Abstract
The cockroach, Leucophaea maderae, can be trained in an associative olfactory memory task by either classical or operant conditioning. When trained by classical conditioning, memory formation is regulated by a circadian clock, but once the memory is formed, it can be recalled at any circadian time. In contrast, when trained via operant conditioning, animals can learn the task at any circadian phase, but the ability to recall the long-term memory is tied to the phase of training. The optic lobes of the cockroach contain a circadian clock that drives circadian rhythms of locomotor activity, mating behavior, sensitivity of the compound eye to light, and the sensitivity of olfactory receptors in the antennae. To evaluate the role of the optic lobes in regulating learning and memory processes, the authors examined the effects of surgical ablation of the optic lobes on memory formation in classical conditioning and memory recall following operant conditioning. The effect of optic lobe ablation was to “rescue” the deficit in memory acquisition at a time the animals normally cannot learn and “rescue” the animal’s ability to recall a memory formed by operant conditioning at a phase where memory was not normally expressed. The results suggested that the optic lobe pacemaker regulates these processes through inhibition at “inappropriate” times of day. As a pharmacological test of this hypothesis, the authors showed that injections of fipronil, an antagonist of GABA and glutamate-activated chloride channels, had the same effects as optic lobe ablation on memory formation and recall. The data suggest that the optic lobes contain the circadian clock(s) that regulate learning and memory processes via inhibition of neural processes in the brain.
Numerous studies have documented the importance of circadian systems in the regulation of learning and memory processes in a diversity of organisms, including mollusks, insects, fish, birds, and mammals (reviewed in Gerstner and Yin, 2010; Mulder et al., 2013; Smarr et al., 2014; Page, 2015). Depending on the species and the training procedure, circadian phase may be a determining factor in the ability to form a new memory, or it may function as a contextual cue that affects the ability to recall a stored memory. Furthermore, disruption of the circadian system can also impair memory formation or memory retention. The data naturally raise the questions about the mechanisms by which the circadian system regulates these processes. A first step is to identify the anatomical location of the circadian oscillators involved and begin to identify the signaling pathways by which the oscillator controls memory formation or recall.
In cockroaches subjected to classical conditioning in an olfactory discrimination task, the ability to form new memories depends on the circadian phase of training while the ability to recall a learned memory is independent of circadian time (Decker et al., 2007). In contrast, animals trained to a similar task via an operant conditioning protocol can learn and consolidate the memory at any time of day. However, the ability to recall the memory is tied to the circadian phase of training such that the ability to recall recurs at 24-h intervals after training (Garren et al., 2013). A variety of circadian rhythms in the cockroach are regulated by a circadian pacemaking system located in the optic lobes of the brain, including locomotor activity (Nishiitsutsuji-Uwo and Pittendrigh, 1968; Page, 1982; Homberg et al., 2003), sensitivity of the compound eyes (Wills et al., 1985), mating behavior (Rymer et al., 2007), and sensitivity of olfactory receptors in the antennae (Page and Koelling, 2003; Saifullah and Page, 2009). Thus the optic lobes are a prime candidate for the location of the clock that controls learning and memory rhythms. Here we show that ablation of the optic lobes “rescues” circadian deficits of olfactory memory formation following classical conditioning and deficits in memory retrieval following operant conditioning. Furthermore, we show that an antagonist of GABA and glutamate-activated chloride channels will also rescue the learning and memory rhythms. The results suggest that a circadian clock(s) in the optic lobes provides the phase information for the regulation of learning and memory processes and that the clock acts through the inhibition of the processes at the appropriate time.
Materials and Methods
Animals
Colonies of Leucophaea maderae (also known as Rhyparobia maderae) were maintained in 12-h light/dark cycles (LD 12:12). Prior to testing, males were isolated in groups of 8 in a circular, plastic chamber that was 24 cm in diameter without food or water 1 week prior to the pretraining test. After the 7 days of isolation, the animals began the pretraining test to determine their initial peppermint preferences and remained in constant darkness for the remainder of testing.
Training/Testing Procedures
Training and testing procedures were adopted from Decker et al. (2007) and Garren et al. (2013). Naïve L. maderae show a strong preference toward vanilla when given a choice between the 2 odors of vanilla and peppermint. Thus, training either through operant (Garren et al., 2013) or classical conditioning (Decker et al., 2007) involved pairing a positive unconditioned stimulus (apple) with the peppermint odor and a negative stimulus (20% saline solution) with the vanilla odor, leading to a switch in their odor preference.
