Abstract
Newborn infants’ circadian systems are not completely developed and rely on external temporal cues for synchronizing their biological rhythms to the environment. In neonatal intensive care units (NICUs), lighting is usually continuous or irregular and infants are exposed to artificial light at night, which can have negative health consequences. Therefore, the aim of this study was to evaluate the impact of the use of individual light protection equipment at night on the development and growth of preterm neonates. Infants born at less than 37 gestational weeks who no longer needed constant intensive care were admitted into a newborn nursery and randomized to either use eye masks at night (intervention, n = 21) or not (control, n = 20). Infants who used eye protection at night were discharged earlier than those in the control group (8 [5] vs 12 [3.75] days; p < 0.05). A greater variation within the day in heart rate was observed in the intervention group, with lower values of beats per minute at 1400 and 2000 h. There was no significant difference in weight gain between groups. In view of our results and of previous findings present in the literature, we suggest that combining a darkened environment at night with individual light protection devices creates better conditions for the development of preterm infants in the NICU. In addition, eye masks are an affordable and simple-to-use tool that can reduce hospitalization costs by decreasing the number of days spent in the NICU.
Preterm birth is defined by the World Health Organization (WHO) as delivery before the completion of 37 gestational weeks (Vogel et al., 2018). Worldwide, approximately 15 million infants are born prematurely every year, with varying rates among countries that range from 5% to 18% of total births (World Health Organization et al., 2012). Premature infants are at higher risk of neonatal respiratory and neurological conditions, sepsis, visual and hearing problems, and poorer neurodevelopmental outcomes (Vogel et al., 2018). Given that specialized care is critical during the first weeks or months of a preterm infant’s life, when proper health infrastructure is available, the newborns are admitted to a neonatal intensive care unit (NICU). In this environment, infants are often exposed to constant lighting conditions and artificial light at night (ALAN), in contrast to the uterine environment in which their development takes place (Hazelhoff et al., 2021).
During the gestational period, maternal circadian signals, such as temperature variations, activity-rest cycle, feeding schedule, and melatonin secretion are responsible for entraining fetal rhythms (Serón-Ferré et al., 2012). After delivery, in the absence of these maternal signals and with an immature circadian system, the newborn uses social interaction patterns, feeding schedules, and, especially, environmental light variations as timing cues (Thomas et al., 2014). The light-dark cycle is the main external cue responsible for the entrainment of circadian rhythms (Aschoff, 1965). In adults, lighting variations are perceived by intrinsically photosensitive ganglion cells in the retina, which send this information to the suprachiasmatic nucleus (SCN) of the hypothalamus through the retinohypothalamic tract (RHT). The SCN acts as a master clock, synchronizing peripheral circadian oscillators present in the whole organism (Welsh et al., 2010). Although it is currently unknown in which developmental stage the RHT becomes functional in humans, studies in non-human primates indicate that the SCN is responsive to light since the equivalent to 24 weeks of gestation in humans, suggesting that lighting can also influence the rhythms of premature infants (Hao and Rivkees, 1999).
When timed correctly, exposure to bright light can synchronize the circadian clock to the external environment, regulating behavior and physiology (LeGates et al., 2014). However, studies in adults indicate that exposure to even very low levels of ALAN (<10 lux) can interfere with melatonin biosynthesis, alter sleep structure, and aggravate poor sleep quality (C. H. Cho et al., 2016; Phillips et al., 2019; Stebelova et al., 2020; Tähkämö et al., 2019). Exposure to ALAN has been associated in adults with several negative health outcomes, such as psychiatric and metabolic disorders and higher cancer risk (Bechtold et al., 2010; Dauchy et al., 2015; Haus and Smolensky, 2013). Unfortunately, creating a completely dark environment during the night is not always possible, as in the case of intensive care facilities where some light is necessary at all times for visibility reasons. According to Brazilian regulations, light levels for nighttime observation should be around 20 lux (Associação Brasileira de Normas Técnicas, 2013).
Several studies documented improvement in clinical outcomes of preterm infants exposed to light-dark cycles in NICUs, when compared with the typical constant light or constant near-darkness conditions (Hazelhoff et al., 2021; Morag and Ohlsson, 2016). The cycled light condition, characterized by light-dark cycles with decreased light exposure at night, has been shown to accelerate weight gain and the beginning of oral feeding (Miller et al., 1995; Vásquez-Ruiz et al., 2014), shorten the stay in the NICU, and improve peripheral oxygen saturation and heart rate stability throughout the days (Vásquez-Ruiz et al., 2014), decrease activity, respiratory rate, and heart rate during the night (Blackburn and Patteson, 1991; Shiroiwa et al., 1986), and accelerate the emergence of circadian activity-rest rhythms (Rivkees et al., 2004). Cycled light has also been shown to improve weight gain when compared with continuous near darkness (5-10 lux) (Brandon et al., 2002) and to reduce fussing and crying in very preterm infants when compared with dim lighting (97.6 ± 45.3 lux during the day and 20.8 ± 20.7 lux at night) (Guyer et al., 2012). Other studies, however, have found no difference between cycled and continuous dim light conditions (Boo et al., 2002; Mirmiran, Baldwin, et al., 2003).
In these studies, the light-dark pattern is typically achieved through changes in the unit’s lighting system and eventually with the use of individual helmets (Vásquez-Ruiz et al., 2014), curtains (Guyer et al., 2012), or incubator coverings (Brandon et al., 2002; Guyer et al., 2012). Although designed to resemble day-night alterations, the cycled condition usually consists of dim light exposure (~25 lux) during the “lights-off” phase (Guyer et al., 2012; Vásquez-Ruiz et al., 2014). The use of individual light protection equipment, such as eye masks, offers a potential solution to this problem (Hu et al., 2010).
In this context, the aim of the present randomized interventional study was to investigate the impact of the use of individual light protection equipment at night on the development and growth of preterm infants.
Materials and methods
Participants
The sample consisted of 41 infants born under 37 gestational weeks (average gestational age of 31.99 ± 2.09 weeks) that no longer required intensive care and were admitted at a nursery adjacent to the NICU dedicated to clinical stabilization before hospital discharge. The eligibility criteria of the study were weight greater than 1500 g and stability of vital signs for at least 48 h. Infants with congenital malformation relevant to the measurement of the desired outcomes (e.g., pre-chiasmatic blindness; brain, cardiac, and digestive tract malformations; and bronchopulmonary dysplasia) were not included. Moreover, patients did not receive phototherapy during the study, but some of them did receive it while in the NICU. This information was included in the analysis.
