Abstract
This work studied the properties of poly(lactic acid) (PLA) and chemically crosslinked poly(lactic acid) (CrPLA) in the presence of different types of silica (SiO2): SiO2 from rice husk ash (RhSiO2) and commercially available SiO2 (CoSiO2). A mixture of triallyl isocyanate and dicumyl peroxide was used as a chemical crosslinking agent. The rice husk was burnt in a furnace to extract RhSiO2. The thermal and mechanical properties of the PLA and CrPLA composites were investigated as a function of the SiO2 content. The presence of C–H functional groups supports the compatibility of stearic acid and RhSiO2, thereby facilitating the esterification reaction observed in the Fourier transform infrared-attenuated total reflectance spectra. This played an important role in changing the properties of the PLA-based composites. The concave fracture surfaces were consistent with the tensile results, the thermal properties, and the degree of swelling, which suggested that there was a better reaction between stearic acid and RhSiO2. However, the heat deflection temperatures of both the PLA and the CrPLA composites were not significantly affected by blending with either CoSiO2 or RhSiO2.
Introduction
Recently, international attention on and demand for biodegradable polymers has been increasing, due to their potential to replace conventional polymers. Most of the plastics that are used in many industries are derived from petroleum-based resources and are not environmentally friendly because they are not biodegradable. The huge demand for plastics and issues with degradability are leading to problems with plastic pollution and management. This promotes biodegradable polymers as new solutions for greener and more economical industries. Among many candidates for biodegradable polymers such as poly(butylene succinate), poly(ethylene succinate), poly(hydroxyl-alkanoate), and thermoplastic starch, poly(lactic acid) (PLA) is considered to be one of the most promising because of its processability, biocompatibility, renewability, and good mechanical properties. As a result, PLA can be used for many applications. 1 –3 Despite its potential, some poor properties of PLA, especially its low heat deflection temperature (HDT), have hindered its adoption in many applications and this issue needs to be addressed. Many approaches have been employed to overcome this limitation, including creating polymer composites, 4 –6 forming crosslinked structures, 7 and chain extension. 8
There are various types of fillers that are used to create polymer composites, such as wood, 9,10 clay, 11,12 and silica (SiO2). 4 However, SiO2 is widely available, easily obtained as a reinforcing filler and can be used to improve the properties of polymers. SiO2 can be obtained from natural resources, for example, rice husk and fly ash. Using the controlled burning of rice husk, amorphous SiO2 with a large surface area can be obtained in approximately 20 wt%. 13 Amorphous SiO2 has been found to act as a nucleating agent for thermoplastics at a very low load. 14 –16 Wu et al. 17 demonstrated that the addition of 1.5 wt% of SiO2 increased the HDT of SiO2/PLA to 25°C. The presence of a crosslinked structure has been shown to improve the thermal stability of PLA. 18 –20 Radiated PLA mixed with 3 wt% triallyl isocyanate (TAIC) showed improved heat stability and still retained its original shape at over 200°C. 21 However, the radiation cost is high. Thus, the incorporation of chemical crosslinking agents is an alternative and a more cost-effective method for generating crosslinked structures. It has been reported that the gel fraction of PLA reached 100% when dicumyl peroxide (DCP) was used at a very high concentrations. 22 Moreover, a mixture of DCP and TAIC increased the thermal stability, Young’s modulus (YS), and tensile strength (TS) of PLA compounds. 23
As has been previously mentioned, several approaches have been employed to enhance the thermal properties of PLA. However, improving the thermal properties of PLA is still a topic of interest. The focus of this study is the effect of a combination of chemical crosslinking agents with SiO2 on the thermal and mechanical behavior of PLA based-composites. SiO2 extracted from rice husk and a commercial grade SiO2 were used as inorganic filler sources. A mixture of TAIC and DCP was selected as a chemical crosslinking agent. The effects of the SiO2 concentrations blended with PLA and crosslinked poly(lactic acid) (CrPLA) on the mechanical and thermal properties were determined. The degree of swelling, differential scanning calorimetry (DSC), and scanning electron microscopy (SEM) were performed to examine the physical, thermal, and morphological properties of the PLA and CrPLA composites.
Experimental
Materials
A commercial PLA (PLA 2003D, NatureWorks, Minnetonka, MN, USA) was used in this work. The chemical crosslinking agents TAIC and DCP were supplied by PI industry Co., Ltd (Bangkok, Thailand). Commercial synthetic precipitated SiO2 and stearic acid were used as an inorganic filler and surface treatment agent, respectively.
SiO2 preparation
Rice husk was first rinsed with tap water three times, then 100 grams of the husk was soaked in 1 liter of a 2 M HCl solution for 4 h. The husk was washed with tap water until its pH was neutral and dried overnight at 100°C in a hot air oven. To extract the SiO2, the dried husk was burned in a furnace at 800°C for 6 h. Afterward, two types of SiO2, the husk ashes (RhSiO2) and the commercial SiO2 (CoSiO2), were ground in a ball mill for 12 h to obtain SiO2 with a particle size under 75 µm. To improve the compatibility of SiO2 and PLA, both types of SiO2 were surface-modified. First, stearic acid (2 wt% of SiO2) was dissolved in isopropyl alcohol and then magnetically stirred at 35°C for 1 h. Afterward, the SiO2 was mixed with the stearic acid solution for 4 h using a magnetic stirrer. Finally, the surface-modified SiO2 was dried in a hot air oven at 80°C before use.
Sample preparation
PLA, TAIC, DCP, and SiO2 (RhSiO2 or CoSiO2) were mixed in an internal mixer (MX500-TQ, Chareon Tut, Co., Ltd, Thailand) at 190°C for 10 min using a 50 r/min rotor speed. The PLA compounds were hot pressed using a compression molder (PR2D, Chareon Tut, Co., Ltd, Thailand) at 190°C for 5 min to form testing samples. The composite formulations, including their five chemical compositions, are given in Table 1.
Composite formulations.
