Abstract
With time-place learning, animals link a stimulus with the location and the time of day. This ability may optimize resource localization and predator avoidance in daily changing environments. Time-place learning is a suitable task to study the interaction of the circadian system and memory. Previously, we showed that time-place learning in mice depends on the circadian system and Cry1 and/or Cry2 clock genes. We questioned whether time-place learning is Cry specific or also depends on other core molecular clock genes. Here, we show that Per1/Per2 double mutant mice, despite their arrhythmic phenotype, acquire time-place learning similar to wild-type mice. As well as an established role in circadian rhythms, Per genes have also been implicated in the formation and storage of memory. We found no deficiencies in short-term spatial working memory in Per mutant mice compared to wild-type mice. Moreover, both Per mutant and wild-type mice showed similar long-term memory for contextual features of a paradigm (a mild foot shock), measured in trained mice after a 2-month nontesting interval. In contrast, time-place associations were lost in both wild-type and mutant mice after these 2 months, suggesting a lack of maintained long-term memory storage for this type of information. Taken together, Cry-dependent time-place learning does not require Per genes, and Per mutant mice showed no PER-specific short-term or long-term memory deficiencies. These results limit the functional role of Per clock genes in the circadian regulation of time-place learning and memory.
Time-place discrimination or time-place learning (TPL) refers to the ability to anticipate and exploit resources or avoid hazardous situations that display spatiotemporal regularity. Most organisms follow daily programs in physiology and behavior. Guiding future behavior by encoding when and where biologically significant events occurred in the past will lead to a significant advantage in natural selection. This is strengthened by the fact that TPL has been shown in many species, including insects (Gould, 1987; Harrison and Breed, 1987), fish (Reebs, 1996), birds (Krebs and Biebach, 1989), and mammals (Mistlberger et al., 1996; Van der Zee et al., 2008).
Gallistel (1990) stated that animals automatically remember not only the nature of biologically significant events but also the location and time of day at which the events occurred and that memory must be organized in such a way to encode these factors. Another prerequisite for TPL is that animals must have a sense of time. Most organisms have evolved circadian systems to time internal physiology and behavior in daily cycles (Van der Zee et al., 2009). In mammals, the suprachiasmatic nucleus (SCN) is recognized as the “master clock” in the brain. The SCN oscillator synchronizes to the environmental light/dark (LD) cycle and in turn synchronizes many subordinate clocks in the brain and periphery (Dibner et al., 2010). On a cellular/molecular level, circadian rhythms are predominantly generated by clock genes and their protein products, forming a transcriptional-translational feedback loop. In short, CLOCK (circadian locomotor output cycles kaput) and BMAL1 (brain and muscle ARNT-like protein 1) form a heterodimeric complex, which acts as a transcription activator for PER (Period) and CRY (Cryptochrome) proteins, which in turn inhibit CLOCK-BMAL1 transcription (Ko and Takahashi, 2006).
Previously, we introduced a novel paradigm for mice to study TPL in laboratory settings (Van der Zee et al., 2008). This paradigm reflects the natural situation in which foraging animals have to evaluate risks connected with feeding locations, depending on the time of day. We showed that wild-type mice learned the paradigm using a circadian clock (rather than external cue–based strategies) and that Cry1 and Cry2 double knockout mice were unable to learn the task. These results confirmed the use of a circadian clock and have highlighted TPL as a unique paradigm to study the participation of the circadian system in cognition (i.e., decision making and episodic-like memory formation) and as a model to study the role of clock genes on a functional behavioral level.
Clock genes have recently been implicated in cyclical processes underlying long-term memory (LTM) formation and maintenance (Gerstner et al., 2009; Gerstner and Yin, 2010; Kondratova et al., 2010). Altered LTM in Per mutant animals has been demonstrated in Drosophila (Sakai et al., 2004) and mice (Jilg et al., 2010; Wang et al., 2009). Here, we set out to investigate if TPL is solely Cry gene specific or also requires Per clock genes and whether Per genes play a role in memory formation in the context of TPL.
Materials And Methods
Animals and Housing
Experiments were performed using 4-month-old male mPer1Brdm1/Brdm1;mPer2Brdm1/Brdm1 homozygotic double mutant mice (N = 9 in total), backcrossed 11 times to generate a full C57BL6/J genetic background, 5 generations before the cohort used in this study. The Per1 and Per2 mutations are considered null mutations. Generation of these mutant mice has been described before (Zheng et al., 1999, 2001). In short, mutated alleles were obtained in AB2.2 embryonic stem cells (derived from an XY 129s7 embryo) and used to generate chimeric mice. Intercrosses between heterozygous (C57BL/6Tyrc-Brd × 129S7) F1 offspring gave rise to F2 homozygous mutants. The mPer1Brdm1/Brdm1 and mPer2Brdm1/Brdm1 mutant mice were crossed to obtain double mutant mice, hereafter referred to as Per mutant mice. Per mutant mice, together with age-matched male C57BL/6JOlaHsd wild-type mice (N = 8) (Harlan, Horst, the Netherlands), were housed individually in Macrolon type II cages (Bayer, Germany; length of 35 cm × width of 15 cm × height of 13.5 cm) with sawdust as bedding and shredded cardboard as nesting material. The mice were kept in a climate room with controlled temperature (22 °C ± 1 °C) and humidity (55% ± 10%). An LD schedule (12-h light:12-h dark; light onset at 0700 h GMT + 1 h) was maintained, except in the constant light (LL) period. A light intensity of 20 lux was used, as measured from the top of the cages. Food (standard rodent chow, RMHB/ 2180, Arie Block BV, Woerden, the Netherlands) was available ad libitum, except during food deprivation. Normal tap water was available ad libitum. Cages were cleaned at least once every 2 weeks. All efforts were made to minimize animal suffering and to reduce the number of animals used. All procedures were in accordance with the regulation of the ethical committee for the use of experimental animals of the University of Groningen, the Netherlands (license no. DEC 5583A).
Activity Recording
Cages were enriched with a plastic running wheel (diameter = 13.5 cm). Activity, measured by running-wheel revolutions, was recorded continuously throughout the experiment. Revolutions were counted per 2 min and processed into actograms and average daily activity profiles using ACTOVIEW for Microsoft Excel (written by C. Mulder, University of Groningen, the Netherlands).
