Abstract
Introduction
Electronic patient-reported outcome measures (e-PROMs) offer advantages in palliative cancer care, including rapid completion, improved data quality and direct storage, improving clinical decision-making. The electronic Integrated Palliative Care Outcome Scale (e-IPOS) in this context enables thorough self-assessment by patients, enhancing symptom management and self-reflection of their current situation.
Aim
To evaluate the feasibility of implementing the e-IPOS in home palliative cancer care.
Outcomes
The primary outcomes included the enrollment consent rate, study retention rate, e-IPOS completion rate and response completeness, and the number of clinical assessments and interventions performed during home visits. The secondary outcomes were the number of unscheduled visits and patients’ perceived quality of life.
Design
A two-group quasiexperimental clinical pilot study. The control group received standard palliative care, the intervention group received standard care along with weekly e-IPOS completion during home visits. Both groups were enrolled for 4 weeks.
Setting/participants
Adults with advanced cancer from the home palliative care unit of the Istituto Nazionale dei Tumori of Milan.
Results
Twenty-three patients were enrolled (74.19%), and 20 completed the study (drop-out: 13.04%). 82.5% of the expected e-IPOS responses were received, of which 96.9% were fully complete. In the intervention group, the Wilcoxon test showed an increase in identified needs and documented interventions (P < .05) and a decrease in unscheduled visits (P < .05).
Conclusion
It is feasible to recruit people via home palliative care for an e-IPOS implementation study. Future fully powered studies should investigate the feasibility and assess patients’ perceptions of its use to better understand its clinical benefits.
Keywords
Introduction
Patient-reported outcomes (PROs) are descriptions of the patient’s health status provided by the patient themselves without interpretation by a clinician or others.1-4 Patient-reported outcome measures (PROMs) are instruments, often in the form of questionnaires, used to collect information about PROs and evaluate aspects of the patient’s experience, such as symptom burden, functional status, and psychological and emotional well-being. 5 Giving patients a voice to involve them in their care highlights the importance of PROMs in assisting healthcare professionals in decision-making.6-8 The use of PROMs has enabled clinicians to identify and address patients’ hidden and unmet needs, allowing patients to become aware of their health condition and their physical, psychological, and social needs.8-10
PROMs are used in oncology throughout all disease stages, from diagnosis to the terminal phases, and their clinical application is increasingly growing in the context of oncological palliative care.11-13 This growth in the use of PROMs aligns with the fundamental principles of palliative care, which adopts a holistic perspective of the patient by considering the physical, mental, social, spiritual, and economic dimensions.14,15 In this context, the use of PROMs has been linked to the prompt monitoring and management of symptoms, such as pain, nausea, fatigue, depression, and nutritional issues, that typically affect advanced-stage cancer patients.15,16 In palliative cancer care, PROMs are essential for providing truly individualized and high-quality patient-centered care. 17
Electronic PROMs (e-PROMs) are favored over paper forms due to their cost-effectiveness, faster completion time, improved data quality, and higher response rates, resulting in reduced administration time and reduced missing data.15,18,19 Moreover, the electronic format allows for the direct and systematic storage of collected data in medical records, which can assist healthcare providers and patients in making clinical decisions that positively influence patient well-being.18,20,21
One PROM used in the palliative care context is the Integrated Palliative Care Outcome Scale (IPOS). This questionnaire has been tested in various inpatient and home care environments as a standardized tool to assess the current and evolving palliative needs of patients 22 and is also used in an electronic format. 23 Burner-Fritsch et al’s study stated that engaging with the e-IPOS during the palliative phase necessitates a thorough self-assessment by patients of their overall health condition, a process that is potentially advantageous for effectively managing symptom burden and reflecting their current situation. 23
Despite evidence of the effectiveness of outcome measures in palliative care, their integration into routine practice presents challenges, influenced by factors at both the patient and organizational levels. Healthcare professional workload and inadequate technological infrastructure are key organizational barriers,4,24,25 while patient-related challenges include health conditions and comfort with internet-enabled devices. 23 The adoption of these measures in palliative care, especially in home settings, remains limited. 15
Additionally, due to the challenges in conducting studies in the field of palliative care arising from patient fragility and disease progression in terminal phases, 26 there is a significant loss of clinically relevant information, as evidenced by the literature; hence, it becomes crucial to conduct preliminary analyses on the feasibility of research procedures to refine techniques and effectively complete studies in this area.27,28
To our knowledge, there is no published evidence regarding the routine use of e-PROMs in home palliative cancer care in Italy, 29 even in the most specialized settings, such as the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan (INT).
Aim
The aim of this study was to evaluate the feasibility of implementing the e-IPOS in routine care processes in home palliative cancer care.
Methods
Study design
This was a two-group, quasiexperimental, single-center clinical pilot study. This study design aims to establish a cause-and-effect relationship between an independent and dependent variable. Distinct from a true experiment, this quasiexperimental approach does not use random assignment for grouping. 30 To ensure thoroughness and transparency, the study followed to the Joanna Briggs Institute’s checklist for quasiexperimental studies and the Template for Intervention Description and Replication (TIDieR) checklist.31,32
The research conducted was part of a larger project titled “Impact assessment of a system e-patient reported outcome measures on home palliative care: Mixed-methods study of feasibility and intervention” aimed at enhancing e-PROMs adoption in at-home palliative care for cancer patients at the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan (INT).
