Abstract
Keywords
Introduction
Though cancer is less common in children compared to adults, it stands as one of the leading causes of death related to diseases in this age group.1–4 The estimated global total number of children in need of palliative care annually is about 4 million, with cancer accounting for 4.1%, and it is expected to increase in the coming decades.5,6 The recently evolving field of pediatric palliative oncology care has been established to assist children with cancer and their families in managing the substantial physical, psychological, social, and spiritual challenges associated with cancer and end-of-life treatment.4,7,8 However, delivering pediatric palliative oncology care is challenging due to the complexity of symptoms and the delicate nature of decision-making.9–11 Indeed, children with terminal cancer often undergo aggressive treatments, experiencing significant physical and emotional distress throughout their illness and toward life's end.4,12
The child-centered care (CCC) approach is crucial in pediatric palliative oncology care, emphasizing the active inclusion of children in healthcare decisions related to their well-being in the face of life-limiting and life-threatening illnesses.13,14 Central to this approach is giving voice to children, which enhances care and improves their quality of life. 14 Over the last two decades, there has been growing acknowledgment in pediatric palliative oncology care that self-reporting is the gold standard for assessing subjective health status indicators, including symptom burden and health-related quality of life.3,15 The literature emphasizes the reliability of children's self-reports, increasingly establishing them as the primary source of subjective information regarding symptoms, toxicity, function, and quality of life.15,16
Patient-reported outcomes (PROs) consist of any direct reports from the patient regarding their health status, behavior, and experiences, without any interpretation by healthcare professionals.17,18 PROs can be collected using standardized, validated questionnaires known as patient reported outcome measures (PROMs), completed by patients themselves.17,19–21 The use of PROMs in pediatric settings helps prioritize CCC, enhances patient–healthcare professional communication about psychological issues, such as social and emotional functioning, increases referrals to psychological support, and improves health-related quality of life.22,23 In clinical practice PROMs can be administered in paper or digital form, in that case, called electronic patient reported outcome measures (e-PROMs). 24 The electronic format improves the collection, processing, and management of data, also allowing the PROMs to be administrated remotely.25,26 Specifically, in pediatric care, utilizing e-PROMs makes it possible to overcome obstacles due to children's abilities and provide more information about them, allowing for the implementation of better-quality personalized assistance.24,27–29
In contrast to the expanding literature that summarizes the benefits of e-PROMs for adult patients’ well-being and symptom management in person-centered palliative cancer care, research findings regarding the principles of CCC and standardized self-reported measures within pediatric palliative oncology care present a heterogeneous and complex landscape.13,26,30
This scoping review aims to map the technological innovations of e-PROMs in pediatric palliative oncology care, and how PRO data collected through e-PROMs can influence the management of symptoms and enable better communication between healthcare professionals and children.
Methods
Arksey and O’Malley's 31 conceptual framework and the recommendations by Levac et al 32 were followed: (a) identifying the research questions; (b) identifying relevant studies; (c) study selection; (d) charting the data; and (e) collating, summarizing, and reporting the results. 31 The Preferred Reporting Items of Systematic Reviews extension for Scoping Review (PRISMA-ScR) checklist guided the reporting of the present review. 33 The protocol was published in the Open Science Framework (OSF) and can be accessed at https://osf.io/jpgef.
Stage 1: Identifying the Research Questions
Objective
This scoping aimed to map the relevant literature on the use of e-PROMs in pediatric palliative oncology care. The following research questions guided the objective:
What does the literature reveal about recent technological innovations in e-PROMs within pediatric palliative oncology care? How does the use of e-PROMs for PRO data collection impact the monitoring, and management of symptoms in pediatric palliative oncology care? What are the effects of implementing e-PROMs on communication between children and healthcare professionals in pediatric palliative oncology care?
Stage 2: Identifying Relevant Studies
Eligibility Criteria
We included primary studies meeting the criteria of employing qualitative or quantitative methods, without language or publication date restrictions. The target study population encompassed children (0-18 years old) 34 with cancer undergoing palliative and/or end-of-life care and assessed using e-PROMs. Exclusion criteria comprised studies exclusively focusing on adults. However, studies that included both adult and pediatric populations were considered if they presented pediatric-specific data separately. Studies investigating palliative care within specific stages of cancer care or treatment (eg, chemotherapy) were deemed beyond the scope and, thus, were excluded. Studies lacking both abstract and full-text articles were excluded from our review.
Stage 3: Study Selection
Information Sources
On 15 September 2023, the research team conducted an extensive search for relevant records in the following databases: CINAHL, Embase, MEDLINE via Ovid, PsycINFO, SCOPUS, and Web of Science. Additionally, gray literature searches were performed utilizing the Google search engine, gray literature databases, and websites of relevant charities and organizations, including Google Scholar, OpenGray, OpenThesis, and PsycEXTRA.
Search Strategy
This review used the PCC (Population, Concept, Context) framework on which we based our search strategy. 35 The final search terms included: “Electronic patient reported outcome”; “e-PROMs”; “Child”; “Cancer”; “Palliative care”; “End of life”. The search strategies used on CINAHL, Embase, MEDLINE via Ovid, PsycINFO, SCOPUS, and Web of Science can be found in the Appendix.
Selection
The final list of records was transferred for study selection management in the Rayyan Q online reviewing system. 36 Two record screenings were conducted by two independent authors blindly, first based on title and abstract, and then on full text, following the established inclusion criteria. In case of disagreement during the study selection, a third reviewer was consulted to resolve it. The interrater reliability of the study selection process was assessed using Cohen's kappa statistic.
Stage 4: Charting the Data
We collected essential information regarding the inclusion studies, such as the publication year, study location, participants, objectives, research methodology, e-PROMs variants employed, the mode of e-PROMs administration (including software or devices used), the frequency of e-PROMs deployment in patient evaluations, outcome measures (covering aspects like care processes, patient needs assessment, goal establishment, shared decision-making, care planning, outcome tracking, e-PROMs feedback, intervention reporting frequency, and communication effectiveness), as well as any observations or findings related to usability, satisfaction, and supplementary utility.
