Abstract
The visible light–driven ZnS–TiO2/RGO nanocomposites, with good visible light–driven photocatalytic activity, utilizing graphene oxide as precursor, were successfully synthesized via a facile solvothermal thermal method with graphene oxide as precursor. Ti4+ ions were derived from titanium dioxide powder (TiO2, P25) and tetrabutyl titanate. S2− ions and Zn2+ ions were provided by sodium sulfide and zinc acetate, respectively. The photocatalytic and absorbance activity of the nanocomposites was investigated through the photocatalytic degradation of methylene blue in aqueous solution. The results showed that the ZnS–TiO2/RGO nanocomposite prepared by tetrabutyl titanate exhibited better photocatalytic and adsorbance activity for methylene blue under visible light irradiation and its photocatalytic efficiency reached 90% in 60-min light irradiation, almost 1.5 times that of the synthesized ZnS–TiO2/RGO by P25, which was attributed to a cooperative reaction because of an increase of photo-absorption effect of graphene sheets and photocatalytic effect of ZnS nanoparticles. The adsorbance activity for ZnS–TiO2/RGO–tetrabutyl titanate) reached 48%, which was almost 1.7 times higher than that of ZnS–TiO2/RGO-P25 and almost 3.7 times higher than that of ZnS/RGO.
Introduction
At present, environmental problems caused by the undegraded organic pollutions and the harmful compounds such as pesticides and dyes have negative effect on the human being survival and development (Kim and Herrera, 2010; Liu et al., 2016; Sookhakian et al., 2014; Zeng et al., 2014; Zhang et al., 2016). It is difficult to solve these problems by the traditional environment technology. Much more attentions have been paid to the photocatalytic degradation and complete mineralization of undegraded organic compounds in water, soil, and air using the semiconductor photocatalyst method (Hamadanian et al., 2010). Since Fujishima and Honda firstly reported the use of titanium dioxide (TiO2) as photocatalyst to decompose water-producing hydrogen (Fujishima and Honda, 1972; Fujishima et al., 2008), various semiconductors have been developed for photocatalytic degradation of pollutants. Photocatalytic activity of semiconductors depends mainly on the oxidation–reduction potentials of the valence band and the conduction band. The semiconductors with oxidation–reduction potential of the conduction band are more negative, while oxidation–reduction potential of the valence band is more positive own strong oxidation and reduction ability of photogenerated electrons and holes, thus the efficiency of photocatalytic degradation of organic contaminants is greatly improved.
Presently, TiO2 is expected to be one of the most promising materials due to its chemical stability, nontoxicity, low price, and superior photoactivity (Azimi et al., 2016; Hu et al., 2005; Minella et al., 2017; Yanagida et al., 1986). However, in practical application, TiO2 is with two main drawbacks: (1) the light absorption of TiO2 with relatively larger band gap (∼3.2 ev) is limited to shorter wavelength in ultraviolet light which is shorter than 10% of the solar spectrum. (2) Photogenerated electron-hole pairs are easier to be recomposed, reducing the photoelectric conversion efficiency, and then decreasing the photocatalytic efficiency (Khanna and Shetty, 2014; Xiang et al., 2012; Wang et al., 2017). In order to suppress carrier recombination and obtain the visible light photocatalyst materials, a great deal of efforts have been paid to dope with nonmetal elements C, N, S etc. (Cao et al., 2013; Malato et al., 2007) and developed new photocatalyst materials with the absorption spectrum in visible range (400–800 nm) (Asahi et al., 2001).
The development of semiconductor composites with nanometer sizes is one of the most effective approaches to effectively utilize the visible light. For example, the semiconductor composites including (MS/TiO2) (M = Pb, Zn, Cd) exhibited fine optical properties (absorption and photoluminescence (PL)) compared with the normal ones because of the quantum confinement effects (Su et al., 2001). Steng et al. (2008) reported the synthesis of ZnS/TiO2 nanocomposites for the photocatalytic decomposition of Orange II dye. The nanocomposites showed efficient visible-light photocatalytic activity, which is higher than that of a commercially available TiO2 (Degussa P25), pure anatase TiO2, or cubic ZnS nanoparticles. Franco et al. (2009) prepared nanocomposites of (ZnS/TiO2) with different ZnS: TiO2 ratios using a chemical deposition method. The presence of small ZnS percentages on the nanocomposite surface (0.5% and 0.2%) promoted an increase on the catalyst photoactivity compared with the original TiO2. Yu et al. (2006) fabricated ZnS/TiO2 nanocomposites exhibiting enhanced visible-light photocatalytic activity for the aqueous parathion–methyl degradation, which is attributed to the formation of the preassociated complex and the synergetic effect resulting from the combination of the cubic ZnS and anatase TiO2. However, how to further improve the photocatalytic property of this kind of composites is still a great challenge.
