Abstract
The circadian clock is a timekeeping system in most organisms that keeps track of the time of day. The rhythm generated by the circadian oscillator must be constantly synchronized with the environmental day/night cycle to make the timekeeping system truly advantageous. In the cyanobacterial circadian clock, quinone is a biological signaling molecule used for entraining and fine-tuning the oscillator, a process in which the external signals are transduced into biological metabolites that adjust the phase of the circadian oscillation. Among the clock proteins, the pseudo-receiver domain of KaiA and CikA can sense external cues by detecting the oxidation state of quinone, a metabolite that reflects the light/dark cycle, although the molecular mechanism is not fully understood. Here, we show the antagonistic phase shifts produced by the quinone sensing of KaiA and CikA. We introduced a new cyanobacterial circadian clock mixture that includes an input component in vitro. KaiA and CikA cause phase advances and delays, respectively, in this circadian clock mixture in response to the quinone signal. In the entrainment process, oxidized quinone modulates the functions of KaiA and CikA, which dominate alternatively at day and night in the cell. This in turn changes the phosphorylation state of KaiC—the central oscillator in cyanobacteria—ensuring full synchronization of the circadian clock. Moreover, we reemphasize the mechanistic input functionality of CikA, contrary to other reports that focus only on its output action.
Most organisms that are exposed to daily light/dark cycles have developed a timekeeping system, called the circadian clock, to anticipate the environmental changes that occur throughout the day. The circadian clock is composed of a central oscillator plus input and output pathways. The central oscillator generates an oscillation with a periodicity of approximately 24 h. The input pathway senses the daily light/dark rhythm and synchronizes the central oscillator to the environmental cycle by aligning the phase of the oscillation to the appropriate time of day. The output pathway controls the physiology of the organism based on the signals produced by the central oscillator. A circadian clock needs to fulfill 3 requirements. First, the rhythm should have approximately 24-h periodicity in constant conditions (free running). Second, the oscillation should be able to be synchronized with the daily rhythm of the environment (entrainment). Third, the period should be stable over a range of temperatures (temperature compensation), which is an important property for precisely keeping track of time despite natural variations in temperature.
A cyanobacterium, Synechococcus elongatus, is a single-celled organism that possesses the simplest circadian clock (Iwasaki and Kondo, 2004). Its central oscillator is composed of 3 proteins, KaiA, KaiB, and KaiC, and can be reconstituted in vitro by mixing these 3 proteins with adenosine triphosphate (ATP) and magnesium (Nakajima et al., 2005). The phosphorylation state of KaiC oscillates with a circadian period, and the structural basis of the mechanism is well known (Tseng et al., 2014; Tseng et al., 2017). KaiA and KaiB activate the autophosphorylation and autodephosphorylation of KaiC, respectively, by changing the A-loop conformation and the binding affinity of magnesium in KaiC (Chang et al., 2015; Jeong et al., 2019; Kim et al., 2008). These antagonistic interactions generate the rhythm of KaiC phosphorylation with a nearly 24-h period (Akiyama et al., 2008). The major contributors of the period of the cyanobacterial circadian oscillator are the reaction speeds of the phosphorylation and dephosphorylation. For a chemical reaction, the speed is dependent on temperature, and the Q10 value is used as an indicator of the temperature dependency of the reaction (Sweeney and Hastings, 1960). The period of the cyanobacterial circadian clock, even as the central oscillator—composed of KaiA, KaiB, and KaiC— by itself in vitro has a Q10 value near 1, indicating that, overall, the oscillator compensates for the temperature dependency of individual reactions (Nakajima et al., 2005).
Moreover, the circadian oscillator needs to be adjusted every day to anticipate the precise timing of the sunrise, which varies every day in nature. Entrainment is a process that aligns the phase of the circadian oscillation with the light/dark cycle. When cyanobacteria receive a light or dark signal from the environment, it is converted into biochemical signals, such as quinone or adenosine diphosphate (ADP) signals (Kim et al., 2012; Rust et al., 2011). Both the input component and the oscillator itself can sense the biochemical signals and adjust the phases of the oscillator to achieve entrainment. In most cases, the signals change the kinase or phosphatase activities of KaiC and affect the phase of phosphorylation, which can shift forward (phase advance) or backward (phase delay) depending on the timing of an input signal, so that the endogenous rhythm can be properly reset. Using this clock-resetting ability, phase advances and delays can be generated in vivo by applying a short dark pulse (~4-h dark duration) at certain times of the day (Kiyohara et al., 2005). The resulting phase shifts can be summarized in a phase-response curve (PRC), which is a characteristic marker of the entrainment properties of an organism (Johnson, 1999).
