Abstract
Background:
Specialist paediatric palliative care services are promoted as an important component of palliative care provision, but there is uncertainty about their role for children with cancer.
Aim:
To examine the impact of specialist paediatric palliative care for children and young people with cancer and explore factors affecting access.
Design:
A mixed-methods systematic review and narrative synthesis (PROSPERO Registration No. CRD42017064874).
Data sources:
Database (CINAHL, Cochrane Database of Systematic Reviews, Embase, MEDLINE, PsycINFO) searches (2000–2019) identified primary studies of any design exploring the impact of and/or factors affecting access to specialist paediatric palliative care. Study quality was assessed using The Mixed Methods Appraisal Tool.
Results:
An evidence base of mainly low- and moderate-quality studies (
Conclusion:
Current evidence suggests that children and young people with cancer receiving specialist palliative care are cared for differently. However, little is understood about children’s views, and research is needed to determine whether specialist input improves quality of life.
Specialist paediatric palliative care is promoted as an important component of children and young adult cancer services, but there is uncertainty about the factors that affect access and the benefits for children who receive this specialist input.
Three reviews, which have aggregated evidence for children with all life-limiting conditions, suggest that the benefits of specialist palliative care include less time in hospital and improvements in quality of life and symptom management.
The growing number of studies investigating the role of specialist palliative care for children with cancer report mixed results and varying provision, and there is a need to aggregate this evidence to inform future policy and practice.
Accessing specialist paediatric palliative care is associated with less intensive care at the end of life, more advance care planning and fewer in-hospital deaths for children and young people with cancer, but there is no robust evidence to tell us whether these services lead to improvements in quality of life or symptom management.
Children with haematological malignancies are less likely to receive specialist palliative care than children with other cancers.
Uncertainty about when to introduce palliative care services to families, what it comprises and the added value of specialist input was identified as a key barrier to access, as were perceptions that paediatric oncology teams already meet the palliative care needs of their patients.
Evidence is still needed to determine whether specialist paediatric palliative care improves the quality of life and symptom management for children and young people with cancer.
Exploration of why children with certain cancers are less likely to receive specialist palliative care at the end of life may help address this inequality in access.
A core outcome set study including the views of children and families would help improve future aggregation of evidence in this area.
Background
Approximately 20% of children and young people diagnosed with cancer do not survive despite significant medical advances in recent decades.1,2 The majority of deaths are due to the malignancy, with some attributable to anti-cancer treatments. 3 Distress from symptoms and suffering during the end-of-life phase can be significant,4–6 impacting on the child and their family’s quality of life.7,8 In addition, many children and young people who die from cancer continue to have high-intensity treatments towards the end of life,9,10 with nearly half dying in the acute care setting, 11 despite preferences from the majority of children and their parents for being at home during the end-of-life phase.12,13
Palliative care, defined by the World Health Organization (WHO) as ‘the active total care of the child’s body, mind and spirit . . . [that] begins when illness is diagnosed, and continues regardless of whether or not a child receives treatment directed at the disease’, 14 is recognised as an important component of children and young people’s cancer services.15,16 In addition, and over the last 30 years, specialist paediatric palliative care services for children and young people have been developing in many countries including the United States, Canada, United Kingdom and across Europe. 17 The English National Health Service (NHS) defines specialist paediatric palliative care as ‘a consultant-led multi-professional specialist palliative care team . . . led by a medical consultant working at Paediatric Palliative Care Competency Level 4’. 16 In practice, however, the models of providing specialist palliative care vary within and between countries, including, for example, hospital- and community-based teams which support all children with life-limiting conditions, 18 teams embedded within paediatric oncology departments, joint working with, or hospice-led provision, and specialist nurse-led teams as well as services led by a paediatric palliative care consultant.17,19,20
Even within the developed world, the availability, referral and uptake of specialist palliative care among children and young people with cancer remains low and variable between and within countries and settings, and it is not clear to what extent these services are addressing all aspects of palliative care as defined by WHO.17,21–25 For children with cancer, referral to palliative care also often occurs late in the trajectory of illness, sometimes only days before death.26,27 Recent systematic reviews suggest that access to specialist palliative care services is associated with improvements in quality of life, symptom control, perceived support, reduced time in hospital, less invasive treatment and greater advance care planning.19,28,29 However, these reviews have aggregated the results for children and young people across conditions, and the evidence for those with cancer remains unclear because of conflicting results between individual studies 28 and the lack of work exploring condition-specific factors that may influence access to and benefit from specialist palliative care services.30,31
A rigorous review of the evidence on the impacts of specialist paediatric palliative care for children and young people with cancer and their families is both crucial to informing debates within paediatric oncology regarding the positioning and role of these specialist services and for future service development. This mixed-methods systematic review synthesises the existing evidence on the benefits, drawbacks, facilitators and barriers associated with referral to and uptake of specialist paediatric palliative care for children and young people with cancer and their families.
Methods
The review questions are as follows:
What are the reported benefits and drawbacks of referral to specialist paediatric palliative care for children and young people with cancer and their families?
What are the factors (e.g. barriers, facilitators) affecting referral to and uptake of specialist paediatric palliative care for children and young people with cancer?