Stage 5: Collating, Summarizing, and Reporting the Results
Considering that the aim of the scoping review is to examine a broad range of evidence in the relevant literature, this review included both quantitative and qualitative studies. Due to the heterogeneity of the study methodologies, we used a narrative synthesis approach to summarize their findings, employing frequencies and percentages to describe nominal data. We systematically reviewed the included studies in accordance with our three research questions, analyzing their general characteristics with a particular focus on e-PROMs, and wrote a narrative summary of the literature. The results are categorized into three main sections: (a) technological innovation of e-PROMs in pediatric palliative oncology care; (b) the impact of e-PROMs on symptom monitoring, management, and children's care; (c) the effects of e-PROMs on communication between children and healthcare professionals in pediatric palliative oncology care.
Results
Study Selection
We identified 1435 articles from databases and gray literature which were exported to Mendeley, 37 and 822 duplicates were removed. Out of these, 41 articles met eligibility criteria and underwent full-text assessment. Articles were excluded for inappropriate population focus (eg, not palliative, oncological, or pediatric care), irrelevant outcomes (eg, PROMs development), incorrect publication type (eg, commentary, dissertation), or tool choice (eg, not e-PROMs). After full-text reading, only 12 articles met the eligibility criteria.38–49.
The value of inter-rater agreement between the reviewers was K = 0.88, which indicated excellent concordance.
The PRISMA flow diagram 33 in Figure 1 describes the entire review process.
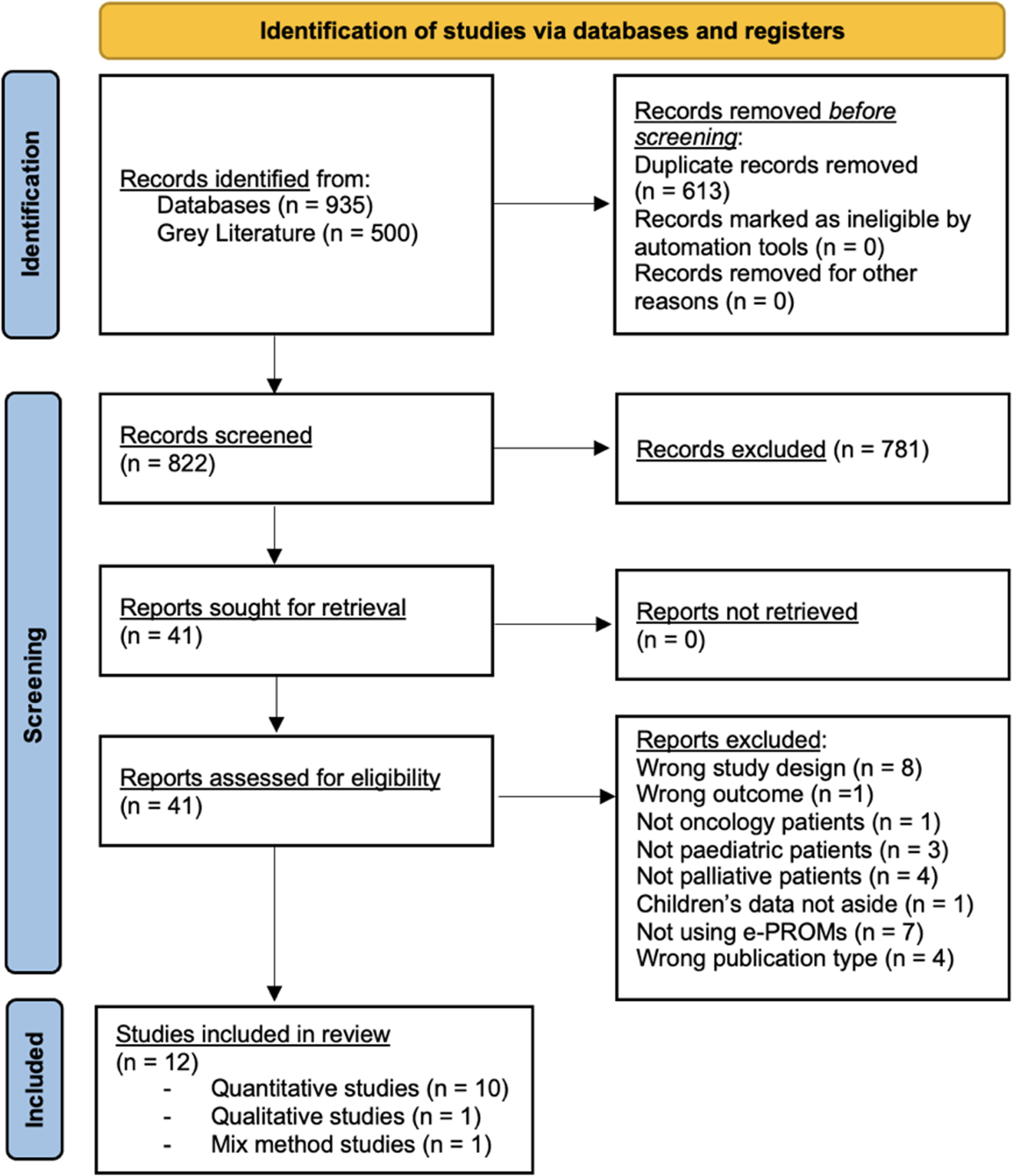
PRISMA flow-diagram.
