Abstract
In this work, magnetic graphene oxide nanocomposites were synthesized by co-precipitation method and used as an adsorbent for removal of arsenic (V) ions from water. The structure and morphology of magnetic graphene oxide nanocomposites were studied by X-ray diffraction, Fourier transform infrared spectroscopy, transmission electron microscopy, Brunauer–Emmett–Teller specific surface area, and vibrating sample magnetometry. Fourier transform infrared spectroscopy, X-ray diffraction, and transmission electron microscopy results of magnetic graphene oxide presented that the Fe3O4 nanoparticles in the size range of 10–25 nm were decorated on graphene oxide nanosheets. The adsorption properties of magnetic graphene oxide nanocomposites for arsenic (V) from water were investigated to study the effects of magnetic graphene oxide mass ratio, contact time, pH, and initial concentration. The suitable magnetic graphene oxide mass ratio of nanocomposites for arsenic (V) adsorption was determined to be 4:1 (FG2). The adsorption process on FG2 followed a pseudo-second-order kinetic and well fitted in to Langmuir isotherm model with the maximum adsorption capacity of 69.44 mg/g at pH 3. Accordingly, FG2 could be used as an effective adsorbent for removal of arsenic (V) from water.
Introduction
Arsenic (As) is considered as one of the most toxic chemicals and a carcinogenic element (Chandra et al., 2010). The most common forms of arsenic species in aqueous environments are arsenite As(III) (as H3AsO3 and H2AsO3−) in ground water system and arsenate As(V) (as H2AsO4− and HAsO42−) in surface water. Arsenic compounds easily accumulate in human organs and cause various health problems (Feng et al., 2012; Vaclavikova et al., 2008). Thus, the removal of As from water has been investigated.
Currently, the research and development of new adsorbent materials is being promoted. Graphene (Gr) with unique characteristics, such as large surface area, mechanical strength, and thermal conductivity, has attracted great interest of many researchers. Gr is a monolayer of carbon atoms, which is packed firmly, forming a two-dimensional structure of honeycomb lattice (Meyer et al., 2007; Zhu et al., 2010). Graphene oxide (GO), a derivation of Gr, is fabricated by exfoliation of graphite oxide, which is synthesized from graphite by oxidizing. GO contains a variety of oxygen-containing groups on the surface like hydroxyl (–OH), epoxy (–O–), carbonyl (–C = O), and carboxylic (–COOH). These groups provide GO with negative charged surface and an ability to interact with positive ions such as heavy metals, dyes, and organic compounds (Stankovich et al., 2006; Zhao et al., 2011). However, the nano size, high dispersion, and difficult separation of GO prevent the direct use of GO as adsorbents. To solve these problems, the magnetic graphene oxide (Fe3O4/GO) nanocomposite has been developed. The unique advantages of Fe3O4/GO such as high adsorption capacity and easy separation of Fe3O4/GO make it a potential adsorbent for the removal of pollutants from water (Fan et al., 2016).
In this work, the Fe3O4/GO nanocomposites were synthesized by co-precipitation method. The characterization of nanocomposites was examined by X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR), transmission electron microscopy (TEM), Brunauer–Emmett–Teller (BET) specific surface area, and vibrating sample magnetometry (VSM). Effects of Fe3O4:GO mass ratio, contact time, pH, and initial concentration on the As(V) adsorption capacity of Fe3O4/GO nanocomposites were studied.
Materials and methods
Materials
Graphite powder (particle size of 20 µm) and arsenic acid (H3AsO4 solution, 1000 mg/l) were purchased from Sigma-Aldrich, Germany. H2SO4 (98 wt%), H3PO4 (85 wt%), H2O2 (85 wt%), NaOH (99 wt%), HCl (36 wt%), FeCl3·6H2O (99 wt%), and FeCl2·4H2O (99 wt%) were purchased from Xilong, China. KMnO4 (99 wt%), NH4OH (30 wt%), and C2H5OH (95 wt%) were purchased from ChemSol, Vietnam.
Synthesis of Fe3O4/GO nanocomposites
GO was prepared by improved Hummers’ method (Marcano et al., 2010). Fe3O4/GO was synthesized by co-precipitation method (Fan et al., 2016). Briefly, 10 ml of FeCl3·6H2O and FeCl2·4H2O solution was slowly added into 50 ml of GO suspension (6 mg/ml). The mixture was stirred and heated to 80°C at pH 10 for 2 h. The black precipitation was collected by using a magnet, then washed with water and ethanol. After drying at 50°C for 24 h, the nanocomposites were obtained. The nanocomposites with different Fe3O4:GO mass ratios of 8:1, 4:1, 2:1, and 1:1 were marked as FG1, FG2, FG3, and FG4, respectively.
Characterizations
The crystal state was analyzed by XRD D2 Phaser machine (Bruker, Germany) with Cu Kα radiation (λ = 0.154 nm). FTIR spectra (Bruker FTIR Alpha-E, Germany) are used to analyze the functional groups on the surface of materials. The morphology of the materials was investigated by TEM (JEM-1400 microscope, Japan). The BET specific surface areas of materials were determined by Nova 3200e through nitrogen adsorption–desorption isotherm method (Quantachrome Instruments, USA). The elemental composition of materials was specified by using scanning electron microscope along with energy-dispersive X-ray analyzer (SEM/EDX) (Jeol JMS 6490, JEOL, Japan). Magnetic properties of the nanocomposites were performed using a MicroSense Easy VSM version 9.13 L machine (MicroSense, USA).
Adsorption experiments
Twenty milligrams of adsorbent was poured into 50 ml of As(V) solution. Effects of mass ratios of Fe3O4 to GO (FG1, FG2, FG3, and FG4), contact time (30, 60, 120, 240, 480, and 1440 min), pH (3, 5, 7, 9, and 12), and initial As(V) concentration (25, 50, 100, 150, and 200 mg/l) were studied. The residual As(V) concentration was determined by inductively coupled plasma mass spectrometry (ICP-MS 7500, Agilent, USA). All experiments were triplicated to estimate the error. The adsorption efficiency (H, %) and adsorption capacity (q, mg/g) were calculated using the following equations (1) and (2)


