Abstract
Heritable variation in the timing of rhythmic events with respect to daily time cues gives rise to chronotypes. Despite its importance, the mechanisms (clock or non-clock) regulating chronotypes remain elusive. Using artificial laboratory selection for divergent phasing of emergence of adults from pupae, our group has derived populations of Drosophila melanogaster which are early and late chronotypes for eclosion rhythm. Several circadian rhythm characteristics of these populations have since been described. We hypothesized that our selection protocol has inadvertently resulted in selection for masking, a non-clock phenomenon, in the early chronotype due to the placement of our selection window (which includes the lights-ON transition). We designed experiments to discriminate between enhanced masking to light versus circadian clock mediated changes in determining enhanced emergence in the morning window in our early chronotypes. Using a series of phase-shift protocols, LD-DD transition, and T-cycle experiments, we find that our early chronotypes have evolved positive masking, and their apparent entrained phases are largely contributed by masking. Through skeleton T-cycle experiments, we find that in addition to the evolution of greater masking, our early chronotypes have also evolved advanced phase of entrainment. Furthermore, our study systematically outlines experimental approaches to examine relative contributions of clock versus non-clock control of an entrained behavior. Although it has previously been suggested that masking may confer an adaptive advantage to organisms, here we provide experimental evidence for the evolution of masking as a means of phasing that can complement clock control of an entrained behavior.
Circadian clocks adaptively schedule behavior and physiology to occur at a specific time of the day. Such scheduling is believed to be critical for maintaining our general health and well-being (Roenneberg et al., 2012; Vaze et al., 2014; Horn et al., 2019). Heritable variations in the timing (or phasing) of rhythmic events with respect to daily time cues result in what is referred to as chronotype variation (Infante-Rivard et al., 1989; Roenneberg et al., 2007). A clear example is that of variation in mid-sleep timing among humans on free days. While most humans will fall in the category of “normal” or “neither” chronotype, some individuals tend to fall asleep relatively early in the evening and wake up early in the morning, hence referred to as “early” chronotypes or “larks” while there are those among us who prefer very late sleep timings and associated late wake timings, also referred to as “late” chronotypes or “owls” (Randler et al., 2017). Chronotypes are primarily controlled by circadian clocks. Studies, including those on humans, have suggested that variation in entrained phases arises due to differences in underlying clock properties such as length of the intrinsic period of circadian clocks, phase/velocity response curves (PRCs/ VRCs), amplitude of the circadian clock, inter-oscillator coupling, and amplitude of the zeitgeber (Aschoff and Pohl, 1978; Bordyugov et al., 2015; Daan and Pittendrigh, 1976; Granada et al., 2013; Johnson et al., 2003; Roenneberg et al., 2012; Swade, 1969).
While light can bring about a change in phase of an entrained rhythm by influencing the phase of circadian clock (Saunders et al., 1994; Schlichting and Helfrich-Förster, 2015), one cannot rule out aspects of direct effects of light. Such exogenous environmental influences on endogenously generated circadian rhythms which obscure aspects of circadian clock expression is referred to as masking (Aschoff, 1960; Fry, 1947; Mrosovsky, 1999). Traditionally, non-involvement of the circadian clock has been considered an essential criterion defining masking, however, Terry Page (1989) and Nicholas Mrosovsky (1999) make a strong case for the importance of masking as a complement to circadian clock regulation of daily rhythms. While there are studies which suggest that masking responses are present in animals without a functional clock (clock mutants or surgical ablation of the central clock) (Redlin and Mrosovsky, 1999; Wheeler et al., 1993), Aschoff had argued that there could be time-of-day dependence in certain masking responses (eliciting activity in blind male hamsters in the presence of mates) (Aschoff and Honma, 1999; Aschoff and von Goetz, 1988b). Positive masking refers to the masking response which elicits the start of a behavior, while negative masking refers to the inhibition or ceasing of the behavior. Traditionally studied in mammals with respect to locomotor activity rhythms, masking has also received some attention in insects, in both locomotor activity and eclosion rhythms (Hamblen-Coyle et al., 1992; Kempinger et al., 2009; Lu et al., 2008; Rieger et al., 2003; Sheppard et al., 2015; Thakurdas et al., 2009; Wheeler et al., 1993).
The act of emergence of a pharate adult fly from its pupal case or eclosion is developmentally gated, and a population level rhythm. It was also among the earliest circadian rhythms to be studied systematically (Chandrashekaran, 1967; Chandrashekaran and Loher, 1969; Engelmann, 1969; Harker, 1965a, 1965b; Pavlidis, 1967; Pittendrigh, 1954, 1967; Pittendrigh et al., 1958; Skopik and Pittendrigh, 1967; Zimmerman et al., 1968). The anatomical and physiological processes underlying eclosion have also been extensively investigated (Johnson and Milner, 1987; Krüger et al., 2015; Peabody and White, 2013; Selcho et al., 2017; Thummel, 2001). It is deemed to be among one of the most critical events in the lifetime of a holometabolous insect (Mcmahon and Hayward, 2016; Zitnan and Adams, 2005). The Drosophila eclosion rhythm has been shown to be regulated by the circadian pacemakers previously implicated in activity rhythms in addition to prothoracic gland clocks (Morioka et al., 2012; Myers et al., 2003; Pittendrigh and Bruce, 1959; Selcho et al., 2017; Zimmerman et al., 1968). In addition, in contrast to several behavioral rhythms, the act of eclosion is expected to be unaffected by motivational state, other behaviors, or interactions among individuals, but susceptible to disruption when mutations are introduced in core clock genes like period (per) or timeless (tim) (Qiu and Hardin, 1996; Ruf et al., 2019; Sehgal et al., 1994) both under constant and cycling conditions. Thus, relative to other rhythms, it appears to be a more reliable indicator of perturbations in the core clock. Eclosion rhythms, their entrainment to light/ dark cycles, temperature cycles, synergistic light and temperature cycles as well as molecular mechanisms of their entrainment have been studied in great details in Drosophilid species (Emery et al., 1997; Kumar et al., 2006, 2007; Morioka et al., 2012; Myers et al., 2003; Nikhil et al., 2014, 2015; Nikhil, Abhilash, and Sharma, 2016; Pittendrigh, 1966; Pittendrigh and Bruce, 1959; Pittendrigh and Minis, 1972; Prabhakaran et al., 2013; Qiu and Hardin, 1996; Vaze and Sharma, 2013). The rhythm in eclosion is modulated by the lights-ON signal (Chandrashekaran and Loher, 1969; Engelmann, 1969; Pittendrigh, 1967; Thakurdas et al., 2009), even though wing-expansion, the last behavioral event of the Drosophila adult eclosion sequence (Fraekkel, 1935), is not affected by the same (McNabb and Truman, 2008), suggesting that the lights-ON signal may have a role specific to the act of eclosion itself. It was suggested previously that the lights-ON signal may have two distinct effects on the timing of eclosion of flies—(a) stimulation of eclosion hormone release and (b) reduction in the latency of eclosion relative to eclosion hormone release (Baker et al., 1999; McNabb and Truman, 2008).
With reference to Drosophila melanogaster, Hamblen-Coyle and colleagues first reported a lights-ON peak in locomotor activity rhythm which they designated as a “startle” effect. It was seen that even though flies carrying core circadian clock mutations adopt distinct phases in evening peak timings, their morning peak (lights-ON peak) phases were not very different (Hamblen-Coyle et al., 1992). Later it was shown that under LD12:12, even without a functional clock, animals exhibited this lights-ON peak, but it was absent when shifted to constant darkness (DD) (Wheeler et al., 1993). Rieger and colleagues showed that under laboratory LD12:12 conditions, the lights-ON peak and the morning peak are indistinguishable as they overlap with each other (Rieger et al., 2003). These two peaks can be separated from each other under different photoperiods, where the lights-ON peak is still phase-locked to the dark-to-light transition, but the morning peak is advanced or delayed under short or long photoperiod respectively (Rieger et al., 2003). Artificial moonlight can make fruit flies nocturnally active, and this nocturnal light is known to induce strong locomotor activity in flies via masking (Kempinger et al., 2009). Recent detailed genetic dissections of masking in Drosophila have revealed complex pathways mediating light-induced masking of locomotor activity (Rieger et al., 2003). Furthermore, Lu and colleagues demonstrated a circadian rhythm in light-induced locomotor activity against a background of DD and showed that the circadian clock genes timeless and clock are involved in regulation of this masking response (Lu et al., 2008; Sheppard et al., 2015). Taken together, the above studies on effect of light on locomotor activity rhythms and eclosion suggest that masking can affect the timing of a circadian rhythm in Drosophila.
