Abstract
The recall of conditioned fear extinction exhibits a circadian rhythm in humans and rodents, with optimal extinction recall occurring during the early active phase. However, it remains unclear whether this rhythm depends on the circadian modulation of mechanisms supporting memory consolidation versus memory maintenance and retrieval. Here, adult male rats underwent conditioned fear extinction at one of four times throughout the day and then, starting 24 h after extinction, were repeatedly tested for extinction recall over the next 24 h. Rats undergoing extinction learning during the early active phase tended toward accelerated extinction learning compared with rats in other groups, pointing to rhythms in mechanisms that support extinction memory encoding. The next day, the strength of extinction recall followed a 24-h cycle that depended not on the time of day of extinction learning but, instead, on the time of day of extinction recall. This latter finding indicates a rhythm in mechanisms supporting extinction memory maintenance and/or retrieval. Subsequent testing for fear relapse in the conditioning context suggested reduced fear in rats tested during the early active phase. These results lay the groundwork for mechanistic investigations of circadian rhythms in fear extinction memory.
The circadian system pervasively modulates learning and memory mechanisms (Krishnan and Lyons, 2015; Rawashdeh et al., 2018; Hartsock and Spencer, 2020; Bolsius et al., 2021). This modulation often manifests at the behavioral level, contributing to circadian rhythms in memory-related behavior (Hartsock and Spencer, 2020). Hence, behavioral experiments can be used to narrow in on learning and memory mechanisms that fall under circadian control.
Memory consists of 4 sequential mechanistic events: encoding (information processing), molecular consolidation (memory formation), maintenance (memory retention), and retrieval (memory recall). At the behavioral level, however, assessments of memory can occur only during learning and recall. Circadian rhythms in memory-related behavior during an initial learning session suggest rhythms in mechanisms of encoding. In recall sessions, rhythms that depend on the time of day of learning suggest rhythms in molecular consolidation, whereas those that depend on the time of day of recall suggest rhythms in maintenance and/or retrieval. In addition, some memories are characterized by time-stamping, whereby rhythms in memory recall occur at regular 12- or 24-h intervals after the learning session. Experiments that can distinguish between these possibilities permit informed hypotheses regarding the mechanisms mediating circadian influences on memory-related behavior (Hartsock and Spencer, 2020).