The kinetic of adsorption process was investigated by pseudo-first-order and pseudo-second-order models. These models can be expressed as equations (3) and (4)


The zeta potential (pHpzc) values of materials were determined by pH drift method (Martinez-Vargas et al., 2018). Besides, to investigate adsorption capacity, the experimental data were applied to the Langmuir and Freundlich isotherm models. The linear equations for isotherm models are expressed as follows (equations (5) and (6))


Results and discussion
Characterization
XRD patterns
The XRD patterns of GO, Fe3O4, and Fe3O4/GO are shown in Figure 1. The diffraction peak at 2θ = 11.12°, ascribed to the crystallographic planes (002), indicates the distance of 0.795 nm between GO sheets. This distance was higher than the interlayer spacing of graphite (0.340 nm) since the oxygen-containing groups were inserted between GO sheets (McAllister et al., 2007). Fe3O4/GO had several peaks at 2θ = 30.15°, 36.27°, 43.32°, 53.89°, 57.13°, and 62.29°, which were respectively assigned to the crystallographic planes (220), (311), (400), (422), (511), and (440) of Fe3O4 (Compton and Nguyen, 2010; Li et al., 2012; Liu et al., 2011). Additionally, the disappearance of GO peak indicates that Fe3O4 nanoparticles were attached on GO surface, thus increasing the interlayer spacing between GO sheets.

XRD patterns of (a) GO and (b) Fe3O4 and Fe3O4/GO. GO: graphene oxide.
FTIR spectra
The functional groups on the surface of GO and Fe3O4/GO were studied by FTIR spectra (Figure 2).

