Abstract
Both the circadian clock and sex hormone signaling can strongly influence brain function, yet little is known about how these 2 powerful modulatory systems might interact during complex neural processes like memory consolidation. Individually, the molecular components and action of each of these systems have been fairly well-characterized, but there is a fundamental lack of information about how these systems cooperate. In the circadian system, clock genes function as timekeeping molecules that convey time-of-day information on a well-stereotyped cycle that is governed by the suprachiasmatic nucleus. Keeping time is particularly important to synchronize various physiological processes across the brain and body, including those that regulate memory consolidation. Similarly, sex hormones are powerful modulators of memory, with androgens, estrogens, and progestins, all influencing memory consolidation within memory-relevant brain regions like the hippocampus. Despite clear evidence that each system can influence memory individually, exactly how the circadian and hormonal systems might interact to impact memory consolidation remains unclear. Research investigating either sex hormone action or circadian gene function within memory-relevant brain regions has unveiled several notable places in which the two systems could interact to control memory. Here, we bring attention to known interactions between the circadian clock and sex hormone signaling. We then review sex hormone–mediated control of memory consolidation, highlighting potential nodes through which the circadian system might interact during memory formation. We suggest that the bidirectional relationship between these two systems is essential for proper control of memory formation based on an animal’s hormonal and circadian state.
Although neuroendocrinologists have long considered sex hormones to be crucial regulators of both neuronal function and animal behavior, many scientists outside of the field have been wary of considering sex hormones in their studies. Historically, females have been understudied, in part due to the perception that fluctuations in hormone levels across the estrous cycle introduce an uncontrollable cofounding variable that increases variability among females (for review see Beery and Zucker, 2011; Shansky, 2019). However, recent meta-analyses have shown that variability within females is no greater than that within males, including in studies of learning and memory (Kaluve et al., 2022; Prendergast et al., 2014). Though the estrous cycle can add additional considerations when using intact females, there are clear sex differences in many disorders such as Alzheimer’s disease (for review see Ferretti et al., 2018; Toro et al., 2019), sleep disorders (for review see Hajali et al., 2019; Mallampalli and Carter, 2014), and learning disabilities (for review see Ferri et al., 2018; Krafnick and Evans, 2018). Similarly, older women are at an increased risk of cognitive decline and dementia largely due to changes in hormone levels caused by menopause (for review see Conde et al., 2021; Scheyer et al., 2018). These clear differences make sex-specific investigations both biologically and clinically relevant.
One major contributor to these sex differences is steroid sex hormones, which serve as potent modulators of many brain functions in both sexes, including memory formation across a variety of species (Gervais et al., 2019; Gresack and Frick, 2006; Hodgson et al., 2008; Koss and Frick, 2019; Orr et al., 2009; Packard et al., 1996). Although various factors contribute to sex differences (e.g., different gene products from X and Y chromosomes, developmental factors, epigenetic factors), sex hormones play a key role in establishing and maintaining sex differences. A more complete understanding of differences in cellular and molecular processes underlying hormonal regulation of learning and memory is therefore necessary to develop sex- and age-specific treatments for many sex-biased disorders.
Sex hormone production has been extremely well-characterized (for review see Frick et al., 2015; Kriegsfeld and Silver, 2006), although it is less clear how sex hormones interface with other regulatory systems. One important regulator for stimulating hormone production is the suprachiasmatic nucleus (SCN) in the hypothalamus. The SCN is the brain’s central circadian pacemaker, which establishes circadian rhythms in many vertebrate species. The SCN encodes both time-of-day and seasonal information, which it then communicates to other nuclei in the hypothalamus to stimulate hormone production and release. While the circadian clock has been well-studied for its role in regulating sex hormone production during development and in reproductive behaviors, much less is known about how the circadian clock itself might be influenced by sex hormones, especially in areas of the brain outside of those responsible for hormone production. Although there are studies considering how sex hormone action could alter timekeeping within the central circadian pacemaker, little is known about how this action could modify the SCN’s downstream afferent projections. Likewise, it is unclear how sex hormones could affect local clock gene expression within almost any brain region outside of those important for regulating hormone production itself. The hippocampus, a key brain region for spatial memory formation, is one such area with potential for a bidirectional relationship between clock genes and sex hormone function, as it is well-documented to be sensitive to both sex hormones and circadian oscillations (for review see Frick et al., 2015; Smies et al., 2022). Furthermore, within the retrosplenial cortex, another memory-relevant brain area, clock genes, such as Period1 (Per1), have been shown to modulate memory formation in a sex-specific manner, suggesting an interaction between the circadian system, sex hormones, and memory consolidation (Urban et al., 2021).
Here, we argue that clock genes and sex hormones synergistically function to regulate memory consolidation. First, we introduce the four core clock gene families that “keep time” across almost all cell types, including those in memory-relevant brain regions. Next, we discuss known examples in which clock gene expression is altered in response to experimental sex hormone manipulations. We then review what is known about sex hormone action in memory-relevant brain regions, specifically emphasizing nodes where clock gene regulation and sex hormone signaling might overlap to modulate memory. Finally, we discuss how clock genes and sex hormones may cooperate via a bidirectional signaling relationship to influence memory formation, potentially in a sex-specific or time-of-day-specific manner.
Clock Genes and the Circadian Cycle
The circadian system plays an essential role in synchronizing an animal’s internal systems with the external time of day. Almost all cells across the body show timekeeping and have at least some rhythmic expression of clock genes. This time-of-day information provided by the circadian system is used to synchronize a variety of physiological functions across the day/night cycle, including sleep (for review Fisher et al., 2013), feeding (for review see Fulgham et al., 2021), and even memory formation (for review see Smies et al., 2022). Despite the distinct features and functions of the tissues that depend upon circadian oscillations, they all require the same basic molecular feedback loop to maintain information about time of day. The molecular clock comprises a series of proteins that are expressed cyclically across the 24-h day. There are 4 central components of the molecular clock: Period (Per), Cryptochrome (Cry), Circadian Locomotor Output Cycles Kaput (Clock), and Brain and Muscle ARNT-Like 1 (Bmal1) (Figure 1). The interactions of Per, Cry, Clock, and Bmal1 form a negative transcription-translation feedback loop (TTFL), which occurs over a roughly 24-h cycle (for review see Yi et al., 2022). The TTFL is initiated by the dimerization of CLOCK and BMAL1 to E-box motifs upstream of the Per and Cry gene families. The CLOCK-BMAL1 heterodimer drives transcription of Per and Cry, which are then translated in the cytoplasm, where they form a PER-CRY heterodimer. Phosphorylation of PER induces trafficking of the PER-CRY complex to the nucleus where it recruits transcriptional repressors to block CLOCK and BMAL1 activity (Aryal et al., 2017; Duong et al., 2011), effectively inhibiting the transcription of the Per and Cry genes. The PER-CRY complex degrades over time to relieve the inhibition of the CLOCK-BMAL1 heterodimer, ultimately initiating a new round of Per and Cry transcription to begin a new cycle.