The randomization was performed as follows: Prior to the start of the study, the research team determined that infants in the control group would be identified by numbers 1-20 and those in the intervention group, by numbers 21-41. The numbers were then randomized by a computer to generate the order in which participants would be included (e.g., the first infant included in the study would receive the number 37 and therefore be part of the intervention group). Envelopes containing information of which group the infant should be assigned to were labeled in the randomized order and sealed. Every time a participant was included in the study, the next envelope in line was opened by the NICU staff, and the infant was allocated accordingly. Infants in the control group were exposed to the typical lighting conditions of the nursery during the day and night, while those in the intervention group wore eye masks during the night (see Figure 1). Twins were included, and each infant was randomly assigned to the intervention (n = 6) or control (n = 4) group, meaning that siblings could either be assigned to the same group or not.
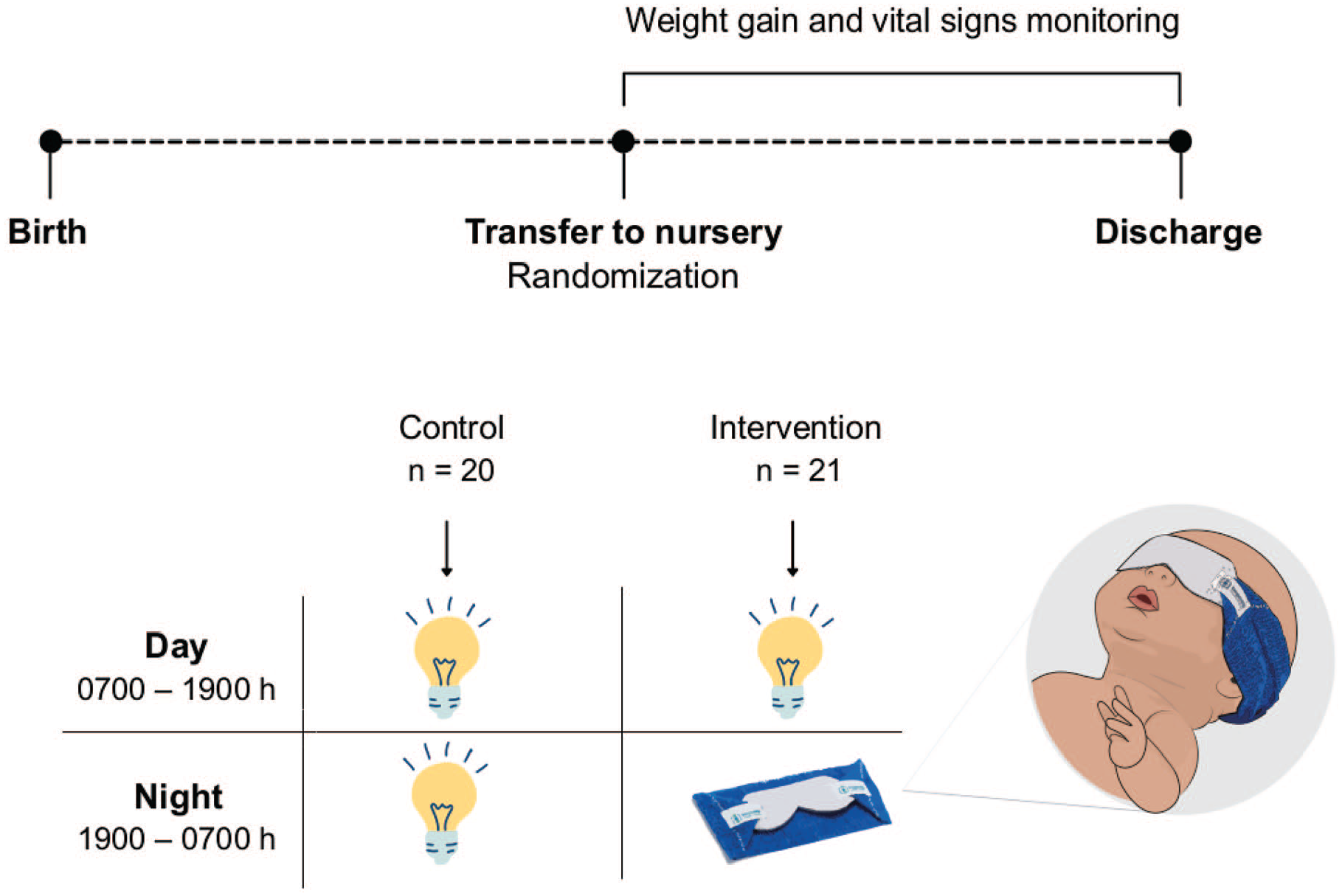
Visual representation of the study design. Time elapsed between birth and transfer to nursery, and from transfer to nursery until discharge varied among participants.
The study was conducted following the Declaration of Helsinki (World Medical Association, 2013) and was approved by the Research Ethics Committees of Hospital de Clínicas de Porto Alegre and Hospital Nossa Senhora de Pompéia (CAAE 65311417.9.1001.5327). Verbal informed consent was provided by a legal guardian.
Eye Masks
Infants in the intervention group were blindfolded from 1900 to 0700 h with an eye protector typically used for phototherapy (Baby Block, Impacto Medical, Brazil). In the case a mother came to breastfeed after 1900 h, the eye mask was removed during breastfeeding to allow eye contact between mother and child.
Room Characterization
The study took place in a five-bed newborn nursery adjacent to the NICU. The room has three windows facing other indoor facilities (one facing a corridor and two facing the nursing station) and no external windows (see Figure 2). Light was provided by two ceiling luminaires with dimmable halogen light bulbs (2700 K) that were mostly on and four fluorescent lamp fixtures (4000 K) that were only switched on if necessary, such as in the case of a medical emergency. Decisions regarding when lights were switched on, dimmed, or switched off were made by the nurse or nurse technician in charge of the room, according to the need for clinical assistance.