We reasoned that one set of our long-term laboratory-selected populations of D. melanogaster could potentially have evolved a masking response due to the nature of the selection regime that had been imposed on them for ~320 generations (early1-4—described below). In this study, we investigated the immediate effects of light on the timing of their eclosion rhythm. These populations are part of an on-going long-term experimental evolution study at the Chronobiology laboratory, JNCASR. The primary goals of creating these populations were—(a) to demonstrate the adaptive significance of phasing of circadian rhythm (here, eclosion rhythm) (b) to then study the associated clock properties mediating phase divergence. Indeed, our populations selected for morning emergence (early1-4) exhibit significantly shorter (22.51 h, ± 95% CI = 0.106) mean free-running period (FRP) compared to those selected for evening emergence (late1-4 mean FRP 23.86 h, ± 95% CI = 0.106) and the control1-4 populations (mean FRP 22.94 h, ± 95% CI = 0.106), which did not undergo any selection for timing of emergence (Kumar et al., 2007; Nikhil, Abhilash and Sharma, 2016). Over the years, several studies from our lab have shown that entrainment of these divergent chronotypes to light-dark (LD) cycles cannot be fully explained by exclusively invoking either the parametric or non-parametric models of entrainment (Abhilash and Sharma, 2020; Kumar et al., 2007; Vaze, Nikhil, et al., 2012). It was also hypothesized that differences in inter-oscillator (A/master/central oscillator and B/slave/peripheral oscillator) coupling might explain the chronotype divergence in eclosion rhythm (Abhilash et al., 2019; Nikhil, Abhilash, and Sharma, 2016).
We reasoned that our selection lines may provide material to examine some aspects of masking and circadian control of a rhythmic phenomenon because the design of our selection regime is such that flies of the early populations have over generations been forced to emerge in a window around lights-ON (3 h prior to lights-ON till 1 h post lights-ON). It is possible that this protocol has inadvertently resulted in selection for the phenomenon of masking. Therefore, among other clock-related factors that have been uncovered to have changed in these populations previously (Abhilash and Sharma, 2020; Nikhil et al., 2015; Nikhil, Vaze, et al., 2016; Vaze, Kannan, et al., 2012) we hypothesized that the phase divergence among early, control and late chronotypes are partly due to differences in masking—specifically, that early populations exhibit a high degree of masking. We designed several experiments to examine whether early flies exhibit enhanced positive masking in eclosion rhythm compared to control, and late flies as shown in Figure 1.
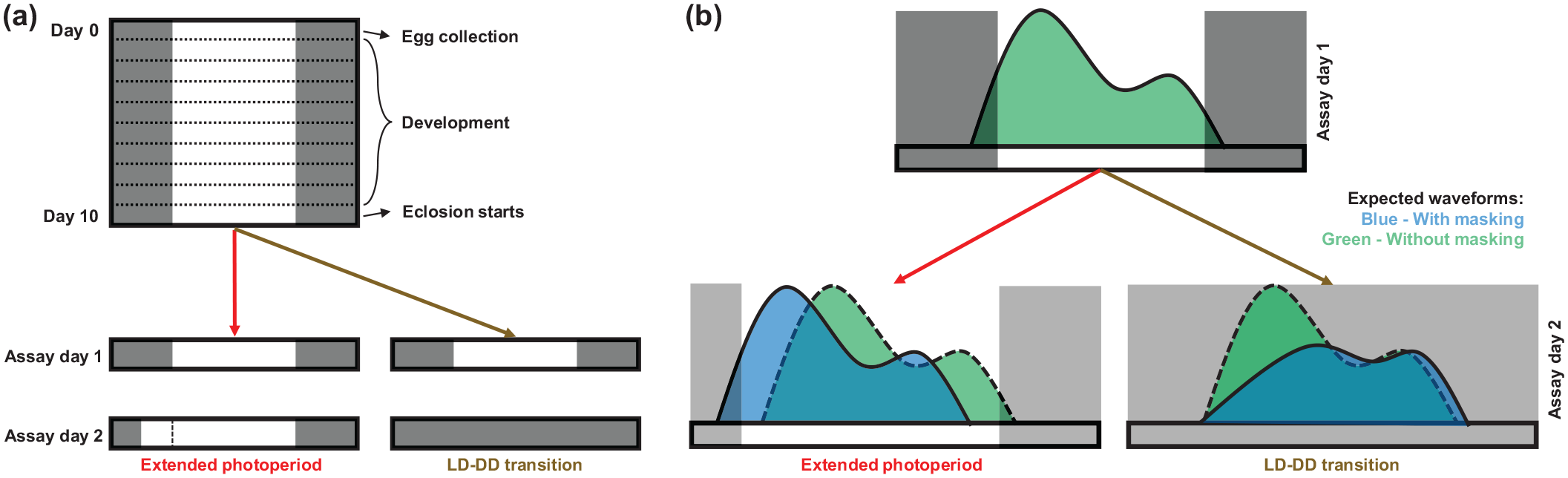
Schematics of experimental design and hypotheses. (a) After egg collection, vials were kept under LD12:12 till 9th day. Emergence assay was started on the 10th day (Assay day 1), and flies were counted every two/half an hour interval depending on experiments and time points. On the 11th day (Assay day 2), vials were placed under a 3 h phase-advanced light schedule (extended photoperiod) or complete darkness (LD-DD transition). (b) The schematic shows expected waveforms in case of complete circadian control (dashed line) or masking (solid line). Dark rectangular shades depict duration of darkness. Abbreviations: LD = light-dark; DD = constant darkness.
Here we demonstrate that—(a) early chronotype flies have indeed evolved significantly more positive masking compared to control and late flies, (b) under full photoperiod, apparent entrained phases of early flies are largely contributed by masking, and (c) under skeleton photoperiod, early flies do show phase lability, and retain advanced phase of entrainment compared to control and late flies to different T-cycles, suggesting that our selection indeed has selected for greater masking alongside selection for advanced phase of entrainment.
Materials and Methods
Selection Protocol and Fly Maintenance
Four sets of genetically independent Drosophila melanogaster populations were used to artificially select for morning eclosion (early populations) and evening eclosion (late populations) timing. Henceforth, denoted as early(1-4), control(1-4) (no selection imposed) and late(1-4). At each generation, ~300 eggs are collected and placed in glass vials which are maintained in a light-proof, temperature-controlled cubicle in a light-dark cycle of 12/12 h- (LD12:12), 25°C ± 0.5°C, and 65% ± 5% RH. Flies emerging from ZT21-ZT01 (ZT0 is
Behavioral Experiments
Before each experiment, ~300 eggs were collected and placed in 10 vials each for all 12 standardized populations. After egg collection, the vials were maintained in different regimes specific to each experiment. Emerged flies were counted every 2 h or 0.5 h after the assay started, depending on the experimental regime. Briefly, all rhythm assays in Figures 2 and 3 were carried out with half hour resolution for 12 h around lights-ON and in assays depicted in Figures 4 and 5 (full and skeleton T-cycle experiments), 2 h resolution was used due to logistic constraints. To account for differences in development time, if any, fly counts from the first emergence cycle were excluded from analysis. All 12 populations were assayed in parallel. In all experiments, temperature (25°C ± .5°C), and light intensity (~70 lux, from a white LED source) were kept constant. Other details of the light regime are mentioned in each experiment separately, and light ON-OFF times (step wise) were programmed with a TM619 timer (Frontier Timer, Pune, India) in the incubators (DR-36VL, Percival Scientific, Perry, USA) in which the experiments were performed.
