Abstract
The suprachiasmatic nucleus (SCN) of the hypothalamus is the brain structure that controls circadian rhythms in mammals. The SCN is formed by two neuroanatomical regions: the ventral and dorsal. Gamma-aminobutyric acid (GABA) neurotransmission is important for the regulation of circadian rhythms. Excitatory GABA effects have been described in both SCN regions displaying a circadian variation. Moreover, the GABAergic system transfers photic information from the ventral to the dorsal SCN. However, there is almost no knowledge about GABA neurotransmission during the prenatal or postnatal development of the SCN. Here, we used whole-cell patch-clamp recordings to study spontaneous inhibitory postsynaptic currents (IPSCs) in the two SCN regions, at two zeitgeber times (day or night), and at four postnatal (P) ages: P3-5, P7-9, P12-15, and P20-25. The results herein show that the three analyzed parameters of the IPSCs, frequency, amplitude, and decay time, were significantly affected by the postnatal age: mostly, the IPSC frequency increased with age, principally in the ventral SCN in both day and night recordings; similarly, the amplitude of IPSCs augmented with age, especially at night, whereas the IPSC decay time was reduced (it was faster) with postnatal age, mainly during the day. Our findings first reveal that parameters of GABA neurotransmission are modified by postnatal development, implying that synaptic adjustments are required for an appropriate maturation of the GABAergic system in the SCN.
The suprachiasmatic nucleus (SCN) of the hypothalamus is the master clock responsible for regulating circadian rhythms in mammals (Stephan and Zucker, 1972). The SCN is neuroanatomically divided into two regions: ventral and dorsal. Both SCN regions abundantly produce and release the neurotransmitter GABA (Moore and Speh, 1993; Abrahamson and Moore, 2001), which synchronizes neurons of the ventral SCN with those of the dorsal SCN, after a shift in the light-dark cycle (Albus et al., 2005). The GABAergic system plays a role in the physiology of SCN neurons and is implicated in the synchronization of circadian rhythms to light (Albers et al., 2017). GABA pulses produce phase shifts similar to those generated by light (Liu and Reppert, 2000), whereas impairment of GABA neurotransmission reduces light-induced phase shifts (Han et al., 2012; McNeill et al., 2018). As part of circadian regulation in the SCN, GABA exhibits a circadian rhythm in releasein release (Aguilar-Roblero et al., 1993), and in the dorsal SCN, the frequency of spontaneous GABAergic inhibitory postsynaptic currents (IPSCs) . . . that peaks at night (Itri et al., 2004).
Although GABA is the prototypical inhibitory neurotransmitter, it is able to exert excitatory effects during development. Excitatory GABA effects are induced when the Cl− equilibrium potential (ECl−) is more depolarized than the resting membrane potential due to an [Cl−]i ≥ 20 mM. In this situation, activation of GABAA receptors elicits Cl− efflux, thereby depolarizing the membrane potential. Na+-K+-2Cl− (NKCCs) and K+-Cl− cotransporters (KCCs) are the main determinants of excitatory or inhibitory GABA effects by regulation of [Cl−]i. NKCCs increase [Cl−]i and KCCs reduce it. During early development, the expression of NKCCs decreases, while that of KCCs augments, resulting in the switching of the role of GABA from excitatory to inhibitory, which occurs between postnatal days (P) 6-12 (Sun and Murali, 1999; Ben-Ari, 2002). Circadian excitatory GABA effects have been described in the adult SCN in some studies. For instance, it has been reported that GABA administration increases spontaneous firing rate in the SCN during the day (Wagner et al., 1997), while similar excitatory effects were observed at night mainly in the dorsal SCN (Choi et al., 2008). Also, a more depolarized ECl− (~ -30 mV) was detected in the ventral SCN during the night and in the dorsal SCN at day (Alamilla et al., 2014). Moreover, a higher [Cl−]i was noticed during the day in both arginine vasopressine (AVP) and vasointestinal polypeptide positive SCN neurons, implying an important role of NKCC1 and KCC2 (Klett and Allen, 2017).