The extinction of cued conditioned fear is a learning and memory process in which a sensory cue previously associated with danger (Fear Conditioning, Figure 1a) is repeatedly presented in a safe environment, reducing fear responding (Extinction Learning, Figure 1b). The recall of an extinction memory can be assessed through a subsequent behavioral session wherein the sensory cue is again presented in a safe environment (Extinction Recall, Figure 1c). After extinction, however, fear can return due to the passage of time (spontaneous recovery) (Pavlov, 1927; Rescorla, 2004), the changing of environmental contexts (renewal) (Bouton and King, 1983), or re-exposure to an aversive stimulus (reinstatement) (Rescorla and Heth, 1975). For this reason, it is common to re-assess the strength of extinction recall also after fear has returned (Fear Relapse, Figure 1d).
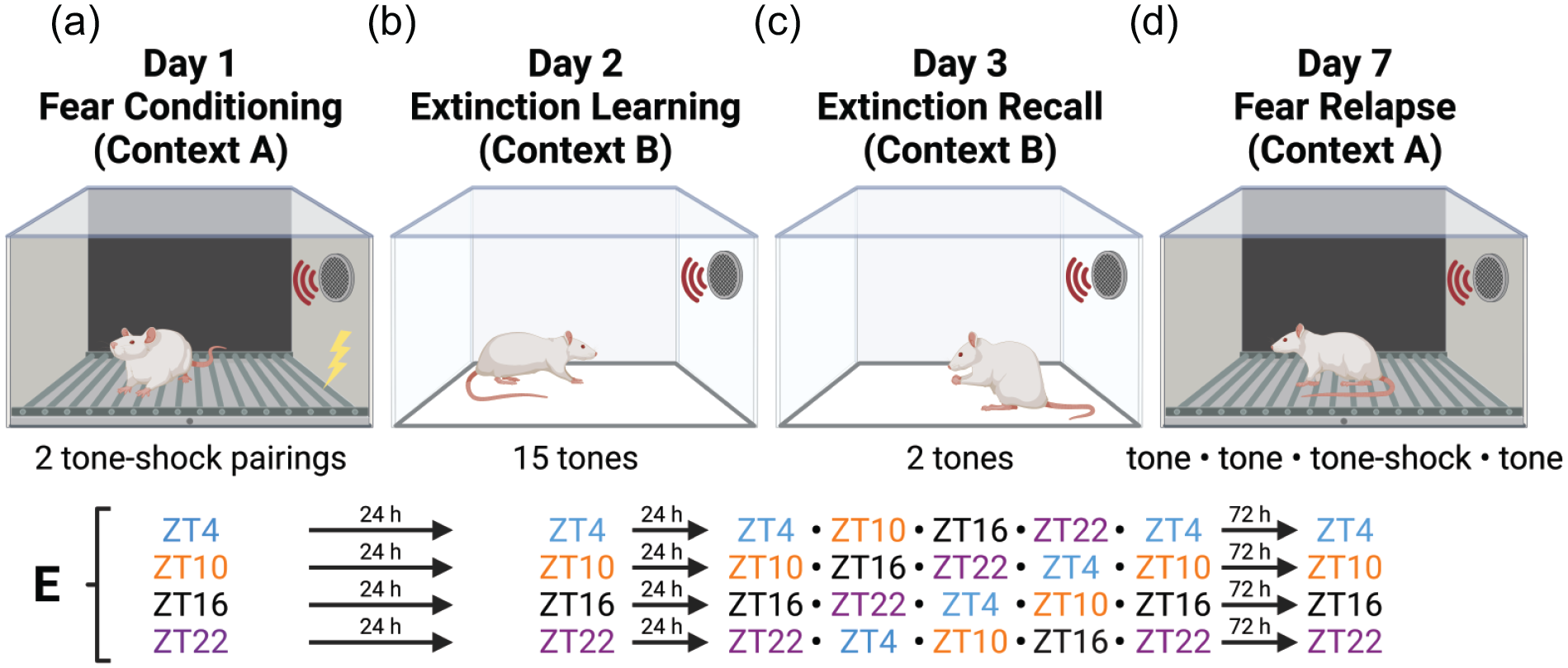
We used a standard cued conditioned fear and extinction paradigm. (a) On Day 1, rats underwent fear conditioning in Context A, with 2 tone-shock pairings at ZT4, ZT10, ZT16, or ZT22. (b) On Day 2, approximately 24 h after fear conditioning, rats underwent extinction learning in Context B, with 15 presentations of the tone alone. (c) On Day 3, beginning 24 h after extinction learning, rats were repeatedly tested at 6-h intervals for 2-tone extinction recall in Context B. (d) On Day 7, rats were returned to Context A for fear relapse testing at the same ZT at which extinction learning originally occurred. Fear relapse consisted of 4 tone presentations, the third of which was paired with shock. (e) Schedule of behavioral sessions. Abbreviation: ZT = zeitgeber time.
The recall of an extinction memory meets multiple criteria for circadian modulation: (1) it exhibits a time-of-day difference, peaking early during the active phase in humans and rodents (Chaudhury and Colwell, 2002; Pace-Schott et al., 2013; Woodruff et al., 2015); (2) this time-of-day difference can be endogenously sustained in constant conditions (Chaudhury and Colwell, 2002); and (3) this time-of-day difference entrains to changes in the environmental light-dark cycle (Chaudhury and Colwell, 2002). Furthermore, the time-of-day difference in extinction recall can be eliminated by local knockdown of Period1/2 clock gene expression in the infralimbic prefrontal cortex (Woodruff et al., 2018), a brain area necessary for extinction recall in rodents (Quirk et al., 2000; Bukalo et al., 2015; Do-Monte et al., 2015). Yet 2 critical questions remain unanswered. First, is this time-of-day difference truly rhythmic when sampling at more than 2 times of day? Second, does this time-of-day difference depend on the time of day of extinction learning or the time of day of extinction recall?
Here, we describe an experiment addressing these questions. Adult male Sprague-Dawley rats (n = 6, N = 24; Envigo, Madison, WI) pair-housed in clear plastic filter-top cages were acclimated for 1 week to a 12 h:12 h light-dark cycle, with lights on at 0440 h Mountain Time. All rats were housed in the same room. Teklad Rodent Diet 8640 (Envigo) and water were available ad libitum. Animal husbandry occurred during the light phase or under dim red light. All procedures were approved by the University of Colorado Institutional Animal Care and Use Committee and conducted in accordance with the ethical treatment of animals.
