Abstract
The catalysts utilized for oxidative desulfurization have acquired significant attention and ability to improve the quality of the fuel oil by removing sulfur. In this work, the catalysts used for oxidative desulfurization include CoWO4 and Bi2WO6 with graphitic carbon nitride (g-C3N4) as support were synthesized by the one-pot hydrothermal method. Graphitic carbon nitride was obtained by thermal polycondensation of melamine at 550°C for 5 h. These catalysts were homogeneously dispersed on the surface of the support and their structure, morphology, and properties were determined by different characterization techniques (Powder X-Ray Diffractometer, Fourier Transform Infrared Spectroscopy, Scanning Electron Microscopy and Energy Dispersive X-Ray Spectroscopy, Specific Surface Area (Brunauer, Emmett and Teller (SBET)). The parameters that affect the efficiency of the desulfurization process such as catalyst amount, amount of oxidizing agent, and reaction temperature have been optimized thoroughly. The oxidative desulfurization reaction was studied in terms of kinetics which shows that reaction is pseudo first order. The thermodynamic studies revealed that the reaction is endothermic and spontaneous in nature. The results determined that the catalytic efficiency for the removal of sulfur (as dibenzothiophene) is more than 90% in the presence of support (g-C3N4) to obtain sulfur free fuel.
Introduction
Raw oil is the world’s largest source of energy that accounts for nearly half of the globe’s total energy resource and its requirements have risen worldwide day by day. Crude oil is mostly made up of straight-chain hydrocarbons, aromatics, and compounds with a heteroatom. The combustion of Sulphur containing compounds that include, benzothiophenes (BT), dibenzothiophenes (DBT) thiophenes (T), and mercaptans produces sulfur oxides and other harmful gases which cause air pollution (such as smog and photochemical haze), soil pollution in form of acid rain and disturb the ecosystem.1,2 The elimination of sulfides is essential for the manufacturing of pollution-free fuel and to meet the requirement of the United States Environmental Protection Agency which has standard criteria of sulfur content (<10 mg/L).3,4 The sulfides are classified into two main categories in fuel: Sulfur, hydrogen sulfide, and mercaptan are categorized as active sulfides whereas thioethers, disulfides, benzothiophenes, and dibenzothiophenes are examples of inactive sulfides. Due to their least complicated structures, thiol, sulfides, and disulfides could be easily removed during the desulfurization process. However, it is hard to eliminate thiophene, benzothiophenes, dibenzothiophene, and its derivatives as they have aromatic and sterically hindered structures. So, the removal of thiophene derivative molecules is considered to be the key goal in desulfurization. 5
Recently, several methods for eradicating sulfur from fossil fuels have been investigated which include oxidative desulfurization (ODS), 6 adsorptive desulfurization, 7 extractive desulfurization 8 and hydrodesulfurization techniques. 9 Hydrodesulfurization (HDS) in combination with carbon rejection technologies, such as coking and fluid catalytic cracking, are the main technologies industrially employed for the desulfurization of heavy oil. 10 HDS is expensive method for the removal of sulfur-containing compounds due to the requirements of high temperature (>600 K) and pressure (>3 MPa). This technique is also not effective for the removal of alkylated organic sulfur compounds and polyaromatic sulfur compounds such as BT and DBT due to steric hindrance and low reactivity of these compounds in hydrogenation treatments.11,12 Comparison with other techniques, ODS has attained more consideration and is supposed to be the most promising owing to cost effective and no high temperature and pressure requirements. Sulfides and other sulfur-containing compounds are oxidized to sulfones and sulfoxides by using a catalyst in ODS and then extracted from the fuel.13,14 In ODS, the choice of oxidant and catalyst is critical. The most promising oxidizing agent include hydrogen peroxide owing to amount of active oxygen increase by 47% by mass unit and also no harmful side product formation. The heterogeneous catalyst is used along with hydrogen peroxide to increase the mass transfer which makes this reaction fast and commercial.
