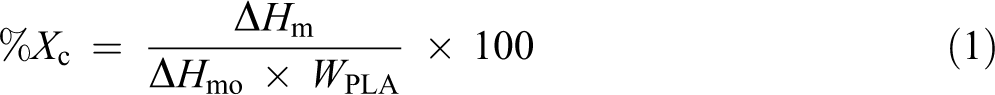Abstract
Biocomposites of poly(lactic acid) (PLA) and cellulose nanofibers (CNFs) extracted from cassava pulp were successfully prepared by melt mixing in an internal mixer. CNFs were prepared from cassava pulp by submitting to alkali hydrolysis, bleaching treatment, and acid hydrolysis. The compatibility between CNFs and PLA matrix was improved using glycidyl methacrylate (GMA) grafted PLA (PLA-g-GMA) as an effective compatibilizer. Higher elongation at break and impact strength of PLA/PLA-g-GMA/CNFs biocomposites was achieved compared to that of neat PLA. PLA-g-GMA shows a strong effect on the crystallization behavior of the biocomposites. The PLA/PLA-g-GMA/CNFs biocomposites induce cold crystallization to take place at lower temperature. Higher degree of crystallinity of PLA/PLA-g-GMA/CNFs biocomposites was obtained compared to PLA/CNFs biocomposites. The mechanical and thermal properties of PLA/CNFs biocomposites at various ratios were investigated. With increasing CNFs contents, the modulus of the biocomposites increases. Thermal stability of PLA/CNFs and PLA/PLA-g-GMA/CNFs biocomposites did not change significantly compared to that of neat PLA.
Keywords
Introduction
Because of their being environmentally friendly and low life-cycle costs, the fully biodegradable composites prepared by a combination of nanofillers and polymer matrix come from renewable agricultural resources have received considerable interest in academic studies. 1,2 Compared to other biodegradable polymers such as starch and chitosan, poly(lactic acid) (PLA) has better mechanical properties. 3 However, the inherent brittleness, low thermal stability, and insufficient impact strength of PLA limited the scope of its applications. So far, besides polymer blends 4 and copolymerization, 5 the addition of natural fibers into PLA matrix has been considered to be an effective strategy for improving the properties of neat PLA. 6 With the renewability and biodegradability of both components, the green PLA/natural fibers biocomposites have a great potential as alternative resource for petroleum-based polymers. Compared to synthetic fibers, natural fibers have many advantages such as renewability, high specific strength, low density, and low cost. 7
Properties of fiber-reinforced biocomposites are strongly affected by interactions between the components. Using fillers with nanoscale leads to the increase in filler/matrix interface area and modified properties at very low loading levels. 8 Up to now, the laboratory-scale preparation of PLA/cellulose nanofibers (CNFs) biocomposites using solvent casting process has been widely investigated. 9 –14 With this method, a good dispersion of hydrophilic CNFs in PLA matrix can be obtained with notably improved mechanical properties. Nevertheless, this method is extremely tedious and time-consuming and has drawbacks for the industrial implementation such as low cost–performance ratio and difficulties in the removal of the entrapped solvent. Melt processing techniques have been considered to be the most suitable methods of PLA/CNFs biocomposites for industrial applications. So far, biodegradable composites of PLA with CNFs prepared by melt compounding have been investigated by Ahmad and Luyt, 15 Espino-Pérez et al., 16 and Yang et al. 17 The addition of CNFs into PLA matrix is expected to have several positive consequences. With the good dispersion of CNFs in PLA matrix, the mechanical properties of PLA/CNFs biocomposites can be improved through stress transfer from the PLA matrix to the CNFs. 18 Moreover, CNFs are expected to act as nucleating agents for crystallization of PLA. Jonoobi et al. 2 prepared PLA/CNFs biocomposites with different CNFs contents by melt compounding using twin screw extrusion. The results of mechanical properties indicated that the tensile strength and modulus of biocomposites were improved with increased nanofiber content. With the addition of 5 wt% nanofibers, a 21% increase in tensile strength was observed. The modulus of PLA was increased from 2.9 GPa to 3.6 GPa. PLA/CNFs prepared using two-roll mill were also reported. 19 Differential scanning calorimetry (DSC) results showed that by the addition of 2.5 wt% CNFs, the crystallinity of PLA was improved. PLA/CNFs biocomposites prepared by melt mixing using an internal mixer were also reported in another study. 20 The results of the mechanical properties of obtained biocomposites revealed that the tensile strength was enhanced with the addition of CNFs. The thermal expansion coefficient was found to decrease considerably when CNFs were used. In addition, using the electrospinning method, the dispersion of CNFs in PLA matrix was improved with significantly enhanced mechanical properties and thermal stability of obtained PLA/CNFs biocomposites. 21
The dispersion of CNFs in PLA matrix during melt processing is the biggest challenge for the preparation of PLA/CNFs biocomposites using melt compounding method. During melt mixing, the agglomeration of CNFs is prone to occur due to the strong fiber–fiber interactions (hydrogen bonding formed through hydroxyl groups on the surface of CNFs) and the poor polymer wetting on the hydrophilic CNFs surface. In order to improve the dispersion of CNFs in PLA matrix during melt processing, some approaches such as using a surfactant, 22 a master batch of PLA/CNFs, 17,23 and a two-step process where solvent mixing and melt mixing were carried out sequentially 18 have been investigated. Using these methods, the dispersion of CNFs in PLA matrix was considerably improved. However, in some cases (using high content of surfactant), degradation of PLA matrix occurred. 22
In this research, the dispersion of CNFs in PLA matrix is improved using glycidyl methacrylate (GMA) grafted PLA (PLA-g-GMA) as a compatibilizer. In our previous publication, 24 the improved dispersion of CNFs in PLA matrix and interfacial adhesion between PLA and CNFs were proved by scanning electron microscopy micrographs. The well dispersed CNFs acted as nucleating agents for the crystallization of PLA. As a result, the crystallization rate of PLA/PLA-g-GMA/CNFs biocomposites studied by synchrotron small-angle X-ray scattering (SAXS) was enhanced significantly as compared to that of neat PLA. The detailed discussion can be found in our previous publication. 24 In this work, the effects of PLA-g-GMA and CNFs on the mechanical and thermal characteristics of PLA were elucidated.
Materials and methods
Materials
PLA (4043D grade) was purchased from NatureWorks, USA. PLA-g-GMA was prepared by melt free radical polymerization using dicumyl peroxide as an initiator. The details of this grafting reaction can be found in one of our previous publications. 25 CNFs were extracted from cassava pulp. The extraction and characterization of CNFs were given in details in another publication. 26
Preparation of PLA biocomposites
Two PLA biocomposites systems with and without using PLA-g-GMA were prepared by direct melt compounding of PLA and CNFs using an internal mixer (Haake Rheomix, 3000p, Thermo Scientific) at temperature of 170°C with the mixing time of 10 min and mixing speed of 60 r min−1. Details of the preparation process of biocomposites can be found in our previous publication. 24 CNFs content was varied into 0.1, 0.3, and 0.5 wt%. PLA-g-GMA with the content of 20 wt% was used as an effective compatibilizer to improve the interfacial adhesion between PLA and CNFs.
Characterization
Thermal properties
Thermal stability of PLA and its biocomposites was evaluated via thermogravimetric analysis (TGA), which was performed using a Mettler Toledo STARe System, USA (TGA/DSC1) by heating the samples from 25°C to 600°C with a heating rate of 10°C min−1 under a nitrogen atmosphere. The sample with a weight between 5 mg and 10 mg was used for each run.