The protocol registration number is CRD42017064874. 32
Eligibility criteria
Primary studies of any design (e.g. experimental, observational, surveys, consensus and qualitative studies) examining either the impact of or factors affecting specialist paediatric palliative care access for children and young people (age 0–24 years) with cancer were included. Studies of a mixed population were included if (1) the majority of the participants were children and young people with cancer and/or (2) data were reported separately.
We defined specialist paediatric palliative care as care provided by multidisciplinary teams or palliative care services which included clinicians (e.g. oncologists, paediatricians, nurses) with paediatric palliative care training, or services who self-identified as providing specialist paediatric palliative care. We included services delivered in different settings (e.g. inpatient, community or home settings) and both liaison services (e.g. supporting the child’s usual care team) and services directly supporting children and their families. Although broad, this reflects the varying provision of specialist palliative care for children within and between different countries and so enabled us to synthesise the evidence about these specialist services.
To understand the different perspectives on referral to specialist palliative care services, studies that included the following participant groups were eligible: children and young people; parents (including bereaved); other family members and health and social care staff. No comparator was required.
We excluded case studies, review articles, descriptive, theoretical or clinical opinion articles, conference abstracts and articles not published in the English language. We also restricted the eligibility to studies conducted in high-income countries (defined as OECD (Organisation for Economic Co-operation and Development) 33 member countries) because of the very different healthcare infrastructure and status of specialist paediatric palliative care in developing countries. 17
Search strategy
Electronic databases (CINAHL, Cochrane Database of Systematic Reviews (CDSR), Embase, MEDLINE, PsycINFO) were searched on 27 June 2017 (from 2000, in line with increasing availability of specialist paediatric palliative care services internationally 19 ). The search strategy consisted of terms and synonyms for [malignancies] AND [children] AND [specialist paediatric palliative care] (see Supplementary Information Appendix A.) Reference lists of included studies and relevant literature reviews were checked, and backward and forward citation searching of included studies and PubMed-related articles link searches were undertaken. An update search in MEDLINE (which identified all the eligible studies in the original search) was performed on 13 September 2019.
Study selection
Titles and abstracts were screened in Covidence, 34 and relevant full-text articles were retrieved and independently assessed for eligibility by two reviewers with disagreements resolved via a third reviewer. 35 Reasons for exclusion at full text were recorded.
Data extraction
Data on study characteristics, methods, study focus (e.g. impact and/or factors affecting access) and quantitative outcome data were extracted into Microsoft Excel using a pre-piloted data extraction template. Qualitative data, including author-reported results, direct quotations and results tables, were imported into NVivo version 11 36 for analysis.
Data extraction was carried out by one reviewer and checked by a second.
Critical appraisal
The Mixed Methods Appraisal Tool (MMAT)37,38 was used to appraise the methodological quality of all included studies. MMAT comprises two generic screening questions and an additional four criteria for use with specific study designs. Criterion assessments (e.g. Is the sample representative of the population under study?) are categorised ‘Yes’, ‘No’ or ‘Can’t tell’. An overall quality score (of 0%, 25%, 50% or 100%) based on how many study design specific criterion were met (those categorised as ‘Yes’) was calculated for each study.
The quality assessment was undertaken independently by two reviewers and informed the synthesis methods and reporting of the review results, along with identifying needs for future research. We did not exclude studies based on quality assessment.
Data synthesis
An integrative narrative synthesis was planned drawing on interpretative review methodology 39 with thematic analysis as the principal method of synthesis. 40 The synthesis plan outlined in the protocol 32 was modified following an assessment of the potential for aggregation, configuration and integration of study findings. The final method involved separate syntheses of data reporting the impact of specialist paediatric palliative care and factors affecting access (both included quantitative and qualitative data). Each comprised the following steps:
Results
A total of 8549 unique records were screened by title and abstract, 626 full-text articles were retrieved and reviewed, and 49 articles describing 42 studies23,43–90 were included in the review (see Figure 1).
