Abstract
Functional magnetic resonance imaging (fMRI) is widely used by researchers to noninvasively monitor brain-wide activity. The traditional assumption of a uniform relationship between neuronal and hemodynamic activity throughout the brain has been increasingly challenged. This relationship is now believed to be impacted by heterogeneously distributed cell types and neurochemical signaling. To date, most cell-type- and neurotransmitter-specific influences on hemodynamics have been examined within the cortex and hippocampus of rodent models, where glutamatergic signaling is prominent. However, neurochemical influences on hemodynamics are relatively unknown in largely GABAergic brain regions such as the rodent caudate putamen (CPu). Given the extensive contribution of CPu function and dysfunction to behavior, and the increasing focus on this region in fMRI studies, improved understanding of CPu hemodynamics could have broad impacts. Here we discuss existing findings on neurochemical contributions to hemodynamics as they may relate to the CPu with special consideration for how these contributions could originate from various cell types and circuits. We hope this review can help inform the direction of future studies as well as interpretation of fMRI findings in the CPu.
Introduction
Functional magnetic resonance imaging (fMRI) is widely used to study human brain function and networks. fMRI gives an indirect measure of neuronal activity through measuring cerebral hemodynamics and its data are interpreted according to the presumed relationship between neuronal and vascular responses, termed neurovascular coupling. It is suggested that regional neuronal and hemodynamic activities are scalable under physiological conditions – a phenomenon attributed to the vascular signaling cascades following neuronal activation. However, even in healthy subjects, hemodynamics are not regulated uniformly throughout the brain, which complicates fMRI data interpretation. This has implications for clinical science as dysregulated hemodynamics can be brain region-specific and lead to energy deficits and eventually brain pathologies. 1 Exclusively considering neuronal activity in fMRI data interpretation omits modulatory influences from neurochemicals, many of which are vasoactive. It is therefore important to consider how neuronal activity and neurochemicals collectively produce hemodynamic responses. Such knowledge will help us understand hemodynamics in the healthy brain and differentiate between healthy and dysregulated hemodynamics in clinical conditions where neurotransmission is also dysregulated.
One of the most used fMRI contrasts is the blood-oxygenation-level-dependent (BOLD) contrast. Rather than reflecting a single physiological metric, BOLD signal is the concerted result of cerebral blood volume (CBV), cerebral blood flow (CBF), and the cerebral metabolic rate of oxygen (CMRO2),2,3 all of which are regulated via signaling within and between various cell types. Blood vessels are enveloped with perivascular cells that include endothelial cells, smooth muscle cells, and pericytes that can modulate vascular tone and propagate signals along vessels. 4 There is some debate as to whether pericytes are contractile,5–7 but more recent studies reveal that pericytes can directly modulate capillary blood flow. 4 Additionally, perivascular cells can initiate vascular change directly or through indirect signaling via astrocytes. 8 Several vascular signaling cascades from neurons or astrocytes have been identified, such as vasodilative nitric oxide (NO) and certain arachidonic acid (AA) derivatives (e.g., prostaglandin) and vasoconstrictive neurochemicals such as the AA derivative 20-HETE. 8 Further, the vasomodulatory impact of various neurotransmitters must be considered because expression of their receptors has been reported on perivascular cells. 9 Examining how specific neurochemical signaling impacts the hemodynamic response at the level of individual brain regions will pave the way towards more accurate fMRI data interpretation.
The dorsal striatum or caudate putamen (CPu) serves as an integrative hub for cortical, limbic, and motor inputs, and is involved in motor learning, decision making, reward, and habit formation. Aberrant CPu function and connectivity are hallmarks associated with neurological disorders such as Parkinson’s and Huntington’s disease, which have dysregulated hemodynamics (Supplemental Material; Figure 2).10–12 Notably, the CPu represents a brain region with distinct vascularization and circuit-level organization compared to the cerebral cortex.13–15 Recent fiber photometry studies reported distinct hemodynamic response functions between the CPu and the motor cortex, 16 but highly consistent hemodynamic response functions across multiple cortical regions. 15 In support, the lateral and medial lenticulostriate arteries, and recurrent artery of Heubner, which respectively branch from the anterior and middle cerebral artery, and anterior choroidal artery, supply the CPu but do not send blood to the cortex. 13 Further, unlike the columnar organization in cortex, the CPu is divided into matrix and striosome compartments, where the matrix is more vascularized than striosome.13,14 These compartments are neurochemically distinguishable, where striosomes, which constitute 15% of total CPu volume, punctuate the much larger matrix. 17 In primates, the CPu is also divided by a white matter tract, the internal capsule. Although white matter fMRI contrast is relatively poor, there is evidence of internal capsule BOLD activation during finger tapping, 18 and negative composite BOLD during mild hypercapnia that is likely the result of vascular steal. 19 While neurovascular coupling is not explicitly shown in the internal capsule, these data indicate that it is a possibility. Though the CPu and cortex have similar blood flow at rest 20 and respond similarly to hypoxic and hypercapnic challenges, 21 these regions exhibit different monoamine concentrations. 22 Hypoxia, hypercapnia, or a combination of the two can modulate the regional concentration of monoamines including dopamine, serotonin, noradrenaline, and their metabolites.23,24 Such modulatory effects are brain-region dependent. For example, hypoxia with hypercapnia significantly reduces dopamine levels in the rodent CPu but not in the cortex or hippocampus compared to controls. 25 The differences in CPu hemodynamics mentioned above, aside from the distinct vascular source, may result from projection densities (i.e., mid-brain dopamine projections), non-columnar circuit structure, and unique neuronal composition.
Unlike cortex, which is composed largely of glutamatergic neurons, the majority of CPu is composed of inhibitory, γ-Aminobutyric acid (GABA) neurons including medium spiny neurons (MSNs) and interneurons. Additionally, CPu GABAergic neurons release neuropeptides known to have vasomodulatory properties in the cortex, 26 but their respective contributions in the CPu are incompletely documented. 16 Further, the CPu contains the highest expression levels of cholinergic markers in the brain 17 as well as the highest density of dopamine receptors, 27 both of which are vasomodulatory. Given the unique chemical and neuronal composition of CPu outlined above, this review aims to discuss how these factors can influence local neuronal and vascular responses and ultimately affect fMRI signal. As many prominent neurochemical systems have not been widely investigated within the CPu, we also aim to present information relevant to this area from studies on other brain regions, including, but not limited to, the cortex, hippocampus, and cerebellum.
GABA
Origins of GABA in CPu
MSNs are divided into two subclasses based predominately, but not exclusively, on receptor expression and terminal projection site. Approximately half of MSNs express the dopamine type 1 receptor (D1R) and project directly to the basal ganglia output nuclei (i.e., substantia nigra pars reticulata and the internal globus pallidus), these neurons will be termed direct projecting MSNs. The remaining half of MSNs express the dopamine type 2 receptor (D2R) and project indirectly to the basal ganglia output nuclei via the external globus pallidus and the subthalamic nucleus, these neurons will be termed indirect projecting MSNs. The GABAergic MSN projections from the CPu play a major role in the basal ganglia circuit and CPu microcircuit, but these are not the only cells contributing to GABAergic tone in the CPu. Additionally, a minority of the CPu cells are GABAergic interneurons.