DSC (model: DSC204F1 Phoenix, NETZSCH) was used to obtain the thermal properties of specimens by heating the samples from 40°C to 200°C at a rate of 5°C min−1 (first heating scan). After keeping the specimens at 200°C for 5 min, they were cooled to 40°C with a rate of 5°C min−1. Afterward, they were heated again to 200°C at 5°C min−1 (second heating scan). The degree of crystallinity (%X c) of PLA and biocomposites was determined by the following equation
In which ΔH m is the measured melting enthalpy (J g−1) from the second heating scan, ΔH mo is the theoretical melting enthalpy of completely crystalline PLA (93.7 J g−1), and W PLA is the PLA weight fraction in the biocomposites. 27
Mechanical properties
Compression molding (LabTech, LP20-B, Thailand) was used to prepare the test specimens for mechanical properties testing. After drying in an oven at 70°C for 2 h, all samples were compression molded at 170°C for 10 min. Tensile properties of PLA and its biocomposites were measured according to ASTM D638 using an Instron Universal Testing Machine (UTM 5565) with a load cell of 5 kN. The unnotched Izod impact strength of PLA and its biocomposites was obtained according to ASTM D256 using Instron CEAST 9050 Pendulum Impact System, Italy.
Results and discussion
Thermal properties
Thermal stability of PLA and its biocomposites was investigated by TGA. The residual mass % as a function of temperature for neat PLA and the biocomposites at various fiber contents is shown in Figures 1 and 3. As observed from Figure 1, the TGA and DTG curves of neat PLA and PLA/CNFs biocomposites show only one degradation step between 310°C and 390°C. It was observed that the thermal stability of PLA/CNFs biocomposites was not significantly different compared to that of neat PLA. A slight decrease in thermal stability of PLA/CNFs biocomposites was observed as CNFs content was increased from 0.1 wt% to 0.5 wt%. This might be attributed to the higher water content induced by the presence of the cellulosic nanofiller. With the increase in fiber content, the agglomerations of fiber on the surface due to poor dispersion could accelerate the water absorption of biocomposites. This leads to lowering the thermal stability of obtained PLA/CNFs biocomposites. 