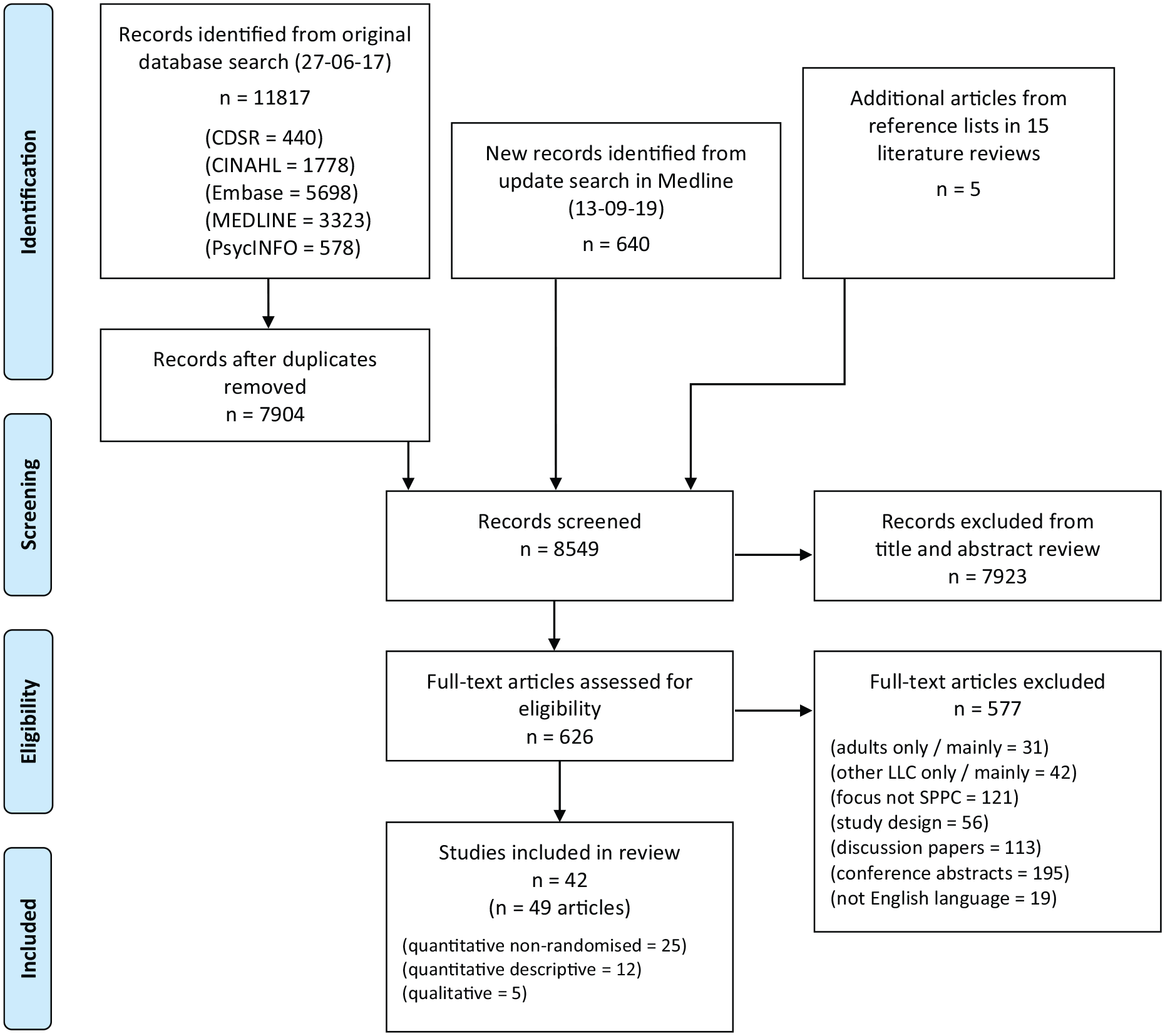
Study flow diagram.
Study characteristics
Of the 42 included studies, 11 examined the impact of specialist paediatric palliative care,44,48,57,58,62,64,66,70,73,74,88 14 explored factors affecting access23,43,45,56,65,68,71,75,76,78–83,89 and 17 studies investigated both46,47,49–55,59–61,63,67,69,72,77,84–87,90 (see Table 1 for study characteristics). Using the MMAT, 25 studies were categorised as quantitative non-randomised, 12 as quantitative descriptive and 5 as qualitative studies. The majority of studies were conducted in the United States (
Characteristics of included studies.
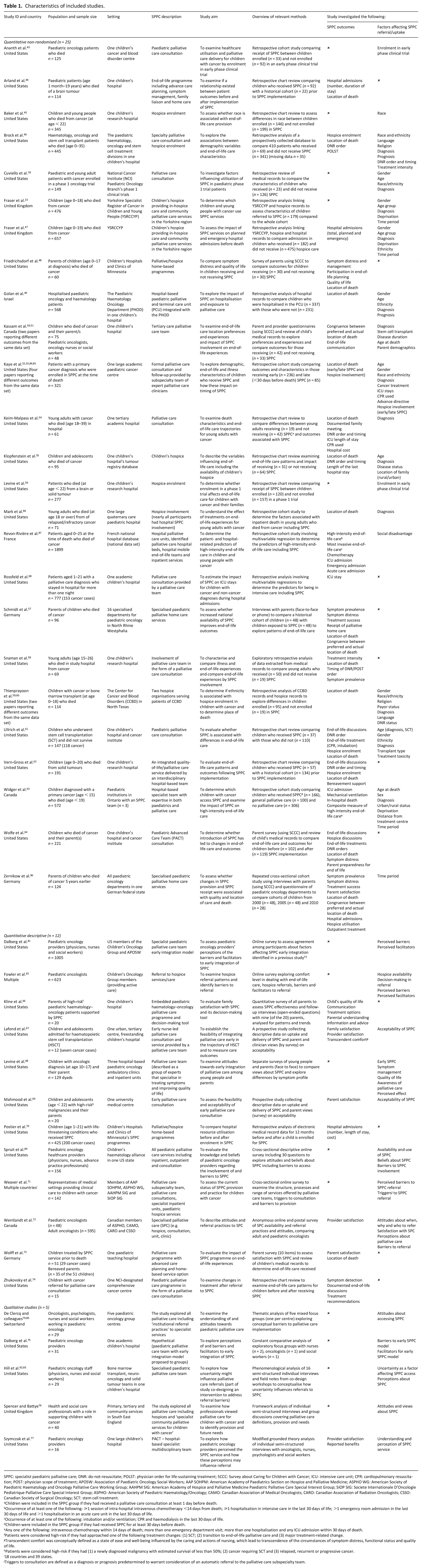
SPPC: specialist paediatric palliative care; DNR: do-not-resuscitate; POLST: physician order for life-sustaining treatment; SCCC: Survey about Caring for Children with Cancer; ICU: intensive care unit; CPR: cardiopulmonary resuscitation; POST: physician scope of treatment; APOSW: Association of Paediatric Oncology Social Workers; AAP SOHPM: American Academy of Paediatrics Section on Hospice and Palliative Medicine; ASPHO WG: American Society of Paediatric Haematology and Oncology Palliative Care Working Group; AAHPM SIG: American Academy of Hospice and Palliative Medicine Paediatric Palliative Care Special Interest Group; SIOP SIG: Societe Internationale D’Oncologie Pediatrique Palliative Care Special Interest Group; ASPHO: American Society of Paediatric Haematology/Oncology; CAMO: Canadian Association of Medical Oncologists; CARO: Canadian Association of Radiation Oncologists; CSSO: Canadian Society of Surgical Oncology; SCT: stem cell treatment.
