Abstract
Artificial light at night disturbs the daily rhythms of many organisms. To what extent this disturbance depends on the intensity and spectral composition of light remain obscure. Here, we measured daily activity patterns of captive blue tits (
Light pollution is defined as the alteration of natural light levels in the outdoor environment owing to artificial light sources (Cinzano et al., 2000). Light pollution is not only a problem for humans—as the night sky brightness damages our perception of the starry sky (McNally 1994) and can have severe impacts on human health (Cho et al., 2015)—but artificial light at night can also have a strong impact on wildlife (Rich and Longcore 2006; Hölker et al., 2010). In the Western world, there are few places left where nights are truly dark (Cinzano et al., 2001); the natural light-dark cycle under which animals evolved is disturbed by anthropogenic light at night.
Birds align their activities to the appropriate time of the day and year through the stimulation of photoreceptors by daylight, which synchronizes their internal circadian and circannual clocks (Dawson et al., 2001). Recent studies on bird populations in the wild have shown that artificial light at night can affect many aspects of a bird’s life: the foraging activity of blackbirds (
Research interest in the biological impact of artificial light at night has grown in recent years (Gaston et al., 2015), and options to reduce the effects of night-time light pollution on ecosystems are being investigated. Simply illuminating the environment less would obviously reduce these effects greatly and save energy costs, but such a change might not always be feasible. Adapting the light spectrum is another option (Gaston et al., 2012). The use of modern light-emitting diode (LED) lamps in outdoor lighting has economic advantages, but, more importantly, their color compositions can be custom designed, potentially mitigating the impact of light on flora and fauna through adaptation of the spectrum (Spoelstra et al., 2015). Ultimately, it should be possible to advise the use of lamps with certain characteristics (such as spectral composition and light intensity) for a specific setting, in such a way that the emitted light has sufficient quality to support human activities while having minimal effects on the biological processes of the organisms or communities nearby (Musters et al., 2009).
To eventually use scientific results to advise outdoor lighting, we set the light levels for the different treatments in our experiments to the same lux levels, as this is the measure of illumination relevant for human vision (lamps of different colors with the same intensity in lux are perceived by humans as equally intense). Lamps with different spectra will, however, differ in intensity for birds, as the spectral sensitivity of birds differs from that of humans. Birds are sensitive to a range of wavelengths to which humans are blind (Bennett and Théry 2007). Many birds are UV sensitive and most birds perceive colors through 4 single cone types (Bennett and Cuthill 1994; Osorio and Vorobyev 2008). Also, they have extra-ocular light perception through photoreceptors in the pineal gland and brain (Cassone 2014). Hart et al. (2000) measured the spectral absorption characteristics of the photoreceptors in the eyes of the blue tit and the blackbird, which are similar to those described in other passerines. The authors found that there are small differences in the properties of their cones, and this may reflect differences in the visual ecology of these 2 species (Hart et al., 2000). Using lamps with different spectra would therefore have different effects on birds simply because some colors are perceived as less intense.
It is not yet clear whether lights with different spectral compositions affect the behavior and fitness of birds differentially; the results so far have been inconsistent. We may expect different spectra to have different effects on birds because they perceive various intensities differently, even at the same lux levels. Furthermore, there may be differences in how these different spectral lights of the same intensity affect bird behavior: little is known about these effects. In general, compared with shorter wavelengths, long wavelengths are known to penetrate the skull more easily, and are more effective at inducing a seasonal photoperiodic response (Hartwig and van Veen, 1979), stimulating gonadal development, and promoting body fattening (Malik et al., 2002). Yadav et al. (2015) showed that blackheaded buntings (
In a previous study, we have shown that the effects of nocturnal light on daily rhythms in birds are not only apparent once a light threshold is reached, but increase progressively with light intensity (de Jong et al., 2016). This means that it is important to study intensity-dependent effects of artificial light. The effects of different colored light at night on daily activity patterns are currently unknown, as is the interaction between light intensity and color. Therefore, in the current study, we focused on both the effects of light color and light intensity on daily rhythms in a common songbird, the blue tit. We made use of light colors that are suitable for application in outdoor lighting (green, red and white light; see Spoelstra et al., 2015). In a first experiment, we studied the activity patterns of blue tits under green, red, and white light at night as compared with a dark control. Based on the results of this experiment, we performed a second experiment, in which blue tits were exposed to different intensities of green and white light at night.
Methods
Birds
In the 2 experiments described here, we studied 28 and 35 male blue tits, respectively. The 35 birds used in the second experiment were largely the same birds as in the first experiment, with extra birds included in the group. All birds originated from Corsica and hatched in spring 2011. They were then moved to The Netherlands, where they were hand-reared and housed at The Netherlands Institute of Ecology (NIOO-KNAW), following the procedure described in detail by Reparaz et al. (2014). Before both experiments, birds were kept on 9-15 LD cycle for 6 weeks, and then the photoperiod increased up to a 14-10 LD cycle by 1 week before the start of the experiment. During the experiments, birds were housed in individual cages (90 cm × 50 cm × 40 cm). Each cage had 2 separate light sources for day- and night-time illumination, and external light was completely excluded using covers that were placed on the cage fronts. Cages were therefore tight to light coming from outside or from other cages. Cages were ventilated and temperature was maintained around 15 °C, with no structural variation between day- and night-time. Birds had access to food and water ad libitum. During both experiments, birds were kept on constant 14-10 LD. To reduce possible disturbances by outside noise and neighboring birds, white noise was played continuously at a low level. All experimental procedures were carried out under licenses NIOO 12.16 and NIOO 13.11 of the Animal Experimentation Committee of the Royal Netherlands Academy of Arts and Sciences.