Setting
The research was conducted at INT’s Palliative Care - Hospice, Pain Therapy, and Rehabilitation Complex Unit, which includes a home palliative care service. Annually, approximately 100 patients use home-based care supported by a team of physicians, nurses, social health workers, physiotherapists, psychologists, a nursing case manager, and a nursing coordinator.
Participants
The study enrolled patients from the home palliative care unit, including those who were older than 18 years, who had a good grasp of the Italian language, and who had metastatic or advanced cancer (stage III or IV). Patients with a prognosis of fewer than 30 days, those experiencing cognitive impairments that hindered their self-assessment abilities, and those with physical disabilities that could prevent the use of electronic devices were excluded.
Recruitment Procedures
From March to September 2023, INT palliative home care nurses and physicians identified potential participants during routine clinical work. The team initially approached potential participants, and if they agreed, the principal investigator (LC), a PhD student, discussed the study in more detail with them by telephone. After being confirmed to meet the inclusion criteria, patients were contacted by a researcher for a meeting where the study objectives and procedures were outlined and written consent was obtained. The enrolled patients were allocated 1:1 to the control or intervention group.
Sampling
Given the typical recruitment challenges in the palliative care context, 28 the study aimed to include at least 20 participants, chosen for practical purposes to assess the study’s feasibility, acceptance, and potential dropout rate. This aligns with guidelines for the sample size of an internal pilot study, which involves analyzing early data from a two-group study. This requires a minimum of twenty degrees of freedom. This interim analysis functioned as a preliminary assessment of the study’s methods and anticipated outcomes.33,34
Measures
• The IPOS is a patient self-report and staff proxy-report outcome measure for advanced illness patients in palliative care that assesses physical symptoms, emotional symptoms, and communication/practical issues through ten questions (17 items)35-37 focusing on symptom burden. The overall IPOS score is the sum of the scores for each of the questions and can vary from zero to 68.
36
We used the electronic format of the Italian version,
38
required at https://pos-pal.org. • The European Organization for Research and Treatment of Cancer (EORTC) questionnaire, called EORTC-QLQ-C15-PAL,
39
is a 15-item questionnaire used to assess the quality of life of cancer patients in palliative care, required at https://www.eortc.org. • The Australian Karfnosky Performance Status (AKPS)
40
is a quality of life rating scale compiled by healthcare professionals that yields a value from 0 to 100 in percentages, with numerical ratings in intervals of 10, accounting for 4 parameters: limitations in daily activities, self-care, autonomy and work activity.
Intervention
After being allocated to the control or intervention group, participants received standard palliative care (control) or standard care plus e-IPOS completion (intervention). Both groups were enrolled for 4 weeks. In each group, a research team member administered the EORTC-QLQ-C15-PAL questionnaire at home at two time points. The first administration occurred simultaneously at the first home visit by health care professionals after the patients provided informed consent (T0). The second administration was after 4 weeks (T3) in conjunction with the visit of health care professionals. Concurrently, the AKPS scores completed by clinicians were extracted from the medical records. Figure 1 shows the study flowchart. Additionally, weekly data for both groups, including sociodemographic data, clinical information and details on physiological needs, pain, symptoms, and emotional aspects, were extracted from medical records as outlined by the Canadian Hospice Palliative Care Association (CHPCA).41,42 Study flowchart. Legend: EORTC, European Organization for Research and Treatment of Cancer; e-IPOS, electronic version of Integrated Palliative Care Outcome Scale; AKPS, Australian Karfnosky Performance Status.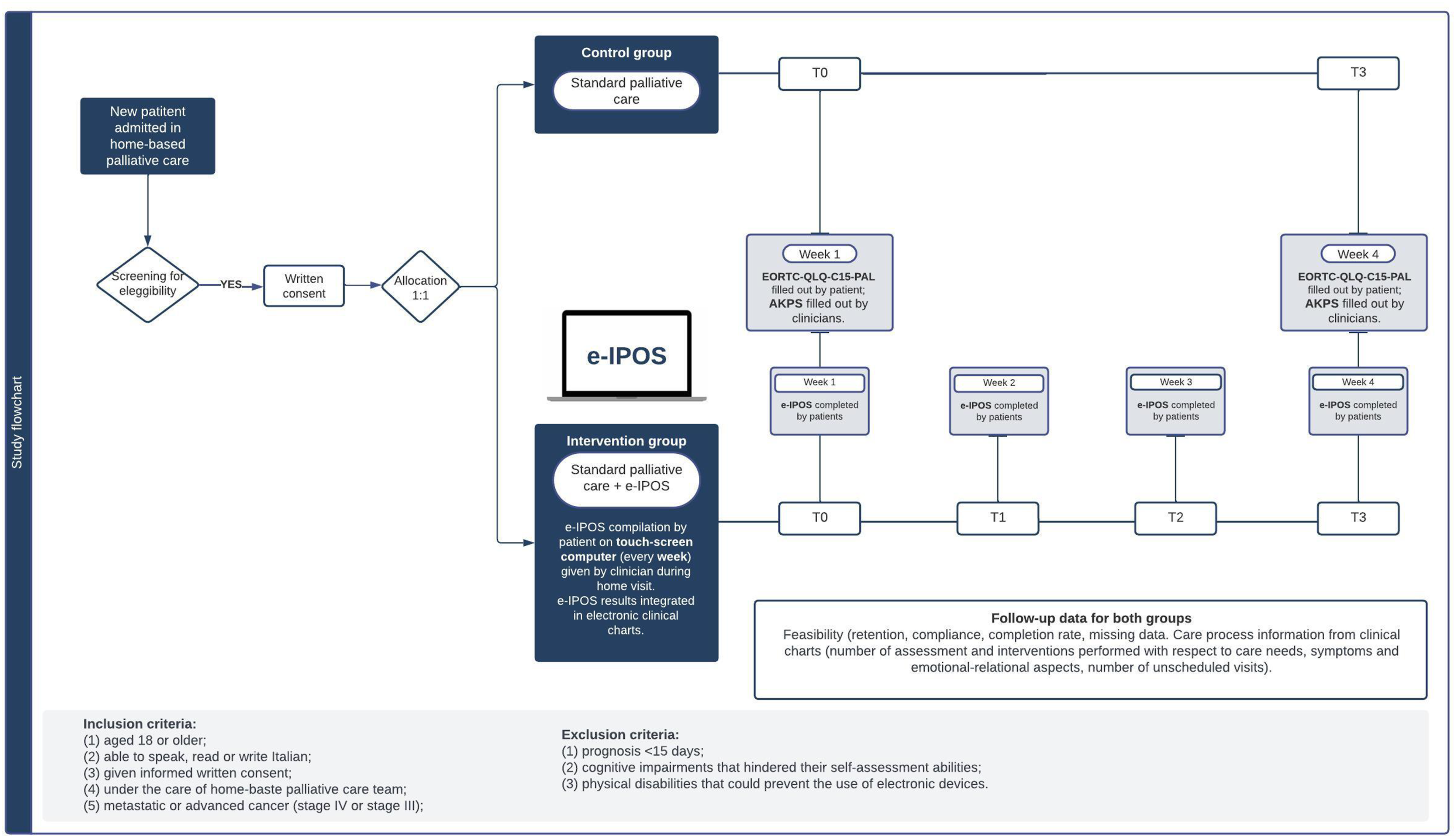