Graphene, a two-dimensional single layer of sp2 hybridized carbon atoms with large specific surface area, performs excellent thermal conductivities, high charge carrier mobility, and remarkable mechanical strength and flexibility. It has been a potential candidate to facilitate the adsorption capability and photocatalytic activity of TiO2 and ZnS due to their effective electron transfer and interaction effects (Chakraborty et al., 2016; Qin et al., 2014, 2017b). Photocatalysts with graphene composites have been carried out by using various methods. Zhang et al. (2010) obtained a chemically bonded TiO2 (P25)–graphene nanocomposite photocatalyst with graphene oxide (GO) and P25, using a facile one-step hydrothermal method. Park et al. (2014) reported that ZnS combined graphene/TiO2 (ZGT) composites were prepared by a sol–gel method. Wang et al. (2012) compounded the single crystal TiO2 nanoparticles exposing high energy with graphene by one-step solvothermal method, simultaneously, GO was reduced in the formation of RGO. The prepared composite materials showed enhancing photocatalytic degradation property compared with pure TiO2.
According to our previous work, the nanocomposite prepared with sodium sulfide as the S2− source exhibited much better photocatalytic degradation efficiency than that with thiourea (Qin et al., 2017a). Herein, we proposed a facile and reproducible solvothermal route to obtain ZnS–TiO2/RGO (ZTR) nanocomposites for the first time. In the present study, graphene was added into ZnS–TiO2 nanophotocatalyst in order to improve their photocatalytic activity. The effect of different Ti4+ ions sources on structural, morphological, optical, and photocatalytic properties of the nanocomposites has been figured out. Methylene blue (MB) was selected as a typical dye to evaluate the photocatalytic activity of the nanocomposites under visible light.
Experimental
Materials
Zinc acetate (Zn (Ac)2·2H2O), sodium sulfide (Na2S·9H2O), tetrabutyl titanate (TBOT), MB, ethylene glycol, and absolute ethanol were obtained from Chinese Medicine Group Chemical Reagent Co. Ltd. GO aqueous solution was purchased from Shanxi Coal Research Institute of Chinese Academy of Sciences, and TiO2 powder (P25) was obtained from Degussa Specialty Chemicals Co. Ltd. All of these chemical reagents were of analytical grade and used as received without any further purification, and deionized water was used to wash samples in all experiments.
Preparation of ZTR photocatalysts using P25 (ZTR–P25)
At first, 0.5 mmol of Zn (Ac)2·2H2O was dissolved in 20 mL ethylene glycol ((CH2OH)2) and then 10 mL GO aqueous solution (2mg/L) was added into the solution, forming the light brown solution after sonication for 0.5 h. Next, 0.5 mmol Na2S·9H2O and 0.5 mmol TiO2 powder were dissolved in 20 mL ethylene glycol respectively, following ultrasonic vibration for 0.5 h, and then added into the above-mentioned light brown solution. The mixture was treated by ultrasonic vibration and magnetic stirring for 1 h and transferred into 100 mL Teflon-lined stainless steel autoclave and heated to 180°C for 10 h. The reaction mixture was cooled down to the room temperature and the precipitate was filtered, washed with deionized water and ethanol for several times, and dried in a vacuum oven at 60°C.
Preparation of ZTR photocatalysts using TBOT (ZTR–TBOT)
In a typical reaction procedure, 0.5 mmol Zn(Ac)2·2H2O was dissolved in 20 mL absolute ethanol and 20 mg GO powder was added to the solution. The mixture’s color becomes light brown after sonication for 0.5 h. Then, 0.5 mmol Na2S·9H2O was dissolved in 20 mL absolute ethanol with sonication for 0.5 h. The above two systems were then mixed together and 3 mL TBOT was added into the mixture under the condition of agitation, following ultrasonic vibration and magnetic stirring for 1 h. Then the mixture was transferred into an autoclave. The sealed autoclave was kept at 180°C for 10 h. The obtained precipitates were filtered and washed several times with deionized water and absolute ethanol, then they were dried at 60°C.