Previously, some entrainment experiments have been performed in vitro with the central oscillator and the signaling molecules but without the input pathway components. Quinone was reported as a signaling molecule for the entrainment of the cyanobacterial circadian clock because KaiA senses the redox state of quinone, which is oxidized at the onset of darkness and reduced at the onset of light in cyanobacteria (Kim et al., 2012). However, this entrainment study was insufficient because it was performed without the input pathway components, and its PRC did not exhibit any phase delays. Because of the absence of the input component, the signal was sensed only by an oscillator component, KaiA, resulting in an impaired entrainment. This is contrary to the ADP signal that can entrain the circadian oscillator in a complete manner even in the absence of input components (Rust et al., 2011). In this case, KaiC detects the ADP signal directly and adjusts its phosphorylation state by dephosphorylating itself.
CikA, circadian input kinase A, was initially proposed to be an input pathway component (Schmitz et al., 2000), but this was considered as a misnomer after CikA’s function in the output pathway as a phosphatase for RpaA was revealed (Gutu and O’Shea, 2013). Few studies on the input functionality of CikA have been performed, since it was shown that CikA has output functionality. CikA senses the redox state of quinone by selectively binding to oxidized quinone. This binding disrupts the structure of the pseudo-receiver domain and makes CikA levels plummet in vivo (Ivleva et al., 2006). Recently, it was reported that CikA directly interacts with the central oscillator, competing with KaiA for binding to KaiB (Tseng et al., 2017). The possibility of its role in the input pathway arises from CikA’s nature of competitive binding, since the addition of CikA in the circadian oscillator mixture affects the phosphorylation state of KaiC and shortens its period in vitro (Kaur et al., 2019).
In this article, we use the reconstituted cyanobacterial circadian clock with CikA as an input pathway component in vitro to propose the molecular mechanism underlying quinone entrainment.
Materials And Methods
Phosphorylation Assay of the Circadian Clock In Vitro
Cloning, purifications, and phosphorylation assays were performed as described previously without modification (Kaur et al., 2019; Kim et al., 2015). CikA was cloned into the pET41a(+) vector, expressed in Escherichia coli (BL21DE3) and purified with a GST affinity column using the same method as for KaiC (Kim et al., 2015). The in vitro oscillation mixtures were prepared with the Kai proteins and CikA in the circadian oscillation buffer (150 mM NaCl, 20 mM Tris-HCl, 5 mM MgCl2, 0.5 mM EDTA, 1 mM ATP, pH 8.0). Each 20-μL oscillation mixture was collected at 2-h intervals for 2 or 3 days. The collected samples were denatured immediately by adding sodium dodecyl sulfate (SDS) gel loading dye (100 mM Tris·HCl, pH 6.8, 4% SDS, 0.2% bromophenol blue, 20% glycerol, 400 mM β-mercaptoethanol) and kept at −20 °C. To separate the phosphorylated and unphosphorylated KaiC, 6.5% SDS–polyacrylamide gel electrophoresis was used. ImageJ (NIH) was used for the densitometry (Schindelin et al., 2012). Periods and phases were estimated using BioDare2 (https://biodare2.ed.ac.uk/) implementation of the fast Fourier transform–nonlinear least squares procedure with linear detrending (Zielinski et al., 2014).
Statistical Analysis of the Periods
Dunnett’s test was used to compare the period of oscillations under different treatments (CikA concentrations) to a control (absence of CikA). This test was performed using the DunnettTest function in the R package “DescTools” with R version 3.5.1 (Signorell et al., 2019). Tukey’s honest significance difference (HSD) method was used to perform pairwise comparisons of the period of oscillations at different temperatures. This test was implemented using the Tukey HSD function in the R package “stats” (R Core Team, 2018).
Quinone Entrainment Assay
Quinone entrainment assays were performed as described previously (Kim et al., 2012) with the following modification: for the oscillation mixture, we used 10 mM ATP concentration to remove the effect of the ADP/ATP ratio (Rust et al., 2011). The oscillation mixtures were incubated overnight at 30 °C without KaiA and CikA to make KaiC fully dephosphorylate. The time 0 sample was collected immediately after adding KaiA and CikA. Q0 (2,3-dimethoxy-5-methyl-p-benzoquinone) was added to the oscillation mixtures to mimic the beginning of the dark pulse. After 4 h, dithionite (200 μM final concentration) was added to reduce Q0 to mimic the termination of the dark pulse. The final concentration of quinone was 9.6 µM for every entrainment experiment. Data from the later 2 days (from 28 h to 72 h) were uploaded to BioDare2 (https://biodare2.ed.ac.uk/) developed by Andrew Millar (Zielinski et al., 2014) for phase estimation using the cosine fitting method. Because each estimated phase angle is converted to the circadian time (CT) in BioDare2, each phase shift was calculated simply by subtracting the entrained phase (CT 8 or CT 22) from the nonentrained phase (control).
