Abstract
In this study, the preparation of nylon 6/talc nanocomposites was studied by melt blending in a co-rotating twin-screw extruder having length/diameter ratio of 32:1 at a screw rotation speed of 100 r min−1. Concentration of nanotalc was varied from 1 phr to 5 phr in nylon 6. The composite samples were subjected to a series of three extrusion cycles, and the effect of reprocessing on the structural properties of materials was investigated. Properties such as mechanical (tensile flexural and impact), thermal (differential scanning calorimetry), morphological (scanning electron microscopy), rheological (viscosity vs. shear rate), and colour spectrophotometry were investigated. This study would enrich the knowledge about the recycling of nylon 6, with the additional aspect of the use of collected data from more complex system, that is, composite materials, where the nanotalc particles play a role in the interactions initiated by repeated extrusion processing. Mechanical, thermal, rheological and colour properties improved with the increase in nanotalc in nylon 6 and remained nearly unchanged up to second extrusion processing. However, all the properties decreased on third extrusion processing due to the degradation of nylon 6 matrix.
Introduction
Nylon 6 is one of the largely used engineering plastics. In order to improve its performance properties, such as strength and morphology, various reinforcing agents had been used. Nanoclay takes the forefront position mainly due to its better compatibility, uncomplicated modifications and easy exfoliation happening in the nylon 6 matrix. 1 –12 Yoon et al. prepared nylon 6/clay composite fibres and investigated the effect of annealing and drawing on the crystal structure and morphology of the composite. 13 Dabrowski et al. prepared a kinetic model for the effect of heat on the properties of pristine nylon 6 and nylon 6-reinforced with nanoclay. 14 Fornes et al. prepared nylon 6/clay nanocomposite using different molecular weight nylon 6 and found that the strength and viscosity of the nanocomposite increased with the increase in both the molecular weight of nylon 6 and the concentration of nanoclay. 15 Nanoparticles of silica (to improve strength and morphology), 16 –18 graphite (to prepare electrically conducting nanocomposite), 19 carbon nanotubes (for improving strength and electrical conductivity) 20 and silver (to induce antimicrobial activity) 21 had also been used to improve the performance properties of nylon 6.
Recently nanotalc has been used as a potential reinforcing material for improving the strength of nylon 6, 22 as a substitute for clay and silica. Earlier microtalc had been added in nylon 6 to prepare their microcomposites, 23,24 in order to improve its mechanical properties. Nylon 6/talc nanocomposite, containing talc of higher aspect ratio, was prepared by Maiti and Balamurugan. 25 Sakthivel and Pitchumani synthesized nanosized talc using stirred ball mill and studied its effect on the mechanical properties of nylon 6. 26
However, polymer recycling is increasingly common for economic and environmental reasons. 27,28 Researchers have reported the effect of repeated injection-moulding cycles on the properties of nylon 66/short glass and carbon fibre composites, 29 polypropylene/flax composites, 30 nylon 6/clay nanocomposites 31,32 , whereas the effects of extrusion reprocessing have been reported for poly(acrylonitrile–butadiene–styrene)/montmorillonite nanocomposites. 33
Despite the high and increasingly applied and scientific interest in nylon 6/talc nanocomposites, there are no studies reporting the effect of extrusion reprocessing on the properties of nylon 6/talc nanocomposites. This work examines the impact of reprocessing on the properties of nylon 6/talc nanocomposites, by means of repeated extrusion cycles. Nylon 6/talc nanocomposites were prepared by melt processing and reprocessing in a co-rotating twin-screw extruder. The prepared pristine and reprocessed nanocomposites were investigated for mechanical, thermal, rheological and morphological properties. This would be one of the significant contributions to the recycling of engineering plastic containing nanoparticle reinforcements.
Experimental
Materials
Nylon 6 (NXE-01 NC) was obtained from Next Polymers Ltd (Mumbai, Maharashtra, India). Nanosized talc (approximately, particle size = 200 nm, specific surface area = 50 m 2 g−1, density = 2.76 g cm−2) was procured from Nippon Talc Co. Ltd (Osaka, Japan). Nanotalc was used as obtained, without any purification or chemical modification. Finalux G3 (wetting agent; oleochemical derivative, acid value: 3 mg potassium hydroxide g−1 sample, specific gravity: 0.85, setting point: < 20°C) was obtained from Fine Organics Pvt. Ltd (Mumbai, Maharashtra, India), which helped the nanosized talc to adhere on to the surface of nylon 6 granules during dry blending to minimize the loss brought about by its sticking to the walls of the dry blender equipment.
Methods
Dry blending
Nanotalc and nylon 6 were dried in an oven at 100°C for 12 h to remove the adsorbed and absorbed moisture. Dry blending of nanotalc and nylon 6 was performed in a tumbler mixer for 10–15 min, using Finalux G3 as the wetting agent.
Melt blending
Dry-blended nylon 6/nanotalc mix was melt blended in a co-rotating twin-screw extruder (Lab Tech Engineering Co. Ltd, Germany) having length/diameter ratio of 32:1 and temperature profile from the hopper to the dye as 165, 180, 190, 205, 215, 225, 235 and 250°C. 34 The feeder screw rotation speed was maintained at 10 r min−1, whereas the twin-screw rotation speed was kept constant at 100 r min−1. Vacuum was applied in the vent of the extruder to facilitate easy removal of moisture (if any present). Vacuum was maintained at 20 mbar. Rod profile dye was used in the extrusion process, having a diameter and a land length of 6 mm and 15 mm, respectively. Extruded strands were water cooled (30°C) and pelletized. As a simulation of the recycling process, the nylon 6/talc nanocomposites were subject to repeated cycles of extrusion till degradation in the performance properties was observed.
Injection moulding
Pellets were dried in an oven at 100°C for 10 h to remove the adsorbed and absorbed moisture. Injection moulding (Boolani Machineries India Ltd, Mumbai, Maharashtra, India) was performed by maintaining the temperature profile as 210, 230 and 250°C from the hopper to the ejection nozzle. Standard ASTM samples for tensile (ASTM D638), flexural (ASTM D790) and impact (ASTM D256) testing were obtained from injection moulding. Injection pressure, packing pressure and cooling time were maintained constant at 115 MPa, 50 MPa, and 1 min, respectively.
Samples for impact testing were notch cut before testing. The formulations prepared are shown in Table 1. Concentration of nanotalc was varied from 0 phr to 5 phr of nylon 6, whereas the concentration of the wetting agent (Finalux G3) was maintained constant at 2 phr of nylon 6.
Prepared nylon 6/talc nanocomposite formulations.