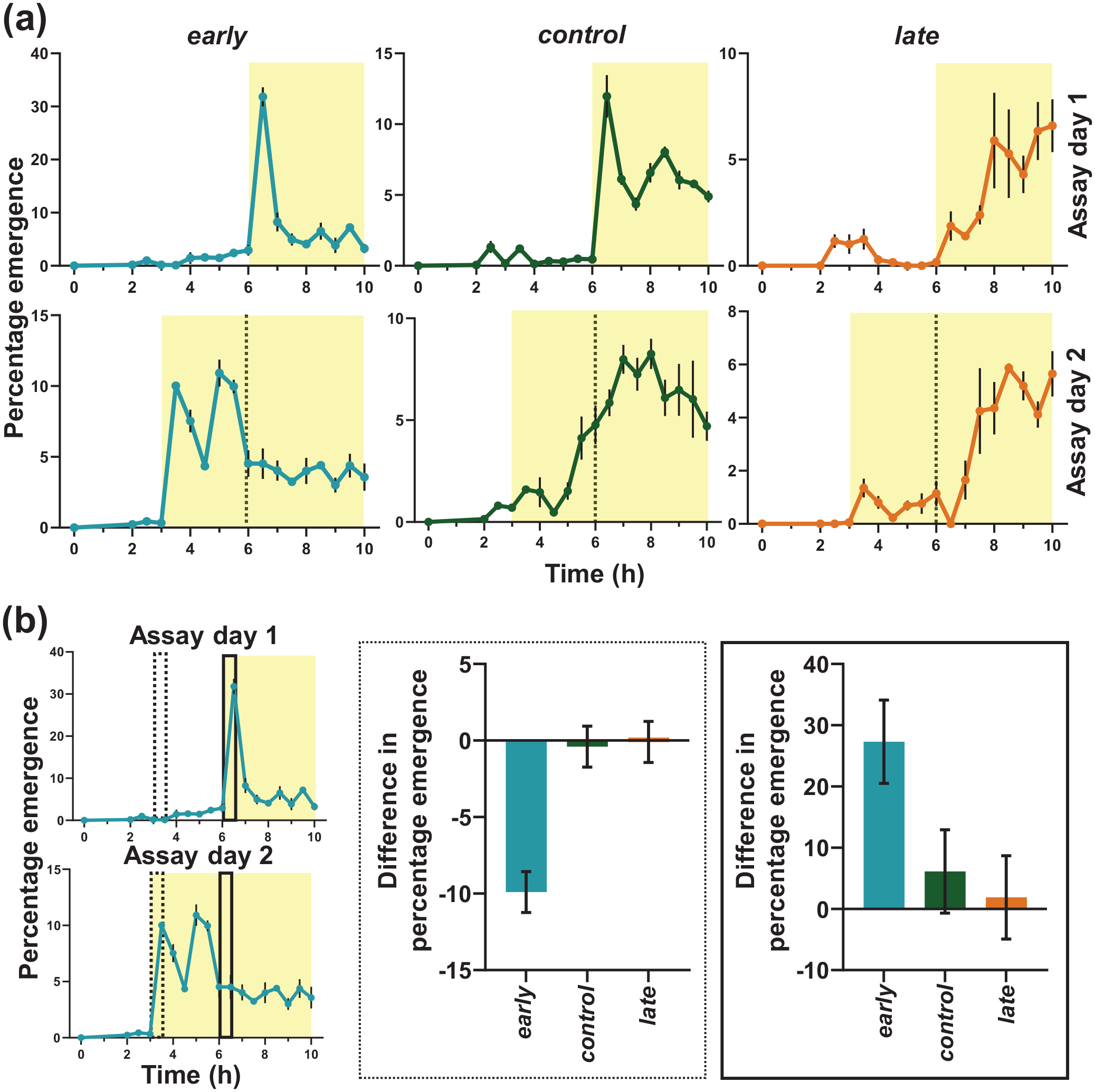
Profile during first 10 h on day 10 (post egg collection) in pre-shift (Assay day 1) and post-shift (Assay day 2) days and difference in percentage emergence between pre-shift (Assay day 1) and post-shift (Assay day 2) days in two 0.5 h time windows. (a) early flies advance their emergence waveform to emerge immediately after lights-ON on Assay day 2. The control and late flies conserve their waveform on both days by not advancing their emergence in response to sudden advancement of the lights-ON stimulus. The shading indicates the photophase in the LD cycle each day. Error bars are ± SEM. Gray dotted lines in the bottom panel indicates lights-ON time on previous day. (b) The left panels show time windows used for analysis (dashed and solid rectangles respectively). In the same time windows, early flies show significantly higher (dashed rectangle region) or lower (solid rectangle region) emergence than that of control and late flies on Assay day 2 than Assay day 1, showing this high emergence immediately after lights-on is specific to early flies. Abbreviations: LD = light-dark; SEM = standard error of the mean.
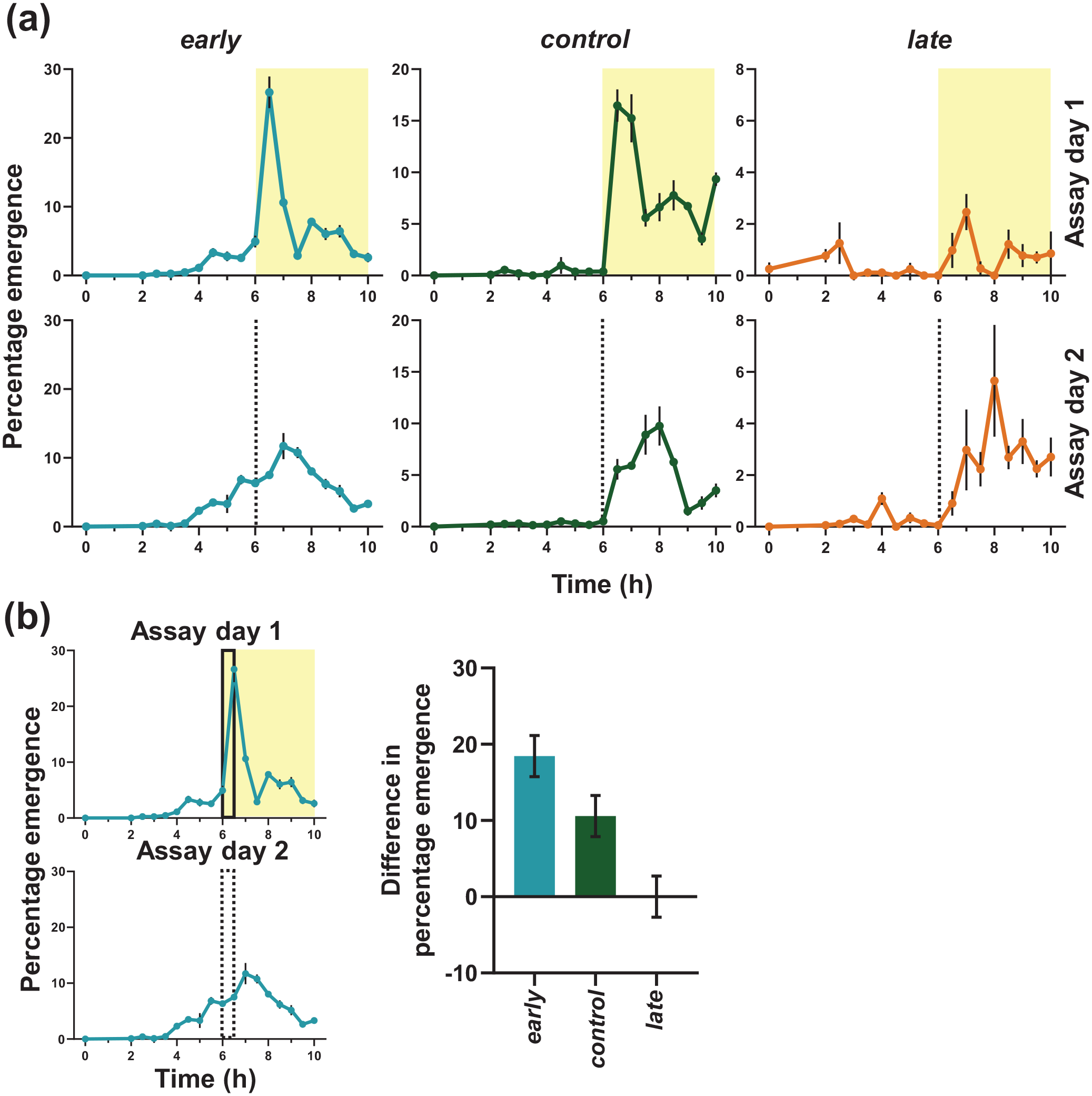
Profile during first 10 h on day 10 (post egg collection) in LD (Assay day 1) and DD (Assay day 2) days and quantification of difference of percentage emergence between pre-shift and post-shift days from ZT0-0.5 (lights-ON phase followed from LD cycle). (a) early flies attenuate their emergence to a great extent in absence of light stimulus in the earliest part of the subjective photophase, unlike their waveform in LD12:12. However, control and late flies, emerge with waveforms like under LD12:12, albeit with lower amplitude. Error bars are ± SEM. Gray dotted lines in the bottom panel indicates lights-ON time on previous day. (b) early flies show significantly higher difference than control and late flies. This suggests the high emergence of early flies immediately after lights-on was a masking response rather than a clock mediated response. Abbreviations: LD = light-dark; DD = constant darkness; SEM = standard error of the mean.
