Abstract
Over the years, various types of techniques have been used for the synthesis of nanocomposites. In this work, melamine-based polyamide (PA) was synthesized using a one-pot polycondensation method under mild conditions. carboxyl graphene (CG)/PA nanocomposites (CGMPA) were prepared by CG nanofiller loadings of 1, 3, and 5 wt.% via delamination/adsorption approach. The prepared CGMPA nanocomposites were characterized using different analyses, such as Fourier transform infrared techniques (FTIR), field emission scanning electron microscopy (FESEM), X-ray diffraction (XRD), differential scanning calorimetric (DSC), and thermogravimetric analysis (TGA). The effects of the CG on the thermal properties of the CGMPA nanocomposites were significant. The results showed that the melting temperature (Tm) of neat PA and CGMPA were increased from 378°C to 393°C suggested better dispersion of CG in PA matrix. The decomposition temperature of PA was increased from 451°C to 463°C in CGMPA nanocomposites indicates the better thermal stability of PA matrix by addition of CG.
Introduction
Graphene is a two-dimensional (2D) substance made of structured carbon atoms as hexagonal honeycomb in lattice with impressive mechanical, thermal, electrical and barrier properties. It has shown considerable attention since its discovery in 2004 by KS Novoselov and AK Geim.1,2 One of the most desirable application of graphene is in the preparation of graphene-based polyamide nanocomposite (GPA) materials. 3 In polyamides (PAs), graphene has been considered as an excellent reinforcing nanofiller with novel and improved properties. 4
PAs are widely used important class of engineering thermoplastic material with remarkable properties and characteristics. 5 However, poor dimensional stability, low electrical conductivity, and low thermal stability limit the use of PA in the high-performance applications. 6 At very low filler content, graphene has greatly improved the physical properties of PA, making it highly stimulating in the preparation of high-performance materials. 7 As a result, various experiments have been performed in order to enhance the properties of PA using graphene nanofiller. The GPA nanocomposites have been prepared by several methods, including melt blending, solvent mixing, in situ polymerization, electrospinning, and others, etc. The key challenges in the preparation of GPA nanocomposites are uniform graphene dispersion and strong interfacial interactions between graphene and PA.8,9
Many studies have clearly shown that the even dispersion of graphene in composites depends on synthesis methods. 10 The most common methods for preparing GPA nanocomposites are in situ polymerization, solvent mixing and melt blending. The complex processes and long treatment time of in situ polymerization method affects the large-scale production of GPA nanocomposites. The in situ polymerization is not suitable for the large-scale production of graphene reinforced nanocomposites. 11 Dixon et al. prepared graphene oxide based PA6 nanocomposites and showed the effects of GO on PA6 properties. The addition of GO also influenced the crystallinity and reduced the average molecular weight of PA6. 12 O’Neill et al. reported thermally stable multifunctional PA6/GO nanocomposites with noted improvements in the mechanical properties. 13 The solution mixing and melt blending methods are used for a large-scale fabrication of GPA nanocomposite materials. The solution mixing technique for GPA nanocomposites is a simple scale-up technique that involves mixing of graphene and PA in a suitable solvent. By using a solution mixing process, Kausar created PA1010/PTA reinforced graphene nanoplatelets nanocomposites with improved mechanical properties for automotive part application. 14 Liu et al. also used a solution mixing method to build graphene/CO-PA nanocomposites with good properties, such as electrical, thermal, mechanical and especially a low percolation threshold properties. 15 Recently, Li et al. studied the PA6/GO-HP nanocomposites, the results showed that the strong interfacial interaction and hydrogen bonding between GO-HP and PA matrix. 16 Wang et al. predicted the thermal conductivity of expanded graphite (EG) sheets filled polymer composite. EG is the perfect nanofiller material to produce composites with high thermal conductivity. 17 Liu et al. prepared PA6/GO nanocomposites; the thermal stability of the nanocomposite was significantly improved compare to neat PA6. 18 Tang et al. fabricated PA6/GO composite fibres; the results showed the thermal properties. 19 Rafiq et al. enhanced mechanical properties of PA12 by addition of small content of ultra-toughened functionalized graphene. 20 The solvent-assisted melt blending technique is a simple and easy way to disperse graphene into PA matrix. 21 Melt blending technique has been optimized in order to improve dispersion efficiency. 22 Jin et al. prepared high-performance nanocomposites by direct melt blending process. 23 Wu et al. studied graphene-filled PA6 nanocomposites and found that the melting temperature of the composites increased as the concentration of graphene increased. 24 Lee et al. prepared G-PA6 composites by melt compounding with enhanced mechanical properties. 25 Korkees et al. studied recycled PA6/PA6,6 blends-graphene nanocomposites; the results showed that the thermal conductivity of nanocomposites was improved using functionalised graphene. 26 As a result, it appears that combination of solution mixing and melt blending is the only way to achieve better graphene dispersion in polymeric materials with exceptional properties. 9
In this study, the delamination/adsorption method18,27 is applied in which dimethylacetamide solvent is used to make novel carboxyl graphene/melamine-based polyamide (CGMPA) nanocomposites. The AD-Carboxyl Graphene (CG) is used as a nanofiller in the PA matrix, resulting in strong interfacial interactions with PA. The delaminated dispersion of CG sheets are intercalated by melamine-based polyamide matrix to obtain CGMPA nanocomposites. The technique is efficient, simple to scale-up for preparation of nanocomposites. The good dispersion of graphene and its distribution into the polymer matrix have a significant effect on the thermal properties of CG reinforced PA nanocomposites which is highlighted in this research work.
