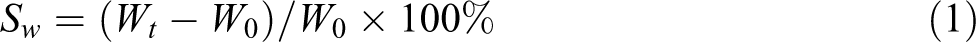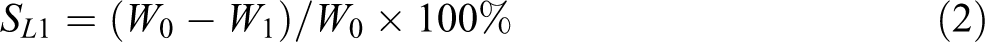Abstract
A novel water-swellable rubber (WSR) has been prepared by dynamically vulcanized ethylene–vinyl acetate copolymer (EVA)/chlorinated polyethylene (CPE)/nitrile butadiene rubber (NBR) blends where the cross-linked poly(sodium acrylate) (CPNaAA) was used as a super water-absorbent resin and dispersed in the NBR rubber. The mechanical properties, water-swelling behavior, weight loss and crystallization behavior of the prepared WSRs were investigated systemically. When the CPNaAA content was 20 phr above, the increase in CPNaAA dosage contributed to the decrease in mechanical properties. However, the WSRs showed strong water-swelling behaviors and the water-swelling ratio of WSRs with 50 phr CPNaAA was 669.3% at 120 h. The WSRs with 60 phr CPNaAA showed high water-swelling rate and achieved the equilibrium swelling at about 566.3% in 23 h. The secondary and third water-swelling behaviors of WSRs showed significantly rapid equilibrium swelling and remarkable decrease in weight loss. X-ray diffraction results revealed the increase in CPNaAA content would result in the reduction of crystalline structure content in EVA. Morphological study of etched surface showed that the CPNaAA particles were dispersed evenly in the WSRs and coated with cross-linked NBR rubber; moreover, field-emission scanning electron microscopic graphs showed the crystalline structure of EVA and clusters of flakes were dispersed on the etched surface of WSRs.
Keywords
Introduction
Thermoplastic elastomers (TPEs) are a class of polymeric materials with elastic and mechanical properties similar to those of conventional vulcanized rubbers but processable as thermoplastics. A unique kind of TPEs is thermoplastic vulcanizates (TPVs) that are prepared using the method of dynamic vulcanization. 1,2 Dynamic vulcanization, first described by Gessler 3 and then developed by Fisher 4 , Coran and Patel 5 and Coran et al., 6 is a procedure in which curing agents are used to cross-link rubber in situ during the process of mixing with molten thermoplastics. The productions are able to offer outstanding mechanical properties and be melted and recycled repeatedly. Morphologically, the achieved TPVs consist of small cross-linked elastomeric domains embedded in a continuous thermoplastic matrix. 7,8
Water-swellable rubbers (WSRs) are a new kind of elastomeric functional materials, and they possess both water-swelling ability and properties of general rubber such as outstanding resilience and excellent tensile strength. 9 –11 Once the WSRs are immersed into water, the water molecules will enter into the WSRs by physical actions such as diffusion, capillarity and surface adsorption, connecting with the WSRs and forming a strong affinity with hydrophilic water-absorbent resins. 12 The hydrophilic components in WSRs are able to absorb large quantities of water and an expansion force is exerted; meanwhile, the rubber itself has resistance to the force of the deformation. When the two forces reach a balance, the equilibrium swelling is achieved. WSRs are capable of maintaining their elasticity and retaining water even under direct pressure, so they are widely used in dams, caulking, sealing of gaps and construction works.
In recent years, a number of literatures about WSRs have been reported. The general methods to prepare WSRs are physical blending 13,14 and chemical grafting modification. 15,16 However, we know of few reports on WSRs based on TPVs. Water-swellable TPVs is a multicomponent product. The dispersed rubber phase in TPVs offers elastic and mechanical properties similar to those of conventional vulcanized rubber, while the thermoplastic resin phase in TPVs gives the strength properties including processability and recyclability. However, in order to reduce the extraction of super water-absorbent resin from the rubber matrix of WSRs, super water-absorbent resin was dispersed in the dispersed rubber phase which was surrounded by the thermoplastic matrix.
The purpose of this research was to explore a new approach to the preparation of WSRs. In our previous report, we had prepared TPVs based on the blends of ethylene–vinyl acetate copolymer (EVA) and nitrile butadiene rubber (NBR) rubber which were compatibilized by chlorinated ployethylene(CPE).The prepared products showed excellent mechanical properties. 17 In this article, we reported water-swellable EVA/CPE/cross-linked poly(sodium acrylate) (CPNaAA)/NBR TPVs, while the CPNaAA particles were dispersed in the NBR phase. The mechanical properties, water-swelling behaviors and weight loss of the dynamically vulcanized blends were investigated systemically as a function of the CPNaAA dosage. Morphology of the dynamically vulcanized EVA/CPE/CPNaAA/NBR WSRs was observed by field-emission scanning electron microscopy (FE-SEM). Crystallization behavior of the WSRs was studied by x-ray diffraction (XRD).
Experimental
Materials
NBR rubber, grade 3305 (acrylonitrile content, 35 wt.%), was commercially manufactured by Lanzhou Petrochemical Co., Ltd (Lanzhou, China). EVA copolymer, grade 630 (vinyl acetate content, 15 wt.%), was supplied by Tosoh Co. (Tokyo, Japan). CPE rubber, type 135 (chlorine content, 35 wt.%), was commercially manufactured by Qingdao Hygain Chemical Co., Ltd (Qingdao, China). CPNaAA resin was supplied by DongGuan Anxin Water Retaining Co., Ltd (Dongguan, China). Sulfur, used as a vulcanizing agent, was obtained from Hengye Zhongyuan Chemical Co., Ltd (Beijing, China). N-cyclohexyl-2-benzothiazole sulfenamide (CZ) and tetramethyl thiuram monosulfide (TS), used as accelerators, were manufactured by Northeast Auxiliary Chemical Industry Co. (Beijing, China). Zinc oxide (ZnO) was used as an activator and obtained from NewLe Qinshi Zinc Co., Ltd (Shijiazhuang, China). Stearic acid was used as an activator and obtained from Wanyou Co., Ltd (Shenzhen, China). Poly(1,2-dihydro-2,2,4-trimethyl-quinoline) (antioxidant RD) was used as an antioxidant and obtained from Shengao Chemical Co., Ltd (Taizhou, China).
Preparation of dynamically vulcanized EVA/CPE/CPNaAA/NBR blends
Commercially available NBR, EVA and CPE, as above, were used for the WSRs. The concentrations for cross-linking the NBR system are expressed in parts per hundred NBR rubbers by weight (phr). The sulfur-containing accelerating system recipe consisted of the following ingredients: 100 phr NBR, 1.0 phr sulfur, 1.5 phr CZ, 1.2 phr TS, 5.0 phr ZnO, 1.5 phr stearic acid and 1.0 phr antioxidant RD.
