Abstract
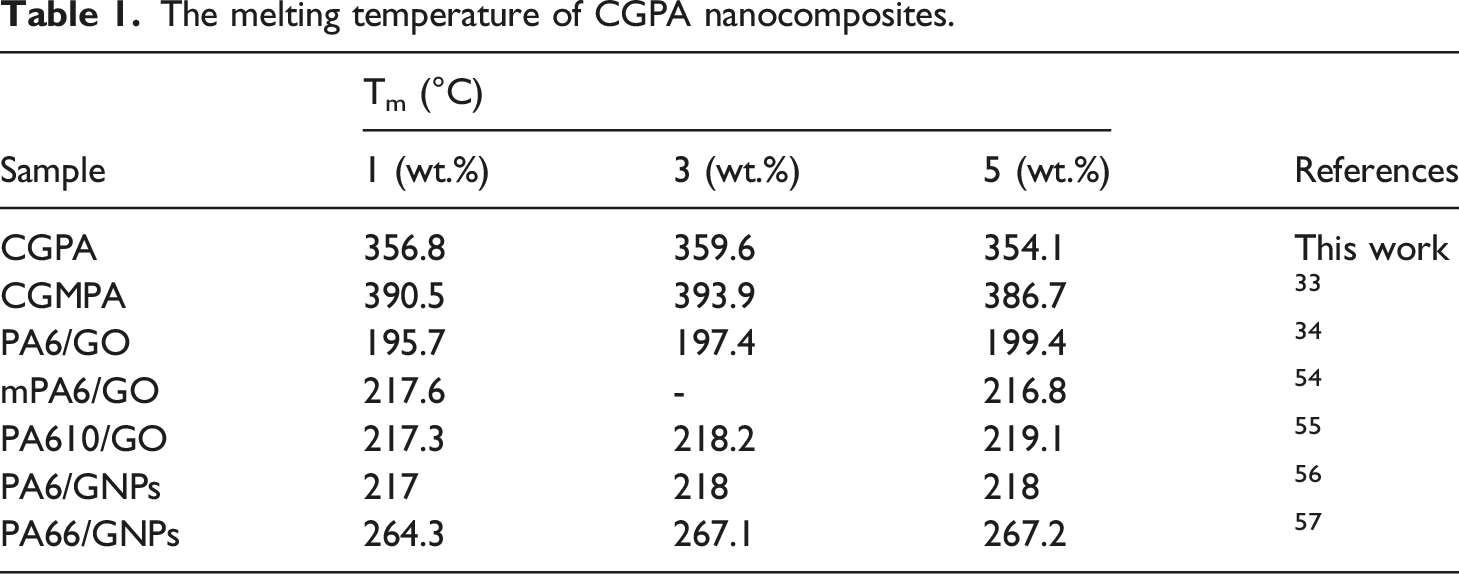
Carboxyl-functionalized graphene (CG)reinforced aromatic polyamide (PA) nanocomposites (CGPA) were prepared with CG nanofillers (1, 3 and 5 wt.%) by the delamination/adsorption method. PA was synthesized by 2,6-pyridine dicarboxylic acid with aromatic diamine via direct polycondensation reaction. The prepared CGPA nanocomposites were evaluated through Fourier transform infrared techniques (FTIR), field emission scanning electron microscopy (FESEM), X-ray diffraction (XRD), differential scanning calorimetric (DSC) and thermogravimetric analysis (TGA). The results showed that the melting temperatures (Tm) of CGPA nanocomposites were significantly increased from 289°C to 359°C. The thermal decomposition temperature was improved by ∼13°C with the incorporation of CG nanofiller. CG nanofillers play a significant role in improving the thermal properties of CGPA nanocomposites.
Introduction
In the past years, graphene-based polymer nanocomposite materials have attracted research and commercial interest due to the unique properties of graphene such as mechanical, electrical, thermal and optical properties. Advancements in polymer nanocomposites are rapidly expanding, and the possibility of developing materials with enhanced properties using graphene and its derivatives are particularly of interest.1–4
Graphene is a one-atom thick sheet with two-dimensional (2D) honeycomb structure and novel properties. 5 It has been used as one of the promising nanofiller material to produce graphene-based polymer nanocomposites. The most interesting part of graphene is that it requires a small quantity to enhance the characteristics of a polymer matrix due to its large surface area. 6 Several characteristics such as type and aspect ratio of nanofiller, degree of dispersion, interfacial bonding of nanofiller and polymer matrix affect the thermal performance of resultant polymer nanocomposites. Among these aspects, the degree of dispersion and interfacial interaction between nanofiller and polymer matrix play an important role in the thermal performance of the resultant polymer nanocomposites.7,8 Polyamide (PA) is one of the important class of engineering thermoplastic polymers due to their excellent properties, low cost and ease of processing.9–11 However, PAs have low electrical conductivity, thermal and dimensional stability which limit its applications. 12 Hence, many studies have been started to increase its physical and thermal properties through the reinforcement of nanofiller material to create polymer nanocomposites with promising applications.13-16
Graphene-based PA nanocomposites have been considered as a new type of high-performance material that blends the advantages of both PA and graphene.12,17 These nanocomposites have drawn attention for its significant improvements in the thermal and other physical properties at low-loading content of graphene in PAs.18–20 Many synthesis methods were reported for graphene-based polymer nanocomposites in the literature.3,21–23 Among them, melt blending or melt compounding, in-situ polymerization, and solvent blending or solution mixing were used to prepare graphene-reinforced PA nanocomposites.12,24 The types of synthesis methods for graphene-based PA nanocomposite were important for the performance and reliability of the nanocomposite. Methods such as solvent blending and melt blending can provide uniform dispersion of graphene in PA. Zhang et al. 25 prepared graphene/PA6 composites by melt processing and showed the effect of graphene nanofiller on crystalline temperature of PA6 was increased due to heterogeneous nucleation. Xu et al. 26 produced PA6/PA66/GNP thermal composites by the melt-blending method with improved thermal properties of composite compared to the neat PA6. Roy et al. 27 also improved the thermal stability of PA12/polyethylenimine (PEI)-RGO composites by the melt-mixing process. Li et al. 28 developed PA6/GO-ghindered phenol nanocomposites by the melt-blending method. The nanocomposite showed increase in the thermal degradation temperature. Gong et al. 29 prepared PA6/graphene composites by the solvent-mixing method. Song et al. 30 have prepared PA6/graphene nanocomposites by solution mixing and found the effect on thermal conductive property of PA6/graphene nanocomposites. Evie et al. 31 have also developed PA66/graphene nanoplatelet composite films by the solution-blending technique. Liu et al. 32 developed graphene/CO-PA nanocomposites by the easy scale-up solution-mixing method with improved properties. Therefore, a combination of solution mixing and melt blending is the better way to get good dispersion of graphene in PA matrix to increase the properties of nanocomposite.