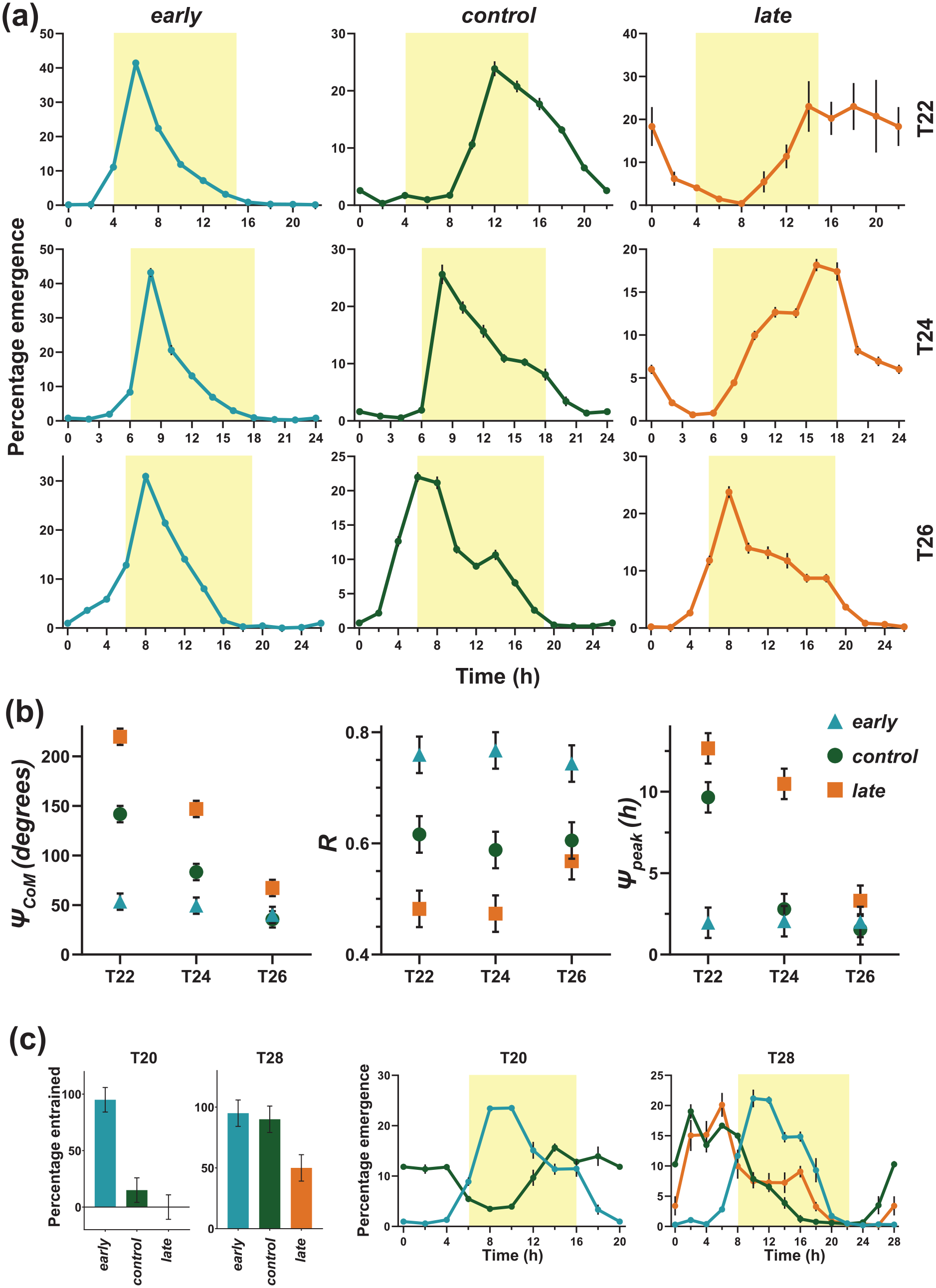
Profile of early, control, and late flies under T20, T22, T24, T26, and T28 cycles and eclosion rhythm parameters under T-cycles. (a) early flies do not change their emergence profile depending on T-cycles, whereas control and late flies shift their emergence profile in expected directions (phase advance in longer than 24-h cycle and phase delay in shorter than 24-h cycle). Shading depicts the photophases of respective T-cycles. Error bars are ± SEM. (b) Left panel: Center of Mass (ΨCoM—in degrees), middle panel: Consolidation of emergence (R), right panel: Peak in ZT (ΨPeak, in hours). Error bars are ± 95% CI. early flies do not change their ΨCoM, ΨPeak, and R under any T-cycle, showing they are phase-locked to the lights-ON stimulus. (c) Percentage entrainment and emergence profiles of early, control and late flies under T20 and T28. Error bars are ± 95% CI for percentage entrainment and ± SEM for profiles. SEM = standard error of the mean; ZT =
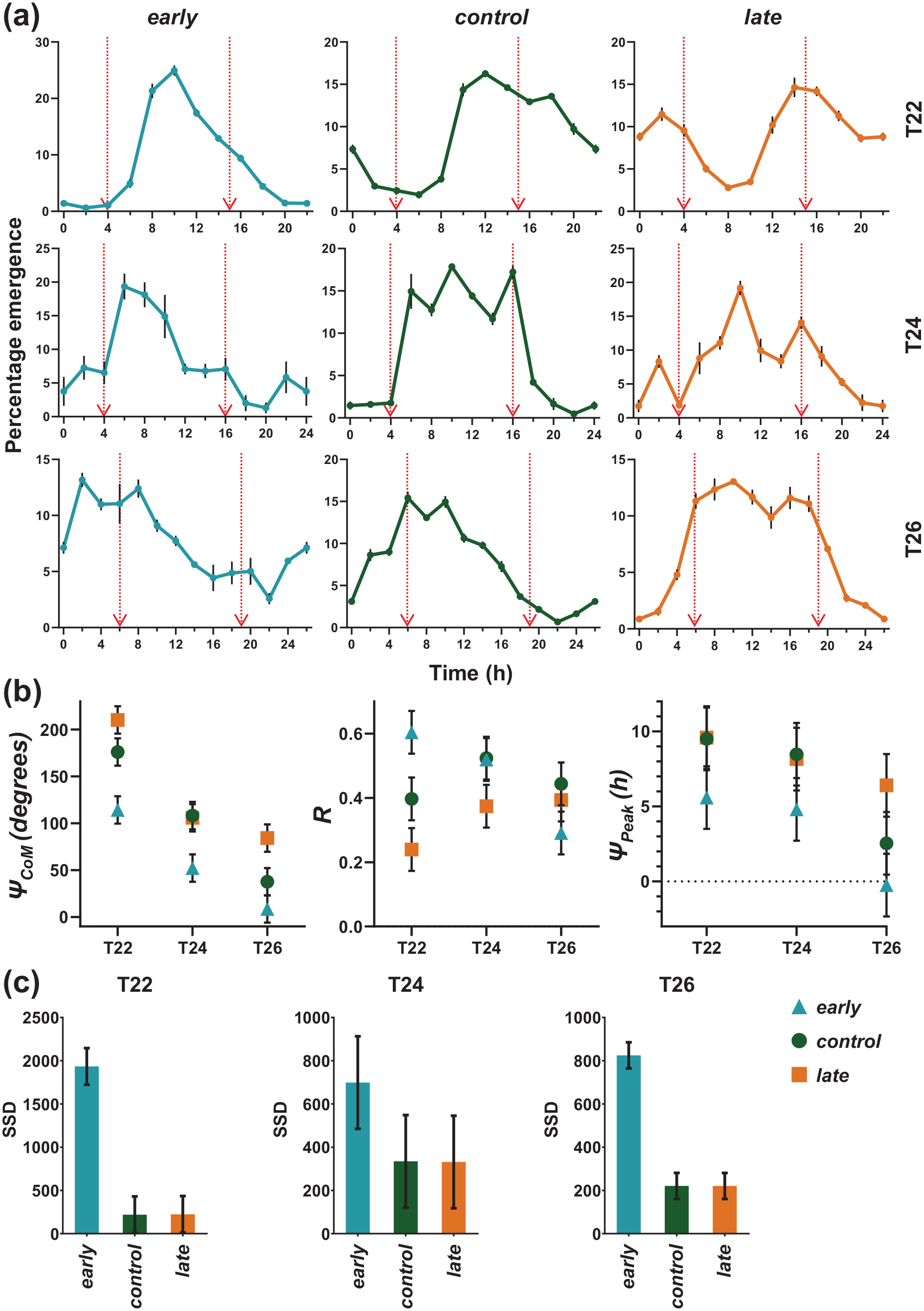
Profile of early, control, and late flies in T22, T24, and T26 skeleton photoperiods and quantification of different eclosion rhythm parameters under different skeleton T-cycles. (a) early flies change their emergence profile depending on skeleton T-cycles as do control and late flies. Arrows depict the 15 min of the day when flies get light in respective skeleton photoperiods. Error bars are ± SEM. (b) Left panel: Center of Mass (ΨCoM—in degrees), middle panel: Consolidation of emergence (R), right panel: Peak in ZT (ΨPeak, in hours). Error bars are ± 95% CI. (c) Sum of Square Difference (SSD) of waveforms under full and skeleton photoperiods. Significantly high SSD in early flies depict large changes in waveform and phases between full and skeleton photoperiods. Error bars are ± 95% CI. SEM = standard error of the mean; ZT =
Analysis of Data and Statistics
Average profiles were constructed first by averaging over multiple cycles for a vial, and then by averaging over vials for each population. All statistical comparisons were made using either a 2-way or 3-way randomized block design analysis of variance (ANOVA) with selection regime and T-cycle (as applicable) as fixed factors and blocks (replicates) as the random factor. Results were deemed significant for main effect or interaction as applicable at α < 0.05. Post hoc comparisons were carried out by a Tukey’s Honest Significant Difference (HSD) test. In the main text, we report significant main effects or interaction effects while supplementary tables provide the full details of the ANOVA. All mean values and the 95% CI used for post hoc comparisons are also reported in supplementary tables. All statistics were performed in Statistica 7 (StatSoft, Tulsa, USA). Standard errors of means have been plotted as error bars in average profiles for ease of visualization. 