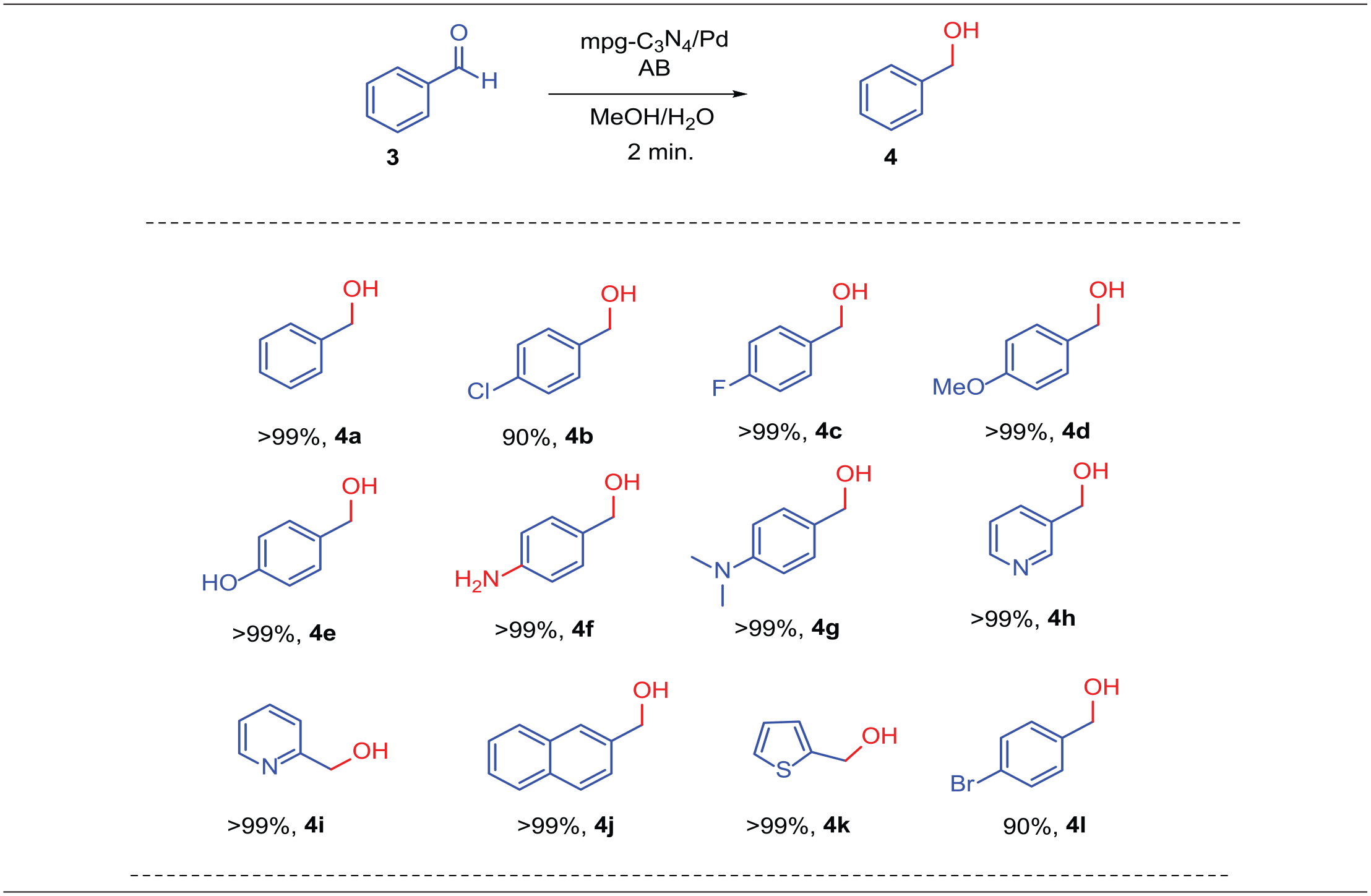
Abstract
A novel transfer hydrogenation methodology for the reduction of ketones (14 examples) and benzaldehyde derivatives (12 examples) to the corresponding alcohols using Pd nanoparticles supported on mesoporous graphitic carbon nitride (mpg-C3N4/Pd) as a reusable catalyst and ammonia borane as a safe hydrogen source in an aqueous solution MeOH/H2O (v/v = 1/1) is described. The catalytic hydrogenation reactions were conducted in a commercially available high-pressure glass tube at room temperature, and the corresponding alcohols were obtained in high yields in 2–5 min. Moreover, the presented transfer hydrogenation protocol shows partial halogen selectivity with bromo-, fluoro-, and chloro-substituted carbonyl analogs. In addition, the present catalyst can be reused up to five times without losing its efficiency, and scaling-up the reaction enables α-methylbenzyl alcohol to be produced in 90% isolated yield.