Influence of GABAergic transmission in CPu
The electrophysiological properties of GABAergic MSNs as compared to glutamatergic neurons may contribute to differences in hemodynamics between CPu and cortex. MSNs are largely quiescent and their membrane potentials transition between a resting down state and a depolarized up state. 17 These states determine the effects of GABA on MSNs: GABA is depolarizing in the down state, 17 in sharp contrast to its common hyperpolarizing influence on pyramidal neurons. Therefore, it is likely that the metabolic demand per neuronal activation might be different. Electron and light microscopy studies show GABA terminals in the synaptic range of pericytes and some capillaries within the cortex. 28 Optogenetically stimulating GABAergic cortical parvalbumin interneurons in parvalbumin-cre mice anesthetized under ketamine and xylazine increased CMRO2, but responses were generally weaker than from optogenetic GABAergic interneuron activation paired with sensory stimulations or from sensory stimulations alone. This contrasted with excitatory pyramidal neuron stimulation, where optogenetic or combined optogenetic-sensory stimulations evoked greater CMRO2 changes than sensory stimulation alone. 29 These results suggest that GABAergic activity requires less oxygen than glutamatergic activity. While GABAergic signaling may not have intense metabolic demand, there is still evidence for GABA-mediated changes in vascular tone. Interestingly, GABAergic interneuron stimulation in cortex and hippocampus produces either vasodilation, constriction, or a combination of both26,30,31 and stimulating direct and indirect projecting MSNs can also induce condition-dependent BOLD responses in the CPu.32–34 These divergent effects on vascular tone could be a result of GABAergic signaling because specific types of GABAergic neurons and interneurons co-release different vasoactive neurotransmitters 26 and the activation and expression of GABA receptor subtypes is not uniform across brain regions. In support of the potential vasomodulatory role of GABAergic transmission, a recent study reported GABAergic receptor expression on multiple types of perivascular cells, including but not limited to pericytes, smooth muscle cells, endothelial cells, and astrocytes (Figure 1(a)). 9
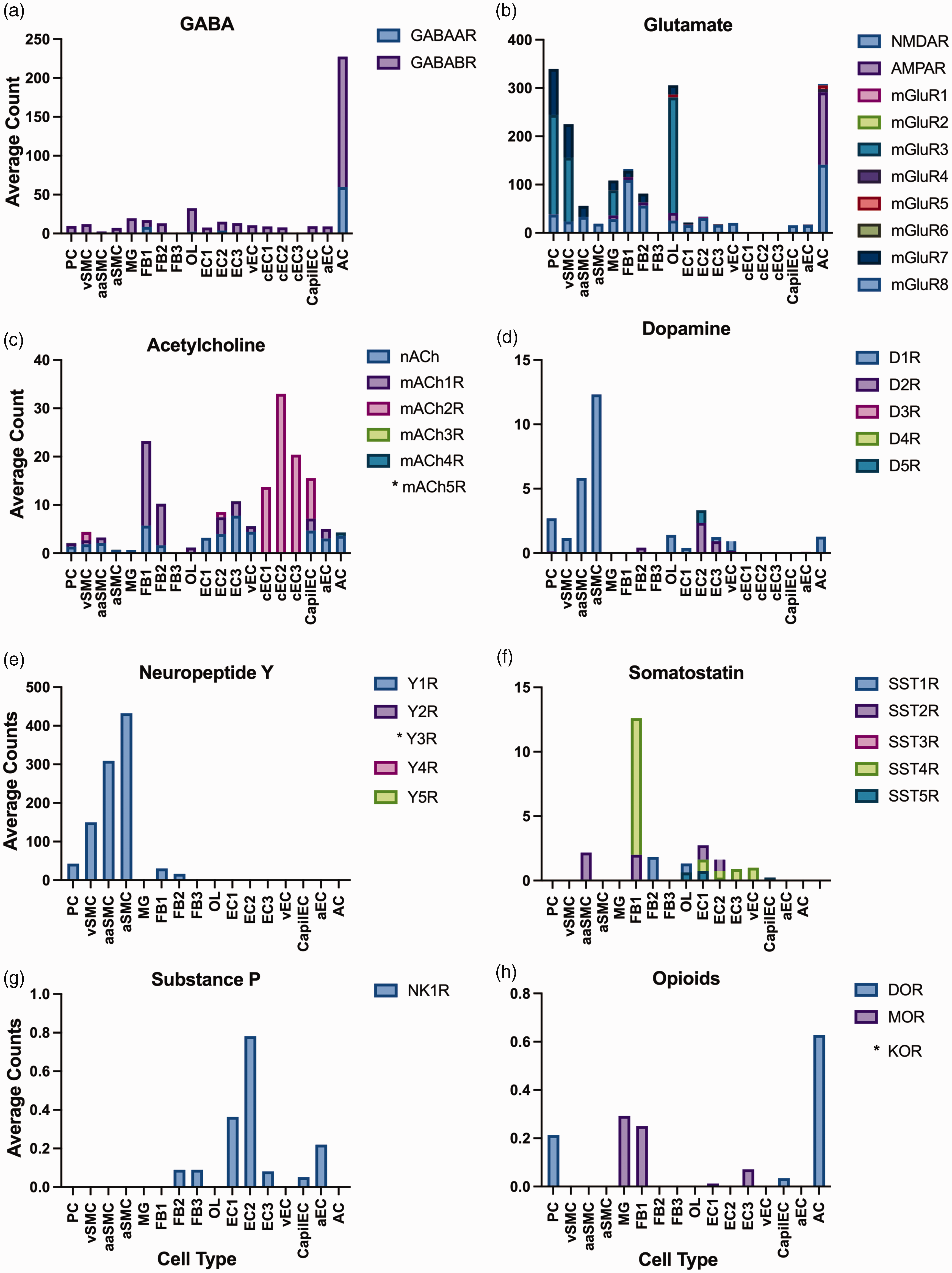
Receptor expression on perivascular cell types. All data were calculated from open-source, single-cell gene-expression data at https://betsholtzlab.org/VascularSingleCells/database.html. For all panels: (a) GABA, (b) Glutamate, (c) Acetylcholine, (d) Dopamine, (e) Neuropeptide Y, (f) Somatostatin, (g) Substance P, and (h) Opioids, average counts reflect the total number of sequence counts for a target gene divided by the total number of cells counted by fluorescent expression of cell-type marker. As several receptor subunits were independently targeted in this database, the average count for each receptor subtype was computed by summing the averages of each subunit and dividing by the number of subunits that make a functional receptor (dimer, pentamer, etc.). Cell type abbreviations: Pericytes (PC), venous smooth muscle cells (vSMC), arteriolar smooth muscle cells (aaSMC), arterial smooth muscle cells (aSMC), microglia (MG), fibroblast-like type 1 (FB1), fibroblast-like type 2 (FB2), fibroblasts-like type 3 (FB3), oligodendrocytes (OL), endothelial cell type 1 (EC1), endothelial type 2 (EC2), endothelial type 3 (EC3), venous endothelial cells (vEC), capillary endothelial cells (cEC), arterial endothelial cells (aEC), astrocytes (AC). A detailed methodology on the classification of each cell type can be found in the manuscript published by He et al., 2018. 9 * denotes receptor expression data not available.