FTIR spectra of Fe3O4 and Fe3O4/GO. GO: graphene oxide.
For Fe3O4/GO, several peaks at 3355.43, 1600.63, 1399.45, and 1059.30 cm−1 correspond to the vibration of hydroxyl (O–H), carbonyl (C=O), alkoxy (C–O), and aromatic C=C bonds, respectively (Metin et al., 2014; Yang et al., 2009). The peaks at 630.98 and 584.13 cm−1 can be ascribed to Fe–O bonds of Fe3O4 (Sheng et al., 2012). This result indicates the existence of distinctive functional groups of GO and Fe3O4.
TEM images
The morphologies of the materials were analyzed by TEM images as shown in Figure 3. GO shows high transparency with small wrinkles, which indicates effective exfoliation during sonication. The effect of Fe3O4:GO mass ratio on the size and the distribution of Fe3O4 on GO surface is shown in Figure 3(b) to (e). The Fe3O4 nanoparticles with the average size of 10–15 nm were distributed uniformly on GO surface. However, Fe3O4 nanoparticles in FG1 were larger with average size of 15–25 nm. The Fe3O4 nanoparticles in FG2 were highly dispersed and showed an excellent interaction with GO sample. Besides, TEM images of FG3 and FG4 present fewer decoration of Fe3O4 nanoparticles compared to FG2 and FG1.

TEM images of (a) GO, (b) FG1, (c) FG2, (d) FG3, and (e) FG4. GO: graphene oxide.
The formation mechanism of Fe3O4/GO nanocomposite is shown as follows: (1) Fe2+ and Fe3+ ions linked with functional groups on the surface of GO and then (2) hydrolyze to form Fe3O4 NPs.
BET specific surface areas
Table 1 shows the BET surface areas of Fe3O4/GO nanocomposites and other materials. The Fe3O4 nanoparticles were attached on the surface of GO, decreasing the aggregation of Fe3O4 and stacking of GO, leading to the increase in specific surface area. Besides, the surface area of FG2 was higher than that of other nanocomposites due to a larger number of active sites on the surface. FG3 and FG4 had fewer Fe3O4 nanoparticles, leading to the reduction in the number of sites, which resulted in the decrease in surface area.
The BET specific surface areas of Fe3O4/GO nanocomposites and other materials.

BET: Brunauer–Emmett–Teller; GO: graphene oxide.
SEM/EDX
The composition of elements in Fe3O4/GO was determined through SEM/EDX data as shown in Figure 4 and Table 2. The Fe3O4 nanoparticles of FG2 were evenly dispersed on GO surface, indicating GO could reduce the agglomeration of Fe3O4 nanoparticles effectively. In EDX results, the weight percentage of Fe3O4 on FG2 was approximately 69.98%, which was consistent with experiment (66.67%).

The EDX results of FG2.
The composition of elements in FG2.

VSM
The saturation magnetization (Ms) values of Fe3O4/GO nanocomposites were calculated to be 41.13, 36.34, 28.52, and 18.54 emu/g, respectively. The nanocomposites can be easily separated by using external magnetic field. The Ms values of Fe3O4/GO and other materials are shown in Table 3. The Ms values of nanocomposites are less than that of the bulk Fe3O4 (92 emu/g) due to the smaller size of Fe3O4 particles and the amount of loading of GO in the nanocomposite.
The Ms values of Fe3O4/GO nanocomposites and other materials.

Effects of factors on the As(V) adsorption capacity of Fe3O4/GO nanocomposites
Fe3O4:GO mass ratio
The As(V) adsorption capacities of GO, Fe3O4, and Fe3O4/GO nanocomposites are shown in Figure 5. The presence of Fe3O4 nanoparticles on GO surface increased the adsorption capacity of Fe3O4/GO. Compared to GO and Fe3O4, the adsorption capacities of nanocomposites were also higher due to the following reasons: (1) the electrostatic interaction between the negative charged surface of GO and As(V) ions, and (2) the complex interaction between metallic ions and oxygen-containing groups on the surface of Fe3O4/GO (Wang et al., 2013).

The As(V) adsorption capacities of GO, Fe3O4, FG1, FG2, FG3, and FG4. GO: graphene oxide.
The FG2 has the highest As(V) adsorption capacity due to its largest surface area and the uniform distribution of Fe3O4 nanoparticles on the surface of GO. Therefore, FG2 was selected as an optimal adsorbent for the following experiments.
Contact time
Figure 6 shows the increase in As(V) adsorption capacity of FG2 as contact time increased. The As(V) adsorption process occurred rapidly due to a substantial amount of active sites. After 480 min, As(V) ion concentration slightly changed due to the reduction of empty active sites on the surface of FG2. Therefore, the adsorption time of FG2 for As(V) was 480 min. The adsorption process of As(V) by FG2 well fitted to the pseudo-second-order model, with a correlation coefficient value close to 1 (R2 = 0.9987) (Figure 7).