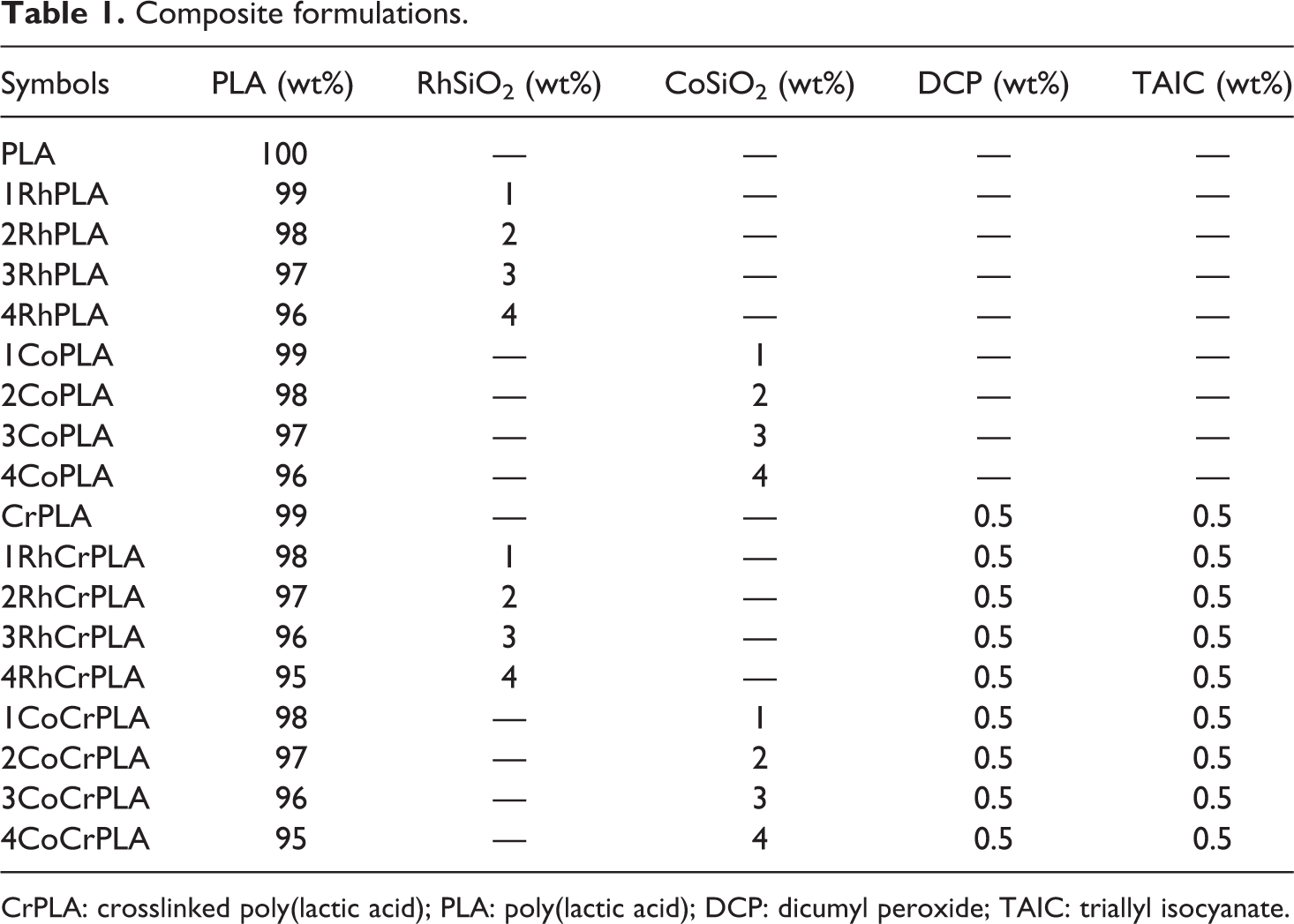
CrPLA: crosslinked poly(lactic acid); PLA: poly(lactic acid); DCP: dicumyl peroxide; TAIC: triallyl isocyanate.
Mechanical properties
The mechanical properties of PLA, CrPLA, and their composites were determined according to the ASTM D638 (2008) standard using a universal testing machine (M350-10CT, Testometric, Co., Ltd, UK) with a crosshead speed of 5 mm/min at room temperature.
Fourier transform infrared spectroscopy
Fourier transform infrared-attenuated total reflectance (FTIR-ATR) spectra were obtained using a Spectrum Spotlight 300 instrument (PerkinElmer, Inc., Waltham, MA, USA) using the ATR mode in the range of 4000–400 cm–1. The FTIR-ATR was performed to observe the effect of the SiO2 surface treatment with stearic acid.
Degree of swelling
The CrPLA and CrPLA composites were immersed in chloroform at room temperature for 48 h. Then the degree of swelling was calculated using equation (1)
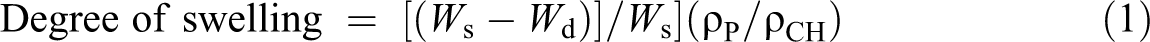
where W s is the weight of the sample after immersion in chloroform for 48 h, W d is the weight of the dry gel component after immersion in chloroform for 48 h, and ρP and ρCH are the densities of PLA and chloroform, respectively. The tests were carried out at room temperature.
Analysis of the fractured surface
The fracture surface morphologies of PLA, 4RhPLA, 4CoPLA, CoPLA, 4RhCoPLA, and 4CoCrPLA were studied by SEM (SEM; Zeiss LEO 1450 VP, Germany) using the secondary electron mode and an acceleration voltage of 10 kV under vacuum. All the samples were coated with gold before the experiment.
Thermogravimetric analysis
The thermal stabilities of PLA and CrPLA filled with RhSiO2 and CoSiO2 were determined using a thermogravimetric analyzer (TGA 55, TA instrument, Inc., New Castle, DE, USA). The temperature was increased from 30°C to 700°C at a heating rate of 20°C/min under a nitrogen atmosphere.
Heat deflection temperature
The HDT was determined in compliance with ASTM D648 using a distortion temperature tester (Ceast 6510, Torino, Italy). The rectangular sample was tested by immersing it in silicone oil under an applied constant flexural load of 0.46 MPa. The temperature of the oil medium was increased at 2°C/min. The HDT was recorded when the tested sample was deflected by 0.25 mm.
Differential scanning calorimetry
DSC (DSC822, Mettler-Toledo, Columbus, OH, USA) was used to study the thermal properties and degree of crystallinity (X c) of PLA, CrPLA, and their composites. The temperature was increased from 30 to 200°C, cooled down to 30°C, and then increased to 200°C at a rate of 10°C/min under a nitrogen atmosphere.