All tests of odor preference were conducted inside a closed dark box housed in an environmental chamber. An infrared-sensitive CCD camera (Sony XC-77; Sony, Tokyo, Japan) was used to monitor the animal’s behavior. The interior of the box was illuminated with a darkroom safelight that limited visible light wavelengths to greater than 600 nanometers (Kodak 1A or GBX-2; Eastman-Kodak, Rochester, NY). Light intensity was adjusted with a rheostat to a final intensity at the floor of the testing arena less than 0.1 µE · m−2 · sec−1 (LiCor Photometer, Li-Cor Biosciences, Lincoln, Nebraska).
Measuring Odor Preference
Individuals were placed in the center of a circular Plexiglas arena that was 30 cm in diameter. Odors were provided by using a 1.5-cm-square piece of filter paper to soak up 20 µL of either vanilla or peppermint extract. The pieces of paper were inserted into a 1.5-mL microfuge tube that contained 24 small holes drilled into the sides to allow the odor to spread through the arena. Each tube was fixed to a modified Petri dish lid 3.1 cm in diameter. For odor preference testing, 1 dish of each odor was used, and these were placed on opposing sides of the arena. A visit to either odor dish consisted of the animal placing its mouth parts on and investigating the dish.
Initial odor preference was evaluated prior to training. In this “pretraining test,” each animal was given 12 min to make 10 total visits. To eliminate inactive or unhealthy animals, those who failed to make at least 8 visits were eliminated from further training or testing (approximately 20% of animals failed this criterion). After the pretraining test, a peppermint preference index (PPI) was calculated by dividing the number of visits to the peppermint dish by the total number of visits. The animal was then taken out and placed back in the group isolation chamber in constant darkness. Individuals were identified by unique patterns of dots on the pronotum made with white paint. Training, either through classical or operant conditioning, was conducted 24 h after the pretraining test. Posttraining tests for odor preference were carried out in the same manner as the odor preference test prior to training.
Training: Operant Conditioning
For training, a small cup in each Petri dish lid held either a slice of apple (paired with the peppermint odor) or 75 µL 20% saline solution (paired with the vanilla odor). A “standard” apple reward was prepared by inserting glass tubing, 0.5 cm in diameter, through the apple. The cylindrical core of apple retrieved from the tube was sliced with 2 razor blades separated by the 1-mm width of a microscope slide to produce 1-mm-thick apple slices that weighed approximately 0.01 g. The dishes were placed on opposite sides of the circular arena. A training trial ended after the animal reached and consumed the apple reward. The animal was then covered with a Petri dish and placed in the center of the arena for 5 min, until the next trial began. Three trials were conducted for each animal, and only animals that reached the apple reward during all 3 training trials were included in the data analysis. All training sessions were initiated in the early subjective night at circadian time 14 (CT 14), and training of a group of 8 animals took 2 h on average (CT 14-16). The number of visits to the vanilla dish and the time taken to reach the apple reward were recorded so that the development of short-term memory could also be determined and analyzed.
Training: Classical Conditioning
Training via classical conditioning was carried out on animals that had been restrained with tape to the back of an inverted Petri dish so that their head and mouth parts were easily accessible during training. To present the odorant stimuli, a piece of filter paper treated with 20 µL of either vanilla or peppermint extract was wrapped around the end of a small metal rod. As in operant conditioning, the apple reward was paired with the peppermint odor while a droplet of 20% saline solution was paired with the vanilla odor. For each trial, the odorant was brought up to the antennae, and 2 sec after, either the paired reward or punishment was applied to the mouth parts of the animal. Five minutes separated each trial, and 3 trials of both the peppermint and vanilla odors were conducted in an alternating fashion between peppermint and vanilla. After training, the animals were removed from the Petri dishes and placed back in their isolation chamber. Training was initiated at either CT 2 when intact animals are unable to learn the task or at CT 14, the peak of the animals’ ability to acquire new short- and long-term memories.
Injections
Injections were performed using a 25-µL syringe in which 5 µL of the desired solution was injected into the thoracic cavity through a joint of the metathoracic leg. The fipronil solution was made by dissolving fipronil in roach saline containing 0.1% acetone, creating a solution with a 5-ng/µL concentration of fipronil.
Optic Lobe Ablations
Surgical procedures were carried out with CO2 anesthesia. Access to the brain was obtained by removing a small square of cuticle from the head capsule. The ocelli and superficial fat bodies were taken out with forceps to reveal the optic tracts, which were then cut using microsurgical scissors, and the optic lobes were removed with forceps. Once both optic lobes were removed, the piece of exoskeleton was put back in place and sealed using melted wax. The animals were placed in an isolation container with food and water for 1 week to recover before being completely isolated for another week without food or water prior to testing. After the posttraining test, animals were dissected to confirm the successful removal of the optic lobes. Sham surgeries were carried out in a similar manner, without the cutting of the optic tracts and removal of the optic lobes.