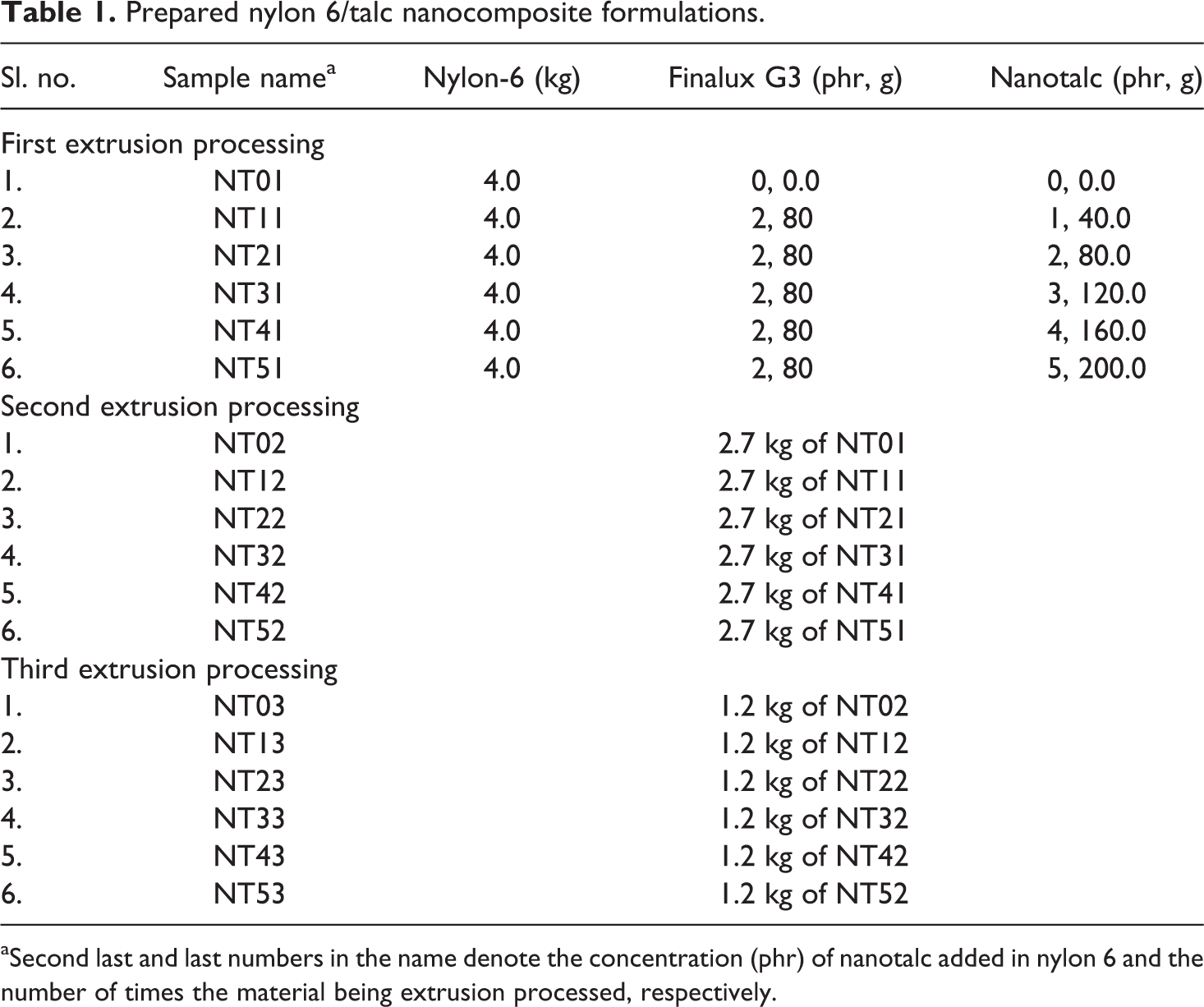
aSecond last and last numbers in the name denote the concentration (phr) of nanotalc added in nylon 6 and the number of times the material being extrusion processed, respectively.
Characterization and testing
Mechanical properties
Tensile properties (tensile strength, tensile modulus and percentage elongation at break) and flexural properties (flexural strength and flexural modulus) were measured at ambient condition using a universal testing machine (LR-50 K, Lloyds Instrument, UK), according to ASTM procedures D638 and D790, at a crosshead speed of 50 and 2.8 mm min−1, respectively. The notch for impact test was made using a motorized notch-cutting machine (Polytest model 1, Ray-Ran, UK). Cutter height was adjusted such that it cuts a notch that leaves 10.16 ± 0.05 mm of the material remaining under the apex of the notch in a single pass and a radius of curvature at the apex of 0.25 ± 0.05 mm with a notch angle of 45°. Notched Izod impact strength was determined at ambient condition according to ASTM D256 standard, using impact tester (Avery Denison, UK) having striking velocity of 3.46 m s−1, employing a 2.7-J striker. During the test, surrounding temperature and relative humidity were maintained constant at 23 ± 2°C and 50 ± 5%, respectively. A minimum of five specimens were tested for each reported value.
Thermal properties
Differential scanning calorimetry (DSC; Q 100 DSC, TA Instruments Ltd, Bengaluru, Karnataka, India) characterization was done to investigate the crystallization and melting behaviour of the prepared nanocomposites. Two consecutive heating scans were performed to minimize the influence of possible residual stresses in the material due to any specific thermal history. A scanning rate of 10°C min−1 was maintained for both heating and cooling cycles, whereas nitrogen gas purge rate was maintained at 50 ml min−1. Melting temperature (Tm, peak maximum) and enthalpy of melting (Hm, melting peak area) were determined from the second heating scan, whereas crystallization temperature (Tc, peak minimum) and enthalpy of crystallization (Hc, crystallization peak area) were determined from the only cooling scan. Crystallinity was calculated assuming an Hm of 190.6 J g−1 for 100% crystalline nylon 6 using equation (1).
31

Rheological properties
The melt viscosity (pascal second) was measured using a rotational rheometer (MCR101, Anton Paar, Gurgaon, Haryana, India) with parallel-plate assembly, having a diameter of 35 mm. Samples were predried before analysis. Viscosity was determined for shear rates from 0.01 s–1 to 100 s–1, at a constant temperature of 250°C.
Morphological properties
Scanning electron microscopic (SEM) analysis was performed using JEOL 6380 LA (Japan). Samples were fractured under liquid nitrogen to avoid any disturbance to the molecular structure and then coated with gold before imaging. Accelerating voltage was maintained constant at 15 kV. Images were captured at a resolution of 35,000×. This study helps to understand the dispersion of nanosized talc in nylon 6.
XRD analysis
The XRD analysis was carried out to determine the percentage crystallinity of the prepared composite. A normal focus copper (Cu) X-ray tube was operated at 30 kV and 15 mA. Sample scanning was done from 10° to 60° at a rate of 3° min−1. The data processing was done using Jade 6.0 software. The X-ray of the Cu Kα radiation filtered by a nickel filter has a wavelength of 1.54178 Å. 35
FTIR analysis
The Fourier transform infrared (FTIR) spectra were recorded with PerkinElmer Spectrum GX equipment (Waltham, Massachusetts, USA). Samples were compression moulded at 250°C to prepare thin films of about 0.1 mm thickness. These thin films were subjected to FTIR analysis. Samples were scanned with a resolution of 2 cm−1 in the scan range of 450–4000 cm−1.
Colourimetric properties
International Commission on Illumination (CIE) L*, a*, b* values for the composites were determined using colour ppectrophotometer (Color Eye 7000, Optiview Light Quality Control 1.9, Gretag Macbeth, Germany). Illuminant used was D65. Observer was placed at 10°.
Results and discussion
Mechanical properties
Mechanical properties such as tensile strength (MPa), tensile modulus (MPa), elongation at break (%), flexural strength (MPa), flexural modulus (MPa) and impact strength (J m−1) obtained for nylon 6/talc nanocomposites on first, second and third extrusion processing are listed in Table 2. Tensile strength, tensile modulus, elongation at break, flexural strength, flexural modulus and impact strength for pristine nylon 6, extrusion processed only once, were found to be 49.0 MPa, 572.3 MPa, 315.8%, 48.2 MPa, 1420.7 MPa and 57 J m−1, respectively.
Mechanical properties obtained for nylon 6/talc nanocomposites on extrusion reprocessing.