GABAA receptors
GABAAR is an ionotropic receptor found on both neurons and astrocytes. GABAAR activation induces rapid inhibitory postsynaptic membrane hyperpolarization in receptor-expressing cells. 17 GABAARs can be activated either tonically or phasically to generate gamma frequency oscillations associated with hemodynamic responses or decrease the likelihood of neuronal firing, 35 respectively. GABAAR-mediated lateral inhibition also assists in tuning synchronous neuronal activity within the CPu, 17 an activity pattern known to play a role in hemodynamics. 35
Takayasu and Dacey have shown with isolated pressurized arterioles taken from rodent cortex that GABA alone had no effect on arteriole diameter, 36 this is in contrast to increased CBF observed regionally after intravenous administration of GABAAR agonist muscimol in rats anesthetized with halothane, 37 suggesting GABAergic signaling effects on hemodynamics are likely receptor specific, and that activating both receptor types could result in a cancellation of effect. In support of GABAAR specific effects, direct application of muscimol onto rat hippocampal slices dilates hippocampal microvessels and decreases LFPs. 30 To assess if the GABAA vasodilation is mediated by NO, a powerful vasodilator, the NO synthase inhibitor Nω-nitro-L-arginine (L-NNA), was applied but had no effect on hippocampal vasodilation, suggesting this effect is independent of NO. 30 Intriguingly, this conflicts with results from a more recent study using intrinsic imaging and laser doppler flowmetry, where L-NNA significantly diminished the vasodilatory response in mice following optogenetic stimulation of vesicular GABA transporter-expressing cortical neurons without attenuating neuronal activity. 38 As these experiments did not selectively manipulate GABAARs, the discrepancy may be related to downstream NO release signaling via other pathways. To determine how glutamate contributes to the vascular response during optogenetic stimulation of GABAergic cortical interneurons in mice, Anenberg et al. applied a cocktail of ionotropic glutamate receptor (iGluR) antagonists 2,3-dioxo-6-nitro-7-sulfamoryl-benzo[f]quinoxaline (NBQX) and dizocilpine (MK-801) and reported no effect on CBF measured using laser speckle contrast and intrinsic imaging, in mice anesthetized with ketamine and xylazine. 39 When GABAAR antagonist picrotoxin was added to the cocktail, the peak CBF response was slightly attenuated, but not to the level of statistical significance. 39 These results were replicated by Vazquez et al. in mice anesthetized with ketamine and xylazine, suggesting that GABAAR-mediated vasodilation is independent from iGluR signaling. 38 Further, Mueggler et al. and Reese et al. both showed increased CBV fMRI signal in the primary somatosensory and motor cortices that scaled with elevating concentrations of the systemic GABAAR antagonist bicuculline. Both studies also showed that the response profile to bicuculline is brain region-specific, 40 with cortex vasodilating in a time-dependent manner and CPu responding with a more complex two-phase pattern. 41
GABAB receptors
GABABRs are G protein-coupled receptors (GPCRs) found on axons and dendrites. GABABRs exert their actions by inhibiting neurotransmitter release and modulating neuronal excitability. GABABRs reduce glutamatergic transmission via cortical and thalamic inputs within the CPu. 17 Additionally, MSNs elicit inhibitory feedback control onto each other via GABABR-mediated inhibition. 17 Perfusion of the GABABR agonist baclofen onto rat hippocampal slices constricts microvessels, but perfusion of the GABAAR agonist muscimol dilates microvessels even in the absence of cell firing. 30 However, such an effect could be region dependent, as electrical stimulation of Purkinje cells and climbing fibers in the cerebellum under applied baclofen altered synaptic activity but not CBF, as measured by laser doppler in rats anesthetized with halothane or isoflurane. 42
GABAAR and GABABR activations primarily inhibit neuronal activity and may induce vasodilation and vasoconstriction, respectively. Several studies highlight that GABAergic receptor signaling can modulate neuronal activity, vascular tone, and/or metabolism in a concentration- and brain region-dependent manner. These studies must be interpreted cautiously, as most are slice preparations that may damage synapses 43 and are known to have a lack of myogenic tone, blood flow and maintenance of pressure within arterioles. 44 These factors can significantly influence neuronal and hemodynamic activity in these preparations and should be considered in the interpretation of results. The induced vascular responses could result from relatively direct (e.g., GABAergic receptor activation on perivascular cells) or indirect (e.g., via GABAergic receptors on neurons and astrocytes) transmission. Closer examination of direct versus indirect influences of GABA on vascular tone in a predominately GABAergic brain region such as the CPu may yield divergent results.
Glutamate
Origins of glutamate in CPu
Glutamate is a key regulator of MSN activity and exerts its effects via ionotropic receptors (iGluRs) and metabotropic receptors (mGluRs). The CPu lacks a local source of glutamate; instead, glutamate predominately comes from cortical and thalamic projections. Corticostriatal projections originate from sensory, motor, association, and prefrontal cortical regions, and their corresponding striatal targets are topographically organized. 17 The thalamic input to the CPu is often thought of as a relay to convey information from the basal ganglia to the cortex and among cortical nuclei. Like the corticostriatal projections, thalamostriatal projections are organized topographically. 17
Influence of glutamatergic transmission in CPu
The relationship between glutamatergic signaling and vascular activity has been examined by fMRI in rats anesthetized with urethane. Studies have reported local tissue oxygen and CBF increases in cortex and hippocampus following glutamate microinfusions, 45 and that picomole glutamate ejections via iontophoresis increase neuronal spiking but decrease local tissue oxygen in cortex and CPu. 46 Meanwhile, synaptic glutamatergic transmission is known to evoke vasodilation 47 in several ways, such as: binding to neuronal iGluRs to initiate neuronal NO synthesis and release as a volume transmitter, 48 binding to astrocytic mGluRs to promote signaling cascades that release AA metabolites, and binding with gliotransmitter co-agonist d-Serine on endothelial N-methyl-D-aspartate receptors (NMDARs) to produce endothelial NO. 49 In rats anesthetized under α-chloralose, activation of cortical inputs increases local CPu neuronal activity, but promoting activity at the cortical region or driving CPu neuronal activity does not consistently result in corresponding positive BOLD, CBF, or CBV increases in the CPu.32,34,50–56 Though both iGluRs and mGluRs influence vascular tone, the underlying mechanisms differ depending on receptor subtype and location. In support of the potential vasomodulatory role of glutamatergic transmission, a recent study has reported glutamatergic receptor expression on multiple types of perivascular cells including but not limited to pericytes, smooth muscle cells, endothelial cells, and astrocytes (Figure 1(b)). 9
Ionotropic glutamate receptors
The two predominant iGluRs are NMDARs and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptors (AMPARs), which are cation permeable, predominately postsynaptic, and responsible for transmitting quick excitatory responses. Stimulating glutamatergic projections in prefrontal cortex evoked biphasic tissue oxygen changes in CPu, but iGluR blockade via 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX) and (2 R)-amino-5-phosphonovaleric acid (AP5) iontophoresis attenuated the initial oxygen decrease with no effect on the subsequent oxygen increase. 46 The following section highlights the individual contributions of each iGluR type to vasodilation.
A known mechanism by which glutamatergic NMDAR activation induces vasodilation is via the synthesis and release of NO by NMDAR-expressing neurons. 45 Pharmacologically evoked vasodilation, measured via cranial window microscopy in the parietal cortex of rabbits anesthetized with pentobarbital, was abolished in the presence of either NMDAR antagonist MK-801 or tetrodotoxin. 57 Similarly, Yang and Chang blocked increases in CBF, measured by laser doppler flowmetry, evoked in response to NMDA but not acetylcholine in the parietal cortex of rats anesthetized with pentobarbital by applying either MK-801, tetrodotoxin, or 7-nitroindazole, which inhibits NO synthase from neuronal sources. 58 However, NMDARs also express on endothelial cells, and isolated pressurized middle cerebral arteries dilate via endothelial NO synthase in response to the co-binding of gliotransmitter D-serine and glutamate to NMDARs. 59 To elucidate the involvement of NO synthase from endothelial sources in this cascade, Stobart et al. evoked vasodilation in murine cortical slices via astrocytic Ca2+ uncaging and determined that endothelial NO synthase inhibition by L-N5-(1-iminoethyl)ornithine significantly decreased this vasodilatory response. 60 Together, these results suggest that while NMDAR activation leads to vasodilation, additional studies are needed to determine whether such dilation is attributed to downstream neuronal NO synthase signaling, endothelial NO synthase signaling, or both.