28 In addition, the increased thermal conductivity of the polymer after CNFs addition was also suggested as the reason for reduced thermal stability of the biocomposites. 29 Figure 1 also reveals that the char yield increased with the addition of CNFs.
TGA and DTG curves of neat PLA and PLA/CNFs biocomposites with different fiber loadings. TGA: thermogravimetric analysis; DTG: derivative thermogravimetry; PLA: poly(lactic acid); CNF: cellulose nanofiber.
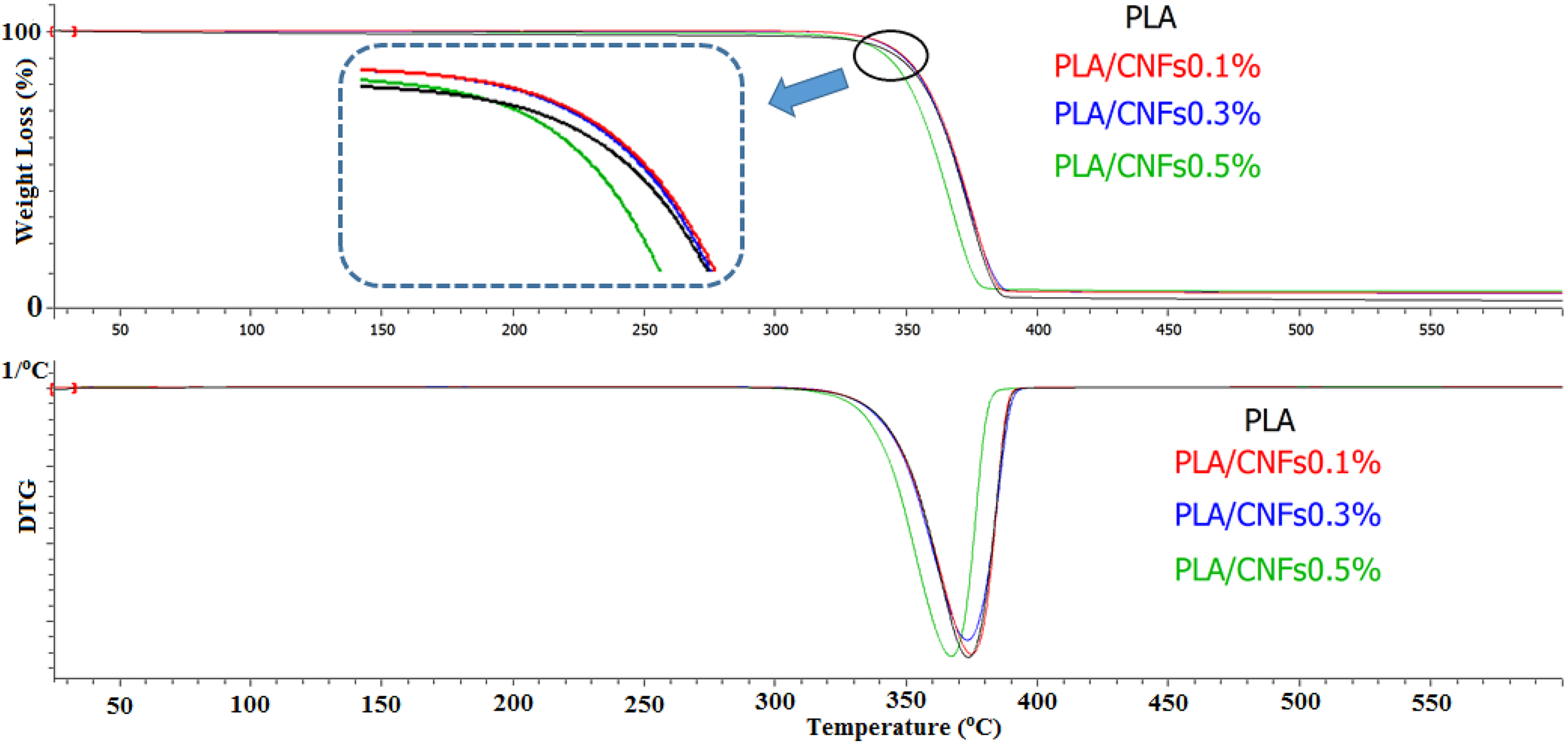
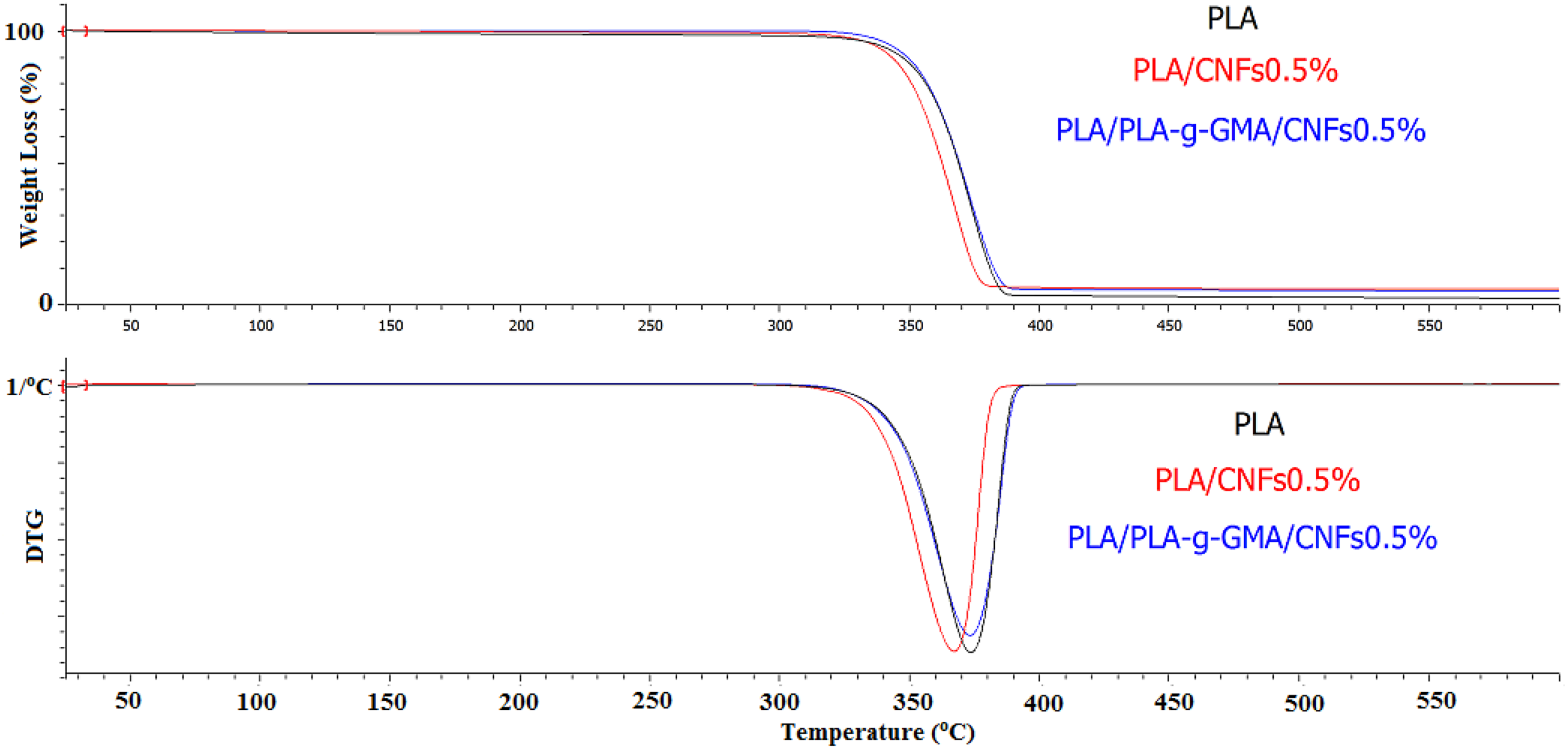
In the case of using PLA-g-GMA as a compatibilizer, the thermal stability of PLA/PLA-g-GMA/CNFs biocomposites was found to be higher than that of PLA/CNFs biocomposites (Figure 2). This could be due to the better dispersion of CNFs and the improved interaction between PLA and fibers, which led to reduce the chain mobility. As a result, the thermal stability was enhanced. This result was in agreement with another study in which the strong hydrogen bonding between hydroxyl groups of cellulose and the polyvinyl alcohol (PVA) matrix was suggested to improve the thermal stability of the host polymer. 30 Additionally, it was observed from Figure 3 that there was no considerable difference in the thermal stability of PLA/PLA-g-GMA/CNFs biocomposites with the increase in CNFs content. The char yield of PLA/PLA-g-GMA/CNFs biocomposites was higher than that of neat PLA.
TGA and DTG curves of neat PLA, PLA/CNFs 0.5 and PLA/PLA-g-GMA/CNFs 0.5 biocomposites. TGA: thermogravimetric analysis; DTG: derivative thermogravimetry; PLA: poly(lactic acid); CNF: cellulose nanofiber; PLA-g-GMA: glycidyl methacrylate (GMA) grafted PLA.
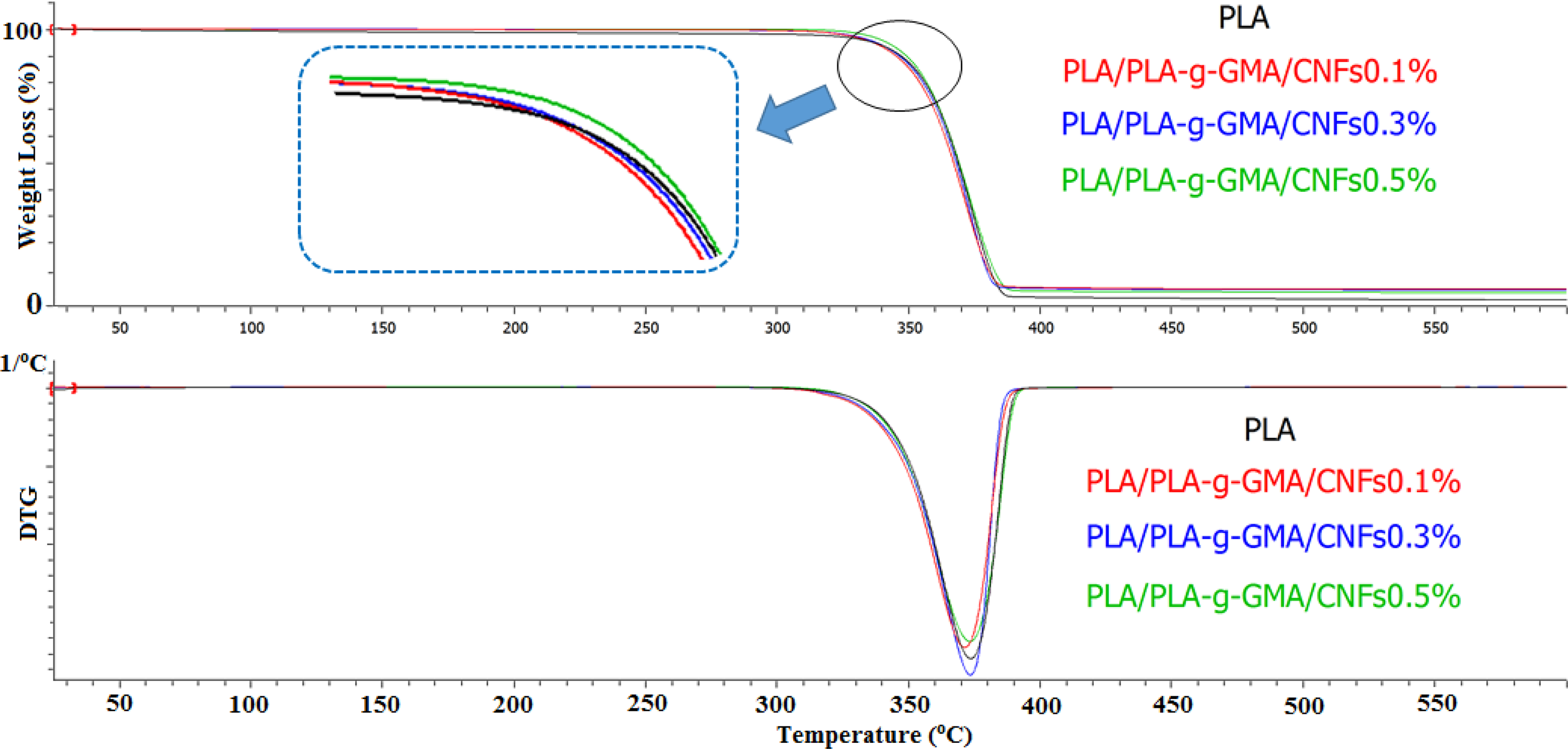
TGA and DTG curves of neat PLA and PLA/PLA-g-GMA/CNFs biocomposites with different fiber loadings. TGA: thermogravimetric analysis; DTG: derivative thermogravimetry; PLA: poly(lactic acid); PLA-g-GMA: glycidyl methacrylate (GMA) grafted PLA; CNF: cellulose nanofiber.