95% confidence intervals from the Tukey’s HSD are plotted in all other graphs and used for quantitative comparisons for visual hypothesis testing. Basic data processing and calculations were done with Microsoft Excel 365, and all graphs were plotted with Graphpad Prism 8. Criteria for “apparent entrainment” was Tobserved must equal to Tenvironment (Tobserved is the observed period of the eclosion rhythm and Tenvironment is the duration of the light/ dark cycle). Tobserved was calculated with JTK-cycle (Hughes et al., 2010) employed in MetaCycle2d (Wu et al., 2016) with percentage eclosion data for each vial. Vials showing a “JTK_pvalue” less than 0.05 were considered to be rhythmic, and among the rhythmic vials, the ones with “JTK_period” within a range of ± 1 h of Tenvironment were considered to be “apparently entrained.” All calculated phases are essentially phase relationships with the lights-ON signal. Peak phase is denoted by ΨPeak and measured in hours, and phase of Center of Mass is denoted by ΨCoM and measured in degrees. ΨPeak was calculated as the time where maximum number of flies emerged in each vial and ΨCoM was calculated as a measurement of mean phase of emergence in polar coordinates for each vial (corrected for different lengths of T-cycles). Consolidation of emergence/ normalized amplitude (R) was also calculated in a polar coordinate system for eclosion data averaged over cycles, details of computation and usefulness of which can be found in a recent publication from our lab (Abhilash et al., 2019).
Results
Lights-ON Elicits an Immediate Response in the early Chronotypes
Previously, all eclosion rhythm assays on our populations were carried out with a maximum resolution of 2 h and thus far no difference was detected in the peak phase (ΨPeak) of early and control flies, both of which were found to emerge maximally at (ZT2) (Kumar et al., 2007; Nikhil, Abhilash, and Sharma, 2016). To examine whether there are subtle changes in the onset or peak of emergence among stocks we increased the resolution to 0.5 h for the first half of the day (Assay day 1 and 2, Figures 2a and 3a). In fact, we now find that peak (ΨPeak) emergence for both early (~30%) and control (~15%) flies occur at ZT0.5, that is, immediately after lights-ON. Onset of emergence is similar to that obtained by 2-h resolution assays done previously (Kumar et al., 2007; Nikhil, Abhilash, and Sharma, 2016) across stocks.
When lights-ON was advanced by 3 h on the following day (Assay day 2, Figure 2a), early flies exhibited high emergence in the first 0.5 h, whereas very few flies emerged in the early part of the light phase in both the control and late populations—they mostly emerged at similar time as the previous cycle (Assay day 1, Figure 2a). Peak emergence of early flies occurred at ZT2 on Assay day 2. On both assay days, majority of early flies emerged immediately after lights-ON (Figure 2a). The phase of peak emergence for control flies remained similar to Assay day 1, even after the light phase was advanced on Assay day 2 (Figure 2a). Emergence waveform of late flies did not change from Assay day 1 to Assay day 2 (Figure 2a).