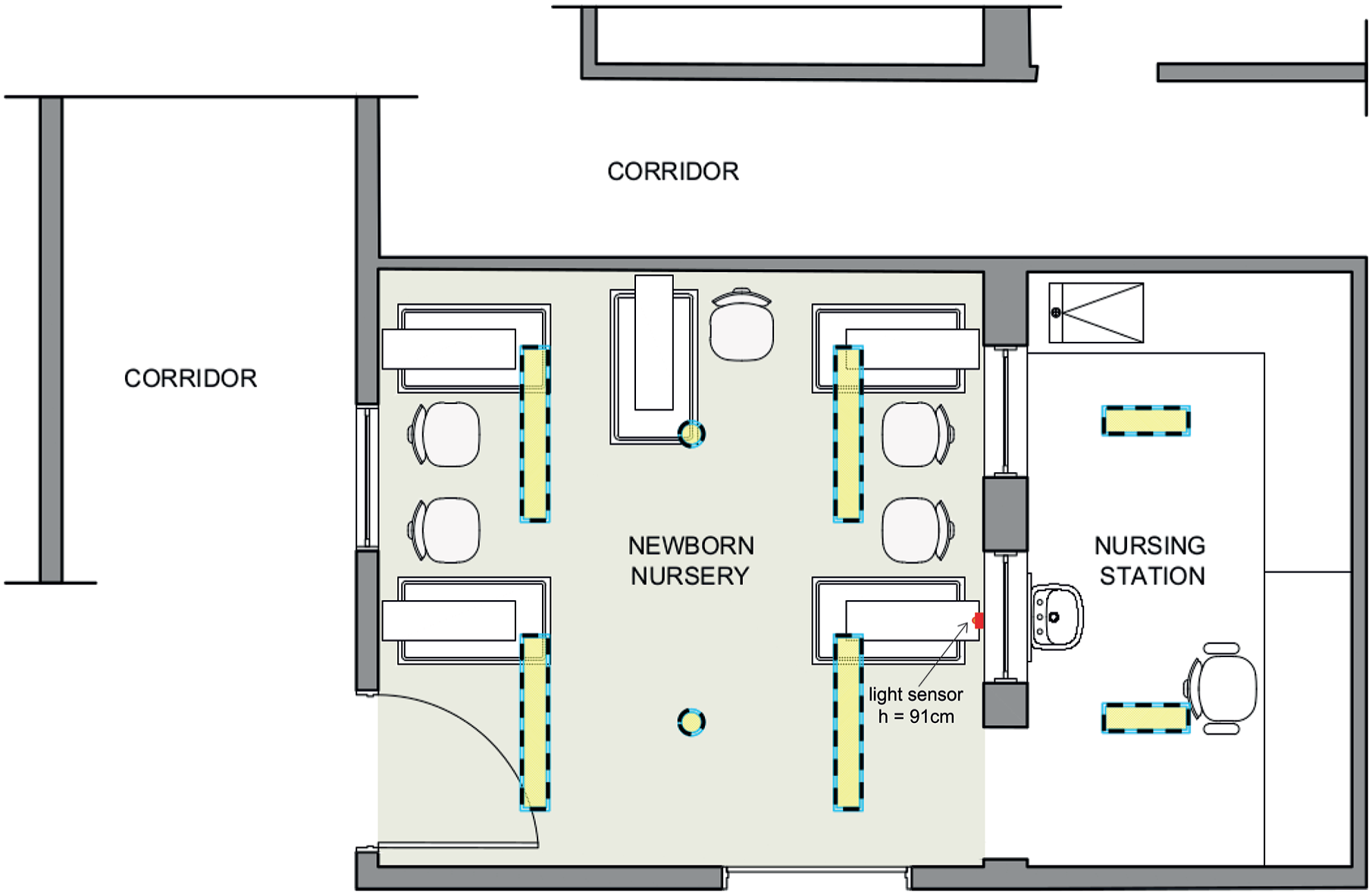
Architectural plan of the five-bed newborn nursery where the study took place. The location of the luminaires is shown in dashed lines.
Illuminance, temperature, and humidity were measured every 10 min for 15 consecutive days by a sensor developed by the Biomedical Engineering Department of Hospital de Clínicas de Porto Alegre. The sensor was attached to a wall near the height of the head of an infant’s bed, at 92 cm from the ground, facing the opposite wall (see Figure 2). The average temperature in the room was 24.9 ± 0.78 °C, and the average humidity was 62.73% ± 6.28%. The illuminance detected at the point where the sensor was located ranged from 0 to 47 lux (average illuminance ranged between 0.6 and 12.5 lux during the day and 0.4 and 4.5 lux during the night) (see Figure 3).
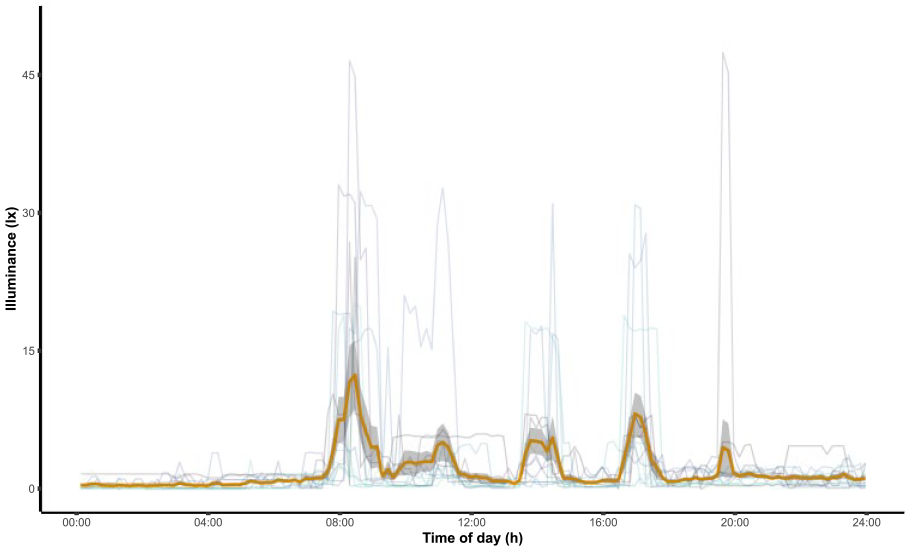
Daily profile of illuminance. Each line represents illuminance measurements from 1 day. Means of illuminance (in lux) are presented according to clock time in a thicker orange line, and standard error is presented in gray.
Measures
Days Until Discharge
The primary outcome was the number of days spent in the room until discharge. Discharge criteria included stable weight gain (15-30 g per day), weight greater than 1900 g, the removal of the orogastric tube and complete oral feeding established for at least 48 h, and the absence of other health conditions that required constant supervision.