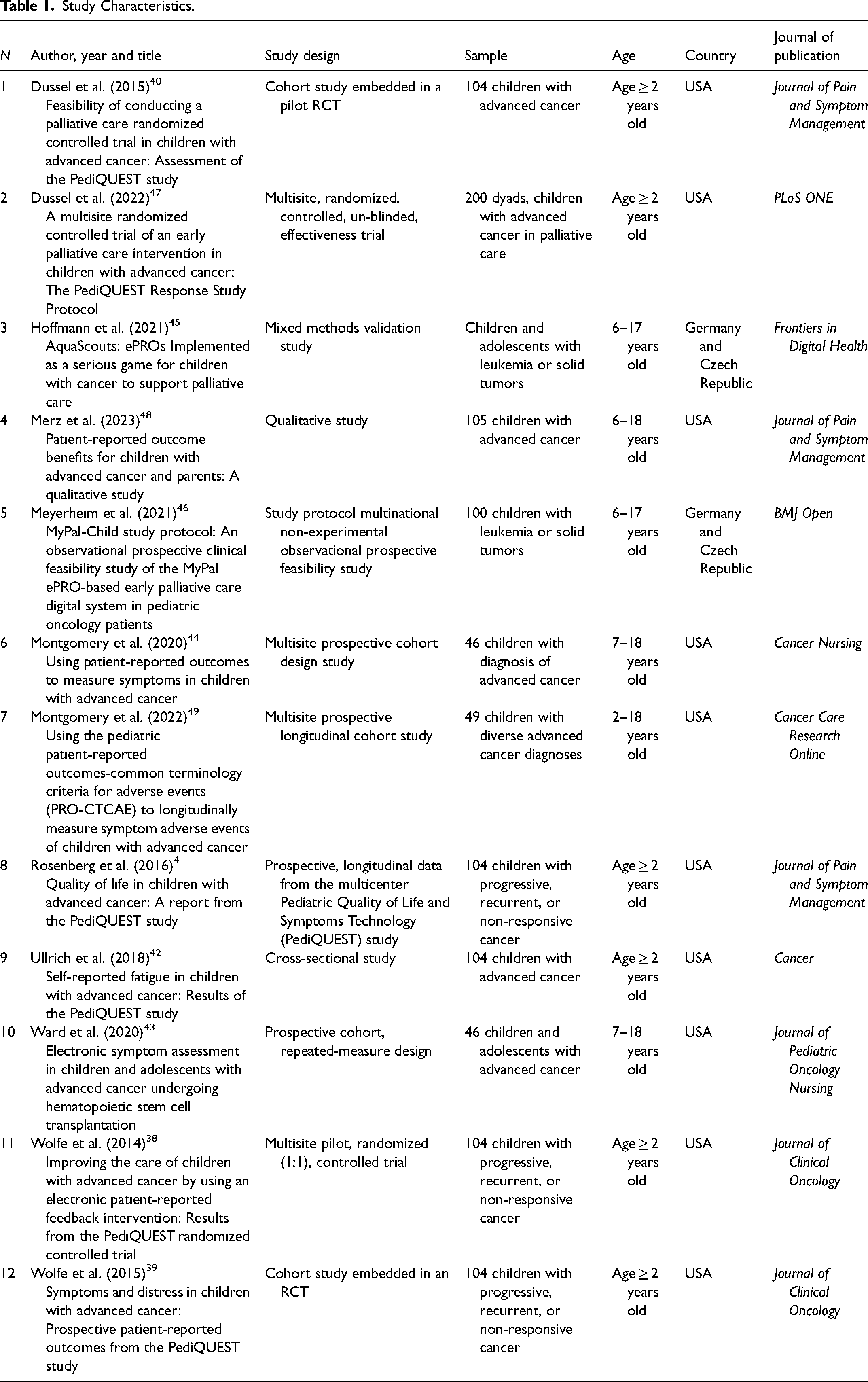
Study Characteristics
In Table 1, we report the key characteristics of the included studies. These published between 2014 38 and 2023 48 encompass research from a variety of global locations, including both European and non-European countries. Notably, the United States stands out as the most prolific contributor to this field, accounting for a total of 10 records.38–44,47–49 The age considered for the sample ranges from a minimum of 238–42,47,49 to a maximum of 18.43,44,48,49
Study Characteristics.
Technological Innovation of e-PROMs in Pediatric Palliative Oncology Care
Table 2 presents a summary of the characteristics of the e-PROMs, along with their associated software and devices. The participants, encompassing inpatients, 42 outpatients,44,48,49 both,38–41,43,46,47 or unspecified categories, 45 engaged with the e-PROMs at various frequencies. These included weekly,38–42,44,47,48 biweekly,43,49 monthly,38–42 or as often as deemed necessary by the child,45,46 ensuring a comprehensive overview of their usage across different settings and schedules.
e-PROMs Characteristics.

Notes: EORTC PATSAT C33: European Organization for Research and Treatment of Cancer Satisfaction with Cancer Care—Core questionnaire; EQ-5D-3L: European Quality of Life Five Dimension; PediQUEST: Pediatric Quality of Life and Evaluation of Symptoms Technology; Ped-PRO-CTCAE: Pediatric Patient Reported Outcomes version of the Common Terminology Criteria for Adverse Events; REDCap: Research Electronic Data Capture; PedsQL: Pediatric Quality of Life Inventory; PQ-MSAS: Pediatric Quality of Life and Evaluation of Symptoms Technology Memorial Symptom Assessment Scale; SSPedi: Symptom Screening in Pediatrics Tool.
Concerning the administration of e-PROMs, the methods varied across studies. Four studies used electronic devices for e-PROM administration without specifying the type.40,47–49 Rosenberg et al. and Wolfe et al. in their two studies opted for administration via tablet or computer,38,41 while the study of Montgomery et al. allowed access through either a smartphone or a computer. 44 Additionally, one study highlighted the flexibility of e-PROMs across tablet, smartphone, and computer platforms. 43 In another two studies, administration was conducted through either a smartphone or a tablet.45,46 Lastly, the two studies of Wolfe et al. and Ullrich et al. exclusively used tablets for e-PROMs administration.39,42
The most frequently used e-PROMs included the Pediatric Quality of Life and Evaluation of Symptoms Technology Memorial Symptom Assessment Scale (PQ-MSAS)38,40,41,47 and its versions adapted for different age groups,39,41–44 alongside the Pediatric Quality of Life Inventory (PedsQL).38,40,41,45–47
In four studies,39,41,42,49 the option of completing the e-PROMs in proxy format by parents was provided, in case the child was unable to (eg, in the age group between 2 and 6 years) or refused to fill out the e-PROM. In the study of Montgomery et al., 99% of participants reported no or little difficulty completing the e-PROMs electronically, 45 as also emphasized in the study by Ward et al. 43 However, a lack of energy among participants was mentioned as an obstacle to compilation of e-PROMs. 40
In three studies,43,44,49 instead of developing dedicated software for e-PROMs, the Research Electronic Data Capture (REDCap) platform was used to distribute e-PROMs electronically. In seven studies,38–42,47,48 the Pediatric Quality of Life and Evaluation of Symptoms Technology (PediQUEST) web system, an electronic data collection platform, was employed also to provide reports, feedback, and alerts for healthcare professionals. It was accessible on different device and tracks symptom trends over time. In two studies,45,46 e-PROMs were administered through MyPal digital health platform, which is divided into two applications: one dedicated to children (MyPal Child App) and one for parents–caregivers (MyPal Carer App). In the research conducted by Hoffmann et al, the MyPal Child app introduced AquaScouts, a serious video game, as an innovative approach for gathering e-PROMs data from children. 49 The MyPal platform also offered healthcare professionals a dashboard for real-time data monitoring and with reports on symptom progression.