Activity Rhythms
We monitored the activity of male roaches in running-wheel cages (e.g., see Page and Barrett, 1989) in constant darkness. Animals had free access to food and water. Rotation of the running wheel caused by the animal moving activated a switch closure that was counted and recorded in 6-min intervals by a computerized data acquisition system (Actimetrics, Willmete, Illinois).
Data Analysis
Learning and memory data were analyzed with Sigma Plot statistical software (version 11; Systat Software, Chicago, IL). The PPI, calculated by dividing the number of peppermint visits by the total number of visits made to either odor, was analyzed with a Kruskal-Wallis 1-way analysis of variance (ANOVA) followed by Dunn’s pairwise comparisons to identify significant differences between treatment groups.
Activity rhythm data were analyzed using Clocklab (Actimetrics). Activity onset time was used as a reference point for calculating the phase shift. The animals were monitored for at least 7 days and were then injected at their respective onset time. They were then monitored for another period of at least 7 days. To calculate the magnitude of the phase shift, we measured the interval between the extrapolated regression lines of activity onset pre- and postinjection on the day of the injection.
Results
Optic Lobe Ablations Restore Memory Acquisition following Classical Conditioning
We used a classical conditioning protocol to train both optic lobe ablation (OLX) and sham-operated animals at CT 2, when animals exhibit a severe deficit in memory acquisition, or CT 14, the peak of the circadian rhythm of memory formation (Fig. 1). Sham-operated animals trained at CT 14 were still able to perform when tested at 48 h later, while animals trained at CT 2 showed significant deficits in learning. In contrast, following optic lobe ablation, the animals were able to learn and recall in the olfactory memory task at both CT 2 and CT 14. The increase in PPI for both OLX groups as well as the sham animals trained at CT 14 were significantly greater than the change observed in the sham group trained at CT 2. This suggests that the removal of the optic lobes was able to restore the ability to form long-term memories during early subjective day (CT 2) without impact on memory acquisition at CT 14.
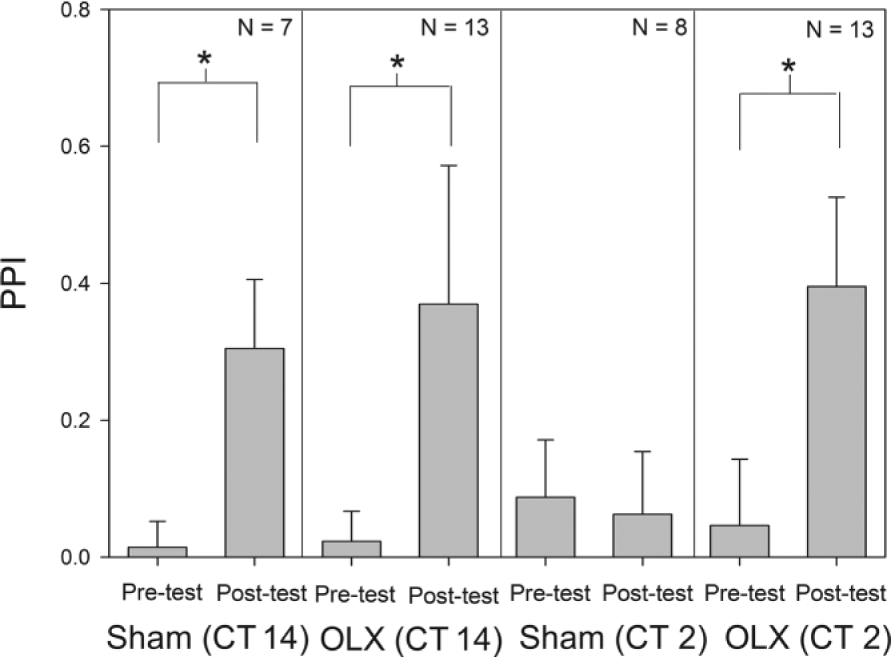
Plots pretest and posttest PPI values (mean ± SEM) for animals that received optic lobe ablations or a sham surgery and were subsequently trained via classical conditioning at either CT 2 or CT 14. Results indicate that all groups except for the sham control trained at CT 2 exhibited a significant increase in posttest PPI compared with their corresponding pretest values. The p value for the analysis of variance was significant (p < 0.001). Brackets marked with a (*) indicate a significant difference (p < 0.05) in PPI between the 2 indicated groups (Dunn’s post hoc test). OLX, optic lobe ablation; PPI, peppermint preference index.