Characterization
X-ray power diffraction (XRD) patterns of samples were recorded on D/max 2400 X-ray diffractometer (Neo Confucianism, Japanese) using CuKa radiation source with wavelength of 1.54056 Å to obtain the crystal phases of the composite photocatalysts. Transmission electron microscopy (TEM) images were taken with a F20 microscope (FEI, America) operating at 200 kV, by depositing a drop of sample dispersion onto 300 mesh Cu grids coated with a carbon layer. Raman and PL spectra of samples were taken on a Finder Vista spectrometer (Zolix, China) to observe the chemical structures of the materials. The UV–vis absorption spectrum was recorded with a homemade UV–vis spectrometer.
Photocatalytic tests
The photocatalytic performances of all nanocomposites were evaluated by the degradation curves in a 20 mg/L MB aqueous solution under visible light for different interval times. The experiment of photocatalytic reaction was carried out using a 500 w xenon lamp (λ > 420 nm). In each experiment, the photochemical reactor contained 20 mg ZTR catalysts and 50 mL of 20 mg/L MB aqueous solution. Before starting the illumination, the reaction mixture was stirred for 30 min in dark to reach the adsorption–desorption equilibrium between the MB and the photocatalyst. Three milliliters aliquots were withdrawn from the suspension and then centrifugally separated to essentially remove all the catalyst after a given time interval of irradiation. The concentration of the remnant dye was spectrophotometrically monitored by recording the variation of the absorption band maximum in the UV–vis spectrum of MB (663 nm).
Results and discussion
Characteristics of nanocomposites
Figure 1 shows the XRD patterns of both ZnS/RGO and ZTR nanocomposites. It can be seen that ZnS (Figure 1(a)) reveals three major peaks at 2θ = 28.97°, 48.3°, and 57.1° corresponding to the crystal planes of (111), (220), and (311), respectively, which makes a good agreement with the face-centered cubic phase of ZnS (JCPDS card No. 05–0566). That is, (101), (004), (200), (105), (211), (204), and (116) crystal planes are originated from the anatase TiO2 phase (JCPDS file No. 21–1272). XRD spectrum of the ZTR composites prepared using P25 (Figure 1(b)) shows the characteristic diffraction peaks of ZnS, because of the absorption of Zn2+ ions on the surface of GO and the initial formation of ZnS micronucleus on the surface of RGO; however, TiO2 powder hardly exists in the preparation process with the state of Ti4+ ions due to its indissolubility which leads to a small amount of TiO2 attaching to the RGO surface. While the XRD spectra of the composites prepared using TBOT (Figure 1(c)), clearly shows the diffraction peaks of anatase TiO2 phase. The diffraction peaks of ZnS cannot be observed. These experimental results indicate that TiO2 nanoparticles can be loaded on the RGO surface through hydrogen bonding, and the negative charge characteristics of GO powder dissolved in ethanol is weakened or even disappeared, resulting in a few ZnS particles adhered to the RGO surface.

XRD patterns of ZnS/RGO composites and ZTR ternary composites.
The morphologies of these two nanocomposites were characterized by TEM, as shown in Figure 2. Figure 2(a) shows that the graphene is a homogeneous and smooth sheets. While, Figure 2(b) presents that only TiO2 nanoparticles locate on the graphene sheet and these particles are heavily aggregated in the composites by TBOT because GO powder is not completely dissolved and dispersed in absolute ethanol that results in few ZnS particles adhering to the surface of GO. high resolution transmission electron microscope (HRTEM) images of these two nanocomposites are suggested as shown in Figure 2(c) and (d). The lattice distances of 0.308 nm and 0.352 nm correspond to the interplanar distance of the (111) plane of cubic ZnS phase and the (101) plane of anatase TiO2 phase, respectively.

TEM and HRTEM images of ZTR ternary composites prepared by P25 (a, c) and TBOT (b, d).
Figure 3 displays the Raman spectra of the GO, graphite, and ZTR nanocomposites. The two typical peaks of GO can be found at 1345 cm–1 and 1589 cm–1, which correspond to the D bands assigned to the E2g phonon mode of sp2-bonded carbon atoms and G bands are attributed to the breathing mode of A1g symmetry from phonon interaction near the K zone boundary (Shen et al., 2006). With the reduction of GO, the D bands of ZTR composites shift to lower wave numbers around 1330 cm−1, and the G bands change to higher wave numbers of 1594 cm−1 compared to GO. However, the movement of D and G bands indicates that there is an interaction between semiconductor nanoparticles and graphene. In addition, the disorder degree of composites synthetized by P25 and TBOT can be respectively estimated from the intensity ratio of the D to G bands (ID/IG). As shown in Figure 3(c), the ratio of the ZTR-P25 composite is higher than that of the ZTR–TBOT composite.