The non-isothermal crystallization and melting behavior of PLA and its biocomposites were investigated by DSC. DSC thermograms of neat PLA and biocomposites during the cooling scan are shown in Figure 4. From this graph, one can see that neat PLA and PLA/CNFs biocomposites with various fiber contents did not show melt crystallization upon cooling. In contrast to that, with using PLA-g-GMA as a compatibilizer, the melt crystallization peaks were observed for all PLA/PLA-g-GMA/CNFs biocomposites. This indicates that PLA-g-GMA induced the melt crystallization of PLA upon cooling. Furthermore, as seen in Figure 4, there was no significant change in crystallization temperature of the biocomposites as fiber content was increased from 0.1 wt% to 0.5 wt%.
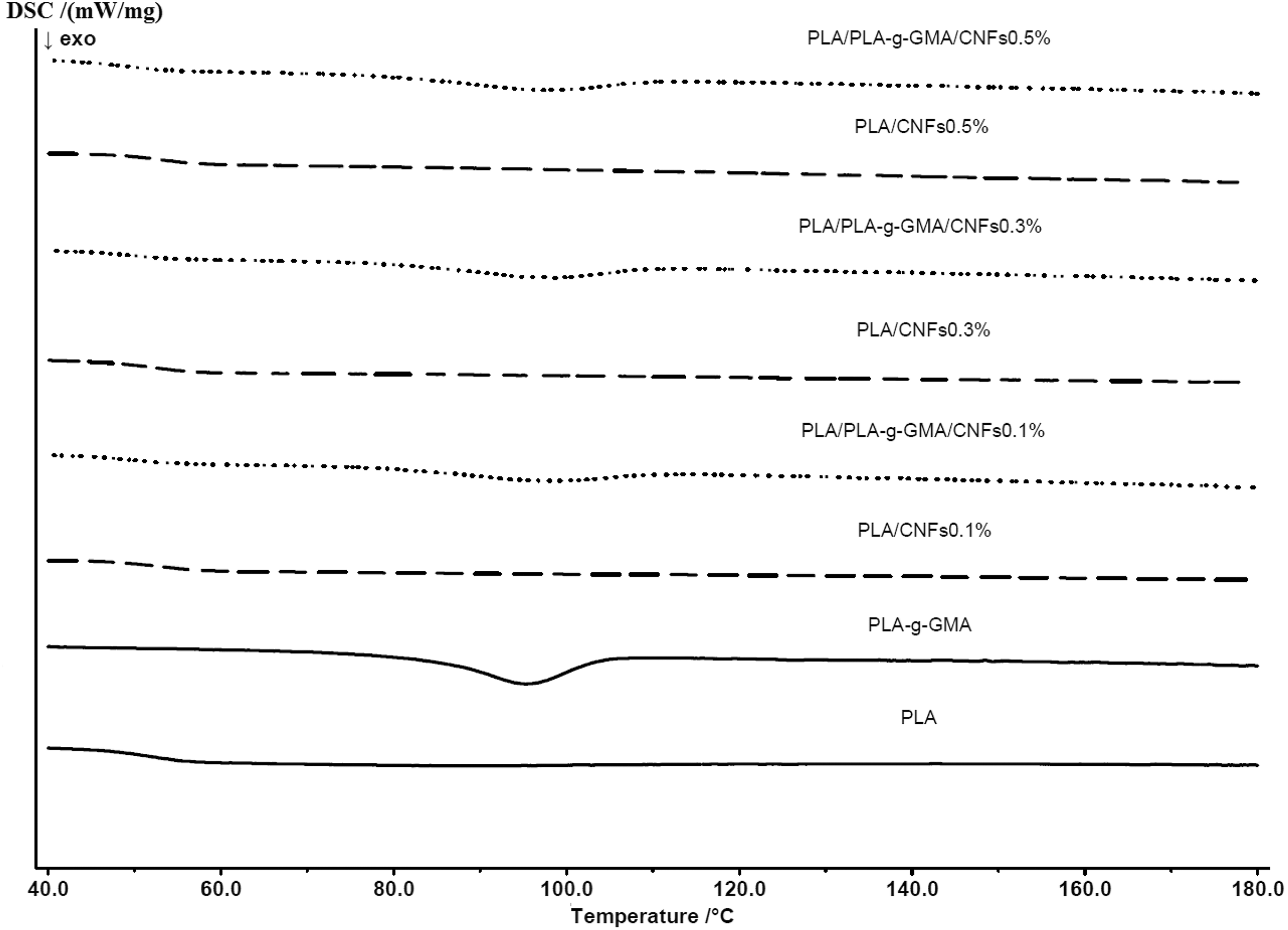
DSC thermograms of PLA and biocomposites with various fiber loadings (cooling cycle). DSC: differential scanning calorimetry; PLA: poly(lactic acid).
Figure 5 shows the DSC thermograms of neat PLA and its biocomposites during the second heating scan. As observed in Figure 5, with the addition of CNFs, the glass transition temperature (T g) of PLA/CNFs biocomposites slightly increased in comparison with that of pure PLA. It is due to the restriction of chain mobility with the presence of CNFs. The T g values of PLA/CNFs biocomposites were found unchange significantly with increasing fibers content from 0.1 wt% to 0.5 wt%. Using PLA-g-GMA as a compatibilizer, a slight decrease in T g values of biocomposites was observed.
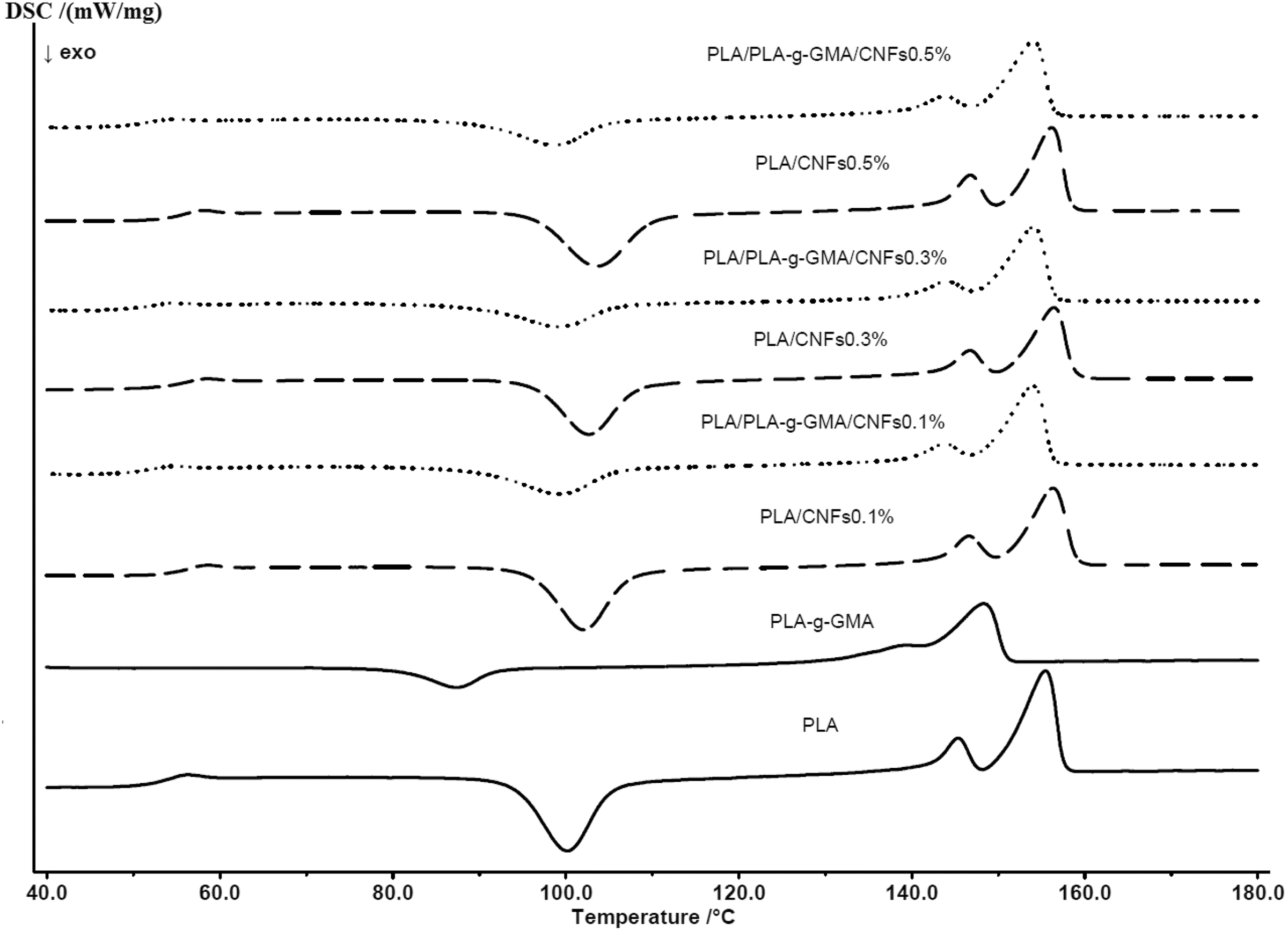
DSC thermograms of PLA and biocomposites with various fiber loadings (second heating cycle). DSC: differential scanning calorimetry; PLA: poly(lactic acid).