Glutamate can also mediate vasodilation via AMPAR activation. Gsell and colleagues showed the involvement of AMPARs in glutamate-mediated vasodilation via the reversable yet immediate attenuation of increased BOLD and relative CBV changes to forepaw stimulation in the cortex of rats anesthetized under α-chloralose by AMPAR antagonist GYKI-53655. 61 Using in vivo two-photon imaging, Ohata et al. corroborated these results by showing vasodilation following AMPA perfusion in the cortex of rats anesthetized with halothane. 62 In the same experimental preparation, NMDA perfusion also evoked vasodilation, but NO synthase inhibitor L-NNA only attenuated the NMDAR-mediated vasodilation and had no effect on the AMPAR-mediated vasodilation. 62 These studies indicate that AMPARs can be involved in vasodilation in ways that differ mechanistically from NMDAR-mediated vasodilation.
Metabotropic glutamate receptors
mGluRs are GPCRs divided into 3 groups: group 1 (mGluR1 and 5), group 2 (mGluR2 and 3) and group 3 (mGluR4, 6, 7, and 8). Both group 1 and group 2 mGluRs are found on neurons and astrocytes, and group 3 mGluRs express presynatically on neurons. In addition to modulating neuronal excitability, they also influence astrocytic activity. Both cultured murine bEND563 and human hCMEC/D364 brain endothelial cells directly respond to glutamate application by increasing intracellular Ca2+ and releasing NO. These effects of glutamate on endothelial cells can be mimicked by applying a selective group 1 mGluR agonist 64 or blocked by BAPTA or mGluR1 antagonist MCPG.63,64 Further, the mGluR agonist tACPD increases the release of NMDA receptor co-agonist D-Serine, which induces vasodilation, and degradation of D-Serine blocks this effect. 60 In cerebellar slice preparations, bath application of glutamate resulted in vasoconstriction that was reversed by antagonizing mGluR1 and AMPA/kainate receptors, but not NMDA receptors. 65 Inhibiting phospholipase A2, which aids in the release of AA and is found in glia and Purkinje cells, using MAPF also inhibited the glutamate induced vasoconstriction. 65 In addition, firing of Purkinje cells expressing endothelin 1, a potent vasoconstrictor, induced vasoconstriction. 65 Although glutamatergic signaling-induced vasoconstriction is rarely reported, the findings in cerebellum compared to those in cortex or cell culture preparations highlight the regional specificity of glutamatergic neurotransmitter signaling to vasomotility.
In summary, in vivo and in vitro activation of iGluRs and mGluRs generally induce neuronal activation and vasodilation but could also lead to vasoconstriction in select brain regions like the cerebellum. Activation of these receptors induces the production and release of NO, a potent vasodilator. Though iGluR receptor subtypes have been comprehensively studied, the specific contributions of various mGluR subtypes to vascular activity are less well-characterized, and thus more systematic studies of mGluRs are needed to inform how glutamate influences hemodynamics in CPu.
Acetylcholine
Origins of acetylcholine in CPu
Acetylcholine is often considered an excitatory neurotransmitter, but it can also downregulate or inhibit cellular activity due to the complex distribution of acetylcholine receptor subtypes on cells, and the configuration of those cells within local circuits. 66 The CPu has high levels of acetylcholine, cholinergic receptors, and enzymes related to the synthesis and breakdown of acetylcholine; 17 thus, it follows that CPu function is heavily influenced by cholinergic signaling. Acetylcholine releasing neurons are divided into cholinergic interneurons and cholinergic projection neurons. While cholinergic interneurons are the major source of acetylcholine in the CPu, the pedunculopontine and the laterodorsal tegmental nucleus also send spatially organized cholinergic projections to CPu. 67
Influences of acetylcholine in CPu
In the CPu, cholinergic interneurons express dopamine receptors, iGluRs, GABAARs, and cholinergic receptors, and are capable of co-transmitting glutamate or GABA with acetylcholine in a brain region dependent manner. 68 These interneurons switch between autonomous single-spike firing and bursting activity. Cholinergic interneurons maintain tonic levels of acetylcholine via autonomous firing, which also regulates glutamatergic tone within the CPu via pre- and post-synaptic mechanisms. 17 Further, cholinergic interneurons can be inhibited via reciprocal connections with GABAergic interneurons. 17 Inhibiting cholinergic interneurons reduces tonic acetylcholine levels in CPu and may facilitate changes in local microcircuitry that lead to aberrant behaviors or pathology. 66 It is worth noting that brainstem cholinergic afferents to CPu inhibit MSNs and excite cholinergic interneurons, 69 but do not modulate dopamine release. 70
While modern tracing, imaging, and optogenetics studies have shown that acetylcholine can signal via fast synaptic transmission, 71 acetylcholine is classically considered a volume neurotransmitter. Acetylcholine could affect vascular tone directly by extending extrasynaptically to reach blood vessels and perivascular cells in acetylcholine-rich brain regions like the CPu. Axon terminals expressing choline acetyltransferase, the enzyme responsible for acetylcholine synthesis, have been found near endothelial cells, 72 and endothelial cells within the rat cortex were also found to be immunoreactive for choline acetyltransferase. 72 As acetylcholine is easily broken down in blood by acetylcholinesterase, it is posited that endothelial cells likely take up choline and then synthesize acetylcholine.
Zaldivar et al. showed that injecting acetylcholine into the macaque visual cortex elicited a spatially specific response. 73 Near the injection site, baseline CBF and BOLD increased, but visual stimulation evoked vascular response amplitudes decreased, as did LFP and MUA. 73 Farther from the injection site, vascular baselines remained the same but vascular and LFP evoked responses increased. 73 In another fMRI study of rats anesthetized with isoflurane, Hoff et al. reported cortical and hippocampal vasodilation in response to the mAChR agonist pilocarpine, as expected, but also reported robust vasoconstriction in CPu. 74 These differences between brain regions could be related to differences in recruited acetylcholine receptor subtypes and the proximity to acetylcholine release sites. Acetylcholine has two receptor types, nicotinic and muscarinic, which express differently in cortex versus CPu. In support of the potential vasomodulatory role of specific acetylcholine receptor types, a recent study has reported acetylcholine receptor expression on multiple types of perivascular cells, including but not limited to pericytes, endothelial cells, smooth muscle cells, and astrocytes (Figure 1(c)). 9
Nicotinic receptors
Nicotinic acetylcholine receptors (nAChRs) are ionotropic receptors that are responsible for fast, phasic cholinergic signaling. They are composed of alpha (α2-10) and beta (β2-4) subunits. Within the CPu, nAChRs are expressed on corticostriatal terminals, 75 and cholinergic, and GABAergic interneurons. 76 It is well documented that nAChRs expressed on presynaptic DA terminals increase DA release independently from cell firing in midbrain structures. 75 Cholinergic interneurons modulate each other indirectly via nAChR activation on GABAergic interneurons. 76 MSNs do not express nAChRs, but activating nAChRs on GABAergic interneurons leads to di-synaptic MSN inhibition. 76 Further, nAChRs exist on vascular beds throughout the central nervous system. 77 Suarez et al. showed that β2 KO mice anesthetized with ketamine/xylazine exhibited reduced BOLD activation in response to subcutaneous nicotine injection across cortical and subcortical brain regions, including the nucleus accumbens. 78 Similarly, intravenous nicotine injection in rats anesthetized with isoflurane increased BOLD across the brain, including the CPu, in a dose-dependent manner. 79 This effect was blocked by pretreatment with mecamylamine, a nAChR antagonist. 79 Likewise, in another study, activation of nAChRs by intravenous administration of nicotine or α4β2 agonist 5-iodo-3-(2(S)-azetidinylmethoxy)pyridine increased CBV fMRI within the cortex and subcortical nuclei including the nucleus accumbens in rats anesthetized with halothane. 80 These findings are consistent with human imaging studies, as Tanabe and colleagues reported that nicotine administration significantly increases CBF within the human ventral striatum. 81