Weight Gain
Infants were weighed daily by a nurse or nurse technician. Body weight gain per day was calculated by dividing the total weight gain by the number of days until discharge. Because weight can vary according to postmenstrual age (PMA) and sex of the infant, weight Z-scores were also calculated using PediTools (Chou et al., 2020).
Vital Signs
Heart and respiratory rate, blood-oxygen saturation, and body temperature were measured every 6 h (0200, 0800, 1400, and 2000 h) by the nurses using patient monitors (DX 2023, Dixtal Biomédica, Brazil, and Efficia CM120, Philips, Brazil).
Statistical Analyses
Measures of age, sex, weight, head circumference, and body length at birth, days until oral feeding, and days until discharge were compared between groups using chi-square, Student’s t, or Mann-Whitney U tests when appropriate. The variations in heart rate over time were analyzed by generalized estimated equations (GEE), which is one of the methods to analyze repeated measurements using a clustering variable to account for the covariance between data points (Vagenas and Totsika, 2018). Differences between groups at each time point were assessed by Bonferroni’s test for pairwise comparisons, and statistical significance for multiple-factor interactions was set at p < 0.1 as we prioritized the detailing of the interactions. Data were presented as mean ± SD, median (interquartile range [IQR]), or percentage, and statistical significance was set at p < 0.05 for the remaining tests. Analyses were performed on SPSS Statistics Subscription (IBM Corp., Armonk, NY, USA) and RStudio version 1.3.1056 (RStudio, PBC, Boston, MA, USA). Graphs were made on SPSS, RStudio, and Microsoft Excel version 16.47 (Microsoft Corp., Redmond, WA, USA).
Results
A total of 41 infants were included in the study, 21 of which were randomized into the intervention group. No loss or exclusion occurred after randomization. Groups were similar in age and weight at birth, at the time of enrollment, and at discharge. A summary of sample characteristics can be found in Table 1 and details of study outcomes can be found in Table 2.
Characteristics of the study population.
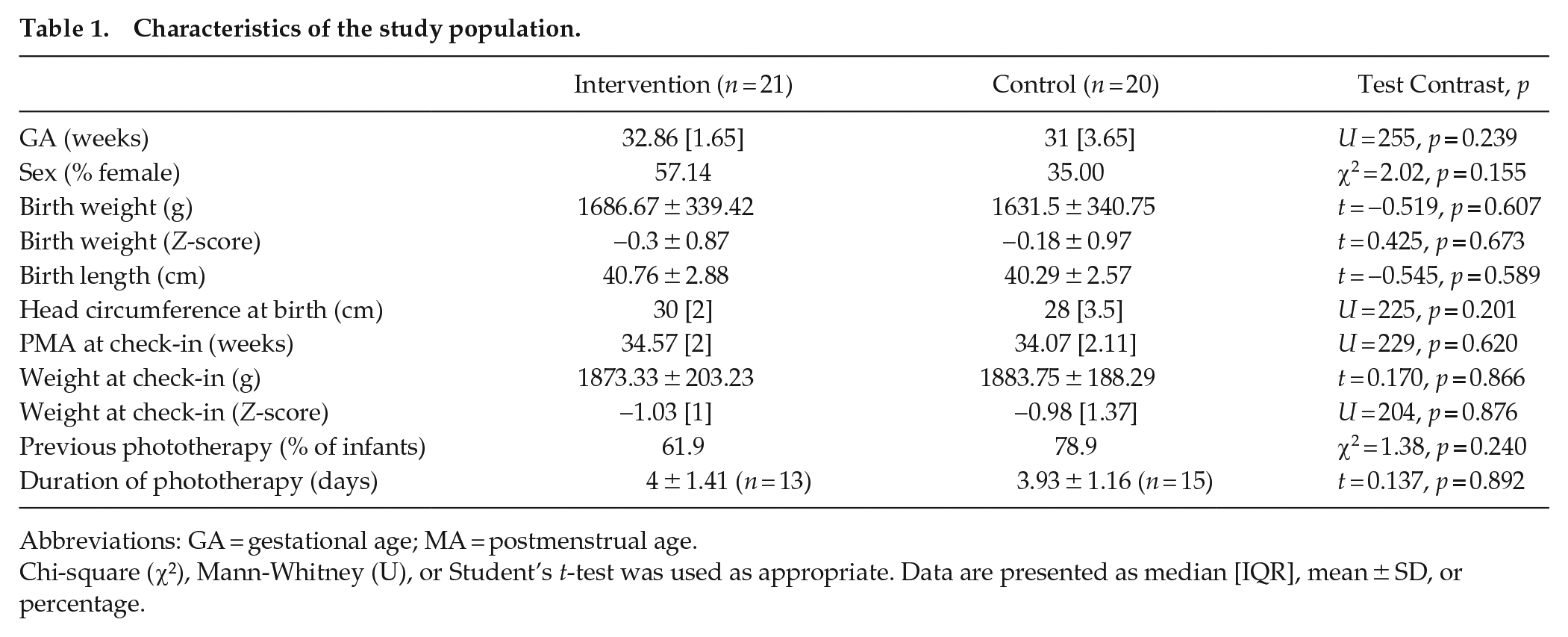
Abbreviations: GA = gestational age; MA = postmenstrual age.
Chi-square (χ²), Mann-Whitney (U), or Student’s t-test was used as appropriate. Data are presented as median [IQR], mean ± SD, or percentage.
Study outcomes.