Materials and characterization
Materials
AD-Carboxyl Graphene was purchased from Ad-Nano Technologies Pvt. Ltd, Shimoga, India. Terephthalic acid (TPA) and melamine (MA) were purchased from Sigma-Aldrich Chemical Company, USA. Dimethyl sulphoxide (DMSO), tetrahydrofuran (THF), Dimethylacetamide (DMAC), acetone and methanol were purchased from Spectrochem Pvt. Ltd., Mumbai, India. All chemicals were used without further purification.
Synthesis of melamine-based polyamide
Melamine-based polyamides were prepared by one-pot polycondensation method. 28 The molar ratio of MA over TPA was set to be 1:3. To obtain polyamide, MA (21 mmol) was dissolved in 100 mL three-neck round bottom flask with 25 mL anhydrous DMSO to make polyamide. TPA (7 mmol) was then added in the flask and the reaction mixture was heated up to 120°C for 3 h. The reaction mixture was further heated to 150°C for 72 h. The resultant white precipitate was filtered, washed with hot water, THF, and methanol for several times, and extracted with acetone for 24 h. Finally, the white powder of PA was dried under vacuum at 80°C for 24 h. Figure 1 shows the synthesis process for PA.
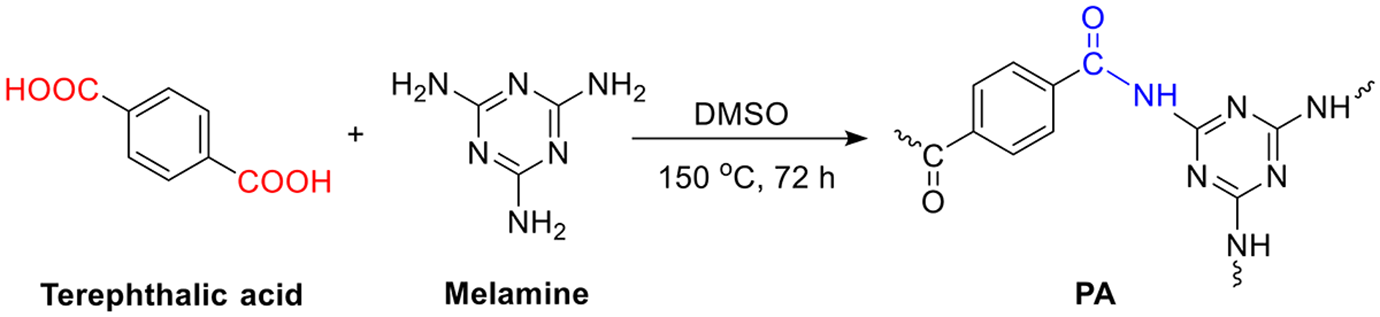
Synthesis of melamine-based polyamide.
Synthesis of CGMPA nanocomposites
The synthesis of CGMPA is depicted in Figure 2. The nanocomposites were synthesized by delamination/adsorption technique listed according to the method in Refs.18,27 To produce CGMPA with 1 wt.% of CG, 10 mg of CG was immersed in DMAC (50 ml) and sonicated for 30 min in bath sonicator to produce suspension. The suspension was transferred quickly into a 100-mL three-necked round-bottom flask which was preplaced in certain boiling DMAC (30 ml) solution of PA (1 g). The flask was equipped with a magnetic stirrer. The resulting mixture was poured into an empty beaker, cooled, filtered, and washed with acetone to extract DMAC after 30 minutes of stirring. The resulting CGMPA nanocomposites were dried at 60°C for 24 h in a vacuum oven before characterization. Pristine PA was treated under the same procedure as for nanocomposites without adding CG. Corresponding to weight fractions of CG the composites are named as CGMPA1 (1% CG), CGMPA3 (3% CG), and CGMPA5 (5% CG).
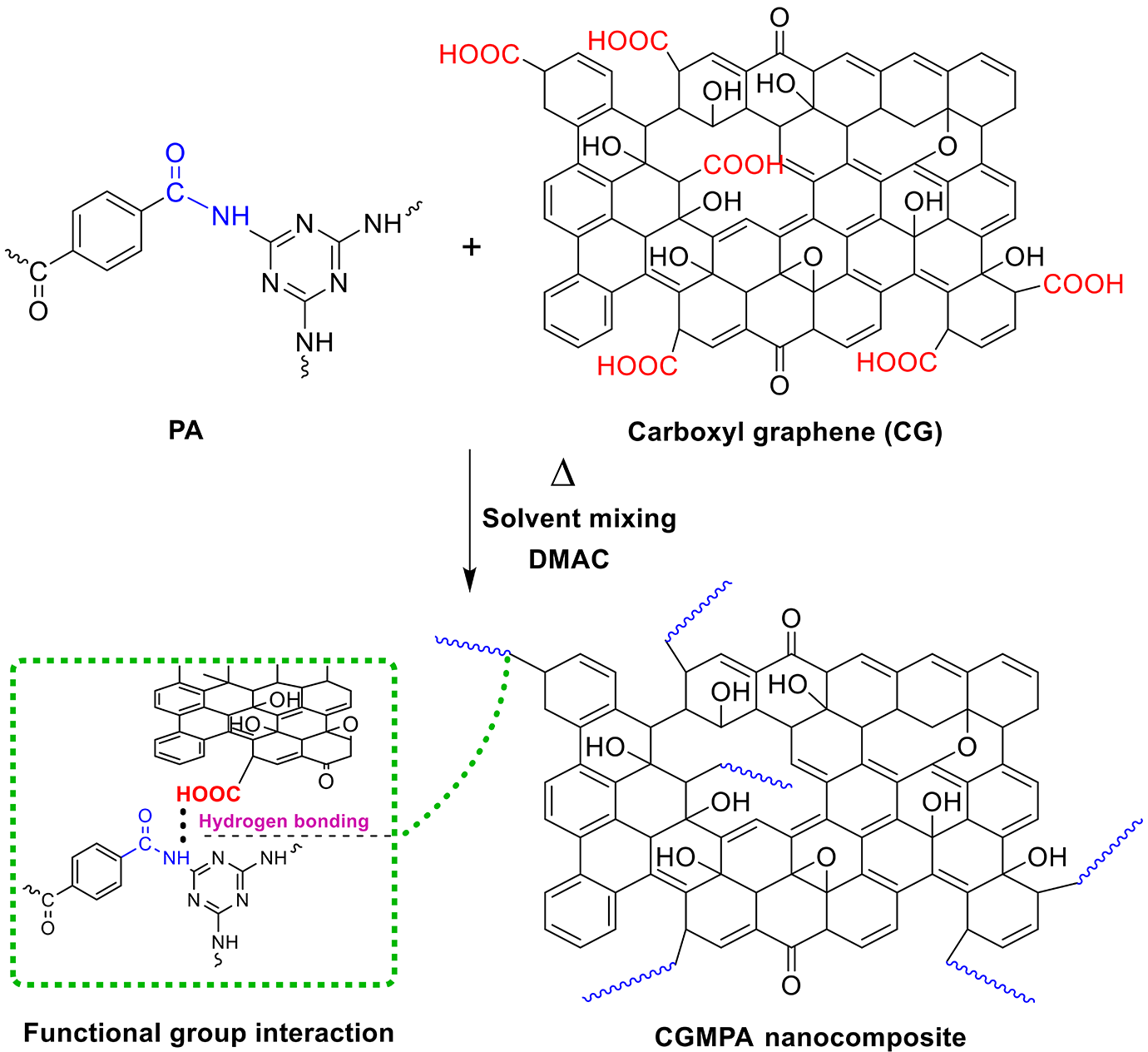
Synthesis of CGMPA nanocomposites.