Moreover, it was observed from Figure 5 that PLA/CNFs biocomposites exhibited a slightly higher cold crystallization temperature compared to neat PLA. When PLA-g-GMA was used, the broader cold crystallization peaks appeared at lower temperatures as compared to the cold crystallization of neat PLA. The lower cold crystallization temperature is an indication of faster crystallization induced by CNFs which acted as nucleating agents for PLA. 31 CNFs allow heterogeneous nucleation mechanism which induces a decrease in the free energy barrier and fastens the crystallization. In addition, it was observed from Figure 5 that the cold crystallization temperatures of PLA/CNFs and PLA/PLA-g-GMA/CNFs biocomposites were not significantly influenced by the increase in fiber content.
The melting temperatures (T m1 and T m2) of PLA/CNFs biocomposites were found to be slightly higher than those of neat PLA. The lower temperature peak (T m1) is attributed to the melting of crystals formed upon cooling from the melt, and the higher one (T m2) corresponds to the melting of crystals generated from cold crystallization upon heating. 32 The PLA/PLA-g-GMA/CNFs biocomposites, however, showed slightly lower melting temperatures compared to neat PLA and PLA/CNFs biocomposites. The degree of crystallinity (%X c) calculated based on equation (1) indicated that PLA/PLA-g-GMA/CNFs biocomposites had higher crystallinity compared to PLA/CNFs biocomposites at all fiber loadings used. This result is consistent with the SAXS result presented in our previous publication. 24 Using PLA-g-GMA as a compatibilizer, the crystallinity of PLA in the biocomposites was enhanced. This enhancement was induced by the improved dispersion of CNFs in the PLA matrix. 24 As a result, more crystals were able to nucleate and grow on the increased surface area of the interfaces due to a growing number of nucleating particles. 33 Besides that, as can be seen from Table 1, the X c values of PLA/CNFs and PLA/PLA-g-GMA/CNFs biocomposites did not change significantly with the increase of fiber content from 0.1 wt% to 0.5 wt%. The degree of crystallinity (%X c) and DSC data such as the T g, crystallization temperature (T c), enthalpy of crystallization (ΔH c), cold crystallization temperature (T cc), enthalpy of cold crystallization (ΔH cc ), melting temperatures (T m1 and T m2), and enthalpy of melting (ΔH m) of neat PLA, PLA-g-GMA, and biocomposites are reported in Table 1.
Thermal properties of neat PLA, PLA-g-GMA, and biocomposites with different fiber contents.
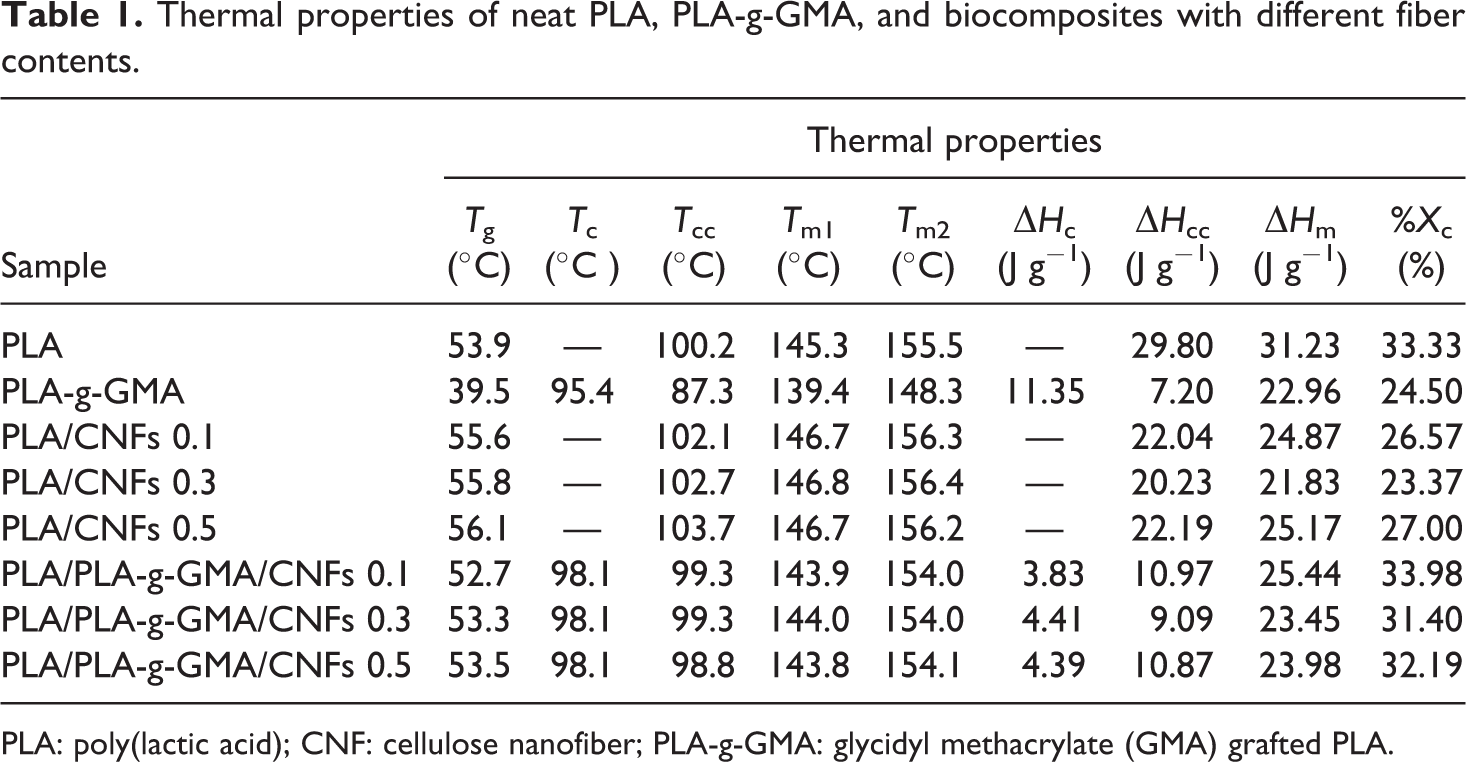
PLA: poly(lactic acid); CNF: cellulose nanofiber; PLA-g-GMA: glycidyl methacrylate (GMA) grafted PLA.