The Impact of e-PROMs on Symptom Monitoring, Management, and Children Care
Table 3 provides a summary of the most frequently identified concepts in the included studies, as detected through e-PROMs. They facilitated a detailed capture of both physical and emotional symptoms in children with advanced cancer, offering a comprehensive and dynamic overview of their condition over time. 49 The most frequently examined symptoms included pain, fatigue, emotional functioning, gastrointestinal symptoms, sleep disturbances, and quality of life, encompassing eight out of the 12 studies included.38–44,47 The concurrent presence of multiple physical and emotional symptoms directly affected children's quality of life, with the burden of uncontrolled symptoms proving to be particularly exhausting.41,42,49 e-PROMs, such as the PQ-MSAS, enabled healthcare professionals to gain a deeper understanding of the complex symptoms affecting children, as illustrated by Ullrich et al's study on fatigue. 42 According to the study of Merz et al, the regular completion of e-PROMs enhanced the monitoring of children's symptoms over time and tracking progress, reflecting on their own situation. Increased awareness and reflection on symptoms encouraged parents and children to address previously neglected and unreported symptoms. 48
Concepts Most Frequently Detected in the Included Studies.
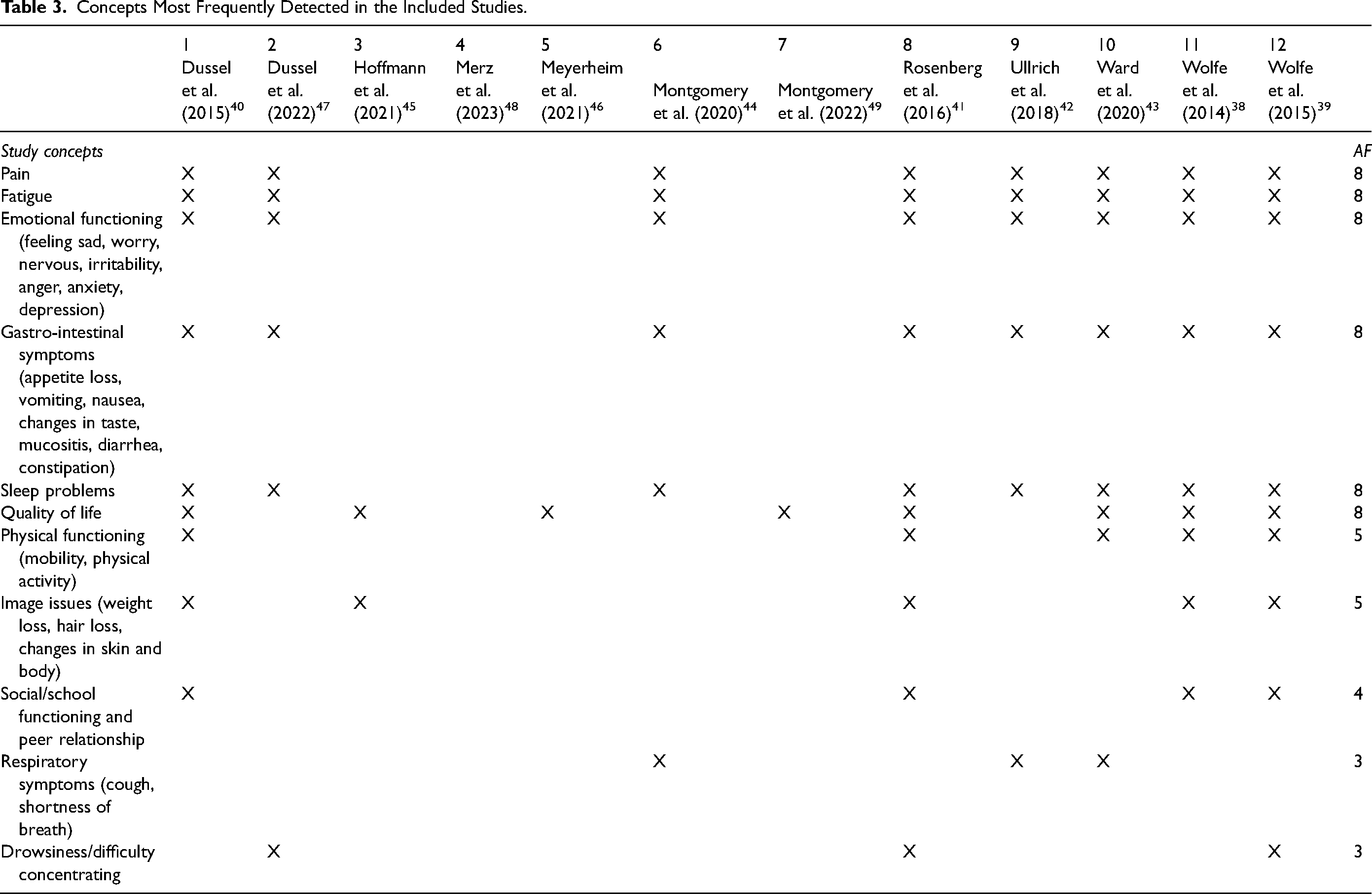
Four e-PROMs also made it possible to investigate aspects such as the relationship with one's peers and school, which is of no small importance for a child.38–41 In five studies,38,40,42,48,49 the change in the body image of these children and how they perceive themselves in the face of these changes was also emphasized, considering hair and weight loss and skin changes. Despite the possibility of receiving feedback from healthcare professionals, possible with only one software, PediQUEST, there were no significant data regarding an improvement in symptoms or quality of life. Feedback significantly improved PedsQL 4.0 emotional functioning and sickness, but only for a limited number of children—specifically, those aged 8 years or older who survived beyond the follow-up period. 38
The Effects of e-PROMs on Communication Between Children and Healthcare Professionals in Pediatric Palliative Oncology Care
Regarding the aspect of communication between children and professionals, Table 4 summarizes the impact of introducing e-PROMs on this aspect. Almost all studies have addressed this aspect.38,39,41–49 Of these, nine highlight that the adoption of e-PROMs significantly improved the decision-making process by providing data that enabled healthcare professionals to make well-informed decisions,38,39,41–45,47,49 thereby ensuring that clinical choices were guided not only by medical tests but also by the patient's personal point of view.39,43,44,48,49 According to the studies of Wolfe et al. and Merz et al., the regular completion of e-PROMs has been shown to significantly improve communication and foster a stronger connection between healthcare professionals and children, facilitating more meaningful discussions.38,48 In fact, e-PROMs could facilitate the discussion of thorny topics such as palliative care or psychosocial issues. 38 This allowed children with advanced cancer and near to death to make their voices heard. 39 Moreover, the study by Merz et al. observed that children began to employ more scientific terminology when describing their condition. 48
Health Professional–Child Communication Reported Characteristics of e-PROMs in Included Studies.