Characterization
IR spectra of composites were recorded on Bruker FTIR (Bruker, Germany) spectrophotometer in the range of 400–4000 cm−1.1HNMR spectra were recorded on Bruker Avance-II 400 MHz (Bruker, Switzerland). The powder XRD was recorded on XPERT-PRO (PANalytical, Netherlands) using Cu Kα radiation (λ = 1.54060 Å). Diffractograms were obtained by varying the angle from 3° to 40° (2θ) at room temperature. The microstructures were observed using a Field emission scanning electron microscope (FESEM; TESCAN, Mira 3-XMU, Tescan Co, Brno, Czech Republic). DSC curves were obtained on a NETZSCH, DSC 204 F1 Phoenix differential scanning calorimeter (NETZSCH-Gerätebau, GmbH). DSC scans were performed from 30°C to 500°C with a heating rate of ±10°C/min in nitrogen atmosphere. The thermogravimetric analysis (TGA) was carried out using NETZSCH STA-2500 thermal analyzer (NETZSCH-Gerätebau, GmbH) from 30°C to 600°C with a heating rate of ±10°C/min in nitrogen atmosphere.
Results and discussion
Morphology and chemical structure of CG, PA, and CGMPA nanocomposites
FTIR Spectra of CG, PA, and CGMPA-x (CGMPA1, CGMPA3, and CGMPA5 nanocomposites) are shown in Figure 3. For PA, the absorption peaks at 1279 cm−1 (C–N stretching), 1542 cm−1 and 1464 cm−1 (C=N stretching), 1534 cm−1 (aromatic C=C stretching), 3133 cm−1 (aromatic C–H stretching), 1676 cm−1 (C=O stretching), 1547 cm−1 (N–H bending), 3392 cm−1 (N–H stretching). The PA’s formation is confirmed by these vibrations. 29 For CG, the absorption peaks at 1037 cm−1 and 1164 cm−1 (C-O-C stretching), 1391 cm−1 (C=C skeleton vibrations of graphene backbone), 1620 cm−1 and 1721 cm−1 (C=O stretching vibrations of carboxyl and ketone groups), 2850–2950 cm−1 (C-H bonds in CH2 groups), and 3336 cm−1 (O-H stretching vibrations of OH groups). The existence of oxygen-containing functional groups on the edges of CG nanosheets was revealed by these vibrations. 30 The addition of CG has no significant effect on the characteristic peaks of PA in CGMPA nanocomposites, but the position and intensity of PA changed slightly as the CG content increased, i.e. the amide II peak at 1547 cm−1 has shifted slightly. These vibrations indicated the interaction between CG and PA. 31 The SEM micro images of the CG (a) and PA (b) are shown in Figure 4. The CG sheets are quite flat and have many stacked layers. The PA micro images show aggregation of small particles and a structure of interconnected micro holes or macropores. 28 The 1H NMR spectra of PA are shown in Figure 5. The absorption signals of aromatic protons appeared in the range of 7.8 to 8.3 ppm. The appeared peak at 13.3 ppm corresponding to the amide protons (C(=O)–NH–).