Determination of Magnesium Concentration in Cyanobacteria
Both wild-type and ΔkaiABC cells of Synechococcus elongatus PCC 7942 were grown in a modified BG-11 medium (Bustos and Golden, 1991) with normal aeration in 24-h light-dark cycles (LD:12,12; 50 µE/m 2/s, cool-white fluorescent light in the light phase) at 30 °C. At each time point, 10 mL of the culture (OD750 = 0.35~0.45) was withdrawn, and the cells were washed with 8 mL of ice-cold 0.25 mM EDTA pH 8.0 once and 10 mL of ice-cold deionized water twice by centrifugation. The washed cells were resuspended in ~500 µL of ice-cold deionized water, and the OD750 and chlorophyll a concentrations (Porra, 2002) of the cell suspensions were determined. To extract total metals from the cells, 50 µL of the cell suspension was mixed with 300 µL of nitric acid, incubated at 60 °C overnight, and diluted to 10 mL with Milli-Q water. The magnesium concentrations of those samples were quantified by inductively coupled plasma mass spectrometry (ICP-MS) at the Mass Spectrometry Research Center, Vanderbilt University, as described previously (Wakeman et al., 2016). The total Mg2+ concentration was normalized to OD750.
Results
Adding CikA Compensates for the Decrease in KaiA Concentration in Maintaining the Period of the Circadian Oscillation Reaction Mixture
Because various amounts of KaiA and CikA are known to affect the period and amplitude of the circadian oscillation of KaiC phosphorylation (Kageyama et al., 2006; Kaur et al., 2019), we tried to find the minimum CikA concentration that gives a statistically meaningful effect on the period. Various amounts of CikA (from 0.3 µM to 1.2 µM) were added into the circadian oscillation mixture with various amounts of KaiA (from 0.3 µM to 1.2 µM), and each phosphorylation state of KaiC was monitored for 2 days. The period of each oscillation mixture was estimated and statistically analyzed to find a concentration that leads to the maximum effect on the period (Fig. 1; Suppl. Table S1). With 1.2 µM and 0.9 µM KaiA, we did not find any statistically meaningful change in the period when increasing CikA concentration (Fig. 1A,B; Suppl. Fig. S1). We also did not consider the 0.3-µM KaiA concentration because the amplitudes of the oscillations were not robust enough (Suppl. Fig. S1). The 0.6-µM CikA and 0.6-µM KaiA concentrations were selected for further experimentation because they show a statistically meaningful change and a relatively conserved circadian period (Fig. 1C; Suppl. Fig. S1). One of the criteria for being considered a circadian oscillator is a period near 24 h in a constant environment, and this reaction mixture fulfills that requirement.
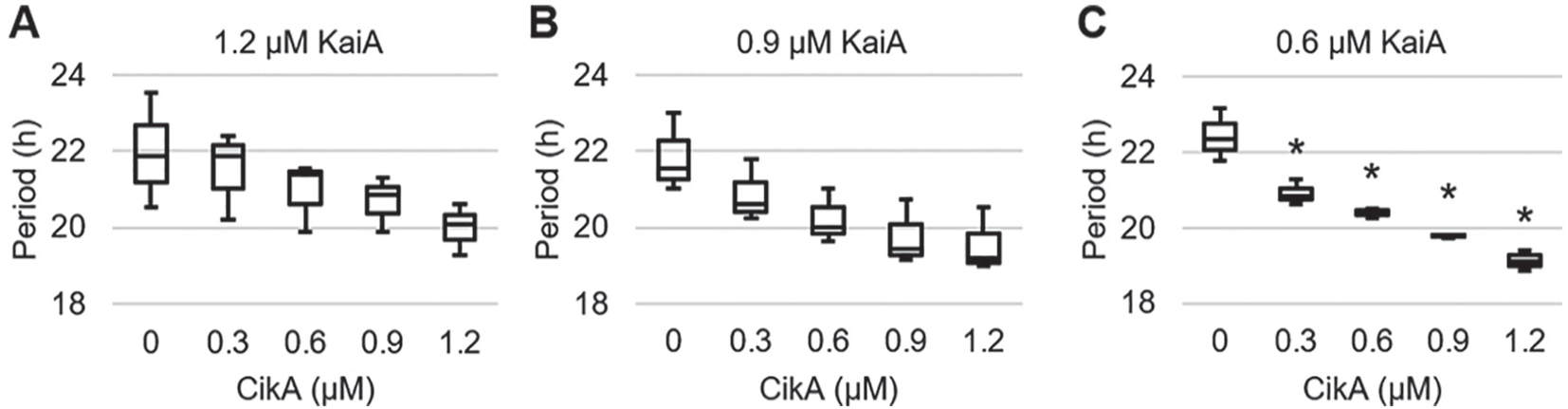
Statistical analysis of the circadian period with various CikA and KaiA concentrations. The concentration of KaiA is labeled on top of the graph, and 3.4 µM KaiB and 3.4 µM KaiC were used to generate the oscillation. (A) The period data are taken from the published article (Kaur et al., 2019). (B) No statistically significant change was observed among the periods. (C) An asterisk indicates that the period is significantly different from that of 0 µM CikA. p < 0.01. n = 3.