Phenotyping and Genotyping
Per mutant mice were phenotyped based on arrhythmic running-wheel behavior over a 12-day period of housing in constant darkness (DD). In addition, wild-type and mutant genotypes were confirmed by PCR. The Per1 and Per2 mutations were detected in 2 separate PCRs. The PCR mixtures contained the following: 6.5 µL H2O, 12.5 µL master mix containing the necessary polymerase enzymes and dNTP (HotStar, Qiagen, Venlo, the Netherlands), 2.5 µL forward primers (Per1: 5′-ACA AAC TCA CAG AGC CCA TCC-3′; Per2: 5′-GCT GGT CCA GCT TCA TCA ACC-3′), 1.25 µL reverse primers (Per1: 5′-ATA TTC CTG GTT AGC TGT AGG-3′; Per2: 5′-GAA CAC ATC CTC ATT CAA AGG-3′), 1.25 µL PKG hprt-reverse primers as the positive mutation marker (5′-CGC ATG CTC CAG ACT GCC TTG-3′), and 1 µL sample DNA. Mixtures were incubated for 15 min at 94 °C and then cycled 35 times for 30 min at 94 °C, 30 min at 56 °C, and 1 min at 72 °C, followed by 10 min at 72 °C. The PCR products were run on a 1% ethidium-stained agarose gel together with a molecular weight marker (1-kb DNA ladder, O’GeneRuler, Fermentas, Burlington, ON, Canada). For Per1, the wild-type genotype was indicated by a Per1-forward/Per1-reverse band at approximately 450 bp; the mutation was indicated by a Per1-forward/PKG hprt-reverse band at approximately 287 bp. For Per2, the wild-type genotype was indicated by a Per2-forward/Per2-reverse band at approximately 381 bp; the mutation was indicated by a Per2-forward/PKG hprt-reverse band at approximately 120 bp.
Spontaneous Alternation (SA) Test
Short-term spatial working memory was assessed by recording SA behavior in a Y-maze paradigm prior to TPL testing. Nine Per mutant mice and 8 wild-type mice, naïve to the maze and matched for age and body weight, were tested. The Y maze consisted of 3 tubular and transparent Plexiglas (Evonik Industries AG, Germany) arms forming the Y. All 3 arms were 4.4 cm in internal diameter, 29 cm long, and at a 120° angle from each other. The experimental room contained visual cues, which served as distal spatial cues. Mice (naïve to the maze) were placed in the center of the Y maze (5-cm internal diameter) and allowed to explore the maze freely during an 8-min session. The series of arm entries was recorded visually. An arm entry was considered to be completed when all 4 paws of the animal had entered a Y-maze arm. The maze was cleaned with paper towels and water between each test. An alternation is defined as successive entries into 3 different Y-maze arms. The alternation percentage (SA performance) was calculated as the ratio of actual to possible alternations, where the possible alternations are defined as the total number of arm entries minus 2 (Anisman, 1975). The number of arm entries was used as a measure for general exploratory behavior.
TPL Apparatus
The TPL test apparatus consisted of a round middle chamber (diameter = 12.5 cm; height = 7 cm) made of gray polyvinyl chloride (PVC) and a transparent Plexiglas lid with ventilation holes. Four round tubes (arms) made of transparent Plexiglas (length = 24.5 cm; inside diameter = 4.5 cm) were connected to this middle chamber. One bottom arm served as a starting point to which a small transport cage could be connected, from which the mice could enter the maze. The other 3 upper arms (60° apart) were equipped with a small food platform (gray PVC; 1.0 × 4.5 cm) and a steel grid (6 × 3.5 cm) at the end, through which a mild but aversive electric foot shock (set to 620 V [0.09 mA]) could be delivered. In order to reach the food in the baited arms, the mice had to step onto the grid. The electric current could be manually delivered to each grid by pressing a button. This device was handmade at the University of Groningen (see Fig. 1 in Van der Zee et al. [2008]).
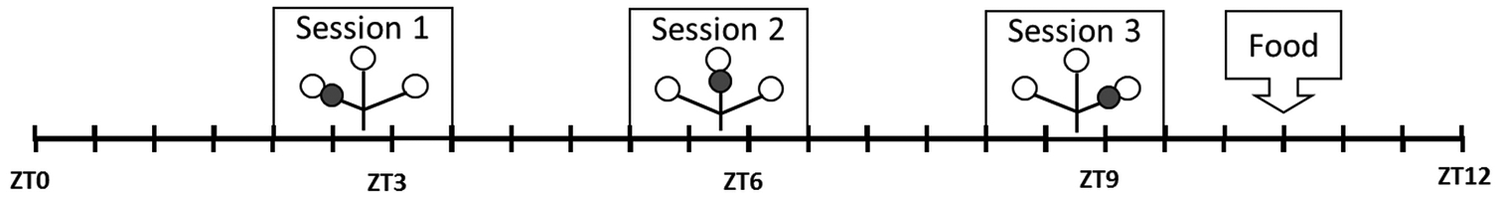
Schematic overview of the daily TPL testing protocol. Mice were tested individually 3 times a day in the light phase (ZT0 indicates light onset), with a 3-h intersession time. Symbols represent the maze: open circles indicate food at the end of an arm of the maze, and gray circles indicate the nontarget (shock) location. Mice received an individual amount of food at the end of each day to regulate food deprivation. Each unit on the scale represents 30 min.
TPL Testing Procedure
Testing procedures have been described before (Van der Zee et al., 2008). Briefly, in each of the 3 daily sessions (lasting maximally 10 min per mouse), mice had to learn to avoid 1 of the 3 feeding locations, depending on the time of day (i.e., session). On visiting the nontarget location, mice received a mild but aversive foot shock (<1 s). To induce food-seeking behavior and voluntary location choices, mice were food deprived to 85% of their ad libitum body weight, as individually determined by the average of 3 daily measurements prior to food deprivation. Mice were weighed before each session and received an individual amount of food at the end of each light phase to maintain food deprivation. A session was considered correct, on an individual level, only when the 2 target locations were visited first, avoiding the nontarget location or visiting it last. Daily performance was calculated for each animal as the percentage of correct sessions (e.g., 0%, 33%, 67%, or 100%), and these performances were averaged and plotted per genotype group, forming a learning curve over multiple testing days. Actual testing was preceded by habituation steps as described previously (see Van der Zee et al. [2008]). In short, target locations were always baited. During the first 4 days, all 3 locations were baited and safe to explore freely. During the next 4 days, the nontarget location was kept unbaited but still safe to visit (no foot shock delivery). During the next 3 days, the shock was introduced at the nontarget location, while still kept unbaited, so that mice could identify the nontarget location based on sight and smell. On day 9, mice were first habituated to first-time foot shock exposure. The nontarget location was kept inaccessible until the mice had first consumed the food rewards in the 2 target locations. This way, in each session, all mice received both the positive food experience, followed by the negative foot shock experience. Because of this manipulation, day 9 was excluded from further analysis. After these habituation steps, actual testing started with all locations baited and foot shock delivery in the nontarget location. A schematic overview of the daily protocol is provided in Figure 1. Four wild-type mice and 4 Per mutant mice were tested. Groups were matched for age, body weight, and SA performance (short-term spatial memory capability).