At present, information about GABA neurotransmission during the development of the SCN is very limited. As far as we know, there are only two studies analyzing developmental changes in this neurotransmission system in the hypothalamus and the SCN. Excitatory GABA effects have been reported in embryonic hypothalamic neuronal cell cultures that decreased after 18 days (Obrietan and Van den Pol, 1995). In the SCN, Ikeda et al. (2003) reported that at P6-7, the GABAergic agonist muscimol increases intracellular Ca2+ equally during day and night, whereas circadian differences (higher [Ca2+]i during the day) are observed at older postnatal ages (P9-14). These studies suggest a developmental maturation of the GABAergic system in the hypothalamus and the SCN. Here, we studied the spontaneous GABA release in both regions of the SCN (ventral and dorsal), at day and night, across early postnatal development (from P3 to P25).
Materials and Methods
Ethics
This study was conducted after the approval of the Institutional Animal Care and Use Committee of the University of Colima according with the Guide for the Care and Use of Laboratory Animals (U.S. National Research Council, 2011). Hypothalamic brain slices (P3-25) were utilized for this investigation. Euthanasia by decapitation was applied on anesthetized (sodium pentobarbitone, 60 mg/kg, i.p.) animals after confirming no pedal withdrawal reflexes.
Animals and Slice Preparation
Wistar rats of either sex were used, N = 62: ~40% males and ~60% females; for some animals, the sex was not recorded (Suppl. Table 1). For the experiments, rats were bred in our animal facilities from parents provided by Harlan Mexico. Rat pups were kept with their mother until the day of the assays or weaned at P21 and housed in pairs. A 12:12 h light-dark cycle (lights-on at 0700 h) was applied.
The brain was quickly removed from decapitated rats to obtain the hypothalamic slices (220-250 µm thick, as previously reported by Irwin and Allen, 2007; Moldavan et al., 2017), where three to four slices (depending on the age of the animals) can be obtained for each brain with a VT1000 microtome (Leica Microsystems, Wetzlar, Germany) in cold (4 °C) artificial cerebrospinal fluid (ACSF), which contained (in mM) 124 NaCl, 1 MgSO4, 5 KCl, 1.25 KH2PO4, 10 glucose, 26 NaHCO3, 2 CaCl2, and supplemented with 0.02 cyclothiazide; the solution was bubbled with 95% O2/5% CO2 and adjusted to pH 7.4 (Reyes-Mendez et al., 2020).
Recording of Spontaneous IPSCs
Spontaneous GABAergic IPSCs were recorded with a similar methodological approach as previously published (Reyes-Mendez et al., 2020). Briefly, whole-cell recordings were performed in ventral and dorsal SCN neurons from rostral to caudal levels (Moore et al., 2002) by inspection of the slices with an Axio Examiner A1 microscope, equipped with differential interference contrast optics with 10× (W Achroplan, NA 0.3) water immersion objectives (Carl Zeiss, Oberkochen, Germany). The recording ACSF solution included CNQX (5 µM) and MK-801 (10 μM). Recordings were made at a holding potential of −60 mV for at least 10 min. For day experiments, brain slices were obtained at ZT 4 and recordings were carried out at ZT 5-7, whereas for night experiments, the brain slices were prepared at ZT 11 and recordings were performed at ZT 16-19. After obtaining the slices, they were maintained in ACSF (at 22-24 °C), bubbled with 95% O2-5% CO2, and transferred to the recording chamber 30 min before the recordings.
Statistical Analysis
Data are reported as mean ± standard error of the mean. Spontaneous IPSCs were analyzed using Mini Analysis 6.0.7 (Synaptosoft, Decatur, GA) and pClamp 10.2 software (Molecular Devices, Sunnyvale, CA). One cell was recorded per brain slice, while measurements of IPSC parameters (frequency, amplitude, and decay time) from the three to four slices per animal were averaged to obtain independent values for these parameters within an animal. Afterwards, statistical analysis was done with Prism 8 (GraphPad Software, La Jolla, CA) and Origin 8.5 software (OriginLab, Northampton, MA). The normal distribution (Shapiro-Wilk test) and the homogeneity of variances (Levene test) of data were checked to perform a three-way analysis of variance (ANOVA) and, if needed, Dunnett’s or Tukey’s post hoc tests. A p < 0.05 was defined as significant.