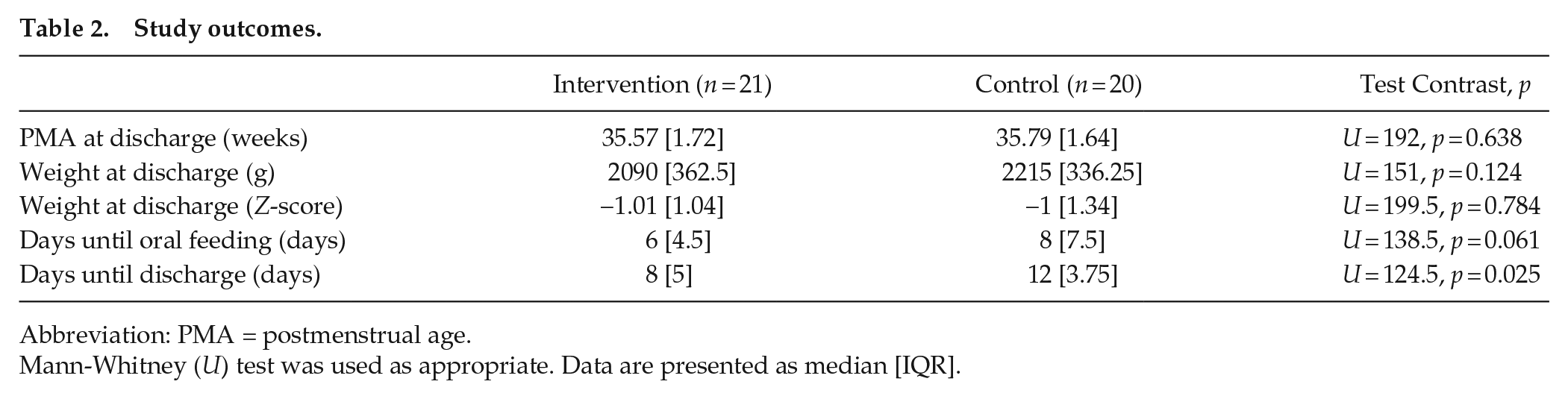
Abbreviation: PMA = postmenstrual age.
Mann-Whitney (U) test was used as appropriate. Data are presented as median [IQR].
Days Until Discharge
Infants stayed a median of 11 [7.5-13] days in the nursery. Preterm infants randomized to the intervention group were discharged earlier than infants in the control group (control, 12 [10.25-14]; intervention, 8 [7-12]; Mann-Whitney U test = 124.5, p = 0.025) (Figure 4). The calculated effect size was intermediate to large (η2 = 0.121). Two infants stayed in the room much longer than the others, one from the control group (29 days) and one from the intervention group (35 days). If removed from the sample, the significant difference between groups was maintained; therefore, we decided to include them in the remaining analyses.
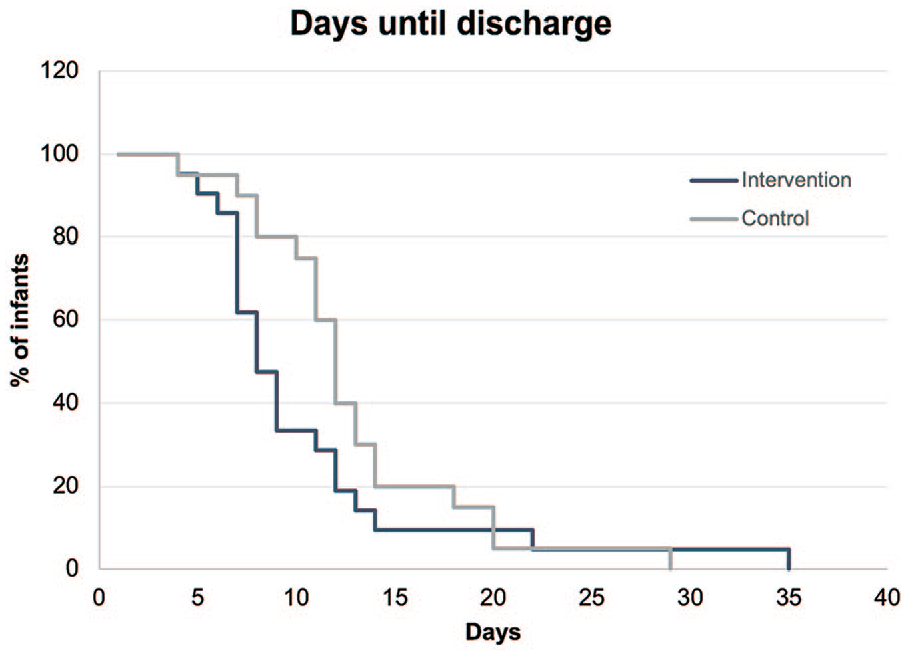
Percentage of infants in the nursery room at a given time point. Infants in the intervention group (black line) were discharged earlier than those in the control group (gray line).
Weight Gain
No significant difference was found between groups regarding average weight gain per day (control, 31.35 ± 8.27; intervention, 27.26 ± 7.7; t = 1.641, p = 0.11) (Figure 5).
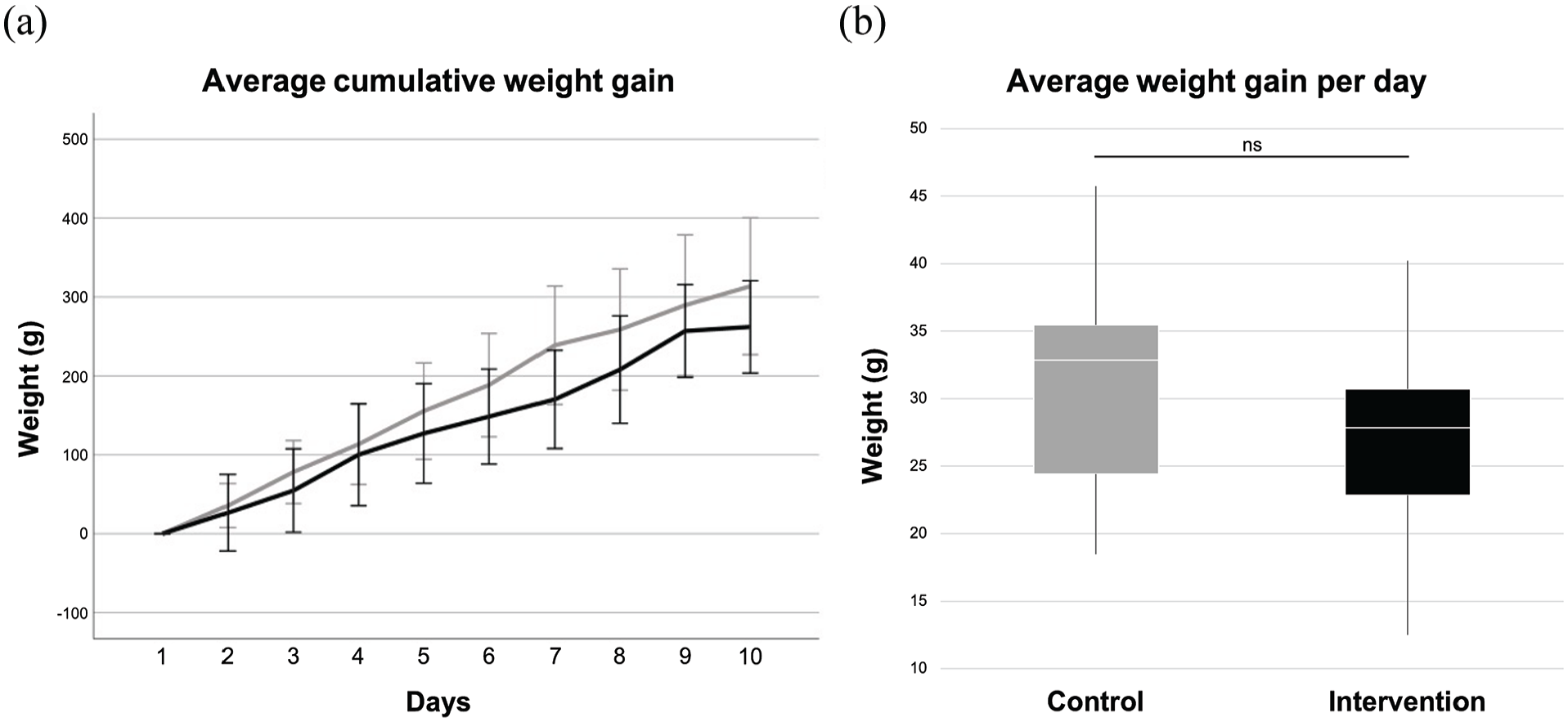
(a) Cumulative weight gain of the intervention group (black line) and the control group (gray line) in the first 10 days (error bars represent one standard deviation). (b) Boxplot of the average weight gain per day during the whole stay in the nursery room. ns, non-significant.