Standard Palliative Care Plus e-IPOS (Intervention)
The intervention group received standard home palliative care via a physician‒nurse pair and completed the e-IPOS every week at 4 time points (T0, T1, T2, and T3); in addition, they participated in visits from the researchers for the completion of the EORTC-QLQ-C15-PAL questionnaire (T0 and T3). The e-IPOS was completed via Ticuro Reply software using a touchscreen portable PC provided by healthcare staff during home visits. 43 This software facilitated the integration of the e-IPOS data with electronic records, which allowed response summaries to be exported and provided graphical representations to monitor progress over time.
Standard Palliative Care (Control)
The control group received standard home palliative care via a physician‒nurse pair, and the researchers visited the patients to complete the EORTC-QLQ-C15-PAL (T0 and T3) questionnaire.
Outcomes
Primary Outcomes
• Enrollment consent rate greater than or equal to 40%44-49; • Study retention rate at the end of follow-up greater than or equal to 80%.44-49 • Percentage of patients with a completed e-IPOS response in the planned follow-up period greater than or equal to 80%44-49; • Percentage of completeness of each e-IPOS response greater than or equal to 50%44,50; • Number of medical and nursing clinical assessments performed at each home visit and tracked in the documentation in relation to end-of-life care needs and problems in the follow-up period41,42; • Number of medical and nursing clinical interventions performed at each home visit and tracked in the documentation in relation to end-of-life care needs and problems in the follow-up period41,42
Secondary Outcomes
• Number of unscheduled visits performed by care staff and recorded in the electronic medical records; • Overall quality of life, physical function, emotional function, and intensity of symptoms perceived by patients (EORTC-QLQ-C15-PAL).
Statistical Analysis
All the data were included and reported, adhering to an intention-to-treat analysis approach. A comprehensive analysis of patient characteristics, including age, gender, nationality, education, and oncological pathology, was conducted, involving calculations of frequencies, means, medians, and standard deviations for various variable types. This included evaluating unscheduled visits and biophysiological, symptomatic, and emotional-relational aspects in the control and intervention groups. Chi-square tests were used to explore the relationships between nominal qualitative variables in these groups. Additionally, the e-IPOS utilization rate and completeness were calculated, as were temporal differences in e-IPOS scores at T0 and T3; these differences were analyzed using Student's t test and the Wilcoxon W test, with normality verified by the Shapiro‒Wilk test (P values compared against an alpha of .05). Similarly, AKPS scores were calculated and compared at T0 and T3. Furthermore, the latent factors of the EORTC-QLQ-C15-PAL were examined after linear scaling (0 to 100),51,52 calculating averages and ranges, and assessing response differences at T0 and T3 in both groups using Wilcoxon test post-normality verification. The software Jamovi (version 2.4) 53 was used for all the analyses.