Results
Figure 1 illustrates the location of cellular recordings performed in each SCN region. GABAergic IPSCs were studied and validated by adding the GABAA receptor antagonist picrotoxin (50 µM) after each assay (Figure 2).
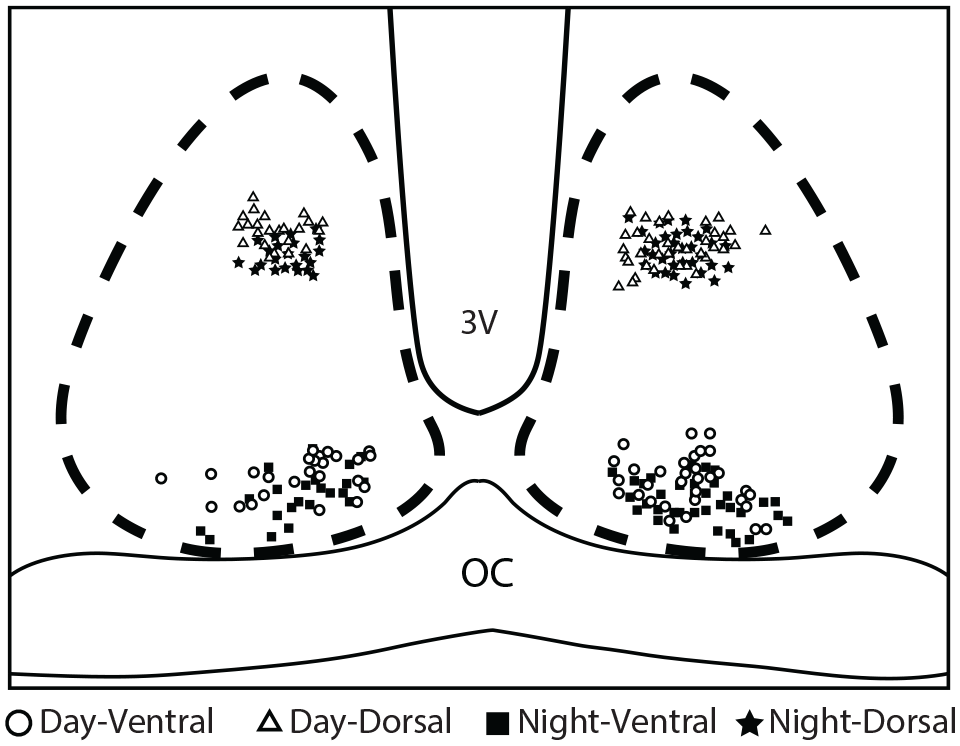
Location of SCN neurons recordings. Cartoon representation depicting the individual neurons recorded in both SCN neurons regions (dorsal/ventral) at day or night. The symbols illustrate the place of recordings in the SCN at the indicated ZT time. Abbreviations: SCN = suprachiasmatic nucleus; 3V = third ventricle; OC = optic chiasm.