Mechanical properties
The mechanical properties of PLA and its biocomposites are shown in Figures 6 to 9. Figure 6 shows the tensile modulus of neat PLA and its biocomposites at various fiber contents. The tensile modulus of PLA increased 5.9% from 846 MPa to 896 MPa with the addition of 0.5-wt% CNFs without using PLA-g-GMA. This can be associated with the restrictions of macromolecular mobility and deformability imposed by the presence of CNFs. The tensile modulus of PLA/CNFs biocomposites increased with increasing CNFs loading because the increase in CNFs content restricted the mobility of the PLA chains. With using PLA-g-GMA as a compatibilizer, the tensile modulus of biocomposites was slightly improved (reached a value of 940 MPa with the addition of 0.5-wt% CNFs). The increased composite stiffness depends on the CNFs content and uniformity of reinforcement dispersion. 34 In this case, the enhanced tensile modulus of PLA/PLA-g-GMA/CNFs biocomposites can be attributed to the better dispersion of CNFs nanofillers in PLA matrix due to the presence of PLA-g-GMA. 24 Moreover, PLA-g-GMA may act as a plasticizer in PLA/PLA-g-GMA/CNFs biocomposites due to its lower T g values, as shown in Table 1.
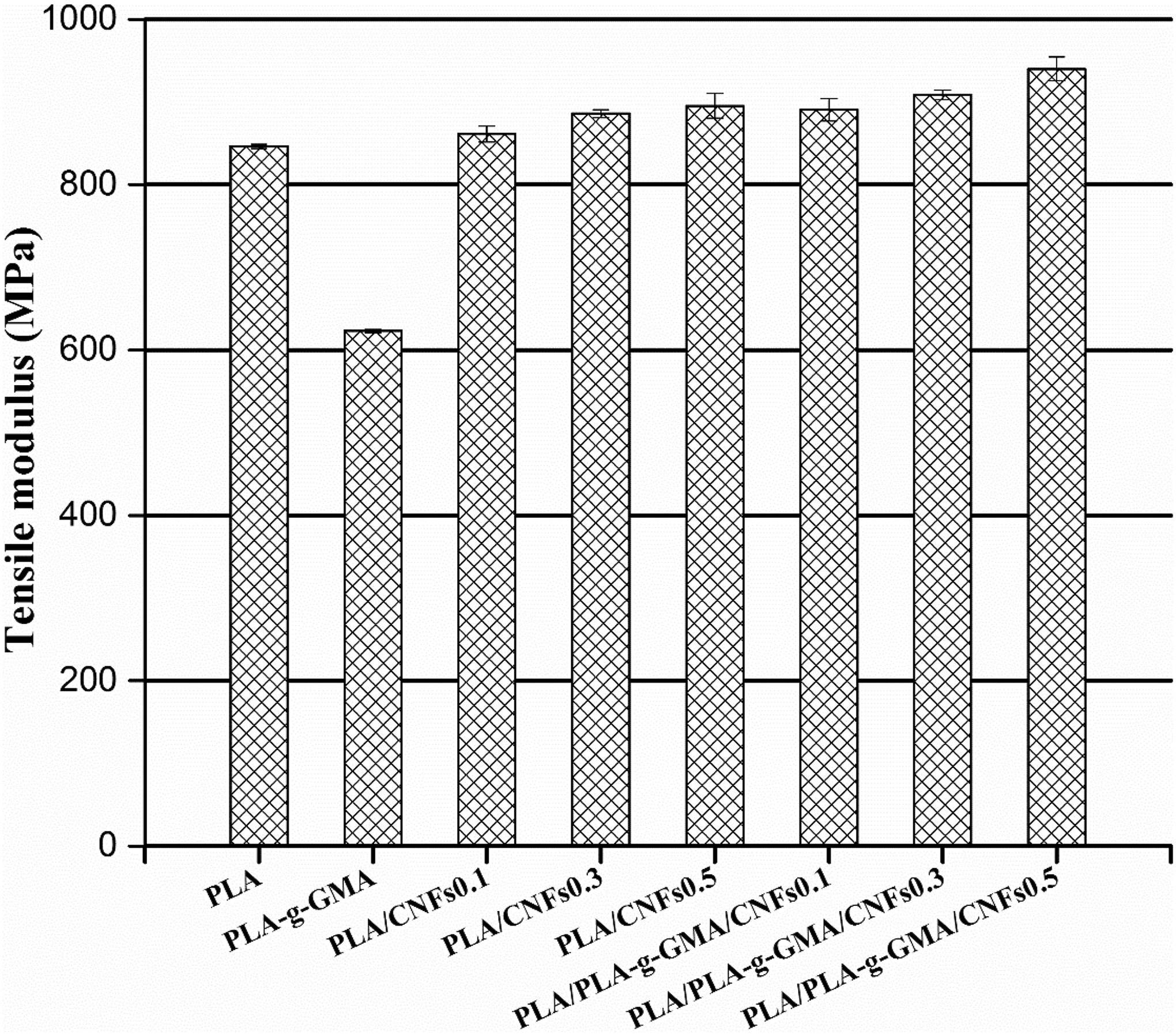
Tensile modulus of PLA, PLA-g-GMA, and biocomposites with different fiber loadings. PLA: poly(lactic acid); PLA-g-GMA: glycidyl methacrylate (GMA) grafted PLA.
