding of g-C3N4 nanoparticles. These induced structural changes alter the behavior of these nanocomposites after gamma irradiation. The SEM images showed that the g-C3N4 content was well dispersed in the polymer matrix, thus indicating that the nanocomposites were successfully prepared. The presence and amount of g-C3N4 nanoparticles improved the tensile strength and considerably decreased the elongation of PVC. The flexural strength and flexural models were enhanced by increasing both the g-C3N4 nanoparticle content and the dose of gamma-ray irradiation.
Studying the electrical characteristics, the prepared sample can be considered as a good candidate material for different applications such as radiation dosimeter with varying the capacitance and/or conductance measurable parameters reflects the radiation dose.
Footnotes
Author contributions
All authors (Abdelhameed Sharaf, Mohamed Elhady, Ahmed Abdel-Galil, Shaban Eladl, Ashraf Nasr); ware equal contributions, namely: Conception and design of study, acquisition of data, analysis and/or interpretation of data, Drafting the manuscript, revising the manuscript critically for important intellectual content, Approval of the version of the manuscript to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