Results and discussion
FTIR-ATR analysis
Figure 1(a) to (d) shows the FTIR-ATR spectra of the samples before and after surface modification. It can be seen that the FTIR spectrum of CoSiO2 is similar to that of RhSiO2 (Figure 1(a) to (b)). The peaks at 3500 cm–1, 1100 cm–1, 950 cm–1, and 470 cm–1 were assigned to the hydroxyl (–OH), siloxane (Si–O–Si), silanol (Si–OH), and Si–O–Si bending modes, 24 respectively. However, an additional C–H stretching peak in RhSiO2 appeared at 2950–2850 cm–1 (dash circle). After the SiO2 surface modification with stearic acid in an alcohol solution, it was expected that the spectra of CoSiO2 and RhSiO2 would have similar peaks corresponding to –OH, –COOH, Si–O–Si, Si–OH, and Si–O–Si blending. Nevertheless, a slight difference was observed in the silanol functional (Si–OH) peak located at approximately 950 cm–1 (Figure 1(c) to (d)). The Si–OH peak for RhSiO2 decreases significantly while the peak for CoSiO2 is still present. It is known that a large number of hydroxyl groups (–OH) on the SiO2 surface is important for the surface modification, which involves the esterification reaction of the carboxylic group (–COOH) of stearic acid with a surface –OH group, that is, the surface modification of fibers (rich in –OH) with stearic acid in an alcohol solution. 25 A possible source of this difference is the C–H groups that are found only in RhSiO2 (Figure 1(b)). The C–H groups could function as facilitators of the interaction between stearic acid and RhSiO2, assisting the esterification reaction on the RhSiO2 surface. Therefore, the change in the Si–OH peak of RhSiO2 confirmed that a chemical reaction occurred between the –COOH group of stearic acid and the –OH group of RhSiO2, as proposed in Figure 2.
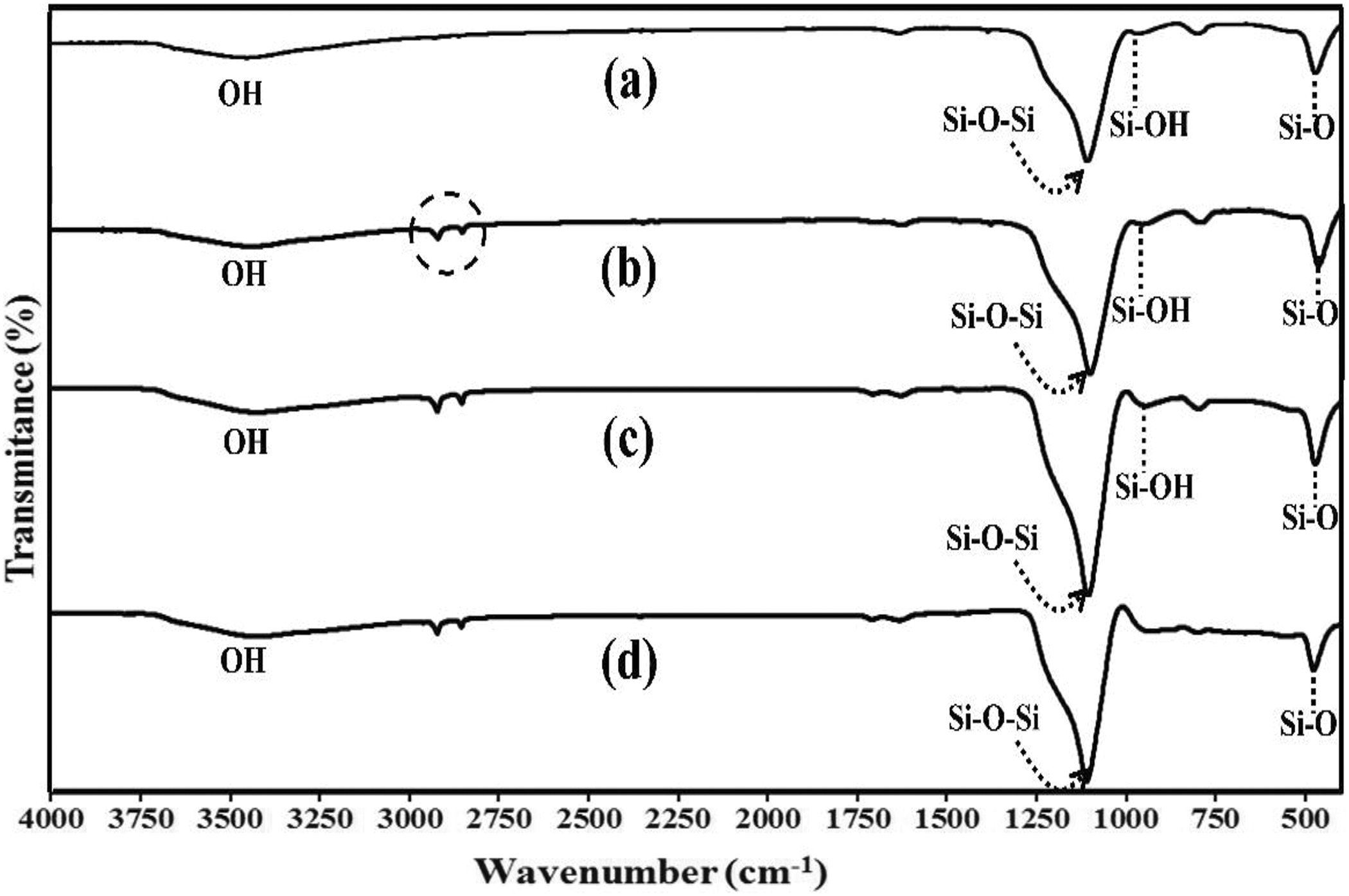
FTIR-ATR spectra of SiO2 before and after surface modification with stearic acid: (a) CoSiO2; (b) RhSiO2; (c) treated CoSiO2; and (d) treated RhSiO2.