These results suggest that the optic lobe is the site of the clock that regulates the circadian rhythm in olfactory memory formation via classical conditioning. In addition, the data suggest that the clock acts via an inhibitory mechanism to suppress memory acquisition in the early subjective day.
OLX Restores Memory Recall following Operant Conditioning
We investigated the effects of optic lobe ablation on memory recall following operant conditioning.
OLX cockroaches that were tested for memory retention and recall 36 and 48 h after training via operant conditioning were compared with sham-operated controls (Fig. 2). The sham group did not have a significant change in PPI when tested at 36 h after training, confirming previous results that showed that cockroaches normally cannot recall memories formed through operant conditioning at times of day other than when the task was first learned (Garren et al., 2013). In contrast, at both 36 and 48 h after training, OLX animals showed significant increases in PPI, indicating that the removal of the optic lobes and the biological clock rescued the inability of animals to recall memories at 36 h after training and did not affect recall at the 48-h test. Figure 2 shows a comparison of the change in PPI before and after training. Performance of the OLX groups tested at either 36 or 48 h was not significantly different and recall of both groups was substantially better than that exhibited by sham-operated controls. The data suggest (a) that the circadian clock that regulates memory recall is located in the optic lobe and (b) that the contextual cue of circadian phase of training that inhibits the ability to recall memories at circadian times other than the time of training acts via an inhibitory mechanism.
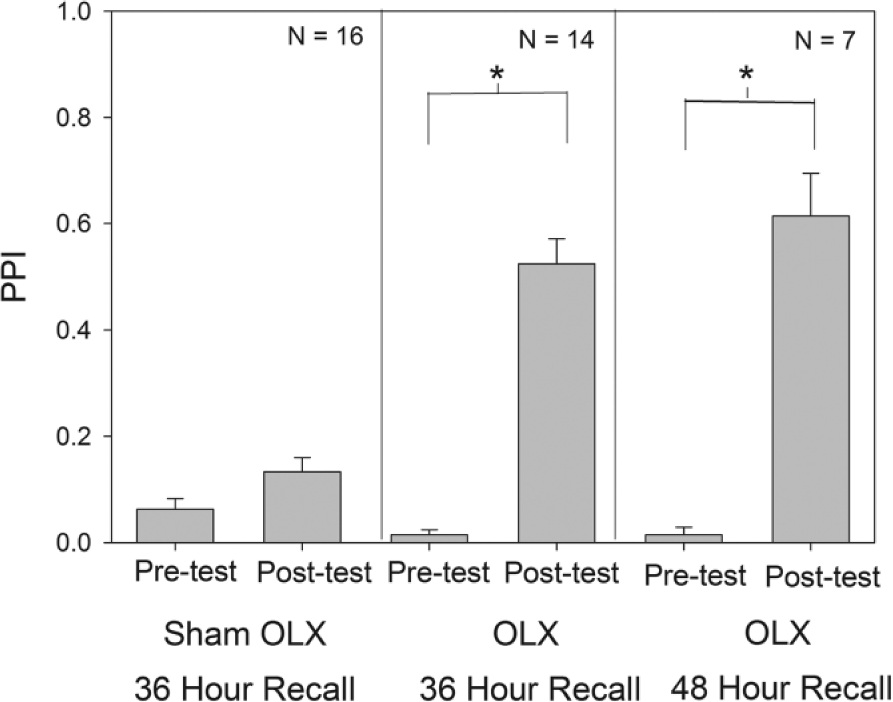
Plots pretest and posttest PPI values (mean ± SEM) for animals receiving optic lobe ablations and tested for memory recall at either 36 or 48 h after operant conditioning with the test values for sham-operated animals tested 36 h after training. During both recall times, animals with optic lobe ablations exhibited significantly higher posttest PPIs, unlike the sham group tested 36 h after training, which exhibited no significant change in PPI. The p value for the analysis of variance was significant (p < 0.001). Brackets marked with a (*) indicate a significant difference (p < 0.05) in PPI between the 2 indicated groups (Dunn’s post hoc test). OLX, optic lobe ablation; PPI, peppermint preference index.
Fipronil Injections
To test the hypothesis that the biological clocks act on memory formation (classical conditioning) and memory recall (operant conditioning) through an inhibitory mechanism, the impact of blocking ionotropic chloride channels on memory formation and recall was explored. In insects, the insecticide fipronil is known to block both GABA and glutamate-activated chloride channels (Ikeda, et al., 2003; Janssen et al., 2007).