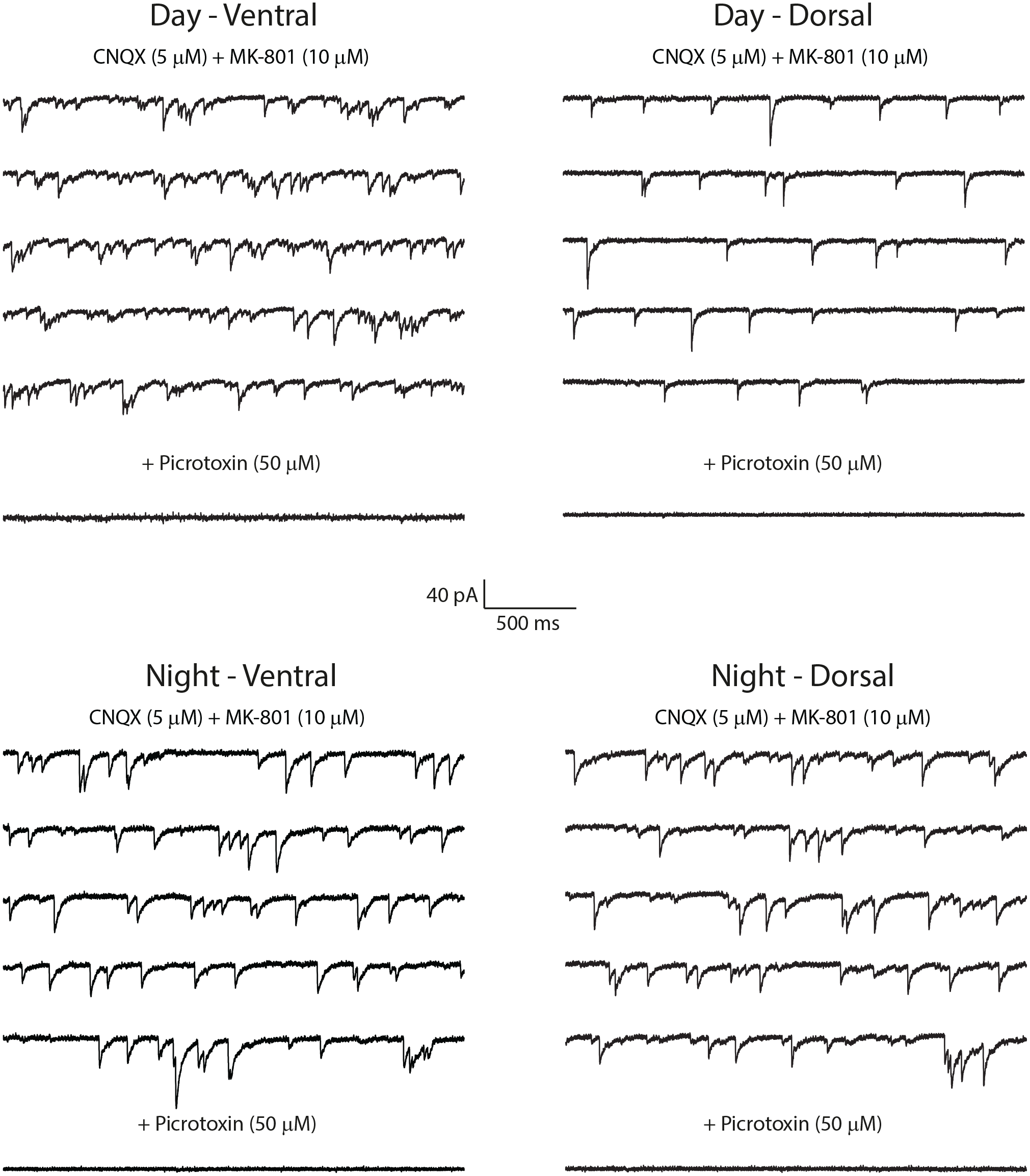
Spontaneous GABAergic IPSCs in the SCN regions and ZT times. SCN regions and times of recordings are indicated above the current traces. All current traces shown in the panels correspond to P20 to P25 animals. Abbreviations: IPSCs = inhibitory postsynaptic currents; SCN = suprachiasmatic nucleus.
A three-way ANOVA was used to examine the statistical differences of the frequency, amplitude, and decay time of the IPSCs between the different groups evaluated. This analysis revealed that the IPSC frequency increased with postnatal age, F3,48 = 15.8, p < 0.0001, and was higher in the ventral than in the dorsal SCN, F1,48 = 6.34, p = 0.015. Also, an interaction between age and SCN regions was detected, F3,48 = 4.9, p = 0.0045; this parameter augmented with age in the ventral SCN, while in the dorsal SCN, age had no influence on IPSC frequency (Figure 3a and Suppl. Table 2). Post hoc tests indicated that in both day and night recordings of the ventral SCN, the frequency was greater at P20-25 than at P3-5 and P7-9. Moreover, in the night/ventral data set, the P20-25 group displayed higher frequency than at P12-15, and comparisons between the P20-25 groups recorded in the day exhibited more frequency in the ventral than in the dorsal SCN (Figure 3a and Suppl. Table 2).