Experimental Set-up
In both experiments, the individuals were assigned randomly to a treatment group and to one of 7 blocks of cages, each block containing all night light treatments. These blocks were divided over 2 rooms. Treatments were allocated to cages using a Latin Squares design. Birds from the same families were distributed evenly across treatment groups (sample size for each treatment group was 7). During daytime, 0600 to 2000 h, birds were exposed to full-spectrum daylight high-frequency fluorescent lights emitting ±1000 lux at perch level (Activa 172, Philips, Eindhoven, The Netherlands). During night-light treatment periods, night-light LED lamps were switched on at 1945 h and off at 0615 h, with a 15-min overlap with the daylight lamps.
In the first experiment (Experiment 1), we tested the effect of different light colors on blue tit activity. This experiment was performed in 2012, and consisted of 2 periods. During the first period (“control”, 14 November to 21 November), birds were not exposed to any light at night. In the second period (“experimental”, 21 November to 10 December), birds were exposed to either green LED light, red LED light, or white LED light (Philips, Eindhoven, The Netherlands) of approximately 5 lux at perch level, or no light (dark control). There were no differences between the light intensities (in lux) for the green, red, or white lamps (
In the second experiment (Experiment 2), we tested the effect of the interaction of light color and light intensity on the activity of blue tits. This experiment was done in 2014, as a follow-up of Experiment 1, and consisted of 5 periods alternating between control (dark nights) and experimental treatments. Based on the results of the first experiment, we chose to compare green and white light. During the first period (“control”, 14 April to 28 April), all birds were kept under dark nights. In the second period (“experimental”, 28 April to 22 May), the birds were divided over 5 treatment groups with different light intensities: dark, 0.15 lux, 0.5 lux, 1.5 lux and 5 lux of green or white light. During the third period (control, 22 May to 6 June), all birds had dark nights. In the fourth period (experimental, 6 June to 30 June), they were exposed to night light of the opposite color (i.e., first green then white, or first white then green), or to no light at night. Finally, in the fifth period (control, 30 June to 8 July), all birds were kept under dark nights again. Spectral composition of the lamps in Experiment 2 was equal to that of the green and white lamps of Experiment 1.
Activity Measurements
Daily activity patterns of each individual bird were measured continuously throughout both experiments. We used the same method as described in detail in de Jong et al. (2016). Briefly, each cage was equipped with one normal wooden perch and one wooden perch fitted with a microswitch that recorded perch-hopping activity. An on/off signal was registered every 0.1 s and software developed by T&M Automation (Leidschendam, The Netherlands) created files in which each 30-s interval was logged. We obtained one level of activity per 2 min. In the analysis, we excluded the first 5 days of each experimental period, because birds had to acclimatize to their new environment or to the change in treatment. ChronoShop 1.1 (written by KS) was used to calculate 4 activity descriptors for each bird in each 24-h cycle. The onset of activity is defined as the first moment the activity exceeds the average activity of the day (Spoelstra et al., 2014). To avoid ‘premature’ onsets, a running mean of 20-min length was fitted to the raw activity data. Activity onset is reported in min relative to the moment the daylight was switched on; ‘start of day’. The offset of activity, the last moment the activity was above the average activity value of the day, was calculated in the same way, and reported as min relative to the moment daylight was switched off; ‘end of day’. Total activity was defined as the total number of min during which the bird has been active in a 24-h cycle. Finally, nocturnal activity was calculated as the proportion of total activity that took place during the objective night (daylight off to daylight on). These 4 measures of daily activity were used as response variables in separate analyses.
Statistical Methods
All statistical analyses were done using R v3.1.2 (R Development Core Team 2014), with a significance level of α = 0.05. All analyses were based on averages of the response variable per individual per experimental period. For Experiment 1, we used linear-mixed-effects models (LMMs) with treatment as fixed effect and block nested within room as random intercepts, to account for possible effects of the location of the cage, for all 4 response variables. First, we tested for an effect of treatment group (a 4-level factor) in the first control period, with dark nights for all individuals. Then, we tested for an effect of treatment in the second period, with light treatment at night. For Experiment 2, we used LMMs with individual nested within block, which is nested within room as random intercepts, to account for multiple measures of the individuals, for all 4 response variables. First, we tested for an effect of period for all individuals over the 3 dark night periods (period 1, 3 and 5) to check for possible temporal changes. For the individuals that were kept under dark nights during one of the experimental periods, we compared those with the previous and next period (either period 1, 2 and 3 or period 3, 4 and 5) to check for carry-over effects of treatments or effects of neighboring birds during experimental periods. Then, we used light color (a 2-level factor), light intensity (a continuous variable), and their interaction as fixed effects to test for the effect of light treatment during the experimental periods 2 and 4. We used actual, measured light intensity as the explanatory variable in our analyses instead of treatment as factor, because light intensities of the green and white LED lights differed (for measurements of all lamps, see Suppl. Fig. S2). If the interaction between light color and light intensity was significant, we subset the data in green and white light and tested for the effect of intensity. Statistical significance was tested with F-tests using Kenward and Roger adjustment (Halekoh and Højsgaard 2014). Significant covariates were kept in the models.