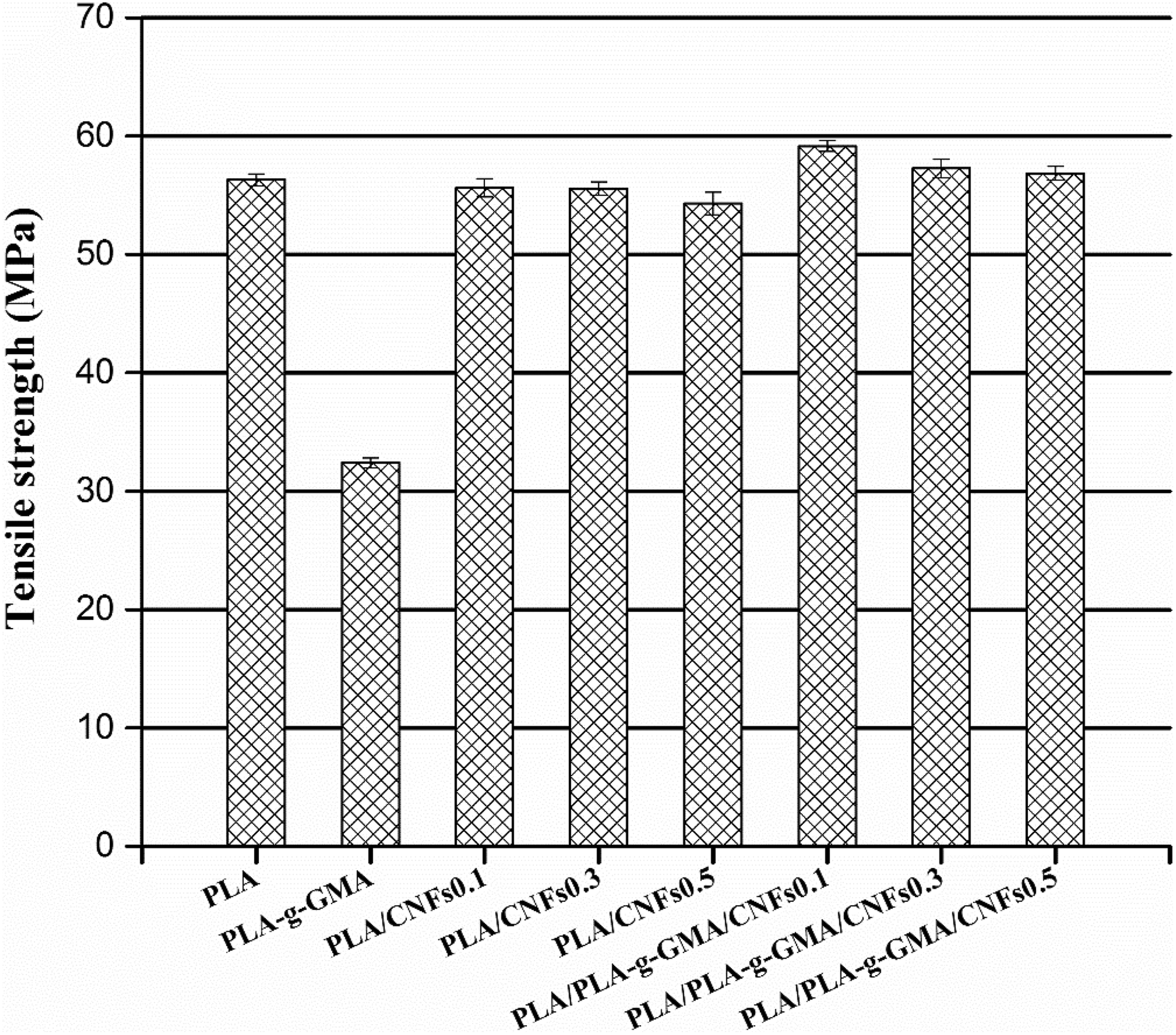
Tensile strength of PLA, PLA-g-GMA, and biocomposites with different fiber loadings. PLA: poly(lactic acid); PLA-g-GMA: glycidyl methacrylate (GMA) grafted PLA.
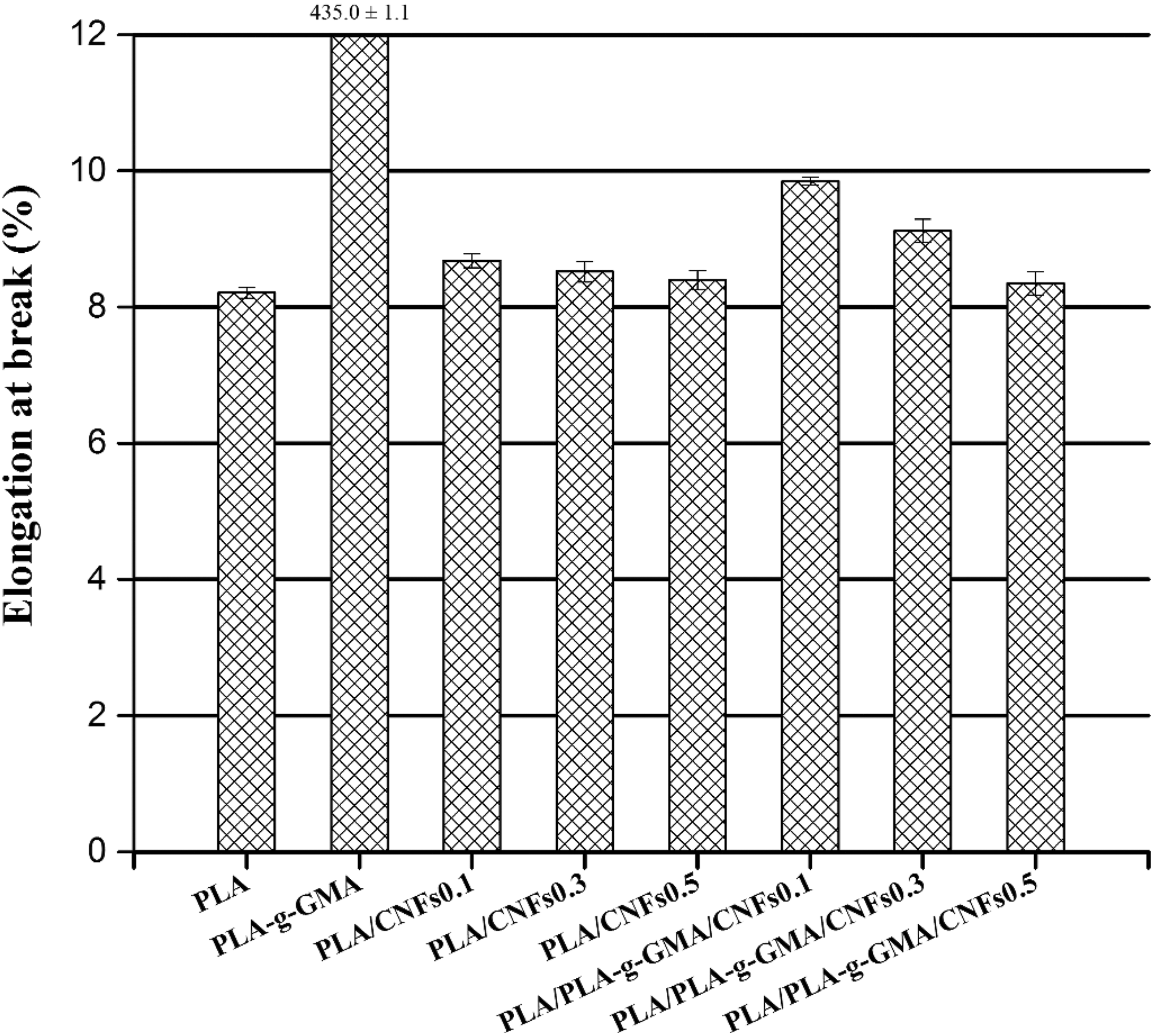
Elongation at break of PLA, PLA-g-GMA, and biocomposites with different fiber loadings. PLA: poly(lactic acid); PLA-g-GMA: glycidyl methacrylate (GMA) grafted PLA.