The dynamically vulcanized EVA/CPE/CPNaAA/NBR blends were produced via a two-step mixing process. In the first step, the preblends containing NBR, the cross-linking ingredients and the CPNaAA powder were compounded in a two-roll mill at room temperature. After 3 min of mixing time, the preblends were removed from the mixer. In the second step, the TPV compounds were prepared by melt-mixing the NBR preblends with CPE and EVA resins using a Brabender PLE 331 Plasticorder (Brabender Gmbh, Duisburg, Germany). The mixer temperature was kept at 160°C with a constant rotor (cam type) speed of 80 r/min. The EVA/CPE/NBR blending ratio was fixed at 30/10/70 and the amount of CPNaAA varied from 20 to 60 phr. The requisite quantity of EVA and CPE was charged into the mixer and allowed to melt. After 2 min, the NBR-based preblend was added. The mixing was continued for another 5 min to allow dynamic vulcanization. Finally, the compound was removed from the mixer and passed through a cold two-roll mill in the molten state to obtain a sheet. The sheet, about 2 mm thick, was compression molded under a pressure of 15 MPa at 160°C for 5 min, followed by cold compression in another molding machine for 8 min at room temperature. Test specimens were die-cut from the compression-molded sheet and used for testing after 24 h.
Measurement and characterization
For the measurement of tensile properties, dumbbell-shaped specimens were prepared according to ASTM D412. The tearing strength was tested according to ASTM D624 using the unnotched 90° angle test piece. Both tensile and tearing tests were performed on a universal testing machine (TCS-2000, Gotech Testing Machines Inc., Taiwan, China) at a crosshead speed of 500 mm/min. The shore A hardness was determined using a handheld Shore A Durometer according to ASTM D2240. All the tests were carried out at 23°C. The average value was calculated for five test specimens.
Morphological study was carried out by FE-SEM (FE-SEM, 6700F, Japan Electron Co., Ltd.,Tokyo, Japan). For the etched surface of specimens, the EVA phase was extracted by immersing the blends into xylene at 100°C for 30 min. Then the samples were dried in a vacuum oven at 40°C for 6 h. The etched surfaces and the fractured surfaces of the specimens were sputtered with thin layers of gold and imaged using the FE-SEM.
The samples were characterized using XRD (XRD with Cu Ka radiation, λ = 1.54178 Å, D/max-2500/PC, Rigaku Co., Ltd.,Tokyo, Japan) at room temperature.
The measurements of the water-swelling ratio were performed by the immersion method with a square sample, with a side length of 30 mm and a thickness of 2 mm. First, the samples were dried for 4 h at 80°C in a vacuum oven, then the sample was immersed in an excess amount of distilled water at 23°C. The weight of the water-swelling sample was determined immediately after the removal of the surface water via blotting with filter paper at different times. The first water-swelling ratio (Sw
) and the first weight loss (SL
1) by mass were calculated as follows:
where Wt and W 0 were the weights of a sample swelled and unswelled with water, respectively; W 1 was the mass of the dried swollen sample, which has reached its equilibrium state in a vacuum oven at 80°C.
The secondary water-swelling ratio and the third water-swelling ratio were measured by the same method as the first water-swelling, and the samples were the dried WSRs after water swelling. The secondary weight loss ratio (SL 2) and the third weight loss ratio (SL 3) were also measured.
Results and discussion
Influence of CPNaAA dosage on mechanical properties of the dynamically vulcanized EVA/CPE/CPNaAA/NBR blends
Figure 1 illustrates the stress–strain behaviors of dynamically vulcanized EVA/CPE/NBR blends with different CPNaAA dosages. As shown in Figure 1, the stress–strain curves of dynamically vulcanized EVA/CPE/CPNaAA/NBR blends present a representative soft and tough characteristic of elastomer. It should be noted that the tensile stress and elongation at break of dynamically vulcanized EVA/CPE/NBR blends with 20 phr CPNaAA dosage reached the maximum value in our experiment. When the CPNaAA content was more than 20 phr, the increase in CPNaAA dosage in the WSRs led to the decrease in the tensile strength and elongation at break obviously.
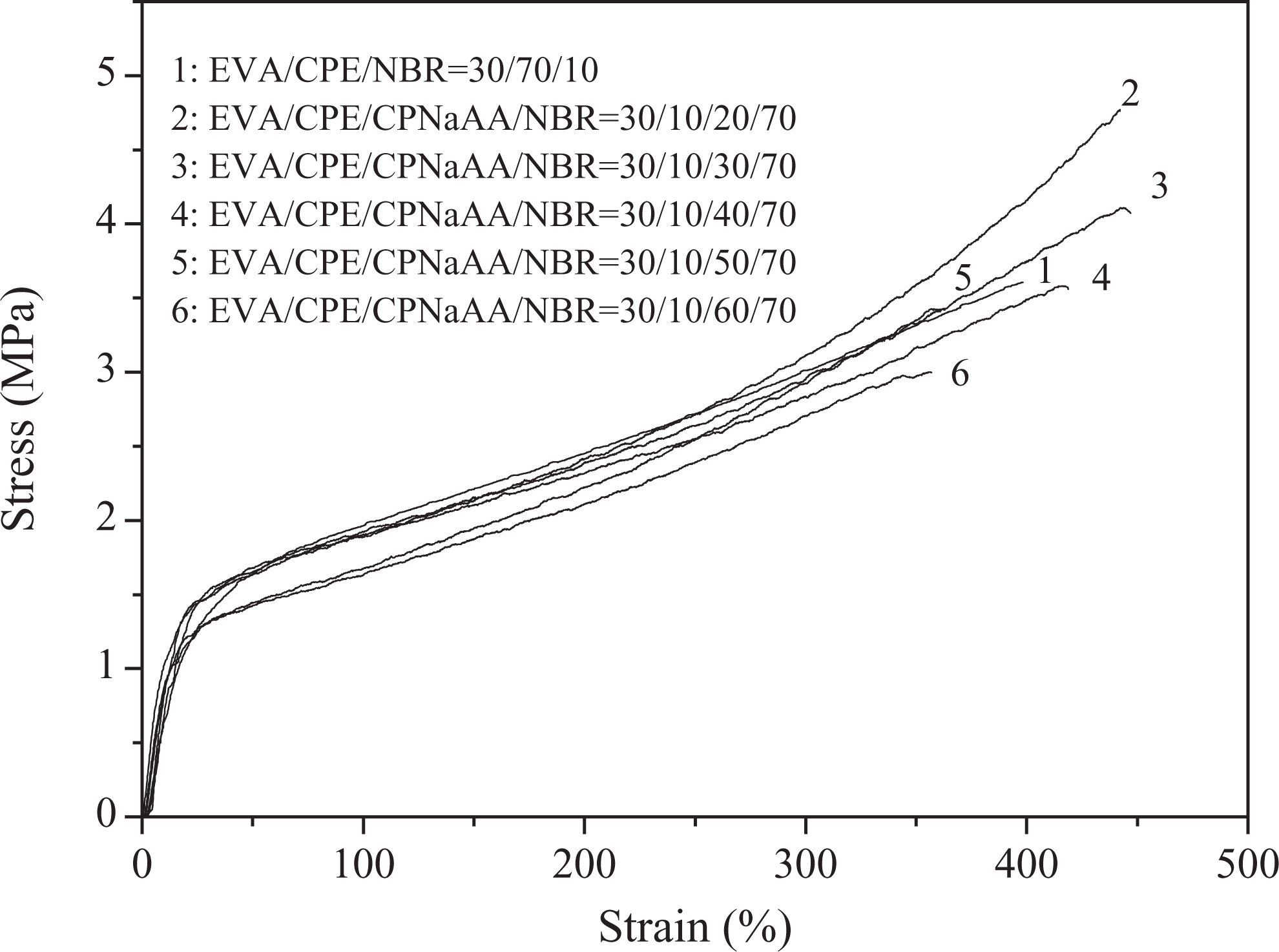
Stress–strain curves of dynamically vulcanized EVA/CPE/NBR blends with different CPNaAA dosages. EVA: ethylene–vinyl acetate copolymer; CPE: chlorinated polyethylene; NBR: nitrile butadiene rubber.