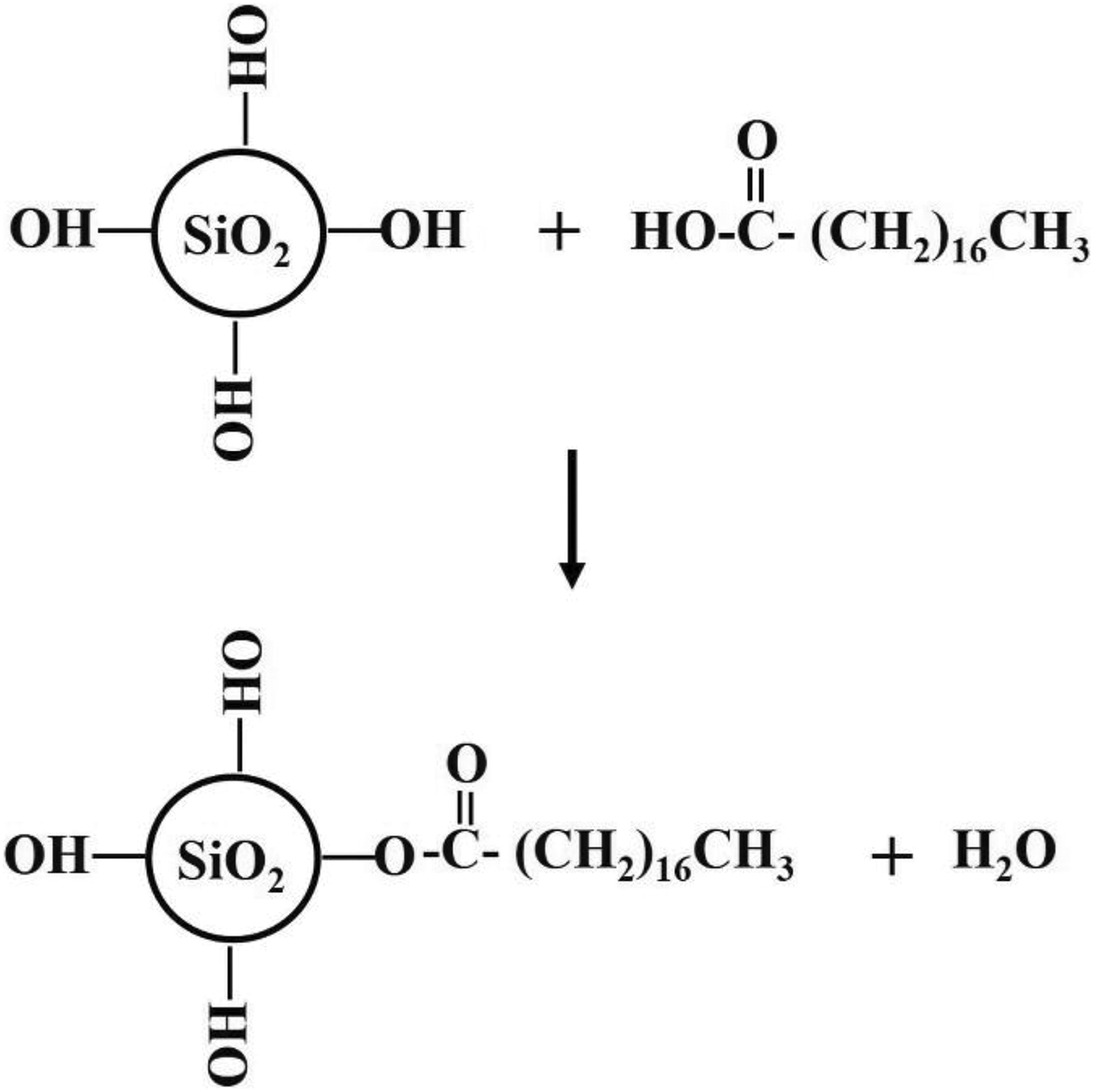
The proposed reaction mechanism for the SiO2 surface modification.
Degree of swelling
The swelling properties of CrPLA are related to the crosslinking density. A possible crosslinking mechanism for CrPLA using TAIC in the presence of DCP have been given by Yang et al. 23 The degrees of swelling for CrPLA containing different concentrations of CoSiO2 and RhSiO2 are shown in Figure 3. The swelling level of the CrPLA composites increased with increasing SiO2 (CoSiO2 and RhSiO2) content. The swelling level of CrPLA is defined as the amount of absorbed chloroform inside the crosslinking points. If the crosslinking density is higher, fewer spaces are available between the crosslinking points to contain chloroform. Therefore, an increasing degree of swelling as a function of SiO2 loading suggests lower crosslinking density in the CrPLA composites. One possible reason for this is that since TAIC and SiO2 are polar molecules, the liquid polar TAIC may be absorbed on the hydrophilic SiO2 surface during the mixing step, affecting the increase in the degree of swelling of the CrPLA composites. However, the swelling observed due to the CoSiO2 was more pronounced. This may be due to the higher hydrophilicity of CoSiO2, due to its lower chemical reactivity with stearic acid (FTIR results in Figure 1(a) to (d)), which facilitates greater absorption of TAIC on the CoSiO2 surface.
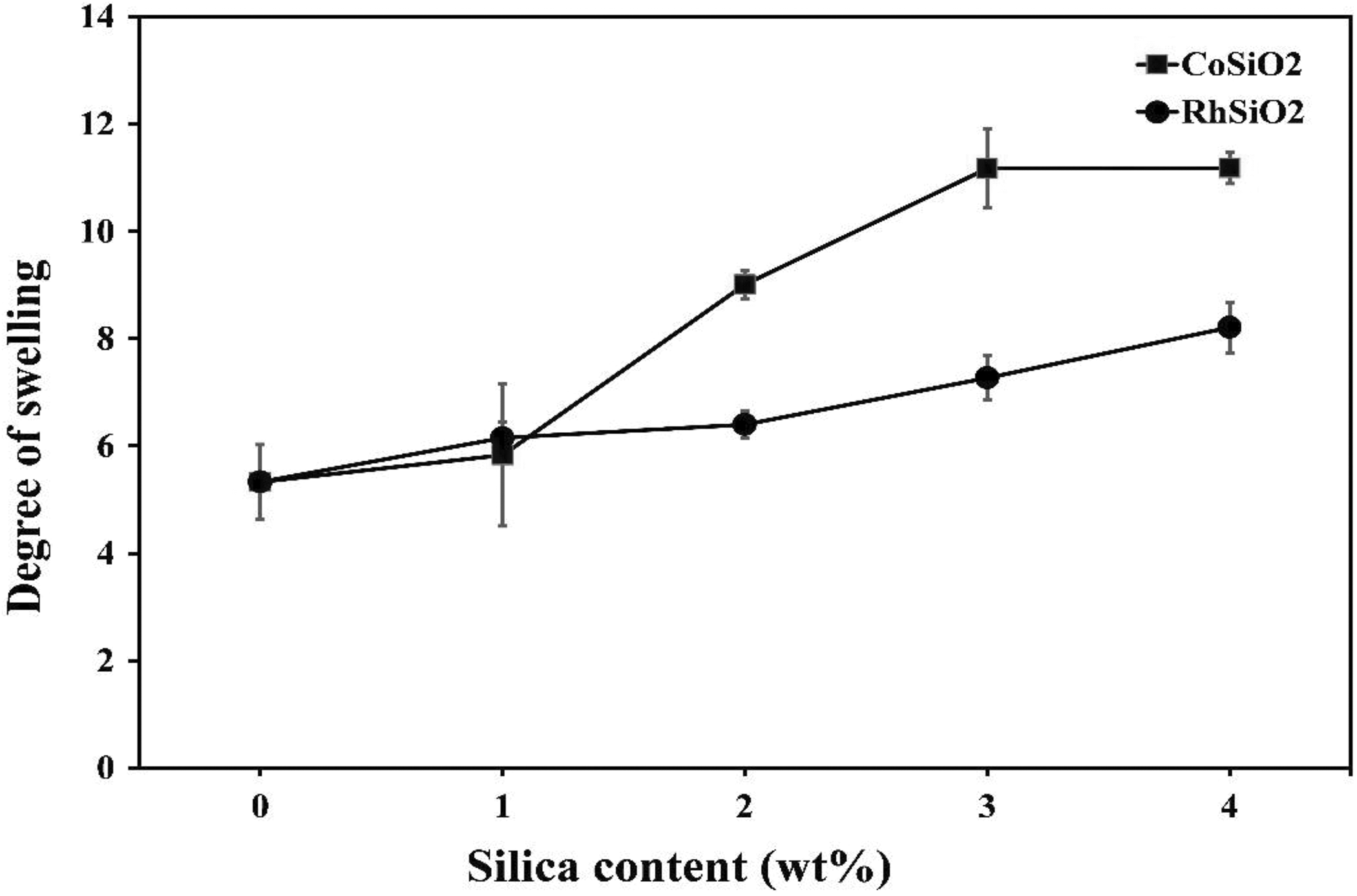
The degree of swelling of CrPLA as a function of CoSiO2 and RhSiO2 loading.