In order to achieve deep desulfurization, the catalyst is essential in the ODS. Organic acids, heteropoly acids, ionic liquids (ILs) and tungstate-based catalysts have been reported for oxidation desulfurization process.15–18 Although homogeneous catalysts work well for the desulfurization of model fuel oil, a portion of the catalyst is lost during the reaction as separation and recovering after the reaction is challenging that results in a greater cost. 19 Heterogeneous catalysts, on the other hand, have a number of benefits that help them overcome these constraints, including the ease with which they can be separated, recovered, and circulated, as well as the fact that they are environmentally beneficial. 20 As a result, heterogeneous catalysts are commonly utilized in ODS. Tungsten oxides-based metal catalysts have significant importance due to easy synthesis, low toxicity, cheap cost, and consistent multifunctional performance. 21 The active components are frequently put on supports to improve the activity and separation of the catalysts. Silica, alumina, activated carbon, zeolite, carbon nitride (g-C3N4), and other supports have all been studied.22–25 Boron nitride was also used as a metal-free catalyst in oxidative desulfurization of fuel with H2O2 as the oxidant. It was postulated that the boron radicals activate the H2O2 is which results in ultra-deep desulfurization. 26 As a carrier, g-C3N4 is usually modified with metals inorganic carbon materials, resulting the formation of heterojunctions which increase its electron conduction and catalytic activity. 27 The use of g-C3N4 as support gets significant importance because of the larger surface area provided by this support. 28
Regarding the above remarks, the present study comprises the synthesis of two novel nanocomposites (Bi2WO6@g-C3N4 and CoWO4@g-C3N4) via hydrothermal method and study its performance for the first time in oxidative desulfurization of model fuel oil. The prepared materials were characterized via Fourier Transform Infrared Spectroscopy (FT-IR), Powder X-Ray Diffractometer (P-XRD), Scanning Electron Microscopy (SEM) and Energy Dispersive X-Ray Spectroscopy (EDX). Optimization, kinetics and thermodynamics parameters were discussed in detail.
Materials and method
Materials
All the chemicals were used of analytical grade; melamine (C3H6N6, 99%, MW= 126.12 g/mol) and sodium tungstate dihydrate (Na2WO4 ·2H2O; 98% MW = 329.85 g/mol) were purchased from DAEJUNG, Korea, Cobalt (II) Chloride hexahydrate (Cl2CoH12O6, 99%, MW = 237.93 g/mol) and Bismuth (III) nitrate pentahydrate (Bi (NO3)3 ·5H2O, 99% MW = 485.07 g/mol) were purchased from Sigma -Aldrich.
Synthesis of g-C3N4, CoWO4@g-C3N4 and Bi2WO6@g-C3N4
Graphitic carbon nitride was synthesized by thermal polycondensation of melamine at 550°C for 5 h. The resultant yellow product was collected and milled into powder for further use as support as well as a catalyst in our reaction 29 The synthesis of CoWO4@g-C3N4 was conducted by mixing sodium tungstate dihydrate (Na2WO4 ·2H2O, 1.0 g) with cobalt (II) chloride hexahydrate (Cl2CoH12O6, 1.5 g) in 100 mL distilled water. Similarly, for the preparation of Bi2WO6@g-C3N4; sodium tungstate dihydrate (Na2WO4 ·2H2O, 1.0 g) was mixed with bismuth (III) nitrate pentahydrate (Bi (NO3)3 ·5H2O, 2.91 g), in 100 mL of distilled water. Then stirred for 15 min for complete mixing followed by sonication for 20 min to obtain the uniform size particles. Afterward, a few drops of hydrazine were added for reduction. This solution was kept in the autoclave at 150°C for 6 h. The product (CoWO4 and Bi2WO6) was then cooled, filtered, and ground. Consequently, CoWO4 or Bi2WO6 (1g each) and g-C3N4 (4g) were taken in 100 mL of distilled water, stirred for 5 min and sonicated for 20 min for uniformly dispersion of particles. It was kept in the autoclave at 150°C for 12 h, filtered and ground via pestle mortar.