Tensile strength, elongation at break, tensile set at break, tensile set at 100% elongation, tearing strength and shore A hardness of the dynamically vulcanized EVA/CPE/NBR blends with different CPNaAA dosages are shown in Table 1. When the CPNaAA content was more than 20 phr, increasing the loading of CPNaAA in the blends led to the increased hardness and tensile set at 100% elongation, whereas the tensile strength, elongation at break, tensile set at break and tearing stress decreased remarkably. The elongation at break of the dynamically vulcanized EVA/CPE/CPNaAA/NBR blends in Table 1 was much higher than 100% and the tensile set at 100% elongation was lower than 50%; according to ASTM D1566-09, the dynamically vulcanized EVA/CPE/CPNaAA/NBR blends should be considered to be TPEs.
Mechanical properties of dynamically vulcanized EVA/CPE/NBR blends with different CPNaAA dosages.
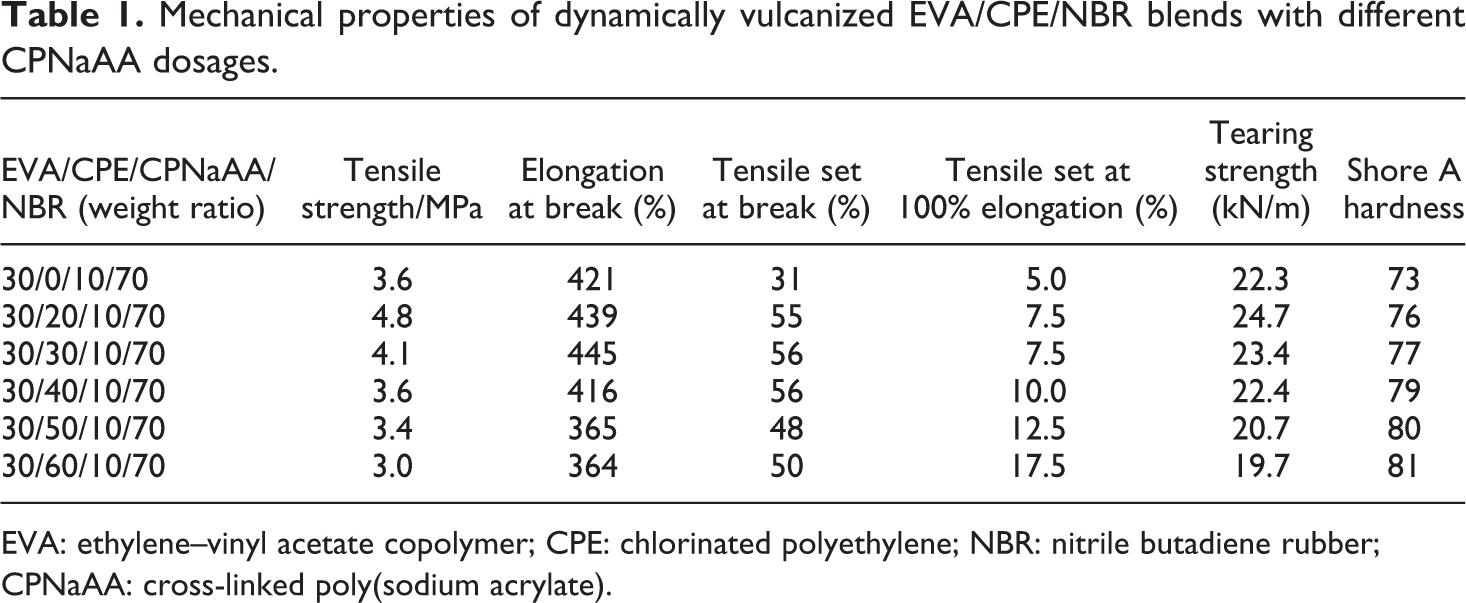
EVA: ethylene–vinyl acetate copolymer; CPE: chlorinated polyethylene; NBR: nitrile butadiene rubber; CPNaAA: cross-linked poly(sodium acrylate).
When the CPNaAA loading is low, the relatively strong interface interaction between the CPNaAA particles and the NBR matrix with strong polar nitrile would contribute to the enhanced mechanical properties. However, with the increasing dosage of CPNaAA, the cross-linking network was destroyed partly by a large number of filling with CPNaAA, which would inevitably decrease the mechanical properties and resilience. This is consistent with the results in Table 1.