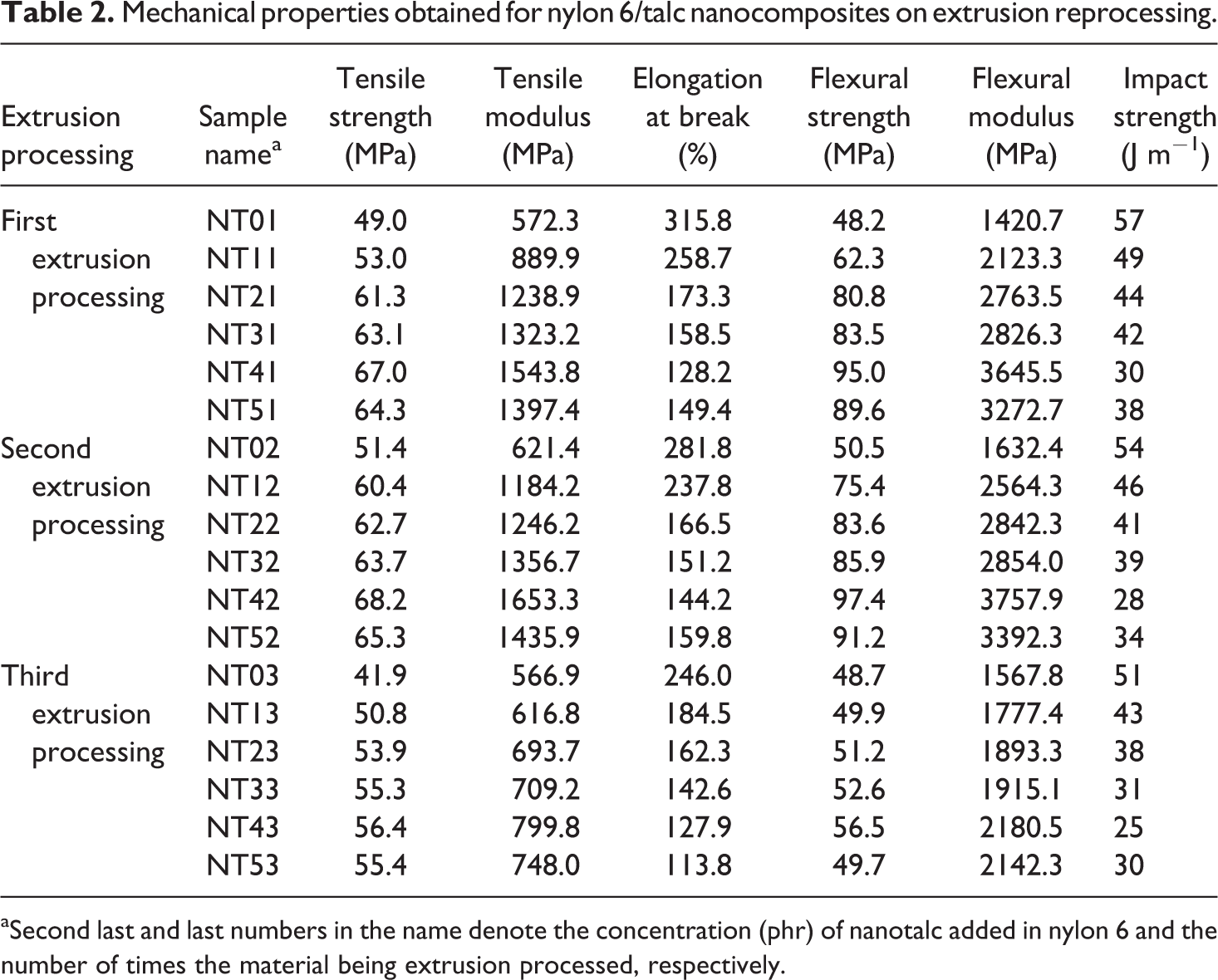
aSecond last and last numbers in the name denote the concentration (phr) of nanotalc added in nylon 6 and the number of times the material being extrusion processed, respectively.
In the first extrusion processing cycle, it was found that tensile strength, tensile modulus, flexural strength and flexural modulus increased by 36.7, 169.7, 97.1 and 156.%, respectively for 4 phr nanotalc-loaded nylon 6 (NT41) as compared to pristine nylon 6. The increase in properties is highly appreciable for 4 phr loading of nanotalc in nylon 6. Both talc and nylon 6 are hydrophilic in nature; thus, they showed better compatibility when brought together to prepare a composite. This compatibility led to better interaction between them, increasing the modulus and strength of the composite. In addition, the nanosize of talc helps in providing very high surface area, compared to the weight added. However, the most important reason is the uniform and proper distribution of nanotalc in nylon 6 matrix. All these factors exponentially increased the number of interaction points between nanotalc and nylon 6. At 5 phr concentration of nanotalc, the properties of the nanocomposite (NT51) degrade due to the formation of aggregates (as evident from SEM shown later). These aggregates led to a decrease in the effective surface area available to interact with the nylon 6 polymeric chains and also generated points of stress concentrates in the region of aggregates, decreasing the performance properties. Percentage elongation at break and impact strength steadily decreased with the increase in the concentration of nanotalc in nylon 6. Due to the increase in modulus, the elongation property of the composite decreased, decreasing the toughness, and thus, the impact strength of the composite. However, for NT51, due to the formation of aggregates and decrease in modulus, percentage elongation at break and impact strength increased as compared to 4 phr nanotalc loaded nylon 6. 36 –38
The second extrusion processing of the prepared nanocomposites slightly increased the tensile strength, tensile modulus, flexural strength and flexural modulus but decreased the percentage elongation at break and impact strength. This happened due to more uniform distribution of nanotalc in nylon 6 matrix (for NT12 to NT42) and breaking of certain aggregates, converting them into nanosize again, due to the re-shearing in the extrusion screws (for NT52).
However, on third extrusion processing of the prepared composites, properties degraded drastically. For NT43, tensile strength, tensile modulus, flexural strength and flexural modulus decreased by 15.8, 48.2, 40.5 and 40.2%, respectively, as compared to NT41. Surprisingly, percentage elongation at break and impact strength also decreased as compared to NT41. However, tensile strength, tensile modulus, flexural strength and flexural modulus increased with an increase in the concentration of nanotalc. This means that nanotalc is still playing its role as a reinforcing agent. But the increase in properties is very less. This gives the direct indication of the degradation happening of the nylon 6 matrix, decreasing its molecular weight. This was confirmed through thermal, rheological and colour tests.
XRD analysis
Percentage crystallinity values obtained for nanotalc and nylon 6/talc (1–5 phr) after first, second and third extrusion processing are listed in Table 3, whilst the X-ray diffractograms are shown in Figure 1.
Crystallinity values obtained for nylon 6/talc nanocomposites on extrusion reprocessing using X-ray diffractometer.
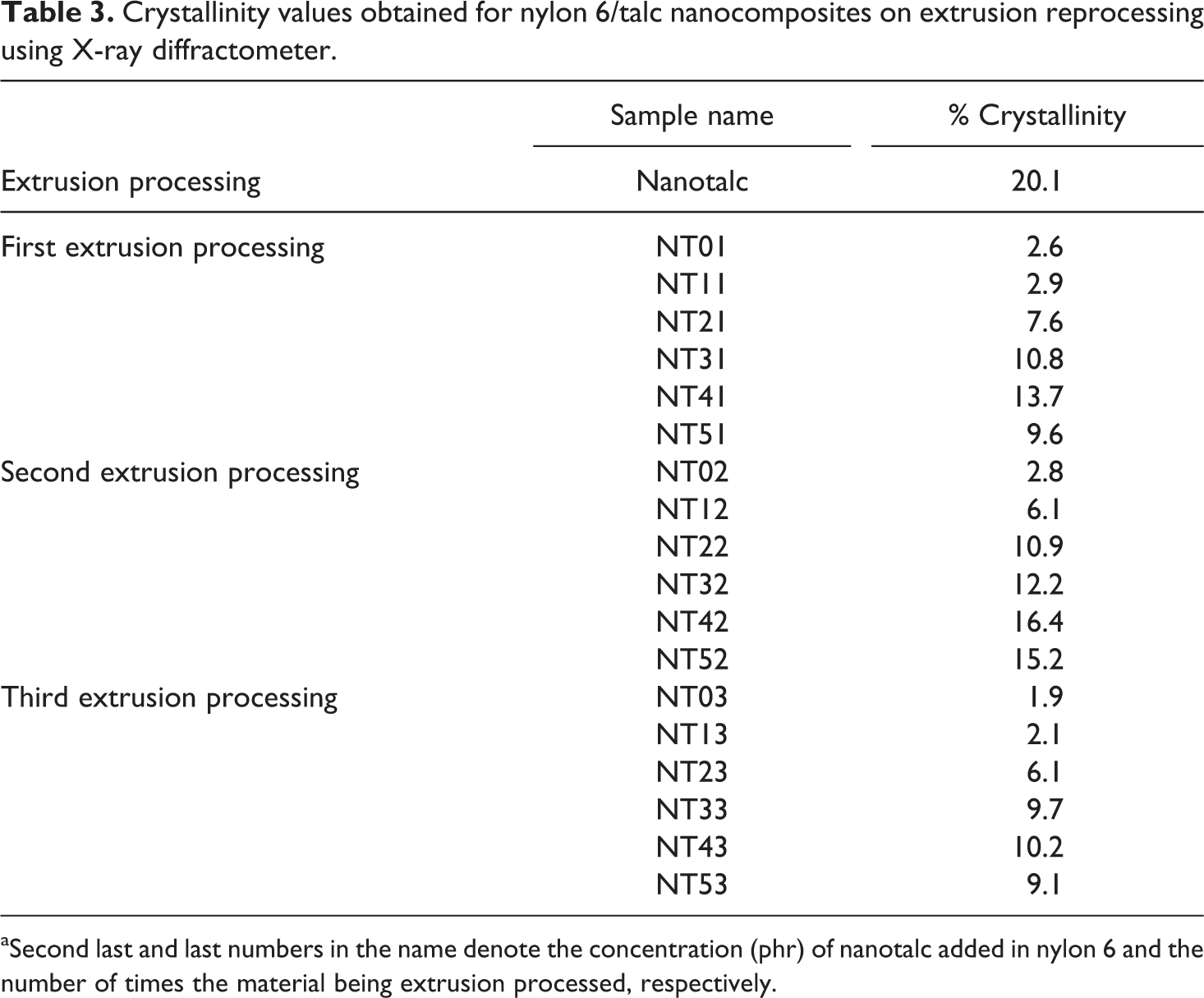
aSecond last and last numbers in the name denote the concentration (phr) of nanotalc added in nylon 6 and the number of times the material being extrusion processed, respectively.