Ethical Consideration
The participants in the study provided written consent. It was also guaranteed that their identity would remain anonymous and that the confidentiality of the data collected would be maintained. Strict data protection procedures were followed, and additional guarantees of confidentiality were provided. All the collected data were stored in a two-tier password-protected file to which the researchers had exclusive access to an encrypted computer. The study received ethical approval from the INT Ethics Committee [ref. 187/21].
Results
From March 2023, a total of 31 patients were found to be eligible, and 23 of them (74.19%) provided consent to participate in the study. Of the patients enrolled, 3 died before the T0 assessment. Of these, 2 patients were in the control group, and 1 was in the intervention group (retention rate of 86.96%)). These results show an enrolment consent rate greater than or equal to 40% and a study retention rate at the end of follow-up greater than or equal to 80%. No patients dropped out of the study during the planned follow-up period (Figure 2). Study enrollment flowchart.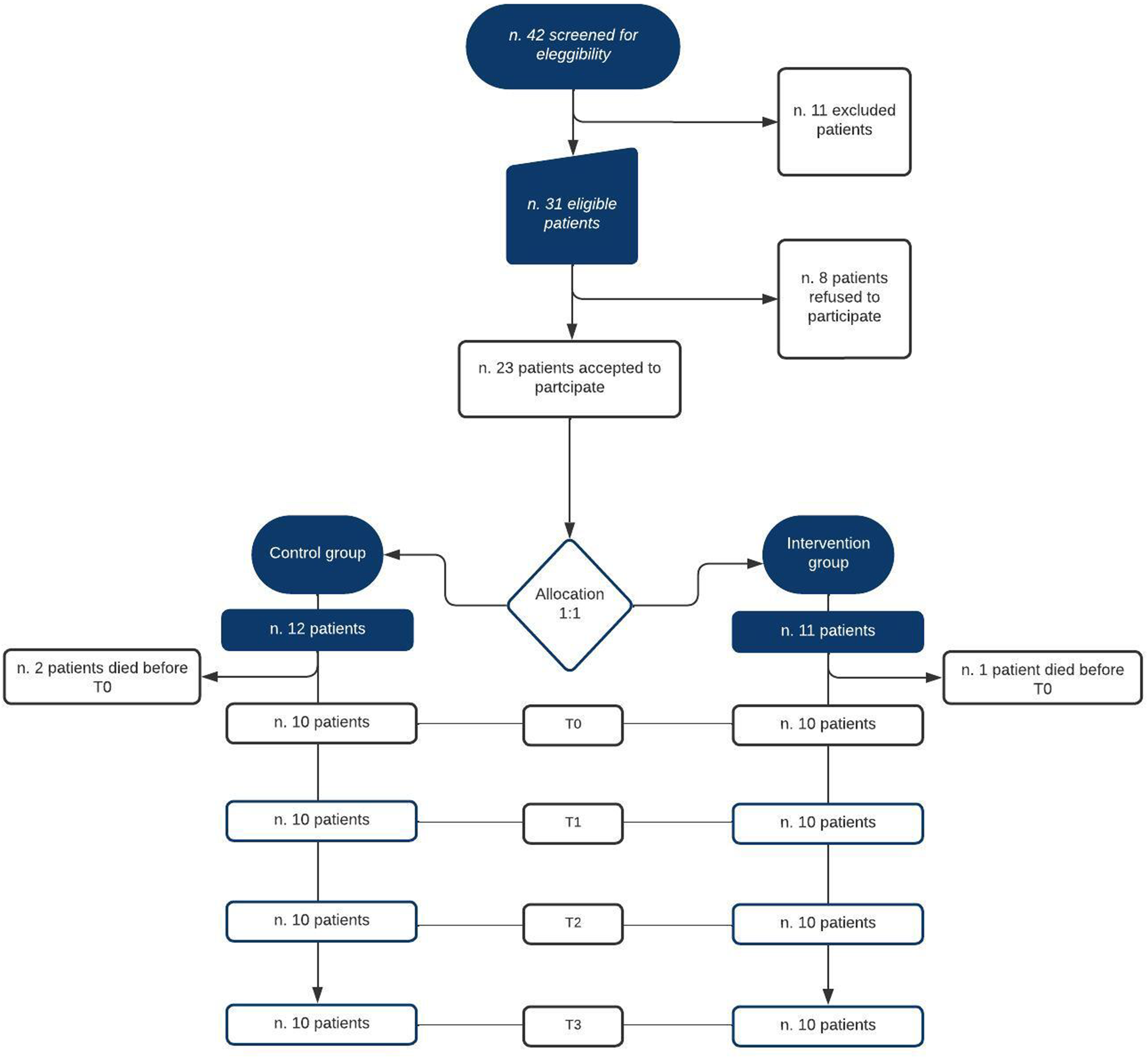
Enrolled Sample Characteristics.