Influence of CPNaAA dosage on the water-swelling behaviors of the dynamically vulcanized EVA/CPE/CPNaAA/NBR blends
The first water-swelling behaviors of EVA/CPE/CPNaAA/NBR WSRs are shown in Figure 2. When the CPNaAA content was 50 phr below, the water-swelling ratio was enhanced obviously with the increase in the CPNaAA content; the swelling ratio of WSRs with 0 phr and 50 phr are 3.2% and 669.3%, respectively. The EVA/CPE/NBR TPV almost did not swell in distilled water and the WSR with 20 phr CPNaAA dosage only showed slight water-swelling behavior. Unlike the conventional WSRs based on thermoset rubber, in our research, the cross-linked NBR rubber particles containing the CPNaAA are dispersed in the EVA/CPE thermoplastic matrix. During the water-swelling course, the water molecules need to overcome more resistance to penetrate into the rubber phase. When the CPNaAA dosage is less than 20 phr, the expansion force exerted by the water-swelling CPNaAA could not surpass the restriction resulted from the thermoplastic matrix and the vulcanized rubber, leading to a weak water-swelling behavior. With the increase in CPNaAA dosage in the WSRs, the expansion force is stronger than the restriction of WSRs, resulting in a high water-swelling behavior. As shown in Figure 2, the sample with 30 phr CPNaAA dosage showed strong water-swelling ratio at 80 h. Furthermore, with the increase in CPNaAA dosage, the water-swelling rate was increased obviously and the WSR with 60 phr CPNaAA could achieve the equilibrium swelling state just at 23 h. However, as shown in Table 2, due to the high weight loss ratio resulting from the extraction of colloidal educts during the process of immersion, the water-swelling ratio of sample with 60 phr CPNaAA is lower than that of WSR with 50 phr CPNaAA. Compared with the data in literatures, it also should be noted that the water-swelling ratio of the WSRs-based EVA/CPE/CPNaAA/NBR TPVs is much higher than that of the CPE/P (acrylic acid–acrylic amide) WSRs 13 and CPE/lithium acrylate (LiAA) WSRs prepared by in situ method; 11 the water-swelling rate is faster than ethylene propylene diene monomer/cross-linking polyacrylate WSRs 12 and CR/crosslinked sodium polyacrylate (CSP) WSRs, 18 indicating the EVA/CPE/CPNaAA/NBR WSRs are new-style WSRs with well application prospects.
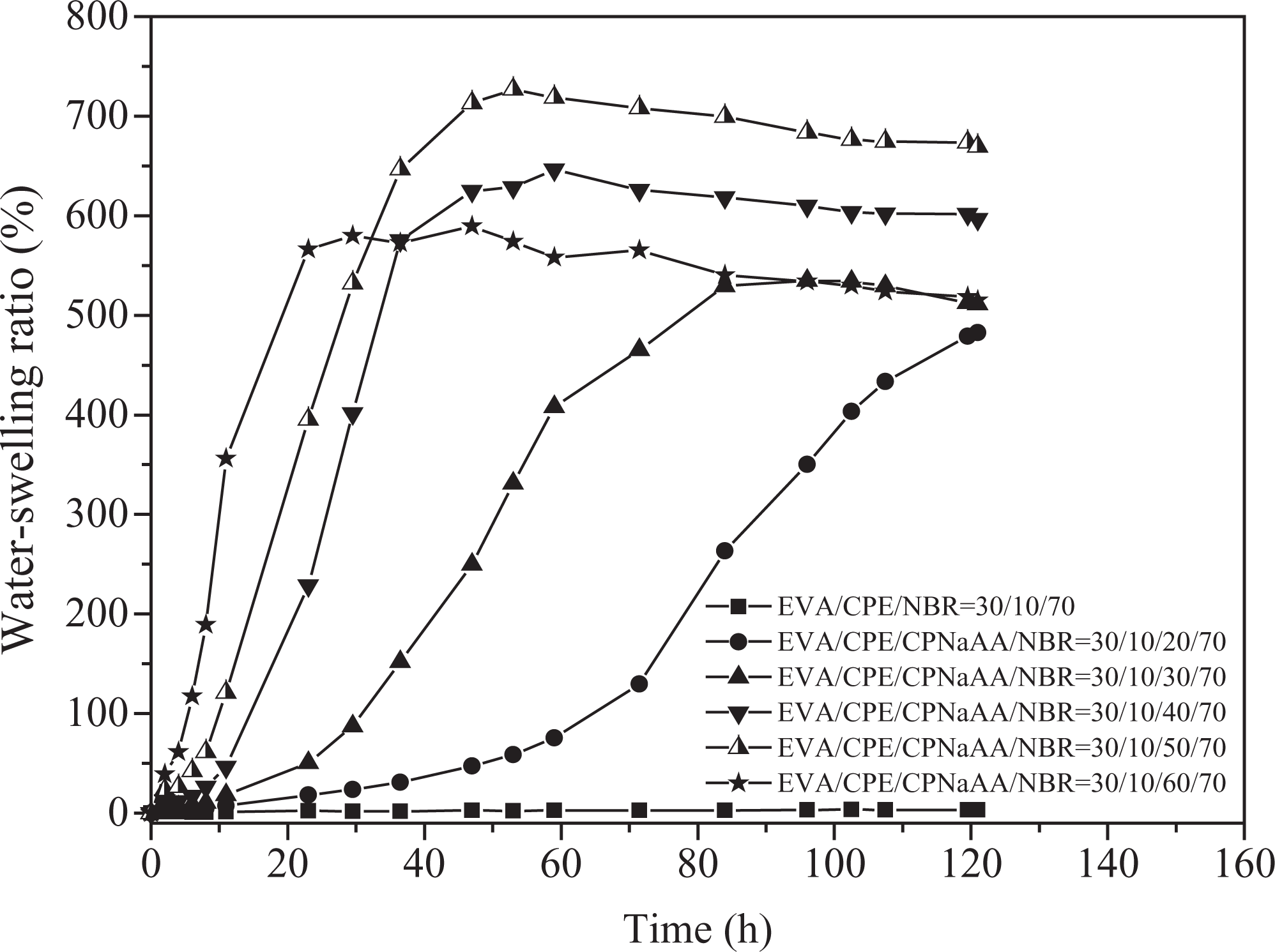
Water-swelling behaviors of dynamically vulcanized EVA/CPE/NBR blends with different CPNaAA dosages. EVA: ethylene–vinyl acetate copolymer; CPE: chlorinated polyethylene; NBR: nitrile butadiene rubber; CPNaAA: cross-linked poly(sodium acrylate).
Weight loss of the dried WSRs after repeated water-swelling process.