Results
Blue tits clearly reacted to the light at night in both Experiments 1 and 2; the actograms for representative individuals from both experiments are given in Figure 1. Birds adjusted their activity patterns, with a strong response in the onset of morning activity. In contrast, during the periods with dark nights, activity patterns were normal, with a sharp on- and offset coinciding with the moments the daylight lamps were switched on and off, respectively. In control periods after experimental periods, patterns went quickly back to normal. We analyzed activity onset (relative to ‘start of day’), activity offset (relative to ‘end of day’), total activity, and nocturnal activity to quantify the behavioral response of birds to artificial light at night.
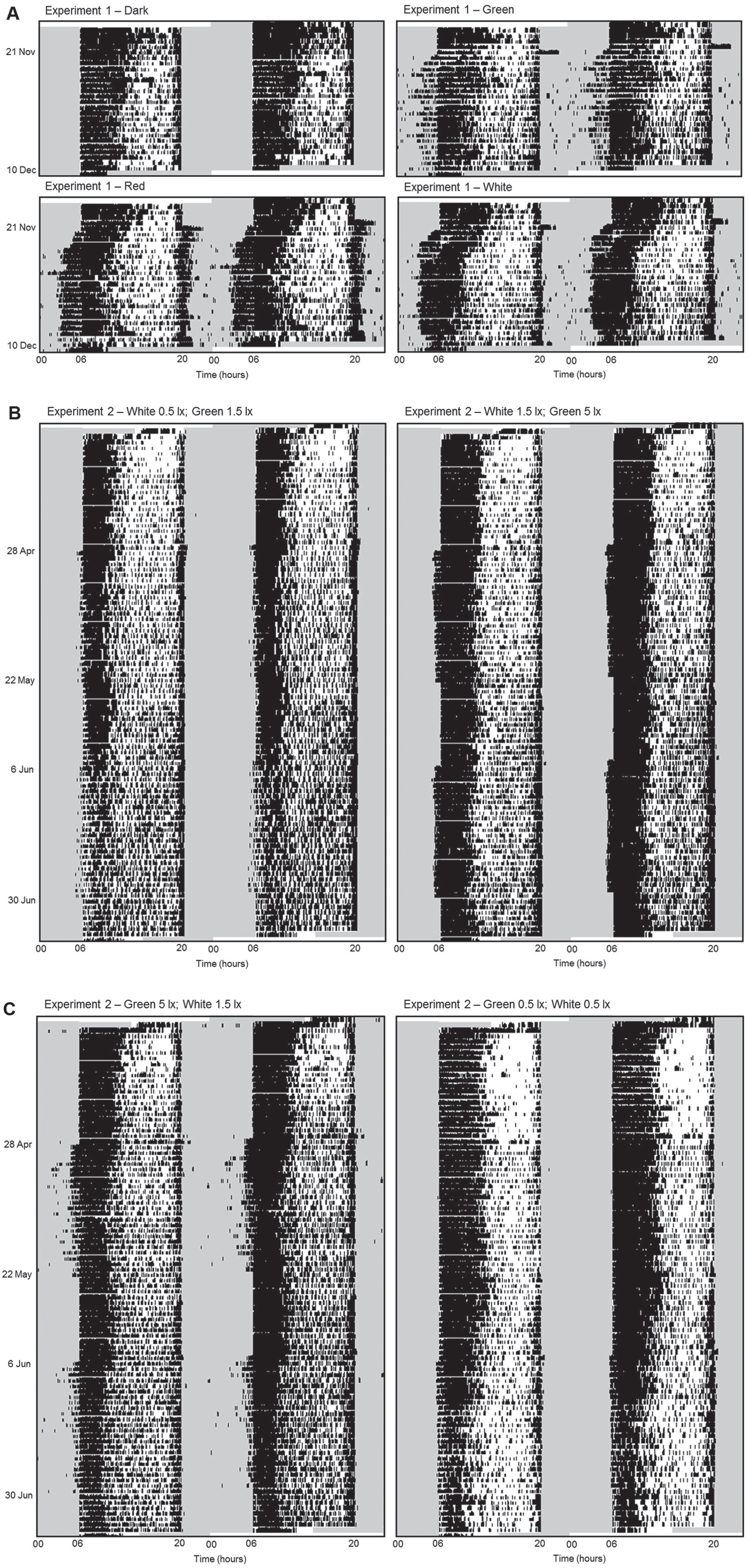
Double plotted actograms of representative individuals from treatment groups (indicated above the actogram) for Experiment 1 (A) and Experiment 2 (B, C). Each actogram shows the activity of one individual bird and each row represents 2 consecutive days. Black bars represent activity in each 2-min bin, where height of the bar is proportional to the amount of activity. Grey areas indicate when daylight lamps were switched off (between 2000 h and 0600 h). Dates at the y-axis give start and end of each experimental period.