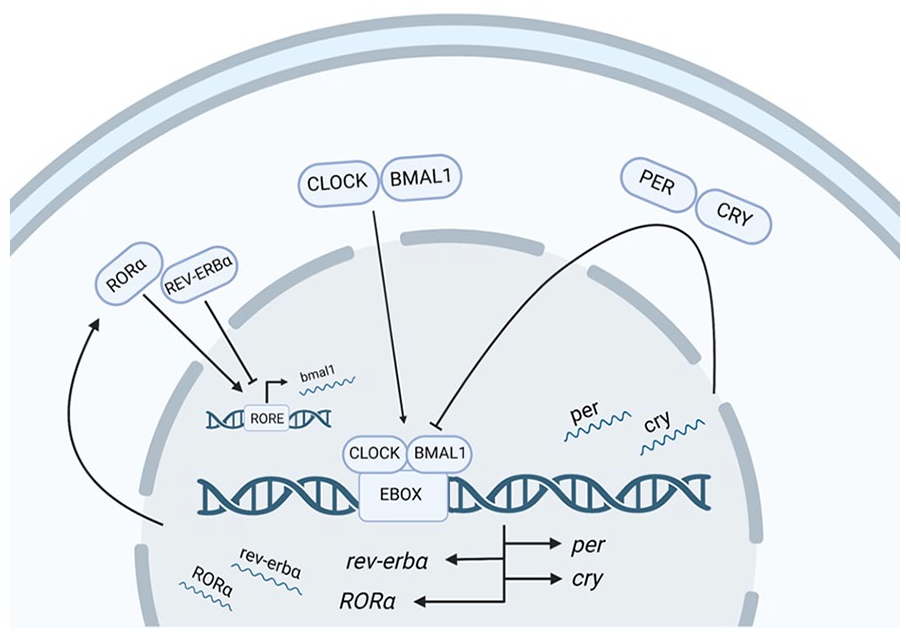
Generalized diagram of transcription-translation feedback loop of the four core clock gene families. Expression of the molecular clock begins when the CLOCK-BMAL1 heterodimer binds to E-box motifs in the Per and Cry promoters, driving their transcription. Once these proteins are translated in the cytoplasm, PER and CRY form a heterodimer that inhibits CLOCK-BMAL activity to block subsequent Per/Cry transcription. Eventually, degradation of PER and CRY alleviates this CLOCK/BMAL1 repression, restarting the cycle. In addition, REV-ERBα and RORα compete to bind retinoic acid–related orphan receptor response elements (ROREs) upstream of Bmal1 to promote (arrowhead) and inhibit (flat head) Bmal1 transcription to fine-tune the molecular clock. Created with BioRender.com.
While much research has been done to study the role of circadian oscillators within the SCN, less is known about how clock genes function in other brain areas and tissue types. For example, it is less clear how these clock genes function in memory-relevant brain regions like the hippocampus. Although it is well-documented that the circadian system can strongly influence memory formation across species (Chaudhury and Colwell, 2002; Jilg et al., 2010; Lyons et al., 2006; Lyons and Roman, 2009; Rawashdeh et al., 2016; Valentinuzzi et al., 2008), it is still unclear exactly how the clock exerts diurnal control over memory. It is likely that core clock genes function outside their role as circadian regulators in the SCN, performing tissue-specific functions within downstream satellite clocks in addition to their role as timekeepers. These tissue-specific functions would likely be enabled by interactions among clock genes and local modulators such as proteins, genes, and even hormones that act locally to control site-specific actions based on the time of day.
Core Circadian Clock Genes and Their Roles in Memory
Each of the four core clock gene families have been broadly implicated in memory. For the purpose of this review, we will briefly introduce some of the most prominent examples, but we encourage interested readers to see relevant reviews for a more comprehensive coverage (for review see Hartsock and Spencer, 2020; Smies et al., 2022). Mice with global deletions of Clock, Bmal1, Cry, or Per all show disrupted activity patterns and have deficits in memory formation (De Bundel et al., 2013; Jilg et al., 2010; Kondratova et al., 2010; Oishi et al., 2006; Van der Zee et al., 2008; Wardlaw et al., 2014). It is important to note that global knockdowns of core clock components typically impact the central circadian system as well as satellite clocks. Therefore, it is impossible to determine from these studies whether clock genes play a direct role in memory-relevant brain regions or whether their effects on memory are secondary to sleep-wake cycle disruptions (for review see Wright et al., 2012). Recently, however, there have been several studies that have specifically tested the effects of site-specific manipulation of clock genes. For example, forebrain-specific Bmal1 knockdown mice show impairments to both short-term and long-term memory formation (Price et al., 2016). Similarly, local manipulation of Per1 in the dorsal hippocampus (Kwapis et al., 2018), ventromedial prefrontal cortex (Woodruff et al., 2018), or retrosplenial cortex (Brunswick et al., 2023; Urban et al., 2021) alters memory performance in mice. This work strongly suggests that clock genes function locally within memory-relevant brain regions to modulate memory in addition to their well-documented roles in the central circadian system.
Other Clock Modulators and Their Implications in Memory Formation
Beyond these 4 core clock components, there are many other accessory feedback loops that fine-tune and regulate this cycle under different conditions and within different cell types (for review see Gallego and Virshup, 2007; Reddy and Rey, 2014; Reinke and Asher, 2019). Notably, in a key accessory feedback loop, the CLOCK-BMAL1 heterodimer can bind to E-box regions upstream of 2 nuclear orphan receptor genes, Rev-erbα and RORα. REV-ERBα and RORα proteins compete to bind to retinoic acid–related orphan receptor response elements (ROREs) in the Bmal1 promoter to inhibit or enhance Bmal1 transcription, respectively. This competition between REV-ERBα and RORα can fine-tune Bmal1 expression and therefore locally adjust the expression of the molecular clock and clock-controlled genes. Recent studies have revealed that REV-ERBα functions in the hippocampus to control neurogenesis (Schnell et al., 2014), excitatory long-term potentiation (Choi et al., 2018), and memory-relevant behaviors (Jager et al., 2014), although no one has yet tested the effects of site-specific manipulation of REV-ERBα or RORα in memory.
Hormones such as melatonin and corticosterone have been shown to serve as an important link in the circadian control of memory formation (Chaudhury et al., 2005; Dana and Martinez, 1984; Rawashdeh et al., 2007). Data from these studies suggest that circulating hormones could operate as a zeitgeber, to convey time of day information in memory-relevant brain regions during memory formation. We predict that fluctuations in sex hormones, both those from circadian-induced changes and those from de novo synthesis may similarly be used to exert control of memory formation. More broadly, we hypothesize there is a bidirectional relationship between sex hormone action and clock gene expression that may exist within memory-relevant brain regions to synergistically modulate memory based on internal and external cues.