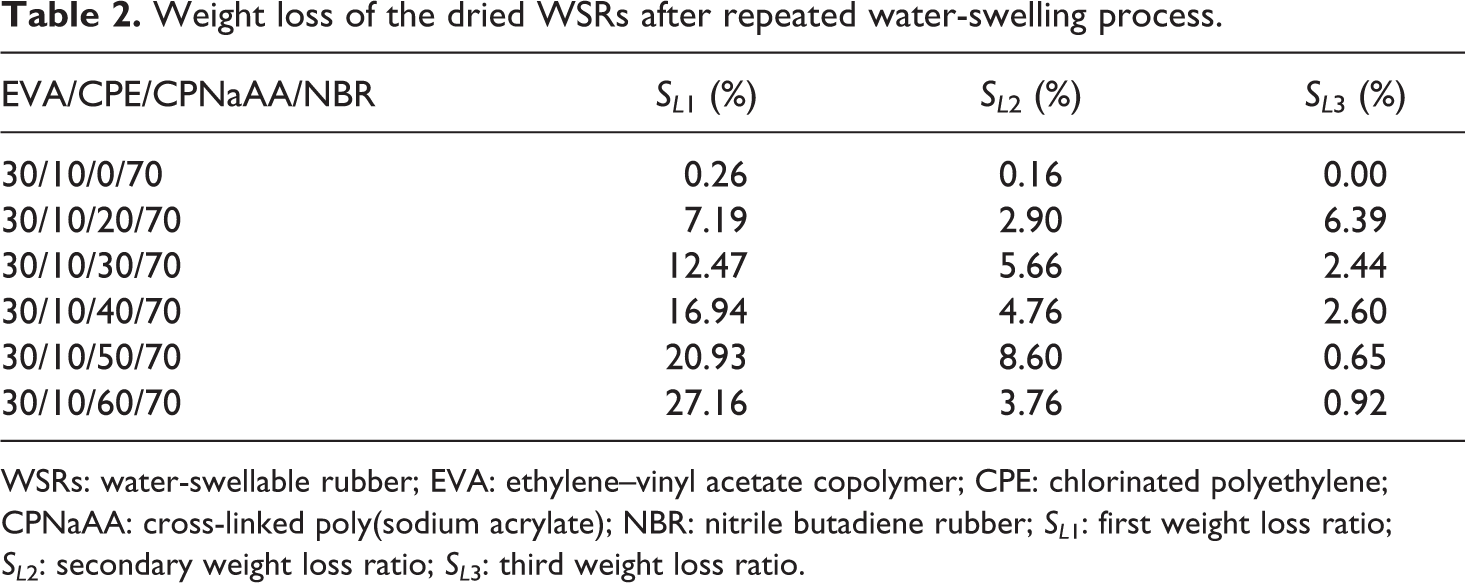
WSRs: water-swellable rubber; EVA: ethylene–vinyl acetate copolymer; CPE: chlorinated polyethylene; CPNaAA: cross-linked poly(sodium acrylate); NBR: nitrile butadiene rubber; SL 1: first weight loss ratio; SL 2: secondary weight loss ratio; SL 3: third weight loss ratio.
Table 2 shows the weight loss of the EVA/CPE/CPNaAA/NBR WSRs calculated by the mass of the dried swollen sample. It is clear that the weight loss increased remarkably with the increase in CPNaAA dosage in WSRs, especially for the samples with 60 phr CPNaAA. In our experiment, high CPNaAA dosage contribute to a high volume expansion ratio and the swollen CPNaAA particles are inclined to drop out from the cross-linked rubber network and migrate into the water through the matrix expansion gap.
In order to further evaluate the repeatability of water-swelling, the secondary and third water-swelling behaviors were researched. As shown in Figures 3 and 4, the secondary and third water-swelling ratios were enhanced obviously with the increasing content of CPNaAA. Compared with the primary water-swelling behavior, the maximum water-swelling ratio of the WSRs was only slightly lower at equilibrium swelling state. However, it was surprising that the secondary and third water-swelling rate were much higher than that of the first water-swelling behavior. The secondary water-swelling ratio of 60 phr CPNaAA sample was 542.0% after being immersed in distilled water for 24 h, and it could achieve the equilibrium swelling just at 16 h. The third water-swelling ratio of 60 phr CPNaAA sample could achieve 446.8% just at 24 h, as shown in Figure 4. For 60 phr CPNaAA sample, the CPNaAA particles in the interior were hard to migrate, thus major water swelling resins would remain during the secondary and third water swelling. In addition, from the high secondary water-swelling ratio, it can be deduced that the weight loss after the first water-swelling (Table 2) is not mere loss of CPNaAA particles.
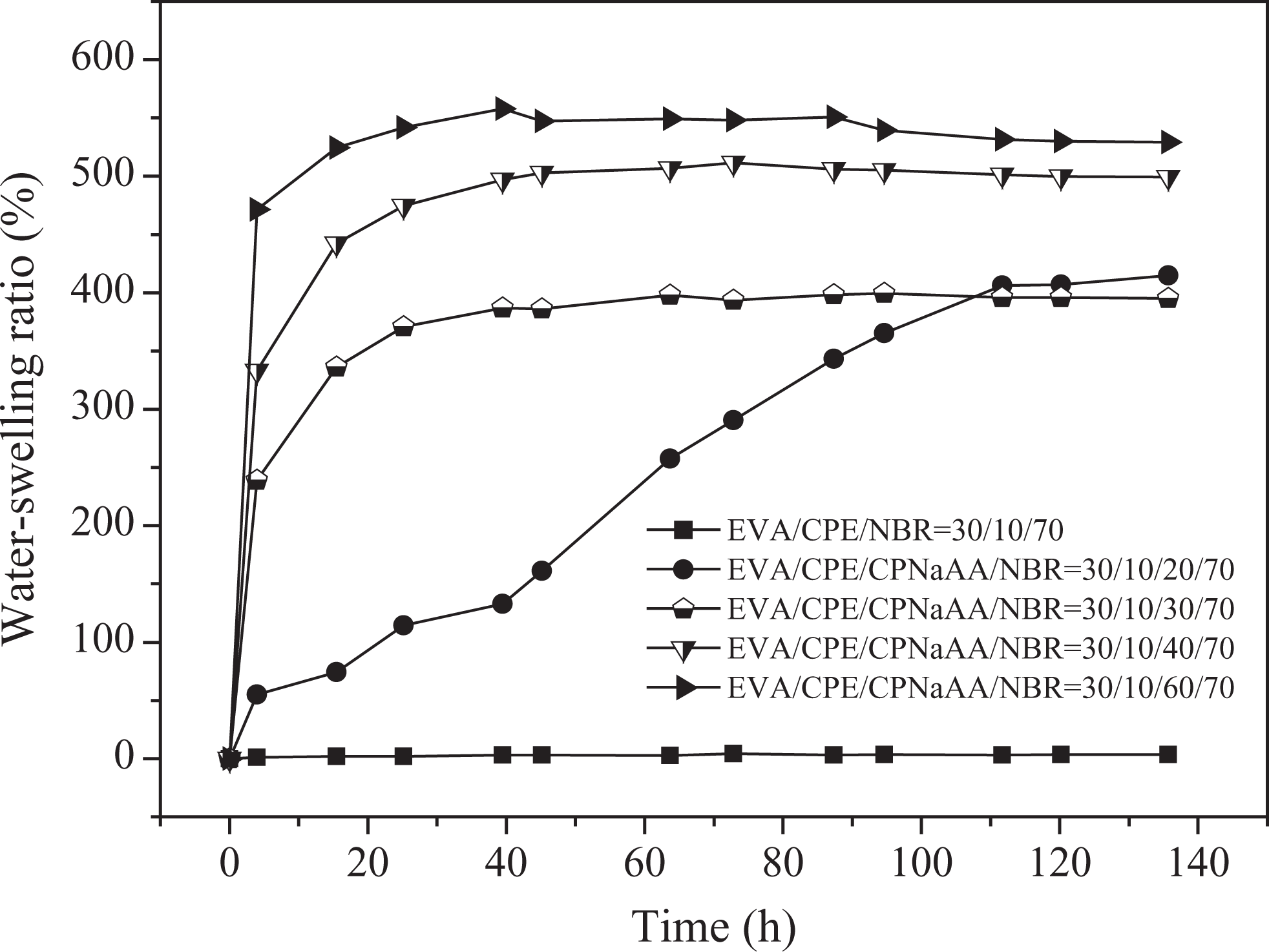
Secondary water-swelling behaviors of dynamically vulcanized EVA/CPE/NBR blends with different CPNaAA dosages. EVA: ethylene–vinyl acetate copolymer; CPE: chlorinated polyethylene; NBR: nitrile butadiene rubber; CPNaAA: cross-linked poly(sodium acrylate).