Introduction
The synthesis of alcohols from ketones and benzaldehyde moieties still attracts great attention for applications in drugs, polymers, pharmaceuticals, fine chemicals, and industrial manufacturing.1,2 An example of the selective hydrogenation of acetophenone (AP) is 1-phenylethanol, which is widely used in the pharmaceutical industry. 3 Several methodologies have been developed for these valuable conversions.4–6 Typical methods for the reduction of carbonyl compounds use hydrides or molecular hydrogen to generate a reducing agent. 7 The transfer hydrogenation (TH) methodology is one of the most effective methods for the synthesis of alcohols. 8 The classical hydrogenation (direct hydrogenation) methods used in industrial and research laboratories usually involve high pressure/temperature conditions in the presence of a noble metal catalyst (Ni, Pd, etc.) with hydrogen gas. 9 Due to their importance, researchers have focused on developing sustainable and environmentally friendly strategies for the synthesis of alcohols. 10
In recent years, the generation of hydrogen in the reaction medium has been used to gain selectivity and safety. For that reason, a range of hydrogen-containing regents, such as isopropanol, 11 glycerol, 12 sodium hydride, 13 hydrazine hydrate, 14 dimethylamine borane, 15 and silanes, 16 have been tested. These methods have some disadvantages, such as the weakness of the hydrogen donor group, toxicity, and the need for high temperature. One of the best hydrogen-containing reagents is ammonia borane (AB). AB is a solid material with a higher hydrogen content compared to other hydrogen donors. Furthermore, it can transfer hydrogen rapidly with the help of transition metals in aqueous solvents. By utilizing AB as a hydrogen reservoir, several efficient and mild conditions have been reported.17–19 In our previous studies, we have presented some protocols using AB as a hydrogen source with reduced graphene oxide–supported bimetallic NiPd alloy nanoparticles (NPs), 20 graphene hydrogel–supported Pd NPs, 21 and CuPt alloy NPs assembled on reduced graphene oxide as a catalyst, respectively. 22 In a recent study, we have also demonstrated that in situ synthesized Pd NPs supported on mesoporous graphitic carbon nitride (mpg-C3N4/Pd) are highly efficient catalysts in the TH of nitroarenes to the corresponding aniline derivatives at room temperature. 23
In order to clarify the efficiency of the newly developed mpg-C3N4/Pd catalyst, we describe herein an efficient, selective, and fast reduction chemistry method for the synthesis of alcohols using AB as a hydrogen source with mpg-C3N4/Pd in a water/methanol mixture (v/v = 1/1). In addition, it should be noted that less AB was used for the hydrogenation reactions compared to our previous studies,20–23 making our new methodology cost efficient and more practical.