Catalytic activity tests and product analysis
25 mL of DBT solution (50–500 mg/L in n-hexane) with acetonitrile (25 mL) was taken in two necks round bottom flask. The ODS procedure was performed by adding catalyst along with oxidizing agent (H2O2) and stirring it at different time intervals. After the equilibrium was reached, the lower layer containing n-hexane with DBT was analyzed by using a UV spectrophotometer and calculated sulfur concentration in fuel oil as DBT shows an absorption (λmax) at 284 nm. 30 The formation of sulfones in acetonitrile was checked by thin-layer chromatography. The formation of sulfones was also confirmed by the melting point of the product obtained (229–232°C).
Results and discussion
Characterization
Fourier transform infrared spectroscopy
The Infrared spectroscopic analysis was performed to confirm the presence of specific functional groups via IR-TRACER-100 in the range of 4000–400 cm−1 with scan rate of 15 cm−1. The g-C3N4 has strong absorption peaks in the range of 1200–1650 cm−1, which could be attributed to the C=N vibration at (1637 cm−1) in the aromatic ring and, the stretching vibration of C–N at (1237,1317, and 1406 cm−1). Moreover, the bending vibration of C–N in the heterocyclic triazine ring system at (810 cm−1) is also noticed. The broad absorption band at 3156 cm−1, can be attributed to the stretching mode of the N−H bond as shown in Figure 1(a).31,32 The pure Bi2WO6 sample demonstrates main absorption bands at 400−800 cm−1, which are ascribed to Bi−O, W−O stretching and W−O−W bridging stretching modes (Figure 1(b)). These peaks are still present in Bi2WO6@C3N4, also suggesting that no structure change of Bi2WO6 appears during the process, which is consistent with the XRD result. Furthermore, all characteristic absorption bands of C3N4 and Bi2WO6 appeared in the spectra of Bi2WO6@g-C3N4, showing the coexistence of catalyst with support as shown in Figure 1(c). The peaks at 800 and 626 cm−1 are the characteristic peaks of CoWO4.
33
The CoWO4 with g-C3N4 support contains all the vibration peaks of both pristine g-C3N4 and pure CoWO4. Hence, we concluded that there were no structural changes in the g-C3N4 during the introduction of the CoWO4. The FT-IR spectra contain the characteristic peaks of CoWO4 (810 and 626 cm−1) and g-C3N4 (1240, 1320, 1403, and 1637 cm−1) in CoWO4@g-C3N4 (Figure 1(d) and (e)). Fourier transform infrared spectroscopy of g-C3N4 (a), Bi2WO6 (b), Bi2WO6@g-C3N4 (c), CoWO4 (d) and CoWO4@g-C3N4 (e).
X-ray diffraction
Powder X-ray diffraction (P-XRD) using Cu-Kα radiation detector having wavelength 0.154 nm with a scanning rate of 2°/min in the range of 2θ from 20–70° by a powder diffractometer and was confirmed by Joint Committee on Powder Diffraction Standards (JCPDS) cards. The crystallite sizes were measured with the help of Sherer’s equation (equation (1)) using the full-width half maximum (FWHM) of the sharpest peak. Powder X-ray diffractometer pattern of g-C3N4 and composites.
Scanning electron microscopy
Scanning microscope scans a centered beam of the electron over a surface to form an image in which the electrons within the beam act with the sample, manufacturing numerous signals that may be used to acquire data regarding the surface topography and composition. The signals which were derived from electron-sample interactions revealed information relating to the sample along with external morphology (texture), chemical composition, crystalline structure, and orientation of materials making up the sample. SEM (NOVA Nano) images show the uniform distribution of the Bi2WO6 in the form of nanoflakes over the g-C3N4 support. The nanoflakes have a very large surface area and are densely interconnected with each other without any small gap as shown in Figure 3(b). These nanoflakes have shown rough morphology with agglomerates of small particles.34,35 SEM images at different resolutions show that CoWO4 nanoparticles were uniformly distributed on the g-C3N4 support.