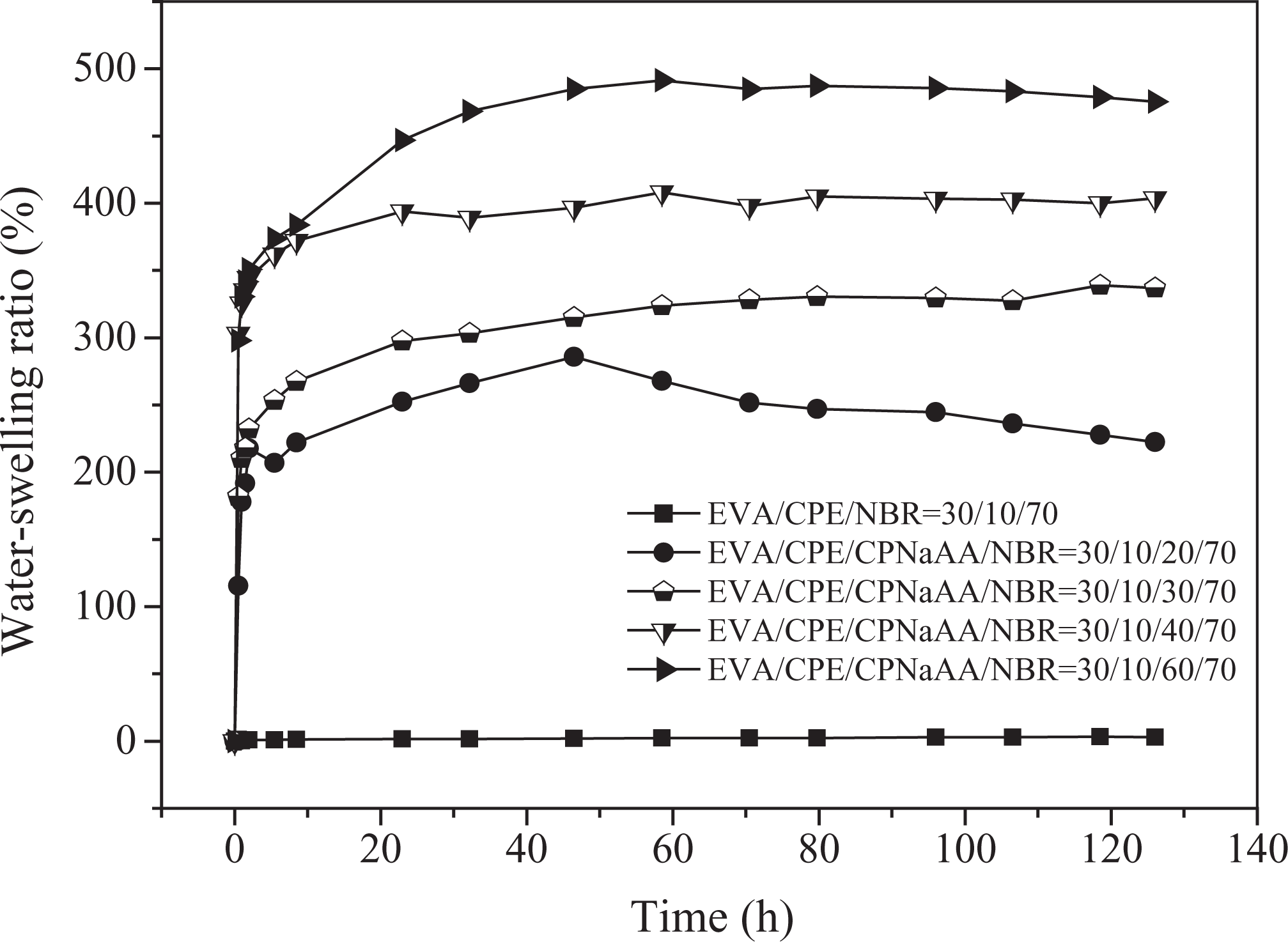
Third water-swelling behaviors of dynamically vulcanized EVA/CPE/NBR blends with different CPNaAA dosages. EVA: ethylene–vinyl acetate copolymer; CPE: chlorinated polyethylene; NBR: nitrile butadiene rubber; CPNaAA: cross-linked poly(sodium acrylate).
The weight loss of the WSRs after the secondary and third water-swelling is presented in Table 2. Compared to the weight loss after the first water-swelling, the weight loss after the secondary and third water-swelling was slight. Compared with the CPNaAA particles in the surface of WSRs, the CPNaAA particles in the inner matrix are wrapped seriously and hard to be migrated. That leads to the lower weight loss of the secondary and third water swelling, which is consistent with the results in Table 2.
Crystallization behavior of the dynamically vulcanized EVA/CPE/CPNaAA/NBR blends
XRD patterns of pure EVA, zinc oxide (ZnO) powder and EVA/CPE/CPNaAA/NBR blends are shown in Figure 5. For the pure EVA, three perceptible diffractive peaks had been observed around 21.280°, 23.619° and 36.078° corresponding to the typical (110), (200) and (020) crystallographic planes of polyethylene, indicating EVA has an orthorhombic unit cell structure. 19 Moreover, the characteristic reflections of EVA/CPE/CPNaAA/NBR blends corresponded to those of EVA and ZnO. Compared to the XRD curves of WSR with low CPNaAA content, the enhanced content of CPNaAA would reduce the intensity of diffractive peak, indicating the decreasing crystalline structure content in EVA. This was generally in agreement with the reduction of mechanical properties in high CPNaAA content WSRs. Also, the morphology of EVA spherical crystals would be observed later.