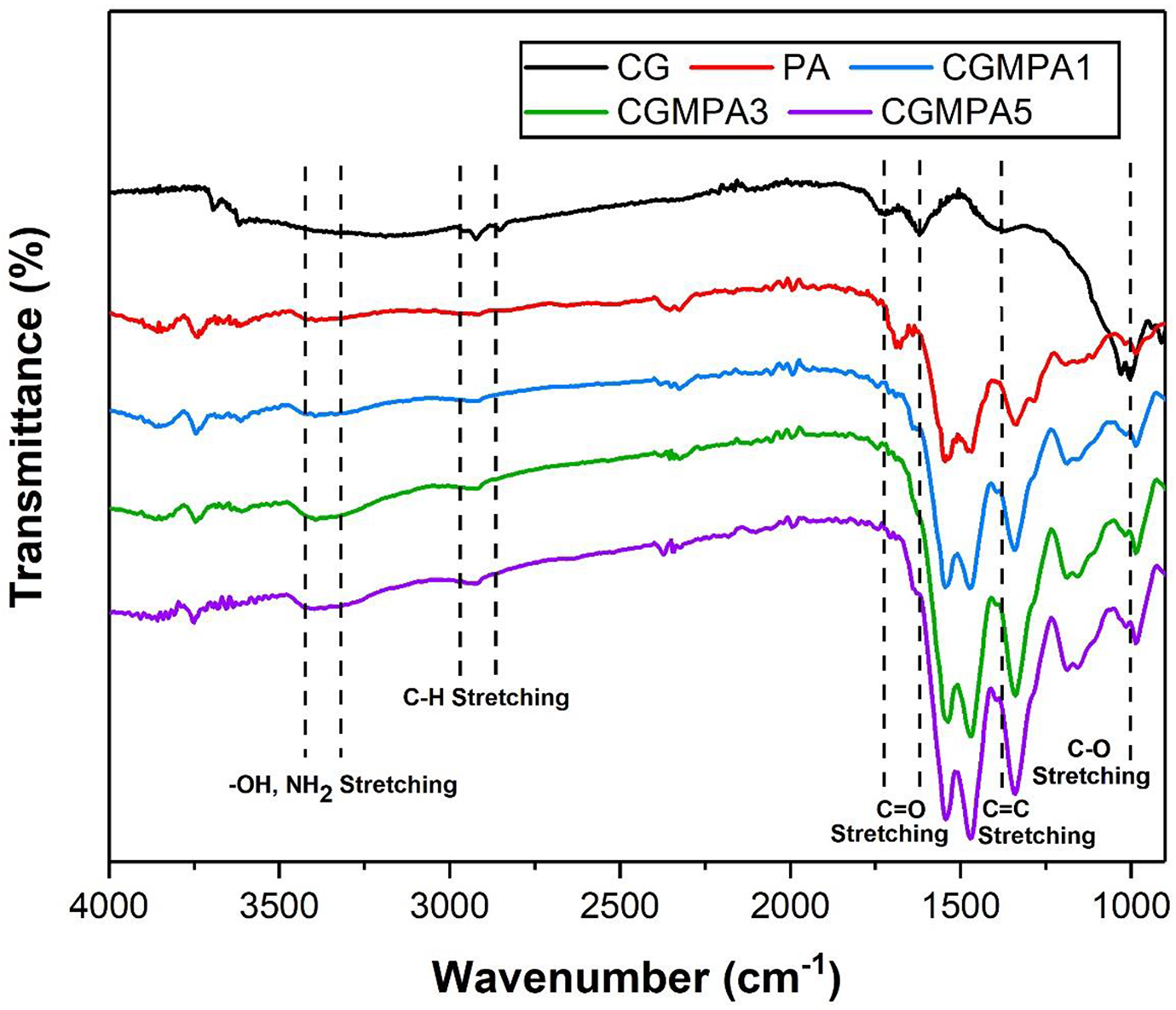
FTIR spectra of CG, PA, and CGMPA-x.

SEM images of CG (a), and PA (b).
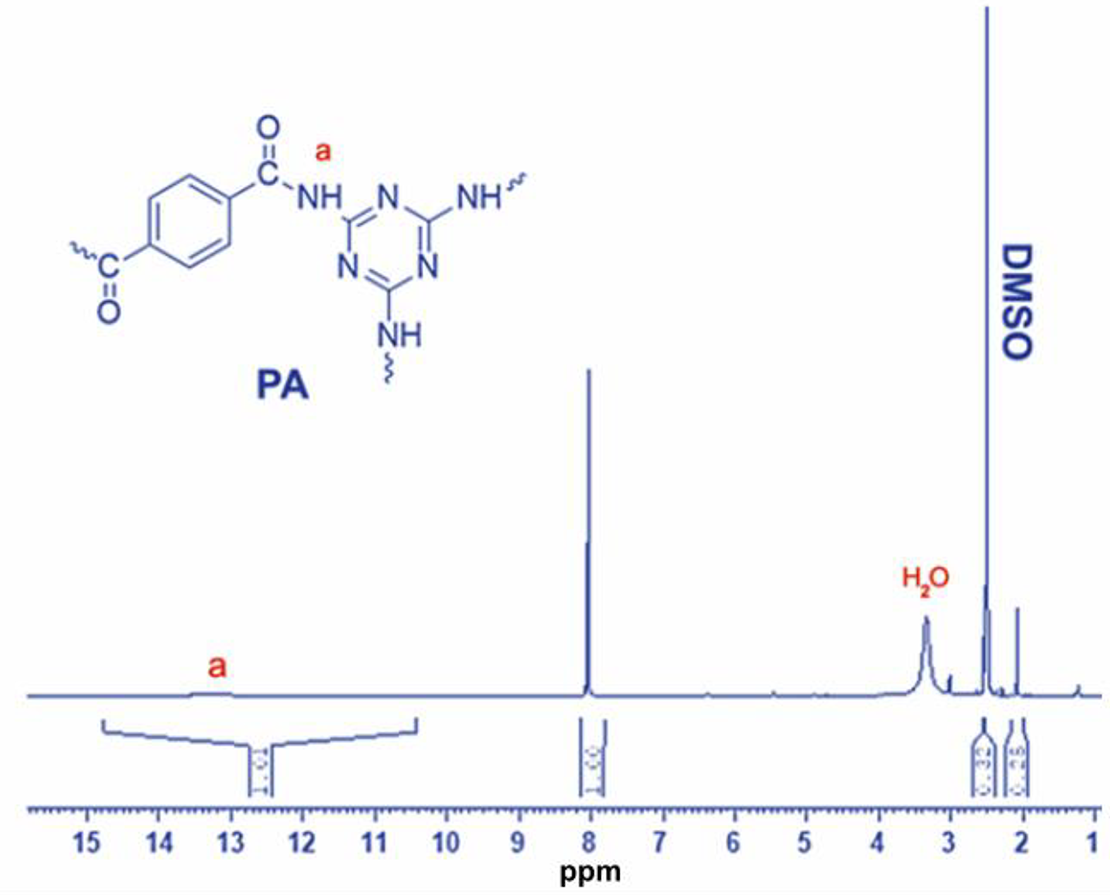
1H NMR spectra of PA.