Children were included in the SPPC group if they had received a palliative care consultation at least 1 day before death.
Occurrence of at least one of the following: ⩾1 session of intra-hospital intravenous chemotherapy <14 days from death; ⩾1 hospitalisation in intensive care in the last 30 days of life; >1 emergency room admission in the last 30 days of life and >1 hospitalisation in an acute care unit in the last 30 days of life.
Occurrence of at least one of the following: intubation and/or ventilation; CPR and haemodialysis in the last 30 days of life.
Children were included in the SPPC group if they had received SPPC for at least 30 days before death.
Any one of the following: intravenous chemotherapy within 14 days of death; more than one emergency department visit; more than one hospitalisation and any ICU admission within 30 days of death.
Patients were considered high-risk if they had approached one of the following treatment changes: (1) SCT; (2) transition to end-of-life palliative care and (3) major treatment-related change.
Transcendent comfort was conceptually defined as a state of ease and well-being influenced by the caring and actions of nursing, which lead to transcendence of the circumstances of symptom distress, functional status and quality of life.
Patients were considered high-risk if they had (1) a newly diagnosed malignancy with estimated survival of less than 50%; (2) cancer requiring SCT and (3) relapsed, recurrent or progressive cancer.
18 countries and 39 states.
Triggers to consultation are defined as a diagnosis or prognosis predetermined to warrant consideration of an automatic referral to the palliative care subspecialty team.
Study populations
Thirty-one studies (all quantitative) examined the impact of and/or characteristics of children receiving specialist palliative care; 24 included a comparator group of children not receiving this,23,43–51,54–64,78,86–88,90 1 compared children receiving late and early specialist input,52,53,84,85 and 6 used a single-group study design.66,67,69,70,73,74 Of the remaining 11 studies (6 quantitative and 5 qualitative), 10 explored the views of healthcare staff65,71,72,75–77,79–83,89 and 1 the views of parents and young people. 68
Of the 31 studies examining outcomes and/or characteristics of children, the majority (

Data collection period for studies examining outcomes or characteristics of children receiving specialist paediatric palliative care.
In total, data for 7933 children and young people, 4289 of whom had received specialist palliative care compared to 3644 who did not, were included. While the majority (
Of the 11 studies exploring stakeholder views, 3 recruited paediatric oncology staff from single hospitals75,77,82,83 and 1 from multiple hospitals,79,80 1 recruited staff involved in providing palliative care to children with cancer from primary, tertiary and community settings, 76 4 recruited paediatric oncologists via professional organisations65,71,72,81 and 1 recruited parent and young person dyads from three hospitals. 68 In total, these studies represented the views of 1133 physicians, 986 other healthcare professionals (mainly nurses, social workers and other staff working in paediatric oncology, but also in palliative care and other settings), 129 parents and 129 young people.
Models of delivery
We identified five broad service types from the included studies: hospital-based palliative care teams with referral triggering an initial consultation46,50–53,58,61–64,67,69,73–75,77,82–85,87,88 (
Very few studies provided specific details about the team or service providing palliative care in terms of skills mix, role and the extent of provision. There were also some anomalies between studies; for example, five studies used hospice discussions or enrolment as an outcome of specialist palliative care,46,61,62,64,90 whereas in other studies hospices were defined as the source of specialist intervention. Not all studies investigating hospice settings specified the characteristics (e.g. adult vs children’s hospice), and it was difficult to determine what ‘home-based services’ might comprise. Among the studies exploring stakeholder views, three offered only ‘hypothetical models’ for participants to consider68,75,76 and three focused primarily on views about early integration of specialist palliative care, which was defined as close to the time of diagnosis.68,75,81
Only two studies accounted for the timing of the initial palliative care consult/enrolment when deciding which children to classify as having received specialist intervention; one only included children who had received specialist palliative care for more than 30 days 63 and the second for more than 1 day. 54 A third study, which compared early and late interventions, included all children receiving specialist palliative care but defined early provision as that received for more than 30 days.52,53 Only two studies explored how the duration of specialist paediatric palliative care exposure affected outcomes.61,70
Study quality
The quality and reporting of studies varied greatly among the 37 quantitative studies, with scores ranging from 0% to 100% (see Table 2).