The Temperature Compensation of the Period Was Conserved in the New Cyanobacterial Circadian Clock Mixture with an Input Component, CikA
To test the temperature compensation of this reaction mixture (0.6 µM CikA, 0.6 µM KaiA, 3.4 µM KaiB, and 3.4 µM KaiC), the oscillation periods were measured at 25 °C and 35 °C. Robust circadian oscillations were observed at all temperatures, and periods were conserved in the circadian range (Fig. 2A; Suppl. Fig. S2). The periods increased when the temperature decreased (Fig. 2B; Suppl. Table S2); however, the change in period was not statistically significant (Suppl. Table S3). This is the same trend that was previously reported without CikA in the reaction mixture (Nakajima et al., 2005). The temperature compensation was examined by calculating the Q10 value of the circadian clock reaction mixtures using the periods at 25 °C and 35 °C. The Q10 value was 0.95 for the new circadian clock mixture, meaning that adding CikA and decreasing KaiA did not change the property of the temperature compensation. Therefore, our reaction mixture with CikA fulfills another requirement of a circadian clock.
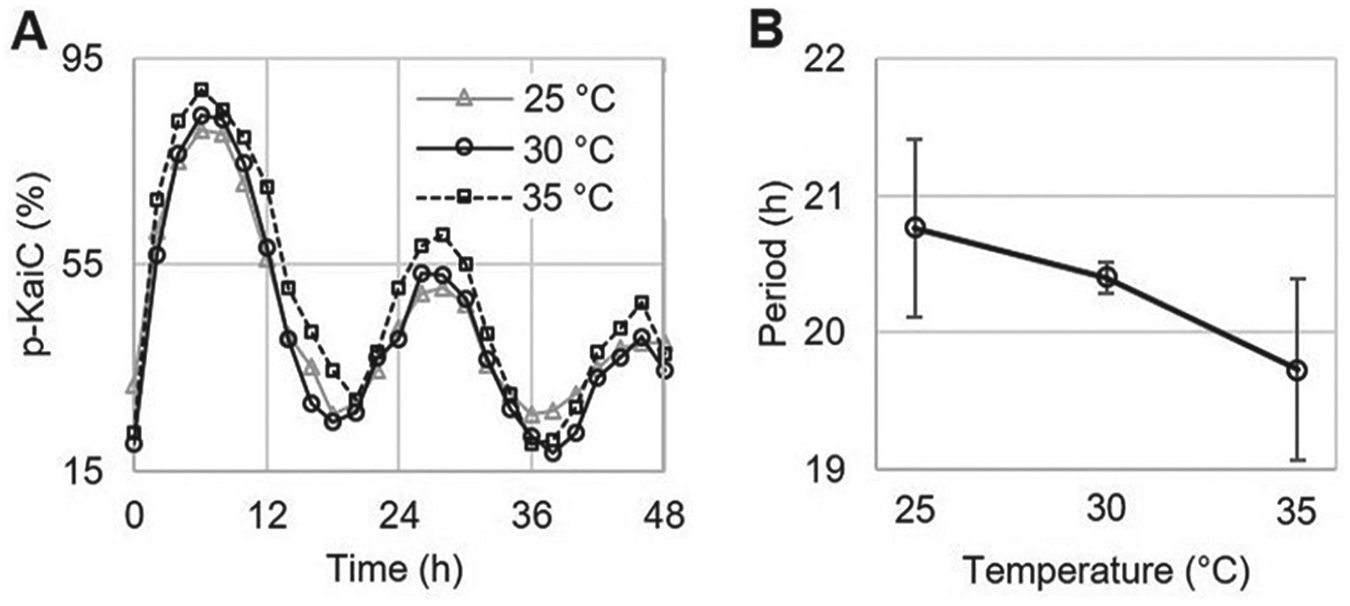
Temperature compensation of the circadian clock in vitro. (A) The circadian oscillations of KaiC phosphorylation at the different temperatures. (B) The circadian periods of the circadian clock in vitro at different temperatures. Error bars are standard deviation (SD). n = 3.