Since memory formation with classical conditioning and memory recall following operant conditioning has been shown to be dependent on the circadian phase of training (Decker et al., 2007; Garren et al., 2013), it was important to show that the drug injections were not causing a significant phase shift in the circadian clock. Injections of 25 ng fipronil at CT 0 to 3 had only a small effect on the phase of the locomotor activity rhythm (ΔΦ = −0.68 ± 0.3, mean ± SEM, n = 8). Similarly, injections at CT 12 to 15 had a minimal effect on rhythm phase (ΔΦ = −0.71 ± 0.45, mean ± SEM, n = 5) (Fig. 3). In neither case would the small phase shift be expected to have a significant impact on learning and memory processes. An additional 3 animals injected with 250 ng fipronil died within 24 h.
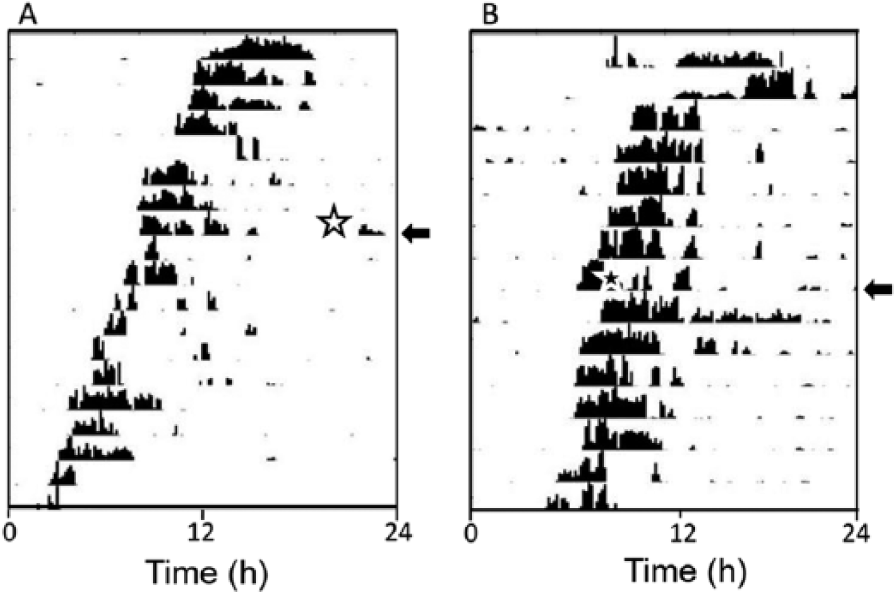
Actograms of 2 cockroaches freerunning in constant darkness showing the effects of fipronil injections (25 ng) on circadian phase. Arrows indicate the day of the injection, and the stars show the phase of the injection. (A) Injection at CT 2.5 caused an estimated phase advance of +0.5 h. (B) Injection at CT 14.1 resulted in an estimated phase delay of −1.2 h.
In an earlier study on the effects of fipronil on olfaction in bees, it was reported that fipronil may interfere with the bee’s ability to distinguish between odors (El Hassani et al., 2009). Thus, we wanted first to establish that the fipronil injections did not affect either the cockroach’s innate preference for vanilla over peppermint or the ability to distinguish between the 2 odors. Effects of fipronil injections on the pretraining peppermint preference test were compared with vehicle-injected controls (Fig. 4). There was no significant difference between the groups as cockroaches still exhibited a strong preference for vanilla, and there was no evidence of an effect on the ability to distinguish between the 2 odors or complete the sequence of testing and training (14 of 16 animals injected with fipronil completed the task).