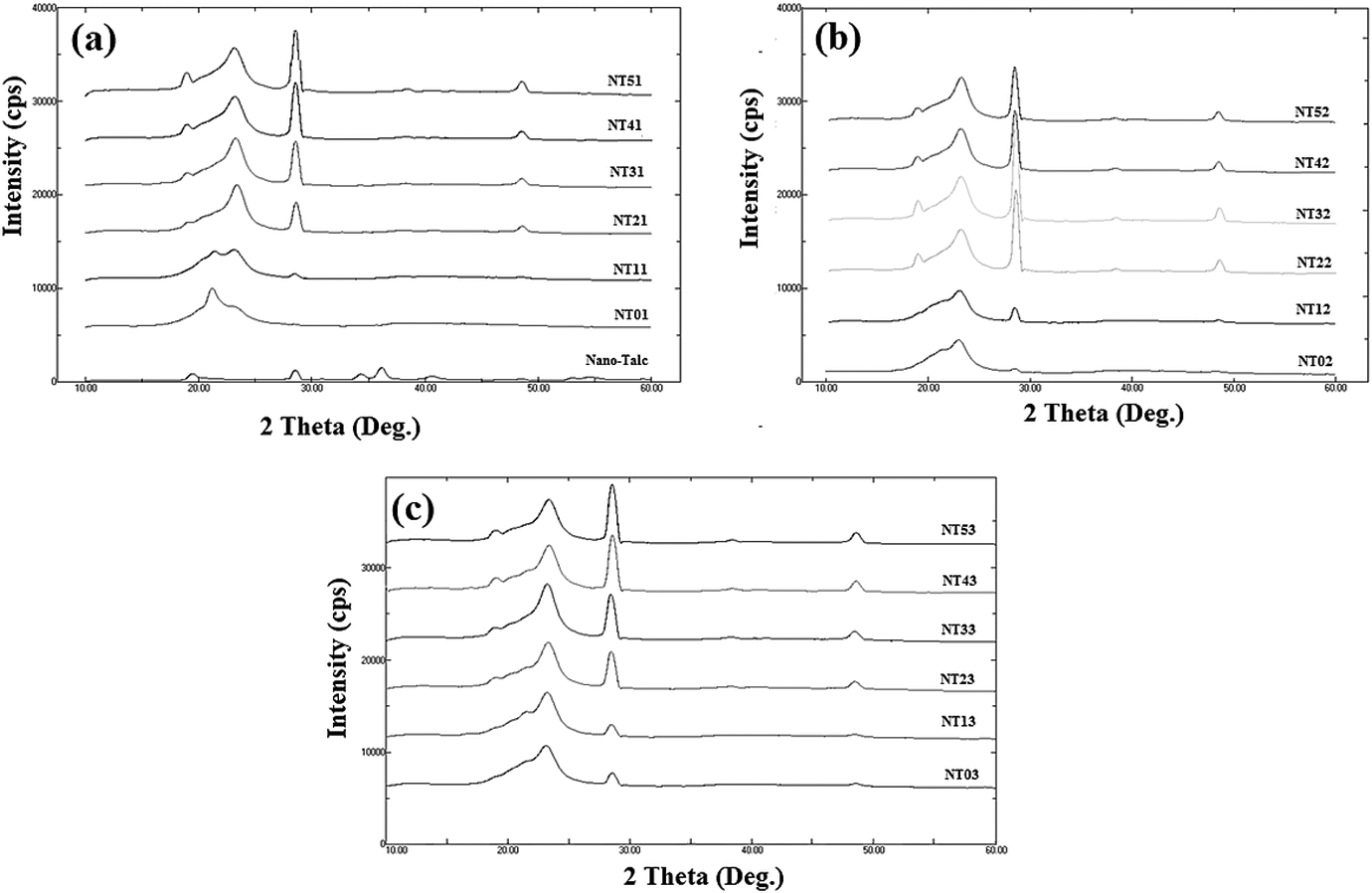
X-ray diffractograms obtained for nanotalc and nylon 6/talc (1–5 phr) nanocomposites after (a) first, (b) second and (c) third extrusion processing.
Nanotalc was found to have crystallinity of about 20% and pristine nylon 6 of about 2.5%. The prepared nanocomposites had crystallinity in between them. All the composites showed talc-specific peaks at about 28° and 48°. Intensity of these peaks increased with increase in the concentration of nanotalc in the nylon 6 matrix. Also the crystallinity of the nanocomposites increased with increase in the concentration of nanotalc.
Nylon 6 and talc, both being hydrophilic, had better interaction with each other. This helped in better alignment of nylon 6 molecules over that of nanotalc, which was the reason for increased crystallinity of the composite compared to nylon 6 without nanotalc. As the concentration of nanotalc increased, surface area available for interacting with nylon 6 also increased. These higher interactions increased the crystallinity of the composite by making the nylon 6 polymer chains get orient about it. Crystallinity increased for the two times extrusion processed samples than the one time extrusion-processed samples. This must be due to the more proper dispersion of nanotalc in the nylon 6 matrix on second time processing in the extruder. However, crystallinity decreased for the three times extruded samples. This happened due to the degradation of the nylon 6 chains. Due to the third processing of the samples in the extrusion, the shearing of the screw and repeated high-temperature exposure caused the polymeric chains of nylon 6 to break down to low-molecular-weight segments, making them stiffer to align about nanotalc. In addition, due to degradation, the number of nylon 6 molecules increased as compared to the available nanotalc particles, decreasing the number of interaction points between nanotalc and nylon 6. This led to decrease in the crystallinity of the samples processed three times in the extrusion machine. Even for the three times extruded samples, it was found that crystallinity increased with the increased concentration of nanotalc. This confirms the above-mentioned (statement mentioned in mechanical properties) statement regarding nanotalc still playing its role as a reinforcing agent in nylon 6 matrix, even in three times extrusion processed samples.
Thermal properties
DSC thermograms, both heating and cooling scans, for the prepared nylon 6/talc (1–5 phr) nanocomposites obtained on one, two and three times extrusion processing are shown in Figures 2 and 3. Values obtained for Tm, Hm, Tc, Hc and crystallinity (C) are listed in Table 4.