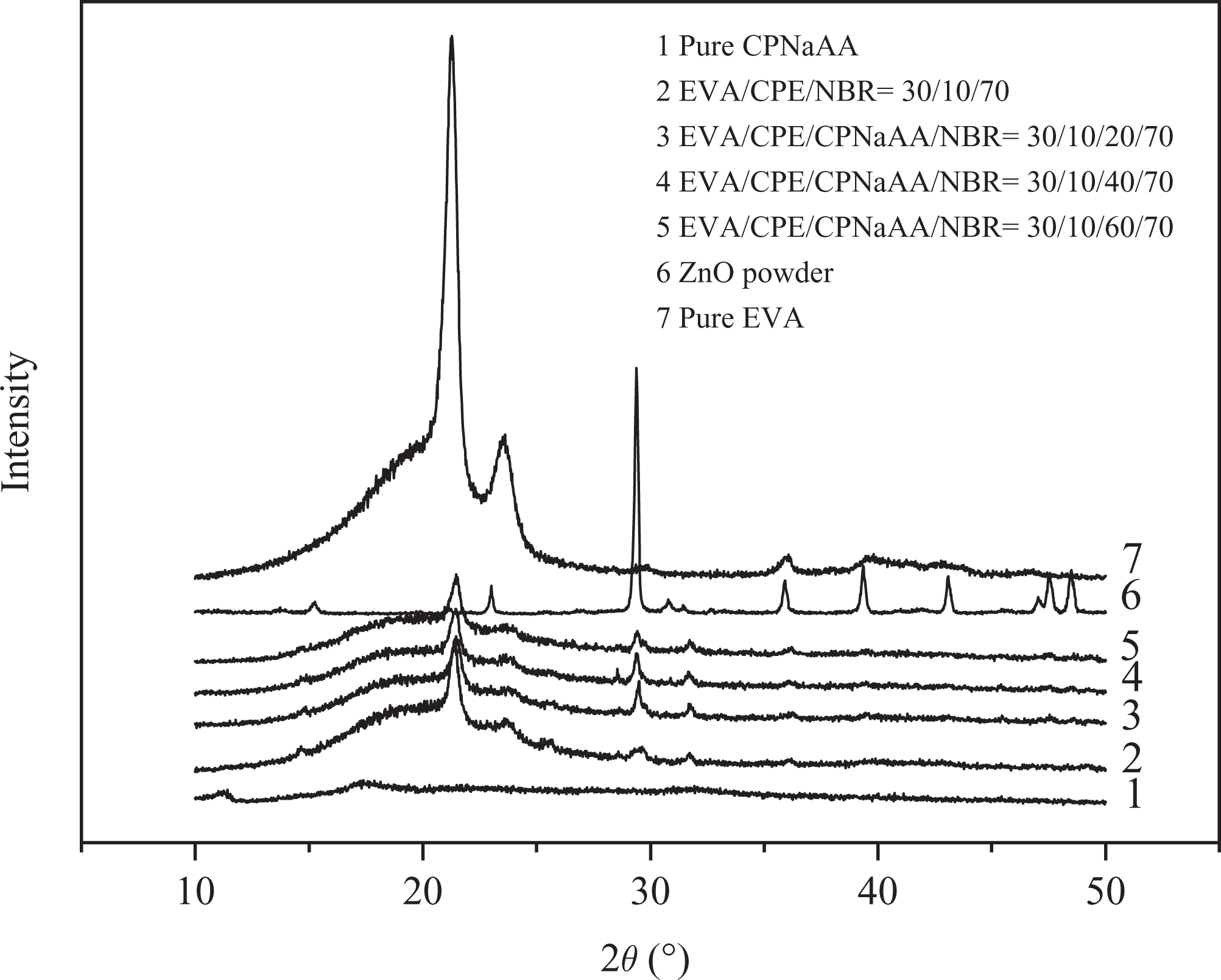
XRD patterns of EVA, ZnO powder and EVA/CPE/CPNaAA/NBR WSRs. XRD: x-ray diffraction; EVA: ethylene–vinyl acetate copolymer; ZnO: zinc oxide; CPE: chlorinated polyethylene; CPNaAA: cross-linked poly(sodium acrylate); NBR: nitrile butadiene rubber; WSRs: water-swellable rubber.
Morphological research of the dynamically vulcanized EVA/CPE/CPNaAA/NBR blends
The FE-SEM photograph of the original CPNaAA particles is shown in Figure 6, where the CPNaAA particles had irregular morphology and the dimensions were in the range of 15–150 μm.

Morphology of scanning electron microscopic image of CPNaAA particles. CPNaAA: cross-linked poly(sodium acrylate).
Figure 7 is the morphology of etched surface of EVA/CPE/CPNaAA/NBR (30/10/50/70 weight ratio) WSRs. From Figure 7(a), it can be observed that the EVA matrix on the surface of WSRs had been etched and the CPNaAA particles were observed clearly. The CPNaAA particles were dispersed evenly in the WSRs and the morphology was similar to that of Figure 6, revealing that CPNaAA particles were stable and not changed even after melt-compounding and dynamic vulcanization. The particles in Figure 7(a) and (b) were coated with cross-linked NBR layers, indicating the relatively strong interface interaction between CPNaAA and NBR vulcanizate. It is noteworthy that the crystalline structures of EVA were dispersed on the etched surface of WSRs, as shown in Figure 7(c) and (d). When the EVA/CPE/CPNaAA/NBR WSRs were etched with xylene at 100°C, the amorphous EVA phase, part crystalline EVA phase and CPE in the surface of WSRs were etched. Thus, the cross-linked NBR rubber, CPNaAA particles and the residual crystalline structure of EVA could be retained. The morphology of the clusters of flakes is spherical, indicating the flakes should be the aggregates of lamellae crystals of EVA spherical crystals, as shown clearly in Figure 7(d).