Experimental Outline
After 10 days of habituation to the climate room and housing conditions, animals were phenotyped during a 12-day DD period. After 2 weeks back on LD, the SA test was performed. Body weights, to determine ad libitum values, were measured on the day of the SA test and the 2 following days. Food deprivation was started on the same day as the last ad libitum body weight measurement, after the measurement was taken. TPL testing was started the next day. Animals were tested daily during 47 days, starting with 11 days of habituation steps, followed by 23 days of testing in LD and then by 13 days of testing in LL. Session skipping was performed on the following days, with the number in parentheses indicating which session was skipped: 22 (2), 24 (1), 29 (2), 32 (1), 36 (1), 39 (2), 41 (3), 43 (1, 2, 3), and 45 (3). On day 43, all sessions were skipped (nontesting day). At the end of the last testing day, animals were put back on an ad libitum food diet but remained on LL for another 2 weeks before being returned to LD. Two months after the initial period of TPL testing, the same animals were again food deprived and retested for 8 days (6 days in LD and 2 days in LL; habituation steps omitted). Food deprivation was started 3 days before retesting, so animals were at least food deprived to below 90% of the ad libitum body weight at the start of retesting. For each mouse, the number of retreats (incomplete arm entries before the first full entry at any of the 3 locations) was counted as a measure of long-term contextual memory for the potential hazard (foot shock) at the locations. A first session was skipped on day 8 of retesting.
Statistical Analysis
Statistical analyses were performed using GraphPad Prism 5.01 (GraphPad Software Inc., La Jolla, CA) and Statistix 8 (JMP Software, NC, USA). Differences between genotype group averages were assessed by 2-tailed unpaired t tests or 1-way ANOVA. The χ2 test was used to assess location preference. Differences from the chance level were assessed by a 1-sample t test. Differences between genotype groups over multiple testing days were assessed by repeated-measures ANOVA with a Bonferroni posttest. Learning curves were compared by a 2-sample t test of logistic regression slopes. Pre-post differences within genotype groups were assessed by a 1-tailed or a 2-tailed paired t test. p < 0.05 was considered to be significant.
Results
Phenotyping and Genotyping
Arrhythmic phenotypes of Per mutant mice were confirmed by assessing running-wheel behavior in DD. Without the presence of an entraining zeitgeber, wild-type mice show a free-running activity pattern (Fig. 2A), while Per mutant mice immediately become arrhythmic (Fig. 2B). Genotypes were additionally confirmed by genotyping (Fig. 2C and 2D).

Phenotyping and genotyping results. Together with the wild-type mice, Per mutant mice were phenotyped for arrhythmic running-wheel behavior in DD. Shown are representative double-plotted qualitative actograms of a wild-type mouse (A) and a Per mutant mouse (B) over 10 days (x-axis). Activity was counted per 2-min bins. In addition, genotypes of all mice were confirmed by genotyping. The wild-type and/or mutated gene fragments (indicated on the right outer side of the figure) were detected for Per1 (C) and Per2 (D) separately. Shown on both panels, from left to right, are the molecular weight marker, a heterozygous mouse (htz), and a wild-type mouse (wt), followed by all 4 homozygous mutant mice (mutant) used for TPL testing.
SA Test
A failure to achieve TPL can be attributed to both/either an internal timing deficiency and/or a spatial memory deficiency. To exclude the latter, an SA test was performed to assess short-term spatial working memory (by SA performance) and general exploratory behavior (by the number of entries). Results are shown in Figure 3. We found no difference in SA performance between wild-type and Per mutant mice (71.0% ± 3.3% and 76.8% ± 4.3%, respectively) (2-tailed unpaired t test: p = 0.31). Wild-type mice showed slightly, but not significantly, more entries (22.4 ± 2.7 and 17.9 ± 1.6, respectively) (2-tailed unpaired t test: p = 0.16).
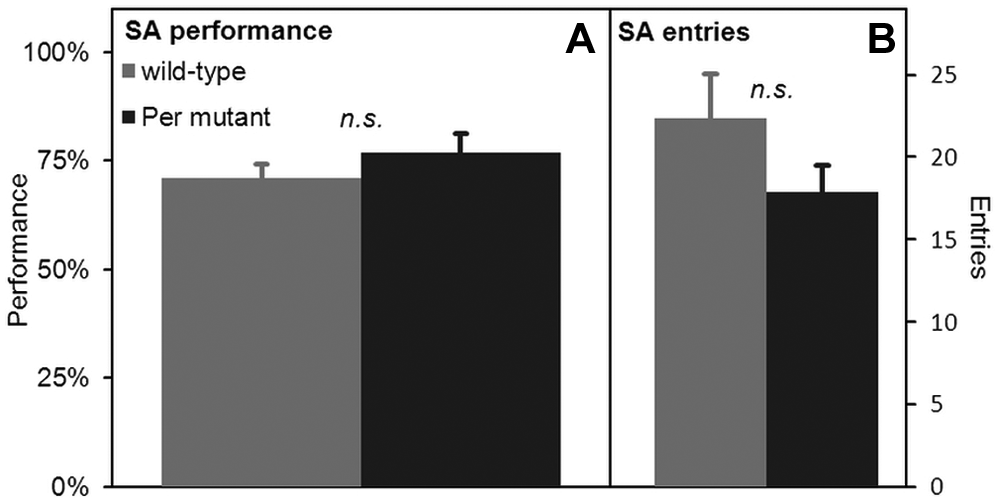
SA results. SA performance (A) and entries (B) of wild-type (N = 8) and Per mutant (N = 9) mice. No statistical differences were found between wild-type and Per mutant mice. Error bars represent SEM.