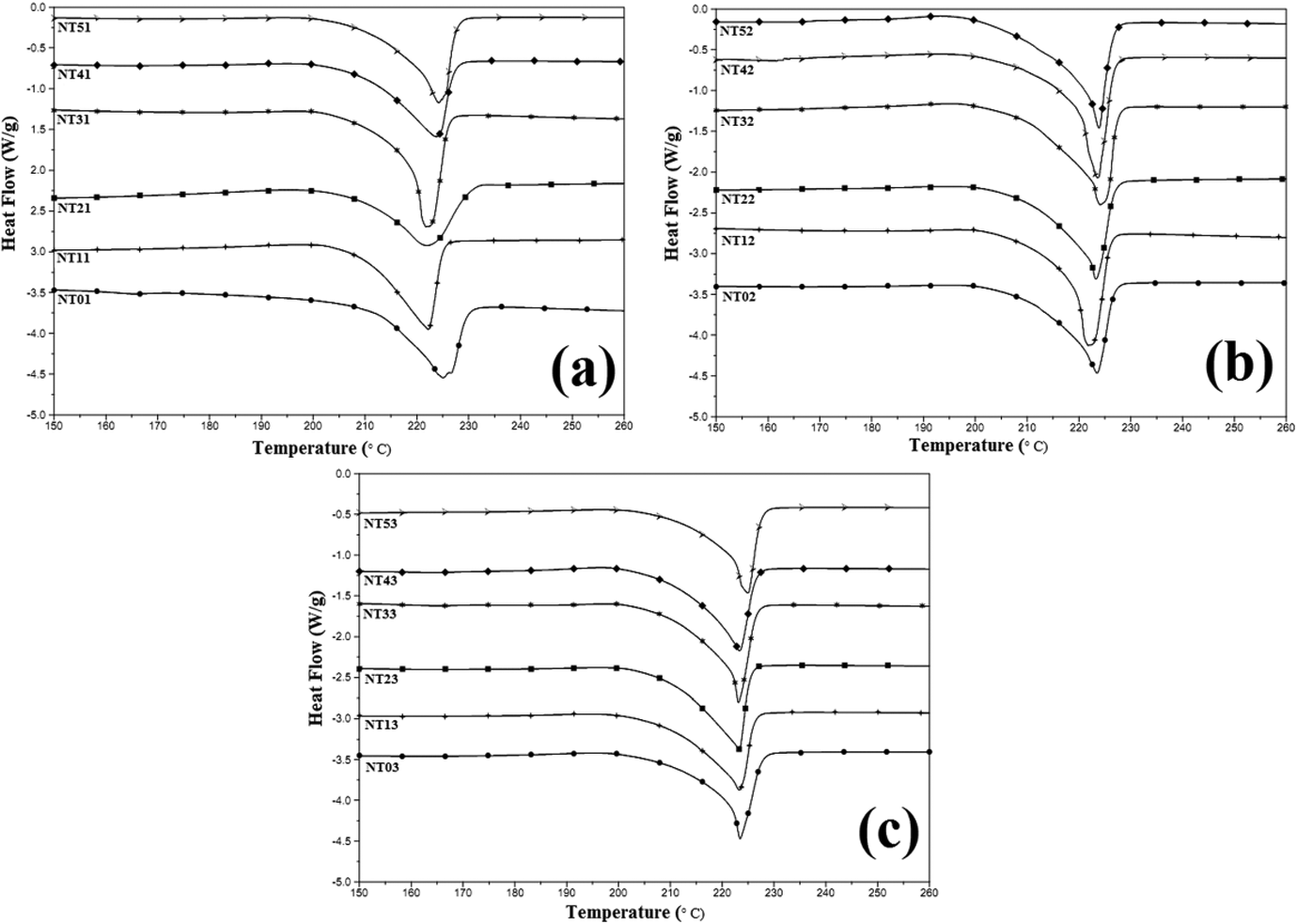
Differential scanning thermograms obtained for nylon 6/talc (1–5 phr) nanocomposites after (a) first, (b) second and (c) third extrusion processing – heating scans.
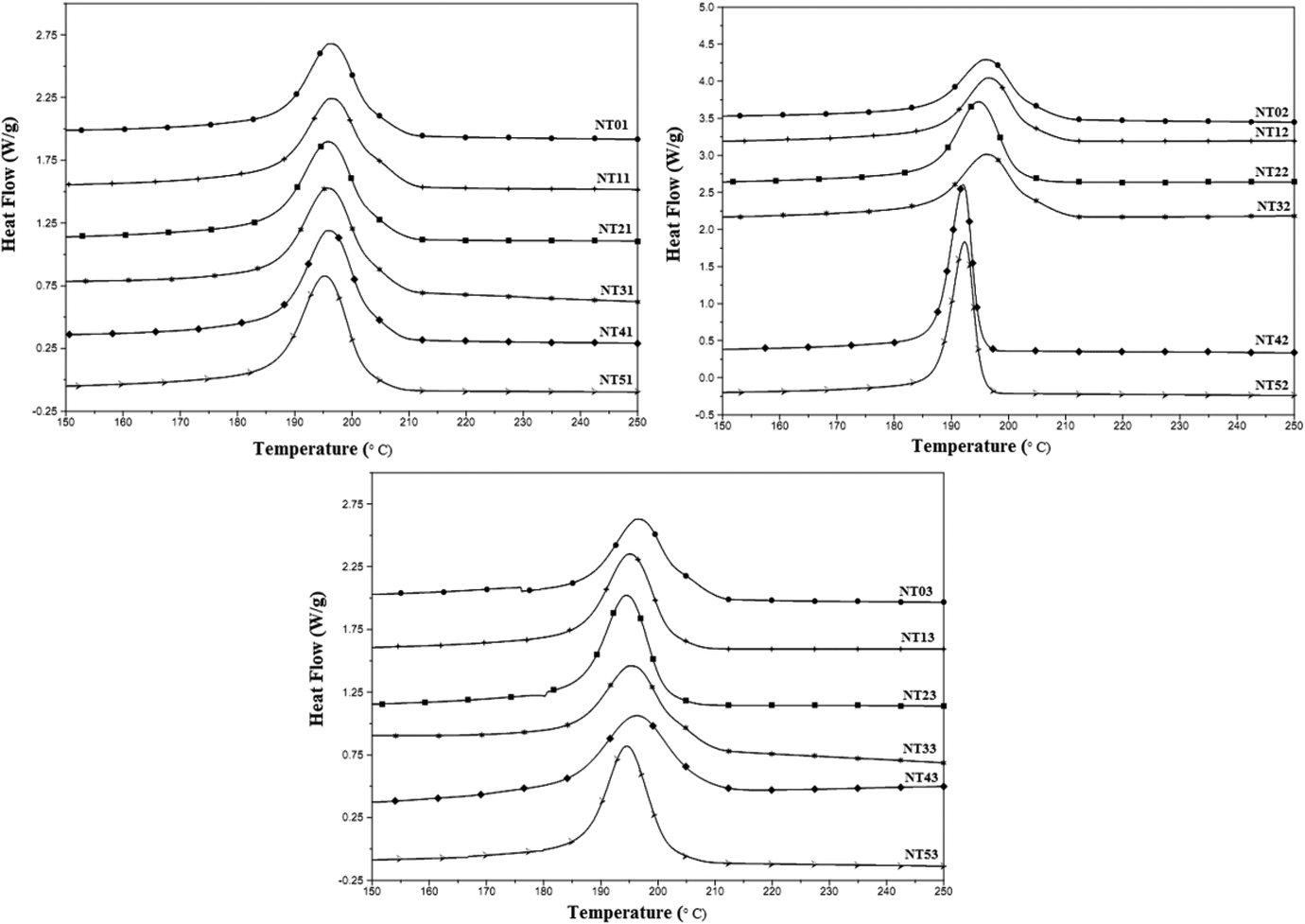
Differential scanning thermograms obtained for nylon 6/talc (1–5 phr) nanocomposites after (a) first, (b) second and (c) third extrusion processing – cooling scans.
Thermal properties obtained for nylon 6/talc nanocomposites on extrusion reprocessing.