Results and discussion
The mpg-C3N4@Pd nanocatalysts were purchased from NANOKAT R&D company, Turkey, and characterized by transmission electron microscopy (TEM), X-ray diffraction (XRD), and Brunauer–Emmett–Teller (BET) surface area analysis. Figure 1 presents the characterization data of the mpg-C3N4@Pd nanocatalysts. As seen in a representative TEM image given in Figure 1(a), Pd NPs with an average particle size of 2.5 nm are dispersed over mpg-C3N4 nanosheets without any agglomeration. The XRD pattern of powdered mpg-C3N4@Pd nanocatalysts (Figure 1(b)) shows five major reflections at 2
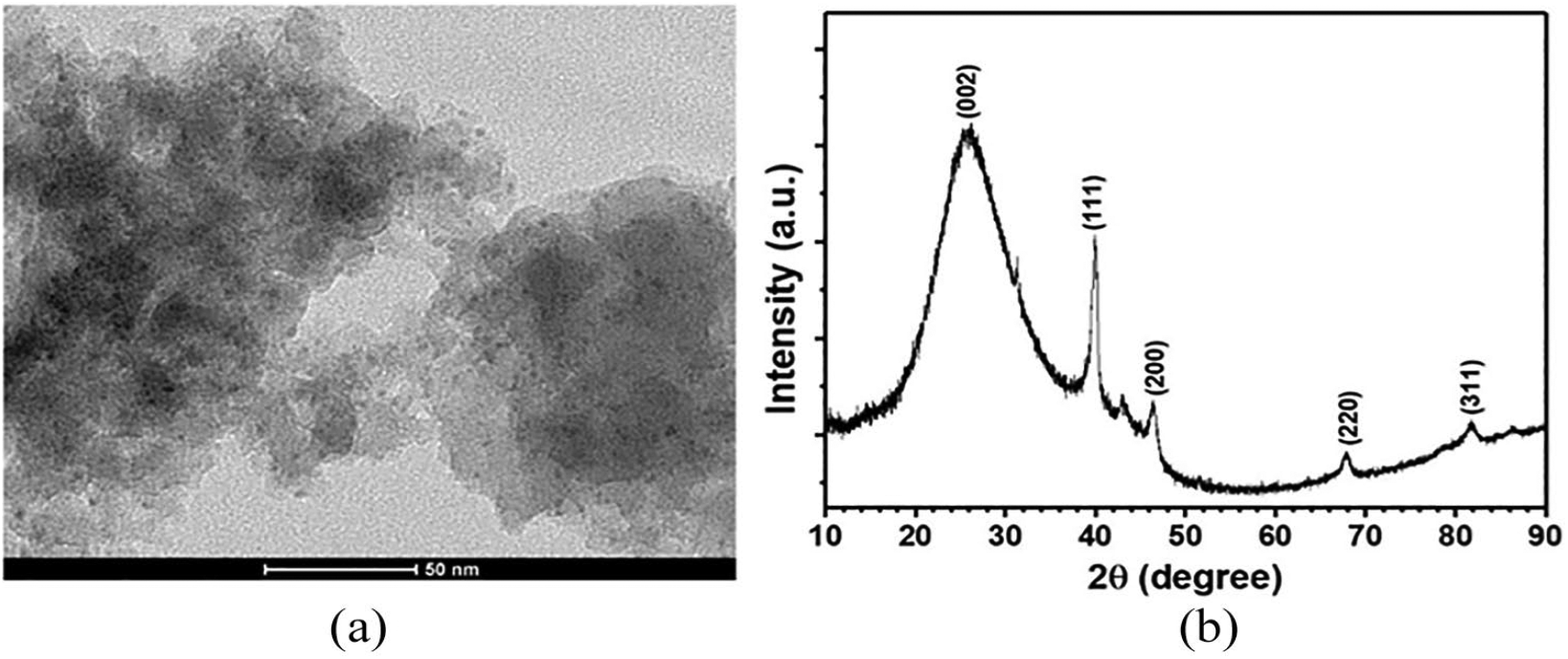
(a) A representative TEM image, and (b) the XRD pattern of mpg-C3N4@Pd nanocatalysts.