Muscarinic receptors
mAChRs are GPCRs that play a role in tuning excitatory and inhibitory synapses. The five mAChR receptor sub-types are categorized as either stimulatory Gq/11 (M1R, M3R, and M5R) or inhibitory Gi/o (M2R and M4R) receptors, and express nonuniformly across most brain regions and cell types. 82 Unlike cortex, which predominantly expresses post-synaptic M1Rs and pre-synaptic M2Rs, 82 the CPu broadly expresses all mAChR sub-types. 83 The predominantly expressed mAChRs in the CPu are M1Rs and M4Rs, followed by M2Rs, M3Rs and M5Rs.84,85
Several studies point to the potential for mAChR modulation of vascular tone. Examination of human cerebral cortex mAChR expression by polymerase chain reaction revealed M2Rs and M3Rs on microvessels, M2Rs and M5Rs on endothelial cells, all mAChRs except M4Rs on smooth muscle cells, and all sub-types on astrocytes. 86 Pharmacologically enhancing acetylcholine increased cortical CBF, as measured by laser doppler flow during whisker pad stimulation in rats anesthetized under urethane. This effect could be attenuated by the nonselective mAChR antagonist scopolamine but not the nAChR antagonists mecamylamine and methylycaconitine. 87 Yamada et al. showed via cranial window that concentration-dependent vasodilatory effects of acetylcholine were absent in M5R knockout mice anesthetized with pentobarbital, but that vasodilative signaling cascades downstream of M5R activation remained functional. 88 Pharmacological injection of the non-specific muscarinic agonist pilocarpine, at a non-seizure inducing dose in awake animals, resulted in region-dependent CBV fMRI responses, where vasodilation was seen in cortex, thalamus, and hippocampus but vasoconstriction was seen in the CPu. 89 fMRI studies have revealed that the M4R positive allosteric modulator VU0467154 significantly attenuated D1R agonist-induced CBV increases in the substantia nigra and primary motor cortex in rat, but had no effect on CBV in CPu, sensory cortex, or cingulate cortex.90 This contrasts with the effect of M1R/M4R preferring muscarinic agonist xanomeline, which increased CPu BOLD and oxygen in mice anesthetized with halothane when administered alone and attenuated BOLD increases that were evoked via the NMDAR antagonist phencyclidine. 91
Acetylcholine produces concentration-dependent, bidirectional responses within the cortex via differential involvement of cholinergic receptors. In summary, activation of nAChRs induces vasodilation in both cortical and sub-cortical regions, whereas broad activation of mAChRs induces region-dependent responses with vasoconstriction specific to the CPu. Intriguingly, some mAChR subtypes (M1Rs and M4Rs) evoke vasodilation instead of vasoconstriction within the CPu when activated. To fully understand acetylcholine influence over vascular responses in the CPu, further studies are needed to determine the mAChR(s) responsible for vasoconstriction.
Dopamine
Origins of dopamine in CPu
The CPu has some of the highest dopaminergic tone in the brain. 27 Dopaminergic neurons project to the CPu primarily from the substantia nigra pars compacta, and to the nucleus accumbens from the ventral tegmental area. Nigrostriatal dopamine neurons fire spontaneously and shift between different modes of firing to control tonic and phasic dopamine release in CPu. 17 Burst firing evokes phasic dopamine release heterogeneously across the CPu depending on the burst duration and frequency, with higher tonic-level evoked dopamine release in CPu but higher phasic-level evoked dopamine release in nucleus accumbens. 92 These properties enable dopamine neurons to continuously alter dopaminergic tone and precisely tune CPu neuronal activity.
Influence of dopamine in CPu
Dopamine influences neuronal activity through synaptic or volume neurotransmission via 5 GPCRs (D1-D5Rs). D1 and D5Rs are excitatory Gq/11-coupled and characterized as D1-like receptors (D1/D5Rs) while D2-D4Rs are inhibitory Gi/o-coupled and characterized as D2-like receptors (D2/D3/D4Rs). D1Rs express postsynaptically on direct projecting MSNs, interneurons, and on microvessels. 27 D2Rs express pre and postsynaptically 93 on indirect projecting MSNs, interneurons, and sparsely on microvessels. 27 D4Rs are expressed on both direct and indirect projecting MSNs, pre and postsynaptically. 17 Endothelial cells express D3Rs and D5Rs, as do cortical microvessels and capillaries, but D5Rs additionally express on microvessels and capillaries in CPu. 27 Finally, all but D4Rs are expressed on astrocytes in both the CPu and cortex.9,27
Dopamine can excite or depress neuronal firing in CPu depending on the receptor type and the concentration of dopamine released. 17 Dopamine also plays a role in inhibitory transmission as midbrain dopaminergic neurons projecting to CPu co-release GABA, 94 as opposed to midbrain dopaminergic neurons projecting to the nucleus accumbens which co-release glutamate. 94 Stimulating dopaminergic neurons evokes neuronal firing, dopamine release, 95 and increased fMRI responses in CPu96–99 with few exceptions. Blocking dopamine synthesis attenuates evoked dopamine and spiking responses 100 but does not affect evoked oxygen increases in rats anesthetized with urethane, 101 which conflicts with observations that broad dopamine receptor antagonism attenuates dopamine-evoked BOLD increases in awake rats. 96 With receptor expression on vasculature, perivascular cells, and neurons, it is possible that dopamine affects hemodynamics in CPu via both neuromodulatory and vasomodulatory mechanisms that differ between experimental paradigms.
Cortical capillaries were found to constrict with bath application of dopamine in slice preparations, 102 whereas both cortex and CPu showed robust vasodilation in response to pharmacological amphetamine challenge, 103 which greatly increases extracellular dopamine via dopamine transporter blockade and reversal. It should also be noted that dopamine depletion has been associated with decreased pain-induced vasoconstriction in the CPu, in rats anesthetized with α-chloralose. 104 These results are therefore likely the result of differential receptor activation. In support of the potential vasomodulatory role of specific dopamine receptors, a recent study has reported dopamine receptor sub-type expression on multiple types of perivascular cells including but not limited to pericytes, smooth muscle cells, and endothelial cells (Figure 1(d)). 9
Dopamine D1/D5 receptors
D1/D5Rs have the highest expression among dopaminergic receptors throughout the brain. Within the rat CPu the D1 to D2R ratio is almost 3:1. 105 D1/D5R activation increases direct projecting MSN and parvalbumin interneuron excitability and GABAA inhibitory post synaptic current amplitudes in postsynaptic neurons. 106 In addition, dopamine was shown to trigger action potentials in persistent low threshold spiking interneurons via D1/D5Rs 107 and dopamine is known to modulate glutamatergic transmission through interactions between D1/D5Rs and NMDARs. 108
Chen et al. observed that administration of the D1/D5R agonist, dihydrexidine increased CBV fMRI globally in the rat brain, with some of the largest increases in the CPu. 109 However, they determined that this effect is developmentally dependent as D1/D5R expression is lower in younger rats. 109 In rats anesthetized with isoflurane, the D1R antagonist SCH-23390 attenuated CBV fMRI increases in CPu evoked via selective dopamine neuron stimulation during fMRI, 97 but a separate study in rats anesthetized with medetomidine found no effect on ventral tegmental area stimulation-induced BOLD increases, 110 though the lack of agreement could be due to differences in drug concentration or stimulus modality.