The XRD patterns of CG, PA, and CGMPA-x are shown in Figure 6. Bragg’s condition for diffraction is λ = 2d sin θ where λ = 1.54 Å (wavelength of X-ray), n = 1 (order of diffraction) and d = interatomic distance. 32 The diffraction pattern for CG gives a sharp peak at 26.5° (2θ) which corresponds to an interlayer spacing of 0.33 nm because of carboxyl group functionalities. PA is a polymorphic polymer, that can produce typical α phase diffraction peak. The diffraction peaks near 25.1° (2θ) features to the inter-chain hydrogen bonded (100) plane of amide groups and 27.7° (2θ) showing the overlapping diffraction peaks for (010) and (110) planes of PA. 6 To compare the shifts, the ratio and height of the diffraction peak were used. The intensity of strong diffraction peak of CG decreased in the nanocomposite, which reveals that the CG layers are stripped into a layer structure and exfoliated in the nanocomposite. 18 The CGMPA3 and CGMPA5 nanocomposites displayed CG diffraction peak at 26.4° (2θ) and 26.5° (2θ), respectively. The CGMPA3 nanocomposite shows only one dominating diffraction peak in place of two peaks because the intensity of peaks decreased due to the changes in d-spacing. 33 The diffraction peaks of CGMPA nanocomposites shifted toward lower 2θ values as the CG wt.% increase compared to neat PA, confirming the major significance influence on the crystallization behaviours of PA chains.23,34
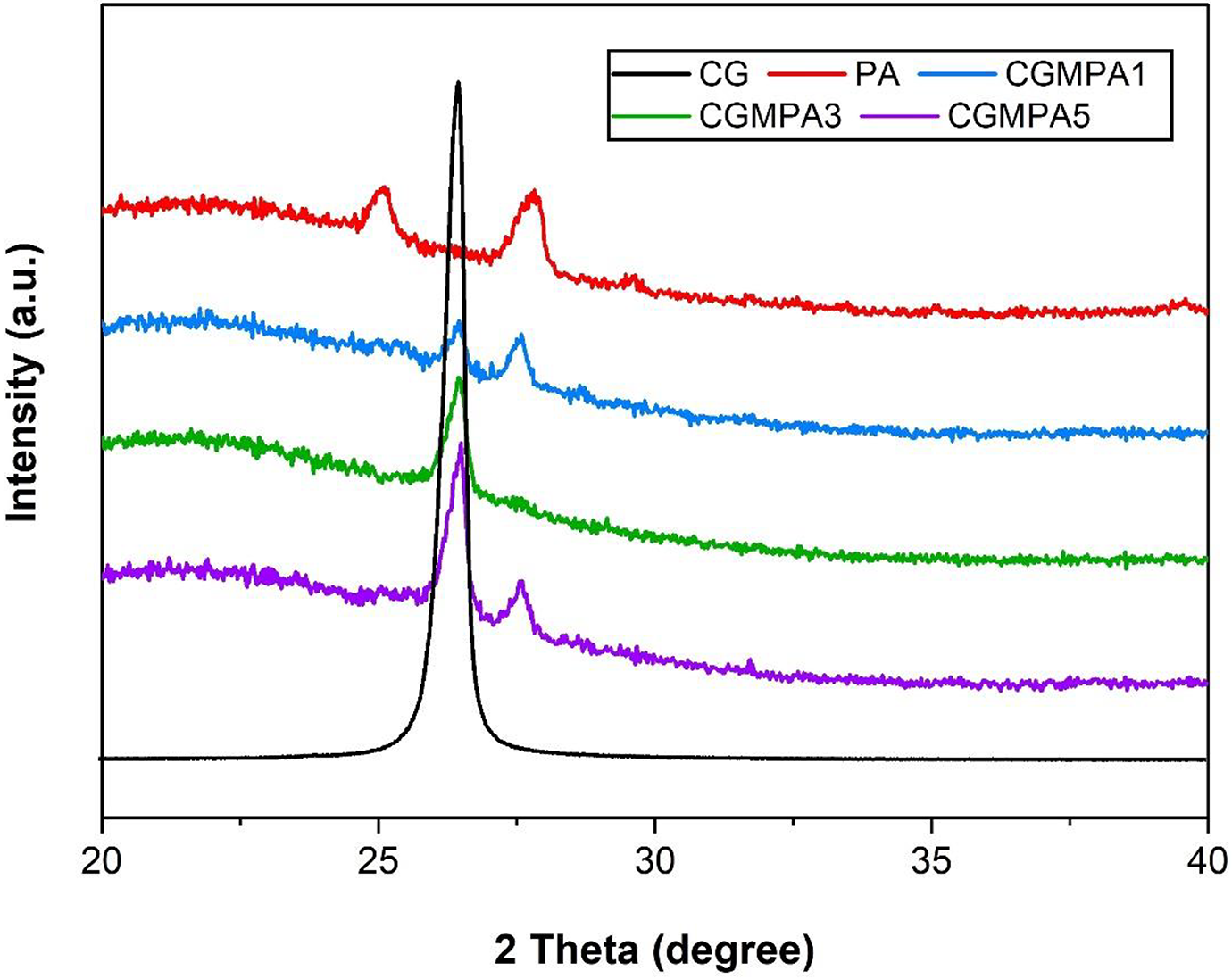
XRD patterns of CG, PA, and CGMPA-x.