Defining Sex Hormones and Their Mode of Action in the SCN
Circadian Control of Sex Hormone Production
There are two main classes of steroid hormones. The first are steroid hormones involved in the stress response, such as corticosterone and aldosterone. The second, which will be the focus of this review, are sex hormones, which are classically defined as regulating reproductive functions. The three main types of sex steroid hormones are progestins, estrogens, and androgens. It is important to note that because of the close chemical relationship of these steroid hormones, many serve as precursors for the production of other steroid hormones. The structural similarity of these molecules has made researching their independent functions challenging. For example, progesterone is the precursor of most androgens and androgens are necessary precursors for the synthesis of estrogens, making it extremely difficult to know whether a given hormone influences a behavior or brain function directly or whether its primary function occurs indirectly via a downstream metabolite. Thus, it is important to consider any off-target or indirect effects that could be occurring when studying the action of a given sex hormone.
Progestins and estrogens were first described for their role in generating female-specific appearance, behaviors, and brain development and have been well-studied for their role in ensuring reproductive success. Similarly, androgens have been classically categorized for their essential role in male development and reproduction. Interestingly, despite sex hormones having sex-specific roles in developmental and reproductive periods, the functions of sex hormones within the brain are not always sex-specific. For example, estrogens are known to enhance memory formation in both male and female rodents, but there are prominent sex differences in the molecular and cellular mechanisms that underlie these hormonal actions (Jain et al., 2019; Koss et al., 2018).
Across the animal kingdom, the circadian system plays a critical role in regulating reproduction. In females, this influence is especially important, as cyclic changes in sex hormone levels throughout the body control sexual receptivity and activity at the physiological and behavioral levels (for review see Blaustein et al., 1994; McEwen et al., 1987). Extensive research in female rodents has characterized how time-of-day output from the SCN directly modulates the female reproductive system (for review see Miller et al., 2014). Summarized briefly, gonadotropin-releasing hormone (GnRH) is released from GnRH neuron terminals in the mediobasal hypothalamus. The release of GnRH induces a surge of luteinizing hormone (LH), coined the LH surge (for review see Tonsfeldt et al., 2022), and follicle-stimulating hormone (FSH), from the anterior pituitary into circulation. In both males and females, the gonads dynamically respond to changing levels of LH and FSH by modulating rates of gametogenesis and sex steroid production across the day-night and reproductive cycle. Sex steroid hormones, likewise, negatively or positively feedback onto neurons in the hypothalamus to control LH and FSH release (for review see Wang and Moenter, 2020). Positive feedback is mediated by kisspeptin neurons in the rostral hypothalamus, whereas negative feedback is modulated by both kisspeptin neurons in the caudal hypothalamus and gonadotropin-inhibitory neurons (Angelopoulou et al., 2021; Chassard et al., 2015; Gibson et al., 2008; Smith et al., 2005; Williams et al., 2011). Interestingly, both of these feedback loops, positive and negative, receive input from the SCN (Vida et al., 2010), suggesting the clock is involved in regulating sex hormone concentrations. Thus, there seems to be a bidirectional relationship between the circadian system and sex hormone function. We predict that this relationship is not unique to the central circadian pacemaker but is also present in memory-relevant brain regions.
In female rodents, the LH surge is released from the anterior pituitary specifically during the late afternoon or early evening of proestrus (Legan and Karsch, 1975). Interestingly, rats treated chronically with estradiol will have an LH surge on multiple successive days, but this surge is always restricted to the late afternoon, indicating that the circadian system controls reproductive timing on a stereotyped timescale (Legan and Karsch, 1975). Unsurprisingly, deletions of core clock components can alter reproductive timing. For example, female mice with inhibited Bmal1 expression are deficient in both LH and FSH release during proestrus, as well as being infertile (Chu et al., 2013; Ratajczak et al., 2009). Within males, testosterone synthesis is known to fluctuate across the 24-h day, as LH release stimulates a peak of testosterone production in adult male rodents at 8:00 a.m. and a trough of production between 7:00 p.m. and 9:00 p.m. (Lucas and Eleftheriou, 1980). Furthermore, Bmal1 knockout male mice have low testosterone levels, high LH levels, and are infertile, again showing the interconnectedness of the circadian system and steroidogenesis.
Sex Hormone Receptors in the SCN
Most commonly, the SCN is classified into two main subregions which are defined largely by the inputs they receive (van den Pol, 1980; van den Pol and Tsujimoto, 1985). The first subregion is the core, which receives the greatest density of retinal inputs (Lokshin et al., 2015), and is identified by its vasoactive intestinal polypeptide- and gastrin-releasing peptide-containing neurons (Moore et al., 2002). The other is the shell, which sparsely receives retinal input and has a high density of arginine vasopressin–expressing neurons (Abrahamson and Moore, 2001). Androgen receptors have been identified in a variety of species including humans (Fernandez-Guasti et al., 2000; Kruijver and Swaab, 2002). Within rodents, androgen receptors are localized to the core region of the SCN and are sensitive to light stimulation (Karatsoreos et al., 2007). Unlike androgen receptors, estrogen receptors are expressed in both the shell and core of the SCN, with one type of receptor, estrogen receptor-β being expressed more frequently than estrogen receptor-α (for review see Hatcher et al., 2020; Yao and Silver, 2022). Progestin receptors are also present in the SCN, but very little has been characterized about their localization (Kruijver and Swaab, 2002).
It is important to recognize that most studies reporting sex differences come from more recent literature, since traditionally most neuroscience investigations have primarily focused on males. In fact, female subjects were included in less than 20% of circadian studies before 2013 (Kuljis et al., 2013). One exception in which sex differences have been clearly investigated is anatomical work measuring the distribution of androgen and estrogen receptors within the SCN. Androgen receptors in the SCN are normally expressed at much higher levels in intact males than in intact females. Interestingly, androgen treatment in orchiectomized (ORX) male or intact female mice increases androgen receptor concentration (Iwahana et al., 2008). In the human SCN, there are also sex differences in ovarian hormone receptors since estrogen receptor-α expression is higher among women, whereas there are no sex differences in the expression of estrogen receptor-β or progesterone receptors (Kruijver and Swaab, 2002). Sex differences in receptor distribution may explain why several circadian genes are differentially expressed between males and females, such as the increased Bmal1 expression observed in the central and medial amygdala in female rats compared to males (Chun et al., 2015). Evidence from the studies above should encourage future research to investigate both the biological basis for sex differences in sex hormone receptor distribution and differences in the molecular response to receptor activation, which influences the maintenance of rhythmic clock gene expression.