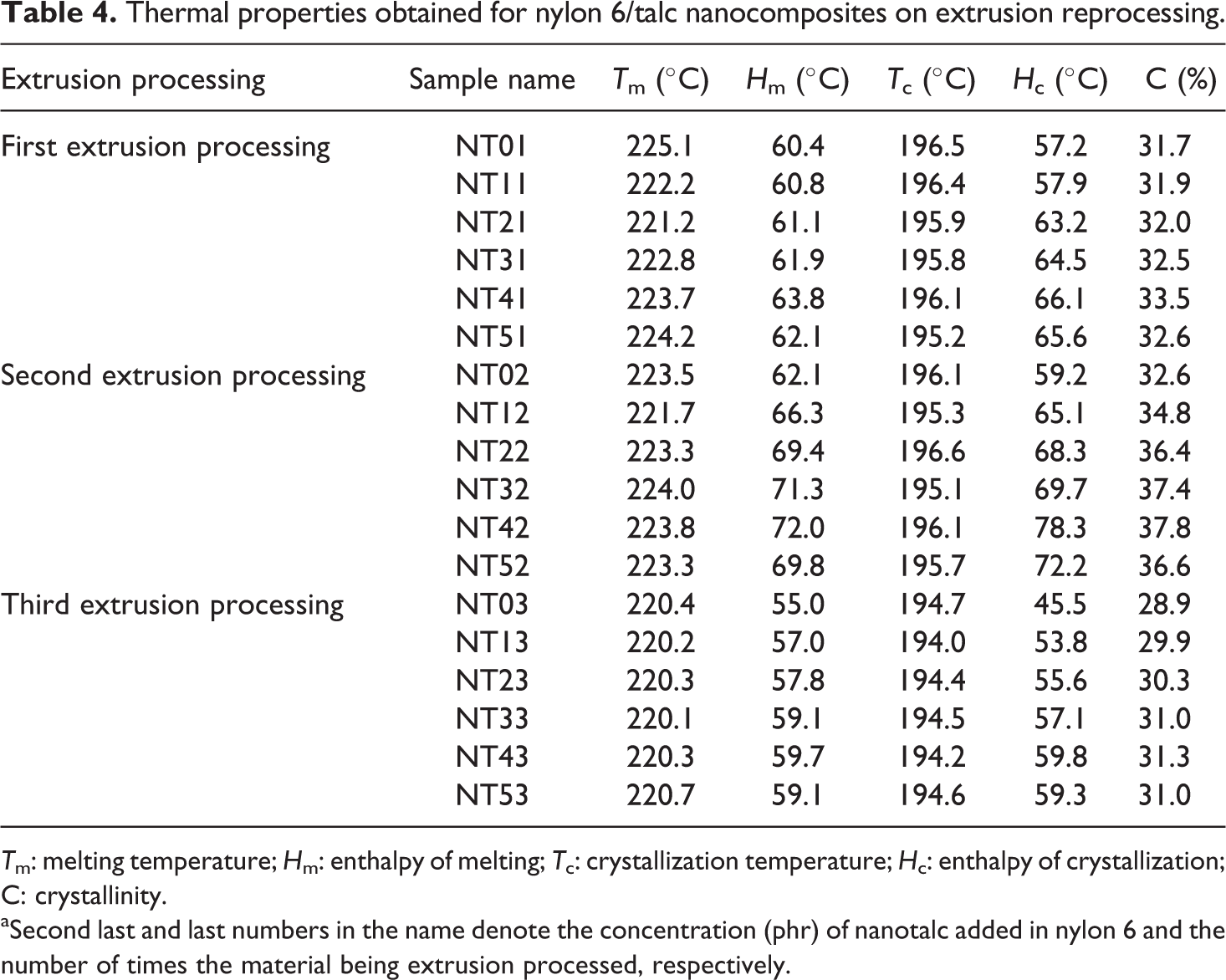
Tm: melting temperature; Hm: enthalpy of melting; Tc: crystallization temperature; Hc: enthalpy of crystallization; C: crystallinity.
aSecond last and last numbers in the name denote the concentration (phr) of nanotalc added in nylon 6 and the number of times the material being extrusion processed, respectively.
For the one-time extrusion processed composites, it was found that the Hm, C and Hm increased with increase in the concentration of nanotalc in the nylon 6 matrix, with no such effect on the Tm or Tc of the composite. This proves the reinforcing effect of nanotalc in nylon 6. Due to better compatibility and interaction between nylon 6 and nanotalc, nylon 6 polymer chains got oriented about nanotalc particles, increasing the crystallinity of the system. This led to increase in the heat required for breaking the strong intermolecular forces of attraction, generated due to increased crystallinity, thus increasing the Hm. Similarly, during cooling scans, as the composite was being cooled (after the melting stage), the higher crystallinity induced due to the nanotalc particles increased the heat to be released during crystallization. This phenomenon became more pronounced with an increase in the concentration of nanotalc. But at 5 phr concentration, the properties decreased slightly, which was due to the formation of nanotalc aggregates, leading to the decrease in the effective surface area for bonding with nylon 6. This led to a decrease in crystallinity of the system, and thus, the properties.
For the two times extrusion processed nanocomposites, Hm, C and Hc increased as compared to the one-time extrusion processed nanocomposites, with no effect as such on Tm and Tc. This must have happened due to the more proper distribution of nanotalc in the nylon 6 matrix, due to the re-shearing happening in the extrusion process. Again, Hm, C and Hc decreased for 5 phr nanotalc loaded nylon 6, which was again attributed to the formation of nanotalc aggregates, decreasing the effective surface area for interacting with nylon 6 matrix. This led to decrease in the heat required for melting and released during crystallization. But Hm, C and Hc of NT52 is higher than NT51. Second extrusion processing broke down the aggregates (if any) back to the nanosize, decreasing the number of aggregates. This increased the effective surface area for interacting and bonding with nylon 6 polymer chains, increasing the C value and thus the Hm, and Hc.
However, the values of Hm, C and Hc decreased appreciably for the three times extrusion processed composites. Even Tm and Tc values decreased for the reprocessed composites. However, the addition of nanotalc had the same effect as discussed above. This proves that the nanotalc has not got aggregated on reprocessing, but is still playing its role as a reinforcing agent. However, the appreciable decrease in the values of Hm, Hc, Tm and Tc was due to the degradation of the nylon 6 matrix. This degradation led to a decrease in its molecular weight (increasing the number of nylon 6 molecules) and decreased the number of interaction points between nylon 6 and nanotalc. Thus, crystallinity decreased, decreasing the thermal properties. Even rheological studies proved the observed behaviour.
Rheological properties
Graphs of viscosity versus shear rate obtained for the prepared nylon 6/talc (1–5 phr) nanocomposites on one, two and three times extrusion processing are shown in Figure 4.
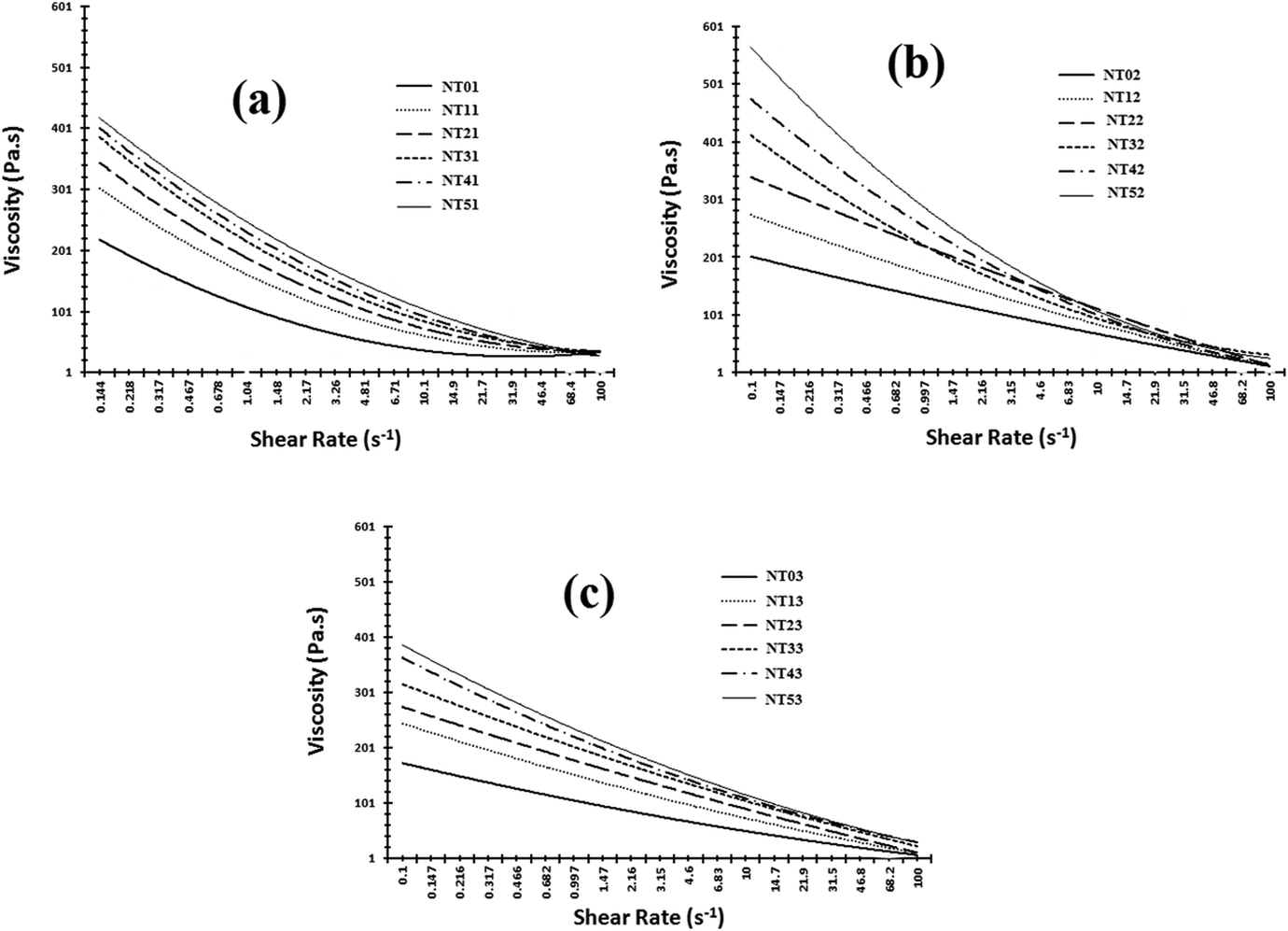
Rheological properties (viscosity vs. shear rate) obtained for nylon 6/talc (1–5 phr) nanocomposites after (a) first, (b) second and (c) third extrusion processing.