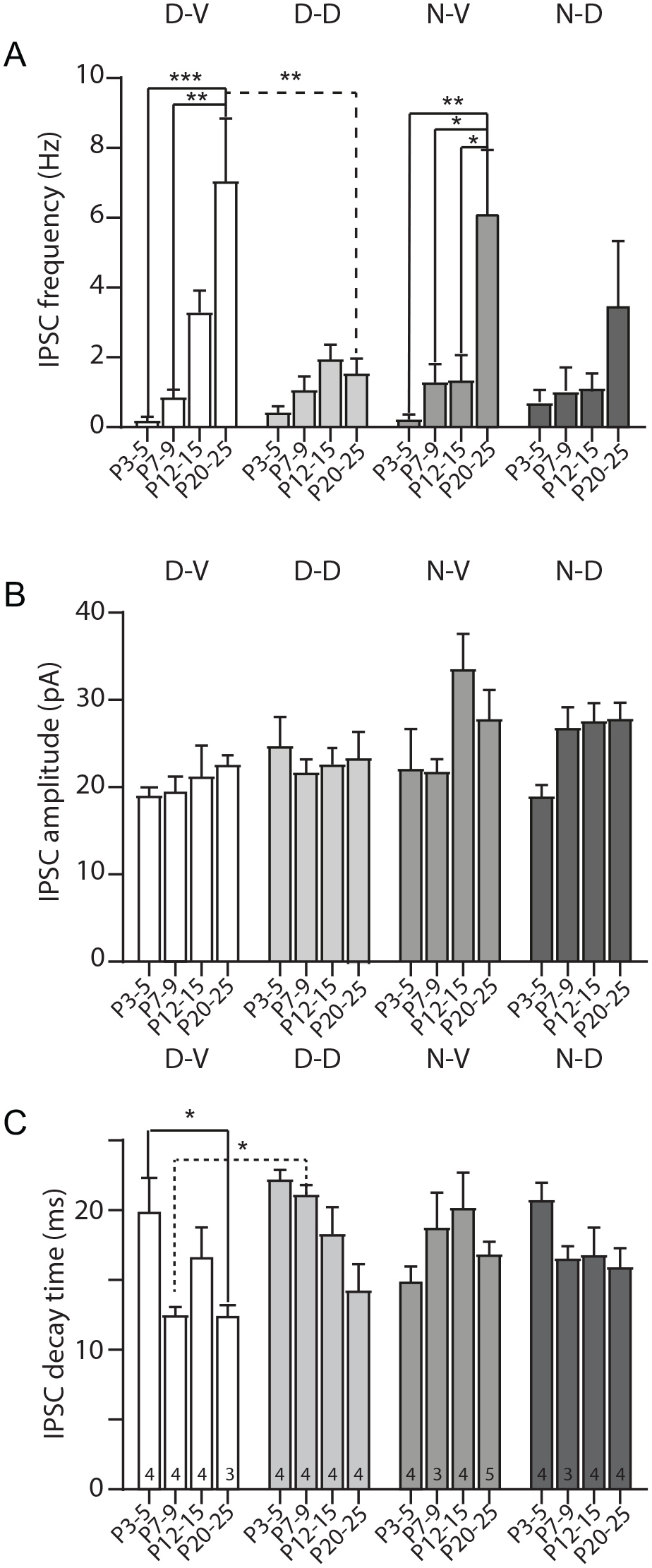
GABAergic release along postnatal development in the SCN regions and ZT times. Summarized measurement of the frequency (a), amplitude (b), and decay time (c) of IPSCs recorded at different ages of postnatal development, SCN zones, and ZT times. The number of neurons recorded for each group (n) is indicated at the bottom inside of each bar of panel c. For all panels, Tukey’s post hoc test, *p < 0.05. **p < 0.01. ***p < 0.001. Abbreviations: SCN = suprachiasmatic nucleus; IPSCs = inhibitory postsynaptic currents.