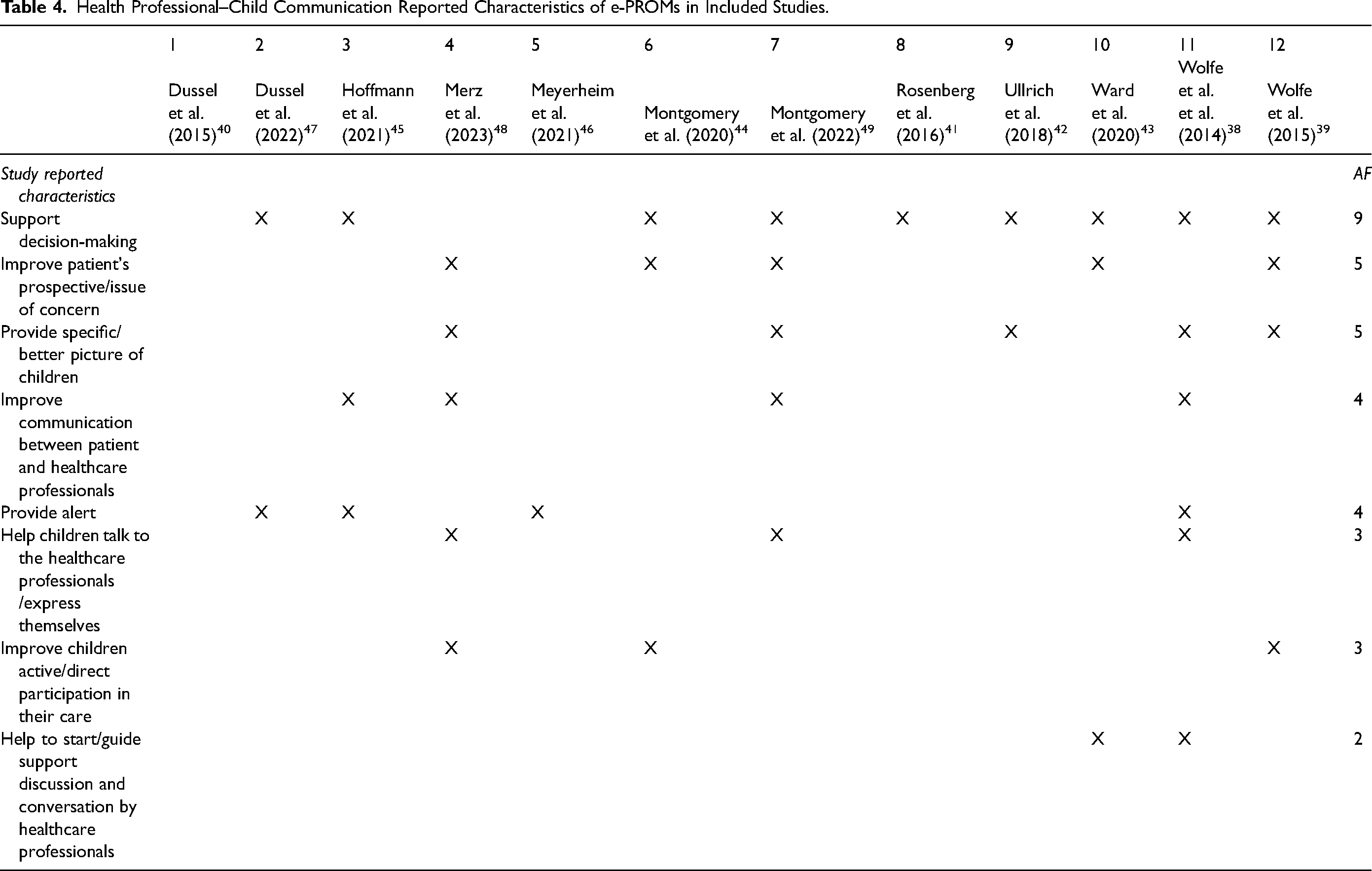
Four studies described a software that allowed people to send alerts to healthcare professionals on health aspects of particular concern and worthy of attention.38,45,46,48 In the protocol of Hoffmann et al, the e-PROMs, designed in alignment with serious games, were found to enhance agile communication with professionals while simultaneously amplifying the child's role in their own care journey. 45 From the perspective of healthcare professionals, the use of e-PROMs did not result in increased consultation times, as revealed in the study by Wolfe et al. 38
Discussion
This scoping review aimed to map the existing literature regarding technological innovations supporting e-PROMs in the context of pediatric palliative oncology care. It also focused on evaluating the influence of them on managing symptoms in children, and facilitating communication between young patients and healthcare professionals. According to the literature, e-PROMs for children have proven to be highly effective in empowering them to articulate their perspectives and actively participate in their healthcare, especially during their illness and in end-of-life care.3,15,39,44
The software and devices used for e-PROMs showed exceptional versatility across different age groups. In situations where children were unable or not in a position to complete the assessments themselves, parents intervened to complete them on their behalf.39,41,42,44,49 Such adaptability guarantees that the collected data is thorough and representative of all participant experiences, irrespective of a child's capacity to interact directly with the device.