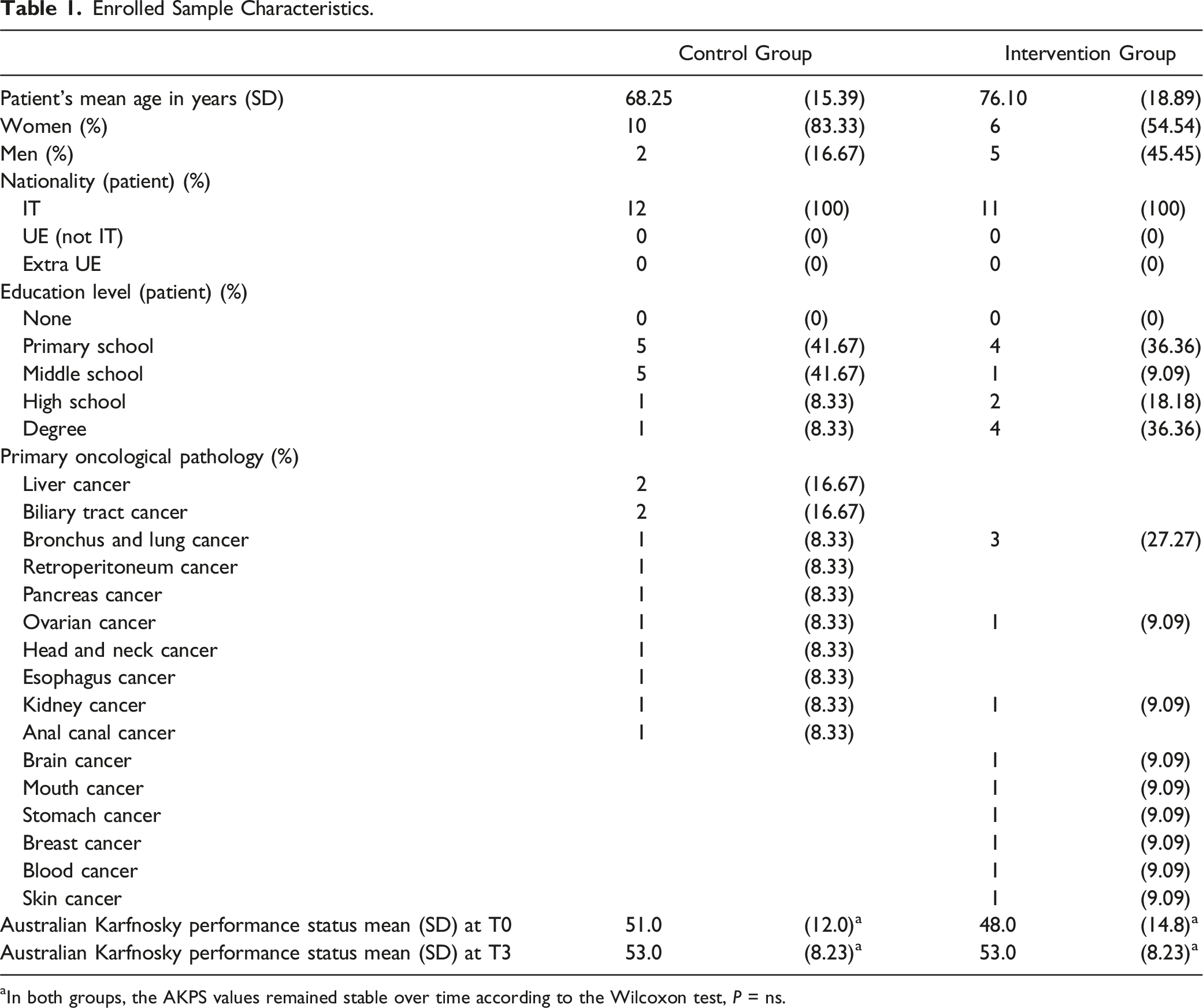
aIn both groups, the AKPS values remained stable over time according to the Wilcoxon test, P = ns.
There were 16 women (83.3% in the control group and 54.54% in the intervention group), with an average age of 72 years (68 in the control group and 76 in the intervention group). According to Wilcoxon, the AKPS remained stable over time (T0 – T3) in both groups with no statistically significant differences detected. Specifically, with values of 51 vs 48 in the control group and 53 vs 53 in the intervention group. Although, in the control group, there was a slight worsening of patient conditions over time, moving from T0, where the score recorded by clinicians on AKPS indicated patients who “require considerable assistance” to T3, which indicated patients “in bed more than 50% of the time”.
Acceptability and Feasibility of Using the e-IPOS
e-IPOS use Characteristics.