Evidence for Sex Hormone Control of the Circadian Clock
Traditionally, research on the intersection between sex hormones and the circadian cycle has focused on the implications for organismal development, sexual behavior, and reproduction. Much less is known about the influence that sex hormones have on circadian regulators or how these interactions might alter downstream functions like memory formation, despite numerous potential commonalities between these systems. At the molecular level, sex hormones interact with at least two circadian gene families, Per and Cry, within the SCN (Table 1). At the cellular level, sex hormones affect the SCN’s functioning similar to how they are known to act in memory-relevant brain regions, for example, altering firing activity (Fatehi and Fatehi-Hassanabad, 2008) and synapse formation (Karatsoreos et al., 2011). The presence of sex hormone receptors within the SCN suggests that these pacemaker neurons are directly modulated by sex hormone signaling (Blattner and Mahoney, 2014, 2015; Iwahana et al., 2008; Karatsoreos et al., 2007). The following sections will highlight the current state of knowledge about the bidirectional interactions of clock gene expression and sex hormone action.
Summary of sex hormone effects on clock genes expression in rodents.
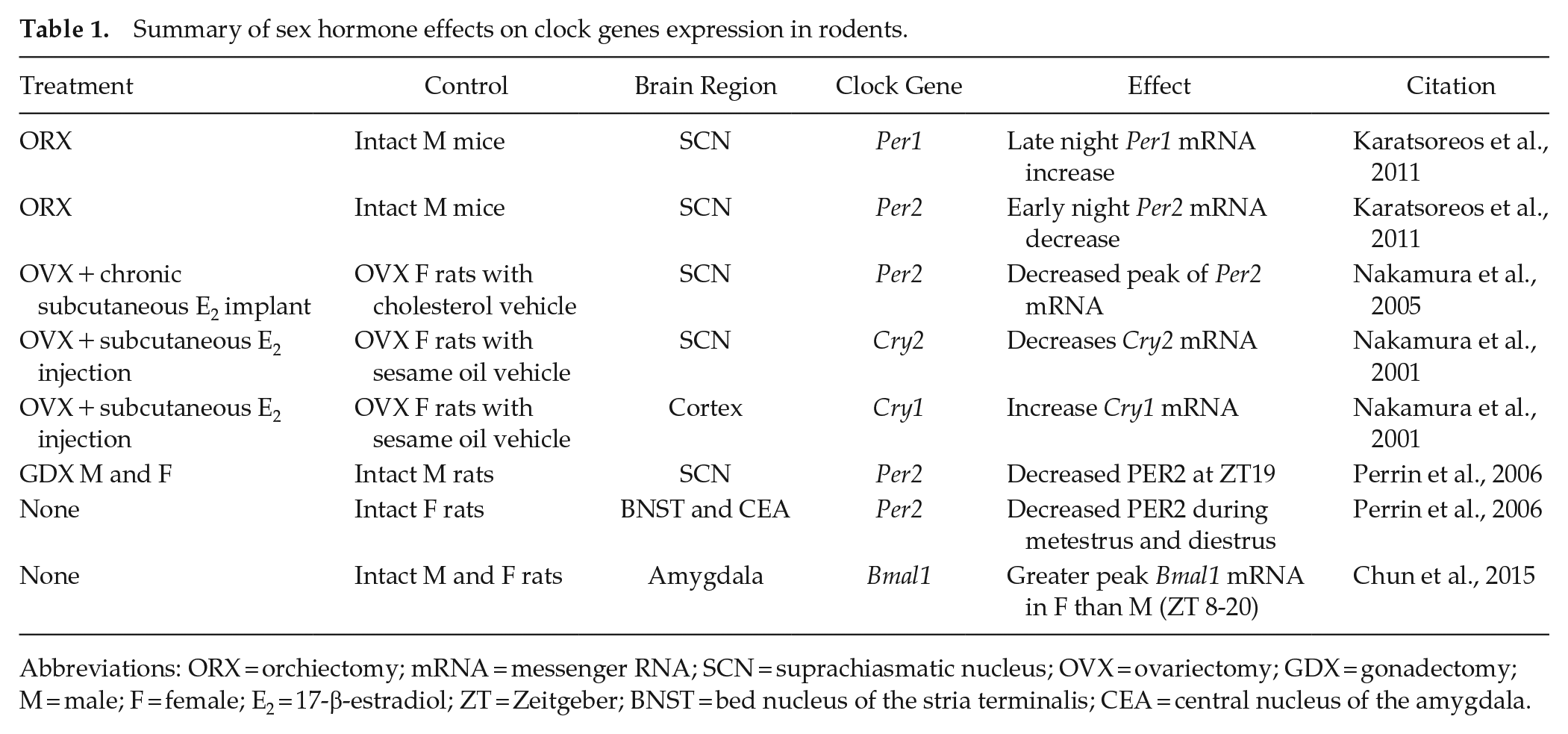
Abbreviations: ORX = orchiectomy; mRNA = messenger RNA; SCN = suprachiasmatic nucleus; OVX = ovariectomy; GDX = gonadectomy; M = male; F = female; E2 = 17-β-estradiol; ZT = Zeitgeber; BNST = bed nucleus of the stria terminalis; CEA = central nucleus of the amygdala.
Androgenic Control of the Circadian Clock
The most common method for studying the role of androgens in males is the use of orchiectomy to remove the testes, the major sites of male androgen synthesis. Experimenters can then systemically or intracranially administer exogenous infusions of testosterone or dihydrotestosterone, a nonaromatizable form of testosterone (i.e. a form that cannot be converted into estrogens). At the molecular level, reducing systemic concentrations of androgens through ORX in mice changes circadian clock gene oscillations in the SCN, reducing Per2 expression in the early night and increasing Per1 expression in the late night in response to a light pulse (Karatsoreos et al., 2011). Androgen receptor expression in the SCN of mammals is sensitive to changing androgen levels in either sex, indicating that the central circadian pacemaker is acutely sensitive to fluctuations in androgen levels. Consistent with this idea, hypothalamic implants of testosterone in ORX male mice increase AR expression in the SCN and lengthen free-running activity rhythms (Model et al., 2015). Neuronal structure and activity are also modulated by androgens in the SCN. Orchiectomized male mice show increased glial fibrillary acid proteins and decreased synaptophysin expression, suggesting changes to both the structure and function of the SCN in response to the reduction of sex hormone production (Karatsoreos et al., 2011). Neuronal firing in the SCN of rodents is modulated by testosterone application, as assessed by electrophysiological recordings on multiple hypothalamic nuclei (Jansen et al., 1993; Orsini, 1981). At the behavioral level, the frequency of activity bouts in ORX male mice is dramatically reduced but can be restored with the addition of exogenous testosterone (Iwahana et al., 2008; Morin and Cummings, 1981). Although the exact mechanisms of androgen action on the SCN are not known, the examples cited above suggest that manipulation of androgen concentrations can alter the circadian system at multiple levels.