Habituation to TPL
The results are shown in Figure 4. During the first habituation step (days 1-4), mice could freely explore the 3 baited locations. No significant preference for a single location was found (χ2 test: p = 0.3 and p = 0.6 for wild-type and Per mutant mice, respectively) (data not shown). During the second habituation step (days 5-8; target locations baited, and nontarget location unbaited without foot shock delivery), the performance of both genotypes did not significantly differ from the chance level (1-sample t test: p = 0.08 and p = 0.06 for wild-type and Per mutant mice, respectively). Although, at this step, Per mutant mice showed better performance than wild-type mice, this is likely to be an artifact of the low sample size to which we were bound. Performance typically varies largely during the first 2 habituation steps (mice make random choices) but becomes very congruent once the foot shock is introduced. On day 9, mice were habituated to first-time foot shock exposure (excluded from analyses). During the third habituation step (days 10 and 11; target locations baited, and nontarget location unbaited with foot shock delivery), both genotypes significantly learned to avoid the nontarget location, demonstrating performance significantly above the chance level (1-sample t test for day 11: p = 0.02 and p = 0.01 for wild-type and mutant mice, respectively), with no significant difference between genotypes (2-tailed unpaired t test: p = 0.2).
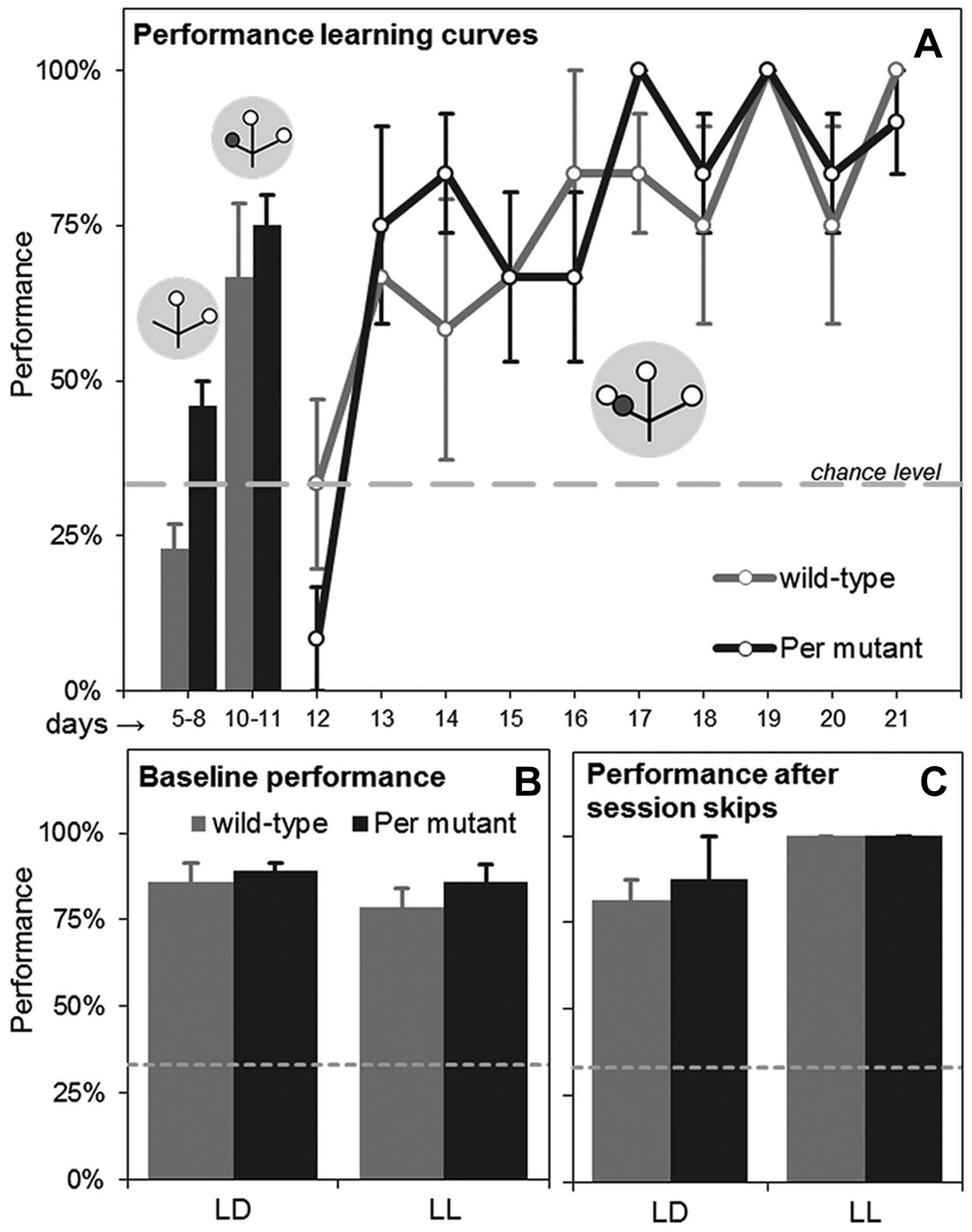
TPL results. (A) Average performances for wild-type and Per mutant mice during the last 2 habituation steps (bar graphs, left) and the first 10 days of TPL testing (learning curve). The gray circular symbols represent the TPL maze. Within, small open circles indicate food at the end of an arm of the maze, and small dark gray circles indicate the nontarget shock location. Note that only the test situations of the first session are depicted. The nontarget location (nonbaited and non–shock reinforced during habituation days 5-8, nonbaited and shock reinforced during habituation days 10 and 11, and baited and shock reinforced during actual testing) changes with the time of day (i.e., session). (B) Baseline TPL performance under LD (average over 14 normal test days, excluding the learning phase and days on which manipulations were performed) and LL (average over 7 normal test days). Testing in LL had no significant effects on performance for either genotype. (C) Session skipping did not reduce performance for either wild-type or Per mutant mice under either LD or LL. Performances were measured in the next (single) session after the skipped session. For LD, the average is shown of the 2 first session skips and 2 second session skips. For LL, the average is shown of 1 first session skip and 1 second session skip. No differences were found between skipping a first or a second session. In all panels, the theoretical chance level is indicated by the gray horizontal broken line at 33%. Error bars represent SEM.