Based on our earlier attempts at hydrogenation and reductive amination,20–25 we tried to optimize the hydrogenation conditions by experimenting with different parameters, such as solvent, temperature, time, the catalyst, and the amount of AB. 4′-Methoxyacetophenone was selected as the model reagent for the synthesis of the corresponding alcohol under optimized conditions. The data in Table 1 demonstrate some of the key features of the TH chemistry. Primarily, it was aimed to carry out TH reactions with the smallest amount of the catalyst possible (Table 1, entries 1 and 2). With the use of water as a solvent, a 44% yield was obtained with 2 mg of mpg-C3N4/Pd (Table 1, entry 1), which was increased to 77% with the addition of methanol (Table 1, entry 2). By increasing the amount of the catalyst used, a significant improvement in the yield of the TH reaction in water was observed (Table 1, entry 3), while a quantitative conversion was obtained in the water/methanol mixture (Table 1, entry 4). Reducing the time from 5–3 min resulted in a relatively small decrease in the yield (Table 1, entry 5). Using 1 equiv. (0.35 mmol) of AB provides moderate performance (Table 1, entry 6). Decreasing the amount of methanol led to a moderate yield (Table 1, entry 7). Furthermore, no reaction was achieved in the catalyst-free medium (Table 1, entry 8). Following these optimization experiments, it can be concluded that the best results are obtained using 0.7 mmol of AB as a hydrogen source and 4 mg of mpg-C3N4/Pd as a nanocatalyst with a reaction time of 5 min using 2 mL of methanol/water (1:1) as the solvent (Table 1, entry 4).
Optimization of the conditions for the mpg-C3N4/Pd-catalyzed transfer hydrogenation of 4-methoxyacetophenone (
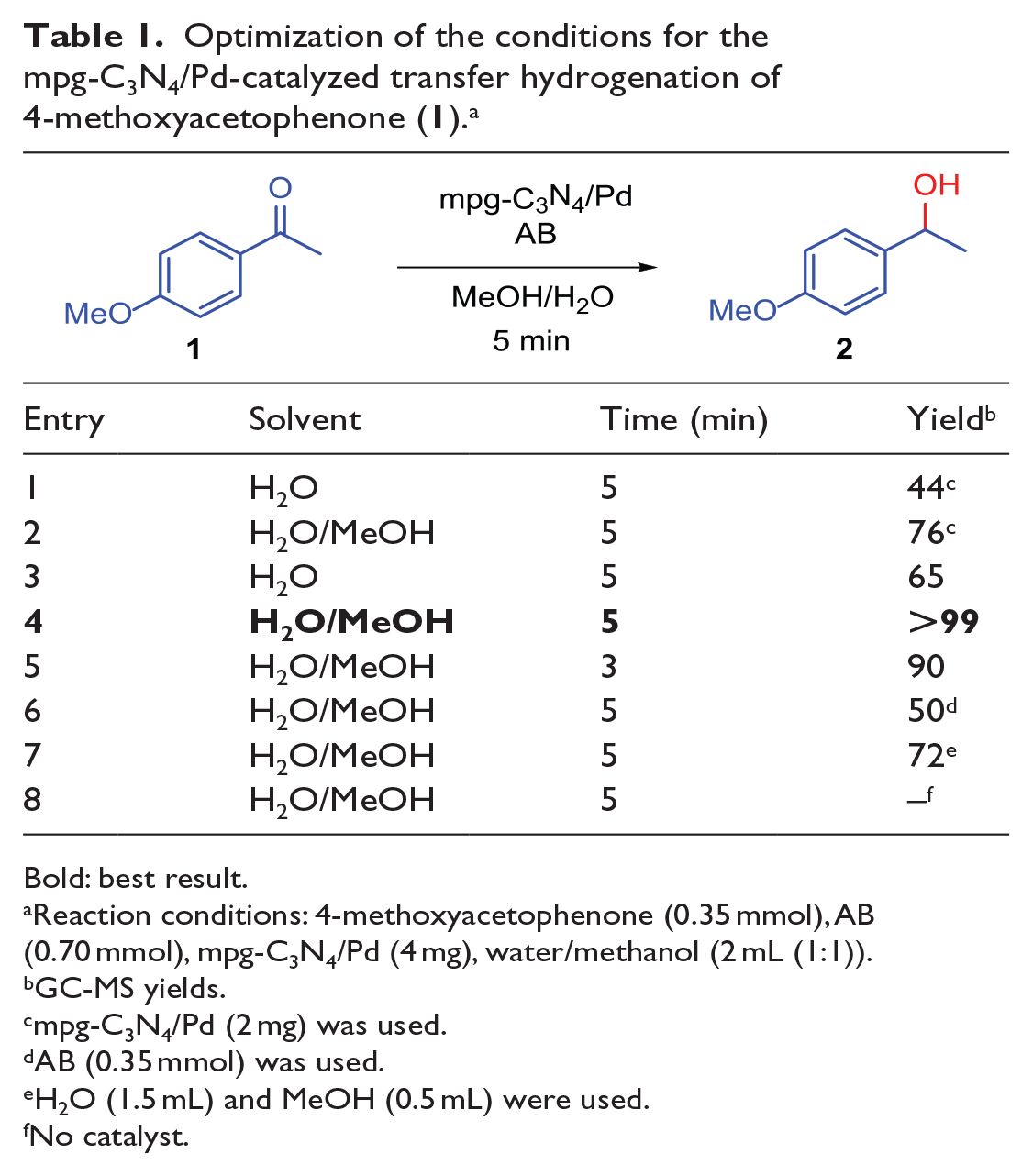
Bold: best result.