Dopamine D2/D3/D4 receptors
D2Rs are located on all cell types within the CPu and can decrease glutamatergic, GABAergic, cholinergic, and dopaminergic transmission when activated. 111 D4Rs are found exclusively on MSNs and MSN terminals, 112 and D3Rs are preferentially expressed in nucleus accumbens compared to CPu. 113 Dopamine binds to D2/D3/D4Rs with higher affinities compared to D1/D5Rs. This is important for interpreting fMRI data acquired during development when D2/D3/D4Rs outnumber D1/D5Rs, as drugs that increase endogenous dopamine evoke CBV decreases in juvenile rats anesthetized with halothane, but evoke CBV increases in adult rats. 109 D2/D3/D4R agonist administration results in negative CBV fMRI responses,27,103,109 while D2/D3D4R antagonist administration reduces CBV or augments CBV fMRI changes in the cortex and CPu.27,50,103 Few studies have examined the effects of D3R binding on the vascular response due to the lack of pharmacological agent specificity for the receptor; however, the effects of D3R binding have been studied using agonists and antagonists at concentrations preferable to D3R binding. In rats anesthetized with halothane, intravenous D3R antagonist administration evoked stronger CBV fMRI responses in the nucleus accumbens than in the CPu, 114 while D3R agonist decreased CBF across both cortical and sub-cortical areas during PET imaging of non-human primates sedated with 70% N2 and 30% oxygen. 115 Lastly, high doses of D3R agonist result in differential responses across cortical layers, with layers 2/3 and 4 showing negative CBV changes and layers 5 and 6 showing positive CBV changes with fMRI. These response differences may be related to relative D1/D5R to D2/D3/D4R densities. 114
Compared to other neurotransmitter systems, the dopamine system is relatively well studied with regards to its influence on vascular tone. The influence of dopamine on vascular responses depends on dopamine concentration, differential activation of receptor subtypes, receptor densities, and developmental stage. D1/D5R agonism results in vasodilation while D2/D3/D4R agonism results in vasoconstriction, however the contributions of specific receptor subtypes have not been fully explored within the limitations of current pharmacological manipulations. Further, dopamine modulates numerous neurotransmitter systems in the CPu via either D1/D5Rs or D2/D3/D4Rs. Therefore, it is of interest for future studies to investigate how these interactions shape vascular tone in CPu.
Neuropeptides
Origins of neuropeptides in CPu
Neuropeptides are large neurotransmitters released from dense core neuronal vesicles that can travel microns away via volume transmission to bind to GPCRs. Neuropeptidergic signaling in CPu is of interest for addiction and pain research, as well as neuropathologies such as Parkinson’s and Huntington’s disease. 17 Of relevance is that nociceptive stimuli have been consistently shown to induce robust negative CBV fMRI changes in the CPu of rats anesthetized with α-chloralose or isoflurane.50,54 MSNs and persistent low threshold spiking interneurons in CPu are known to release one or more neuropeptides. By co-releasing with glutamate or GABA, 17 neuropeptides allow neurons to chemically modulate neuronal and vascular systems on both rapid and slow time scales that vary depending on the neuropeptide identity and receptor subtype to which it binds. Below we review neuropeptides according to their origin within the CPu.
Somatostatin and neuropeptide Y
Neuropeptide Y and somatostatin are expressed throughout the central and peripheral nervous systems. Within the brain, neuropeptide Y has relatively high expression levels in the cortex and CPu, 116 whereas somatostatin has much higher expression levels in the hypothalamus and amygdala versus the CPu. 117 These neuropeptides are released by GABAergic interneurons via Ca2+-dependent mechanisms. Neurogliaform interneurons in the CPu express only neuropeptide Y, whereas over 75% of persistent low threshold spiking interneurons express somatostatin in addition to neuropeptide Y. 118 Cholinergic interneurons and MSNs also receive inputs from somatostatin-expressing GABAergic projection neurons located in primary and secondary motor cortex, respectively. 119 Most persistent low threshold spiking interneurons in CPu fire spontaneously, but neurogliaform interneurons do not. 17 Both persistent low threshold and neurogliaform interneurons inhibit MSNs via synaptic GABAARs. 17
Substance P, dynorphin, and enkephalin
Substance P is highly expressed in axon terminals of direct projecting MSNs within the CPu and is released following high frequency firing of direct projecting MSNs that synapse onto indirect projecting MSNs, cholinergic interneurons, and NO synthase-expressing persistent low threshold spiking interneurons. 17 The periaqueductal gray also sends a small percentage of substance P expressing axons to the ventral CPu. 120 Though substance P is expressed elsewhere in the brain and periphery, the mechanisms controlling substance P within the CPu are largely considered the same as those that regulate direct projecting MSNs.
Dynorphin and enkephalin are endogenous opioid receptor ligands that are expressed highly within the CPu and dopaminergic inputs to CPu. 17 Dynorphin is released from direct projecting MSNs and enkephalin is released from indirect projecting MSNs. Dynorphin and enkephalin release in CPu is regulated by the same mechanisms that regulate direct projecting MSNs and indirect projecting MSNs, respectively. Dynorphin and enkephalin expression is enriched in striosome and matrix compartments of the CPu, respectively. 17
Influence of neuropeptides in CPu: Somatostatin and neuropeptide Y
Somatostatin and neuropeptide Y each bind to multiple GPCR subtypes that are heterogeneously expressed throughout the central nervous system. Somatostatin binds to SST1-5 receptors (SST1-SST5Rs), and SST2Rs have A and B isoforms. Human endothelial cells express SST1 and SST2Rs, smooth muscle cells express SST2 and SST4Rs, and astrocytes express SST2Rs. 31 Within the CPu, SST1Rs express presynaptically, SST2 and SST5Rs postsynaptically, and SST3Rs extrasynaptically. 121 Neuropeptide Y binds to Gi/o-coupled Y1-8 receptors (Y1-Y8Rs), though only Y1, Y2, Y4, and Y5Rs are expressed and maintain functionality in mammals. Only the Y1R appreciably expresses on human vascular cells. 122 In mice, Y1R has been found on perictyes and smooth muscle cells (Figure 1(e)),9 and all SSTRs except SST3R are present on endothelial cells, with SSTR also expressed on smooth muscle cells (Figure 1(f)),9 suggesting potential direct and indirect influences of somatostatin and neuropeptide Y on vascular tone.
Somatostatin and neuropeptide Y can indirectly influence vascular changes by interacting with other neurotransmitter systems in CPu. Bath-applied neuropeptide Y was found to decrease glutamate release in rat CPu slices. 123 Somatostatin has been shown to dose-dependently increase dopamine, acetylcholine, GABA, and glutamate release in CPu via glutamatergic mechanisms in rats anesthetized with urethane. 124 Inhibition of GABAergic transmission 125 and decreased glutamate release have also been observed at high neuropeptide Y concentrations within the CPu in rats anesthetized with chloral hydrate. 125 SST2Rs may be responsible for somatostatin-mediated dopamine release in CPu. 124
Stimulation of neuropeptide Y-expressing interneurons in cortex induces vasoconstriction in slice preparations. 31 Stimulating the superior cervical ganglion resulted in a robust CBF reduction within the CPu as measured by audioradiography in rats anesthetized with halothane, though local glucose metabolism was unaffected. 126 More recently, two-photon imaging in awake mice and mice anesthetized with α-chloralose and pancuronium showed that optogenetic stimulation of GABAergic cells within the sensory cortex induced biphasic vascular responses in local arterioles, where vasodilation was followed by Y1R-mediated vasoconstriction. 26 Using a Y2R agonist, Lee et al. reduced hypoperfusion and neuroinflammation in a rat model of cardiac arrest, 127 making the neuropeptide Y autoreceptor a potential target to improve blood flow to the brain following ischemic insult.