36
In less resolution, the particles have given the rock-like structure. While in the case of high resolution the particles have shown cauliflower-like morphology with high surface area and more porosity as shown in Figure 3(c). This gives the sites for catalytic activity with good results. Additionally, SEM images of g-C3N4 show the coral-reef type morphology with high surface area to provide the site for catalytic activity as shown in Figure 3(a). Scanning micrographs of g-C3N4 (a), Bi2WO6@g-C3N4 (b) and CoWO4@g-C3N4 (c).
Energy-dispersive X-ray spectroscopy
The elemental composition of the prepared catalyst is being justified by performing EDX analysis, where a beam of X-ray is being interacted with the sample surface. As a result, a unique set of peaks is generated depending upon the characteristic composition of each element. The prepared catalysts have a combination of various elements that gave their characteristic peak. According to the EDX micrograph of Bi2WO6@g-C3N4, the successful preparation of nanocatalyst is confirmed by the existence of Bi, W, C, and N elements with an approximated percentage quantity of 15.03, 6.72, 37.01, 41.24 wt. %, respectively as is evident from Figure 4. Additionally, the EDX micrograph of CoWO4@g-C3N4 shows the existence of Co, W, C, and N elements with an approximated quantity of 3.39, 12.07, 52.59, 31.95 wt. %, respectively as illustrated in Figure 4. Comparatively, the EDX of g-C3N4 only shows two peaks of C and N with 34.85 and 65.15 wt. % respectively. Energy dispersive X-ray spectroscopy spectrums of g-C3N4, Bi2WO6@g-C3N4 and CoWO4@g-C3N4.
Specific surface area analysis
The surface area analysis (Brunauer, Emmett and Teller) was performed from the nitrogen adsorption isotherms at 77 K with a Micromeritics Gemini VII instrument. The nitrogen adsorption-desorption curves of the catalysts are displayed in Figure 5. The Brunauer, Emmett and Teller (BET) surface areas of synthesized g-C3N4, Bi2WO6@g-C3N4 and CoWO4@g-C3N4 catalysts were found to be 19.8, 31.5, 35.8 m2/g respectively. The specific surface areas of the g-C3N4 increased after composites formation with Bi2WO6 and CoWO4. This finding may be related to the catalyst being well dispersed on the g-C3N4 nanosheets, entering the interlayer, and interacting with triazine units of the nanosheets, resulting in expansion of the interlayer, which further leads to an increase of the surface areas of the composites. These finding are in similar to the previously reported studies.37,38 The nitrogen adsorption-desorption curve of the synthesized g-C3N4, Bi2WO6@g-C3N4 and CoWO4@g-C3N4 catalysts.
Catalytic oxidative desulfurization studies
The graph (Figure 6(a) and (b)) exhibited the catalytic activity of catalysts with and without support and it was observed that the catalytic activity enhanced prominently when support was added because of increased surface area and by using support catalyst is well dispersed on the support and provides more active sites for the reaction. Bi2WO6 and CoWO4 oxidized the sulfur-containing compounds up to 41.17% and 48.22% respectively without support, while using the support of g-C3N4 the percentage oxidation of sulfur compounds increased up to more than 90% and 80% respectively. Comparative studies with or without support via Bi2WO6@g-C3N4 (a) and CoWO4@g-C3N4 (b), Optimization for oxidative desulfurization process: Effect of time (Bi2WO6@g-C3N4 (c) and CoWO4@g-C3N4 (d)), Effect of catalyst loading (Bi2WO6@g-C3N4 (e) and CoWO4@g-C3N4 (f)), Effect of temperature (Bi2WO6@g-C3N4 (g) and CoWO4@g-C3N4 (h)) and Effect of oxidant (Bi2WO6@g-C3N4 (i) and CoWO4@g-C3N4 (j)).