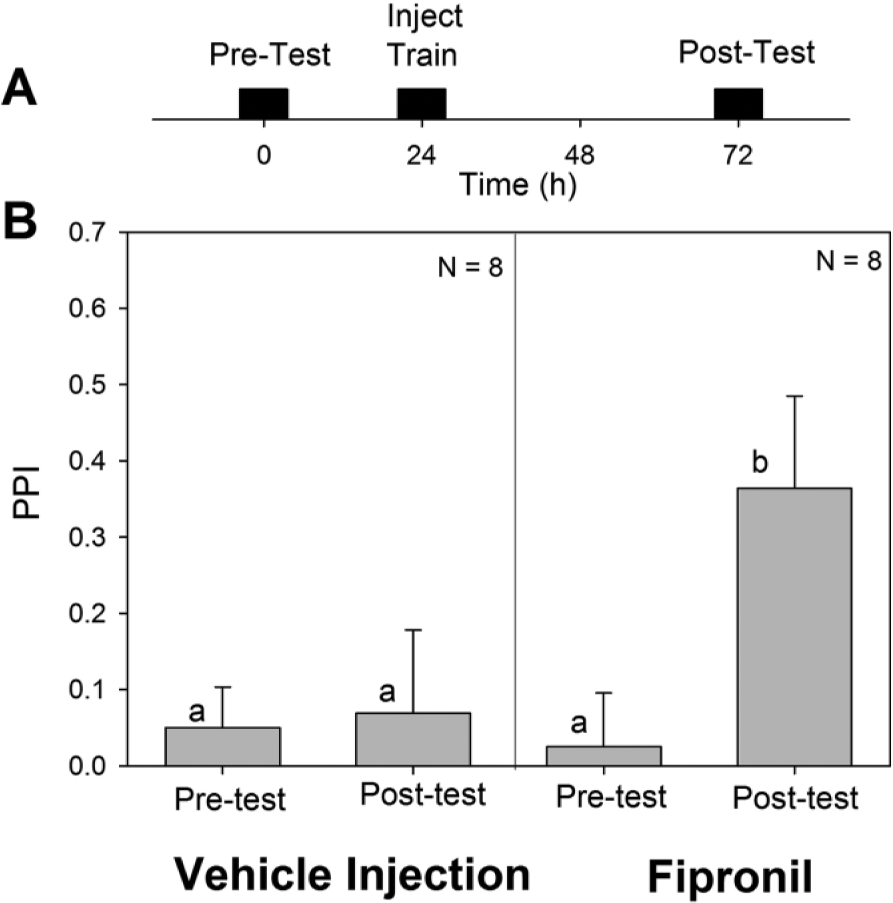
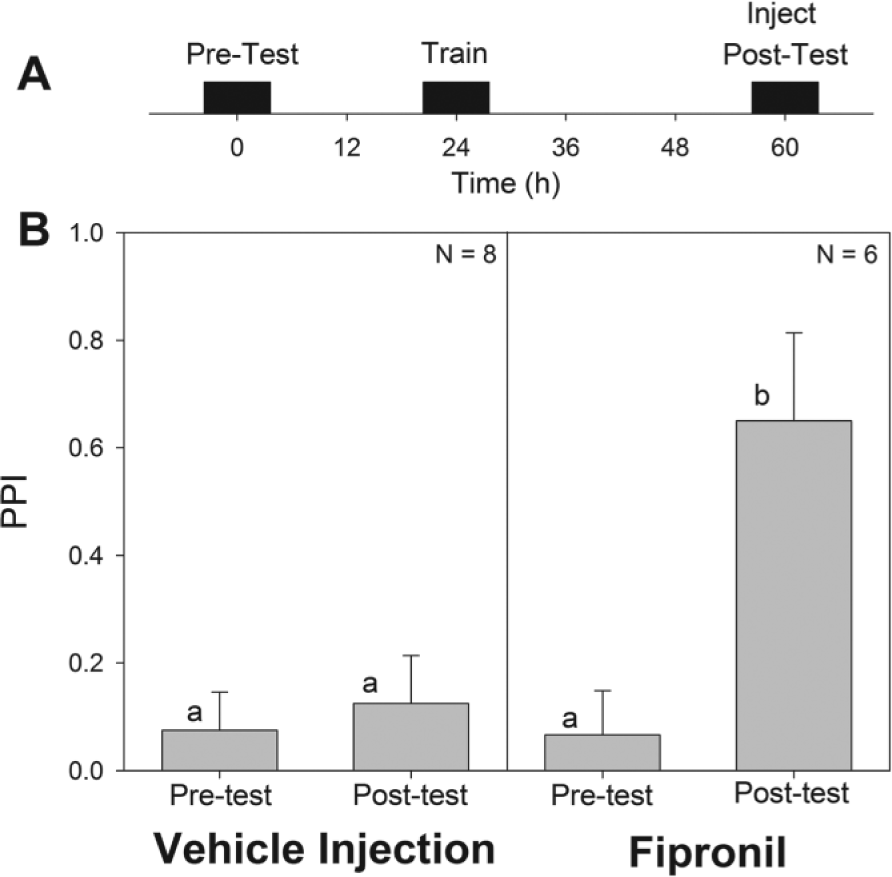
(A) Time course of experimental procedure. (B) Plots pretest and posttest PPI values (mean ± SEM) for animals trained at CT 2 via classical conditioning and injected with either fipronil or a control 30 min prior to training. Control animals showed no significant difference between pretest and posttest PPI, while the animals that received the fipronil injection exhibited a significantly higher posttest PPI. The p values for the analysis of variance were significant (p < 0.001). Bars marked with similar letters represent similar values, while different letters indicate a statistically significant difference (p < 0.05) between bars (Student-Newman-Keuls post hoc test). PPI, peppermint preference index.