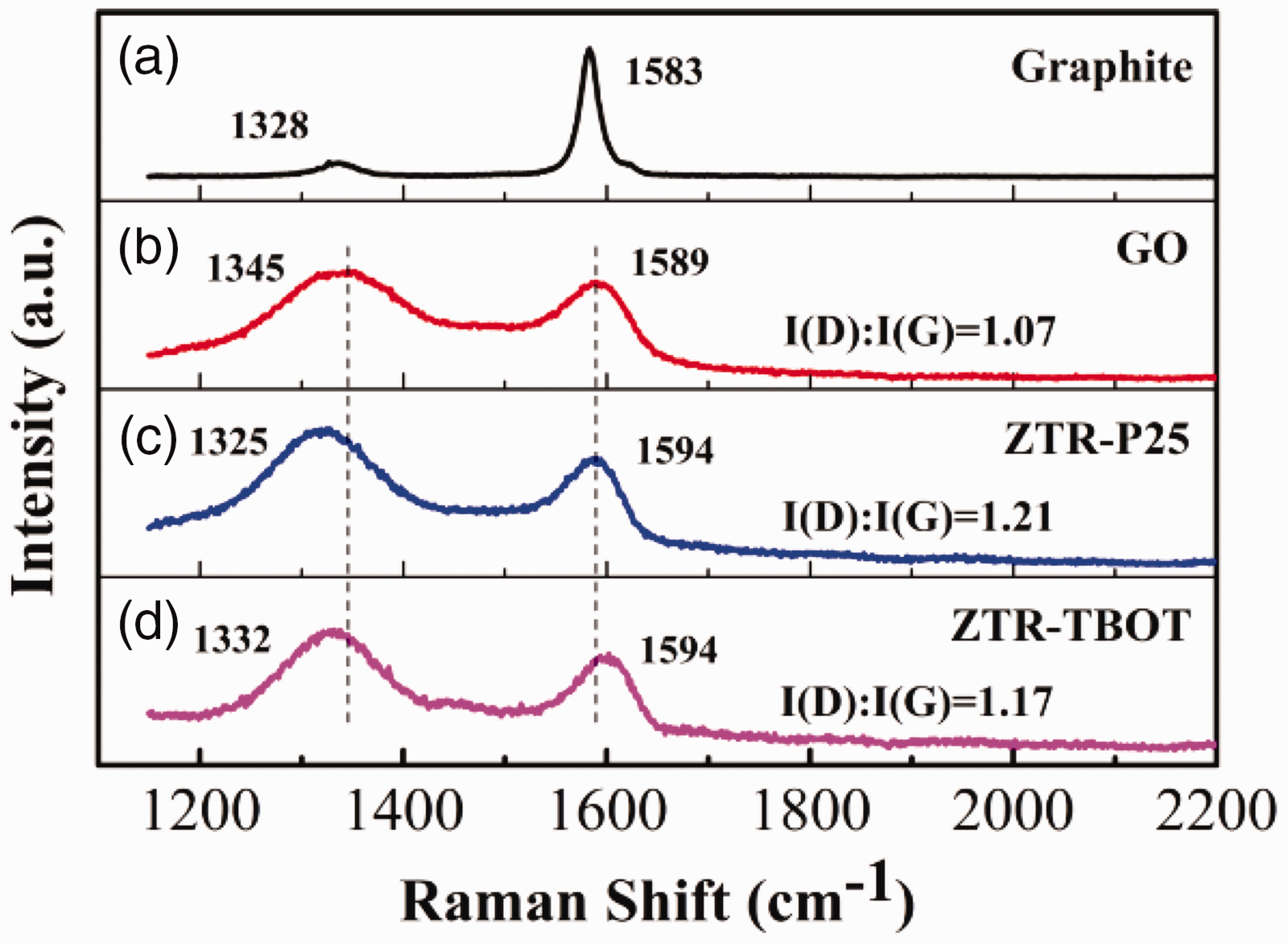
Raman patterns of Graphite (a), GO (b), ZTR-P25 (c), and ZTR-TBOT (d) ternary composites.
Photocatalytic properties of nanocomposites
As shown in Figure 4, PL spectra of the as-prepared samples were investigated with the excitation wavelength of 325 nm. It was reported that the photocatalytic degradation efficiency of materials was related to the efficiency of charge carriers trapping, migration and transfer of the photogenerated electrons and holes (Pant et al., 2016). The PL spectra of ZnS exhibits a sharp green emission peak, which is ascribed to self-activated defect center relating to the presence of Zn vacancies in ZnS/RGO nanoparticles. In Figure 4, it is noticeable that the luminescence efficiency of the ZTR–P25, ZTR–TBOT and ZnS/RGO are all lower than that of bare ZnS. In particular, ZTR–TBOT composite performs the lowest PL intensity, which verifies that the recombination of electron-hole pair is effectively reduced along with the prolongation of lifetime of photogenerated carriers due to the synergistic effect of ZnS and TiO2 on RGO (Pant et al., 2016).