Critical appraisal summary for included studies.
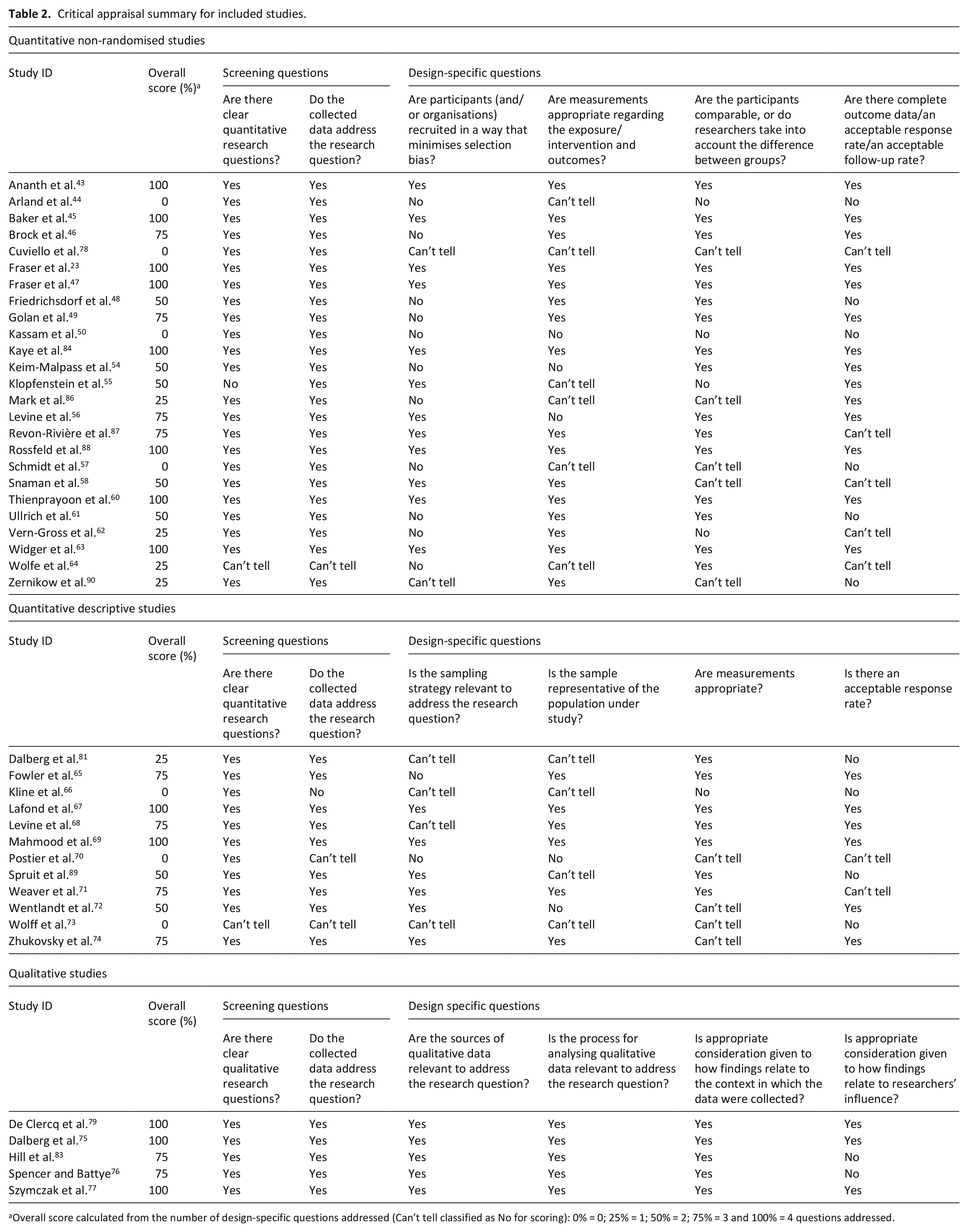
Overall score calculated from the number of design-specific questions addressed (Can’t tell classified as No for scoring): 0% = 0; 25% = 1; 50% = 2; 75% = 3 and 100% = 4 questions addressed.
There were concerns about the representativeness of samples in 14 of the quantitative studies, due to the single-site design of many studies, long study period and inappropriate participant selection or recruitment methods.44,46,48–51,54,57,61,62,64,70,72,86,89 Assessment of representativeness was not possible in another seven studies because of poor reporting.66,68,73,78,81,89,90 In eight studies, there were concerns about the comparability of groups (e.g. use of historical cohorts, different participant characteristics), which were not accounted for in the analysis.44,50,51,54,55,57,58,62,90
Although inappropriate measures were identified in only three studies,50,51,56,66 wider concerns about measurement were evident across studies. These included how receipt of specialist palliative care was determined (e.g. from day of initial palliative care consultation); whether outcomes could be attributed to specialist provision or care from the primary oncology team; potential recall problems in studies using bereaved parent-reported outcomes and failure to take account of the chronology of variables. For example, having a do-not-resuscitate (DNR) order was a predictor of specialist palliative care in some studies46,59,60 and an outcome in others.54,58 Determining the extent of these limitations was hindered by poor reporting of study methods.