Figure 1e illustrates our behavioral timeline. Before and after each session, cages were placed into double-lined Contractor Clean-Up Bags (Husky, Grand Prairie, TX) under dim red light and transported on a wheeled cart between the vivarium and the anteroom of a behavioral testing suite. All testing occurred under dim red light to control for acute suppression of exploratory behavior by white light (Warthen et al., 2011). Freezing, defined as the cessation of all movements except respiration for a full 1-sec duration, was visually assessed at 10-sec intervals throughout each session as a behavioral correlate of fear. Two-way mixed-model analyses of variance (ANOVAs) were performed in Prism (Version 9.1.1). Cosinor analysis was performed using Cosinor.Online (Molcan, 2019).
On Day 1 of the experiment, rats underwent fear conditioning at zeitgeber time (ZT) 4, ZT10, ZT16, or ZT22 ± 30 min. Fear conditioning occurred in a plexiglass chamber measuring 29.2 × 21.0 × 25.4 cm with shock grid flooring (Context A; Med Associates, Saint Albans, VT). Context A was cleaned with 70% ethanol and allowed to dry before each rat. Conditioning consisted of a 5-min baseline and two 30-sec tones (3 kHz, 80 dB), each co-terminating with a 2-sec shock (0.8 mA) and followed by a 120-sec interval. All groups exhibited minimal freezing at baseline, indicating no time-of-day differences in baseline fear (Figure 2a). We observed robust increases in freezing with repeated tone-shock pairings (main effect of shock trial: F2,40 = 346.2, p < .0001). As in our prior work (Woodruff et al., 2015, 2018), we found no time-of-day differences in freezing after shock delivery.
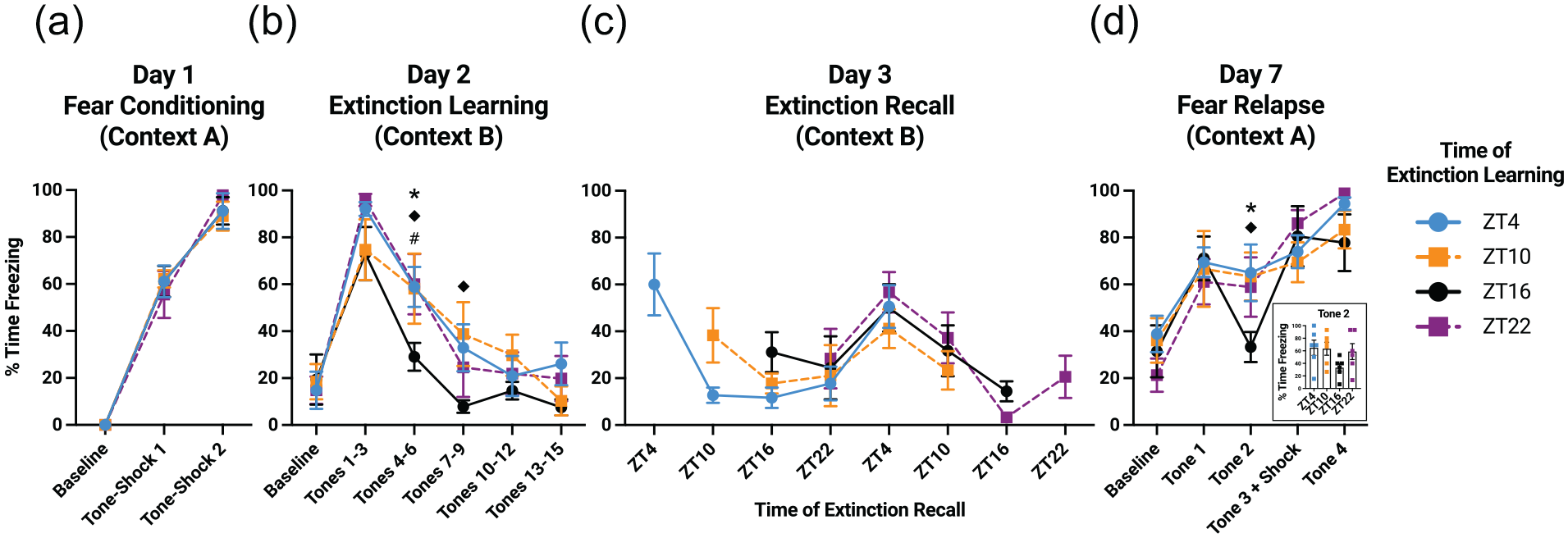
Circadian rhythms in fear extinction recall depend on the time of day of extinction recall, not the time of day of extinction learning. (a) On Day 1, rats froze more after repeated tone-shock pairings, regardless of the fear conditioning ZT. (b) On Day 2, all groups recalled fear and learned extinction to the same extent, although ZT16 rats learned extinction more quickly than rats at other ZTs (p < .05 when comparing ZT16 with ZT4*, ZT10◆, and ZT22#). (c) On Day 3, freezing was greatest at ZT4 and least at ZT16, regardless of the time of extinction learning. (d) On Day 7, all groups showed comparable fear renewal in response to the first tone, but ZT16 rats showed decreased freezing in response to the second tone (p < .05 when comparing ZT16 with ZT4* and ZT10◆). All groups then showed comparable fear reinstatement after shock re-exposure. Data are presented as mean ± SEM. Abbreviation: ZT = zeitgeber time.