Mechanical properties and SEM observations
The mechanical properties of PLA and CrPLA with different SiO2 contents are shown in Figure 4 (a) to (c). The TS of the PLA and CrPLA (Figure 4(a)) decreased with increasing RhSiO2 content. The TS of PLA and CrPLA decreased from 58.1 MPa and 56.0 MPa to 51.8 MPa and 44.9 MPa, respectively, with 4 wt% of RhSiO2. The effect was more pronounced for the samples loaded with CoSiO2. For example, the TS of PLA and CrPLA dropped from 58.1 MPa and 56.0 MPa to 38.8 MPa and 40.6 MPa, respectively, when 4 wt% CoSiO2 was loaded. The obvious decrease on the TS for the samples loaded with CoSiO2 may be the result of poor bonding interactions between the CoSiO2 and the PLA matrix. 26 The elongation at break (EB) of PLA and CrPLA containing SiO2 showed similar behavior that observed in the TS, namely a decrease in the EB as a function of the SiO2 content. In addition, a significant drop in the EB was observed for the samples composed with CoSiO2. The EB of PLA and CrPLA decreased from 3.3% and 2.8% to 1.6% and 1.6%, respectively, with the addition of 4 wt% CoSiO2, corresponding to a 51.51% and 42.86% decrease in the EB compared to the unloaded samples (Figure 4(b)). The effects of SiO2 on the YS of the PLA composites are given in Figure 4(c). It can be observed that the addition of SiO2 did not affect the YS of the crosslinked samples significantly, indicating that the crosslinking is the main factor in determining the YS and not the SiO2 addition. For the noncrosslinking samples, however, a low dose of RhSiO2 (1 wt%) shows a greater YS than neat PLA, but when the dose of RhSiO2 is greater than 1 wt% the YS began to decrease. This is due to the effective stearic acid surface modification as discussed in FTIR-ATR results. The higher the dose of RhSiO2, the greater the amount of stearic acid on the SiO2 surface. This leads to the drop in the YS of the PLA containing RhSiO2. The changes in the tensile properties due to the two sources of SiO2 indicate that RhSiO2 more efficiently bonds to the PLA and CrPLA matrix than does CoSiO2, which is also revealed by the SEM image analysis. Figure 5(a) to (f) shows the fracture surface morphologies of the PLA, CrPLA, and their composites. Two different types of fracture surface can be clearly seen: (i) flake-like morphologies and (ii) concave morphologies. The flake-like morphology of PLA, 4CoPLA, CrPLA, and 4CoCrPLA demonstrates brittle fracture behavior, while all the samples composited with RhSiO2 show concave fracture patterns, suggesting less brittleness in the materials. The SEM fracture surfaces were consistent with the tensile properties, suggesting better bonding of RhSiO2 onto the PLA and CrPLA matrix as a result of a better reaction between stearic acid and RhSiO2, as discussed in the FTIR results.
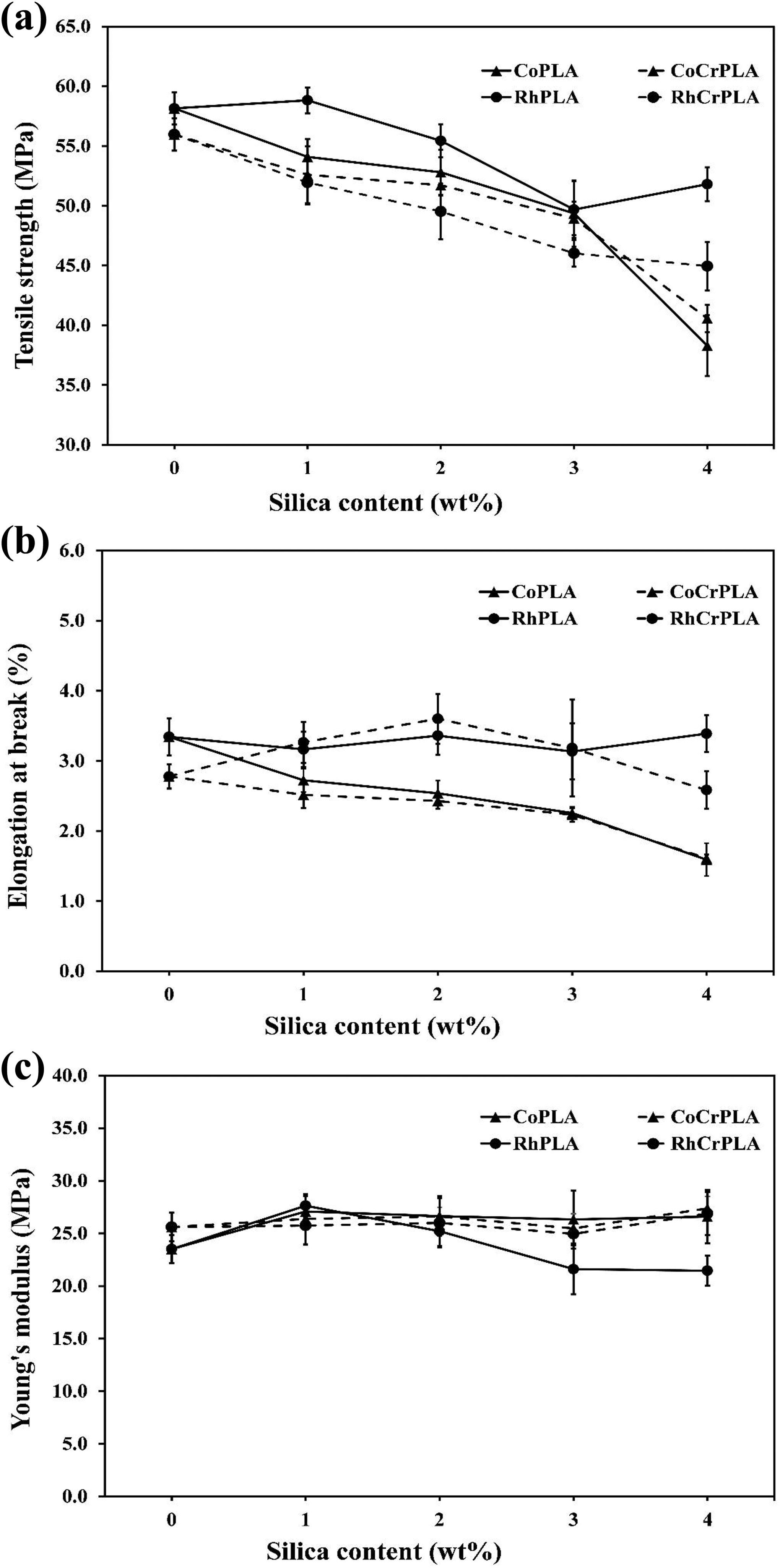
The mechanical properties of PLA and CrPLA with different SiO2 loading: (a) TS; (b) EB; (c) Young’s modulus.