To analyze the effect of the inhibition of GABA and glutamate-activated chloride channels on long-term memory formation in the cockroach, we injected animals with 25 ng fipronil 30 min prior to training via classical conditioning at CT 2 and compared with cockroaches receiving a vehicle injection (Fig. 4). It was clear that while vehicle-injected controls did not learn the classical conditioning task at CT 2, fipronil, applied just before training, restored the roach’s ability to form long-term memories at CT 2. Therefore, fipronil injections and optic lobe ablation had similar effects on long-term memory formation of classical conditioning tasks.
The use of an inhibitory mechanism to modulate memory recall in cockroaches was also analyzed by comparing cockroaches trained using an operant conditioning paradigm at CT 14, which received either a control or fipronil injection 30 min prior to a posttraining test at CT 2 (Fig. 5). While vehicle-injected control animals were unable to recall the memory of an operant conditioning task when tested 36 h after training, the cockroaches that received the fipronil injection showed a significant increase in PPI—fipronil restored the ability of cockroaches to recall memories 36 h after operant conditioning training. Therefore, the biological clock appears to modulate memory recall of operant conditioning tasks through an inhibitory mechanism, blocking recall at times other than when the task was first learned. In conjunction with the effects of fipronil on classical conditioning, this suggests the probable involvement of either GABA or glutamate in the biological clock’s inhibitory regulation of both memory formation and memory recall in the cockroach.
(A) Time course of experimental procedure. (B) Plots pretest and posttest PPI values (mean ± SEM) for animals trained at CT 14 via operant conditioning and injected with either fipronil or a control 30 min prior to a posttest conducted 36 h after training (at CT 2). Results indicate that while control animals showed no difference between pretest and posttest peppermint preference, the animals that received the fipronil injection exhibited a significantly higher posttest PPI. The p values for the analysis of variance were significant (p < 0.001). Bars marked with similar letters represent similar values, while different letters indicate a statistically significant difference (p < 0.05) between bars (Student-Newman-Keuls post hoc test). PPI, peppermint preference index.
Discussion
The results presented here address 2 issues with regard to the circadian regulation of olfactory learning and memory in the cockroach. The first concerns the location of the clock that regulates the rhythms, and the second concerns the mechanism by which phase information is imposed by the clock on learning and memory circuits.
Circadian Regulation of Memory Acquisition
The circadian regulation of the formation of associative memories has been demonstrated in a phylogenetic diversity of organisms, including mollusks (Lyons et al., 2005), insects (Decker et al., 2007; Lyons and Roman, 2009; Lehman et al., 2011), fish (Rawashdeh et al., 2007), mice (Chaudury and Colwell, 2002; Valentinuzzi et al., 2001), and Siberian hamsters (Ruby et al., 2008). A central question that arises from these studies is what is the mechanism by which the circadian system suppresses memory formation at some times of day or, alternatively, promotes memory formation at other times?
In L. maderae, it has been shown that the circadian system regulates the early stages of memory formation via classical conditioning (Decker et al., 2007). When trained in the early subjective day at CT 2, animals show a severe deficit in the ability to acquire new memories. Performance was severely impaired even when animals were tested only 5 min after training—the cockroaches trained in the early subjective day behaved the same as naïve, untrained animals—clearly indicating a deficit at the acquisition stage. The results presented here indicate that this deficit arises from a strong inhibition of memory acquisition by the circadian system in the optic lobe. Removal of the optic lobes eliminated the deficit in acquisition so that animals performed as well when trained at CT 2 as either sham-operated controls or optic lobe–ablated animals did when trained at CT 14. We further showed that injections of fipronil, an antagonist of ionotropic GABA or glutamate-activated chloride channels, could also rescue the learning deficit at CT 2. The data lead to the hypothesis that the circadian clock in the optic lobe regulates formation of olfactory memories via the inhibition of neural circuitry involved in early stages of memory acquisition. It is interesting to note in this context that in 2 other phylogenetically distinct animals, zebrafish (Rawashdeh et al., 2007) and hamsters (Ruby et al., 2008; Fernandez et al., 2014), circadian regulation of associative memory formation also appears to involve inhibition of the neural circuity involved in early stages of memory acquisition. In zebrafish, data from surgical lesion and pharmacological experiments showed that melatonin released from the pineal gland suppressed nighttime memory formation in active avoidance conditioning. In hamsters, lesions of the SCN or treatment with the GABAA receptor antagonist pentylenetetrazol (PTZ) were shown to rescue memory deficits in a novel object recognition task that result from abolition of circadian cycling by brief exposure to an abnormal lighting regimen. Here the hypothesis is that this method of disruption of the circadian rhythm results in constant elevation of inhibitory tone by the arrhythmic circadian circuitry (SCN) that can be alleviated by PTZ treatment. This is further consistent with the observation in rats that surgical ablation of the SCN, which would eliminate any SCN-driven inhibition, also rescues circadian deficits in a passive avoidance task (Stephan and Kovacevic, 1978). A similar story has been developed in the mollusk Aplysia, where results suggest that the circadian clock suppresses formation of long-term memory of nonassociative sensitization at night through repression of training-induced increases in MAPK and ApC/EBP (Lyons et al., 2006). The data on mollusks, fish, rodents, and cockroaches are consistent with the general view that circadian systems regulate memory formation by inhibition at those times of day when, presumably, the formation of some types of memories is, for one reason or another, inappropriate.