Activation of the broad range of somatostatin receptors expressed on vascular cells often induces vasoconstriction, as has been measured both in vitro and in vivo using microscopy, but this effect can differ depending on the vessel source. 128 Colocalized SST4Rs and GABABRs in cortical murine astrocytes responded to bath-applied somatostatin with increased Ca2+ activity in brain slices, even in the presence of a GABABR antagonist. 129 Selective stimulation of somatostatin expressing interneurons during laser doppler flowmetry in awake, head-fixed mice produced a local CBF increase, sometimes surrounded by vasoconstriction, followed by an undershoot, 130 but it remains obscure whether these changes are specific to downstream somatostatin, neuropeptide Y, or GABAergic signaling. In support of somatostatin and neuropeptide Y-selective vasoactive effects, both neuropeptide Y-expressing interneuron activation and direct neuropeptide Y or somatotostatin perfusion evoked local vasoconstriction in rat cortical slices, whereas activation of other GABAergic interneuron types and perfusion of their corresponding neurochemicals produced vasodilation. 31
Influence of neuropeptides in CPu: Substance P, dynorphin, enkephalin
Substance P binds to the type 1 neurokinin receptor (NK1R), a GPCR that has been proposed to couple to various intracellular pathways. NK1R quickly internalizes after activation and leads to morphological changes in the expressing cell. NK1Rs express postsynaptically on cholinergic interneurons and NO synthase-expressing persistent low threshold spiking interneurons 17 but have also been found on rodent endothelial cells (Figure 1(g)). 9 Thus, substance P could lead to neurovascular changes via different mechanisms depending on the cell type of the activated receptor.
Presynaptic NK1Rs have been found on asymmetric boutons from glutamatergic afferents, providing a mechanism through which substance P can modulate and potentiate excitatory neuronal activity in CPu. 17 Direct-projecting MSN stimulation was found to excite cholinergic interneurons via substance P release, which could be replicated via bath application of substance P and blocked via an NK1R antagonist. 131 With widespread transmission throughout the central nervous system, these mechanistic differences must be taken into consideration when studying the effect of substance P on neuronal activity in CPu cell types.
Studies examining the effect of substance P on major vessels extracted from felines reveal that substance P does not show vessel preference and is a more moderate vasodilator compared to acetylcholine, histamine, and adenosine. 132 In addition, studies in both isolated human cortical and pial arteries and isolated canine carotid and basilary arteries show that substance P dilates vessels with a endothelial-dependent mechanism that is partially NO dependent.133,134 NO synthase antagonist L-NNA completely blocks substance P-induced relaxation in approximately half of vessels and partially blocks substance P-induced relaxation in the remainder, hinting at the involvement of NO-mediated substance P vascular relaxation. 133 Together this evidence suggests that the dilatory effects of substance P binding are in part mediated by NO.
Substance P release may also lead to vasodilation through other vasoactive neurotransmitters. NK1R activation in the CPu via local substance P perfusion in freely moving rats was shown to increase extracellular, vasodilative, acetylcholine. 135 A microdialysis study found that substance P evoked acetylcholine release with a bell-shaped dose-response curve followed by a long-lasting desensitization against subsequent substance P evoked acetylcholine release, 136 perhaps due to rapid NK1R internalization. A systemic substance P injection increased extracellular dopamine measured by microdialysis in the CPu of freely moving rats over the timescale of hours; 137 however, no increase in dopamine was observed with intra-striatal substance P infusion. 136 These conflicting results may be due to differences in the route of substance P administration or recording location. Direct projecting MSNs that release substance P constitute approximately 70% of cells within substance P immunoreactive striosomes. 138 Thus, neurovascular modulation mediated through substance P may be heterogenous across different CPu domains.
The CPu and its dopaminergic afferents express some of the highest levels of opioid peptide receptors in the brain, though they are heterogeneously distributed. Dynorphin is the primary ligand of κ opioid receptors (KORs) and enkephalin is a ligand for both δ and μ opioid receptors (DORs and MORs, respectively), which are predominately Gi/o- and to a lesser extent Gz-coupled inhibitory GPCRs; however, dynorphin and enkephalin can have functional effects through all three receptors with varying affinities. Direct projecting MSNs highly express MORs over DORs while the opposite is true for indirect projecting MSNs, and KORs preferentially express on direct projecting MSNs versus indirect projecting MSNs. 139 The diversity of opioid receptors with respect to spatial distribution, cellular expression, and ligand affinity could give dynorphin and enkephalin a wide range of potential effects on neuronal activity and hemodynamics in the CPu.
Dynorphin and enkephalin signaling generally inhibits neuronal activity. In the CPu presynaptic MOR and DOR activation inhibits excitatory potentials in MSNs. However, the predominant influence of opioid receptors on CPu microcircuitry may be indirect and dependent upon other neurotransmitter systems. Opioid receptors affect dopamine and acetylcholine release in the CPu, which can continue signaling cascades according to their own neuro- and vaso-modulatory effects. This is demonstrated when administration of naloxone, a general opioid antagonist, or eticlopride, a D2R antagonist, reversed morphine-induced and abolished nociception-induced negative CBV fMRI signals the CPu, highlighting the opioid-dopamine relationship and possible effects on vasomodulation. 50 This contrasts with the increased BOLD activation seen across most of the brain in response to the MOR agonist, oxycodone. 140 Opioids are suspected to be involved in vascular regulation in both the periphery and central nervous system, but how is still poorly understood. In isolated rat cerebral arteries, dynorphin evoked vasoconstriction that was partially mediated through KORs. 141 Conversely, in rats anesthetized with halothane, femoral artery cannulation showed that local dynorphin infusion to the hippocampus decreased mean arterial blood pressure via KORs, 142 suggestive of vasodilation. It is likely that signaling downstream of hippocampus, potentially in hypothalamus, could be responsible for the observed systemic effect. 143 However, it should be noted that the hippocampus has a higher ratio of KORs relative to other opioid receptors than the CPu, 143 so the hemodynamic influence of dynorphin could be different. There is limited evidence that these vascular effects via dynorphin and enkephalin could be through direct signaling to astrocytes or the vasculature. For one, activating DORs expressed on human and rat smooth muscle cells, with DOR agonist [H]-[D-Ala2, Met5] enkephalinamide, has been found to evoke vasoconstriction. 144 In addition, activating a specific MOR subtype (µ3) found on cultured human arterial and rat microvascular endothelial cells evokes vasodilation via endothelial NO release, an effect that was blocked in isolated rat aortic rings with naloxone pretreatment, 145 but the low expression levels of MORs and lack of enkephalin neuropeptide-evoked NO release at the MOR µ3 subtype 146 make it an unlikely means of direct vascular regulation in the CPu. Astrocytes in the CPu also reportedly express messenger ribonucleic acid for all three receptors, highest for DORs and lowest for MORs, with relative expression ratios that differ from cortical, hippocampal, and cerebellar astrocytes in rats. 143 Nonetheless, a recent study did not find brain-wide opioid receptor expression on astrocytes and other perivascular cells (Figure 1(h)), 9 highlighting the need for additional investigations on potential perivascular action sites for dynorphin and enkephalin.