The New Circadian Clock Mixture Showed Both Phase Advance and Phase Delay on the PRC in Response to the Oxidized Quinone Signal
The last requirement is that a circadian clock must be entrainable. To test this with quinone, we selected the 0.6-µM CikA and 0.6-µM KaiA concentrations that conserve the temperature compensation of the circadian period. The phosphorylation of KaiC can be suppressed by adding 9.6 µM quinone (Suppl. Fig. S3). Quinone entrainment experiments were performed at CT 8 (Fig. 3A; Suppl. Fig. S3) and CT 22 (Fig. 3B; Suppl. Fig. S3) to observe the phase advance (at CT 8) and delay (at CT 22) by adding oxidized quinone in the circadian clock mixture for 4 h. When oxidized quinone was applied at CT 8, a 4-h phase advance was observed (Fig. 3C). This phase advance was smaller than that in the previous report, which used the circadian oscillator without an input component (Kim et al., 2012). The comparatively less KaiA present in our new reaction mixture may generate less of a phase advance in response to quinone because it is known that KaiA deactivation through quinone binding leads to a phase advance (Kim et al., 2012). On the other hand, applying oxidized quinone at CT 22 generated a 6-h phase delay (Fig. 3C), which was not observed in the experiments without an input component (Kim et al., 2012). This 6-h phase delay was 2 h longer than the PRC generated in vivo (Kiyohara et al., 2005). We thought that this difference might to be due to excess CikA present in our reaction mixture. In accordance with our observations, we hypothesized that KaiA and CikA are involved in phase advances and delays, respectively. If we increase KaiA and decrease CikA in our circadian clock mixture, we may observe a PRC that more closely matches the in vivo PRC.
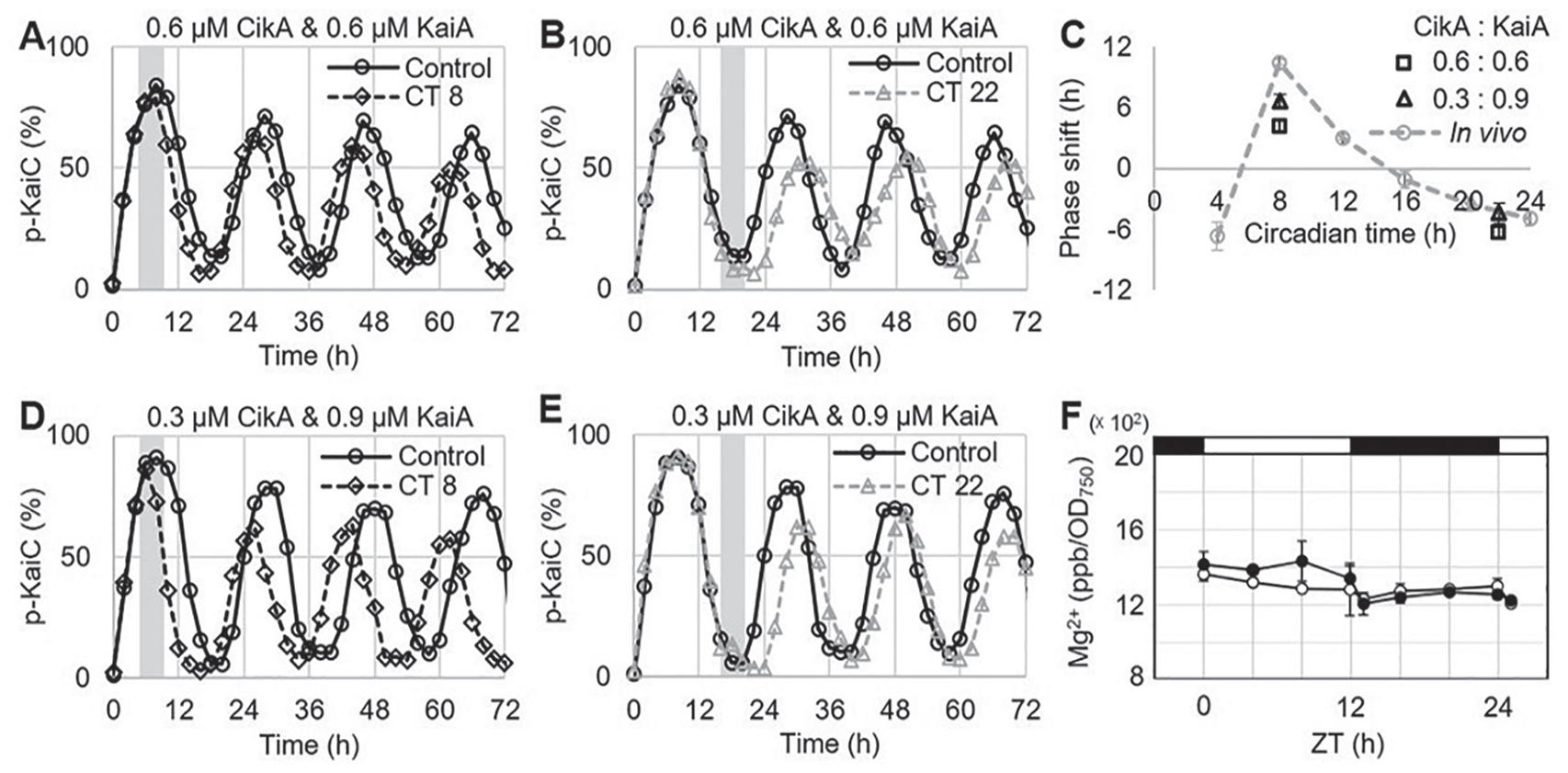
Entrainment of the circadian clock in vitro and total magnesium concentration in vivo. (A) The quinone entrainment of the KaiC phosphorylation at CT 8. The gray bar represents the duration of adding oxidized quinone in the circadian clock mixture in vitro. (B) The quinone entrainment of the KaiC phosphorylation at CT 22. (C) The phase-response curve of the cyanobacterial circadian clock in vitro and in vivo. Error bars are standard errors of the mean (SEM). n = 3. (D) The quinone entrainment of the KaiC phosphorylation at CT 8. (E) The quinone entrainment of the KaiC phosphorylation at CT 22. (F) Total magnesium concentration in cyanobacteria: wild type (○) and ΔkaiABC (•). Error bars show the standard deviation (SD). n = 3.
KaiA and CikA Are Involved in Phase Advance and Delay, Respectively
To test our hypothesis, we performed the quinone entrainment experiments with a different ratio of KaiA and CikA. KaiA concentration was increased to 0.9 µM, while CikA concentration was decreased to 0.3 µM because we assume that the amount of the phase shift is positively correlated with the amount of each protein. The summed amount of KaiA and CikA was maintained at 1.2 µM to conserve the circadian period (Kaur et al., 2019). In this mixture, a 7-h phase advance and