Circadian Regulation of Memory Recall
The linkage between the circadian phase of training and the performance of a memory task (referred to variously as Zeitgedachtnis, time-memory, time-stamping, or time-place learning) was first documented in honeybees (Beling, 1929) and has since been studied extensively in bees (reviewed in Moore, 2001) and a variety of other organisms, including birds, fish, and mammals (reviewed in Mulder et al., 2013). The data raise the prickly, and as yet unanswered, question of precisely how the circadian phase of training is encoded in memory circuits (along with the other contextual cues associated with a particular memory) and how the subsequent rhythmic expression of memory recall is temporally regulated.
Two potential hypotheses to explain the learned connection between circadian phase and performance have been suggested (e.g., Moore, 2001; Mulder et al., 2013; Ralph et al., 2013). In the first, it is suggested that there is a circadian oscillator somewhere in the animal whose phase is set by the training procedure. Because some animals at least are able to associate memories with multiple times of day (e.g., Kolterman, 1971; Wagner et al., 2013), there must be multiple oscillations that can be independently set, perhaps within the cells that encode memory (Ralph et al., 2013). These oscillators subsequently regulate the ability to recall specific memories in a phase-dependent manner. The second hypothesis suggests that animals are able to formulate an association between some readout of the phase of a “continuously consulted” clock and the training procedure. Here the idea is that the animal has the ability to “read” some feature of the phase-dependent output of a circadian clock and tie that phase to a particular memory as a contextual cue for recall.
In hamsters, a substantial body of evidence suggests that memory for time of day in a conditioned place avoidance task depends on one or more oscillators that reside outside the SCN (Cain and Ralph, 2009; Cain et al., 2012; Ralph et al., 2013). The proposal is that training sets the phase of a context-entrainable oscillator (CEO) in much the same way that time-restricted feeding sets the food-entrainable oscillator (FEO) (Stephan, 1983, 1989, 2002). Central to this hypothesis is the observation that lesions of the SCN do not prevent these time-of-day associations (e.g., Stephan, 1983; Mistlberger et al., 1996; Cain and Ralph, 2009). However, recent results do suggest that the SCN can entrain the CEO (Ralph et al., 2013), indicating the involvement of both the central pacemaking system in the SCN and extra-SCN oscillators in timing performance.
In contrast to the mammalian case, in the cockroach, we find that the time-of-day association for memory recall that occurs with operant conditioning is removed following ablation of the optic lobes, the site of the central pacemaker that is functionally analogous to the mammalian SCN. We have also shown that treatment with GABA/glutamate-activated chloride channel antagonist fipronil eliminates the performance deficit at “inappropriate” times. Together, these results lead to speculation that the optic lobes can drive inhibitory circuits that prevent memory recall in a phase-dependent fashion following operant conditioning. Whether this timed inhibition depends on the phase of a continuously consulted “master pacemaker” in the optic lobes or if it is regulated by additional “context entrainable oscillators” that either reside in the optic lobes or depend on optic lobe structures to be effective will require further investigation.
In summary, it is clear that many animals possess the remarkable ability to form associative memories that includes time of day as a key element and that the “clock” used for the readout of time has the properties of a circadian oscillator. However, much remains to be learned about how and where circadian phase is encoded within the memory circuits to regulate behavior.
Footnotes
Acknowledgements
The research reported here was supported by National Science Foundation grant IOS-1145605 to T.L.P.
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