Time-Place Learning
On the experimental days (days 12-47), testing was performed with food in all locations and foot shock delivery in the nontarget location, which changed according to the time of day. Hence, mice had to use the time of day to predict the hazardous nontarget location. Although, initially, performance dropped back to the chance level (day 12) (Fig. 4A), all mice gradually learned to avoid the nontarget location. Per mutant mice performed as well as age-matched wild-type mice (learning curves are shown in Fig. 4A). On average, over all experimental days, Per mutant mice even performed slightly better than wild-type mice (84.6% ± 2.9% SEM v. 81.1% ± 2.8% SEM, respectively) but not significantly (2-tailed unpaired t test: p = 0.40). No significant differences were found between genotypes overall or on any of the experimental days (2-way repeated-measures ANOVA: F = 0.43, df = 1, p = 0.54; Bonferroni posttest: p > 0.05 for all days). Learning curves did not differ between Per mutant and wild-type mice (logistic regression slope: T = −0.14, df = 6, p = 0.90). All individual mice performed significantly above the chance level (average over experimental days) (1-sample t test: p < 0.0001 for each mouse). We found no differences between genotypes in overt behavior.
The potential use of alternative strategies, based on external cues rather than a circadian clock, can be identified by skipping sessions and testing in the absence of an LD cycle (Mulder et al., 2013). Skipping of any session in LD did not affect performance in either wild-type or Per mutant mice (paired t test before v. after session skipping: p = 0.50 and p = 1.00 for wild-type and Per mutant mice, respectively) (Fig. 4C). Performances of both genotypes were also unaffected when animals were tested in LL (Fig. 4B) or when sessions were skipped in LL (paired t test before v. after session skipping: p = 0.39 for both wild-type and mutant mice) (Fig. 4C).
The potential use of an hourglass mechanism, which depends on daily resetting by a zeitgeber, was investigated by a nontesting day in LL. TPL performance, measured on the following day, remained significantly above the chance level (1-sample t test: p = 0.01), with no difference between genotypes (1-way ANOVA: p = 0.99). This excludes the testing procedures and the light cycle as potential zeitgebers. Furthermore, in a follow-up TPL experiment using the same animals as in the current study, mice received no food at the end of a test day. Again, TPL performance, measured on the following day, remained significantly above the chance level (1-sample t test: p = 0.0001), with no difference between genotypes (1-way ANOVA: p = 0.54). This excludes the food provided at the end of testing days as a potential zeitgeber. In the same follow-up study, mice showed no affected performance after a complete nontesting day in LD (no testing and no food provided at the end of the day) (1-sample t test: p = 0.0002), with no difference between genotypes (1-way ANOVA: p = 0.72). This excludes the combination of testing procedures and the food provided at the end of the testing days as potential zeitgebers for an hourglass mechanism.
LTM Assessment
We tested if Per mutant mice show impaired LTM for contextual features of the maze (the mild foot shock) and for the learned time-place associations. After 2 months without training, the previously acquired time-place associations were lost. Both wild-type and Per mutant mice had to relearn the test, with performance starting below the chance level (Fig. 5A).
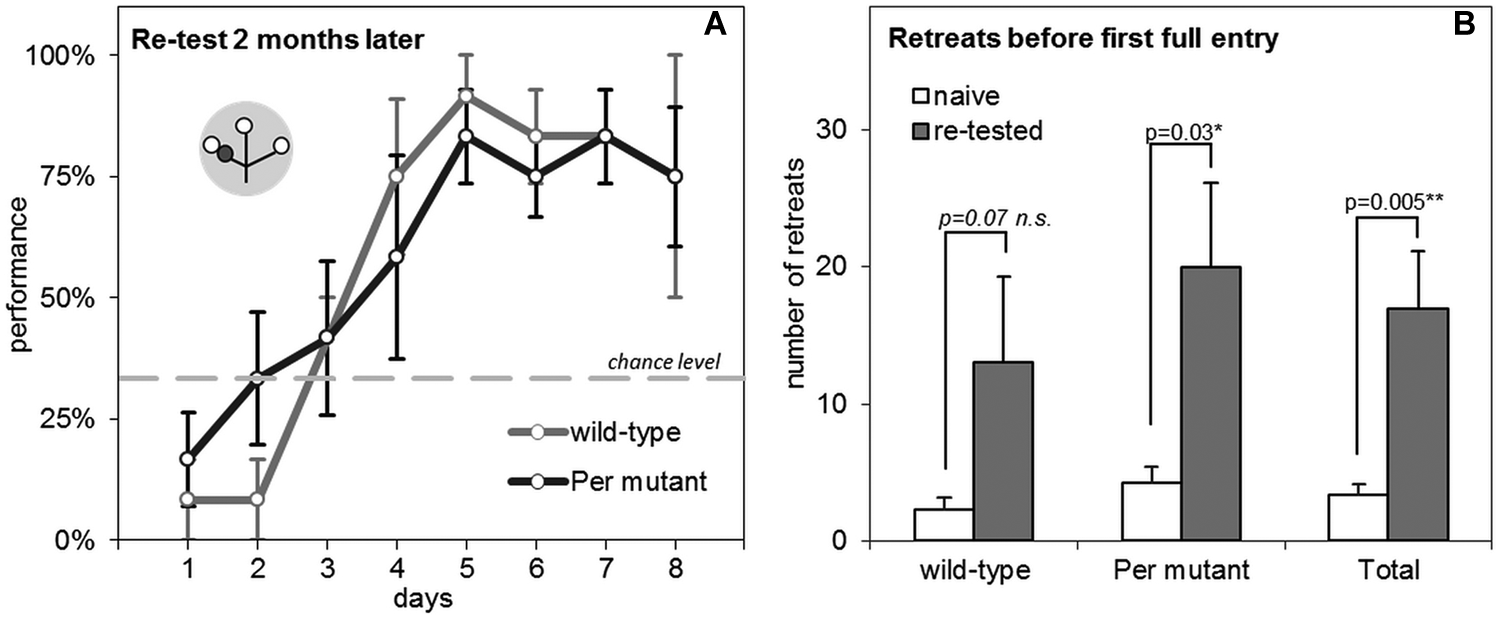
TPL retesting results and LTM assessment. (A) Learning curves of the retest 2 months after initial testing. On days 1 through 6, animals were tested in LD; on days 7 and 8, animals were tested in LL. The theoretical chance level is indicated at 33%. The gray circular symbol represents the TPL maze. Within, small open circles indicate food at the end of an arm of the maze, and small dark gray circles indicate the nontarget (shock) location. Note that only the test situation of the first session is depicted, while the nontarget location changes with the time of day. (B) The number of retreats (incomplete arm entries before the first full entry) of wild-type and Per mutant mice, when the mice were naïve to the maze and at retesting. Error bars represent SEM.