Fracture surface morphologies: (a) PLA; (b) 4CoPLA; (c) 4RhPLA; (d) CrPLA; (e) 4CoCrPLA; (f) 4RhCrPLA.
Thermogravimetric analysis
Figure 6 shows the thermal degradation behavior of PLA and CrPLA with and without 1 wt% SiO2 studied by thermogravimetric analysis (TGA) under a nitrogen atmosphere. The TGA curves show that all the samples decreased in weight until the temperature reached 350°C. Table 2 shows the degradation temperatures for the loss of 10 wt% (T 10) and 50 wt% (T 50). The use of the two types of SiO2 increased the thermal stability of the PLA and CrPLA composites. The T 50 value of PLA increased from 321°C to 339°C and 342°C when 1 wt% of CoSiO2 and RhSiO2, respectively, were added. Furthermore, the use of a chemical crosslinking agent also improved the degradation temperatures of the PLA-based samples, as shown by the increase in the T 50 for CrPLA, 1CoCrPLA, and 1RhCrPLA. However, the effect of the addition of RhSiO2 on the thermal resistance of the CrPLA composites was obviously greater than observed for CrPLA containing CoSiO2. Thus, the RhSiO2 is a more effective filler for improving the thermal stability of PLA and CrPLA.
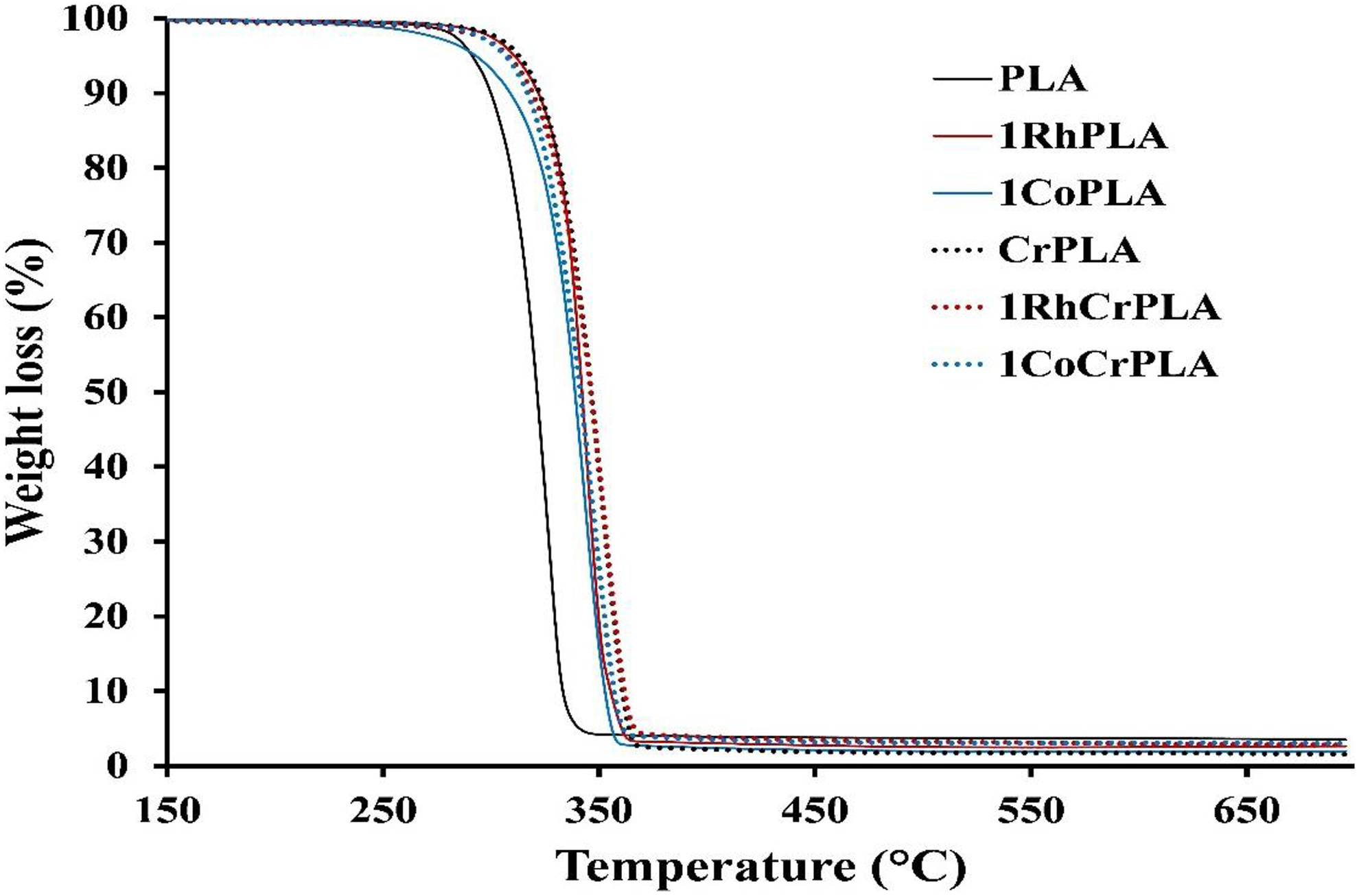
TGA curves for PLA, CrPLA and their composites.