Neuropeptides are released by all MSNs and GABAergic interneurons in the CPu and can affect local vasculature in several ways, including: (1) interacting with various neurotransmitter systems, (2) binding to astrocytes, and (3) binding directly to vasculature and influencing vascular tone. Neuropeptide interactions with other vasoactive neurotransmitter systems can be heavily influenced by the differential expression of receptors throughout the CPu (e.g., matrix versus striosome). Thus, it is unsurprising that neuropeptides have documented concentration and/or region-specific effects. Given the limited evidence for opioid receptor expression on astrocytes and perivascular cells, these complex vascular effects may indicate indirect opioidergic signaling to the vasculature via other cell types or yet undiscovered perivascular action sites. While it is important to determine the direct and indirect effects of neuropeptides on vasculature in the CPu, future studies should also take circuit level effects into account in order to include the influence of other neurochemical systems.
Conclusion
Most studies examining the relationship between neuronal activity and hemodynamics have focused predominantly on the cerebral cortex, where the principal neurons are glutamatergic. Little is known about this relationship in the CPu, where the principal neurons are GABAergic. Additionally, the CPu has been shown to contain distinct cell types and neurochemical distributions compared to cortex. Given the role of the CPu in various behaviors and disorders (Figure 2) and widespread utilization of fMRI to investigate the CPu, a better understanding of hemodynamic influences in the CPu is needed. Here, we summarize how glutamate, GABA, acetylcholine, dopamine, and neuropeptides may have divergent influences on hemodynamics in CPu, as opposed to cortex, due to regional differences in relative concentration, receptors expression levels, and their empirically observed influences on neuronal and vascular activity. While we focus predominately on acute effects, these neurochemicals are crucial for synaptic plasticity and may also gradually tune CPu hemodynamics. In addition to dopamine, there is widespread involvement of serotonin in CPu neurovascular activity and related diseases. 147 Further, the involvement of astrocytes, adenosine, and K+ in the neurovascular unit and neurochemical signaling of AA derivatives which directly influence hemodynamics8,148 cannot be overstated. Nonetheless, these key contributors were omitted to manage the scope of this review, and because detailed discussion of each can be found elsewhere. Other neurochemicals with only sparse distribution in CPu that were not covered herein, such as norepinephrine8,149,150 and vasoactive intestinal peptide, 130 could also have considerable influence on neuronal and vascular activity and should be considered in future studies. It is clear that contributions to neuronal and vascular activity include the confluence of multiple neurochemical systems, where each may produce different effects within a brain region depending on proximal cell types, receptor densities, and concurrent activity. This review points to the need for future studies to examine hemodynamics in areas such as the CPu, to better understand the mechanisms controlling local vascular tone, improve interpretation of fMRI data, and guide preclinical and clinical fMRI investigations of disease states with abnormal neurochemical signaling.
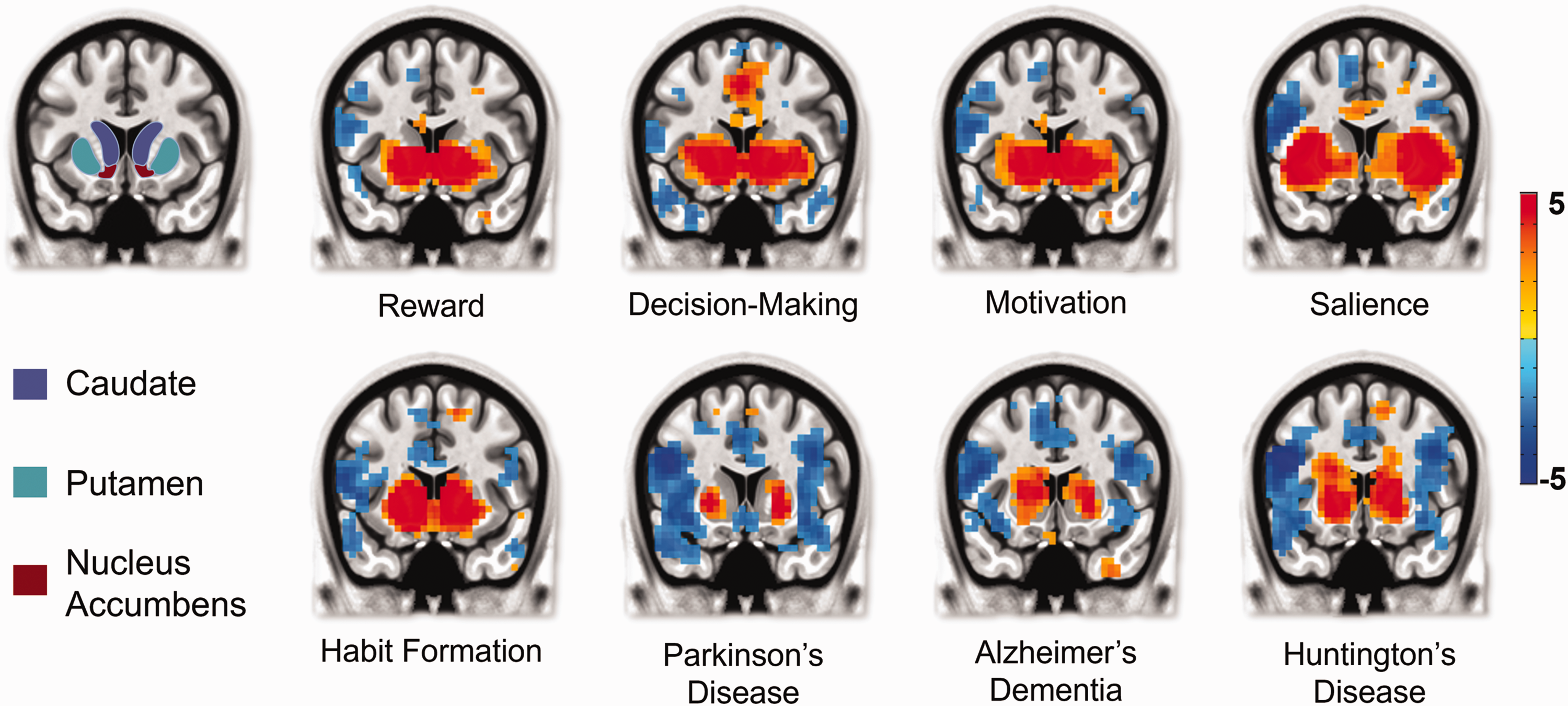
Involvement of striatum in various behavioral and disease states. Meta-analysis was performed by searching key words listed under each behavior or disease using NeuroQuery at https://neuroquery.org. The first image is an anatomical map highlighting the specific areas that compose the striatum in the human brain, specifically the caudate, the putamen and the nucleus accumbens. The following heatmaps make use of NeuroQuery’s ability to scavenge its library of literature and highlight the likelihood that a particular brain region is involved (warm colors) or not involved (cool colors) in a particular behavior or pathology. Colorbar unit is in Z score, representing how likely a given brain location is to be reported in neuroimaging studies tagged with the selected keyword(s). For more detail, please refer to Dockes et al. 2020. 12
Supplemental Material
sj-pdf-1-jcb-10.1177_0271678X221142533 - Supplemental material for Putative neurochemical and cell type contributions to hemodynamic activity in the rodent caudate putamen
Supplemental material, sj-pdf-1-jcb-10.1177_0271678X221142533 for Putative neurochemical and cell type contributions to hemodynamic activity in the rodent caudate putamen by Brittany M Katz, Lindsay R Walton, Kaiulani M Houston, Domenic H Cerri and Yen-Yu Ian Shih in Journal of Cerebral Blood Flow & Metabolism
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Extramural Research Programs of U.S. National Institutes of Health, NIMH (RF1MH117053, R01MH126518, and R01MH111429), NINDS (R01NS091236), NIAAA (P60AA011605 and U01AA020023), and NICHD (P50HD103573).
Acknowledgements
The authors thank Drs. Donita Robinson, Kafui Dzirasa, Thomas Kash, and Eran Dayan for their inputs.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