In this study, the effect of AD-carboxyl graphene (CG) nanofiller on the properties of PA has been investigated. The delamination/adsorption method33,34 is used to prepare novel CG-reinforced PA nanocomposites (CGPA) which are obtained by intercalation of delaminated dispersion of CG in the PA matrix. The nanocomposites were characterized through structural, morphological and thermal analysis to determine the influence of the CG nanofiller on the thermal properties of the nanocomposites.
Experimental
Materials
2,6-Pyridinedicarboxylic acid; 1,4-phenylene diamine (PPDA); calcium chloride (CaCl2); triphenyl phosphite (TPP) and pyridine were purchased from Sigma-Aldrich, USA. AD-carboxyl graphene (CG) was purchased from Ad-Nano Technologies Pvt. Ltd., Shimoga, India. N-methyl-2-pyrrolidone (NMP), dimethylacetamide (DMAc) and methanol were purchased from Spectrochem Pvt. Ltd., Mumbai, India. All chemicals were used without further purification.
Synthesis of aromatic polyamide
Aromatic polyamide was prepared by the direct polycondensation method. Into a 100-mL round-bottom flask fitted with a magnetic stirring bar, 2,6-pyridinedicarboxylic acid (14 mmol), 1,4-phenylene diamine (14 mmol), calcium chloride (0.5 g, 4.5 mmol), TPP (8.7 mL, 28 mmol), NMP (9 mL) and pyridine (1 mL) were mixed and heated at 120°C in an oil bath for 8 h under N2. Subsequently, the reaction mixture was poured into 150 mL of methanol. The product was collected by filtration and rinsed thoroughly with hot methanol. Finally, the product was dried at 60°C for 24 h inside an oven (Figure 1). Synthesis of polyamide (PA).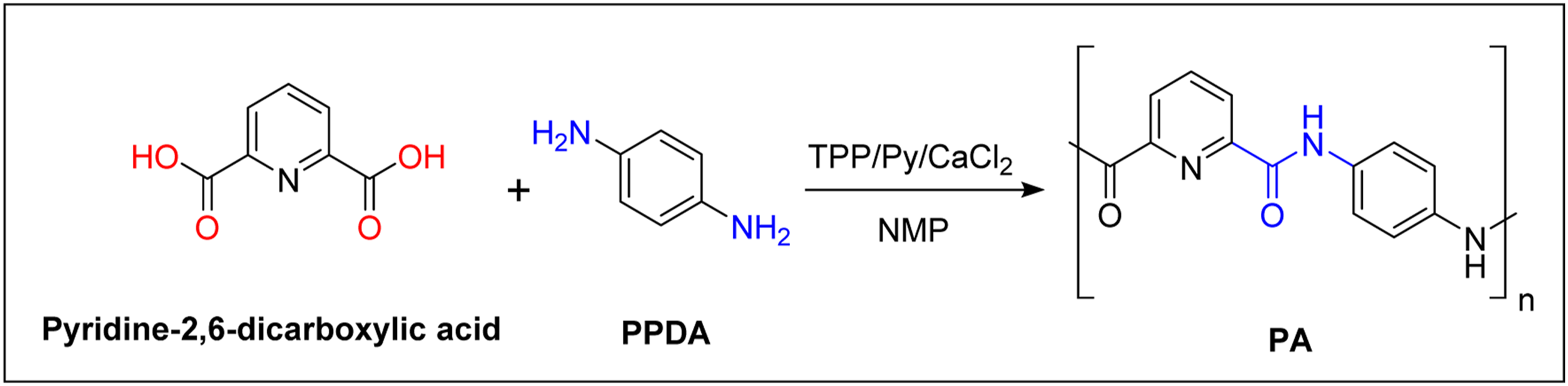
Synthesis of CGPA nanocomposites
According to our previously reported study,
33