Ovarian Hormone Control of the Circadian Clock
The estrous cycle in rodents is defined by fluctuations of estrogens and progestins across a roughly 4- to 5-day cycle, and these fluctuations drive direct changes in clock gene expression in the brain. Early evidence suggests a reciprocal interaction between ovarian hormones and the clock through observation of “scalloping” behavior. Scalloping was first described as rodents showing a phase advance in wheel running behavior during days of estrous, when estradiol levels are highest (Albers et al., 1981; Morin et al., 1977). Since activity phase is known to be driven by the SCN, where there is a documented presence of estrogen receptors in humans and rodents (Kruijver and Swaab, 2002; Vida et al., 2008), these studies suggest that ovarian hormone signaling in the SCN can impact SCN function. More recent research has suggested that clock gene expression is sensitive to estrous phase (Perrin et al., 2006), most likely due to changes in ovarian hormone concentrations. For example, daily Per2 expression in the bed nucleus of the stria terminalis and central nucleus of the amygdala is blunted during metestrus and diestrus, suggesting that ovarian hormones can modulate clock genes in brain structures outside the SCN (Perrin et al., 2006). Naturally occurring fluctuations in estrogens and progestins may therefore have region-specific impacts on clock gene expression outside the SCN, although the functional significance of these changes is not yet clear.
To study the effects of estrogen and progestin levels on female physiology in a more controlled setting, investigators use ovariectomized (OVX) females in which bilateral removal of the ovaries drastically reduces circulating levels of these hormones. Ovarian hormones, such as progesterone or the most potent form of estrogen, 17β-estradiol (E2), can then be administered systemically or intracranially to temporally control their reintroduction to the system. Compared to androgenic action in the SCN, much less is known about the role progestins and estrogens have in changing circadian gene expression. Work in the field of environmental toxicology has shown that chronic progesterone treatment in male and female zebrafish can dramatically change expression of several notable circadian genes, including members of the Per and Cry family within whole brain extracts (Zhao et al., 2015), suggesting there is a relationship between progesterone and clock gene expression within neural tissue. Furthermore, tissue explants from the SCN of female rats treated with progesterone or a combination of progesterone and E2 had significantly longer Per1 expression compared to explants from mice treated with just E2 or cyclo-β-dextran, suggesting a specific role for progesterone in modulating clock gene expression (Murphy et al., 2013). For estrogens, systemic injection of E2 leads to significantly lower Cry2 mRNA expression in the SCN of OVX rats (Nakamura et al., 2001). The same study found that Cry1 mRNA expression is increased in the cortex in response to E2 administration, suggesting that estrogens can differentially alter clock gene expression in a site-specific manner (Nakamura et al., 2001). Likewise, chronic systemic E2 administration in OVX rats significantly advances the peak of Per2 mRNA expression in the SCN (Nakamura et al., 2005). Subcutaneous E2 implants can increase both immediate early gene expression and cAMP response element–binding protein (CREB) phosphorylation in the SCN, implying that more than just circadian genes may be altered in response to estrogens (Abizaid et al., 2004). As we will review later, CREB is a notable molecular intersection between the circadian cycle and sex hormone signaling. Interestingly, CREB initiates Per1 transcription independently of the canonical TTFL that induces Per1 transcription via the E-box element (Travnickova-Bendova et al., 2002). This secondary mechanism for transcriptional control of Per1 may enable it to function in roles outside of the circadian system, including in long-term memory formation across a variety of memory-relevant brain regions (Bellfy et al., 2023; Jilg et al., 2010; Kwapis et al., 2018; Rawashdeh et al., 2014; Urban et al., 2021; Woodruff et al., 2018). The SCN, as well as many of its direct afferent targets, expresses both estrogen and progestin receptors, indicating that ovarian hormones likely act through multiple brain regions to regulate circadian gene expression and circadian behavior. Neurotransmission within the SCN is also impacted by estrogens, as bath application of E2 significantly increases the spontaneous firing frequency of neurons in the SCN of intact male rats (Fatehi and Fatehi-Hassanabad, 2008). Thus, estrogenic signaling is regulated by circadian factors to support normal behavior in both female and male animals.
Sex Hormones Modulate Learning and Memory
A considerable literature has amassed over the past four decades demonstrating the importance of sex hormones in regulating memory formation (for review see Garcia et al., 2018; Hsu et al., 2021; Spritzer et al., 2021; Taxier et al., 2020). Within memory-relevant brain regions, sex hormones induce rapid biochemical changes that influence cell-signaling cascades (for review see Frick et al., 2020), changes in gene expression (for review see Fortress and Frick, 2014), neuron morphology (for review see Murakami et al., 2018), and neuronal excitability (for review see Ooishi et al., 2012; Smith and Woolley, 2004). This review will focus on the role of sex hormones in memory consolidation, since this phase of memory is intricately linked to dynamic changes in gene expression, including the transcription of clock genes (Bellfy et al., 2023; Guzowski et al., 2000; Hall et al., 2000; Linnarsson et al., 1997; Rawashdeh et al., 2014). The consolidation phase of memory is a common target for sex hormone manipulations because sex hormones can be manipulated immediately after memory acquisition to affect consolidation without interfering with the learning event itself. In one of the first studies to test for rapid sex hormone action during memory consolidation, a single post-training intraperitoneal injection of E2 into gonadally intact male rats immediately after Morris water maze training drove enhanced spatial memory, as measured 24 h later (Packard et al., 1996). Follow-up studies in OVX rats showed similar post-training spatial memory enhancements after systemic E2 injection or dorsal hippocampal E2 infusion (Packard and Teather, 1997a, 1997b). Since these seminal studies, researchers have replicated E2’s ability to enhance memory consolidation among both sexes in the Morris water maze and a variety of other behavioral tasks such as context fear conditioning, object placement, and object recognition (Chen et al., 2014; Fernandez et al., 2008; Frye and Walf, 2008; Gresack and Frick, 2006; Inagaki et al., 2010; Jacome et al., 2016; Luine et al., 2003; Lynch et al., 2014; Vedder et al., 2013). Similarly, sex hormones have been shown to enhance memory in multiple memory-relevant brain regions, including the medial prefrontal cortex, amygdala, perirhinal cortex, and hippocampus (Fernandez et al., 2008; Gervais et al., 2013; Koss and Frick, 2019; Maeng et al., 2017; Tuscher et al., 2019). In contrast, the loss of sex hormones through gonadectomy or pharmacological manipulations has been linked to changes in neuron structure and function that typically restricts an animal’s ability to form memory (Aubele et al., 2008; Bredemann and McMahon, 2014; Koss and Frick, 2019; Zhao et al., 2018; Zhou et al., 2010). In general, sex hormone action within memory-relevant brain regions is known to impact several systems necessary for memory formation ranging from genomic changes to neuron structure and activity (for review see Baudry et al., 2013; Colciago et al., 2015; Luine and Frankfurt, 2020).