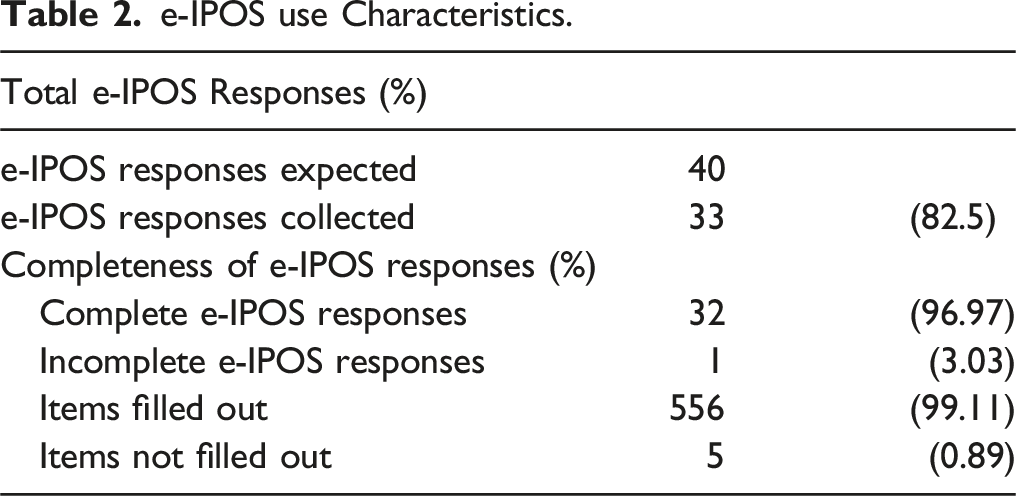
Out of the 40 expected e-IPOS (4 per each patient within 4 weeks), 82.5% were completed. 96.9% of them were complete in all their parts. In 87.8% of e-IPOS, at least 1 optional open-ended question was answered, with pain identified as the main issue and other primary concerns including the course of the disease, relationships, family issues, and the clinical pathway.
Traceability in the Clinical Documentation of Assessments and Interventions Performed
Frequencies of Unscheduled Visits, Assessments and Interventions Performed in the Control and Intervention Groups.
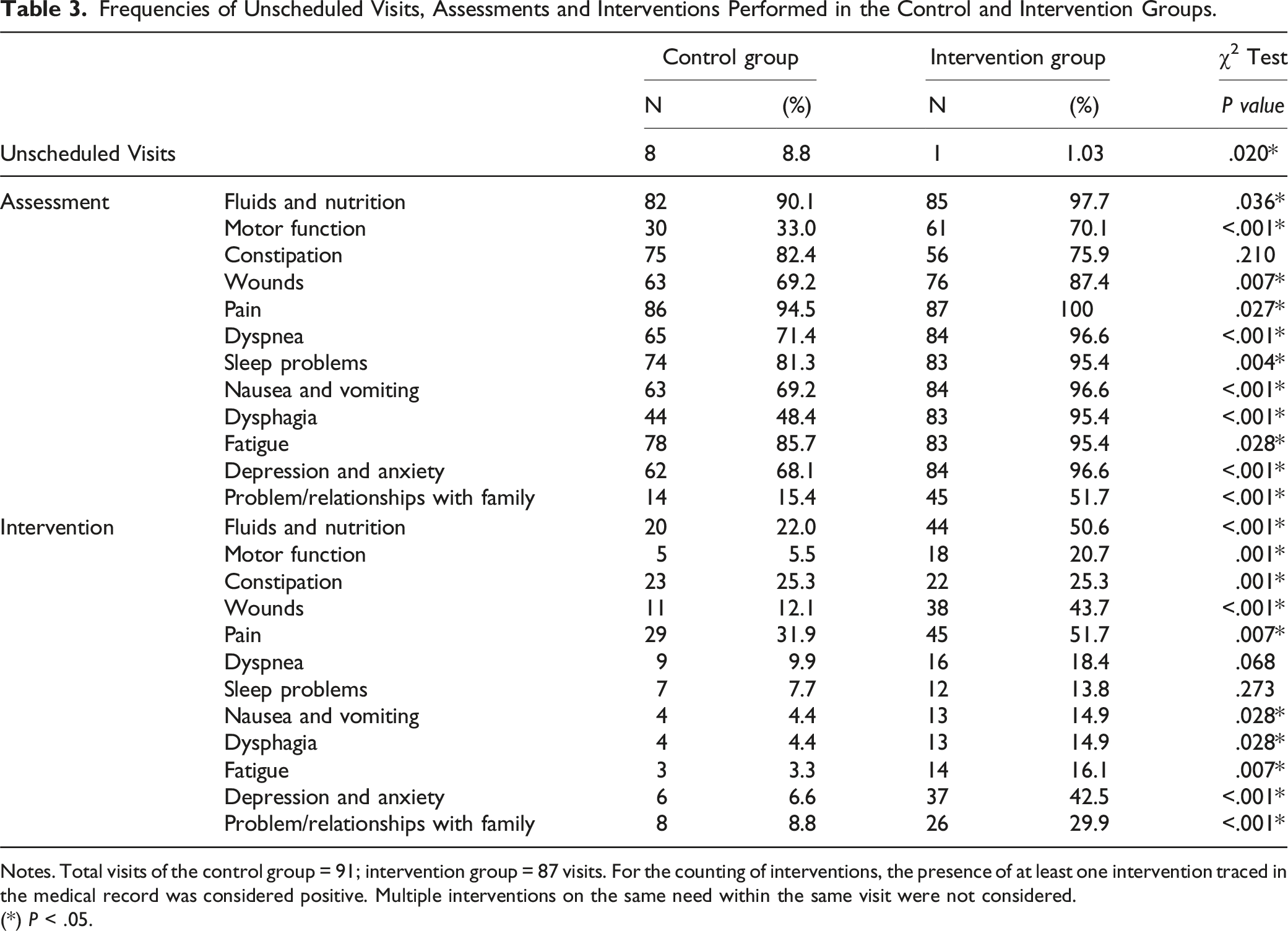
Notes. Total visits of the control group = 91; intervention group = 87 visits. For the counting of interventions, the presence of at least one intervention traced in the medical record was considered positive. Multiple interventions on the same need within the same visit were not considered.