Reaction conditions: 4-methoxyacetophenone (0.35 mmol), AB (0.70 mmol), mpg-C3N4/Pd (4 mg), water/methanol (2 mL (1:1)).
GC-MS yields.
mpg-C3N4/Pd (2 mg) was used.
AB (0.35 mmol) was used.
H2O (1.5 mL) and MeOH (0.5 mL) were used.
No catalyst.
Following these optimization studies, TH of different ketones (14 examples) was carried out to test the viability of this fast, selective methodology, and the results are outlined in Table 2. As can be clearly seen from Table 2, all the ketone derivatives used were converted into alcohols in high yields in 5 min at room temperature. Not only electron-donating ketones but electron-withdrawing analogs were also quantitatively converted into the corresponding alcohols. Fluorine-substituted ketones, on the other hand, were found to be reduced selectively without dehalogenation (Table 2,
The substrate scope of the mpg-C3N4-catalyzed TH of ketones. a
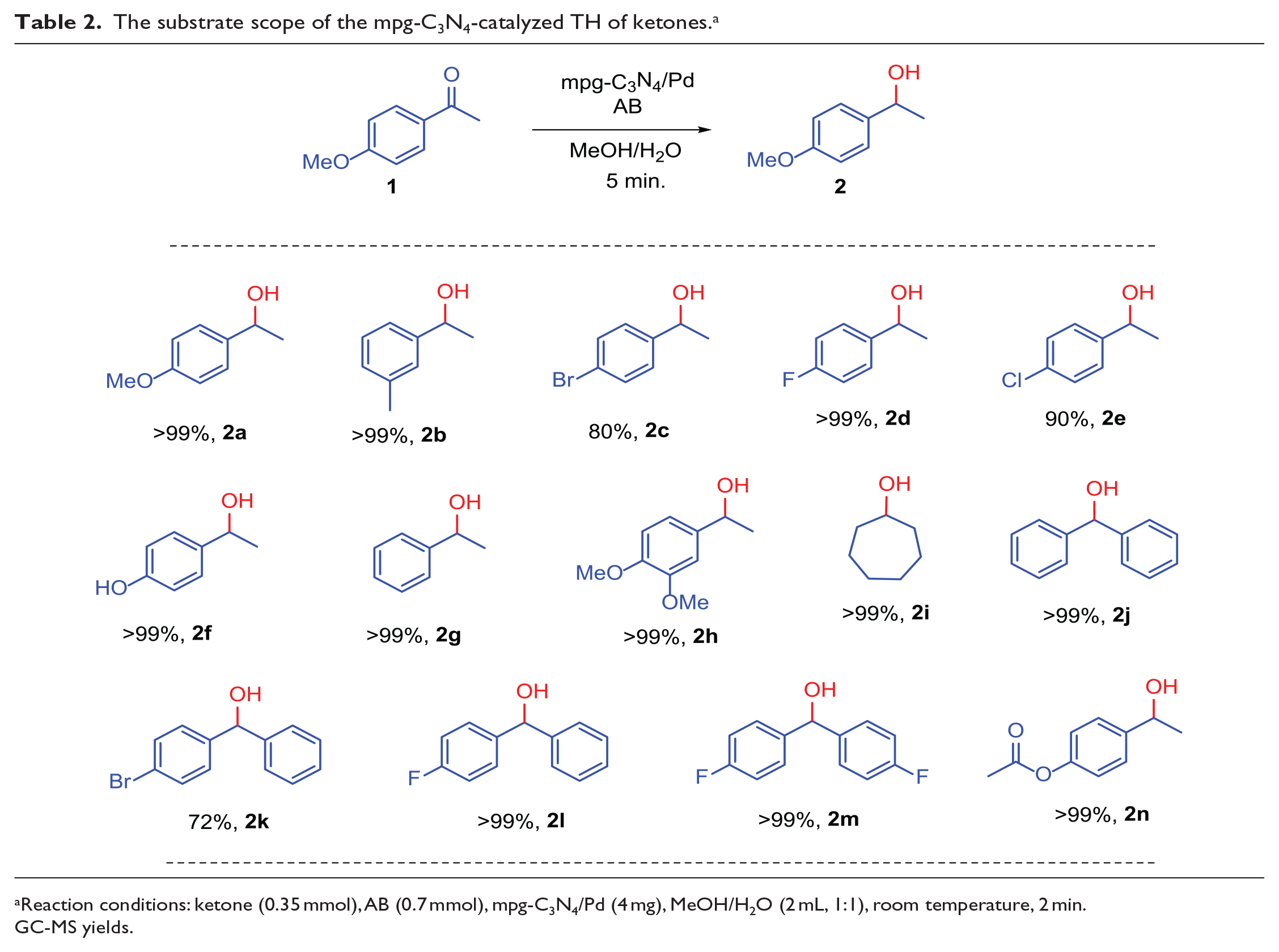
Reaction conditions: ketone (0.35 mmol), AB (0.7 mmol), mpg-C3N4/Pd (4 mg), MeOH/H2O (2 mL, 1:1), room temperature, 2 min.
GC-MS yields.
In the second part of the study, aldehydes were hydrogenated in order to extend the scope of the TH process (Table 3). Many aldehyde derivatives with a variety of substituents (12 examples) were successfully reduced by mpg-C3N4/Pd NPs. On account of the high reactivity of aldehydes, the reactions were complete in only 2 min. As with ketones, fluoro-, bromo-, and chloro-substituted products were synthesized in excellent yields (Table 3,

The TH reaction of 4-iodobenzaldehyde.
Hydrogenation of aldehydes with the commercially available catalyst mpg-C3N4/Pd. a

Reaction conditions: ketone (0.35 mmol), AB (0.7 mmol), mpg-C3N4/Pd (4 mg), MeOH/H2O (2 mL, 1:1), room temperature, 2 min.
GC-MS yields.
Hydrogenation was carried out on a larger scale to verify the practicality of the presented method (Scheme 2). A sample experiment with the ketone derivative acetophenone was performed on a 3.5 mmol scale (10-fold increase), and α-methylbenzyl alcohol (