The conversion of sulfur-containing compounds from sulfides to sulfoxides and sulfones increased with the passage of time. All sulfur-containing compounds are oxidized within 2 h of time duration. The CoWO4@g-C3N4 removes the sulfur up to 84.7% and Bi2WO6 @ g-C3N4 removes the sulfur-containing compound up to 91.3% as shown by Figure 6(c) and (d). The same trend was observed for Fe2W18Fe4@NiO@CTS hybrid nanocatalyst and polysilicotungstate supported β-cyclodextrin composite reported previously.1,39
The catalyst loading has a crucial impact on the removal of sulfur-containing compounds from the fuel oil. With the increase of catalyst amount from 0.02 g to 0.06 g, the percentage oxidation of sulfur also increased. The optimum amount of catalyst was taken as 0.06 g for better catalytic results as with the increase of catalyst amount, more active sites are available for the catalytic activity to convert sulfur-containing compounds into sulfones as shown in Figure 6(e) and (f). The same trend is observed in the case of Fe2W18Fe4@NiO@CTS hybrid nano catalyst and polysilicotungstate supported β-cyclodextrin composite.1,39
The oxidation of sulfur compounds in fuel oil has been increased by the increase of temperature (up to 60°C). The optimum temperature for the best ODS result was taken as 60°C and by increasing temperature to a certain limit catalytic activity has increased. At high temperatures, the ODS process decreases because at high temperatures decomposition of H2O2 starts and this will reduce the oxidative conversion of sulfides into sulfones as displayed in Figure 6(g) and (h). Effect of temperature on percentage oxidation was evaluated for WO3/mesoporous ZrO2, Fe2W18Fe4@NiO@CTS hybrid Nano catalyst show the comparable result with CoWO4@g-C3N4 and Bi2WO6@g-C3N4. 40 The same trend was observed as in the case of CoWO4@g-C3N4 and Bi2WO6@g-C3N4 which shows that the percentage oxidation of sulfur increases when we increase temperature up to 60°C.
Many types of oxidants have been used and for ODS process. The oxidants such as peroxyacids (performic acid, pertrifluoroacetic acid, mixture of formic acid or trifluoroacetic acid, tert-butyl hydroperoxide, and hydrogen peroxide) reported to be the most efficient oxidants38,41. In the current study, H2O2 is used due to its strong oxidizing potential, cheap and easy availability. The increase of oxidizing agents enhanced the ODS process itself. Although by increasing H2O2 (as an oxidizing agent), the ODS increases as represented by Figure 6(i) and (j) but H2O2 can be used up to a specific limit. The optimum amount of hydrogen peroxide used for ODS in presence of given catalysts is 0.6 mL, as higher amount of H2O2 can cause self-decomposition that would not be effective for the oxidation process. These results are comparable with some other catalysts such as [P2W18O62]6- and [P5W30O110].14,1
Oxidative desulfurization mechanism
In the presence of metal-based nanocomposites and hydrogen peroxide as an efficient oxidizing agent, ODS process has been done by oxidizing a sulfur atom in DBT to DBT-sulfone, which was aided by Bi2WO6 and CoWO4 catalysts over g-C3N4 as support (Figure 7). A simplified chemical process implies that oxygen radicals from hydrogen peroxide (H2O2) attack the electron-rich sulfur atom of DBT in succession, resulting in the formation of a DBTO2 molecule. According to the literature and our experimental data, the oxidation of DBT to the corresponding sulfone may be explained by the process. The following is the suggested ODS process for sulfur-containing compounds catalyzed by the heterogeneous catalysts Bi2WO6@g-C3N4 and CoWO4@g-C3N4. The active peroxo intermediate species are generated when the oxidizing agent reacts with the metal oxide catalyst in a reversible process. The nucleophilic attack of peroxo species on a sulfur atom changes the aromatic sulfur compound (DBT) in the form of sulfides into the corresponding sulfoxide (DBTO). In the next step, another active peroxo species oxidizes the sulfoxide (DBTO) to the correspondent sulfone (DBTO2). The inclusion of the tungsten aids in the donation of electron density to peroxo species and provides the metal atoms bismuth and cobalt a more nucleophilic nature. The reaction is carried out in a two-phase system in this study. The oxidation and extraction phases are carried out simultaneously in this desulfurization process. The extraction process is quick, and the oxidation process is the rate-determining slow step, according to the findings. Both the oxidation of DBT to its sulfone (DBTO2) and the extraction of the oxidized DBT take place at the same time. The oxidation of dibenzothiophene takes place at the interface between the oil phase and the extraction phase (acetonitrile). Because of their polarity, the reaction products aggregate in the polar phase, which is advantageous for extraction using an acetonitrile solvent.3,4,39,42,43 Oxidative desulfurization mechanism for model fuel oil.