General Sex Hormone Presence in Memory-Relevant Brain Regions
As with the SCN, sex hormone receptors are commonly expressed in memory-relevant brain regions. The presence of these receptors has fueled much research into the genomic and nongenomic signaling that sex hormones can have in neurons, some of which will be reviewed below. As the presence and characterization of these receptors in memory-relevant brain regions has already been thoroughly reviewed (Brinton et al., 2008; Sarkey et al., 2008; Torres-Reveron et al., 2020), here we will only briefly detail receptor expression in the rodent hippocampus, a focal point for studying the intersection between memory formation and sex hormones. The hippocampus expresses receptors for all the major sex hormones: estrogens, progestins, and androgens. There are 3 types of estrogen receptors expressed in the hippocampus. The first 2 types of receptors, estrogen receptor-α and estrogen receptor-β, can be either nuclear or extranuclear (Milner et al., 2001, 2005). When located on a neuron’s membrane, estrogen receptor-α and estrogen receptor-β commonly colocalize with G-protein-coupled receptors to modulate second messenger signaling (Boulware et al., 2013; Hart et al., 2001), and ultimately memory formation. The third type of estrogen receptor is G-protein-coupled estrogen receptor 1 (Brailoiu et al., 2007), which has been linked to memory formation among other brain functions (for review see Hadjimarkou and Vasudevan, 2018). Progestin receptors are classically defined as ligand-activated transcription factors, which bind progesterone (or other ligands) and undergo confirmational changes that allow them to interact with progestin response elements on the promoter of target genes (Edwards et al., 1991; Guerra-Araiza et al., 2001). Some progestin receptor subtypes have nongenomic mechanisms, such as activation of second messenger signaling cascades (Nilsen and Diaz Brinton, 2003). Androgen receptors are also known to function inside or outside the nucleus (Kerr et al., 1995; Tabori et al., 2005). Interestingly, testosterone is not the only ligand to bind to and activate these receptors; many testosterone metabolites, such as dihydrotestosterone, actually have a higher binding affinity for these receptors than testosterone itself (Grino et al., 1990). Therefore, a diverse set of sex hormone receptors are expressed within the hippocampus, offering a range of possible interactions between sex hormones and memory formation.
Evidence for Sex Hormone Action in Memory-Relevant Brain Regions
Numerous studies have demonstrated that sex hormone administration either systemically or directly infused via cannulation into memory-relevant brain regions can enhance memory consolidation in both male and female rodents (Babanejad et al., 2012; Fernandez et al., 2008; Koss et al., 2018; Orr et al., 2012; Packard et al., 1996; Tuscher et al., 2019). Although both sexes typically show overarching memory enhancements with sex hormone administration, many sex differences have been identified in the cellular and molecular mechanisms that support these memory improvements, including male-female differences in receptor distribution, neuronal excitability, and spine density (for review see Bauer, 2023; Frick et al., 2015). Sex, hormonal stage, and age-related differences in sex hormone receptor distributions could all contribute to changes in neuronal activity and therefore memory consolidation, making this a complex process. For example, E2 potentiates hippocampal glutamatergic synaptic transmission via different estrogen receptor mechanisms in ORX male and OVX female rats (Jain and Woolley, 2023; Oberlander and Woolley, 2017). Some memory-relevant mechanisms induced by sex hormones are identical in both sexes, like the changes in hippocampal spine density and dendritic structure observed following removal of circulating sex steroid hormones in rats (Mendell et al., 2017). Female CA1 dendritic spines are so sensitive to changing ovarian hormones that spine density varies across the estrous cycle (Gould et al., 1990; Kato et al., 2013; Woolley et al., 1990). Sex hormone effects on spine density are unique in that all 3 steroid hormones have been tested to see how their application affects neuron morphology. Progesterone, testosterone, and E2 all increase spine density in hippocampal neurons, though there are some differences in sex of the model and cell type studied (Gould et al., 1990; Hatanaka et al., 2015; Jacome et al., 2016; Kretz et al., 2004). Follow-up studies found that E2 infused directly into the dorsal hippocampus increased spine density not only in the CA1 region but also in the medial prefrontal cortex (Tuscher et al., 2016). Interestingly, inhibition of extracellular signal–regulated kinase (ERK) phosphorylation and mammalian target of rapamycin (mTOR) activation is necessary for the E2-induced spinogenesis in either the dorsal hippocampus or medial prefrontal cortex, suggesting that estrogens increase spine density via changes in transcription and translation in multiple memory-relevant brain regions (Tuscher et al., 2016). There are many more examples of sex hormones inducing rapid changes to cell-signaling cascades, which we will discuss in the next section.
Induction of Cell-Signaling Cascades and Downstream Gene Expression in Memory Consolidation
The most well-studied mechanisms underlying sex hormone regulation of memory consolidation involve rapid activation of dorsal hippocampus cell-signaling cascades. As described previously, sex hormones can impact circadian gene expression, so it is not surprising that sex hormones can alter memory-relevant gene expression as well. Though androgen and progestin signaling can induce cell-signaling cascades, E2 has been the most thoroughly investigated sex hormone in terms of sex hormone signaling during memory consolidation. In both young and middle-aged female mice, estrogen-dependent memory consolidation for both object placement and object recognition tasks relies on rapid activation of cell-signaling cascades, including the ERK signaling cascade (Koss et al., 2018; Tuscher et al., 2016), phosphatidylinositol-3 (PI3K) (Fan et al., 2010), protein kinase A (PKA) (Lewis et al., 2008), and mTOR pathways (Fortress et al., 2013). Activation of ERK is a key regulatory point that can activate many downstream cell-signaling molecules. In the female hippocampus, phosphorylated ERK activates CREB, a transcription factor that is critical for both long-term memory formation (Boulware et al., 2005) and clock gene expression (Frank and Greenberg, 1994; Lee et al., 2010). Hippocampal CREB phosphorylation shows rhythmic diurnal oscillations, with peak activation occurring during the day (Rawashdeh et al., 2016), again implicating CREB’s activity being controlled by the circadian clock.
Interestingly, CREB activation can be regulated by either estrogens or androgens (Boulware et al., 2005; Nguyen et al., 2009), implying that it is a key regulatory mechanism for gene expression in either sex. Moreover, the memory-enhancing effects of E2 in ORX or gonadally intact male mice do not depend on ERK activation but may be associated with CREB phosphorylation and are mediated by androgen receptors (Koss and Frick, 2019). Therefore, the ultimate activation of CREB offers a compelling potential interface between sex hormones, clock gene expression, and memory formation (Figure 2).