Experiment 1
In the dark night period (period 1) of Experiment 1, groups of birds did not differ in their onset (
Results of the linear-mixed-effects models (LMMs) on the 4 activity response variables for the effect of night light color in experimental period 2 of Experiment 1.
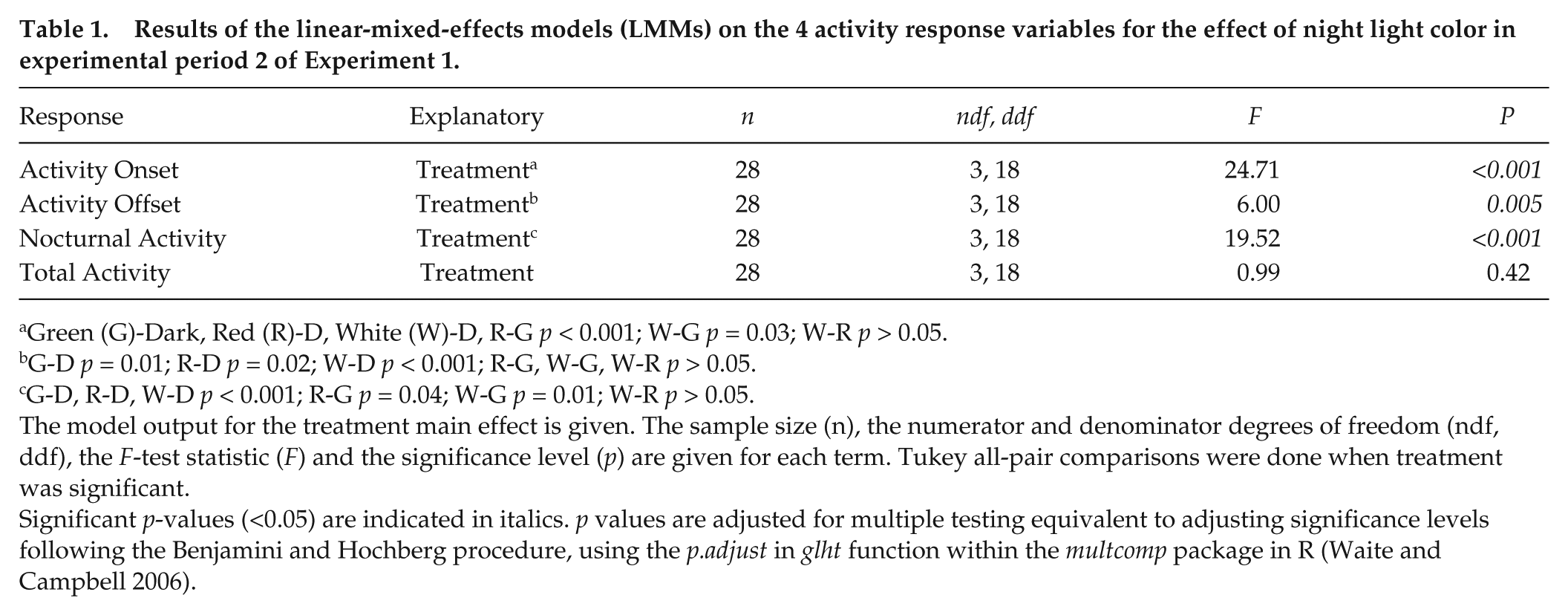
Green (G)-Dark, Red (R)-D, White (W)-D, R-G
G-D
G-D, R-D, W-D
The model output for the treatment main effect is given. The sample size (n), the numerator and denominator degrees of freedom (ndf, ddf), the
Significant
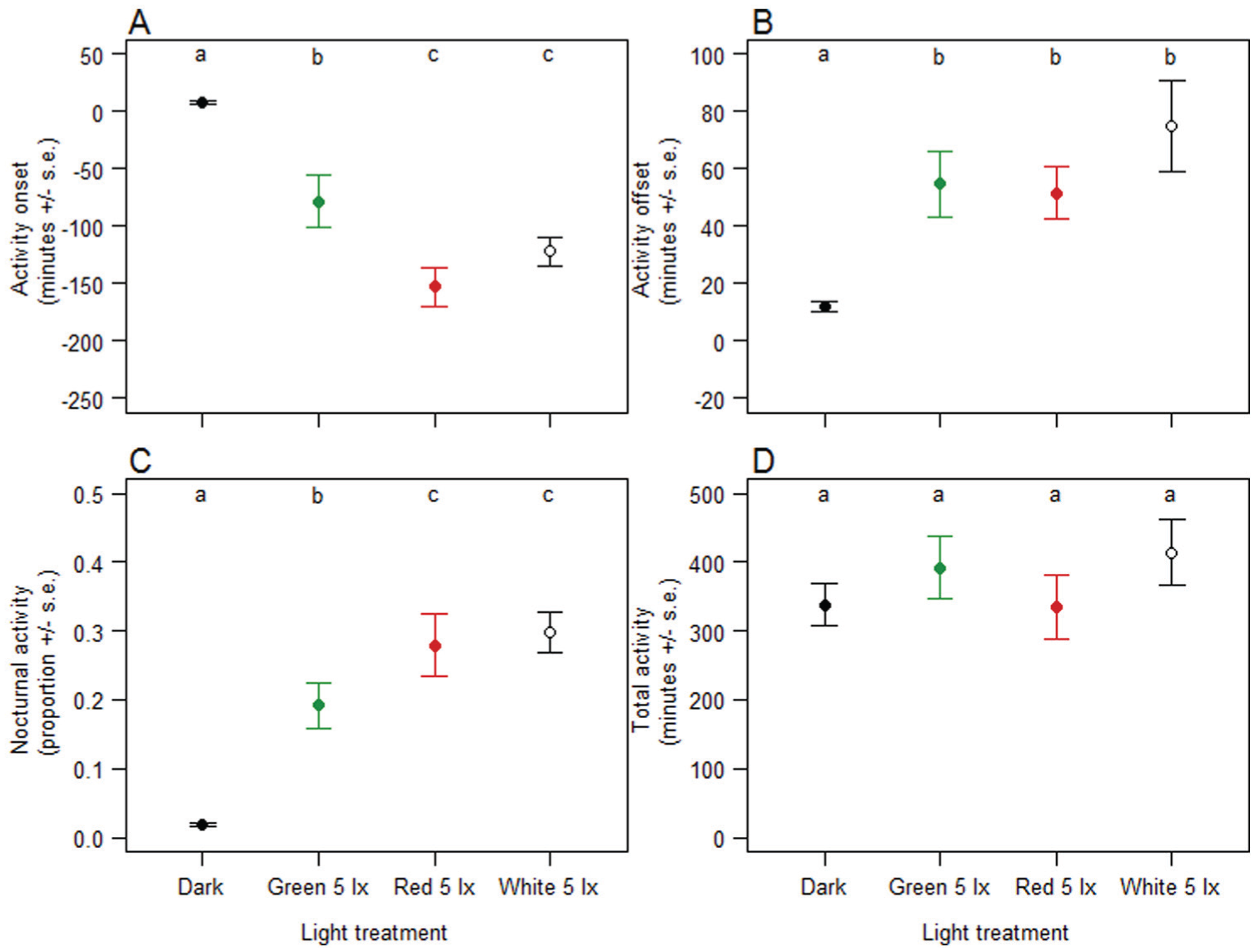
Behavioral response of blue tits to the 4 light color treatments in Experiment 1 and during experimental period 2 of Experiment 1. Data were averaged across individuals. (A) Onset of activity in min relative to ‘start of day’ (min after daylight was switched on). (B) Offset of activity in min relative to ‘end of day’ (min after daylight was switched off). (C) Nocturnal activity; part of the total activity that took place during the night (from daylight off till daylight on). (D) Total active min in 24-h cycle. Letters indicate the results of the Tukey post-hoc tests (see Table 1).
Experiment 2
In Experiment 2, birds exposed to green or white light at night changed their activity onset in a different manner with increasing light intensity. For both colors, activity was advanced more with increasing intensity. The effect was smaller in the green light than in the white light at low intensities, but increased faster in green than in white as intensity increased (Table 2, Fig. 3A). Activity offset was affected by light intensity in the same way for the 2 light colors; birds delayed their offset further into the night with increasing light intensity, up to 30 min in both green and white light (Table 2, Fig. 3B). As in Experiment 1, the increase in the daily activity that was moved into the night with increasing light intensity was proportional to the advancement of onset in both light colors (Table 2, Fig. 3). Birds did not change the total amount of time they were active in each 24-h cycle in response to light intensity or color (Table 2, Fig. 3D) and the variation in total activity was large. By comparing the individual response variables over the dark night periods of Experiment 2, we found no major temporal effects, carry-over effects of treatments, or effects of neighboring birds during experimental periods; although, there were some significant differences (for details, see Suppl. Fig. S3 and Suppl. Table S1).
Results of the linear-mixed-effects models (LMMs) on the 4 activity response variables, for the effect of night light color and intensity in experimental periods 2 and 4 of experiment 2.