The uniformity in the dispersion of suspended CG nanoparticles in the PA matrix was investigated using FESEM imaging. The FESEM images of PA and CGMPA nanocomposites PA (a), CGMPA1 (b), CGMPA3 (c), and CGMPA5 (d) are shown in Figure 7. The CGMPA nanocomposite has a different surface morphology than CG. It was observed that PA showed aggregation of small particles and plentiful interconnected holes or macropores structure with bright contrast, 28 whereas the CG produced sheet type wrinkled structure morphology with dark and bright region. The surface morphology showed dispersed CG sheets in 1 wt.% CGMPA1 and 3 wt.% CGMPA3 nanocomposites due to the strong binding force from the interactions between CG and PA. 35 The 1 wt.% CGMPA1 and 3 wt.% CGMPA3 nanocomposites exhibited well distributed and exfoliated morphology. However, The CG distribution in 5 wt.% CGMPA5 nanocomposite was less optimal with large aggregates present in the PA matrix. 24 It shows overlapped patterns of CG sheets in the PA matrix. It is also found that the agglomerations increase when increasing the CG addition. The better dispersion of graphene in the polymer is one of the key features to enhance the physical properties of the polymer, 36 because the agglomeration of the graphene leads to structural defects of that reduce the reinforcing efficiency the nanocomposites. 37

FESEM images of PA and CGMPA nanocomposites PA (a), CGMPA1 (b), CGMPA3 (c), and CGMPA5 (d).
Thermal and crystallization properties of PA and CGMPA nanocomposites
The DSC heating plot results of PA and CGMPA nanocomposites are shown in Figure 8 and Table 1. According to the findings, the melting temperature (Tm) of the CGMPA nanocomposites were changed significantly. The Tm of CGMPA nanocomposites are higher than that of neat PA matrix. It showed the addition of CG can improve heat resistance of the PA matrix. The Tm of nanocomposites were increased due to the strong nucleation effect of CG and increased interfacial interactions between CG and PA in nanocomposites.38,39 Similar behaviours have been reported with other nanocomposites.18,40–42 The neat PA has a melting peak at about 378°C. The CGMPA1, CGMPA3, and CGMPA5 nanocomposites showed melting peak at 390°C, 393°C and 386°C, respectively. However, CGMPA5 nanocomposite showed lower Tm than other two nanocomposites, because excessive addition of CG could cause re-aggregation and re-stacking of CG sheets which leads to less interfacial interactions and hydrogen bonding between CG and PA matrix.43,44
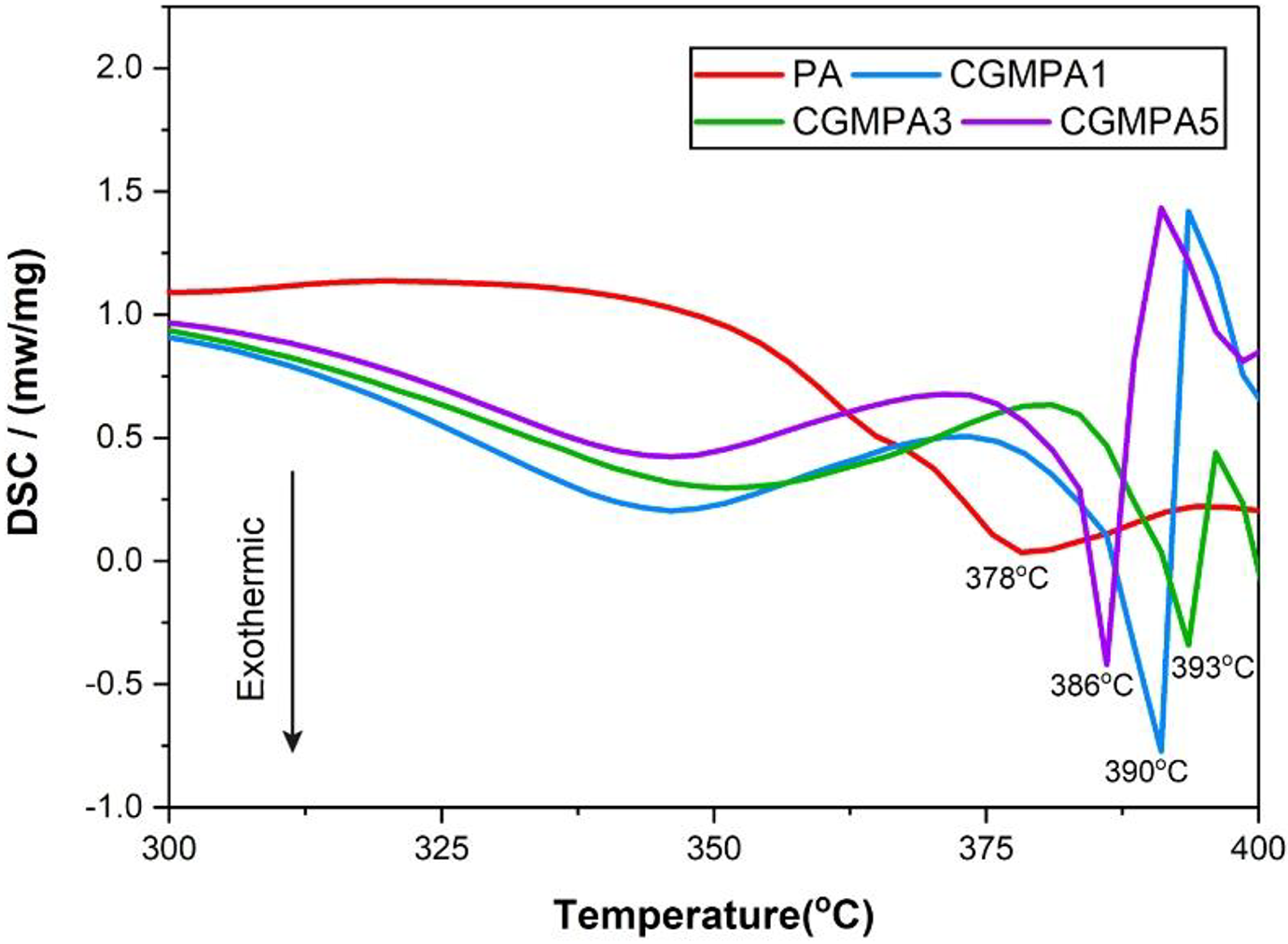
DSC curves of PA, and CGMPA-x.