Collecting feedback directly from children using devices that are integral to their daily lives presents an effective strategy to engage them without imposing the burden of completing e-PROMs, which might seem tedious or uninteresting. Today's children, whose interests are predominantly centered around play and other activities, may find traditional methods of data collection unappealing. Therefore, offering technology familiar to them, such as tablets or smartphones, can make the process more interactive and enjoyable. For instance, developing age-specific software tailored to children's interests, like the AquaScouts software, represents a strategy to facilitate data collection in a manner that does not burden the child. 45 As the child enters the world of play, they simultaneously delve deeper into the realm of their illness, responding to the questionnaire's questions. Therefore, it is increasingly necessary to consider specific challenges in implementing these tools for children compared to adults. These factors include the need for child-centered language, and tools that accommodate their varying cognitive and developmental abilities. 15 This necessity seems to be confirmed by Hoffmann et al, who created two versions of e-PROMs systems for adults and children, differing in design, content, and mode of use. 45 Children with terminal cancer often face significant physical and emotional challenges throughout their illness, with these challenges intensifying as they near the end of life.4,12 e-PROMs serve as a crucial tool in comprehensively capturing both the physical and emotional symptoms these children experience, facilitating a better understanding and management of their condition, enabling healthcare professionals to track symptom progression over time. 44
Caring for a child on their palliative care journey means embracing their entire world. This encompasses not only the physical symptoms wrought by the illness, which undeniably require meticulous attention, but also the broader spectrum of their lives. This includes their interactions with peers and their educational experiences, both of which are significantly altered by the new, disease-centric reality that they are navigating.38–41 Such care ensures a focus not just on the illness, but on maintaining the essence of a childhood that, despite the circumstances, continues to deserve moments of normalcy and joy.
Emotions are deeply subjective, and the way a child processes them is unique to their experience of life. Those around the child can only attempt to understand what they are feeling; truly, a child's emotional experience is indescribable except by the child themselves. This underscores the importance of using tools like e-PROMs to directly capture the child's own report of their symptoms and emotional state, thereby enabling a more nuanced understanding and management of their condition.38–44,47
Another critical aspect, intimately connected to emotions and far from secondary, concerns the detection of children's perceptions of the physical changes caused by the disease and its treatment.38–41,45
Despite adults and children expressing themselves differently due to emotional maturity and various life experiences, e-PROMs enable a more equitable and informed dialogue between healthcare providers and young patients. They facilitate conversations on an equal footing, ensuring that the voices of children are heard and valued in the management of their care. By translating the nuanced experiences of children into data that adults can understand and act upon, e-PROMs focus on the needs and unique perspectives of children.
Implications
The findings of this study have important implications for healthcare professionals and researchers in pediatric palliative oncology care. Our results suggest that e-PROMs can empower children to express their perspectives and actively participate in their care. By integrating familiar technology like tablets and smartphones, healthcare professionals can engage children more effectively, gain a nuanced understanding of their conditions, and foster an equitable and informed dialogue that prioritizes the child's voice.
While the potential of e-PROMs in pediatric palliative oncology care is evident, further research is needed to explore their long-term impact on symptom management, quality of life, and communication outcomes. Additionally, efforts should focus on addressing barriers to implementation and ensuring that e-PROMs are tailored to meet the unique needs of pediatric patients and their families.
Limitations
This review has several limitations. By conducting a scoping review, our goal was to capture a broad spectrum of available information, leading us to include studies without conducting a formal quality evaluation. We analyzed a total of 12 studies from the primary literature, highlighting the limited resources available for children with advanced cancer receiving palliative care. Additionally, a significant portion of the studies we included were focused on e-PROMs development, rather than direct patient involvement. Another limitation is the geographical origin of the included studies, predominantly from the United States, providing a constrained view of the phenomenon.
Conclusions
The incorporation of e-PROMs into pediatric palliative oncology care marks a significant step forward in enhancing symptom management, improving communication, and elevating the overall quality of care for children with life-limiting conditions. This innovative approach offers a thorough understanding of the physical and emotional challenges these children face, providing essential insights for healthcare professionals. By utilizing a range of electronic devices and platforms, e-PROMs facilitate regular and adaptable data collection. Importantly, e-PROMs give children a voice in their own care process, enabling meaningful conversations and fostering a patient-centered approach in decision-making to align interventions more closely with their needs and expectations. Integrating software into the familiar aspects of children's lives proves to be an effective strategy, making them less medical and closer to them, tailored to their language and reality, which is also composed of fantasy. Despite some challenges, such as energy limitations, e-PROMs have been largely well-received.
Footnotes
Availability of Data and Materials
Search strategy is available in the Appendix.
Authors’ Contributions
LC, ML, and DR conceptualized and designed this scoping review, led the project coordination. The search strategy was developed by FR, a research librarian with expertise in systematic searches in medical research databases, in collaboration with the entire research team. SC and LA screened first the titles and abstracts and then the full text for eligibility and extracted data from the included studies. DR and IB took the lead in analyzing and synthesizing the data and were involved in the interpretation of results. The writing and reviewing process saw contributions from DR, LC, and IB. MLP and ML offered insightful critiques on earlier drafts. All authors read, commented on, and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was a scoping review and did not involve participants directly; therefore, it did not require ethics approval or consent.