Based on the emergence profile of the early populations we quantified the difference in emergence during two specific time windows on the two experimental days. The first window (solid rectangle, Figure 2b) depicts the time window of maximum emergence for early flies on Assay day 1 (LD) and the second window (dashed rectangle, Figure 2b) depicts the time window for maximum emergence (immediately after lights-ON) for early flies on Assay day 2 (extended photoperiod). If early flies show more masking than circadian control of their emergence, one expects a large difference in levels of eclosion between the 2 days because they are strongly modifying their waveforms in response to lights-ON. We observe that the difference in percentage emergence during the first window (between Assay days 1 and 2) is significantly higher for early flies than for control and late flies, the latter two showing almost no change across days (solid rectangle, Figure 2b; Suppl. Table S1). Similarly, in the second window, early flies show significantly higher change across Assay days 1 and 2 compared to control and late populations (dashed rectangle, Figure 2b; Suppl. Table S2). This suggests that on Assay day 2, the circadian response of early flies is overridden by the immediate lights-ON response, thus exhibiting higher positive masking response compared to control and late flies. When considering two larger windows each of 2.5 h (around lights-ON of pre-shift day) similar differences prevail (Suppl. Fig. S14), showing that high emergence immediately after lights-ON is specific to early flies.
early Flies Attenuate and Delay Their Emergence Under DD
Since the above experiments suggested that masking plays a prominent role in the timing of eclosion of early flies we then attempted to parse the relative contribution of circadian clock control on the emergence profile of early flies. Therefore, one set of cultures were shifted to constant darkness (DD—Assay day 2, Figure 1a right panels; LD-DD transition). On Assay day 2, very few flies of the early populations emerged in the 2 h duration corresponding to immediately after lights-ON of Assay day 1, resulting in a large difference in emergence between two consecutive cycles (Figure 3a). This difference in emergence between cycles was lesser in control and almost non-existent in late flies (Figure 3b right panel). If the circadian clock has a strong control over the eclosion rhythm, the early flies should show high emergence in the early part of the subjective photophase in DD, which was not observed. The sharp morning peak, an identifiable marker for our early flies was absent in DD (bottom left panel, Figure 3a).
We quantified the difference in percentage emergence from ZT0-0.5 of first day (LD, Assay day 1) and the same phase in second day (DD, Assay day 2), and find that early flies show significantly larger difference than control and late flies, suggesting attenuated emergence immediately after starting of subjective photophase in DD (Figure 3b; Suppl. Table S3). While a reduction in amplitude is expected under DD, it is significantly greater for early flies than control flies. This suggests the high emergence of early flies immediately after lights-ON is largely a masking response and less of a clock-controlled phenomenon.
early Flies Consistently Emerge Close to Lights-ON Under Both Short and Long T-cycles
T-cycles shorter than the FRP of the organism are expected to delay phases of circadian rhythms (phase relationships with Zeitgeber), while T-cycles longer than the FRP of the organism advance phases of the circadian rhythms (Aschoff, 1965; Wheeler et al., 1993; Yadav et al., 2015). Therefore, we subjected our populations to a series of short (T20; LD10:10 and T22; LD11:11) and long (T28; LD14:14 and T26; LD13:13) T-cycles (Figure 4a-4c). Under the two extreme T-cycles T20 and T28, although early flies showed higher percentages of “apparent” entrainment, very small fraction of control and late flies entrained, thus rendering comparison among stocks inappropriate. Under T20, significantly few vials of control and late flies show entrained rhythms compared to early flies, and under T28, significantly low number of vials of late flies entrained compared to control and early flies (left panel Figure 4c; Suppl. Tables S19 and S20); hence, the results of these extreme T-cycles T20 and T28 were not considered for further analyses and quantification of phases. Under T20 and T28 cycles, early flies showed high emergence immediately after lights-ON compared to control and late flies (middle and right panel Figure 4c) suggesting that, early flies mask successfully and show an apparent entrained behavior. We expected that eclosion rhythm of control and late flies, owing to strong control by circadian clocks will shift their phases in the predicted directions. We hypothesized that eclosion rhythm of early flies, if strongly governed by masking and less by the circadian clock, or controlled by parametric changes in the circadian clock, will not modify phases of the eclosion rhythm in the predicted directions—delay under T<24 h and advance under T>24 h.
Under T22, early flies maintain similar waveform as under T24 (left panel top row, Figure 4a). The control flies showed clear phase delay (middle panel top row, Figure 4a), whereas late flies showed even greater phase delay in response to a T22 cycle (right panel top row, Figure 4a) compared to their waveforms in T24 cycles.
Under T26, early flies showed similar emergence waveform as observed under T24 (left panel third row, Figure 4a). The control flies showed phase advance (middle panel third row, Figure 4a) and late flies showed even larger phase advance in response to T26 cycle (right panel third row, Figure 4a) compared to their waveforms under T24. There is some degree of anticipation in early flies to lights-ON under all three T-cycles as there is a gradual rise in emergence prior to lights-ON (Figure 4a). This issue is addressed in Supplementary Figure S15. Briefly, the underlying clock of early flies delay or advance under T22 and T26 respectively, still the high emergence immediately after lights-ON is conserved under all three T-cycles due to masking.
We quantified three parameters of the emergence waveform under all three T-cycles: (a) Center of Mass (ΨCoM), which is an estimate of mean phase angle of emergence in a circular scale, (b) peak in ZT (ΨPeak), and (c) R, which is a measure of normalized amplitude of the eclosion rhythm and comprehensively describes consolidation of emergence. All these parameters have been previously used to describe the eclosion rhythm waveform of Drosophila melanogaster (Abhilash et al., 2019).
As seen in Figure 4b, ΨCoM for early flies did not change across the T-cycles, whereas ΨCoM for control and late flies shifted in expected directions for T22 (delayed) and T26 (advanced) compared to T24 cycle (left panel, Figure 4b; Suppl. Table S4). ΨPeak for early flies remained similar across all three T-cycles (right panel, Figure 4b; Suppl. Table S5). Control and late flies showed expected trends in change of direction of peak phase shift in T22 and T26 cycles (right panel, Figure 4b; Suppl. Table S5). In case of control flies, although there was a trend of advancing peak phase in T26 cycle, compared to T24, this difference was not significant, whereas the delay in peak phase in T22 was much larger when compared to T24 cycle (right panel, Figure 4b; Suppl. Table S5). In late flies, the peak phase in T26 is significantly advanced while in T22 it is significantly delayed compared to T24 (right panel, Figure 4b; Suppl. Table S5). Next, we quantified the consolidation of emergence/normalized amplitude of the eclosion rhythm (R) of all populations under all T-cycles. A large R value is characteristic of the early population as evident by their narrow gate-width of emergence (Nikhil, Abhilash, and Sharma, 2016). If the shorter and longer T-cycles had indeed shifted the circadian clock of early flies and the masking component is only responsible for the high emergence in the earliest part of the photophase, then value of R is expected to change among different T-cycles. Also, decrease in R may indicate one clock-controlled peak and one masking peak, as previously reported in case of locomotor activity rhythm in Drosophilid species (Prabhakaran and Sheeba, 2012; Rieger et al., 2003). We observed that R is significantly higher in early populations in all three T-cycles which suggests that they maintain constant high consolidation of emergence as observed previously under T24 (middle panel, Figure 4b; Suppl. Table S6). Control flies do not change R among T-cycles, but late flies show significantly higher R under T26 compared to T24 and T22, primarily because of high amplitude of their emergence under T26.
Taken together, these results indicate that peak phase of early flies does not change when they entrain to short or long T-cycles. The fact that emergence occurs maximally at similar ZT across T-cycles supports the idea of larger masking component to this synchronization to T-cycles than a circadian clock mediated entrainment.
Under Skeleton T-Cycles, early Flies Show Phase Lability
Previous experiments (Figures 2-4) suggest some degree of anticipation (a hallmark of circadian clock-controlled rhythms) under full T-cycles. Therefore, we asked, to what extent circadian clock controls the phase of emergence in our stocks. The non-parametric model of circadian entrainment posits that lights during the dawn and dusk transitions entrain the clock and even short light pulses at these phases are sufficient to reproduce the waveform of the rhythm seen under full photoperiod. Masking responses to light depend both on duration of illumination, and intensity of light (Mrosovsky, 1999). We carried out skeleton photoperiod experiments to examine the extent of circadian clock control over the phase of emergence in our stocks, with short duration (0.25 h) light pulses of ~70 lux and asked if these pulses elicit a masking response as well.
Although all populations showed entrainment under all skeleton T-cycles provided, their eclosion waveforms did not closely mimic those under full photoperiods of respective T-cycles (Figure 4a and 5a). Under skeleton T-cycles, early flies became phase labile (left and right panel, Figure 5b). With long T-cycles they advanced their phases, just as control and late flies (Figure 5a and 5b). The change in ΨCoM across different T-cycles was small for early flies, compared to control and late flies (left panel, Figure 5b).