Regarding the amplitude of IPSCs, the three-way ANOVA disclosed that this parameter also increased with postnatal age, F3,48 = 3.8, p = 0.016; the average of the IPSC amplitude was larger at P12-15 in comparison with P3-5. In addition, the analysis identified that the IPSC amplitude was greater at night than at the day, F1,48 = 10.5, p = 0.002 (Figure 3b and Suppl. Table 2).
Finally, examination of the decay time (decay to 37% of the peak amplitude) of the IPSCs showed that this parameter decreased with age, F3,48 = 6.1, p = 0.0014, and was smaller in the ventral SCN compared with the dorsal SCN, F1,48 = 5.2, p = 0.027 (Figure 3c and Suppl. Table 2). An interaction between age and SCN regions was noticed, F3,48 = 2.8, p = 0.048, and thus, this parameter diminished with age in the dorsal SCN, but not in the ventral SCN where the IPSC decay time was not affected by postnatal age. Interaction between recording time and SCN regions was distinguished as well, F1,48 = 6.2, p = 0.016; the decay time was lesser at night in the dorsal SCN, whereas in the ventral SCN, the recording time (day/night) had no effect on this parameter (Figure 3c and Suppl. Table 2). Furthermore, the analysis showed that the three variables evaluated (postnatal age, SCN regions, and recording time) are interrelated (triple interaction) to modify the IPSC decay time, F3,48 = 3.9, p = 0.015. Specific post hoc tests between groups pointed out that in the day recordings of the ventral SCN, the decay time was reduced at P20-25 than at P3-5, and when comparing the P7-9 animals recorded in the day, a faster decay time was observed in the ventral than in the dorsal SCN (Figure 3c and Suppl. Table 2).
As the hypothalamic slices for day vs night recordings had different acclimation duration (see “MATERIALS AND METHODS”), we asked if this could have influenced our results by possible different cellular conditions due to a deterioration of the brain slices. Hence, we analyzed the passive membrane properties of SCN neurons recorded in both ZT. Our results revealed no significant differences at any parameter evaluated: membrane capacitance, membrane resistance, and access resistance (Table 1).
Passive membrane properties of SCN neurons recorded during day and night.

Abbreviation: SCN = suprachiasmatic nucleus.
Discussion
Here, we studied the spontaneous release of GABA in both SCN regions, during the day and night, in four postnatal ages. We found a developmental modification of the three analyzed parameters, frequency, amplitude, and decay time of GABAergic IPSCs.
We detected no significant day/night change in the IPSC frequency (which reflects the spontaneous GABA release), contrasting with previous investigations reporting a higher GABA release during the night in the SCN (Aguilar-Roblero et al., 1993) and in the dorsal SCN (Itri et al., 2004). In the first reference, authors did not consider the SCN regions and animals were much older (2-3 months) than ours. In Itri et al. (2004), the age of the animals (>P27) and the species (mice C57BL/6) might explain the discrepancy with our results. We observed a greater IPSC frequency depending on the age and SCN region. Similar results have been described in other brain structures (Cohen et al., 2000; Pardo et al., 2018).
Our result that the IPSC amplitude increased with age is consistent with that found in the anterior piriform cortex (Pardo et al., 2018). Also, modification of the IPSC decay time during development has been previously shown in other brain structures (Cohen et al., 2000; Pardo et al., 2018), and this could imply a remodeling of the subunits that compose the GABAA receptor. Indeed, there is a circadian expression of the β1 subunit of the GABAA receptor in the SCN (Naum et al., 2001). The increase of the frequency and amplitude of IPSCs with age implies that postnatal development is required to accomplish a mature level of spontaneous GABA release. Presynaptic (neurotransmitter release probability and/or number of presynaptic terminals) and/or postsynaptic (number of postsynaptic receptors) changes might occur in the GABAergic terminals during postnatal development that allow the increment of the spontaneous activity and the amplitude of IPSCs (Reyes-Mendez et al., 2020).