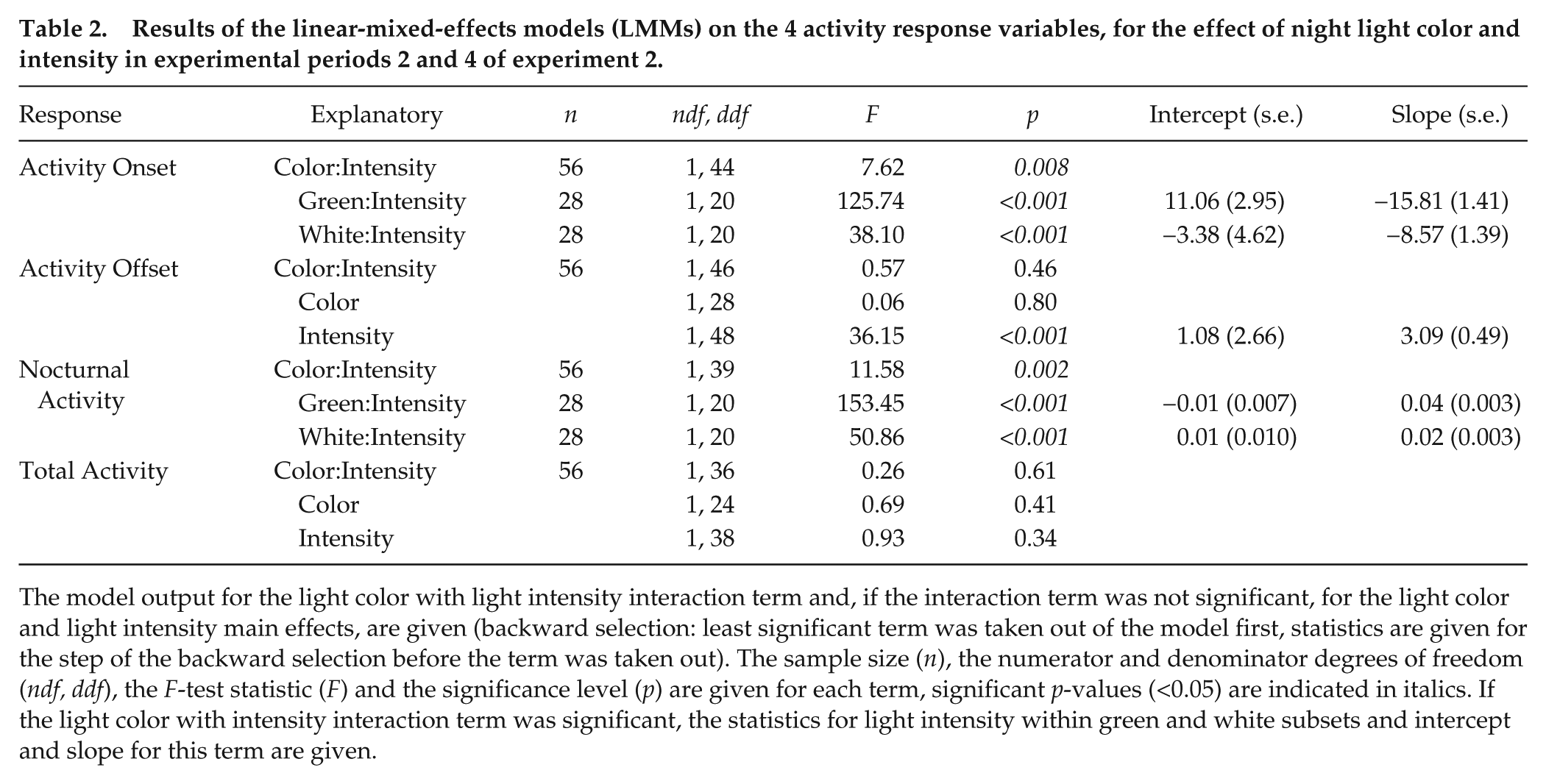
The model output for the light color with light intensity interaction term and, if the interaction term was not significant, for the light color and light intensity main effects, are given (backward selection: least significant term was taken out of the model first, statistics are given for the step of the backward selection before the term was taken out). The sample size (
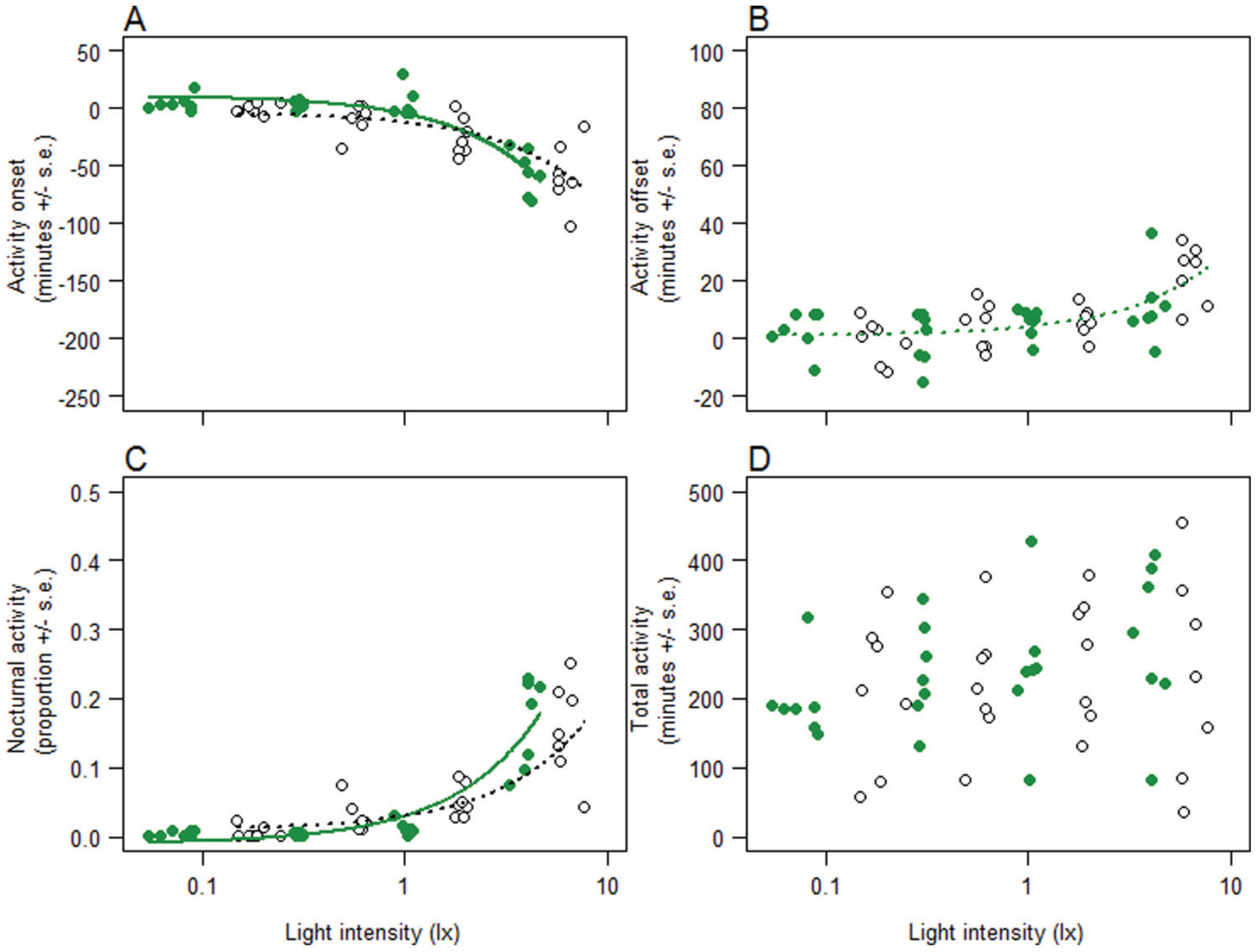
Behavioral response of blue tits to green light (closed dots) and white light (open dots) with different intensities: Experiment 2, during experimental periods 2 and 4. Raw data has been averaged across the individuals (each dot represents one individual). (A) Onset of activity in min relative to ‘start of day’ (min after daylight was switched on). (B) Offset of activity in min relative to ‘end of day’ (min after daylight was switched off). (C) Nocturnal activity; part of the total activity that took place during the night (from daylight off until daylight on). (D) Total active min in 24-h cycle. Predictions of the LMMs with significant terms (see Table 2) are plotted (in A and C as continuous lines for green light and dotted lines for white light; in B, as dotted lines for both light colors). Note that light intensity on the x-axis is plotted on a logarithmic scale.
Discussion
In Experiment 1, the daily activity patterns of the birds were clearly disrupted. Most strikingly, their activity had already started up to 2 h before the day lights went on. This advancement in their onset of activity in the morning was present under all light colors but was most pronounced in red and white light. Their offset of activity was slightly delayed in all light treatments. The total amount of time that birds were active over a 24-h period did not change due to light at night but part of their daily activity was moved into the night, proportional to the advancement of onset. In Experiment 2, there was a clear dose-response relationship for change in activity pattern vs. light intensity. Furthermore, we show that the effect of light intensity on activity onset was smaller in green than in white light in the lower range of intensities, but increased faster with increasing intensity in green light. Activity offset was delayed more with more green or white light, and the results for total activity and nocturnal activity were consistent with those of Experiment 1. The advancement in onset of activity when exposed to low intensity illumination at night appears to be a direct response, as the nightly activity largely disappears as soon as the nights are dark again, and this yields an activity pattern comparable with the situation before light at night. Whether the response to low light levels is a true masking response remains to be tested in constant conditions.