To distinguish the effects of non-parametric or parametric entrainment/ masking we estimated the Sum of Square Differences (SSD) between full and skeleton T-cycles. We also compared the difference between waveforms under full and skeleton T-cycles across T-cycles of different lengths (Figure 5c). If the SSD values are not significantly different from zero, it can be assumed that the light pulses non-parametrically entrained the populations with waveforms similar to full T-cycles, whereas SSD values significantly different from zero will hint toward parametric entrainment or of masking under skeleton T-cycles. Eclosion waveform of early flies showed large changes (in SSD) under skeleton T-cycles, whereas the changes were very small in case of control and late flies (Figure 5c; Suppl. Tables S7, S9—T22 and T26 respectively). We also compared the ΨPeak and ΨCoM of the eclosion rhythm of all populations under different lengths of skeleton T-cycles. ΨPeak of the eclosion rhythm of all populations showed trends similar to ΨCoM but was not significantly different among stocks across T-cycles (right panel, Figure 5b; Suppl. Table S12), mostly due to the fact that the eclosion waveforms were not strictly unimodal in all individual vials. Although, early flies showed phase lability under skeleton T-cycles, they also showed a significantly advanced phase of entrainment (ΨCoM) than control and late flies, across all three regimes (left panel, Figure 5b; Suppl. Table S11), suggesting that in addition to the masking response, advanced phase of circadian entrainment has also been selected for in our early flies. The extent of change in eclosion waveform between skeleton and full T-cycles can be seen in the change in R values (Figure 5b). The early flies show significantly lower R value under T26 skeleton T-cycle, compared to the same under T22 and T24 skeleton T-cycles (middle panel, Figure 5b; Suppl. Table S10).
These results suggest that, in early flies, although positive masking responses to lights-ON strongly influences ΨCoM and ΨPeak of the eclosion rhythm under full photoperiods, under skeleton photoperiods, these phases are clock controlled and significantly advanced (only ΨCoM) compared to control and late flies.
Discussion
While there have been several cases for the existence of masking as a phenomenon mediating biological rhythmicity (Aschoff, 1960; Aschoff and von Goetz, 1988a; Binkley et al., 1983; Redlin and Mrosovsky, 1999), it has been long neglected by circadian biologists; Nicholas Mrosovsky (1999) wrote Nevertheless, as a phenomenon worth study in itself, masking generally has been neglected by circadian biologists. Their attention has been focused more on making sure that interpretations involving masking can be excluded in research on rhythms and on devising ways of eliminating masking effects from measurements of clock phase; these include the use of constant darkness and skeleton photoperiods. (Mrosovsky, 1999)
For the most part, masking has received attention in study of locomotor activity rhythm (Fry, 1947; Hamblen-Coyle et al., 1992; Kempinger et al., 2009; Rieger et al., 2003; Prabhakaran and Sheeba, 2012), although recently it has been invoked to explain aspects of eclosion rhythm in Drosophila (McNabb and Truman, 2008; Thakurdas et al., 2009) and the silk moth Bombyx mori (Ikeda et al., 2019). It is known that the lights-ON signal mediated masking of Drosophila eclosion rhythm is brought about by the release of eclosion hormone as removal of eclosion hormone neurons attenuates the masking response (McNabb and Truman, 2008). A sudden temperature change can also induce masking response in eclosion via the eclosion hormone neurons (Jackson et al., 2005). There is indirect evidence that masking provides some adaptive value (Bloch et al., 2013; Lu et al., 2010), however to the best of our knowledge, there is no experimental evidence for masking evolving in response to selection.
Evolution of Masking in early Populations Along With Advanced Phase of Entrainment
We hypothesized that due to the temporal placement of our selection window for the early flies (ZT22-ZT1), we may have inadvertently selected for individuals evolving a strong masking response to lights-ON (ZT0-ZT1), as we see in Figures 2a and 3a, with very few (<5%) flies emerging in the window of ZT22-ZT0. Also, we observed 25% to 35% flies of early populations emerge immediately within half an hour after lights-ON (ΨPeak = ZT0.5) (Figures 2a and 3a), which led us to think there might be a significant masking component regulating the phase of the eclosion rhythm of early flies.
Unlike control and late populations, upon advancement of lights-ON, many of the early flies eclose immediately after lights-ON rather than what we would have expected if their eclosion rhythm was strongly circadian clock driven (Figure 2a and 2b). Alternatively, this result suggests that the light-sensitive A-oscillator in early flies is stronger or dominant and facilitates re-entrainment in the very next cycle. However, as described in a later part of the discussion on duration and intensity of light, the lights-ON response in eclosion is not as immediate as for other behaviors such as locomotor activity rhythm (as fast as under 5 minutes of lights-ON, Prabhakaran and Sheeba, 2012), so the high emergence immediately after lights-ON on Assay day 2 (Figure 2a) being a result of faster re-entrainment seems highly unlikely. Our hypothesis of strong masking in the early flies was further validated by lower emergence in early population at subjective dawn in DD (Figure 3a and 3b) in contrast to the flies from the other two sets of populations. This result may also be interpreted as—early flies showing higher amplitude expansion under entrainment for eclosion rhythms followed by amplitude reduction in response to transition from LD to DD. However, the high emergence at ZT0.5 (presumably the masking induced peak) under LD is absent in DD, whereas the next highest peak at ZT1 (presumable the clock-controlled peak) under LD is maintained at similar phase under DD (~10% emergence), showing a phase control and is seen in previous experiments as well (Figure 2a). While control and late flies phased eclosion in the expected direction based on circadian clock control under short and long T-cycles (Figures 4a and 5a), the early flies consistently respond by positive masking with high emergence immediately after lights-ON. However, the response of early flies under skeleton pulse-induced T-cycles was remarkable because similar to the control and late flies, they too delayed and advanced emergence phases (ΨCoM) under T22 and T26, respectively (Figure 5).
In the phase advance experiments (Figure 2), although early flies exhibited high emergence immediately after lights-ON at ZT0.5, there was considerable emergence even around ZT1.5—2—this peak was comparable to the peak at ZT1 on Assay day 2 of LD-DD transition experiments (bottom panel, Figure 2a & bottom panel, Figure 3a). We reasoned that the phase advance of lights-ON on Assay day 2 exposed the late night (advance zone) of the PRC of early flies to light (Kumar et al., 2007), thus advancing the clock and hence producing an advanced peak (at ZT 2 in phase advance experiments and ZT1 in LD-DD transition experiments—both at Assay day 2). The exposure to three skeleton T-cycles revealed that indeed early flies maintained significantly advanced ΨCoM compared to control and late flies (Figure 5b), suggesting that our selection protocol has indeed resulted in advanced phase of emergence in early flies along with greater masking which is revealed under full photoperiods. The fact that the control flies show some extent of masking (Figure 3b), and upon selection for advanced phase of entrainment, can give rise to significantly higher masking (early population), suggests that masking-inducing variations are present in populations, albeit in low frequency. Recently, Pegoraro and colleagues have shown that negative masking could evolve when flies were selected for nocturnality (Pegoraro et al., 2019). However, it is important to note that not all short-period flies are expected to show this masking response to lights-ON, for example, pers flies with a period of ~19.5 h, have such an advanced phase of emergence compared to their controls, that their ΨPeak occurs several hours before the lights-ON transition (Qiu and Hardin, 1996). In our populations, this evolved masking response is associated with the early chronotype and their advanced phases after selection under LD12:12 and is unlikely to be driven by the shorter period of the early flies (barely 0.5 h < control).