Previously, it was unknown the developmental age at which GABA displays circadian and regional differences in the SCN. Our findings in rats suggest that this age is ~P12 onwards, which coincides with the consolidation of circadian rhythms and synaptogenesis augment in the SCN (Carmona-Alcocer et al., 2017). Eye-opening occurs after the second postnatal week, although light can still enter into the eyes even when the eyelids are closed. According to our hypothesis, the GABAergic system demands a longer maturation time than that needed by the retinohypothalamic tract glutamatergic neurotransmission (~P7) (Duncan et al., 1986; Reyes-Mendez et al., 2020).
As the ventral SCN synchronizes faster to the light than the dorsal SCN, the GABAergic system communicates this information to this latter (Albus et al., 2005; Han et al., 2012). Our results could explain the day/night and regional differences in electrical activity of SCN neurons. During the day, most ventral SCN neurons have an ECl− ~ −60 mV (Alamilla et al., 2014); thus, it is likely that the increment of IPSCs in this zone hyperpolarizes the membrane potential, reducing the spontaneous firing rate and the amount of GABA inputs that arrive to the dorsal SCN, thereby decreasing the “external noise” in the period of highest activity of clock cells. (Antle and Silver, 2005). In contrast, during the night, most ventral SCN neurons have an ECl− ~ −30 mV (Alamilla et al., 2014); the increase of IPSCs would depolarize the membrane potential and increase the spontaneous firing, allowing intensification of GABA release in the dorsal SCN, which have ECl− ~ −60 mV (Alamilla et al., 2014), reducing spontaneous firing rate in the dorsal SCN and contributing to the circadian activity of these nuclei.
Another possible role of the increment of IPSC frequency in ventral SCN neurons at night could be the participation of the endogenous GABA level in the synchronization to light of circadian rhythms, since administration in the SCN of the NKCC1 transporter blocker, bumetanide, and the antagonist of GABAA receptors, bicuculline both reduce light-induced phase delays, but not phase advances (Albers et al., 2017; McNeill et al., 2018). Thus, it is likely that the spontaneous GABA level during the night is relevant in ventral SCN neurons because most of them are VIP neurons (Antle and Silver, 2005), which help mediate synchronization to light (Jones et al., 2018; Mazuski et al., 2018).
As sexual differences have been reported in brain GABAergic neurotransmission (Auger et al., 2001; Pandya et al., 2019), and as we recorded IPSCs from male and female rats, it is plausible that there exists an indeterminate relationship between our results and the sex of the animals used. However, it is not possible to determine this question with the data set of this research. Another possible limitation was the disparity in the acclimation time of the brain slices used for the day and night recordings. However, our analyses of the passive membrane properties support the idea that this factor had no influence on the parameters of GABAergic IPSCs.
In conclusion, we show for the first time that several parameters of the GABA neurotransmission, such as frequency, amplitude, and decay time of spontaneous IPSCs, are modified along the postnatal development of the SCN. However, more research is needed to determine how excitatory GABA effects and their mechanisms reshape across the SCN development.
Supplemental Material
sj-pdf-1-jbr-10.1177_07487304211048052 – Supplemental material for GABA Neurotransmission of the Suprachiasmatic Nucleus Is Modified During Rat Postnatal Development
Supplemental material, sj-pdf-1-jbr-10.1177_07487304211048052 for GABA Neurotransmission of the Suprachiasmatic Nucleus Is Modified During Rat Postnatal Development by Fernando Osuna-Lopez, Miriam E. Reyes-Mendez, J. Manuel Herrera-Zamora, Jose Luis Gongora-Alfaro, Eloy G. Moreno-Galindo and Javier Alamilla in Journal of Biological Rhythms
Supplemental Material
sj-pdf-2-jbr-10.1177_07487304211048052 – Supplemental material for GABA Neurotransmission of the Suprachiasmatic Nucleus Is Modified During Rat Postnatal Development
Supplemental material, sj-pdf-2-jbr-10.1177_07487304211048052 for GABA Neurotransmission of the Suprachiasmatic Nucleus Is Modified During Rat Postnatal Development by Fernando Osuna-Lopez, Miriam E. Reyes-Mendez, J. Manuel Herrera-Zamora, Jose Luis Gongora-Alfaro, Eloy G. Moreno-Galindo and Javier Alamilla in Journal of Biological Rhythms
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