All the composite samples showed decrease in viscosity with increase in the shear rate, which is a characteristic behaviour of a thermoplastic material, called as the shear-thinning behaviour. 39 –41 The viscosity of the nylon 6 increased with increased addition of nanotalc, whether it was extrusion processed once, twice or thrice. Nanotalc and nylon 6 formed a completely compatible mixture, as both are hydrophilic in nature. This led to a better interaction between them, making nylon 6 molecules to get properly oriented about nanotalc particles, increasing the crystallinity of the system. Crystallinity and intermolecular forces of attraction increased with increase in the concentration of nanotalc in nylon 6, increasing the viscosity. However, no appreciable difference was observed in viscosities between NT4 and NT5 in one and three times extrusion processed samples. But for the two times extrusion processed samples, appreciable difference was observed between the viscosities of NT4 and NT5. This happened due to the shearing action of the extrusion screws, breaking the aggregates of nanotalc and converting them to particulate form. This increased the effective surface area for interacting with nylon 6, bringing about appreciable difference in the NT42 and NT52. Formation of aggregates of nanotalc in one time extrusion processed samples and degradation of nylon 6 matrix in three times extrusion processed samples were the reasons for no appreciable difference in the viscosities of NT4 and NT5 samples. The viscosity of the samples was highly differentiated at lower shear rate but was near about same at higher shear rates. At higher shear rates, the molecular structure of the composite got completely ruptured, leaving no chance to resist the force applied by the rotating spindle of the rheometer.
Three-time extrusion processed samples (NT03–NT53) had lower viscosity than one- and two times extrusion processed samples. This was attributed to the degradation of the nylon 6 matrix. Degradation of the nylon 6 polymeric chains decreased its molecular weight. Shearing and repeated temperature exposure led to the degradation of nylon 6 matrix. Viscosity is directly proportional to molecular weight. Thus, as the molecular weight decreased, viscosity of the samples decreased. However, the viscosity of three times extrusion processed samples increased with increase in nanotalc concentration. But the rate of increase is very low as compared to one and two times extrusion processed samples. Thus, nanotalc still plays its role as a reinforcing agent in nylon 6, but the degradation of the base material (nylon 6) gave no chance to appreciably increase the viscosity with the addition of nanotalc.
FTIR analysis
FTIR graphs obtained for NT41, NT42 and NT43 (batches having optimum mechanical and thermal properties for any extrusion processing cycles) are shown in Figure 5.
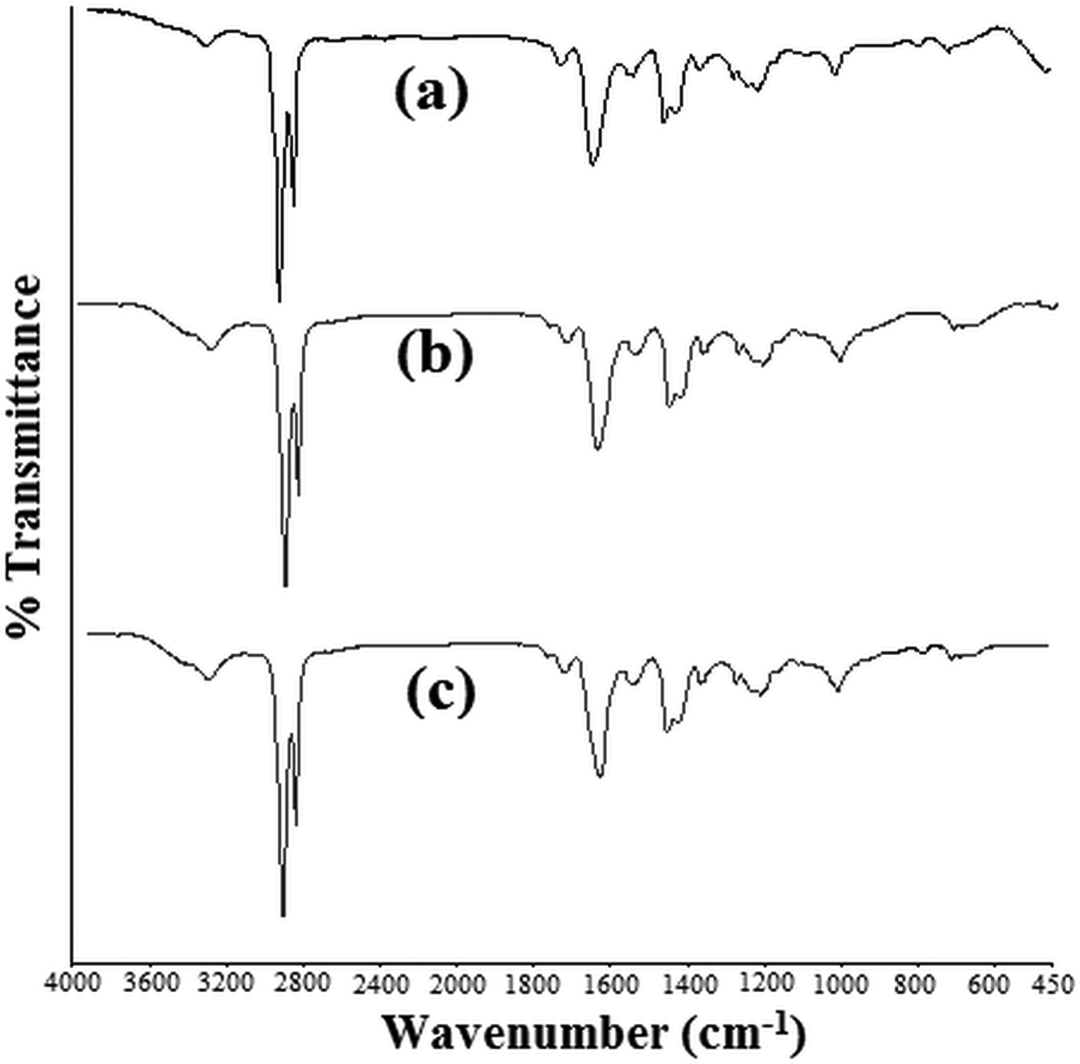
FTIR curves obtained for NT41, NT42 and NT43. FTIR: Fourier transform infrared.