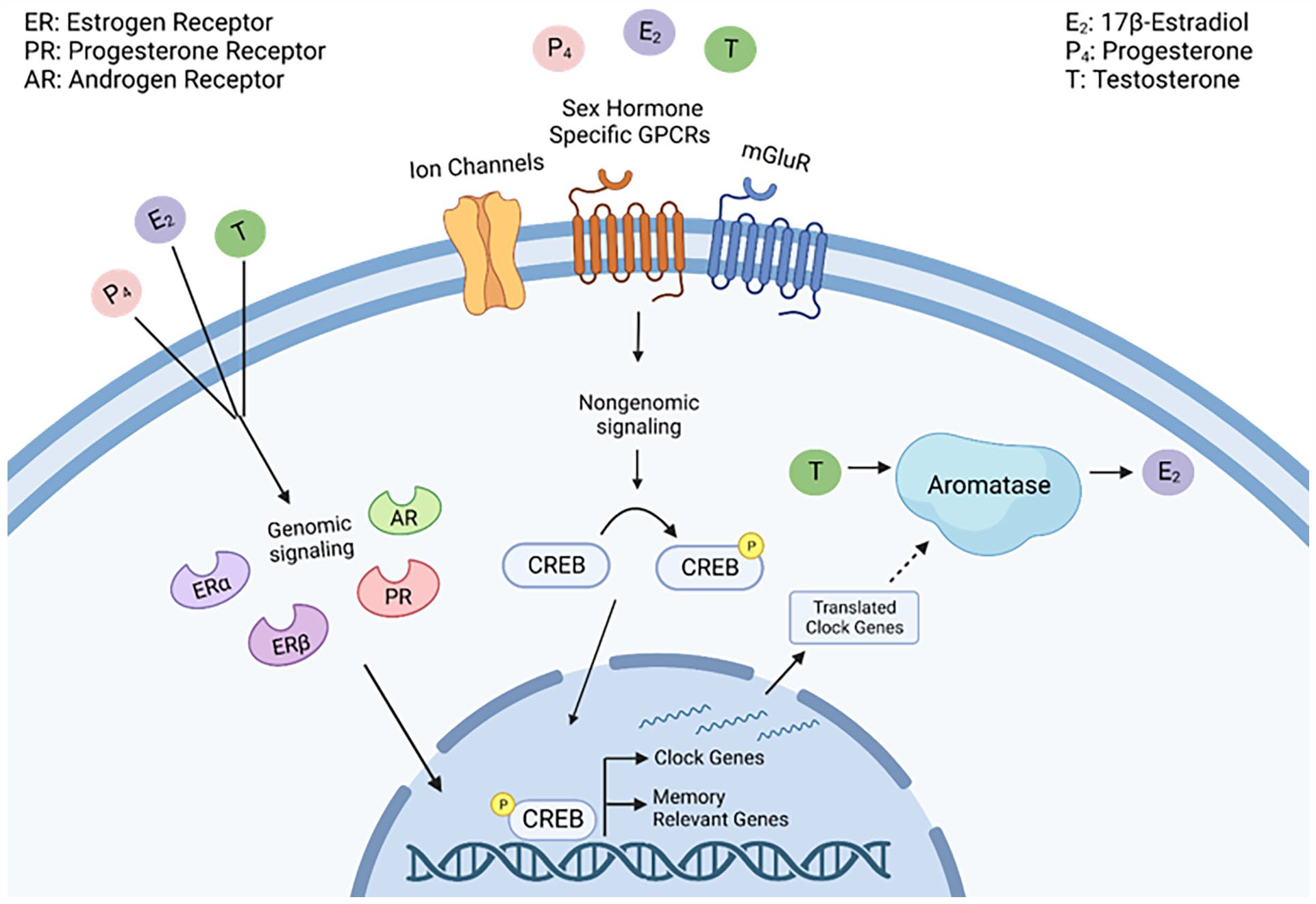
Proposed intersection between sex hormones and clock genes in memory-relevant brain areas. Intracellular processes are initiated by genomic and nongenomic action of steroid sex hormones (17β-estradiol [E2], progesterone [P4], or testosterone [T]). Nongenomic signaling occurs through binding to corresponding G-protein-coupled receptors (GPCRs), metabotropic glutamate receptors (mGluRs), or ligand-gated ion channels. Activation of these receptors initiates sex-specific intracellular signaling cascades to induce cyclic AMP response element–binding protein (CREB) phosphorylation. Activated CREB can then drive transcription of memory-relevant genes and clock genes. Likewise, steroid sex hormone signaling can occur genomically when sex hormones bind to and activate intracellular hormone receptors, such as androgen receptors (AR), progestin receptors (PR), and estrogen receptors (ER). Activation of intracellular sex hormone receptors causes a confirmational change which allows them to be trafficked to the nucleus to modulate gene expression by binding to hormone response elements on DNA. We hypothesize that an increase of clock gene protein products in the cytoplasm then upregulates aromatase activity to induce the conversion of T to E2. Dotted lines denote hypothetical pathways. Created with BioRender.com.
Sex Hormones Regulate Chromatin-Modifying Enzymes
Another potential interface between sex hormones, the biological clock, and memory are epigenetic mechanisms, factors that change gene expression by modulating chromatin structure rather than affecting the underlying DNA sequence itself. Most notably, histone acetylation, a major epigenetic mechanism important for memory consolidation (Barrett et al., 2011; Kwapis et al., 2017; McQuown et al., 2011; Stefanko et al., 2009), is both modulated by E2 and is downstream of the ERK signaling cascade initiated during memory formation (Zhao et al., 2010). Estrogens depend upon ERK activation to increase histone acetyltransferase activity and decrease histone deacetylase (HDAC) activity during memory consolidation (Fortress et al., 2014; Zhao et al., 2012). Sex hormones likely play a key role in regulating learning-induced transcription, as E2 infusions decrease the expression of repressive HDACs in the hippocampus and increase H3 acetylation at Bdnf, a memory-relevant gene (Fortress et al., 2014). Interestingly, clock genes may be regulated by learning-induced changes in histone acetylation. One recent study found that learning increases histone acetylation at the promoter of the clock gene Per1 (Kwapis et al., 2018), which coincides with increased Per1 mRNA expression. Learning-induced changes in histone acetylation may therefore provide an interface between cell-signaling cascades and downstream changes in clock gene expression, possibly serving as a mechanism through which the circadian system can exert local diurnal control over the transcriptional program needed for long-term memory (Kwapis et al., 2018; Rawashdeh et al., 2016). Thus, sex hormones can regulate chromatin modifying enzymes to modulate clock gene expression and ultimately memory formation, serving as a second potential interface between the clock, hormones, and memory.
Role of Brain-Derived Sex Hormone Synthesis on Memory Formation
Beyond the systemic release of sex steroid hormones, de novo brain-derived hormone production has been shown to be intricately linked to memory formation in both sexes. The enzyme aromatase converts testosterone into estrogens within different tissue types, including memory-relevant parts of the brain (Hojo et al., 2004; Prange-Kiel et al., 2003). Several species including rodents, birds, and humans have all been found to have aromatase located in memory-relevant brain regions (Azcoitia et al., 2011; Bailey and Saldanha, 2015; Ivanova and Beyer, 2000), suggesting that rapid E2 synthesis may be important for memory formation. Since males and females produce estrogens at differing levels and on differing oscillatory cycles, unsurprisingly, sex differences have been found in the reliance of animals on systemic versus de novo E2 synthesis. In female mice, E2 levels fluctuate across the estrous cycle, yet remain detectable in the hippocampus after OVX, suggesting de novo E2 synthesis in this brain region (Kato et al., 2013). This local estrogen synthesis is critical for memory (Kretz et al., 2004), though the mechanisms which control aromatase activity and therefore de novo E2 synthesis rates remain poorly understood.