a 4-h phase delay were observed at CT 8 and at CT 22, respectively (Fig. 3C-E; Suppl. Fig. S4). These results are close to the PRC from the in vivo study and indicate that the amount of the phase advance and phase delay was positively correlated with the concentrations of KaiA and CikA, respectively. We also performed the control entrainment experiments with the same reaction mixture above but without CikA. The same as the previous report, a 7-h phase advance and no phase delay were observed at CT 8 and at CT 22, respectively (Suppl. Fig. S5).
Oxidized Quinone Binding Causes a Conformational Change in the Pseudo-receiver Domain of CikA, the Binding Interface of the KaiB Binding
To find the effect of quinone on the binding interaction between CikA and KaiB, we analyzed 2 CikA structures previously published (Gao et al., 2007; Tseng et al., 2017). The pseudo-receiver domain of CikA uses α1 and β2 to bind to KaiB (Tseng et al., 2017). A single mutation on α1 of CikA shows the clock phenotype in the same manner as ΔcikA, and this indicates that the mutation attenuates the binding affinity between CikA and KaiB (Tseng et al., 2017). Interestingly, when oxidized quinone binds to the pseudo-receiver domain of CikA, conformational changes on α1 and β2 were detected through nuclear magnetic resonance spectroscopy (Ivleva et al., 2006). We mapped the disrupted residues on the pseudo-receiver domain of CikA and found that most of the residues are located on α1 and β2 (Suppl. Fig. S6). Therefore, adding oxidized quinone disrupts the binding interface of CikA and dissociates CikA from KaiB.
Magnesium Ion Is Not Used as a Signal for Entrainment in Cyanobacteria
Previously, we suggested that altering the magnesium concentration may be a signal for entrainment of the circadian clock (Jeong et al., 2019). To determine whether the magnesium concentration changes in response to light and dark in cyanobacteria, we measured the total magnesium concentration through ICP-MS. Unlike in other species (Feeney et al., 2016; van Ooijen and O’Neill, 2016), this analysis revealed that the total magnesium concentration remained constant in cyanobacteria under a light/dark cycle (Fig. 3F). This difference may derive from the comparatively high cellular content of magnesium and fast homeostatic response in cyanobacteria. Therefore, we did not perform in vitro entrainment experiments, because it does not appear that magnesium is used as a signaling molecule for entrainment in cyanobacteria
Conclusion
The mechanism of phase advances is well established in the previously reported entrainment experiment with the circadian oscillator composed of KaiA, KaiB, and KaiC (Kim et al., 2012). The phase advance in our new circadian clock reaction mixture also follows the same mechanism, in which KaiA loses its functionality by binding oxidized quinone, leading to the dephosphorylation of KaiC. In the phosphorylation phase, adding oxidized quinone causes the premature dephosphorylation of KaiC, resulting in an earlier transition to the next part of the cycle (i.e., a phase advance; Fig. 4A).