the delamination/adsorption method was applied for synthesis of CGPA nanocomposites.34,35 Calculation for CGPA with 1 wt.% of CG, 10 mg of CG was immersed in DMAc and sonicated to form delaminated suspension for 30 min. The suspension was transferred quickly into a 100-mL three-necked round-bottom flask which was preplaced in certain boiling DMAc solution of PA (1 g). The flask was fitted with a magnetic stirrer. The obtained mixture was poured into an empty beaker after 30 min of stirring, then cooled, filtered and washed with acetone to extract DMAc. The resulting CGPA nanocomposite was dried at 60oC for 24 h in an oven. Pristine PA has been treated without adding CG using the same technique as nanocomposites. Corresponding to weight fractions of CG, the composites are named as CGPA1 (1% CG), CGPA3 (3% CG) and CGPA5 (5% CG) (Figure 2). Synthesis of CGPA nanocomposites.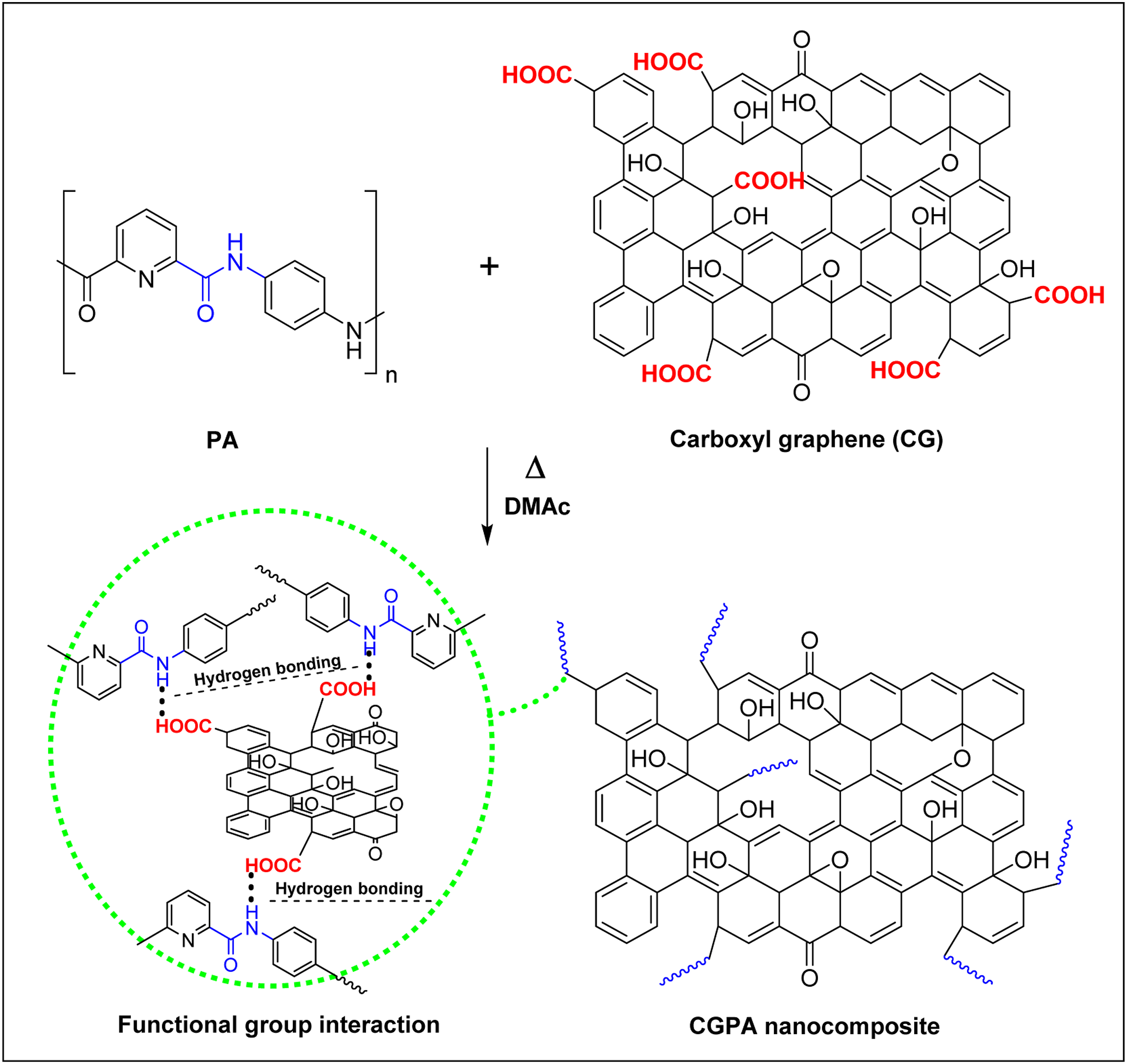
Characterization
IR spectra of composite samples were recorded on the Bruker FTIR spectrophotometer (Bruker, Germany) in the range of 400–4000 cm−1. 1HNMR spectra were recorded on Bruker Avance-II 400 MHz (Bruker, Switzerland). The powder XRD was recorded on XPERT-PRO (PANalytical, Netherlands) using Cu Kα radiation (λ = 1.54060 Å). Diffractograms were obtained by varying the angle from 3° to 40° (2θ) at room temperature. The microstructures were observed using a field emission scanning electron microscope (FESEM; TESCAN, Mira 3-XMU, Tescan Co., Brno, Czech Republic). DSC curves were obtained on a NETZSCH, DSC 204 F1 Phoenix differential scanning calorimeter (NETZSCH-Gerätebau, GmbH). DSC scans were performed from 30°C to 500°C with a heating rate of ± 10 °C/min in a nitrogen atmosphere. The thermogravimetric analysis (TGA) was carried out using NETZSCH STA-2500 thermal analyser (NETZSCH-Gerätebau, GmbH) from 30°C to 600°C with a heating rate of ± 10 °C/min in a nitrogen atmosphere.