A small band of the amide I (–NH2 in primary amides) groups appeared at 3293 cm–1. Peaks at 2925 and 2854 cm–1 were due to asymmetric and symmetric stretching of CH2, respectively. Small peak at 1722 cm–1 was caused due to C=O stretching. The carbonyl peak (–CONH2) of polyamide was around 1640 cm–1 (steep peak). Amide II band/CH2 asymmetric deformation was indicated by the peak of wave number 1560 cm–1. N–H deformation/CH2 scissoring peak was indicated at wave number 1462 cm–1. The peak at 1377.6 cm–1 was due to CH2 wagging. The peak at 1244 cm–1 was corresponded to C–N stretching vibration bond. The peak at about 1000 cm–1 wave number was due to Si–O linkage present in nanotalc. A small peak at 719 cm–1 showed C–C deformation. Position of these peaks can vary slightly for individual spectra of NT41, NT42 or NT43.
No appreciable difference was observed in the FTIR spectra obtained for NT41, NT42 and NT43, even though the degradation of samples in third extrusion reprocessing was claimed. The FTIR spectra proved the presence of no impurities. Also, nylon 6 matrix did not get chemically modified on extrusion reprocessing as no new peaks were formed in the spectra.
Morphological properties
Scanning electron micrographs obtained for NT41 (a), NT51 (b), NT42 (c), NT52 (d), NT43 (e) and NT53 (f) are shown in Figure 6. These strongly correlate with the above-mentioned reasoning.

Scanning electron micrographs obtained for NT41 (a), NT51 (b), NT42 (c), NT52 (d), NT43 (e) and NT53 (f).
NT41, that is, 4 phr nano-talc containing nylon-6 nanocomposites, was found to have uniformly distributed nanotalc particles. These particles maintained their individual characteristics and were thus able to have maximum possible interaction with nylon 6, improving its performance properties. When the concentration of nanotalc was increased to 5 phr (NT51), aggregates were seen to have formed. These aggregates led to a decrease in the effective surface area for interaction with nylon 6, decreasing the performance properties. Also gaps can be seen between the aggregate and the nylon 6, which were the reason for the generation of stress concentrates in the composite, further decreasing the performance properties. 42,43
NT42 was found to have uniform dispersion of nanotalc in the nylon-6 matrix. But the dispersion is more uniform as compared to NT41. This was confirmed by the increase in the distance between the nanotalc particles. Also the size of aggregates formed in NT52 was smaller than NT51. This strongly correlates with the reasoning given above. The re-shearing of the composite in second extrusion processing led to the breakage of the aggregates, decreasing the size of the aggregates. This increased the effective surface area for interacting with nylon 6 compared to NT51.
Again, for NT43, uniform dispersion of nanotalc was seen in the nylon 6 matrix, increasing the interaction. However, in NT53, aggregates were observed, decreasing the effective surface area. This led to decrease in the performance properties.
Colour test
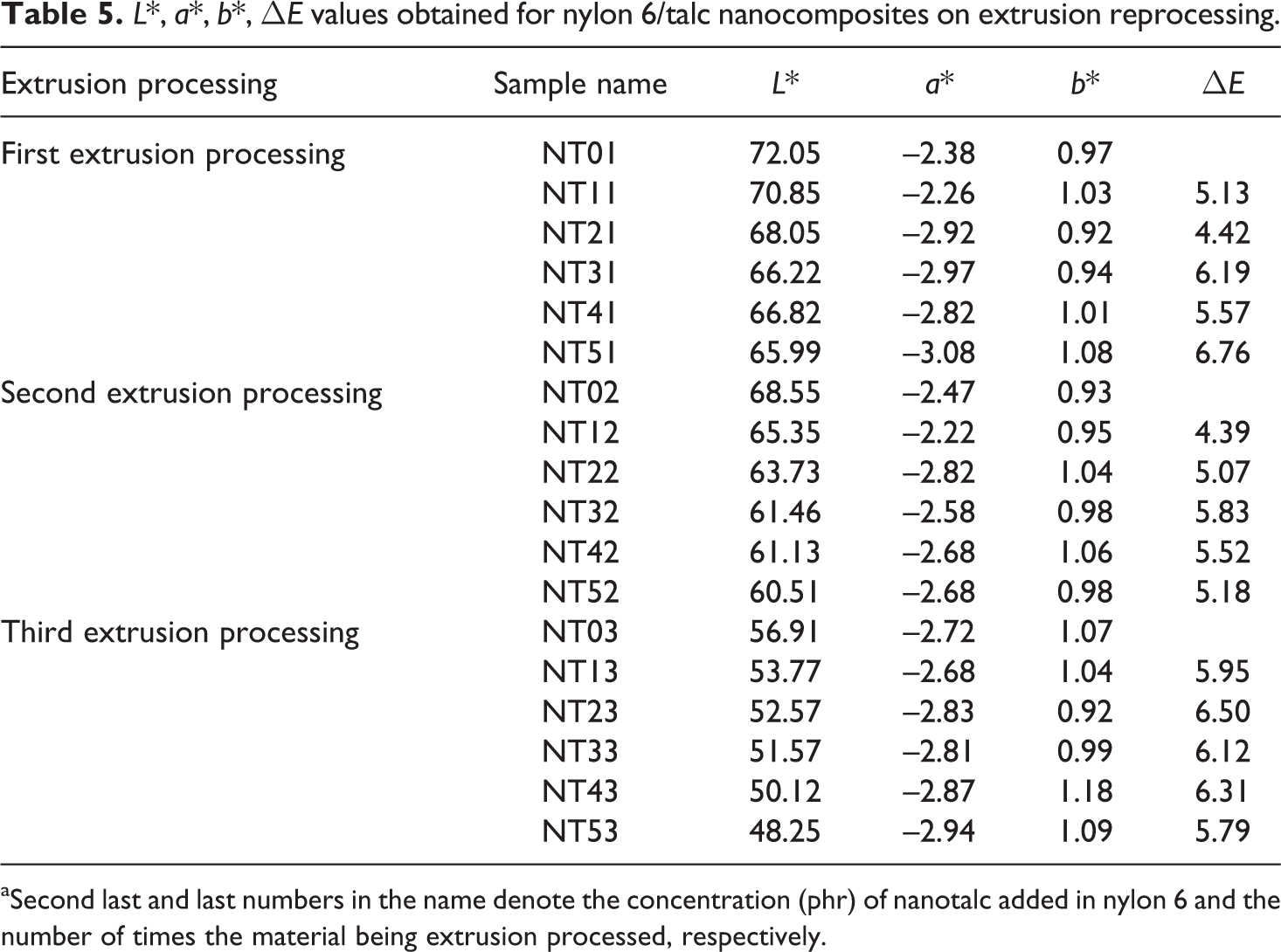
L*, a*, b* and ΔE (L represents lightness; while a and b represents the color-opponent dimensions, based on nonlinearly compressed CIE XYZ color space coordinates) values obtained for nylon 6/talc nanocomposites on first, second and third extrusion processing are listed in Table 5. No appreciable difference were obtained in the values of a*, b* and ΔE. Thus, the composites presented a very stable behaviour during the repeated extrusion. L* value decreased with increase in the concentration of nanotalc in the nylon 6 matrix and also with repeated processing. L* was found to have decreased appreciably for the three times extrusion processed samples. This was attributed to the degradation happening in the nylon 6 due to the repeated exposure to temperature and shearing cycles.
L*, a*, b*, ΔE values obtained for nylon 6/talc nanocomposites on extrusion reprocessing.
aSecond last and last numbers in the name denote the concentration (phr) of nanotalc added in nylon 6 and the number of times the material being extrusion processed, respectively.
Conclusions
Nylon 6/talc nanocomposites were successfully prepared by varying the concentration of nanotalc. Also the prepared composites were subjected to reprocessing, three times, via extrusion in order to simulate the recycling process. On first extrusion, tensile strength, tensile modulus, flexural strength and flexural modulus increased by 36.7, 169.7, 97.1 and 156%, respectively, for 4 phr nanotalc. Second extrusion of the nylon 6/talc nanocomposites lead to better dispersion of nanotalc in nylon 6 matrix, due to the re-shearing, increasing the above-mentioned values. However, in third extrusion reprocessing, all the above-mentioned values decreased drastically due to the degradation of nylon 6 matrix due to repeated shearing and heating in extruder. These observations were confirmed by thermal, rheological and morphological analyses. This study will be helpful in understanding the reprocessing ability of the composite materials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support provided by The University Grant Commission under the SAP-DRS scheme for this work.