PL spectra for ZTR nanocomposites compared to ZnS and ZnS-RGO.
As shown in Figure 5, the intensity of the absorption peak of MB at 663 nm decreases with the increase of irradiation time due to the photocatalytic activity between ZTR–P25 and ZTR–TBOT, which indicates that the MB molecules are degraded by the catalysis. Furthermore, we compared the photocatalytic and adsorptance performance of ZnS–RGO and ZnS with ZTR on MB degradation (Figure 6). It demonstrates that the ZTR nanocomposites have superior photocatalytic activity, particularly prepared by TBOT. After 120 min of light irradiation, the degradation efficiency of bare ZnS and ZnS/RGO catalysts is about 30% and 38% for MB, whereas the nanocomposites show increased degradation efficiency by about 65% for ZTR–P25 and 90% for ZTR–TBOT. The reason is that the surface of ZTR is larger than that of ZnS and ZnS/RGO. On the other hand, the strong π–π conjugated connection between the benzene rings of pollution and the surface of RGO can also enhance the pollutants adsorption (Nawaz et al., 2017). The adsorbance activity for ZTR–TBOT reached 48%, which is almost 1.7 times higher than that of ZTR–P25 and almost 3.7 times higher than that of ZnS/RGO.

Absorption spectra of MB aqueous solution degraded by ZTR nanocomposites: (a) ZTR–P25 and (b) ZTR–TBOT.

Photocatalytic degradation rates of MB for all catalysts.
The photocatalytic degradation process accompanied with pseudo first-order kinetics and the rate constant can be calculated using well-known first-order rate law equation as follows (Soltani and Entezari, 2013)


Photocatalytic rate constant curves and columns of MB for various catalysts.
Comparative performance of the ZnS–TiO2/RGO (ZTR)-tetrabutyl titanate (TBOT) composite with reported photocatalyst for methylene blue (MB) degradation with reaction parameters.

Based on the above analysis, a potential mechanism for photocatalytic degradation of MB over ternary ZTR nanocomposites under light irradiation is proposed in Figure 8, and the detailed process is expressed by the equations (2) to (6). The formation of electron-hole pairs at the semiconductor surface upon light irradiation (energy higher than that of the band energy of semiconductor) will lead to a series of chain reaction mediated via OH· and O2· generation which further results in the photodegradation of dye or pollutants (Kashinath et al., 2017; Thangavel et al., 2016). When the ZTR photocatalyst is irradiated under visible light, the electrons from the valence band are excited into the conduction band of ZnS, leaving the holes in the valence band, and then are transferred to the conduction band of TiO2. Since the extremely high electron mobility of graphene, the electrons transferred from TiO2 to graphene are captured by O2 to produce O2−. Additionally, photogenerated holes will be left in the valance band of ZnS–graphene, migrating to the surface and oxidizing hydroxyl to produce OH, which makes a major contribution to the MB oxidation. The specific process is as follows (Park et al., 2014)






Mechanism for photodegradation of MB by ZTR nanocomposites under visible light irradiation.
Conclusion
In this work, photocatalytic composites of ZTR prepared by using TiO2 (P25) powder and TBOT as precursors respectively were successfully obtained in a simple solvothermal method to improve the photocatalysis effectively. GO was reduced into RGO during this synthesis process. The photocatalytic and adsorptance results illustrate that (i) The tricomponent composites showed high quantum efficiency across the UV–vis light spectrum compared to both the single and bicomponent constructions, highlighting a synergetic charge separation and efficient charge transport channel; (ii) the ZTR–TBOT with the highest rate constant
Footnotes
Author contributions
YLQ and WWZ contributed equally to this work, and they conceived the idea and designed the experiments. WWZ, ZS, GLS, ZYL, and XYL performed the experiments. YLQ and WWZ wrote the paper under the supervision of DRN and ZYM. All authors participated in the data analysis and discussion.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the support of the program of Liaoning Education Department Nos. LG201605, Key Laboratory Open Fund of Shenyang Li gong University Nos. 4801004yb61-d and 4771004kfs49, and the National Basic Research Program of China under Grant Nos. 2011CB932603 and the CAS/SAFEA International Partnership Program for Creative Research Teams.