Results and Discussion
Morphology and chemical structure of carboxyl graphene, polyamide and CGPA nanocomposites
FTIR spectra of CG, PA and CGPA-x (CGPA1, CGPA3 and CGPA5 nanocomposites) are shown in Figure 3. For PA, the absorption peaks at 3306 cm−1 (N–H stretching), 1546 cm−1 (N–H bending), 1656 cm−1 (C=O stretching), 1513 cm−1 (aromatic C=C stretching), 1320 cm−1 (C–N stretching) and 1404 cm−1 (C=N stretching).36,37 The PA’s formation is confirmed by these vibrations. For CG, the absorption peaks at 1030 cm−1 and 1165 cm−1 (C-O-C stretching), 1391 cm−1 (C=C skeleton vibrations of graphene backbone), 1620 cm−1 and 1721 cm−1 (C=O stretching vibrations of carboxyl and ketone groups), 2850–2950 cm−1 (C-H bonds in CH2 groups) and 3334 cm−1 (O-H stretching vibrations of OH groups). These vibrations showed the presence of oxygen-containing functional groups on the edges of CG nanosheets.
38
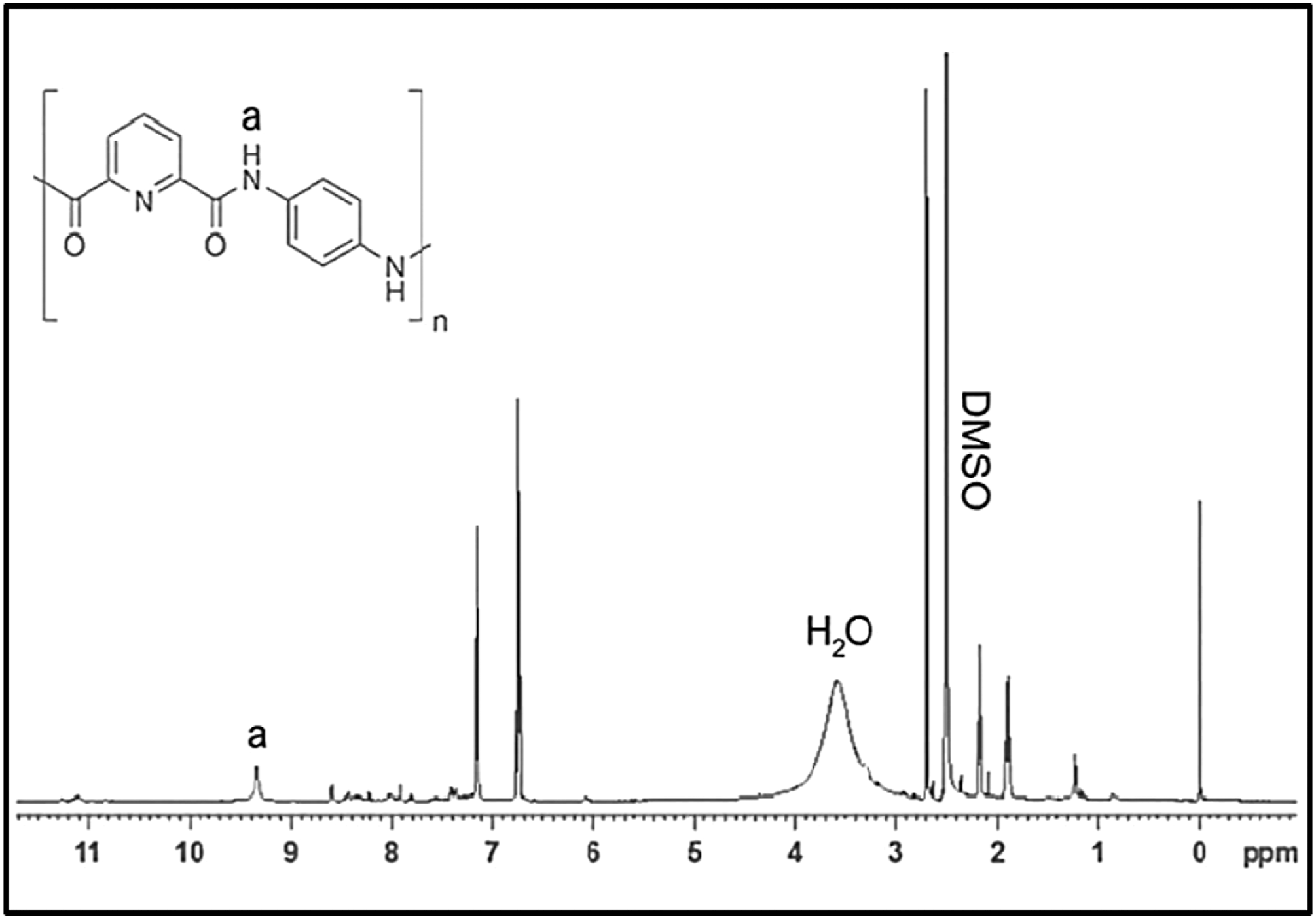
There is no significant impact on the characteristic peaks of PA in CGPA nanocomposites due to addition of CG, but the position and intensity peak of PA varied slightly, that is, the amide II absorption peak located at 1544 cm−1. The 1HNMR spectra of PA are shown in Figure 4. The aromatic protons appeared in the region of 6.5–8.0 ppm. The peak at 9.3 ppm was corresponding to the amide protons (C(=O)–NH–). FTIR spectra of carboxyl graphene, polyamide and CGPA-x. 1HNMR spectra of polyamide.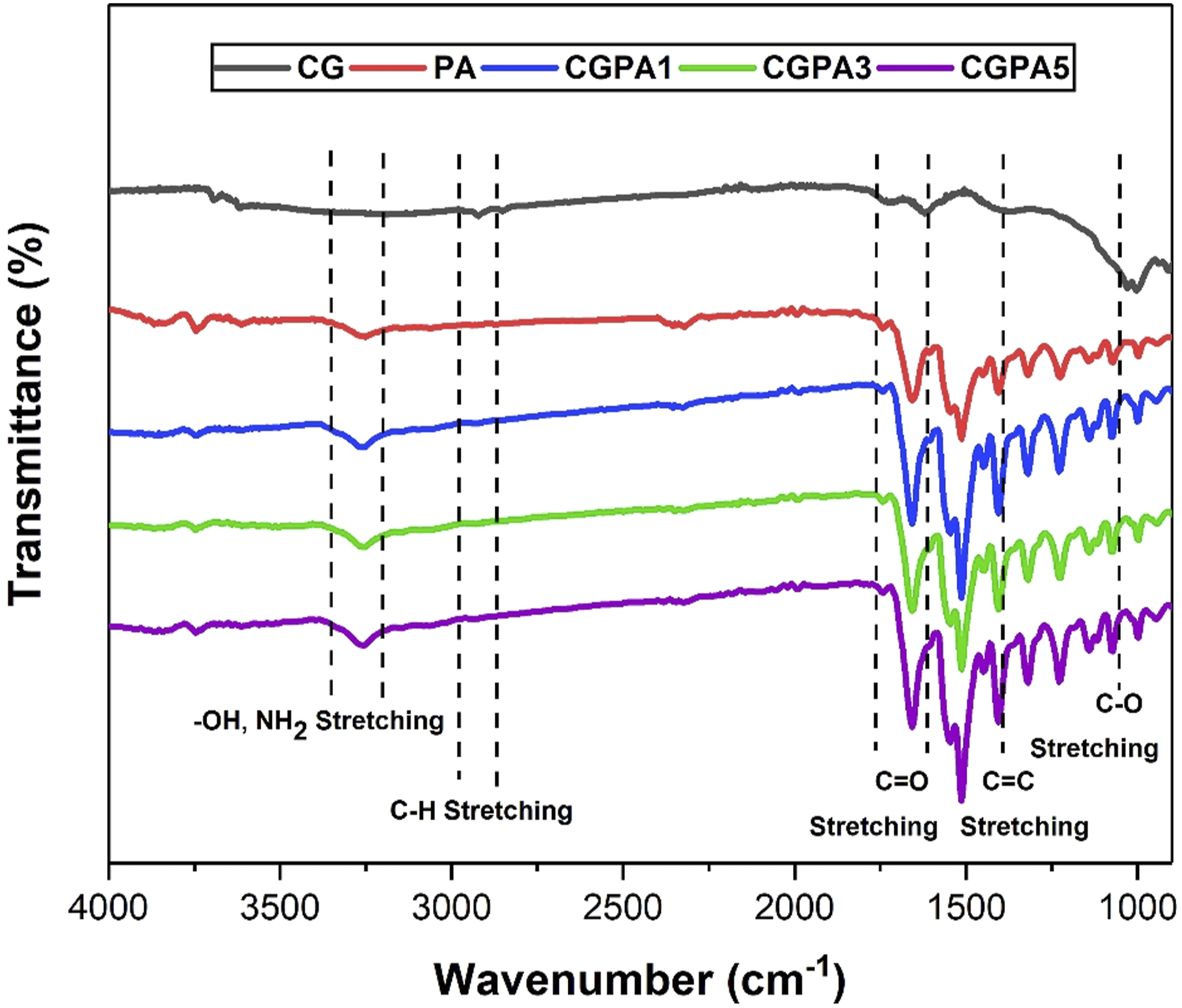