On Day 2, each group underwent extinction learning 24 h ± 60 min after the conditioning session. Extinction occurred in an adjacent room in custom-built plexiglass boxes measuring 42.5 × 42.5 × 62.2 cm (Context B). Context B was cleaned with Sani-Cloth Plus Germicidal Disposable Cloths (Professional Disposables International, Orangeburg, NY) and allowed to dry before each rat. As an additional means of distinguishing Context B from Context A, liquid peppermint extract (Kroger, Cincinnati, OH) was wiped on the ceiling of Context B before each rat, and an open plastic container of liquid peppermint extract was placed just outside each behavior box. The extinction learning procedure consisted of a 3-min baseline, followed by 15 tones identical to those in Context A but with no shock delivery. Intervals following each tone were pseudorandomized between 90 and 120 sec. Freezing was uniformly low at baseline, indicating little generalization of contextual fear from Context A to Context B (Figure 2b). All groups showed robust increases in freezing between baseline and a trial block consisting of the first 3 tones and post-tone intervals (main effect of trial block: F1,20 = 169.0, p < .0001), illustrating strong fear recall at 24 h. All groups showed reductions in freezing with repeated tone presentations (main effect of tone trial block: F4,80 = 78.4, p < 0.0001), indicating strong extinction learning. Fisher’s least significant difference unprotected post hoc tests revealed a significant reduction in freezing at ZT16 compared with ZT4, ZT10, and ZT22 during tones 4 to 6 (p < 0.05) and at ZT16 compared with ZT10 during tones 7 to 9 (p < .05). This suggests the possibility that ZT16 rats learned extinction more quickly than rats at other ZTs. We note that there was no overall main effect of ZT and no interaction between ZT and tone trial block, yet we draw attention to these post hoc results to highlight a trend consistent with our previous findings (Woodruff et al., 2015). We found no differences between groups during the last 3 tones, suggesting that, by the end of the session, all groups learned extinction to the same extent.
On Day 3, each group underwent extinction recall testing starting 24 h ± 60 min after the extinction learning session. Extinction recall occurred in Context B, consisting of a 3-min baseline and two 30-sec tones, each followed by an interval of 120 sec. Using only 2 tones in the extinction recall session enabled us to limit additional extinction learning. Each group was then serially retested at 6-h intervals for the next 24 h (i.e., each group underwent a total of 5 extinction recall tests). Freezing was greatest at ZT4 and least at ZT16, regardless of the time of day of extinction learning (Figure 2c). To run a balanced 2-way mixed-model ANOVA, we averaged freezing values from the first and fifth recall sessions within each group, since these 2 sessions took place at the same ZT. This ANOVA indicated a main effect of extinction recall ZT (F3,80 = 12.9, p < .0001). Importantly, there was no main effect of extinction learning ZT and no interaction between extinction learning ZT and extinction recall ZT. Assuming a 24-h oscillatory period, we then ran a cosinor analysis that aligned the extinction recall ZT freezing data for each group (e.g., all recall measurements at ZT4 were analyzed as one group). This analysis revealed that freezing fit a cosine function with an acrophase of ZT4.7, mesor of 29.6%, and amplitude of 18.3% (zero-amplitude test: F2,117 = 19.2, p < .0001) (Figure 3a). Notably, mean freezing at ZT10 and ZT22 fell between mean freezing at ZT4 and ZT16. The same general pattern emerged also at the level of individual rats (Figure 3b). These data indicate an overall diurnal rhythm in extinction recall that depends on the time of day of extinction recall.