Although the general patterns are quite similar, there are some discrepancies in the results of the 2 experiments. Firstly, the strength of the response to light at night differed for what was basically the same treatment (green 5 lux and white 5 lux treatments). While the birds advanced their onset by up to 2 h in Experiment 1, in Experiment 2, this was only slightly more than 1 h. Also, the delay of offset was larger in the first experiment for the same treatments. Birds stayed active about 1 h longer in the evening in Experiment 1 when daylight was switched off, whereas, in the Experiment 2, this was for only about 30 min in the highest light intensities. Secondly, the difference in the onset of activity to green and white light was not consistent between the 2 experiments. In Experiment 1, green light clearly had less effect than white light, whereas, in Experiment 2, this difference was the case for the lower but not for the higher intensities of light (including the intensity used in Experiment 1). The difference between green and white in the first experiment was the main reason that we chose to have these 2 colors in Experiment 2 to examine light intensity (due to limitations in space and time, we could not have all 3 light colors).
It is unclear why these discrepancies originate; the set-up of both experiments, the experimental procedures and the spectral composition of the lamps were the same. However, birds were older in the second experiment and the experiments were done at different times of the year. We could speculate that the stronger response to light at night in Experiment 1 compared to Experiment 2 originates from the intrinsic, circannual rhythm in light sensitivity of our birds: Experiment 1 was conducted in late autumn, when birds normally reacquire photosensitivity, whereas Experiment 2 was undertaken in late spring to early summer, when birds in temperate regions normally show little sensitivity to further increases in day length (Dawson et al., 2001). This photoperiodic mechanism regulates seasonal breeding but daily rhythms also have a seasonal basis. Dawn song, for example, is performed earlier in early spring and later in the breeding season, and it was shown recently by Da Silva et al. (2015) that effects of light at night on dawn song can be much stronger in early spring.