FESEM images of CG and CGPA nanocomposites CG (a), CGPA1 (b), CGPA3 (c) and CGPA5 (d) are shown in Figure 5. The surface morphology shows dispersed CG sheets in CGPA nanocomposites due to the strong binding force and good interfacial interactions between CG and PA.
39
The surface morphology of CGPA nanocomposite is different from that of CG. It was seen that CG showed wrinkled sheet type morphology with dark and bright areas.
33
CGPA nanocomposites show overlapped patterns of CG stacked sheets in PA. The CGPA1 and CGPA3 nanocomposite showed well distributed and exfoliated morphology, and CG sheets were dispersed in PA with no aggregation. However, the CG was less distributed in CGPA5 nanocomposites with large aggregation in PA.33,40 The good dispersion of CG nanoparticles in the PA is one of the main characteristics to boost the physical properties of the PA because the aggregation of the CG will lead to structural defects of the nanocomposites.33,41 FESEM images of carboxyl graphene(CG) and CGPA nanocomposites CG (a), CGPA1 (b), CGPA3 (c) and CGPA5 (d).
The XRD patterns of CG, PA and CGPA-x are shown in Figure 6. Bragg’s condition for diffraction is λ = 2d sinθ where λ = 1.54 Å (wavelength of X-ray), n= 1 (order of diffraction) and d = interatomic distance.