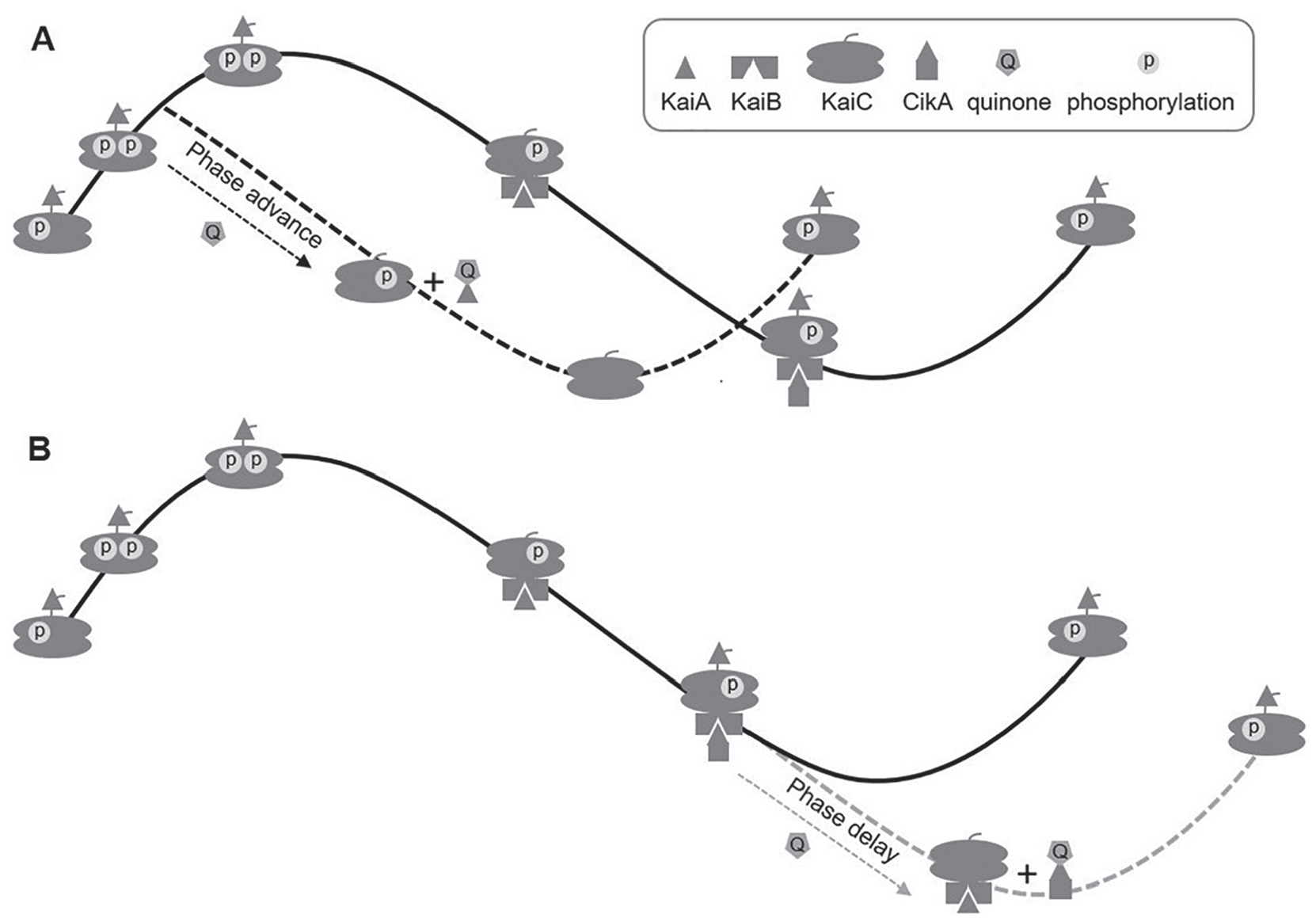
The entraining mechanism of the cyanobacterial circadian clock with oxidized quinone. The circadian oscillation without or with entrainment is shown on the solid or dotted line, respectively. The phosphorylation on the right and the left in KaiC represents amino acid residues S431 and T432, respectively. (A) The mechanism of phase advance. When oxidized quinone is applied on the phosphorylation phase, KaiA dissociates from the A-loop by binding oxidized quinone. The dissociation of KaiA brings early dephosphorylation of KaiC, resulting in phase advance (black dotted line). (B) The mechanism of phase delay. When oxidized quinone is applied in the dephosphorylation phase, CikA dissociates from KaiB by binding oxidized quinone, and KaiA binds to KaiB. This binding extends dephosphorylation of KaiC for a while and results in the phase delay (gray dotted line).