Appendix
CINAHL via Ebscohost, September 15, 2023
S18: S5 AND S14 AND S17
S17: S15 OR S16
S16: (MH “Child+”) OR (MH “Adolescence+”)
S15: TI (KID OR KIDS OR BABY OR BABIES OR BOY OR boys OR GIRL* OR CHILD* OR PRETEEN* OR ADOLESCEN* OR JUVENIL* OR TEEN* OR YOUNG* OR YOUTH OR PEDIATR* OR PAEDIATR* OR SCHOOL* OR PRESCHOOL* OR MINORS OR puber* OR pubescen* OR UNDERAGE OR UNDER-AGE) OR AB (KID OR KIDS OR BABY OR BABIES OR BOY OR boys OR GIRL* OR CHILD* OR PRETEEN* OR ADOLESCEN* OR JUVENIL* OR TEEN* OR YOUNG* OR YOUTH OR PEDIATR* OR PAEDIATR* OR SCHOOL* OR PRESCHOOL* OR MINORS OR puber* OR pubescen* OR UNDERAGE OR UNDER-AGE)
S14: S12 OR S13
S13: S8 AND S11
S12: TI ADVANCED W3 CANCER* OR AB ADVANCED W3 CANCER*
S11: S9 OR S10
S10: TI ((PALLIAT* or end-of-life or EOL-CARE or DYING or HOSPICE* or end-stage* or endstagE* or (terminal* N4 (care or caring or ill* or patient*)))) OR AB ((PALLIAT* or end-of-life or EOL-CARE or DYING or HOSPICE* or end-stage* or endstagE* or (terminal* N4 (care or caring or ill* or patient*))))
S9: (MH “Palliative Care Nursing”) OR (MH “Palliative Medicine”) OR (MH “Hospices”) OR (MH “Terminal Care+”) OR (MH “Terminally Ill Patients+”)
S8: S6 OR S7
S7: TI ((patient* or SELF) N3 report* N3 (outcome* or measure*)) OR AB ((patient* or SELF) N3 report* N3 (outcome* or measure*))
S6: TI (Neoplas* or Tumor* or TUMOUR* or Cancer* or Malignan* or carcinom* or ONCOL* or ONCO-HEMATOL* or ONCOHEMATOL* or HEMATONCOL* or HEMATOONCOL leukem* or leukaem* or aleukem* or aleukaem* or leucem* or leucaem*) OR AB (Neoplas* or Tumor* or TUMOUR* or Cancer* or Malignan* or carcinom* or ONCOL* or ONCO-HEMATOL* or ONCOHEMATOL* or HEMATONCOL* or HEMATOONCOL or leukem* or leukaem* or aleukem* or aleukaem* or leucem* or leucaem*)
S5: S1 OR S2 OR S3 OR S4
S4: TI ((patient* or SELF) N3 report* N3 (outcome* or measure*)) OR AB ((patient* or SELF) N3 report* N3 (outcome* or measure*))
S3: TI ((selfreport* or patientreport*) N3 (outcome* or measure*)) OR AB ((selfreport* or patientreport*) N3 (outcome* or measure*))
S2: TI (proms or prom or epros or epro or eprom or eproms or E-PRO or E-PROS) OR AB (proms or prom or epros or epro or eprom or eproms or E-PRO or E-PROS)
S1: (MH “Patient-Reported Outcomes+”)
Embase
(‘palliative therapy'/exp OR ‘palliative nursing'/exp OR ‘terminally ill patient'/de OR ‘terminal care'/de OR ‘hospice'/exp OR palliat*:ti,ab,kw OR ‘end of life':ti,ab,kw OR ‘eol care':ti,ab,kw OR dying:ti,ab,kw OR hospice*:ti,ab,kw OR ‘end stage*':ti,ab,kw OR endstage*:ti,ab,kw OR ((terminal* NEAR/4 (care OR caring OR ill* OR patient*)):ti,ab,kw)) AND (‘patient-reported outcome'/exp OR ‘patient reported outcome measure'/exp OR ‘patient reported outcome measurement'/exp OR ‘patient reported outcomes measurement information system'/exp OR ‘patient reported outcomes measurement information system physical function'/exp OR ‘electronic patient reported outcome'/exp OR proms:ti,ab,kw OR prom:ti,ab,kw OR epros:ti,ab,kw OR epro:ti,ab,kw OR eprom:ti,ab,kw OR eproms:ti,ab,kw OR ‘e pro':ti,ab,kw OR ‘e pros':ti,ab,kw OR (((selfreport* OR patientreport*) NEAR/3 (outcome* OR measure*)):ti,ab,kw) OR (((patient* OR self) NEAR/3 report* NEAR/3 (outcome* OR measure*)):ti,ab,kw)) AND (‘pediatric oncology nursing'/exp OR ‘childhood cancer'/exp OR ((‘juvenile'/exp OR kid:ti,ab,kw OR kids:ti,ab,kw OR baby:ti,ab,kw OR babies:ti,ab,kw OR boy:ti,ab,kw OR boys:ti,ab,kw OR girl*:ti,ab,kw OR child*:ti,ab,kw OR preteen*:ti,ab,kw OR adolescen*:ti,ab,kw OR juvenil*:ti,ab,kw OR teen*:ti,ab,kw OR young*:ti,ab,kw OR youth:ti,ab,kw OR pediatr*:ti,ab,kw OR paediatr*:ti,ab,kw OR school*:ti,ab,kw OR preschool*:ti,ab,kw OR minors:ti,ab,kw OR puber*:ti,ab,kw OR pubescen*:ti,ab,kw OR underage:ti,ab,kw OR ‘under age':ti,ab,kw) AND (‘cancer pain'/exp OR ‘neoplasm'/exp OR ‘oncology'/exp OR ‘oncology nursing'/exp OR ‘cancer center'/exp OR ‘cancer patient'/exp OR neoplas*:ti,ab,kw OR tumor*:ti,ab,kw OR tumour*:ti,ab,kw OR cancer*:ti,ab,kw OR malignan*:ti,ab,kw OR carcinom*:ti,ab,kw OR oncol*:ti,ab,kw OR ‘onco hematol*':ti,ab,kw OR oncohematol*:ti,ab,kw OR hematoncol*:ti,ab,kw OR hematooncol:ti,ab,kw OR leukem*:ti,ab,kw OR leukaem*:ti,ab,kw OR aleukem*:ti,ab,kw OR aleukaem*:ti,ab,kw OR leucem*:ti,ab,kw OR