The five qualitative studies75–77,79,83 used appropriate methods for sampling, data collection and analysis. However, there were limitations to the transferability of findings due to the single-site design in three studies,75–77,83 and for the third, the time period elapsed since the study was conducted (published in 2001 when specialist paediatric palliative care for children with cancer was at an early stage of development). 76
The impact of specialist paediatric palliative care
A total of 17 distinct outcome domains were identified and these were categorised under one of six overarching categories: advance care planning (
Impact of specialist paediatric palliative care by outcome domain.
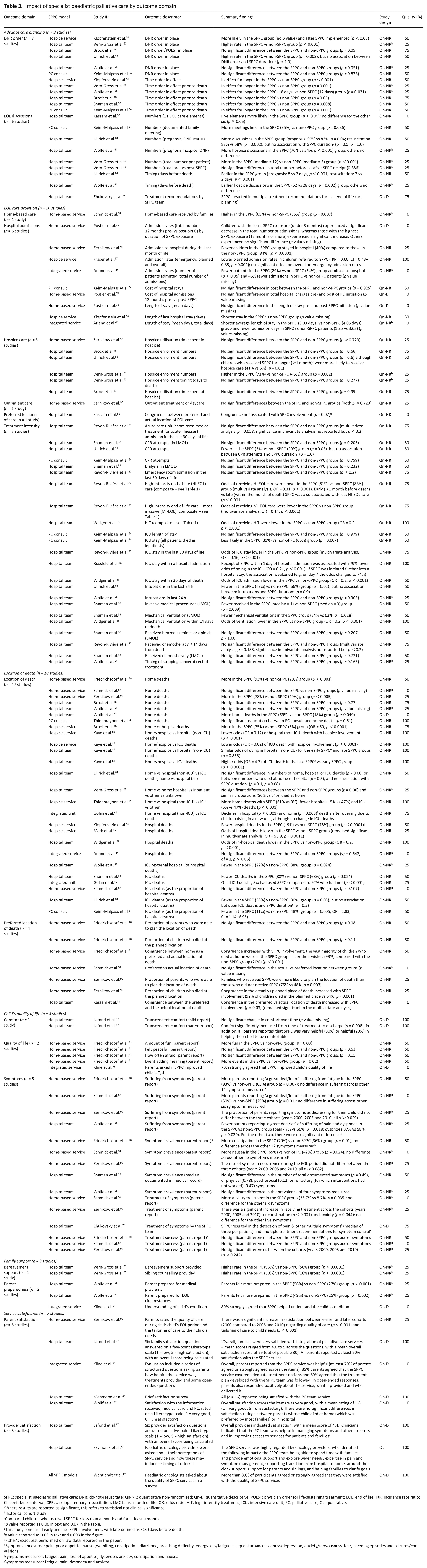
SPPC: specialist paediatric palliative care; DNR: do-not-resuscitate; Qn-NR: quantitative non-randomised; Qn-D: quantitative descriptive; POLST: physician order for life-sustaining treatment; EOL: end of life; IRR: incidence rate ratio; CI: confidence interval; CPR: cardiopulmonary resuscitation; LMOL: last month of life; OR: odds ratio; HIT: high-intensity treatment; ICU: intensive care unit; PC: palliative care; QL: qualitative.
Where results are reported as significant, this refers to statistical not clinical significance.
Historical cohort study.
Compared children who received SPPC for less than a month and for at least a month.
This study compared early and late SPPC involvement, with late defined as <30 days before death.
Fisher’s exact test performed on raw data reported in the paper.
Symptoms measured: pain, poor appetite, nausea/vomiting, constipation, diarrhoea, breathing difficulty, energy loss/fatigue, sleep disturbance, sadness/depression, anxiety/nervousness, fear, bleeding episodes and seizures/convulsions.
Symptoms measured: fatigue, pain, loss of appetite, dyspnoea, anxiety, constipation and nausea.
Symptoms measured: fatigue, pain, dyspnoea and anxiety.
Advance care planning
The outcome domains were of
End-of-life care provision
There was evidence from the seven studies which measured
There was no evidence to suggest that
Location of death
A total of 17 studies examined differences in
Child’s quality of life
This theme included three outcome domains:
Family support
Only three studies investigated whether specialist paediatric palliative care affected provision of support to family members, or the impact of that support. One study found that use of
Service satisfaction
All seven studies (six surveys66,67,69,72,73,90 and one qualitative 77 ) assessing this reported high levels of family and professional satisfaction with specialist palliative care, with reported benefits including expertise in pain and symptom management, time to plan end-of-life care with families and meeting psychosocial and family needs. None of these studies included a comparator group, although one reported increasing parental satisfaction in line with increasing provision of specialist services.
Factors affecting specialist paediatric palliative care access
The synthesis of studies exploring factors affecting specialist paediatric palliative care access identified four overarching categories each containing several linked themes: sociodemographics (
Factors affecting access to specialist paediatric palliative care by theme.