Effect of contact time on As(V) adsorption capacity of FG2.

The linear pseudo-second-order model of FG2 for As(V) adsorption.
pH
The increase of pH values inhibited the As(V) adsorption capacity of FG2 (Figure 8). As(V) ions exist in different forms including H3AsO4 (pH < 2.1), H2AsO4− (2.1 < pH < 6.9), HAsO42− (6.9 < pH < 11.5), and AsO43− (pH > 11.5) (Zhu and Bates, 2013). Based on the experimental data, the pH of zero-point charge (pHZPC) of FG2 was measured to be 5.2. When the pH was lower than 5.2, the surface of FG2 was positively charged, making –OH and –COOH groups on FG2 surface become –OH2+ and –COOH2+ cations, which increased the number of active sites to interact with As(V) ions (Saadi et al., 2015; Zhu and Bates, 2013), leading to the increase in the adsorption capacity of FG2. As pH was increased, the surface of FG2 became less positively charged, the interactions between surface charges and As(V) declined gradually and changed into repulsive forces, decreasing the adsorption capacity. The experimental data showed that the effective removal conditions of FG2 for As(V) adsorption was achieved at pH 3.

Effect of pH on As(V) adsorption capacity of FG2.
Initial concentration
Figure 9 shows the relationship between the adsorption capacity of FG2 and initial As(V) concentration. The uptake positively correlated with the initial concentration of As(V). The parameters of Langmuir, and Freundlich isotherm models are showed in Table 4 and Figure 10. The correlation coefficient of Langmuir model (R2 = 0.9059) was higher than that of Freundlich model (R2 = 0.8199).

Effect of initial concentration on As(V) adsorption capacity of FG2.
Parameters of the Langmuir and Freundlich models of FG2 for As(V) adsorption.