The melting temperature of CGMPA nanocomposites.
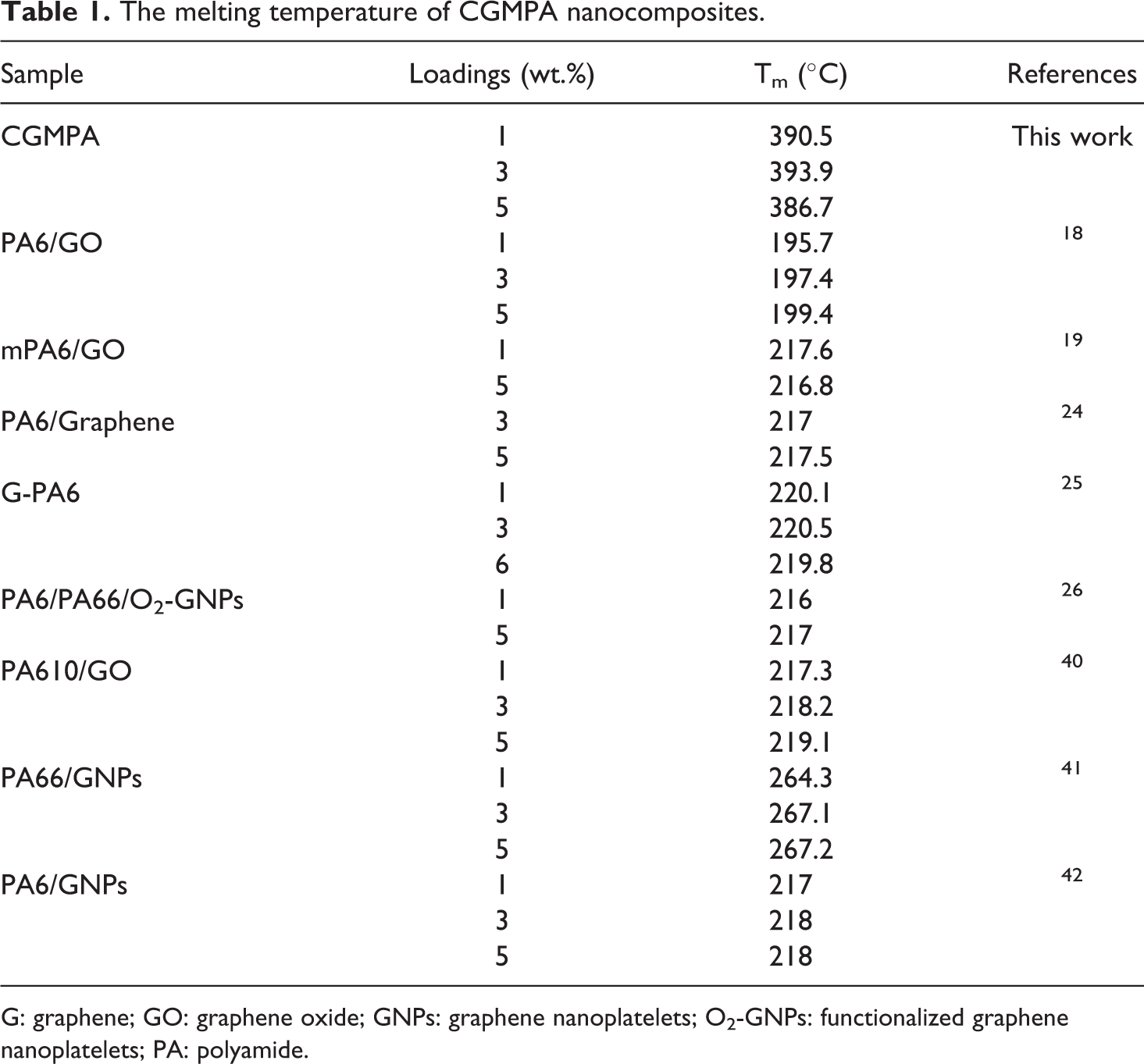
G: graphene; GO: graphene oxide; GNPs: graphene nanoplatelets; O2-GNPs: functionalized graphene nanoplatelets; PA: polyamide.