(*) P < .05.
In the intervention group, a statistically significant decrease in unplanned visits was recorded during the follow-up period (P < .05), and a statistically significant increase in the number of identified needs during each home visit and documented interventions in the medical records was observed. These included those related to emotional and relational aspects, such as depression and anxiety, as well as issues with family relationships (P < .001).
Symptom Burden and Quality of Life
Overall e-IPOS Scores of Patients in the Intervention Group at T0 and T3.

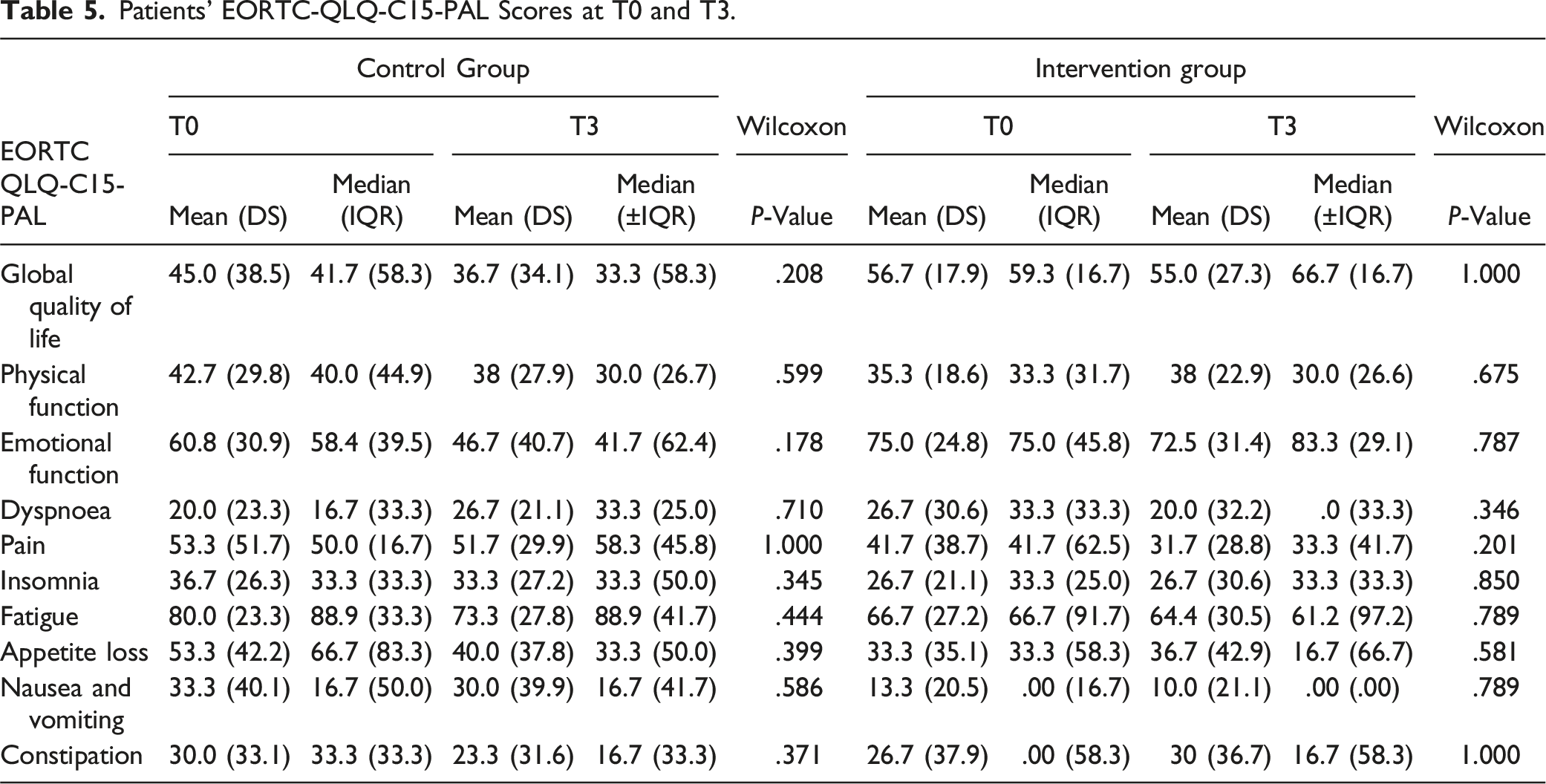
Patients’ EORTC-QLQ-C15-PAL Scores at T0 and T3.