Of the 41 infants, 34 entered the study before transitioning to oral feeding (control, n = 18; intervention, n = 16). No significant difference was found between the groups regarding number of days until oral feeding (control, 8 [7.5]; intervention, 6 [4.5]; Mann-Whitney U test = 138.5, p = 0.06).
Vital Signs
Means of heart rate, respiratory rate, SpO2, and body temperature for each group are presented in Figure 6. Statistical differences were found between groups for average heart rate at 1400 and 2000 h (t = 2.4, p = 0.021 and t = 2.4, p = 0.02, respectively). Mean and standard deviation values can be found in Supplementary Table S1, and the distribution of data and their means through the days can be found in Supplementary Figures S1 and S2.
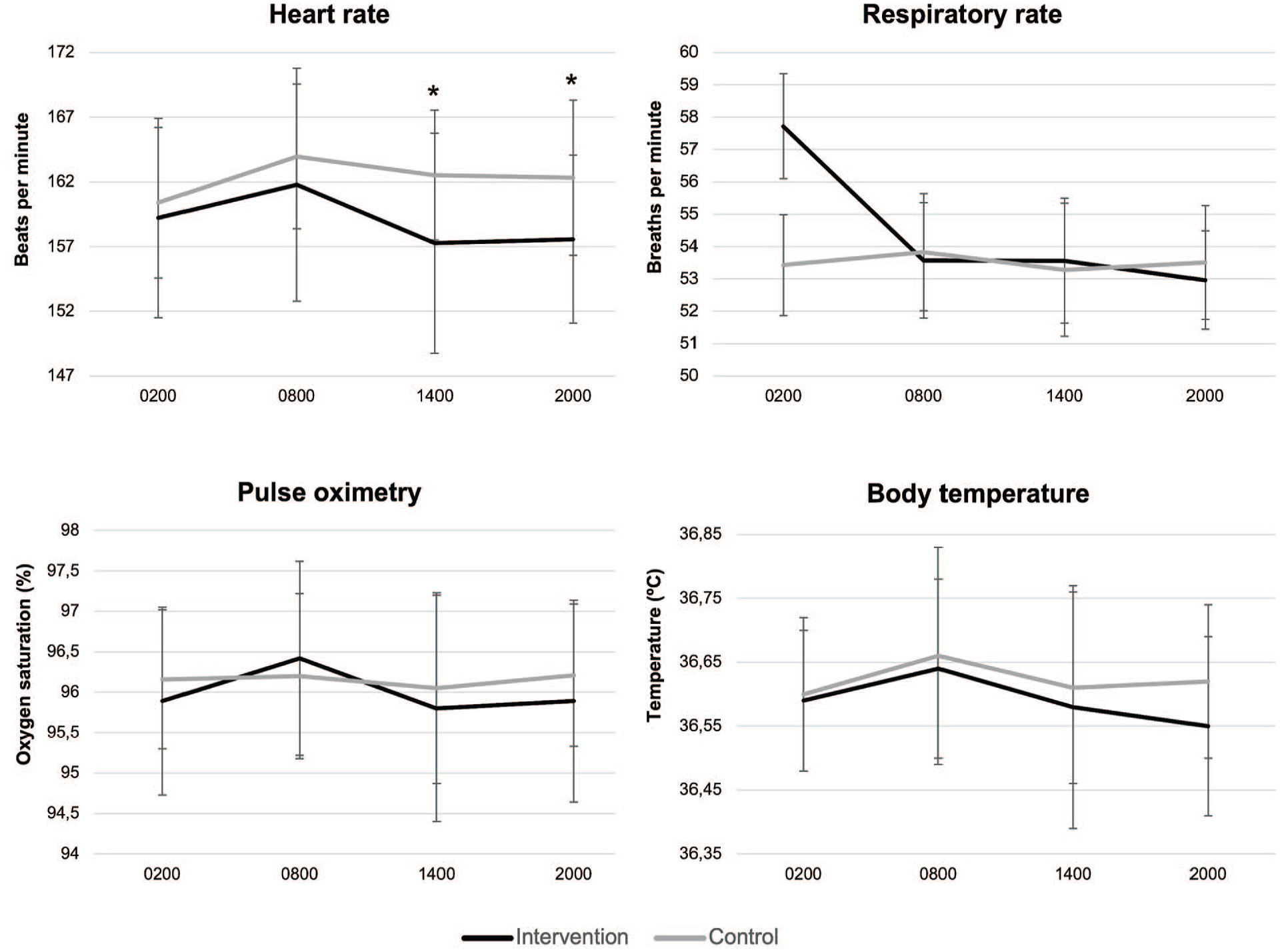
Group means and standard deviations of heart rate, respiratory rate, pulse oximetry, and body temperature at four time points.