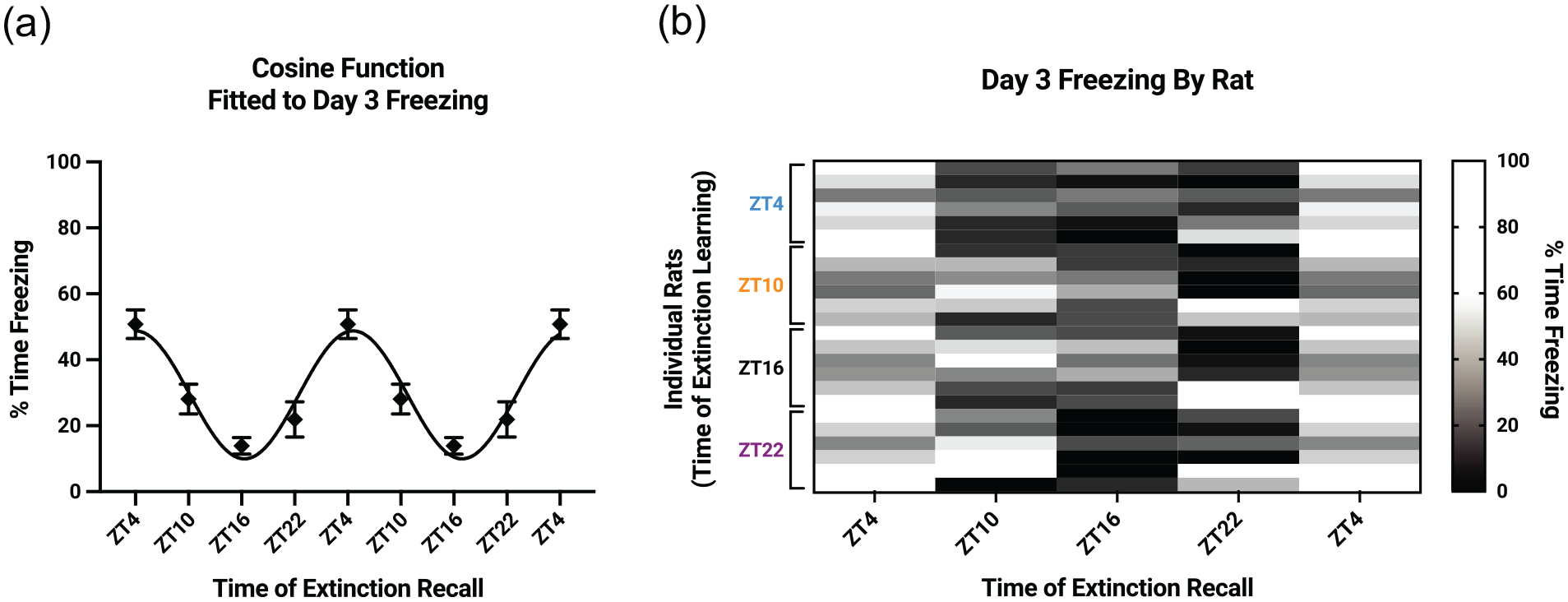
Fear extinction recall follows a cosine function. (a) Day 3 freezing data are collapsed across extinction learning ZT and double-plotted against a cosine function, illustrating a rhythm in extinction recall that depends on the time of extinction recall. ZT4 data are triple-plotted. Data are presented as mean ± SEM. (b) Percent time freezing (black-to-white scale gradient) follows this rhythm at the level of individual rats (rows). ZT4 data are double-plotted. Abbreviation: ZT = zeitgeber time.
On Day 7, rats were returned to Context A and tested for fear relapse. The session took place at the same ZT at which fear conditioning and extinction originally occurred. After a 3-min baseline, four 30-sec tones were presented, each followed by a 120-sec interval. The third tone co-terminated with a 2-sec shock. This allowed us to assess fear renewal during the first 2 tones and fear reinstatement after shock delivery. Freezing was uniformly low at baseline and increased significantly during the first 2 tones and corresponding post-tone intervals (main effect of trial: F2,40 = 17.0, p < .001), indicating strong fear renewal (Figure 2d). For the second tone, however, freezing declined to near-baseline levels in ZT16 rats. Although there was no overall main effect of ZT and no interaction between ZT and tone trial, freezing was reduced in rats tested at ZT16 compared with rats tested at other ZTs (Figure 2d inset), a trend supported by Fisher’s least significant difference unprotected post hoc tests comparing ZT16 with ZT4 and ZT10 (p < .05). This observation suggests that ZT16 rats may exhibit reduced fear renewal after multiple cue-alone presentations in the conditioning context. After shock delivery, freezing was uniformly high across groups, revealing comparable fear reinstatement at all ZTs. At the final tone, there were no differences in freezing between groups. These data open the possibility that, while fear reinstatement does not differ across ZTs, memory for fear extinction may better generalize back to the conditioning context at ZT16.