Interestingly, the locomotor activity rhythm of early flies does not show heightened masking response to lights-ON compared to the control and late flies (up until ~ generation 260, reanalyzed data from Nikhil, Abhilash, and Sharma, 2016; Suppl. Fig. S21). This may be partly due to the fact that, in general, flies show high startle response to lights-ON stimulus (Rieger et al., 2003). Previous studies on these populations have shown that there are no differences in phasing of locomotor activity rhythms under LD12:12, even though there is a significant difference in free running period (Kumar et al., 2007, Nikhil, Abhilash, and Sharma, 2016). For eclosion rhythm, both phase and period have changed significantly among early, control, and late flies (Kumar et al., 2007; Nikhil, Abhilash, and Sharma, 2016). Since period changes are similar for both rhythms while phase divergence among chronotypes is seen only in eclosion rhythm, we speculate that this may be a result of differences in output pathways governing phases of these two rhythms.
Higher Range of “Apparent” Entrainment Achieved by Masking
Our results suggest that apparent phase of an entrained rhythm may be contributed by both masking and the circadian clock, and in some regimes, more by the masking component than the clock (under full photoperiods, Figures 2–4). We introduce the term “apparent entrainment” because under skeleton T-cycles we see that the early flies have evolved an earlier phase of emergence which is clock-driven, along with positive masking. We reason that the early flies have evolved “apparently entrained” phases via changes in both the clock and the masking response. We do not equate this “apparent entrained” state with “entrainment” which is a core property of the circadian clock and its oscillators (Bittman, 2020), but merely mean a “synchronized state” of the overt rhythm with an environmental cyclic cue. We caution that “apparent entrained” states may present as clock driven entrained states, and without targeted experimental designs and adequate temporal sampling resolution, be hard to identify. Our own previous studies with 2 h resolution suggested that the early strains are entrained with an advanced phase of emergence, however, by increasing sampling frequency, we uncovered the effect of masking on this “apparently entrained” emergence profile.
Our studies also show that a spectacularly high range of “apparent entrainment” can be achieved just by masking, at least for the eclosion rhythm. Under extreme T-cycles of T20 and T28 early flies still have high emergence immediately after lights-ON and show an apparent entrained behavior, whereas percentage entrainment of control and late flies under these regimes are significantly lower than early flies. Altogether our experiments make a very strong case that in the process of creating the early chronotypes, in addition to them evolving a faster clock with advanced phase of entrainment, they have also evolved robust positive masking responses to lights-ON. This facilitates the idea that in nature, organisms may use masking—a clock-independent phenomenon, as a mechanism to phase themselves to appropriate times of the day, for example, eclosion being gated to the early part of the day to prevent desiccation and enhance survival rate. It can be argued that masking may be an evolutionary disadvantage in the sense that to compensate for a highly “noisy” clock, masking may evolve to maintain specific phases locked to a zeitgeber(s) and does not have the flexibility of phase lability in complex environments. Indeed, previous work from our lab has shown that under complex zeitgeber conditions (light:dark 12:12 + warm: cool 12:12; in-phase and out-of-phase), early flies show remarkable resilience to shift phases and stay phase locked to the lights-ON signal, whereas control and late flies change their phases more readily (Abhilash et al., 2019). Moreover, empirical evidence suggests that a fully functional clock and masking can co-exist in an animal and in all probability, these are complementary mechanisms for organisms to maintain specific phases (Aschoff and von Goetz, 1988a; Mrosovsky, 1994; Redlin and Mrosovsky, 1999, 2004; Rensing, 1989).
The Duration and Intensity of Light Dramatically Alters Magnitude of Response in Eclosion
The light used in all our experiments was of ~70 lux (to match the long-term selection maintenance regime) and can be considered low intensity compared to majority of the eclosion rhythm studies in Drosophila (which typically used ~750 lux). It is especially so in those studies where relationship between the lights-ON signal and downstream pathways have been analyzed and linked to timing of eclosion (Baker et al., 1999; McNabb and Truman, 2008). They show that lights-ON signal can rapidly induce eclosion of up to ~20% of the waveform within about 10 min. This suggests that a skeleton photoperiod should be able to induce high masking responses, which we did not observe in any of the skeleton T-cycles. This can be explained by the different light intensity used in our experiments (at least 10 times lower). Nevertheless, our regime induces ~35% emergence in a mere 30 min when the lights-ON was advanced (Figure 2b). We propose that similar number of photons integrated over time could induce high emergence in early populations, compared to control and late populations.
Another important difference from previous studies which were mostly aimed to understand the mechanisms of eclosion hormone release and downstream pathways leading to the act of eclosion is, that they used cultures containing similar developmental stages. To observe light-induced emergence, typically experiments were set up such that flies were expected to emerge in the next ~60 min after light pulses were given (McNabb and Truman, 2008). Our studies together with this information from previous reports lead us to hypothesize that photon integration is a part of this masking response, and that as soon as a threshold is crossed, masking response to lights-ON is observed.
Plausible Mechanisms Driving Higher Masking in early Populations
Some hint of possibility of the evolution of masking mechanisms are seen in an in-silico study, where gene regulatory networks were allowed to evolve under selection for correct prediction of phases under light-dark cycles (Troein et al., 2009). It was seen that under a simple LD12:12 condition, only delayed light responses were selected for, while no oscillatory mechanism was found to evolve. This suggests that in a minimal environment, where the only cycling cue is a light-dark cycle, oscillators may not be necessary to achieve particular phases, and a delayed light response may as well do the trick. In our selection regime, environmental conditions are similarly minimal, with only one cycling cue, a ~70 lux LD12:12 square light cycle (abrupt transitions) regime with constant ambient temperature of 25°C ± 0.5°C, and results from Troein and colleagues supports the idea of evolution of simpler phasing mechanisms, such as strong masking. This evolved masking response may also be governed by co-evolved non-circadian photosensitivity. Thus, there are the possibilities of (a) strong photosensitivity and eclosion hormone neurons coupling or (b) heightened photosensitivity of the prothoracic gland itself having evolved in the early flies.
To the best of our knowledge this work is the first experimental demonstration that masking can evolve as a response to selection for phase of entrainment. We propose that masking can be a valid mechanism by which organisms show early chronotype in an environment where strong light transitions are present. Further investigations into the molecular mechanisms and neuronal control of this masking response are needed, for which some plausible targets have been mentioned above. Overall, this work highlights the complex mechanisms of “apparent” light entrainment and provides an experimental framework to dissect out relative contributions of masking and the circadian clock regulating timing of a behavior.
Supplemental Material
sj-pdf-1-jbr-10.1177_0748730421997262 – Supplemental material for Evidence for Co-Evolution of Masking With Circadian Phase in Drosophila Melanogaster
Supplemental material, sj-pdf-1-jbr-10.1177_0748730421997262 for Evidence for Co-Evolution of Masking With Circadian Phase in Drosophila Melanogaster by Arijit Ghosh, Pragya Sharma, Shephali Dansana and Vasu Sheeba in Journal of Biological Rhythms
Footnotes
Acknowledgements
We are extremely grateful to late Professor Vijay Kumar Sharma for providing this beautiful model system to address such questions regarding phase of entrainment of the circadian clock. The maintenance of these populations throughout last ~17 years have seen multiple graduate and undergraduate students working day and night, and we stand on the shoulders of the body of knowledge accumulated by previous lab members. The assays carried out in this study required intense work for 24 h at a stretch of 4-5 days, which would not have been possible but for the able assistance of summer interns (Sankeert Satheesan and Bharathy Nagarajan) during pilot experiments and standardization of methods. We thank Dr. Abhilash Lakshman and 2 anonymous reviewers for carefully reading our manuscript and suggesting some very useful changes. We would also like to acknowledge financial support from the Science and Engineering Research Board, New Delhi, to V.S. (CRG/2019/006802); intramural funding from Jawaharlal Nehru Center for Advanced Scientific Research (JNCASR), Bangalore; the Council of Scientific & Industrial Research (CSIR), New Delhi, India, for a junior research fellowship and a senior research fellowship to A.G.; JNCASR, for a fellowship to P.S.; the Department of Science and Technology, New Delhi, India for an INSPIRE fellowship for masters studies to S.D.; and consumable grant from the Department of Biotechnology (DBT), Government of India, to V.S. (BT/INF/22/SP27679/2018).
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Note
Supplementary material is available for this article online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