The GEE performed for heart rate showed that the interactions time × day and group × time × day were significant (p = 0.09 and 0.08, respectively) and residuals were well distributed (Supplementary Figure S3). This indicates that the heart rate difference between groups varies across days and time points. We then compared the estimated marginal means of heart rates at each time point for the first 10 days, when the majority of infants were still in the nursery, and found that the heart rate was significantly higher in the control group at 1400 h from day 4 onward and at 2000 h from day 1 to day 8 (Figure 7). When we performed the same analysis intra-group, we found significant differences in heart rate from day 7 to day 10 in both groups (Supplementary Table S2).
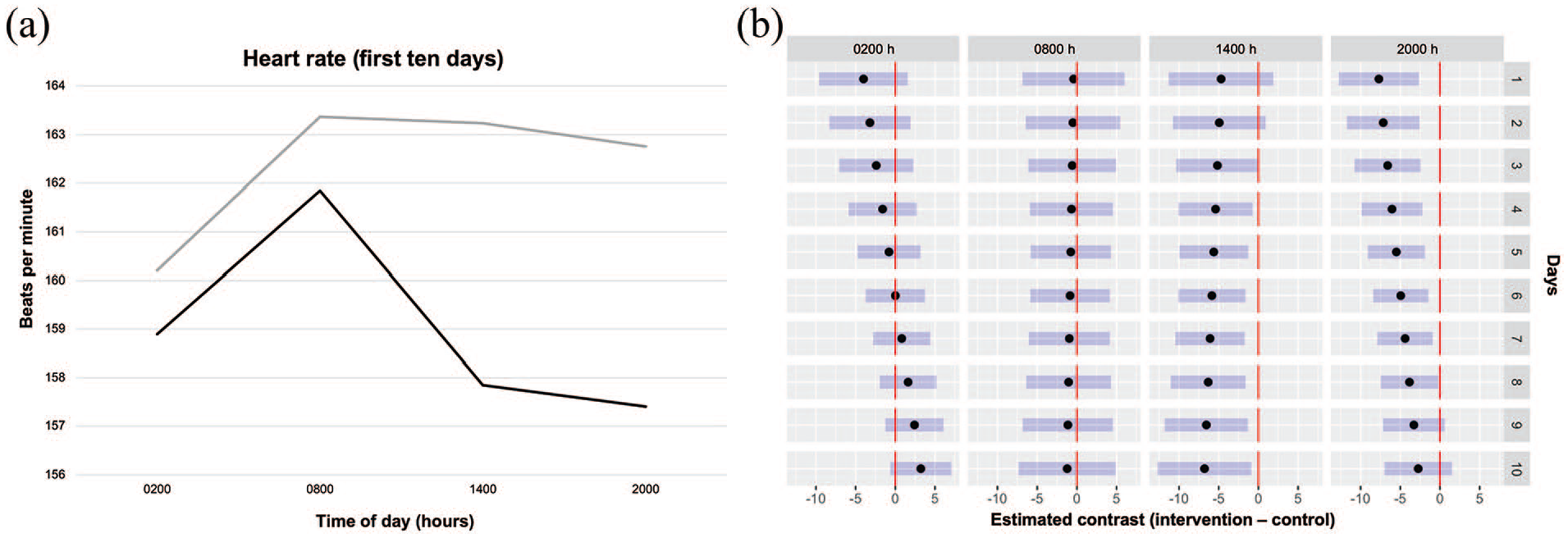
(a) Average heart rate of the intervention group (black line) and the control group (gray line) at different time points during the first 10 days. (b) Multiple comparisons of the estimated marginal means at different time points across days. The black dots represent the difference between the estimated marginal means of the two groups, the blue bars represent 95% confidence intervals for the estimated marginal means, and the red lines represent equal values between groups, allowing for comparison. When a blue bar is not crossing or touching the red line, there is a significant difference between groups. Negative values indicate that the intervention group had lower estimated marginal means than the control group, and positive values indicate that the intervention group had higher estimated marginal means.
Discussion
In this study, preterm infants wearing individual light protection equipment at night were discharged earlier from the hospital than infants exposed to very low levels of light. The intervention group also showed greater heart rate variation throughout the day for several days. Furthermore, we observed no significant differences in weight gain and median of days until the beginning of oral feeding between groups.
Days Until Discharge
Infants wearing eye masks at night were discharged earlier from the hospital than infants in the control group, who were exposed to very low levels of light at night. Light is the main environmental clue responsible for entraining human circadian rhythms (Duffy and Wright, 2005). The timing and duration of the exposure, as well as the intensity and wavelength of light that individuals are exposed to, have physiological and behavioral impacts and can be related to either positive or negative health outcomes (Souman et al., 2018; Vethe et al., 2021). Over time, these parameters of light and dark exposure create a light history that influences human circadian responses to artificial and natural light (Foster et al., 2020). In our study, both groups seemed to be exposed to near darkness, with little variation in light assessment. Here we were able to collect lighting information in the nursery by using a sensor attached to the wall next to one of the infant’s beds. It is important to note that the sensor provides information about lighting levels in that point of the room, not an average of the environment or a precise measurement of light reaching the infants’ eyes. Although the difference in light exposure between the groups was small, the implementation of eye masks that blocked the very low levels of ALAN in the room led to a significant difference in recovery.
Earlier findings pointed to light intensities of >100 lux at night having deleterious health impacts (Y. Cho et al., 2015), but recent research suggests that exposure to less than 10 lux can reduce melatonin biosynthesis, and even 1 lux can aggravate sleep quality in adults (C. H. Cho et al., 2016; Phillips et al., 2019; Stebelova et al., 2020; Tähkämö et al., 2019). Given that melanopsin is present in the eye tissue since early weeks of gestation and that the RHT and the SCN are formed during the second trimester, it is likely that even extreme preterm infants can sense and respond to light variations (Hazelhoff et al., 2021), leading to the results observed in this study.
Heart Rate Variation
Infants in the control group showed a significantly higher heart rate at 1400 h from day 4 onward and at 2000 h from day 1 to day 8 when compared with the intervention group. Inside each group, significant differences in heart rate according to time were observed starting at day 7.