Degradation temperature at 10% and 50% weight loss for PLA, CrPLA, and their composites.
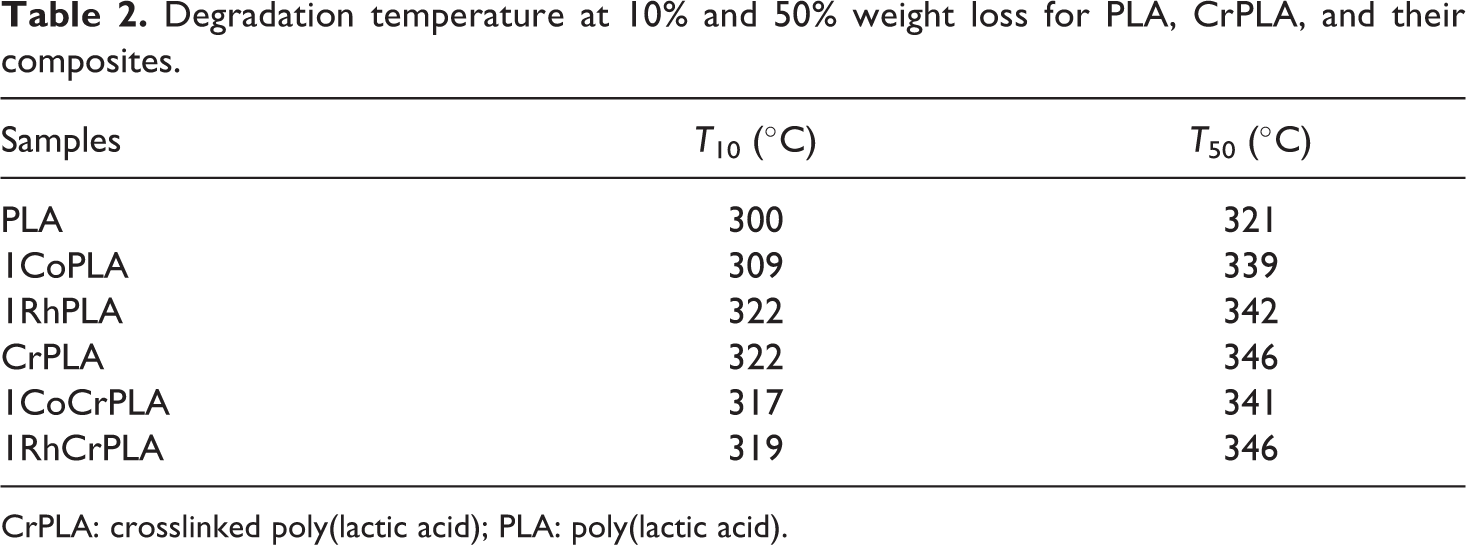
CrPLA: crosslinked poly(lactic acid); PLA: poly(lactic acid).
Differential scanning calorimetry
The thermal behavior of PLA and CrPLA with and without SiO2 was studied using DSC as shown in Figure 7. The second heating cycle was studied in order to erase the prior thermal history of the samples. For the noncrosslinking system, it can be seen that the PLA/RhSiO2 composite exhibited two melting peaks at 148°C and 154°C. This could be attributed to the addition of RhSiO2, which could lead to two different crystalline structures (e.g. a difference in the lamellar thickness or spherulite dimensions). 27 As a result, an imperfect crystal was formed during the heating process. Thus, the second T m peak represents the melting temperature of crystals formed during recrystallization due to the presence of RhSiO2. These two melting points may be related to an increase in the crystallinity of the PLA from 29.08% to 32.02% when 1 wt% of RhSiO2 was added. A similar result was also observed by Pilla et al. 28 who showed that the presence of two melting peaks only occurred for PLA composites with surface modified fibers. For the crosslinked systems, a single melting peak and broad cold crystallization were found for the CrPLA and CrPLA composites. The broad cold crystallization of the chemically crosslinked samples may be due to large chain networks, created by the chemical crosslinking reaction, restricting chain mobility during the recrystallization process, 23 which causes a decrease in the crystallinity of the PLA from 29.08% to 16.59%. The changes in the cold crystallization characteristics of CrPLA have been previously reported in the literature. 19,23,29 However, the incorporation of SiO2 was found to increase the crystallinity of CrPLA, especially for the CrPLA/CoSiO2 composite, for which the crystallinity increased to 19.14%. This can be attributed to fewer crosslinks in the CrPLA/CoSiO2 composite (a greater degree of swelling) associated with the recrystallization process.