42
The sharp diffraction peak appears at 26.5° (2θ) which corresponds to carboxyl group functionalities and an interlayer spacing of 0.33 nm. These suggest that CG has an amorphous structure. PA is a polymorphic polymer that can produce a typical α crystalline-phase diffraction peak. The PA exhibits α crystalline phase which contains two characteristic diffraction peaks (reflection at 21.2° and 25.5°).
43
The diffraction peaks near 21.28° (2θ) features to the inter-chain hydrogen-bonded (100) plane of amide groups and 25.57° (2θ) showing the overlapping diffraction peaks for (010) and (110) planes of PA.
44
To compare the shifts, the ratio and height of the diffraction peaks were used. The intensity of strong diffraction peak of CG was reduced in the nanocomposite, which shows that the CG layers are stripped into a layer structure and exfoliated in the nanocomposite.
34
The CGPA3 and CGPA5 nanocomposites displayed overlapped CG diffraction peaks at 26.43° (2θ) and 26.49° (2θ), respectively. The intensity of peaks decreased due to the changes in d-spacing.
45
The findings support the existence of two components in CGPA nanocomposites. One is for CG, and the other is for PA. The diffraction peaks of CGPA nanocomposites shifted towards lower 2θ values as the CG wt.% increased, confirming the significant effect on the crystallization behaviours of PA.46,47 XRD patterns of carboxyl graphene, polyamide and CGPA-x.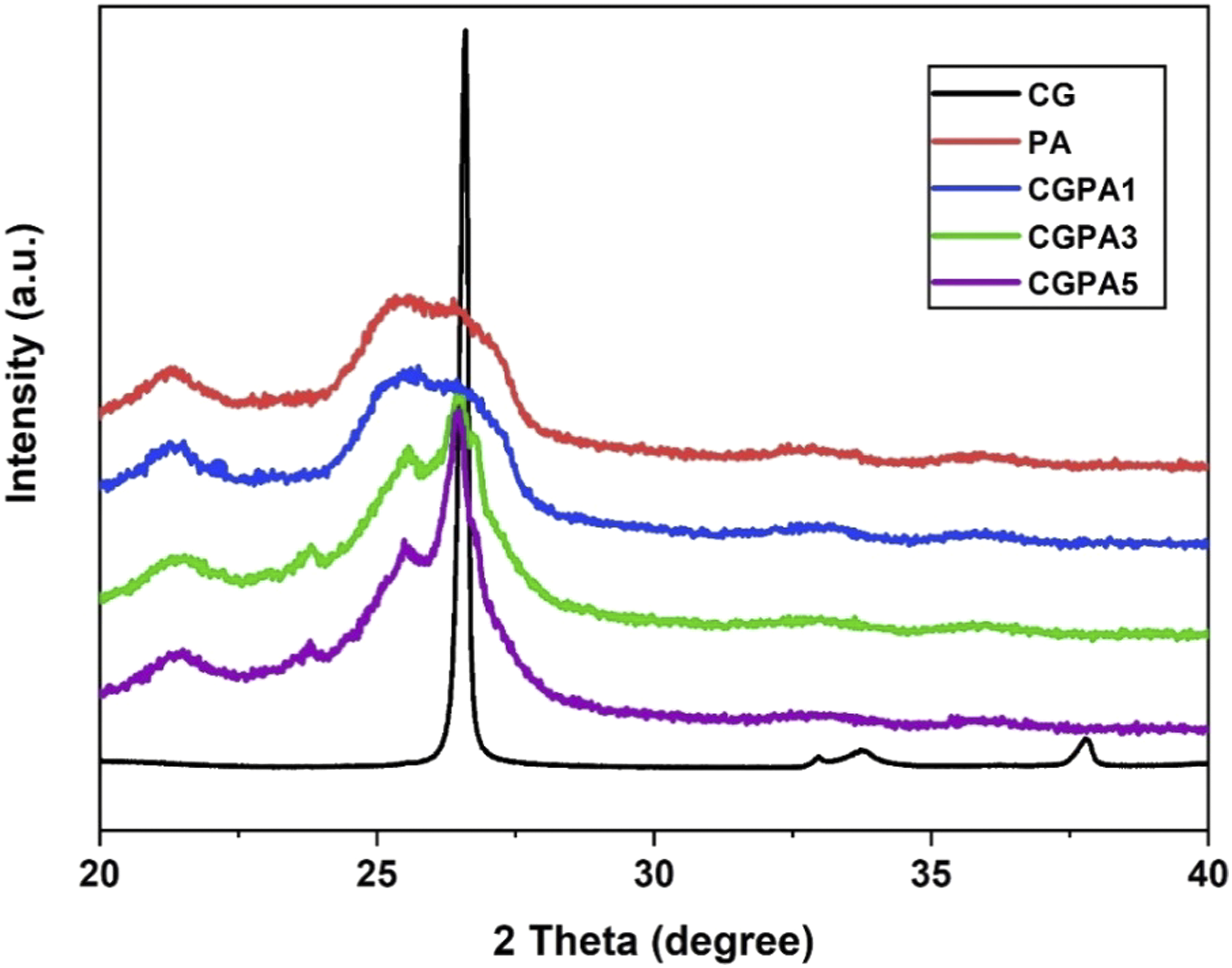
Thermal and crystallization properties of polyamide and CGPA nanocomposites