Studies show that, during the last gestational trimester, a day-night variation of fetal heart rate synchronized to maternal rhythms can be observed (Mirmiran, Maas, et al., 2003). However, research on the postnatal development of circadian heart rate rhythms in preterm newborns is still lacking. Begum et al. (2006) measured physiological variables in preterm and term neonates exposed to a light-dark cycle and reported that the circadian amplitude of pulse rate increased with gestational age. Blackburn and Patteson (1991) observed that preterm infants exposed to cycled lighting had lower heart rates in the period when the light was decreased compared with when lights were on. A year later, D’Souza et al. (1992) published a study with nine extremely preterm infants, in which four of them developed circadian rhythms after being reallocated to a nursery where they were exposed to light-dark cycles. Vásquez-Ruiz et al. reported that neonates exposed to light-dark cycles presented stable heart rate means across days, while those exposed to constant light exhibited an irregular pattern (Vásquez-Ruiz et al., 2014).
Prenatal factors such as intrauterine growth, gestational age, and fetal and maternal illnesses can influence the development of an infant’s circadian rhythms (Mirmiran and Ariagno, 2000), and postnatal environmental conditions such as light exposure, feeding schedules, and nursing care can entrain the rhythms (Bueno and Menna-Barreto, 2016). These factors may also interfere with heart rate rhythmicity development, making it difficult to establish what a “normal” development curve would look like or to determine what to expect at each postnatal week.
In our study, because there was no need for 24-h monitoring of the infants’ vital signs, these were only recorded at four time points. It would be interesting to increase the number of time points in order to achieve a more precise characterization of rhythmic patterns.
Weight Gain and Days Until Oral Feeding
In this study, although not implementing a light-dark cycle, we also observed a decrease in time until discharge in infants wearing light protection devices at night. However, no significant difference was found between groups regarding weight gain and days until the beginning of oral feeding, possibly due to the small variation between day and night light conditions. Daytime bright light exposure has been previously shown to increase nocturnal melatonin levels and maintain the circadian phase in young adults when compared with dim light (Takasu et al., 2006). In our study, since infants were exposed to a near-dark condition during the day, the use of eye masks at night might have had a smaller effect than it would if participants were exposed to daytime bright light.
Factors that can influence the postnatal growth of preterm infants include the degree of prematurity, birth weight (appropriate or small for gestational age), nutrition type (breast milk or formula), food intolerance, and morbidities (Silveira and Procianoy, 2019). We found no statistically significant difference between the two groups regarding the degree of prematurity or birth weight, and infants presenting several morbidities such as brain, cardiac, and digestive tract malformations were not included.
It is also important to note that, given that the study took place in a nursery where infants with stable vital signs are admitted in order to gain weight, transition to oral feeding, and get ready for discharge, the average number of days in the study was lower than if they had been included since birth. The setting, on the other hand, is one of the study’s strengths. Since the nursery is separated by walls and a corridor from the intensive ward, it provides a more controlled environment for investigating the effect of light variations, with less noise and circulation.
Limitations
A methodological limitation of this study is that, since the intervention consists of wearing a mask, the nurses, nurse technicians, and neonatologists who also collect daily data are aware of the intervention allocation, and therefore a double-blind or single-blind study design was not possible. Nonetheless, statistical analyses were blinded.
For the sample size estimation, we used as a reference a similar study carried out by Vásquez-Ruiz et al. (2014). This study has a slightly different methodology, in which infants were included since birth and therefore had a longer stay in the NICU. Considering the p-value of 0.014 found in the reference study for an average difference of 16.74 days of hospitalization time between groups in a total sample of 38 individuals, we calculated the necessary sample size for a significance of 5% and a power of 90%. The calculation resulted in a required sample of 67 individuals. At the end of data collection, our sample consisted of 41 infants. Even though the sample size was smaller than originally estimated, we calculated the effect size of our main outcome (days until discharge), which was intermediate to large (Lenhard and Lenhard, 2016).
Cost Benefit
According to the nursing staff, the masks were easy to wear and did not seem to increase restlessness or bother the infants. They were already available in the hospital, since typically used for phototherapy, and were extremely affordable, costing R$11.68 per participant at the time of this study (R$ = Brazilian real). The costs of a daily stay in the NICU through Brazil’s Unified Public Health System (SUS), on the other hand, ranged between R$139 and R$700 in June of 2022 (Ministério da Saúde Brasil, 2022), and it is not uncommon for premature infants, especially very premature ones, to stay over a month in the NICU (AlJohani et al., 2020; Manktelow et al., 2010; Seaton et al., 2019). The results presented here, together with findings of studies in adults (Hu et al., 2010), suggest that this low-cost intervention has the potential to bring health benefits for patients in an intensive care unit.
Conclusion and perspectives
Lighting needs in the NICU and other intensive care facilities are different among occupants. A cycled pattern consisting of bright light during the day and dim light at night is potentially beneficial for both infants and staff (Rea and Figueiro, 2016), and based on our results, we suggest that coupling a darkened environment at night with individual light protection devices can create the best conditions for the development of preterm infants. Our next step will be the implementation of a dynamic lighting system in the nursery, with color temperature variations that aim to mimic the natural daily ones, in order to investigate the effect of the use of light protection equipment at night coupled with bright light exposure during the day.
Supplemental Material
sj-docx-1-jbr-10.1177_07487304231201752 – Supplemental material for Use of Light Protection Equipment at Night Reduces Time Until Discharge From the Neonatal Intensive Care Unit: A Randomized Interventional Study
Supplemental material, sj-docx-1-jbr-10.1177_07487304231201752 for Use of Light Protection Equipment at Night Reduces Time Until Discharge From the Neonatal Intensive Care Unit: A Randomized Interventional Study by Ana Carolina Odebrecht Vergne de Abreu, Melissa Alves Braga de Oliveira, Tamila Alquati, André Comiran Tonon, Mariana de Novaes Reis, Augusto Camargo Rossi, Fernanda Sbaraini Bonatto and Maria Paz Hidalgo in Journal of Biological Rhythms
Footnotes
Acknowledgements
We thank the staff of Hospital Pompéia’s NICU for their support in data collection, Rogério Boff Borges for statistical consulting, and Luísa Klaus Pilz and Clarissa Gutiérrez for comments on the manuscript. This study was funded by Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES) Finance Code 001 and FIPE-HCPA 17-0208. We also thank CAPES (ACT and FSB) and CNPq (ACOVA, MABO, and MPH) for fellowships.
Conflict of interest statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Note
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