It is interesting to compare our current results with our previous work in another passerine species, the great tit. A similar experiment was conducted (de Jong et al., 2016), in which the experimental set-up, procedures and spectral compositions and intensities of the (white) lamps were the same. Like in blue tits, daily activity patterns of great tits showed a dose-dependent response to light at night. Under white light, both species progressively advanced their activity onset with increasing light intensity. However, great tits advanced faster and much more, up to 5 h in the 5-lux treatment, whereas the maximum advancement of the blue tits under white light of 5 to 7 lux was around 2 h. Although the spectral absorption characteristics of retinal photoreceptors measured in several bird species are broadly similar (Bowmaker et al., 1997), it is possible that great tits and blue tits differ in their spectral sensitivities and action spectra, by differences in spectral transmission through oil droplets and ocular media, and hence perceive the different light colors as different intensities. In fact, another experiment suggested that blue tits were less able to find their food than great tits, when kept under the same, very low light levels (Caro et al., unpublished data). For blue tits, the wavelengths of maximum sensitivity are known for the rods (503 nm), single cones (372 nm [ultraviolet-sensitive], 449 nm [short-wavelength-sensitive], 502 nm [medium-wavelength-sensitive] and 563 nm [long-wavelength-sensitive]) and double cones (563 nm [principal] and 565 nm [accessory]) (Hart et al., 2000); for great tits, these variables are yet to be measured. However, this does not mean that the visual action spectrum of the blue tit directly explains changes in behavior; i.e., it is unknown how the relative abundance of light of specific wavelengths causes changes in, for example, daily activity patterns.
Apart from the bird species, photoperiod also differed between the 2 experiments: here we used 14-10 LD, whereas great tits were kept under shorter days (0815 h of light, 1545 h of D). The difference in advancement of activity could also be due to the difference in day length; under long nights and short days, birds have only a short window to forage and obtain enough energy. The nocturnal illumination could provide an opportunity to start foraging already before daylight and to spread the workload over a longer period (de Jong et al., 2016). Birds under long nights might therefore make use of the extra light at night more than birds under shorter nights and longer days. The results in the advancement of the onset of activity for blue tits in the current study are in line with those found for city blackbirds (1 h; Dominoni et al., 2014) and songbirds close to streetlights (2 h; Kempenaers et al., 2010).
Our finding that green light at low intensities disturbs the daily rhythm of blue tits less than white light at the same intensities (in lux) is in line with earlier studies that show that long wavelength light (red and white) penetrates the skull more easily (Hartwig and van Veen 1979) and has a larger effect on gonadal development (Malik et al., 2002) and stress hormones (Ouyang et al., 2015). It remains unclear why we do not find this difference in higher light intensities, and more studies are needed to provide a decisive answer about whether green light is truly less disturbing for birds. If this indeed turns out to be the case, a next step would be to see whether the effects on daily activity patterns have any fitness consequences; for example, through disturbance of sleep (Steinmeyer et al., 2013; Raap et al., 2015). A field experiment in previously dark, natural areas, using lamps with the same spectral compositions did not show clear, unidirectional effects of experimental nocturnal illumination on fitness so far; but this underlines the need for further research on fitness consequences (de Jong et al., 2015). The same experiment did show an advancing effect of both white and green light on lay date in great tits. So, while daily rhythms might be affected less by green light, this seems not to be true for seasonal breeding, and thus different behavioral and physiological responses might respond differently to different wavelengths of light.
The dose-response relationship of change in activity pattern vs. light intensity which we show here, indicates that decreasing light intensity could be a measure to reduce the effects of artificial light at night. Gaston et al. (2012) indicated 5 main management options to reduce the effects of night-time light pollution, among which are changing the intensity of lighting as well as changing the spectral composition of lighting. The fact that the light spectra used in this study enable full color vision for humans, because they cover a continuous and broad spectrum (Spoelstra et al., 2015), and that the range in light intensities used is comparable with light levels regularly found in natural or rural areas (for example, near roads), makes our results suitable for application in outdoor lighting. We have shown that there are differences in effects on avian daily rhythms between light colors and that green light at low light intensities disturbs less the daily activity patterns of these birds. This means that the strength of the disturbance of behavior of wild birds can be mitigated by changes in the characteristics of outdoor lighting, and this opens up the possibilities for conservationists and landscape planners to use alternative lighting in their daily practice.
Footnotes
Acknowledgements
We thank Marylou Aaldering and Franca Kropman for taking good care of our experimental birds and Ab Wijlhuizen, Gilles Wijlhuizen, Jeroen Laurens and Cock van der Meer for technical assistance during the study. We are grateful to Bart van Lith, Koosje Lamers and Jenny Ouyang for helping with a lot of practical work. This research is supported by the Dutch Technology Foundation STW, which is part of the Netherlands Organization for Scientific Research (NWO), and which is partly funded by the Ministry of Economic Affairs. The project is supported by Philips and the Nederlandse Aardolie Maatschappij (NAM).
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