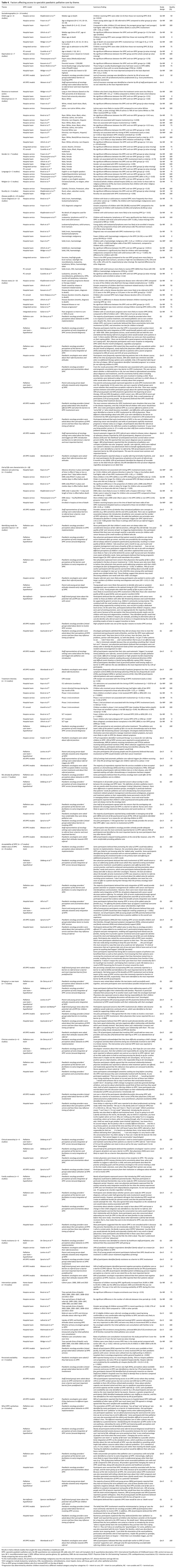
Results in Italics indicate studies that sought the views of families or healthcare staff.
SPPC: specialist paediatric palliative care; Qn-NR: quantitative non-randomised; OR: odds ratio; SCT: stem cell transplant; Qn-D: quantitative descriptive; ICCC: International Classification of Childhood Cancer; CNS: central nervous system; NF-1: neurofibromatosis type 1; DIPG: diffuse intrinsic pontine glioma; QL: qualitative; DNR: do-not-resuscitate; POLST: Physician Order for Life-Sustaining Treatment; CPR: cardiopulmonary resuscitation; ICU: intensive care unit.
Study comparing late versus early SPPC.
In the multivariable analysis, the presence of a hematologic malignancy was the only factor that remained significant; SCT, disease duration and age did not.
ICCC categories include leukaemia, lymphoma, CNS, neuroblastoma, retinoblastoma, renal, hepatic, bone, soft tissue, germ cell, other epithelial and other.
The no-SPPC group included children who first received SPPC on the day of death.
Progression was determined by whether children moved into a different medical category using four categories: IC – intent to cure, ICP – intent to cure palliative, NC – non-curable and TC – terminal care.
Sociodemographics
Multiple quantitative studies which investigated the sociodemographic profile of children and/or their families receiving and not receiving specialist palliative care consistently showed that access was not associated with a child’s
Disease profile
Out of 12 studies examining
Uncertainty about a child’s prognosis and about the benefits of introducing specialist palliative care earlier in the disease trajectory (e.g. around diagnosis) were identified as key barriers to a timely referral,65,71,75,77,79,81,83,89 although non-physician professionals75,81 and families 68 were more receptive to early integration than physicians. Automatic referral triggers were used by the majority of providers in one study. However, in line with reported practice, most encouraged referrals for children during the end-of-life phase of care. 71
End-of-life care characteristics
The end-of-life care provided to children was found to influence whether or not children received specialist palliative care. Four studies consistently found that
There was conflicting evidence about whether
Acceptability of specialist paediatric palliative care
Staff acceptability of specialist palliative care was reported to influence access in 16 studies. Family acceptability was identified in 13, although families’ views were sought in only three of these.67–69 Eight studies measured
Staff uncertainties about the benefits of specialist palliative care, and about how a specialist service differs from care provided within oncology (the
Staff in eight studies identified the association of palliative care with end of life (
Oncology staff reported that
Discussion
Main findings of the review
This systematic review found evidence that children and young people with cancer who receive specialist paediatric palliative care are more likely to be engaged in advance care planning, receive less intensive care at the end of life and are less likely to die in hospital, compared to those who do not receive this. Some of the included studies also indicate that these differences may be more marked when children receive specialist input for a longer duration before they die. The review did not find that receipt of specialist palliative care is associated with improved quality of life or symptom control; however, no conclusions can be drawn because of the significant methodological limitations of the seven studies investigating this.48,57,58,64,66,67 Importantly, only one study sought young people’s views about the impact of these specialist services across the 28 studies which examined this, and this was a feasibility study with no comparator group. 67
The review also found that the type of cancer and whether or not paediatric oncology teams themselves engage in palliative care practices may affect access to specialist palliative care services. Thus, studies reported fewer children with haematological malignancies receiving specialist palliative care, and involvement more likely where the oncology team were proactively addressing palliative care needs (e.g. evidence of advance care planning). There was no indication from the studies included about why children with certain cancers are less likely to receive specialist palliative care, and a recent review of barriers to access did not explore this. 31 Evidence from adult cancer 91 indicates that the remitting and relapsing trajectories of haematological malignancies, a more aggressive approach to treatment and greater difficulties predicting prognosis may contribute to the observed inequity of specialist palliative care involvement. 92 Evidence from our review regarding clinician uncertainty about when to involve specialist palliative care, combined with the commonly reported practice of referring children with a poor prognosis, 27 suggests this might be the case.