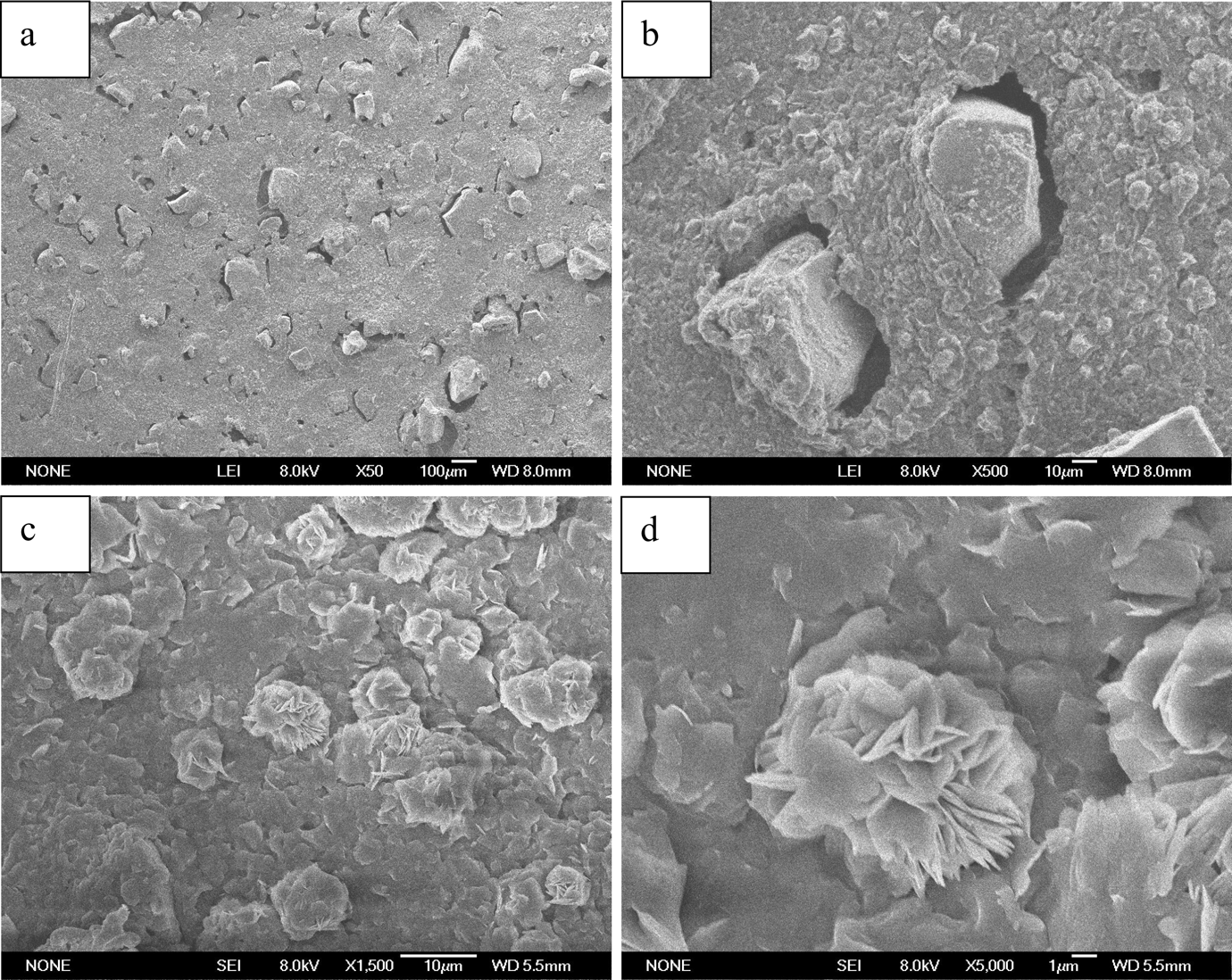
Morphology of etched surface of EVA/CPE/CPNaAA/NBR WSRs (EVA/CPE/CPNaAA/NBR = 30/10/50/70). (a) CPNaAA/NBR particles at low magnification, (b) CPNaAA/NBR particles at high magnification, (c) clusters of flakes at low magnification and (d) clusters of flakes at high magnification. EVA: ethylene–vinyl acetate copolymer; CPE: chlorinated polyethylene; CPNaAA: cross-linked poly(sodium acrylate); NBR: nitrile butadiene rubber; WSRs: water-swellable rubber.
Figure 8 presented the fracture surface of EVA/CPE/CPNaAA/NBR (30/10/20/70 weight ratio) WSRs and a loose scrap-like surface was observed. It could be found that the CPNaAA particles inserted in the fracture surface and the morphology of fracture surface implied that the interface interaction between CPNaAA and NBR was destroyed partly; moreover, some CPNaAA particles in the WSRs were separated and even pulled out from the NBR phase and EVA matrix under the high tensile stress.

Morphology of fracture surface of EVA/CPE/CPNaAA/NBR WSRs (EVA/CPE/CPNaAA/NBR = 30/10/20/70). EVA: ethylene–vinyl acetate copolymer; CPE: chlorinated polyethylene; CPNaAA: cross-linked poly(sodium acrylate); NBR: nitrile butadiene rubber; WSRs: water-swellable rubber.
Figure 9 illustrates the morphology of the surface of the dried EVA/CPE/CPNaAA/NBR (30/10/50/70 weight ratio) WSRs after first water swelling. As shown in Figure 9, some significant gaps were found in the surface, which were mainly caused by the expansion of water-swelling partials. The expansion gaps could provide channels for the water molecules to migrate into the WSRs, leading to the high equilibrium swelling rates in the secondary and third water-swelling behaviors. Meanwhile, it was noteworthy that some CPNaAA particles were coated with NBR rubber in Figure 9, some were divorced from the surface and suspended on the surface, and the significant cavities could be found on the surface, as indicated by arrows a, b and c in Figure 9, respectively. The dropped particles would lead to the cavities in the surface of WSRs and the weight loss of dried WSRs after water swelling.

Morphology of fracture surface of the dried EVA/CPE/CPNaAA/NBR WSRs after water-swelling testing (EVA/CPE/CPNaAA/NBR = 30/10/50/70). EVA: ethylene–vinyl acetate copolymer; CPE: chlorinated polyethylene; CPNaAA: cross-linked poly(sodium acrylate); NBR: nitrile butadiene rubber; WSRs: water-swellable rubber.
Conclusions
Water-swelling TPVs based on EVA/CPE/CPNaAA/NBR blends were prepared by dynamic vulcanization. The stress–strain curves of the dynamically vulcanized EVA/CPE/CPNaAA/NBR blends showed an obvious elastomer behavior. When the CPNaAA content was more than 20 phr, the increase in CPNaAA content in WSRs led to the increase in hardness and tensile set at 100% elongation, whereas tensile strength, tearing strength, the elongation at break and tensile set at break decreased. Considering the proper elasticity, strength and the high water-swelling behavior, the weight ratio of EVA/CPE/CPNaAA/NBR should be 30/10/50/70 and the water-swelling ratio was 669.3% at 120 h immersion. The WSRs with 60 phr CPNaAA achieved the equilibrium swelling at 23 h with the water-swelling ratio of 566.3%. Moreover, the secondary and third water-swelling behaviors of WSRs showed significantly high equilibrium swelling rate and the remarkable decrease in weight loss. The weight loss of the WSRs at the secondary and third water swelling is much lower than that in the first water-swelling experiment. XRD results showed that the increase in CPNaAA content would result in the reduction of crystalline structure content in EVA. Morphological study of the etched surfaces explained that the CPNaAA particles had irregular morphologies and were coated with the rubber. Moreover, FS-SEM graphs showed the crystalline structure of EVA and clusters of flakes were dispersed on the etched surface of WSRs. The FE-SEM graphs revealed that some significant expansion gaps were found in the surface of the dried water-swelled sample, which contributed to the high equilibrium swelling rates during the secondary and third water-swelling process.
Footnotes
Funding
This work was supported by a Project of Shandong Province Higher Educational Science and Technology Program (grant numbers J12LA15); the Science and Technology Development Project of Qingdao (grant numbers 12-1-4-3-(9)-jch); the Natural Science Foundation of Shandong Province (grant number ZR2012EMM002), China; and the National Natural Science Foundation of China (grant number 51272115).