Our findings lead to several important conclusions. First, fear extinction recall is indeed rhythmic when sampled at 4 ZTs evenly distributed throughout the day. If the time-of-day difference observed in previous work were due strictly to lighting conditions or an activity phase difference, one might expect ZT4 freezing to approximate ZT10 freezing and ZT16 freezing to approximate ZT22 freezing. However, our data show that freezing at ZT10 and ZT22 is intermediate between freezing at ZT4 and ZT16. This provides additional backing for the contention that extinction recall is circadian-modulated.
Second, this rhythm in extinction recall is dependent on the time of day of extinction recall, not the time of day of extinction learning. This points to the circadian modulation of molecular mechanisms involved in the maintenance and/or retrieval of extinction memories. Circadian fluctuations in the excitability of infralimbic prefrontal cortex neurons represent one likely contributor. The activity of infralimbic prefrontal cortex neurons determines the strength of extinction recall (Do-Monte et al., 2015; Kim et al., 2016). The circadian system modulates the expression and activity of the transcription factor CREB (Eckel-Mahan et al., 2008; Fropf et al., 2014; Rawashdeh et al., 2016), CREB regulates neuronal membrane excitability (Dong et al., 2006; Han et al., 2006), and neuronal membrane excitability exhibits circadian rhythms (Cauller et al., 1985; Chaudhury et al., 2005; Parekh et al., 2018). Thus, the circadian modulation of CREB in infralimbic prefrontal cortex neurons could influence how readily these neurons can be engaged—such as by known inputs from the hippocampus (Ye et al., 2017; Qin et al., 2021), amygdala (Senn et al., 2014; Hagihara et al., 2021), or prelimbic prefrontal cortex (Marek et al., 2018)—during the retrieval of an extinction memory.
Third, fear extinction may be learned more quickly during the early active phase than other times of day. This may reflect rhythms in encoding processes that occur during extinction learning. Prior work shows that extinction learning engages both the infralimbic prefrontal cortex and the amygdala (Sotres-Bayon et al., 2007; Sotres-Bayon et al., 2009; Sierra-Mercado et al., 2011; Do-Monte et al., 2015). Thus, circadian modulation could occur in either structure, facilitating more efficient information processing during extinction learning in the early active phase.
Finally, our data suggest that rats re-experiencing cue presentation at ZT16 may be better able to generalize extinction learning back to the conditioning context. Although this effect is subtle, it merits consideration in light of our other findings. For example, enhanced extinction recall during the early active phase may be supported by similar mechanisms across extinction sessions (e.g., Day 3 or Day 7 in our experiment). Although on Day 7 our experiment cannot discriminate whether this effect owes to the time of extinction learning or the time of fear relapse testing, the finding conflicts with past studies on fear renewal. Those studies have led to the widespread view that extinction does not generalize across contexts (Bouton and Bolles, 1979; Lovibond et al., 1984; Bouton and Ricker, 1994; Bouton et al., 2006). Yet virtually all behavioral characterization of renewal in rodents has been conducted during the inactive phase of the activity cycle. Our current findings suggest that fear renewal can be suppressed during the early active phase.
Fear extinction provides the conceptual foundation for exposure therapy, the gold standard treatment for fear-based mental disorders (Rauch et al., 2012; Vervliet et al., 2013; Craske et al., 2018). Our findings indicate that the recall of exposure therapy may fluctuate depending on the time of day. In addition, they suggest that exposure-related learning may occur most quickly during the early active phase. Finally, they open the important possibility that exposure therapy during the early active phase may facilitate the generalization of exposure memories outside of the therapy setting. Thus, circadian rhythms in fear extinction may represent a pragmatic and economical target for the enhancement of exposure therapy.
Footnotes
Acknowledgements
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