Synthesis of α-methylbenzyl alcohol via the TH scale-up.
Finally, a recycling experiment, which is one of the most important criteria required to highlight the effectiveness of heterogeneous catalysts, was performed. Scheme 3 shows the recycling experiment of 4-methoxyacetophenone in the presence of the mpg-C3N4/Pd catalyst. In order to make an effective measurement, the first experiment was carried out using mpg-C3N4/Pd (4 mg) over 3 min, during which the conversion was 90%. In subsequent reactions, the reaction time was kept at 3 min to demonstrate the actual impact of the catalyst on the TH. At the end of the sequential experiments, it was found that the catalyst had not lost its effectiveness after five consecutive runs.

The recycling experiment of 4-methoxyacetophenone catalyzed by mpg-C3N4/Pd.
Conclusion
The interest in the development of new, efficient, selective, and rapid catalytic methods in organic chemistry is increasing every day because of stringent environmental policies and financial considerations. In this study, we synthesized the corresponding alcohols from ketones and aldehydes in the presence of mpg-C3N4/Pd as a highly efficient and reusable nanocatalyst. The methodology presented herein has the following advantages over the available alternatives: (1) it is one of the fastest and selective TH methodologies in the literature, (2) it enables the synthesis of bromo-, chloro-, and fluoro-substituted alcohols, (3) the reactions occur in aqueous systems, (4) the amount of AB for the TH reaction is less compared to known studies, (5) the reactions can be easily scaled up, (6) the catalyst is recyclable, and (7) the methodology does not need any purification step. Therefore, we believe that this study will play an important role in the synthesis of alcohols and also other TH applications.