leucaem*:ti,ab,kw))) OR (((advanced NEXT/3 cancer*):ti,ab,kw) AND (‘patient-reported outcome'/exp OR ‘patient reported outcome measure'/exp OR ‘patient reported outcome measurement'/exp OR ‘patient reported outcomes measurement information system'/exp OR ‘patient reported outcomes measurement information system physical function'/exp OR ‘electronic patient reported outcome'/exp OR proms:ti,ab,kw OR prom:ti,ab,kw OR epros:ti,ab,kw OR epro:ti,ab,kw OR eprom:ti,ab,kw OR eproms:ti,ab,kw OR ‘e pro':ti,ab,kw OR ‘e pros':ti,ab,kw OR (((selfreport* OR patientreport*) NEAR/3 (outcome* OR measure*)):ti,ab,kw) OR (((patient* OR self) NEAR/3 report* NEAR/3 (outcome* OR measure*)):ti,ab,kw)) AND (‘juvenile'/exp OR kid:ti,ab,kw OR kids:ti,ab,kw OR baby:ti,ab,kw OR babies:ti,ab,kw OR boy:ti,ab,kw OR boys:ti,ab,kw OR girl*:ti,ab,kw OR child*:ti,ab,kw OR preteen*:ti,ab,kw OR adolescen*:ti,ab,kw OR juvenil*:ti,ab,kw OR teen*:ti,ab,kw OR young*:ti,ab,kw OR youth:ti,ab,kw OR pediatr*:ti,ab,kw OR paediatr*:ti,ab,kw OR school*:ti,ab,kw OR preschool*:ti,ab,kw OR minors:ti,ab,kw OR puber*:ti,ab,kw OR pubescen*:ti,ab,kw OR underage:ti,ab,kw OR ‘under age':ti,ab,kw))) AND (‘article'/it OR ‘article in press'/it OR ‘chapter'/it OR ‘review'/it)
Medline via Ovid search strategy
PsycINFO
SCOPUS
(((TITLE-ABS-KEY (proms OR prom OR epros OR epro OR eprom OR eproms OR e-pro OR e-pros) OR TITLE-ABS-KEY ((selfreport* OR patientreport* OR patient*-report* OR self-report*) W/3 (outcome* OR measure*)))) AND (TITLE-ABS-KEY (kid OR kids OR baby OR babies OR boy OR boys OR girl* OR child* OR preteen* OR adolescen* OR juvenil* OR teen* OR young* OR youth OR pediatr* OR paediatr* OR school* OR preschool* OR minors OR puber* OR pubescen* OR underage OR under-age))) AND (((TITLE-ABS-KEY (neoplas* OR tumor* OR tumour* OR cancer* OR malignan* OR carcinom* OR oncol* OR oncohematol* OR oncohematol* OR hematoncol* OR hematooncol OR leukem* OR leukaem* OR aleukem* OR aleukaem* OR leucem* OR leucaem*) AND TITLE-ABS-KEY (palliat* OR end-of-life OR eol-care OR dying OR hospice* OR end-stage* OR endstage* OR (terminal* W/4 (care OR caring OR ill* OR patient*))))) OR (TITLE-ABS-KEY (advanced W/3 cancer)))
Web of Science
# Web of Science Search Strategy (v0.1)
# Database: All Databases
# Entitlements:
- WOS: 1944 to 2023 - BCI: 2009 to 2023 - CABI: 1910 to 2023 - CCC: 1998 to 2023 - DRCI: 2009 to 2023 - DIIDW: 2009 to 2023 - KJD: 1980 to 2023 - MEDLINE: 1950 to 2023 - PPRN: 1991 to 2023 - PQDT: 1637 to 2023 - SCIELO: 2002 to 2023 - ZOOREC: 2009 to 2023
# Searches:
(SELFREPORT* OR PATIENTREPORT* OR PATIENT*-REPORT* OR SELF-REPORT*) NEAR/3 (OUTCOME* OR MEASURE*) (Topic) AND kid OR kids OR baby OR babies OR boy OR boys OR girl* OR child* OR preteen* OR adolescen* OR juvenil* OR teen* OR young* OR youth OR pediatr* OR paediatr* OR school* OR preschool* OR minors OR puber* OR pubescen* OR underage OR under-age (Topic) neoplas* OR tumor* OR tumour* OR cancer* OR malignan* OR carcinom* OR oncol* OR onco-hematol* OR oncohematol* OR hematoncol* OR hematooncol OR leukem* OR leukaem* OR aleukem* OR aleukaem* OR leucem* OR leucaem* (Topic) AND palliat* OR end-of-life OR eol-care OR dying OR hospice* OR end-stage* OR endstage* OR (terminal* near/4 (care OR caring OR ill* OR patient*)) (Topic) advanced near/3 cancer (Topic) #2 OR #3 #4 AND #1 #4 AND #1 and Preprint Citation Index (Exclude – Database) and MEDLINE® (Exclude – Database) Results: 28 (KP = ((SELFREPORT* OR PATIENTREPORT* OR PATIENT*-REPORT* OR SELF-REPORT*) NEAR/3 (OUTCOME* OR MEASURE*)) AND KP = (kid OR kids OR baby OR babies OR boy OR boys OR girl* OR child* OR preteen* OR adolescen* OR juvenil* OR teen* OR young* OR youth OR pediatr* OR paediatr* OR school* OR preschool* OR minors OR puber* OR pubescen* OR underage OR under-age)) (KP = (neoplas* OR tumor* OR tumour* OR cancer* OR malignan* OR carcinom* OR oncol* OR onco-hematol* OR oncohematol* OR hematoncol* OR hematooncol OR leukem* OR leukaem* OR aleukem* OR aleukaem* OR leucem* OR leucaem*) AND KP = (palliat* OR end-of-life OR eol-care OR dying OR hospice* OR end-stage* OR endstage* OR (terminal* near/4 (care OR caring OR ill* OR patient*)))) (KP = (advanced near/3 cancer)) #3 OR #9 OR #8 #10 AND #7 #11 OR #6