The kinetic studies for oxidative desulfurization
The rate of reaction can be determined by the reaction kinetics. The following equation (equation (2)) is used to calculate the rate constant of the reaction.
The reaction mixture has two layers; the upper oily layer contains DBT along with n-hexane and the lower layer consists of the catalyst with an oxidant. The results revealed that the oxidative desulfurization follows first-order kinetics, and a straight line is obtained between ln C0/C versus time of reaction (t) at 60°C as shown in Figure 8(a) and (b) and Table 1. The trend shows a better result as compared to other metal-based catalysts such as PMoCu@MgCu2O4--PVA and Fe2W18Fe4@NiO@CTS composites.30,39 Pseudo first order kinetics of Bi2WO6@g-C3N4 (a) and CoWO4@g-C3N4 (b) and Thermodynamics of Bi2WO6@g-C3N4 (c) and CoWO4@g-C3N4 (d). Kinetics and thermodynamic parameters for desulfurization of dibenzothiophenes at (333 K).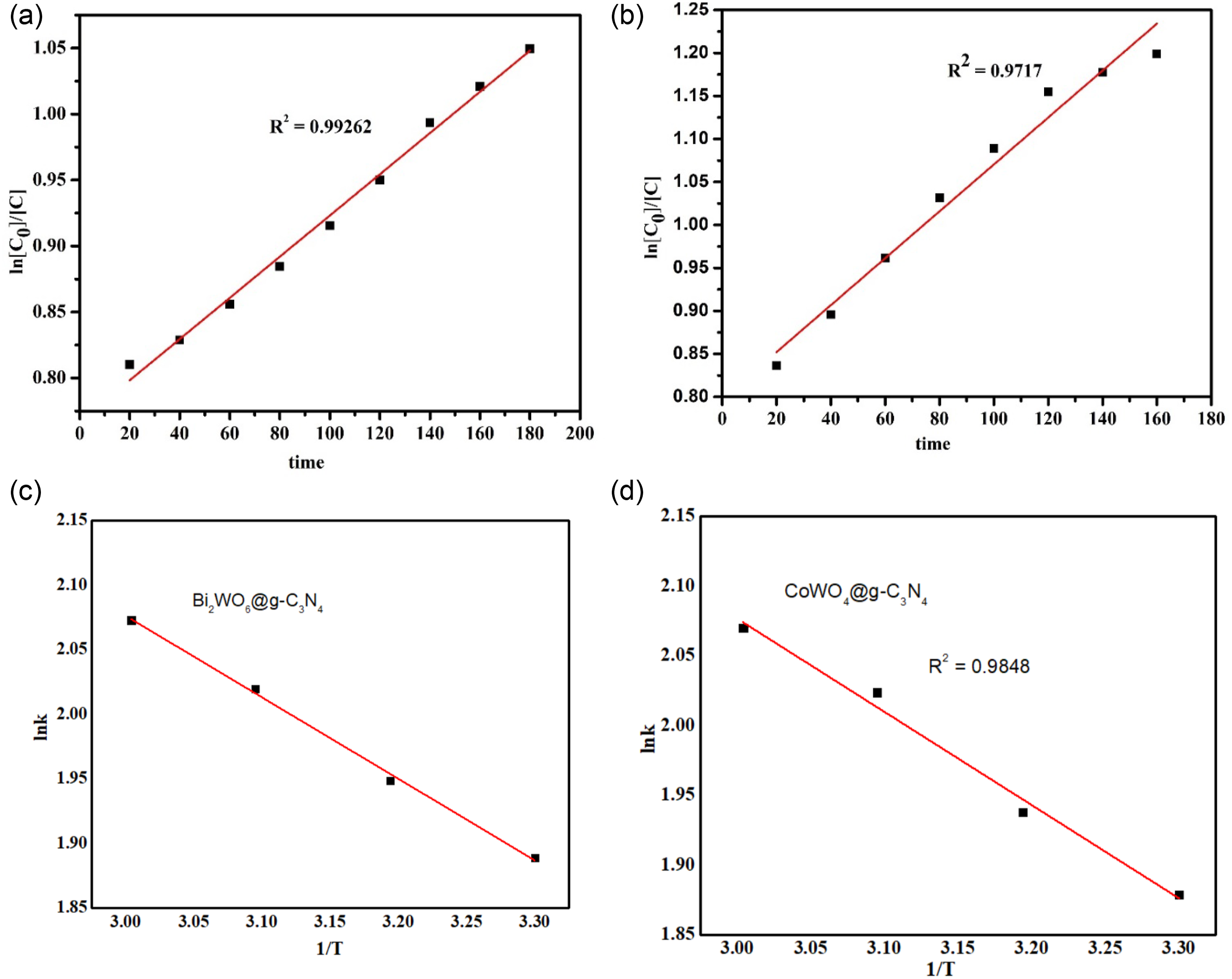

Thermodynamics studies
The Eyring equation used to calculate the basic thermodynamic of the reaction system
44

This equation is obtained by comparing the following equations
45


The reaction aspects can be determined by thermodynamic parameters that give information about Gibbs free energy (ΔG), entropy (ΔS), and enthalpy (ΔH) of reaction. The negative value of Gibbs free energy shows that the reaction is spontaneous and thermodynamically feasible, and it can proceed quickly with good desulfurization results as it is evident from Table 1 and Figure 7(c) and (d). The positive value of enthalpy represents that the reaction is endothermic. The positive value of entropy shows that the thermodynamic feasibility of reaction is due to the increase of entropy, which represents the increase of disorder of the system.