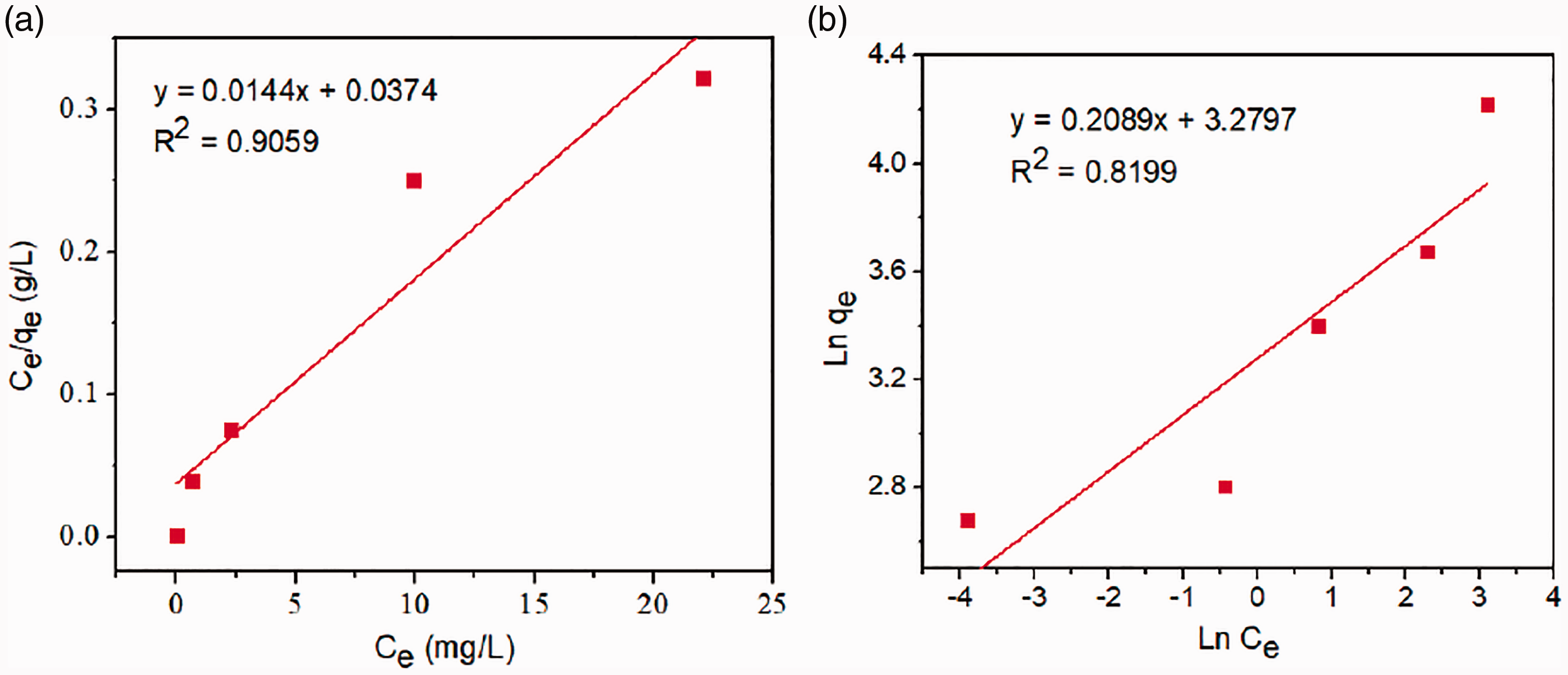
Langmuir (a) and Freundlich (b) isotherm model of FG2 for As(V) adsorption.
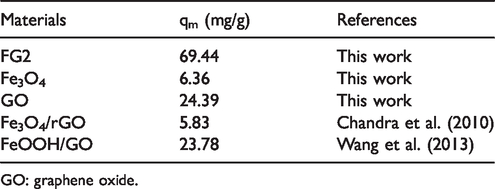
The adsorption of FG2 followed the Langmuir isotherm model with qm = 69.44 mg/g. Besides, the As(V) adsorption capacity of FG2 was in comparison with other materials (Table 5). The result could be explained as the linkage of Fe3O4 nanoparticles on the GO surface, leading to an increase in active sites and surface area, resulting in the increase in adsorption capacity.
Maximum adsorption capacity qm (mg/g) of FG2 and other materials for As(V).
GO: graphene oxide.
Besides, the As(V) adsorption capacity of FG2 was compared with other materials as shown in Table 5. The result can be elaborated that Fe3O4 nanoparticles linked on the GO surface, leading to an increase in active sites and surface area, resulting in the adsorption capacity being increased.
Adsorption mechanism
The As(V) adsorption mechanism of Fe3O4/GO was interpreted via the surface complexation between As(V) ions and functional groups on nanocomposite surface, consisting of two types: outer-sphere and inner-sphere. For outer-sphere complexation, the interaction between As(V) ions and adsorption sites on nanocomposite surface depended on the electrostatic interaction. The in-sphere complexation was formed in Fe3O4/GO structures, the As–O–Fe bonds were formed between As–O(H) groups from H2AsO4− and HAsO42− as ligands and –OH, –COOH groups of Fe3O4/GO structure (Huong et al., 2018; Kumar et al., 2014).
Based on the results of the survey of pH effects, the adsorption capacity of Fe3O4/GO reduced while pH values increased. At low pH, the concentration of H+ ions in the solution increases, –OH and –COOH groups become –OH2+ and –COOH2+ cations, which is advantageous for the adsorption of As(V) (Huong et al., 2018). The main mechanism of adsorption depends on the outer-sphere complex as follows

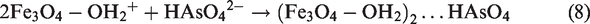




Conclusions
Fe3O4/GO nanocomposites were successfully fabricated by co-precipitation method. XRD, FTIR, TEM, and SEM/EDX results showed that the Fe3O4 particles were anchored on the surface of GO sheets. The Fe3O4/GO with mass ratio of 4:1 (FG2) showed highest adsorption capacity of As(V) compared with other nanocomposites. The adsorption process of FG2 well fitted to the pseudo-second-order and Langmuir isotherm models with the maximum adsorption capacity of 69.44 mg/g at pH 3. Therefore, FG2 could be applied as an adsorbent to remove As(V) from water.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Ho Chi Minh City University of Technology, VNU-HCM, under grant number BK-SDH-2020-1880319.