Inhibiting aromatase activity in the dorsal hippocampus impairs memory consolidation in object location memory, object recognition, and the Morris water maze in OVX and ORX mice (Koss and Frick, 2019; Zhao et al., 2018). Likewise, spine density (Kretz et al., 2004), synapse formation (Prange-Kiel and Rune, 2006), and long-term potentiation (Vierk et al., 2012) are all negatively impacted by aromatase inhibition. The extent to which males and females rely on gonadal versus brain-derived sources of sex hormones remains an active area of investigation, and it is currently unclear how local or global sex hormones interact with the clock. We predict that local synthesis is critically important and expect there is likely an interaction between clock gene expression and aromatase activity (Figure 2). Despite not fully understanding how aromatase activity is controlled, recent work suggests autocrine signaling is important in determining its activity level (Fester et al., 2016). Because both hormone levels and clock genes oscillate across stereotyped cycles, there could be a bidirectional relationship between these mechanisms, part of which may include controlling aromatase’s catalytic activity. We hypothesize that systemically circulating sex hormones induce genomic and nongenomic signaling following learning to activate transcription factors and chromatin-modifying enzymes, which, in turn, increases clock gene expression in memory-relevant brain regions. Local increases in clock gene expression within memory-relevant brain regions could then drive the enzymatic activity of aromatase to influence de novo E2 synthesis, which could subsequently enhance memory formation in neuronal populations necessary for memory storage.
Intersection Between Sex Hormones and Circadian Regulators in Memory-Relevant Brain Regions
Despite these clear links between sex hormones and the circadian clock, how clock-hormone interactions might synergistically regulate local functions like memory is currently unclear. Recent work has suggested that clock genes may modulate memory formation directly within memory-relevant brain regions, suggesting that clock gene oscillations and their modulation via sex hormone action may play a key role in regulating memory. Notably, hippocampal memory is tightly controlled by the circadian system, with memory formation oscillating over the 24-h day/night cycle in both animal models and humans (Bellfy et al., 2023; Chaudhury and Colwell, 2002; Eckel-Mahan et al., 2008; Evans et al., 2017; Goode et al., 2022; Groeger et al., 2008; Harrison et al., 2007; Rawashdeh et al., 2014). Furthermore, long-term memory requires expression of at least one of the core clock components, Per1 (Rawashdeh et al., 2016), and bidirectional manipulation of Per1 in the hippocampus can modulate memory; local knockdown in young mice impairs spatial memory, whereas overexpression in old mice ameliorates age-related memory impairments (Kwapis et al., 2018). Thus, Per1 and other clock genes may locally function within memory-relevant brain regions to exert diurnal control over memory processes. Interestingly, the exact role of clock genes in this process may be sex-specific, as Per1 overexpression in the retrosplenial cortex impairs context fear memory in males but has no effect in females (Urban et al., 2021). This suggests there may be a critical interaction between circadian gene expression and sex hormone action during memory formation.
Perhaps the most convincing argument for a potential bidirectional relationship between clock genes and sex hormones comes from work at the molecular level, in which the transcription factor CREB interfaces with both Per1 (a core component of the molecular clock) and the signaling cascade necessary for estrogens’ enhancement of long-term memory (Figure 1). Known rhythmic phosphorylation patterns in CREB activity may be due to stereotyped fluctuations in sex hormone production, either over a 24-h cycle (e.g. testosterone in males) or over multiple days (e.g. the estrous cycle in females). Hormonal fluctuations could therefore change CREB activity, modulating memory consolidation based on hormonal status (Figure 2). Likewise, clock genes may directly or indirectly influence the activity of aromatase to regulate a rhythmic or memory-relevant production of estrogens which, in turn, can feedback onto CREB phosphorylation. Although there is little evidence to document what mechanisms might control local aromatase activity, we hypothesize that clock genes, like Per1, may be involved in regulating de novo E2 synthesis. This is plausible, as a bidirectional relationship between PER1 and CREB across the day/night cycle has already been established (Rawashdeh et al., 2016). Thus, the circadian system can modulate the CREB-mediated transcriptional program necessary for both memory consolidation and local E2 synthesis.
It is likely that sex hormones and clock genes work in conjunction to control memory formation across many biological processes seasonally, throughout reproductive cycles, or in response to aging. Memory-relevant brain regions both express sex hormone receptors and are known to rely on clock gene expression, enabling memory to be modulated by both time-of-day and hormonal information. Although previous work has shown that sex hormones can regulate clock gene expression within the SCN and on circadian behavior (Table 1), it remains unclear whether this is also true in memory-relevant brain regions during memory formation. Current data suggest that hormones and clock genes each affect memory independently, yet it remains unclear how these processes work together to synergistically regulate memory formation. Our laboratories aim to expand upon the hypotheses presented here with the hopes of gaining valuable insight into the bidirectional relationship of clock gene expression and sex hormone action within memory-relevant brain regions.
Conclusion
Active research into the sex hormone and circadian interface is quite relevant to human health, as both systems contribute to multiple human diseases and disorders, such as Alzheimer’s (Fox et al., 2013), insomnia (Zhang et al., 2009), and infertility (Williams and Kriegsfeld, 2012). Disrupting either the circadian system or sex hormone release from the endocrine system can lead to impairments in memory and cognition (Cho et al., 2000; Gibson et al., 2010; Gresack and Frick, 2006; Li et al., 2022; Loh et al., 2010) and biological sex plays an important role in the underlying mechanisms that cause sleep disorders, of which women are more likely to be affected (Mallampalli and Carter, 2014). Similarly, acute or chronic shifts in estrogenic signaling due to pregnancy, menopause, or reproductive disorders are known to increase the likelihood of circadian rhythm disruption and impaired memory performance (Daniel, 2013; Mahoney, 2010; Shechter and Boivin, 2010; Sherwin and Henry, 2008). It is therefore critical to understand how these major modulatory systems interact to control memory formation in a synergistic and sex-specific manner.
Footnotes
Acknowledgements
This work was funded by NIH grants R01AG074041 (J.L.K.), K99/R00AG056586 (J.L.K.), R21AG068444 (J.L.K.), 01MH107886 (K.M.F), and T325T32GM125592-03 (H.M.B). We are additionally funded through the Whitehall Foundation Grant #2020-05-06 (J.L.K) and start-up funds from the Eberly College of Science and Department of Biology at Pennsylvania State University (J.L.K.).
Conflict of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