Comparative study
Comparative study of different metal-based catalysts.

DBT: dibenzothiophenes.
Conclusions
CoWO4@g-C3N4 and Bi2WO6@g-C3N4 were successfully synthesized by hydrothermal method and first time used for the oxidation of sulfur containing compounds in fuel oil. The catalysts exhibited the outstanding performance for the oxidation of DBT under moderate reaction conditions. The excellent results of catalysts for catalytic activity were attributed to g-C3N4 as support which enhanced porosity, surface area, crystallinity, and active sites. The removal rate of DBT from oil enhanced up to 90% by optimization of reaction parameters which include 0.06 g catalyst dose, 0.6 mL hydrogen peroxide as oxidant, reaction temperature 60°C and 2 h of reaction time. The kinetics studies revealed that the reaction was pseudo-first order with apparent rate constant of 0.00345 min−1 (R2 = 0.9926) and 0.00621 min−1 (R2 = 0.9717) for Bi2WO6@g-C3N4 and CoWO4@g-C3N4 respectively, while the thermodynamics studies determined that the reaction is endothermic with apparent enthalpy values of 1.74 kJ/mole and 1.849 kJ/mole for Bi2WO6@g-C3N4 and CoWO4@g-C3N4 respectively. The reaction was spontaneous with higher feasibility with value of Gibbs free energy values of −5.20 kJ/mole and −8.313 kJ/mole for Bi2WO6@g-C3N4 and CoWO4@g-C3N4 respectively. This work demonstrated a very good catalytic activity in ODS system for improving the quality of liquid fuel.
Footnotes
Acknowledgements
The authors are grateful to Pakistan Science Foundation for financial assistance under project no. (PSF-NSFC-IV/Chem/C-QAU (27).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by Pakistan Science Foundation (PSF-NSFC-IV/Chem/C-QAU (27).