Next, we analyzed LTM for contextual features of the maze. Naïve mice had no reason based on recollection to retreat from an arm entry before going all the way to the food at the end, thereby standing on the shocking grid. However, if mice remembered that standing on the shocking grid could be potentially unsafe, they should show hesitative behavior. This would be reflected by an increased number of retreats before the first full entry (i.e., standing on any grid at the end of any arm). We analyzed the number of retreats in the initial TPL experiment and in the retesting experiment (2 months after the last day of testing in the initial TPL experiment). To exclude positive and negative reinforcement influences by either receiving food or the shock at any location, we only analyzed the number of retreats before the mice received either the food or the shock for the first time in these experiments.
Results are shown in Figure 5B. We found a significant increase in the number of retreats of experienced mice compared to naïve mice (1-tailed paired t test: p = 0.005), but we found no differences between genotypes (average increase ± SEM: 10.8 ± 5.4 v. 15.8 ± 6.2 for wild-type and Per mutant mice, respectively) (2-tailed unpaired t test: p = 0.54). Testing genotypes separately also revealed a significant difference for the Per mutant mice (naïve v. nonnaïve) (1-tailed paired t test: p = 0.03) and a trend for the wild-type mice (p = 0.07).
Analysis of Running-Wheel Activity
Characteristically for their genotype, Per mutant mice were rhythmic in LD (masking) but arrhythmic in LL (last 7 days) (Fig. 6E-H) and DD. Wild-type mice were entrained to the LD cycle and showed free running in DD and LL. Food deprivation (hypocaloric timed feeding) slightly advanced the rhythms of both Per mutant and wild-type animals, with less activity at the end of the dark phase and more activity prior to feeding time (at the end of the light phase) and beginning of the dark phase. This shifting effect can be attributed to food deprivation because wild-type and Per mutant home-cage control mice, which were similarly food deprived, showed the same pattern. During TPL testing and food deprivation in LD, both genotypes showed increased activity prior to feeding time (Figs. 6 and 7). When switched to LL, wild-type animals showed 2 components of their activity rhythm: one 24-h rhythm entrained to feeding time and one free-running rhythm with a period longer than 24 h. Per mutant mice almost entirely shifted all their activity to the light phase, with peak activity occurring prior to feeding time (Figs. 6 and 7). When TPL testing was ended in LL and animals were put back on an ad libitum diet of food, only the free-running rhythm component remained in wild-type animals, while mutant animals immediately became arrhythmic (last 7 days) (Fig. 6).
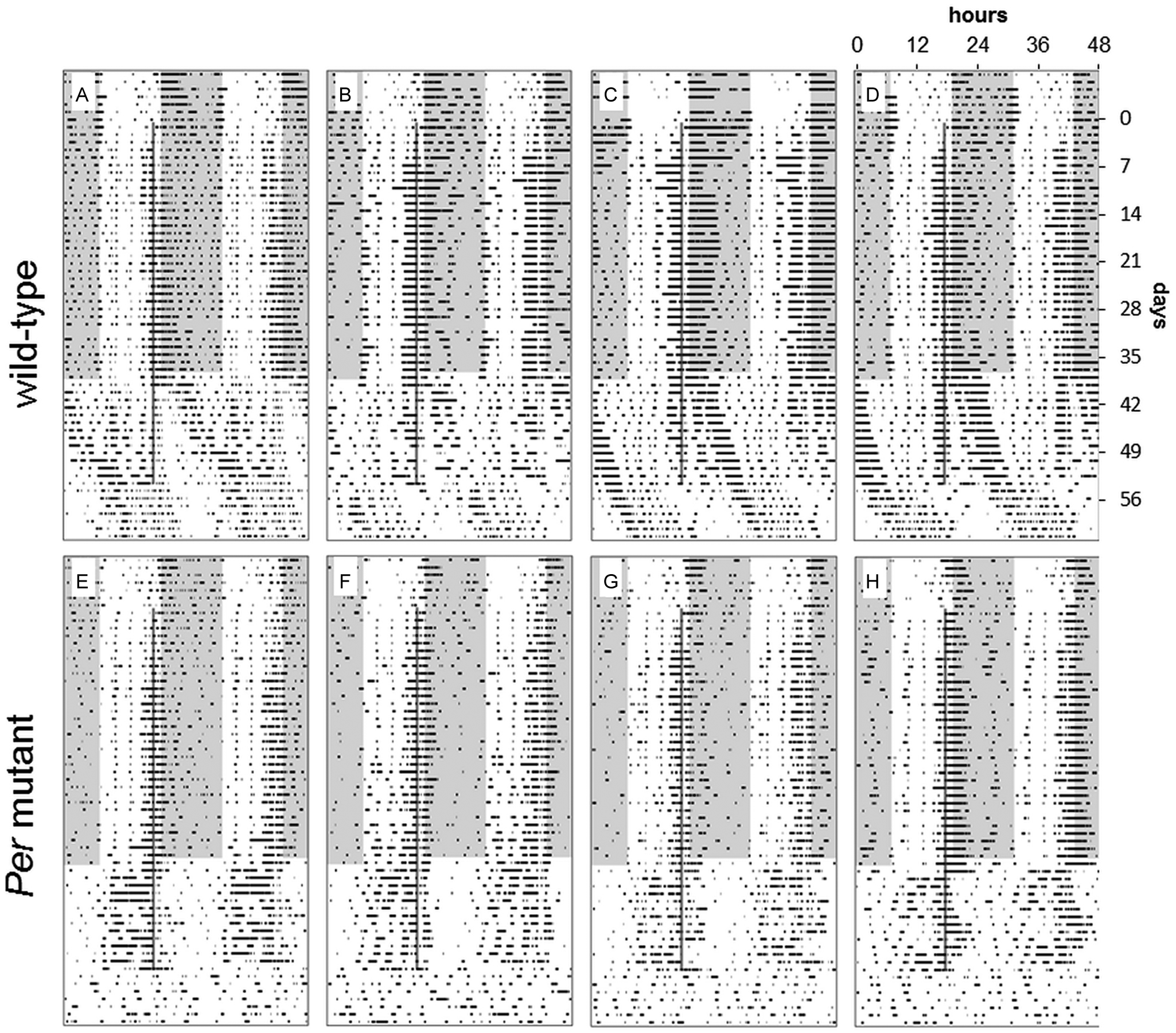
Double-plotted actograms. Food was given 1 h after the last TPL session (indicated in actograms by gray vertical lines). TPL testing started 1 day after the onset of food deprivation, until the last day of food deprivation. Wild-type animals (A-D) showed a free-running rhythm component in LL, while Per mutant mice (E-H) did not. Instead, Per mutant mice showed increased daytime activity in LL and immediate arrhythmic behavior after testing was stopped in LL. Shaded areas indicate darkness.