The TGA analysis defines the relationship between mass loss of samples and temperature; it indicates the variation of the mass with increasing temperature and reveals the thermal decomposition behaviour of materials. The TGA curve presents the thermal stability of polymeric nanocomposite materials. 45 The TGA curve of the PA and CGMPA nanocomposites are shown in Figure 9. The results showed the temperature at the 50% of weight loss of CGMPA1, CGMPA3 and CGMPA5 at 457°C, 463°C, and 455°C, respectively while that of neat PA is 451°C. All nanocomposite have shown less weight loss than neat PA within a temperature range of 300–450°C. The thermal decomposition temperature of CGMPA nanocomposites is higher than that of neat PA matrix, which indicates that CG can increase the thermal decomposition temperature of CGMPA nanocomposites. The addition of CG can enhanced the thermal stability of CGMPA nanocomposites, because the dispersed CG sheets in the PA matrix as a heat transfer medium to conduct heat in time, thus delaying the decomposition process of nanocomposites to a particular extent. The better interfacial interactions decreases the flexibility of the PA chain and hinders movement of macromolecular chains, thus increasing the thermal decomposition temperature of the CGMPA nanocomposites and delaying its decomposition process.43,46,47
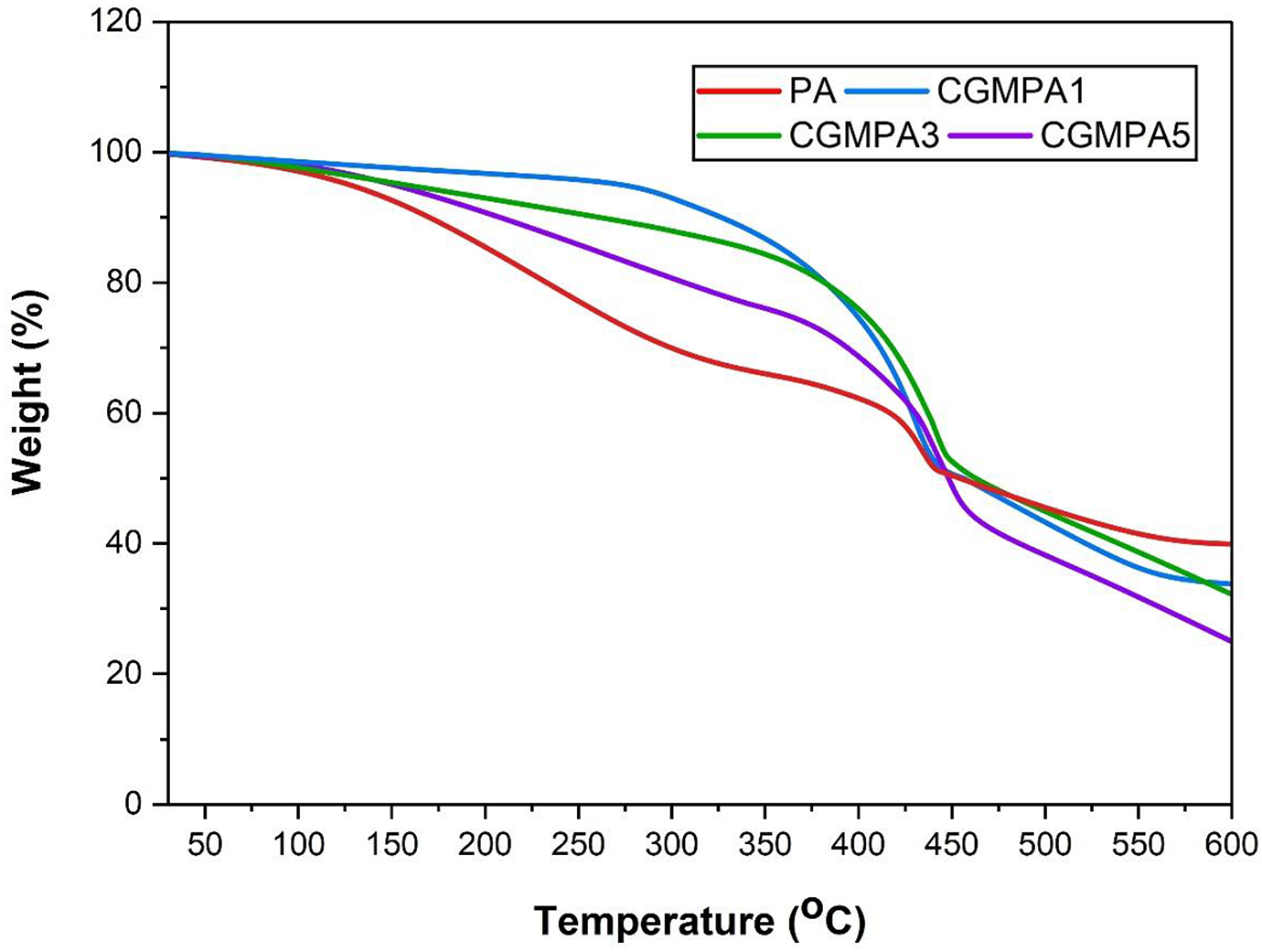
TGA curves of PA, and CGMPA-x.
Conclusion
The novel CGMPA nanocomposites were successfully prepared using CG in PA matrix via delamination/adsorption technique. This technique is efficient and simple to scale-up for synthesis of nanocomposites. The structural, morphological, and thermal properties of the nanocomposites were investigated to study the effect of CG in the properties of the CGMPA nanocomposite. A significant increase of ∼15°C in melting temperature (Tm) was observed upon the addition of 3 wt.% of CG due to the interaction between the CG and PA matrix. A maximum increase about 12°C in thermal decomposition temperature was observed at the 50% of weight loss in CGMPA3 nanocomposite. Owing to the heterogeneous nucleating effect of CG, the thermal stability of CGMPA nanocomposites were improved. The increase in thermal properties must be attributed to the barrier function of the CG sheet and the interfacial interaction between the CG and the PA matrix. The nanocomposite can be considered for promising applications as a thermally stable material, antibacterial coating material, adsorbent for removal of toxic metal ions.
Footnotes
Acknowledgement
The authors are gratefully acknowledge the Department of Chemistry, Gujarat University, Ahmedabad, for providing the essential facilities. The authors are thankful for UGC-Info net & INFLIBNET Gujarat University for providing the resource facilities. The authors also thankfully acknowledge for Department of Biochemistry & Forensic Science, Gujarat University for providing FT-IR facility. SAIF Chandigarh, India, for providing 1H-NMR, SEM and XRD facilities. SAIF, IIT-Madras, Chennai, India, for DSC providing facility. CeNS, Bangalore, India, is acknowledged for providing FESEM and TGA-DTA characterization facilities.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