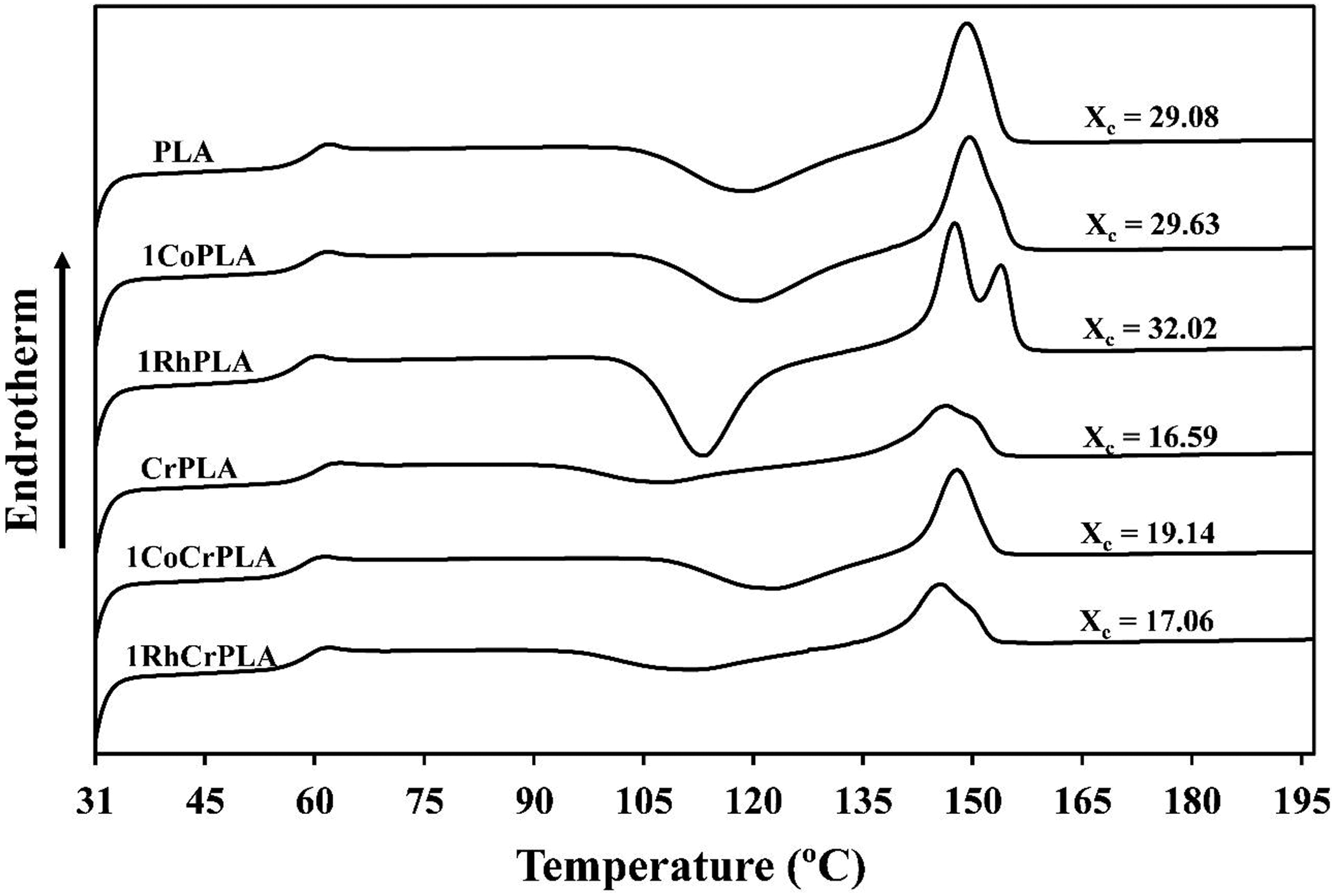
DSC thermograms of PLA, CrPLA and their composites.
Heat deflection temperature
Table 3 shows the HDTs of the PLA and CrPLA composites for different quantities of SiO2. In general, the HDT of PLA was similar to its T g. 30,31 In this work, the HDT of PLA and CrPLA were found to be approximately 56.6°C and 55.5°C, respectively. The incorporation of RhSiO2 and CoSiO2 at 1–4 wt% slightly affected the HDT. A previous study has shown that the HDT can be influenced by an increase in the crystallinity. 32 Thus, this work has shown that the effect of the quantity and type of SiO2 used on the crystallinity did not improve the HDT. However, it can be noted that the chemically crosslinked samples showed a slight decrease in the HDT values, which is in agreement with the decrease in crystallinity. This suggests that the packed crystalline structure of the PLA chain could be the main factor affecting the HDT values compared to the chain networking structure of CrPLA.
HDTs of PLA, CrPLA, and their composites.
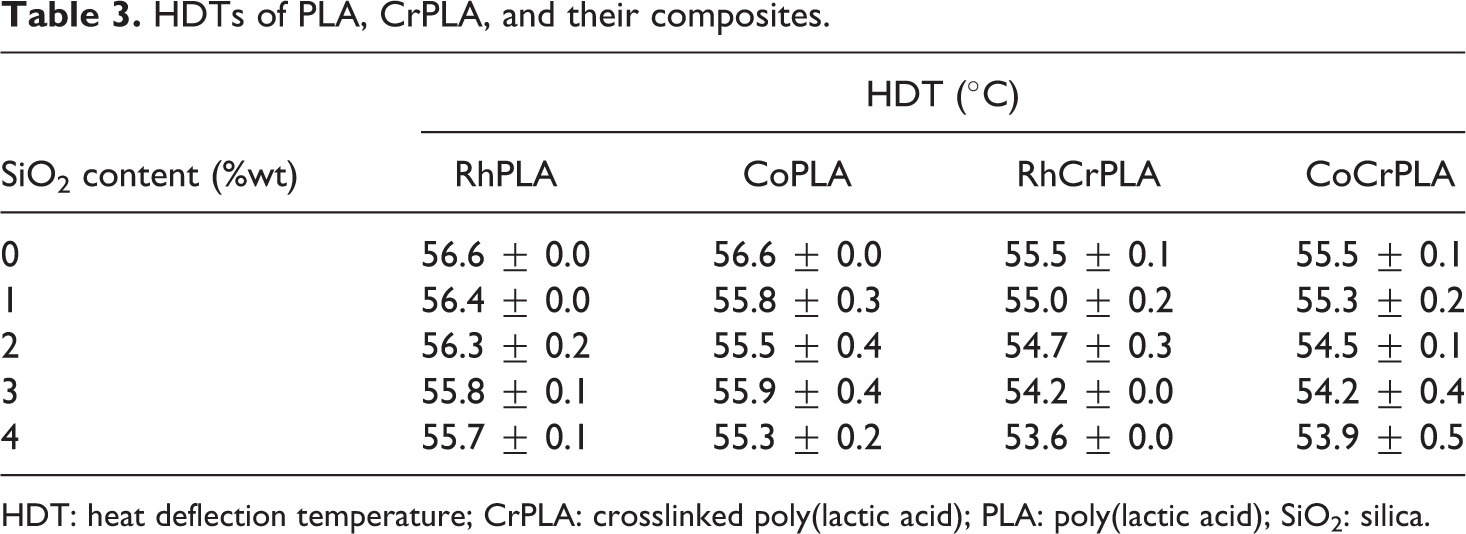
HDT: heat deflection temperature; CrPLA: crosslinked poly(lactic acid); PLA: poly(lactic acid); SiO2: silica.
Conclusions
The FTIR-ATR spectra of RhSiO2 showed that the presence of C–H functional groups assisted the surface modification esterification reaction. This played an important role in altering the properties of the PLA and CrPLA composites. The fracture surfaces, tensile results, degree of swelling, and thermal properties additionally supported the enhanced reaction between stearic acid and RhSiO2. However, the HDT of both the PLA and CrPLA composites were not significantly influenced by mixing with either CoSiO2 or RhSiO2.