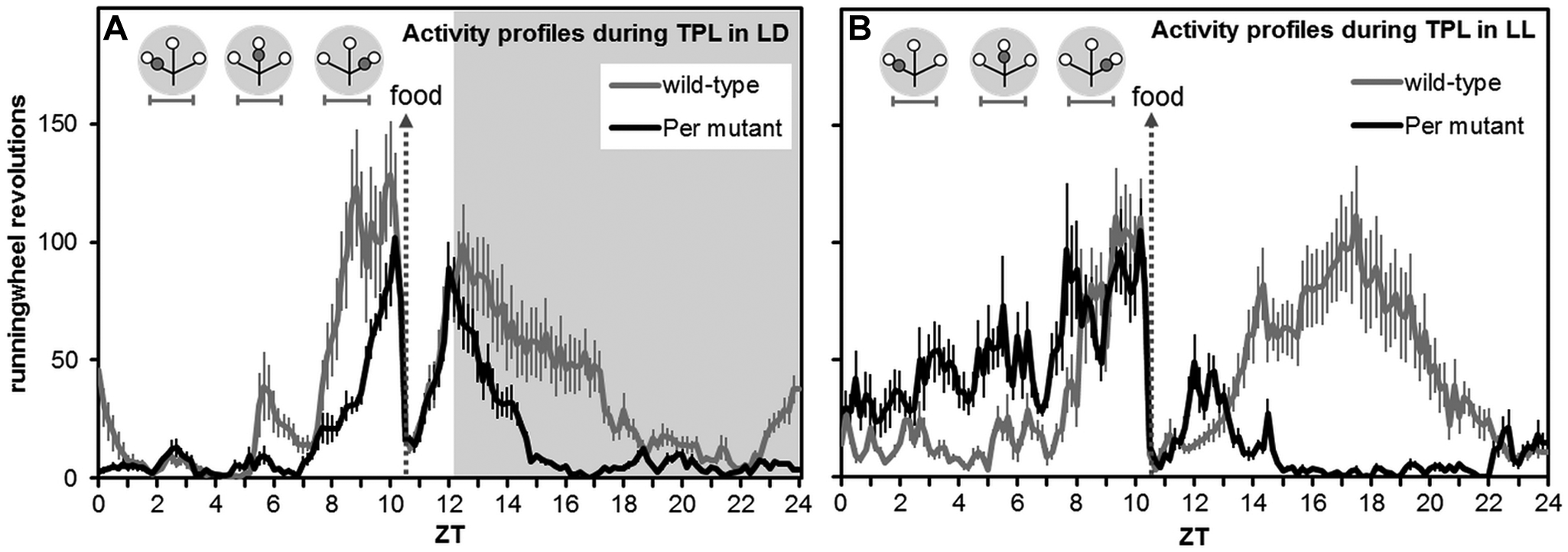
Average activity profiles. (A) Activity profile over all 33 testing days in LD. Zeitgeber time (ZT) is indicated on the horizontal axis. Shaded areas indicate darkness. (B) Activity profile over all 14 testing days in LL. Although a light zeitgeber was absent, the same horizontal axis (ZT) scale was used as in panel A. Per mutant mice showed increased daytime activity. In both panels, the gray vertical arrow indicates when food was provided to manage food deprivation (daily at ZT10.5). Both genotypes show food anticipation. Gray circular symbols represent the daily TPL test session situations. Within, open circles indicate food at the end of an arm of the maze, and gray circles indicate the nontarget (shock) location. Horizontal error bars below the circular symbols indicate session durations. Profiles are plotted in 10-min bins. Error bars represent SEM.
Discussion
Here, we show that Per mutant mice, despite their arrhythmic phenotype, acquire TPL similar to wild-type mice. These results were unexpected given our previous finding that daily TPL depends on intact Crypotochrome (Cry1 and Cry2) clock genes (Van der Zee et al., 2008). Thus, under exactly the same test circumstances, Per mutant mice can acquire TPL, while Cry knockout mice cannot. Apparently, Per mutant mice still exhibit some form of circadian timekeeping functional to TPL. In addition, we found that spatial short-term memory (STM) and contextual LTM are unaffected in Per mutant mice. These findings contribute new insights regarding the role of Per genes in circadian rhythms and memory.
TPL Depends on a Circadian Clock
Several strategies for animals to master a TPL paradigm have been identified in the literature: a stimulus-response strategy, ordinal timing, and interval timing. Alternatively, animals may use a circadian clock, which may be based on an hourglass mechanism or an oscillator (Mulder et al., 2013). The use of a stimulus-response strategy was experimentally ruled out by keeping testing procedures exactly the same for each daily session. With ordinal timing, the animal remembers the sequence of events (e.g., first test session, avoid location A; second test session, avoid location B; etc.). If this strategy is used, then performance will drop to below the chance level after a session is skipped. We found that performance of both genotypes remained intact after session skipping, thus excluding the use of this strategy. With interval timing, an animal tracks or estimates the passage of time relative to a zeitgeber. This strategy may be applied in TPL if animals learn to associate intervals with a specific feeding location (e.g., shortly after light onset [first test session], avoid location A; longer after light onset [second test session], avoid location B; etc.). We investigated if animals use elapsed time after light onset as a timing strategy by testing in LL. Neither the switch to LL nor prolonged testing under LL nor session skipping under LL affected the performance of wild-type or Per mutant mice. These results exclude the possibility that the mice used interval timing. Hence, we can conclude that both genotypes used a circadian clock for TPL. The potential utilization of an hourglass mechanism (a unidirectional process that depends on daily resetting by a zeitgeber) was excluded for all present zeitgebers. Thus, in both genotypes, neither the light cycle nor the testing procedures nor feeding cues are solely responsible for driving an hourglass mechanism. These findings strongly suggest that both genotypes used a circadian oscillator for TPL. Note that this oscillator may still be entrained by any present zeitgeber.