Discussion
This pilot study was conducted to assess the feasibility of integrating the administration of the e-IPOS into the routine care processes of home-based palliative cancer care at the INT, where such an implementation has not been previously attempted. To our knowledge, the current study is the first feasibility study of an e-PROM system involving palliative care patients with cancer at home in Italy.
Despite the numerous challenges faced by end-of-life research, the data from this pilot study showed that the administration of the e-IPOS via touchscreen computers in clinical practice was technically feasible for cancer patients in home palliative care. In fact, there was a high recruitment rate (74.19%) and a drop-out rate of 13.04% related solely to patient death, consistent with studies conducted in the palliative care setting. 44-49 A high e-IPOS response rate and a high level of completeness of the responses were found, confirming that routinely collecting data through this tool was feasible and accepted by terminal cancer patients.
The compilation of the e-IPOS was primarily intended to facilitate the timely assessment and management of patients’ symptoms and problems by health care professionals. Additionally, it aimed to encourage patients to better self-assess their symptoms and the challenges related to their disease.
The integration of the e-IPOS system suggested a significant positive impact on monitoring, assessing, and managing patient symptoms and issues, especially notable in tracking data within electronic clinical records. Although the increase in the number of assessments and interventions due to this integration does not conclusively indicate a qualitative improvement in assessment and clinical actions targeted at the assisted person, it is certain that the introduction of the e-PROM has led to more complete and in-depth clinical documentation. This enhancement in documentation has improved the traceability of patients’ needs, suggesting a potential indirect benefit to the quality of care provided, potentially leading to superior patient outcomes and a more efficient allocation of healthcare resources, as suggested by previous literature.18,20 This approach was particularly effective in addressing emotional and relational issues such as depression, anxiety, and family relationships. The broader range of examined areas with e-IPOS suggested that some symptoms and issues crucial to quality of life are often unmeasured or underestimated by clinicians during visits, as outlined by other previous studies.8-10 Additionally, after 4 weeks of implementation, a statistically significant decrease in unplanned home visits was observed. This could not only underscore the effectiveness of the implemented system in enhancing patient care and monitoring but also imply a more proactive approach to managing patient conditions, potentially preventing acute episodes that necessitate unscheduled visits.
Concurrently, in the intervention group, a significant decrease in the overall e-IPOS score was noted, indicating a reduction in the patients’ perceived burden of symptoms and problems. The e-IPOS system, which also allows patients to answer open questions, led to reports of disease concerns, treatment management, and family tension. The expression of such concerns, along with the decrease in overall symptom burden over time and the significant increase in assessments and interventions recorded for psychological and relational reasons, offers initial insights regarding the extent of the impact of the introduction of the e-IPOS system on patient care in home palliative care. These results align with the existing literature on the effectiveness of e-PROMs for assisting patient communication about emotional, familial, and illness perspectives.8-10
It appears that completing the e-IPOS had no discernible impact on the patients’ perceived quality of life or performance status. The limited follow-up time due to the patients’ terminal status and disease trajectory did not allow the detection of all the potential effects of the e-IPOS on the overall perception of quality of life, as patients’ completion of the relevant questionnaire did not significantly differ.
Strengths and Limitations
This is a novel study in Italy investigating the feasibility of using the e-IPOS for people with a terminal illness assisted at home. This study has several limitations. First, the selection of eligible patients, mainly assessed by clinicians trained in eligibility criteria, might have introduced bias into the sample. Second, study adherence and completion rates may have been influenced by the organizational structure, wherein clinicians prompted patients to complete the e-IPOS weekly during routine home visits. Finally, the study’s quantitative approach restricted thorough investigation of patients’ perspectives on the feasibility, usability, usefulness, and impact of the e-IPOS after usage. Additionally, the nonrandomized nature, single-center nature, and sample size of the present study limit the generalizability of the data about the effects of administration of the e-IPOS on the care process and quality of life.
Conclusion
This research suggested that recruiting people with advanced cancer in home palliative care for a study on the implementation of an e-PROM system is feasible. The preliminary results indicate improved traceability in the assessment and intervention of patients’ symptoms and issues, which are often overlooked in standard care. Notably, the burden of these symptoms and unscheduled visits significantly decreases in patients using the e-IPOS. Future studies should be fully powered to investigate the feasibility of implementing the e-IPOS in routine care and assess patients’ perceptions of its use, aiming to better understand the clinical benefits for patients receiving palliative care.
Footnotes
Author Contributions
All the authors contributed to writing and reviewing the manuscript; LC, ML and TC contributed to the study conception and design and were responsible for the protocol and study procedure design. After the health care team took an initial approach with the participants, the LC discussed the study in more detail with them by telephone. LC, DR and SC recruited the participants, visited them at home to obtain written consent, administered the questionnaires and, if necessary, clarified any doubts about completion. LC, DR and IB were responsible for the data analysis and wrote the paper. ML, AC and TC commented on previous versions of the manuscript. All the authors have read, commented on, and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