The DSC results of PA and CGPA nanocomposites are shown in Figure 7. As indicated by the results, the melting temperatures (Tm) of neat PA and CGPA nanocomposites were shifted considerably ranging from 280°C to 360°C. The Tm of PA, CGPA1, CGPA3 and CGPA5 at 289°C, 356°C, 359°C and 354°C, respectively. The Tm of nanocomposites was enhanced because of the strong nucleation effect of CG and better interfacial interactions among CG and PA in nanocomposites.48,49 These results showed that including CG in PA can improve its heat resistance. Other reported nanocomposites also represent similar behaviour as shown in Table 1. The CGPA5 nanocomposite showed lower Tm than CGPA1 and CGPA3 nanocomposites. Excess adding of CG could cause re-aggregation and restacking of CG sheets which leads to less interfacial interactions between CG and PA matrix.49–51 DSC curves of polyamide and CGPA-x. The melting temperature of CGPA nanocomposites.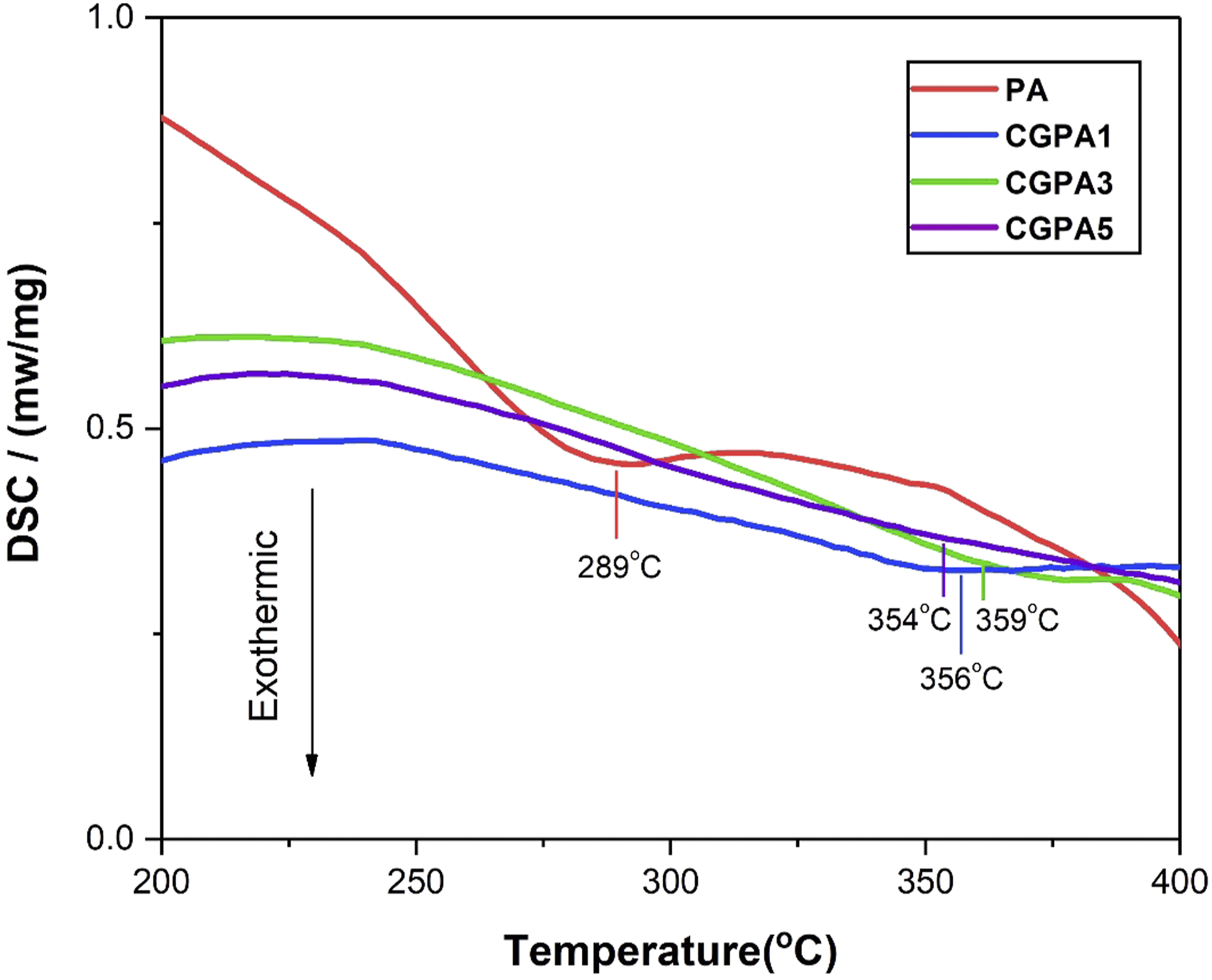
The thermogravimetric (TGA) graph of the PA and CGPA nanocomposites are shown in Figure 8. The TGA analysis was carried out to understand the relationship between mass loss of samples and temperature; the thermal decomposition behaviour of materials. The TGA curve gives the thermal properties of the neat PA and CGPA nanocomposite. The results show the temperature at the 50% of weight loss of PA, CGPA1, CGPA3 and CGPA5 at 549°C, 552°C, 556°C and 562°C, respectively. The decomposition temperature of CGPA nanocomposites is also found higher compared to the neat PA. The TGA thermograms show that the thermal stability of PA increases significantly with the increase of CG wt. % loadings.33,52 It is good to point out that the thermal decomposition temperature is improved by ∼13°C with the incorporation of CG nanofiller. The improved thermal stability of CGPA nanocomposites displays better interfacial bonding between CG and PA matrix.