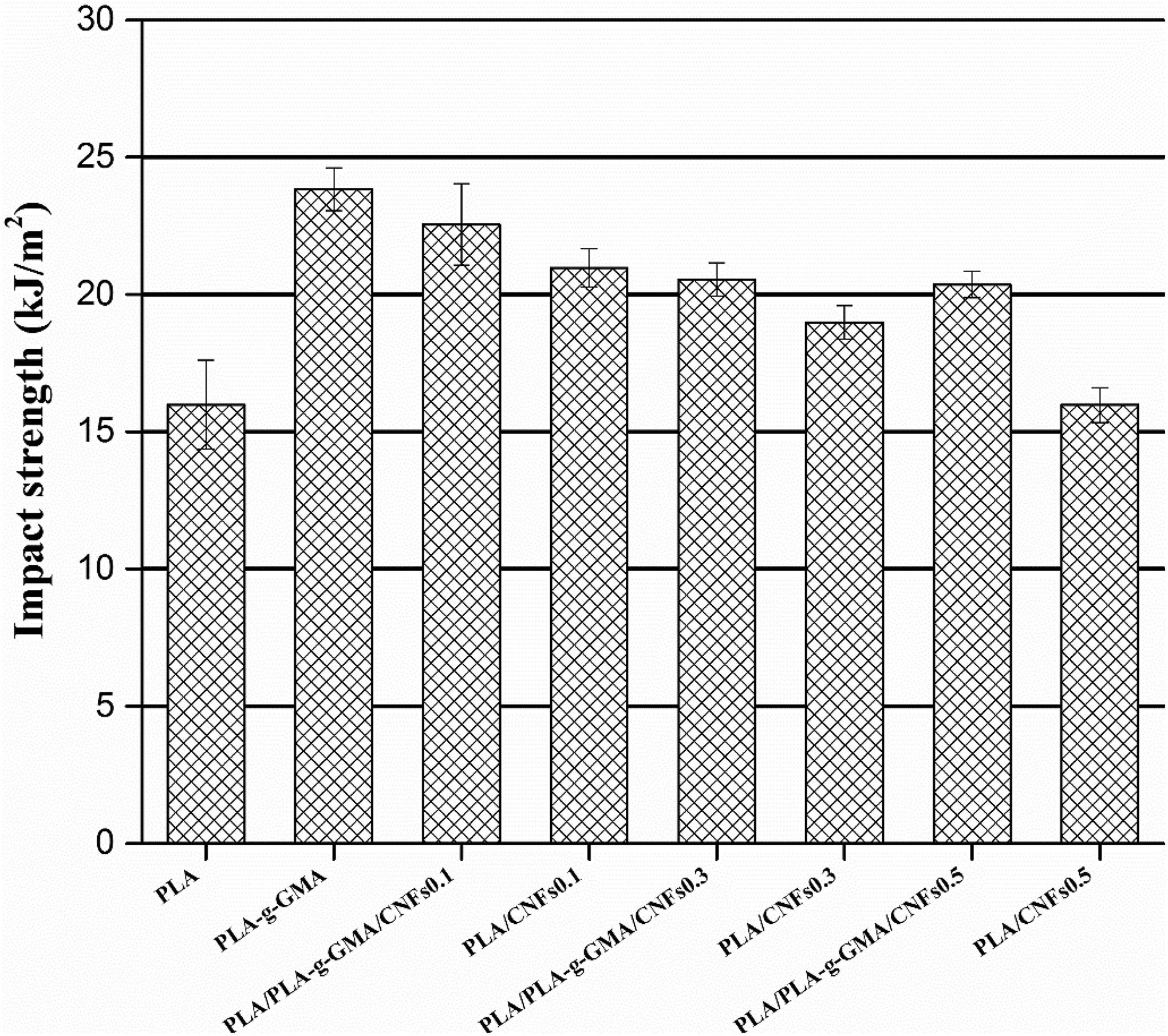
Impact strength of PLA, PLA-g-GMA, and biocomposites with different fiber loadings. PLA: poly(lactic acid); PLA-g-GMA: glycidyl methacrylate (GMA) grafted PLA.
The tensile strength of neat PLA and its biocomposites is shown in Figure 7. Compared to PLA, the PLA/CNFs biocomposites did not show any improvements in tensile strength. This is due to the poor interfacial adhesion between CNFs and PLA matrix. Bitinis et al. 23 also reported that the tensile strength was maintained or lowered with the addition of unmodified and modified CNFs in PLA/natural rubber (NR) blend when NR was fixed at 10 wt% and the CNFs loading was varied from 1 wt% to 5 wt%. As observed in Figure 7, using PLA-g-GMA, the tensile strength of PLA/PLA-g-GMA/CNFs biocomposites was found to be slightly improved as compared to that of PLA/CNFs biocomposites at all used fiber loadings (with the addition of only 0.1 wt% CNFs in PLA resulted in a 5.2% increase in the tensile strength compared to neat PLA). The reason for the improvement in tensile strength can be attributed to the better dispersion of CNFs and the enhanced interfacial adhesion between PLA and CNFs as PLA-g-GMA was used as a compatibilizer. 24 This reduces the voids at the fiber–matrix interface and allows better stress transfer from the PLA matrix to the CNFs. Additionally, the slightly increased degree of crystallinity (Table 1) also contributed to the improvement of tensile strength of PLA/PLA-g-GMA/CNFs biocomposites. Figure 7 also shows that increasing fiber content from 0.1 wt% to 0.5 wt% did not cause a significant change in the tensile strength of PLA/PLA-g-GMA/CNFs biocomposites. The tensile strength of PLA/PLA-g-GMA/CNFs biocomposites slightly decreased with increasing fiber content due to more agglomeration of CNFs.
Figure 8 shows that there was no significant difference in elongation at break of neat PLA and PLA/CNFs biocomposites. The elongation at break of both PLA/CNFs and PLA/PLA-g-GMA/CNFs biocomposites slightly decrease with increasing fiber contents. With using PLA-g-GMA, the elongation at break of PLA/PLA-g-GMA/CNFs biocomposites shows slightly higher than that of PLA/CNFs biocomposites. The grafting of GMA onto PLA chain may cause less intermolecular interaction leading to lower T g of PLA-g-GMA compared to that of PLA. As a result, PLA-g-GMA can act as a plasticizer in the biocomposites and results in the improvement of elongation at break of the biocomposites.
The impact strength of neat PLA and its biocomposites is shown in Figure 9. The impact strength of PLA/CNFs biocomposites was higher than that of neat PLA, particularly for the biocomposites with low fiber content (0.1 and 0.3 wt%). With increasing fiber loading from 0.1 wt% to 0.5 wt%, the impact strength of PLA/CNFs biocomposites decreased. This is ascribed to the poor dispersion of CNFs in PLA matrix as CNFs content was increased. 24 This might result in stress concentration regions that require less energy to initiate cracks. With the addition of PLA-g-GMA, the impact strength of biocomposites was improved. The PLA/PLA-g-GMA/CNFs 0.1 biocomposites achieved the highest impact strength (22.6 kJ m−2) with a 41 and 7.6% increase compared to neat PLA and PLA/CNFs 0.1 biocomposites, respectively. This improvement can be attributed to the better dispersion of CNFs with the presence of PLA-g-GMA. The well-dispersed CNFs acted as a nucleating agent and increased the crystallization rate of PLA, resulting in a reduction in PLA spherulites size. As a result, the impact strength of biocomposites was enhanced. This result was in agreement with SAXS results discussed in our previous publication. 24
Conclusions
The PLA/CNFs biocomposites were prepared by melt compounding using an internal mixer. PLA-g-GMA was shown to be an effective compatibilizer to improve the compatibility between PLA and CNFs. Moreover, PLA-g-GMA with its lower T g compared to PLA was acted as a plasticizer in the biocomposites.
PLA-g-GMA plays an important role in improving the degree of crystallinity of the biocomposites. Cold crystallization temperature of PLA/PLA-g-GMA/CNFs biocomposites was shifted to a lower temperature compared to neat PLA and PLA/CNFs biocomposites.
The improvement in elongation at break and impact strength was achieved using PLA-g-GMA as compatibilizer and plasticizer in PLA/PLA-g-GMA/CNFs biocomposites. Meanwhile, CNFs led to the improvement of modulus of the biocomposites. This results in higher mechanical properties, including tensile modulus, elongation at break, and impact strength of the biocomposites with the addition of only 0.1-wt% CNFs and PLA-g-GMA compared to those of neat PLA.
Footnotes
Authors’ note
Thanh Chi Nguyen is also affiliated with Faculty of Applied Sciences, Ton Duc Thang University, Ho Chi Minh City, Vietnam.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Suranaree University of Technology, Thailand, through SUT-PhD Scholarship for Asian Countries Program.