Experimental
Material and general methods
Pd NPs supported on mesoporous graphitic carbon nitride was purchased from NANOKAT R&D, Turkey. Aldehydes, ketones, AB (90%), CDCl3 (99.8%), and D2O (99.9%) were obtained from Sigma-Aldrich and were used without purification. The reactions were followed using thin-layer chromatography (TLC) by utilizing aluminum-backed Merck Silica-Gel 60 F254 plates. Preparative TLC was performed using Merck silica gel 60 HF254+366. GC-MS was carried out using a SHIMADZU GCMS QP2010 system (70 eV electron impact ionization) with an Rxi-5Sil MS Column (RESTEK, 30 meter). The characterization of the organic products was performed using a 400 MHz Bruker NMR instrument.
Typical procedure for the hydrogenation of ketones and aldehydes
mpg-C3N4/Pd (4 mg) and the ketone or aldehyde (0.35 mmol) were suspended in methanol/water mixture (2 mL, 1:1) in a pressure tube. Subsequently, AB (0.75 mmol) was added and the solution was magnetically stirred for 2 (for aldehydes) or 5 min (for ketones) at room temperature. After completion of the reaction, the catalyst was filtered and washed with methanol for further use. The solvent was removed under the reduced pressure. The yield of each alcohol was determined by gas chromatography–mass spectrometry (GC-MS).
All 1H NMR data of the alcohols were in agreement with the reported spectra.5,26–34
1-(4-Methoxyphenyl)ethan-1-ol (
1-(
1-(4-Bromophenyl)ethan-1-ol (
1-(4-Fluorophenyl)ethan-1-ol (
1-(4-Chlorophenyl)ethan-1-ol (
4-(1-Hydroxyethyl)phenol (
1-Phenylethan-1-ol (
1-(3,4-Dimethoxyphenyl)ethan-1-ol (
Cycloheptanol (
Diphenylmethanol (
(4-Bromophenyl)(phenyl)methanol (
(4-Fluorophenyl)(phenyl)methanol (
bis(4-Fluorophenyl)methanol (
4-(1-Hydroxyethyl)phenyl acetate (
Phenylmethanol (
(4-Chlorophenyl)methanol (
(4-Fluorophenyl)methanol (
(4-Methoxyphenyl)methanol (
4-(Hydroxymethyl)phenol (
(4-Aminophenyl)methanol (
(4-(Dimethylamino)phenyl)methanol (
Pyridine-3-ylmethanol (
Pyridine-2-ylmethanol (
Naphthalen-2-ylmethanol (
Thiophen-2-ylmethanol (
(4-Bromophenyl)methanol (
Recycling experiments
Mpg-C3N4/Pd (4 mg) was suspended in water/methanol (2 mL, 1:1), and 4-methoxyacetophenone (0.35 mmol) was added to a pressure tube. Subsequently, AB (0.75 mmol) was added and the solution was stirred for 3 min at room temperature. Following completion of the experiment, the catalyst was filtered and washed with methanol for further use. Subsequently, a new experiment was established with the recovered catalyst. This TH chemistry was repeated five times. The yields were acquired by GC-MS.
Supplemental Material
Supporting_Information_JCR – Supplemental material for A facile and highly efficient transfer hydrogenation of ketones and aldehydes catalyzed by palladium nanoparticles supported on mesoporous graphitic carbon nitride
Supplemental material, Supporting_Information_JCR for A facile and highly efficient transfer hydrogenation of ketones and aldehydes catalyzed by palladium nanoparticles supported on mesoporous graphitic carbon nitride by Bilal Nişancı and Ziya Dağalan in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We are grateful to Ataturk University BAP (Project No. FBA-2018-6827) for their financial support.
Supplemental material
The spectral characterization of the compounds can be found in the Supplemental material for this article is available online.