7
The good interfacial interactions hinder the movement of macromolecular PA chains, and decrease the flexibility of the PA chain, thus increasing the thermal decomposition temperature of the CGPA nanocomposites and hindering its decomposition process.52,53 TGA curves of polyamide and CGPA-x.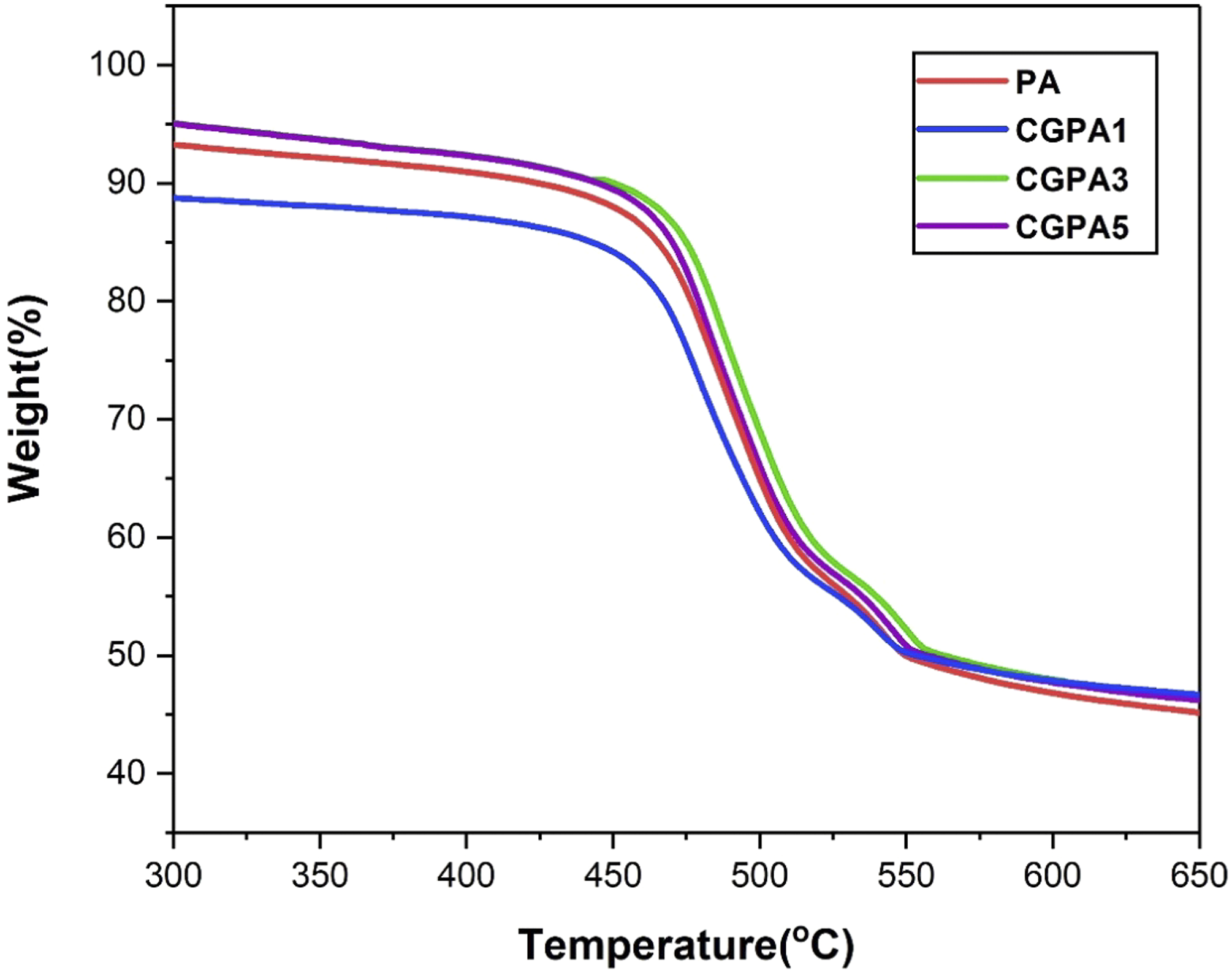
Conclusion
The novel CGPA nanocomposites were successfully synthesized by the delamination/adsorption method. The effects of CG on the structural, morphological and thermal properties of the CGPA nanocomposite were studied. The results showed that the melting temperature (Tm) of PA and CGPA were raised from 289°C to 359°C. The decomposition temperature at the 50% of weight loss of PA was raised from 549°C to 562°C in CGPA nanocomposites, indicating the improved thermal stability of PA matrix by adding of CG. Overall, the thermal analysis results show that CG nanofiller can impact significantly on thermal properties of CGPA nanocomposites. The CG-reinforced PA nanocomposites find promising applications as thermally stable material.
Footnotes
Acknowledgement
The authors are thankfully acknowledging the Department of Chemistry, Gujarat University, Ahmedabad, for providing the essential facilities. The authors are thankful for UGC-Info net & INFLIBNET Gujarat University for providing the resource facilities. The authors also gratefully acknowledge for Department of Biochemistry & Forensic Science, Gujarat University for providing FT-IR facility. SAIF Chandigarh, India, for providing 1H-NMR, SEM and XRD facilities. SAIF, IIT-Madras, Chennai, India for DSC providing facility. CeNS, Bangalore, India, is acknowledged for providing FESEM and TGA characterization facilities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