On the other hand, oxidized quinone binds to the pseudo-receiver domain of CikA and disrupts the binding between CikA and KaiB, which happens during the dephosphorylation phase (Tseng et al., 2017). When oxidized quinone is present during the dephosphorylation phase, CikA dissociates from KaiB, and KaiA takes over its spot. KaiC stays in the dephosphorylation phase until it is fully dephosphorylated. Keeping the dephosphorylation phase longer results in a later transition to the next part of the cycle (i.e., a phase delay; Fig. 4B).
Discussion
Our new data agree with the in vivo PRC, based on the structural and binding information of the clock components and quinone. To understand the cyanobacteria’s ability to align its circadian clock to environmental cycles, entrainment studies have been performed in vitro using only 2 metabolites so far: ADP and quinone. While the ADP signal works with the oscillator itself through bypassing the input pathway, the quinone signal seemed flawed without the input pathway (Kim et al., 2012; Rust et al., 2011). Our in vitro results showed that the input pathway is necessary for producing the expected phase delay in the entrainment using quinone signaling.
In the absence of CikA, applying oxidized quinone cannot generate the phase delay (Suppl. Fig. S5). The inactivation of KaiA at a low level of KaiC phosphorylation may have a minimal effect on the overall KaiC phosphorylation because its phosphorylation level can be recovered after reducing quinone (Suppl. Fig. S7). However, in the presence of CikA, oxidized quinone dissociates CikA from KaiB, and the premature phosphorylation is inhibited (Kaur et al., 2019). Also, the inactivation of KaiA keeps KaiC continuously dephosphorylated. After reducing quinone, the system may follow the oscillation phase trajectory of the one in the absence of CikA generating the phase delay (Suppl. Fig. S7). This idea can be tested by developing an enhanced mathematical model of the system.
In addition, we found that the circadian period becomes shorter (~1 h) after the sequential treatment with oxidized quinone and dithionite, especially when it is applied in the rising phase of the oscillation (Suppl. Fig. S5). However, this period shortening is not the result of incomplete reactivation of KaiA. If KaiA were incompletely reactivated due to the dregs of oxidized quinone, the effective KaiA concentration in the reaction mixture should decrease, and the period should instead become longer (Kageyama et al., 2006; Kaur et al., 2019; Nakajima et al., 2010). Therefore, we suggest that the observed period shortening is a recovery process of unknown mechanism. This phenomenon would be worth looking into in the future to more fully understand quinone’s role in entrainment.
Although we still do not understand why ΔcikA shows the short-period phenotype in vivo (Zhang et al., 2006), CikA is also known to possess an output functionality (Gutu and O’Shea, 2013) that perhaps contributes to the period shortening. This question may be answered if we can reconstitute the entire circadian clock in vitro with both the input and output components. Our findings here will serve as a basis for the reconstitution of the entire circadian clock in vitro.
Supplemental Material
SI_20191113 – Supplemental material for CikA, an Input Pathway Component, Senses the Oxidized Quinone Signal to Generate Phase Delays in the Cyanobacterial Circadian Clock
Supplemental material, SI_20191113 for CikA, an Input Pathway Component, Senses the Oxidized Quinone Signal to Generate Phase Delays in the Cyanobacterial Circadian Clock by Pyonghwa Kim, Brianna Porr, Tetsuya Mori, Yong-Sung Kim, Carl H. Johnson, Casey O. Diekman and Yong-Ick Kim in Journal of Biological Rhythms
Footnotes
Acknowledgements
We thank Hana Nim for insightful discussions, Dr. Yaofang Zhang of the Vanderbilt Mass Spectrometry Research Center for her help in [Mg2+] quantification, and Helene Brochon, Alejandra Lopez-Diaz, Adriana Kaszuba, and Neha Thati for technical assistance. This work was supported by a New Jersey Institute of Technology start-up grant and a faculty seed grant awarded to Y.-I. Kim, the National Science Foundation grant DMS-1555237 and the U.S. Army Research Office grant W911NF-16-1-0584 awarded to C. Diekman, and the National Institutes of Health/National Institute of General Medical Sciences grant R37 GM067152 awarded to C. H. Johnson.
Conflict Of Interest Statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