Observations from Activity Data
Just as in the previously tested Cry-deficient mice (Van der Zee et al., 2008), Per mutant mice showed rhythmic activity during TPL testing in both LD and LL. However, these mice immediately became arrhythmic during phenotyping in DD and after the last test day of the initial TPL test period, when the mice remained in LL. Moreover, Per mutant mice became almost entirely diurnal as soon as testing proceeded in LL. The absence of transient cycles in Per mutant mice indicates masking due to the testing procedures rather than a circadian clock, which may have regained functionality because of the regularity of the testing procedures. In contrast, wild-type mice showed a free-running rhythm component during testing in LL, which was not present in the Per mutant mice. These observations support the presence of a dysfunctional circadian clock in Per mutant mice. Another noteworthy observation is that Per mutant mice, like wild-type mice and the previously tested Cry-deficient mice, showed food anticipatory activity (FAA). Although in agreement with other studies (Storch and Weitz, 2009), FAA here was probably influenced by the testing procedures, as mice could have learned to receive food relatively shortly after the last test session. Therefore, FAA was not further analyzed.
A Learning- and Memory-Based Oscillator
The discrepancy in TPL ability between Cry- and Per-deficient mice does not fit known oscillator types. It is well established that the SCN, as a light-entrainable oscillator (LEO), depends on both Per and Cry genes. The current findings thus predict that the SCN is not the circadian oscillator underlying TPL. Indeed, TPL has been shown in SCN-lesioned rats (Mistlberger et al., 1996), although some studies have suggested time-stamp encoding of events on the level of the SCN via vasopressin signaling (Biemans et al., 2003; Hut and Van der Zee, 2011; van der Veen et al., 2008). Besides the LEO, a separate food-entrainable oscillator (FEO) has been identified (Stephan, 2002). Despite many studies, the locus and neural substrates of the FEO have not been conclusively identified (Mistlberger, 2011). The current theory is that the FEO is composed of a network of interconnected brain structures, likely also involving (non–entrainment based) memory (Carneiro and Araujo, 2009; Silver et al., 2011). While some studies have found FAA deficiencies in both Cry- and Per-deficient mice (Feillet et al., 2006; Iijima et al., 2005), others have concluded that FAA is independent of known clock genes (Honma and Honma, 2009; Storch and Weitz, 2009). A separate methamphetamine-sensitive circadian oscillator (MASCO) has also been identified and has been generalized as an arousal-based oscillator, which could underlie TPL. However, it has been shown that the MASCO does not require the SCN or known clock genes (Masubuchi et al., 2001; Mohawk et al., 2009). Although we cannot definitely rule out any LEO and/or FEO mechanisms, the current data suggest a different type of oscillator that functions in the domain of learning and memory and depends on Cry, but not Per, genes. This is plausible, as it has become clear over recent years that clock genes can have tissue-specific functions. Moreover, a critical role for PER proteins as negative regulators in the core molecular clock mechanism is debatable. PER proteins only modestly inhibit CLOCK:BMAL1-mediated transcription (Jin et al., 1999; Kume et al., 1999; Sangoram et al., 1998). CRY proteins, on the other hand, are potent inhibitors of CLOCK:BMAL1-stimulated transcription, and their activity as negative regulators was shown not to require PER proteins (Griffin et al., 1999; Kume et al., 1999; Shearman et al., 2000b). Taken together, while critical for light entrainment, PER proteins may not be critical in all oscillators in the brain and periphery, as advocated by the current findings.
The Role of PER Proteins in Learning and Memory
Clock genes have been found to be rhythmically expressed in many brain areas, including those involved in learning and memory. Circadian rhythms modulate the formation, consolidation, and retention of memory (Chaudhury and Colwell, 2002; Devan et al., 2001; Eckel-Mahan et al., 2008; Fekete et al., 1985; Fernandez et al., 2003; Holloway and Wansley, 1973; Lyons et al., 2006). Recently, it has been proposed that similar molecular mechanisms may underlie both circadian oscillations and behavioral plasticity (Gerstner and Yin, 2010; Wang et al., 2009). In Drosophila, PER proteins were found to play a key role in LTM (but not STM) formation (Sakai et al., 2004). In Per1 knockout mice, the expression of hippocampal clock genes was found to be disordered, and these mice showed deficiencies in hippocampus-dependent long-term spatial learning (Jilg et al., 2010). Likewise, hippocampal long-term potentiation (cellular correlate of LTM) was found to be abnormal in Per2 mutant mice, and these mice showed deficits in the recall of trace (but not cued) fear conditioning (Wang et al., 2009). However, no deficiencies were found in Per1 or Per2 mutant mice in a water-maze place navigation task and in contextual fear conditioning (Zueger et al., 2006). Here, we add to these results and report that Per mutant mice do not show deficits in TPL, in the SA test for short-term spatial working memory, or in long-term contextual memory. The latter demonstrates memory storage in Per mutant (and wild-type) mice over an extended period of 2 months. In contrast, time-place associations were lost after 2 months in both wild-type and Per mutant mice and took a similar duration of training to reacquire. Apparently, these associations are not stably stored in LTM, which is most likely adaptive.
Paralog Compensation
Per mutant mice carry mutated Per1 and Per2 genes but still have functional Per3 genes. Under normal circumstances, Per3 is not essential in circadian rhythms (Shearman et al., 2000a). Genetic network analysis showed, however, that Per3 expression is augmented when Per1 (and to a lesser degree, also Per2) is knocked down (Baggs et al., 2009). Although incapable of rescuing behavioral rhythmicity, this paralog compensation mechanism could play a role in rescuing the reduced functionality of Per1 and Per2 in learning- and memory-specific functions. Whether such mechanisms actually occur in Per mutant mice and whether Per3 can play a compensatory role in TPL remain to be confirmed in future studies.
Conclusion
We found no mutation-specific short-term memory or LTM deficiencies in Per mutant mice. Taken together with other literature, clock genes may only play a role in specific types of learning and memory. We have shown that Cry-dependent TPL is independent of Per genes. This demonstrates the presence of an intact circadian timekeeping mechanism in Per mutant mice. Although we cannot definitely rule out compensatory mechanisms in Per mutant mice, these data predict the existence of an oscillator that functions in the domain of learning and memory, requiring Cry but not Per genes. The question remains: which circadian system is critically involved in TPL? That of the SCN or those in other brain regions such as the hippocampus?
Footnotes
Acknowledgements
The authors thank Wanda Douwenga for help with the genotyping and Emma Wams for critically reading the article.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