Clinicians’ views regarding the need for specialist palliative care input for their patients and their acceptability of specialist palliative care services were also consistently reported as affecting referral practices. More specifically, views on how these specialist services differ to palliative care provided by the oncology team, perceived drawbacks associated with involving a new team, readiness to accept a change in prognosis and the negative connotations associated with the term ‘palliative care’ were identified as barriers to referral. Concerns among clinicians about how families might react to the offer of a referral was a common theme in several studies, and although the study that explored young people and parents’ views runs counter to this, wider work on this issue reports mixed opinions among young people93,94 and parents73,74 about palliative care and how this should be introduced.95,96
Strengths and limitations
This review is the first to systematically synthesise the available evidence about specialist paediatric palliative care for children and young people with cancer. Strengths of the review include a published protocol, robust search, independent screening and data extraction by two reviewers, and the use of appropriate mixed-methods techniques to synthesise the results. There are, however, limitations in the conclusions which can be drawn from this review due to the heterogeneity of study populations and interventions. This, and the substantial risk of potential bias identified in some of the studies and inconsistency of measurement across studies, meant it was inappropriate to aggregate the results statistically, or to compare results between the different approaches to providing specialist palliative care. In addition, the descriptions of specialist services and the palliative care provided by oncology teams were typically very poor, making interpretation of the differences between these challenging.
What this review adds
Our finding that end-of-life care is different for those who receive specialist paediatric palliative care compared to those who do not broadly aligns with three recent reviews about children with all life-limiting conditions.19,28,29 Two of these reviews19,29 concluded that specialist intervention appears to offer benefit in terms of improved quality of life. By focusing only on children and young people with cancer, our review highlights the lack of robust evidence pertaining to both quality of life and symptom burden in this population. Although the broader literature implies that other differences we observed, such as reduced hospital stays and more advance care planning, are indicative of better care, 63 there is no evidence that these changes in the delivery of care reflect family preferences,48,51,57 or lead to reduced symptom burden, which studies continue to show is significant for children with cancer.5,90,97
The integration of findings from evaluation studies and those which have explored factors affecting access tells us that while end-of-life care may be different for children who receive specialist palliative care, children who receive this are also different to those who do not, particularly in terms of their disease profile and care processes. Synthesis of the qualitative studies go some way to explaining these differences, highlighting in particular the central role of clinicians’ emotions, beliefs and attitudes in shaping referral practices, and the ongoing uncertainty about when to initiate palliative care and whether this should be provided by a specialist service or the oncology team. It also reveals that, in practice, decisions and discussions about no longer pursuing curative treatments and introducing specialist palliative care go hand in hand, and that families who are comfortable discussing an uncertain future or families cared for by clinicians who have this confidence may be more likely to receive specialist palliative care.75,77,83
Interventions that support clinicians to initiate palliative care with families and improve clinical acceptability of specialist palliative care services therefore offer the potential to address these key barriers to access.82,98 Although there are various initiatives to support the delivery of palliative care within oncology services (e.g. palliative care training,99,100 communication tools,101,102 early integration models26,67,69), we know very little about whether these are being implemented in practice and how they might influence referral to specialist palliative care. There is also little available evidence about whether families play an active role in the initiation of palliative care, or whether clinicians’ concerns about how families will respond to a referral are warranted. 68
Future research should therefore investigate the effectiveness, delivery and acceptability of the different models of delivering palliative care for children and young people with cancer, particularly outside of North America. Future research also needs to examine factors affecting uptake from families’ perspectives and to explore the role of socioeconomic factors. This conclusion is supported by the recent priority setting partnership results for teenagers and young adults with cancer, 103 which includes how best to support young people who have incurable cancer and their families. In order to undertake this research, we must first determine what outcomes are the most important to measure and develop appropriate tools to measure them.104–107 Development of a core outcome set would meet this requirement. 108 This too will need to include the views of children and young people and their families, particularly if we are to address the methodological challenges that continue to affect the quality of research in this area, and the lack of evidence about whether specialist paediatric palliative care improves quality of life for children and their families.
Supplemental Material
Appendix_A_-_Medline_search_strategy – Supplemental material for Specialist paediatric palliative care for children and young people with cancer: A mixed-methods systematic review
Supplemental material, Appendix_A_-_Medline_search_strategy for Specialist paediatric palliative care for children and young people with cancer: A mixed-methods systematic review by Johanna Taylor, Alison Booth, Bryony Beresford, Bob Phillips, Kath Wright and Lorna Fraser in Palliative Medicine
Footnotes
Acknowledgements
The authors would like to acknowledge the input of members of the Martin House Research Centre Family Advisory Board, who helped with interpretation of the review findings.
Author contributions
B.B., B.P. and L.F. conceived the original study. J.T., A.B., B.B., B.P., K.W. and L.F. contributed to the design and methods. K.W. prepared the search strategy. J.T., B.B., B.P., A.B. and L.F. screened studies for inclusion. J.T. and A.B. carried out data extraction. J.T., A.B. and L.F. conducted the critical appraisals. J.T. conducted the synthesis with input from A.B., B.B., B.P. and L.F. J.T. drafted the manuscript with contributions from A.B., B.B., B.P., K.W. and L.F. The final manuscript was approved by all authors.
Data management and sharing
Data are reported fully in the included tables and figures and are derived from published sources only.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval and informed consent
This review contains data derived from published sources only.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This article presents independent research partly funded by the Wellcome Trust (Ref. No. 204829) through the Centre for Future Health at the University of York, and the Martin House Research Centre which is a partnership between the University of York and Martin House Hospice Care for Children and Young People. The views expressed are those of the authors and not necessarily those of the Wellcome Trust, the University of York or Martin House Hospice.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
