Abstract
Light is the most important zeitgeber for the synchronization of the Drosophila melanogaster circadian clock. In nature, there is twilight, and the nights are rarely completely dark, a fact that is usually disregarded in lab experiments. Recent studies showed contrary effects of simulated twilight and moonlight on fly locomotor activity, with twilight shifting morning and evening activity into the day and moonlight shifting it into the night. A currently unanswered question is, what may happen to locomotor activity when flies are exposed to more natural conditions in which both moonlight and twilight are simulated? Our data demonstrate that flies are able to integrate twilight and moonlight. However, twilight seems to dominate over moonlight as both, morning and evening activity peaks, take place at dawn or at dusk, respectively, and not during the night. Furthermore, nocturnal activity decreases in the presence of twilight. The compound eyes are essential for this behavior, and by investigating different photoreceptor mutants, we unraveled the importance of photoreceptor cells 7 and 8 for wild-type phases of the activity peaks. To adjust nocturnal activity levels to a wild-type manner, all photoreceptor cells work together in a complex way, with rhodopsin 6 having a prominent role.
In nature, many zeitgebers cycle within the span of a 24-h day, with light being the most important environmental cue for synchronizing the circadian clocks of animals and plants. Lab studies mostly apply rectangular light-dark cycles of 12 h light and 12 h darkness (LD 12:12) to simulate day-night changes, which do not reflect the actual changes of light in nature as dawn, dusk, and moonlight are consequently disregarded. All of these light features cause significant changes in animal behavior and physiology: moonlit nights lead to an accelerated recovery from jetlag, can increase the activity of nocturnal animals, and, in humans, can affect sleep (Erkert and Cramer, 2006; Evans et al., 2009; Cajochen et al., 2013). Moreover, the simulation of twilight by nature-like increases or decreases of light intensity improves entrainment compared to abrupt lights-on and lights-off in many species and is able to advance Per1, Per2 and PER1, PER2 oscillation profiles under long photoperiods in mice (Boulos et al., 1996a; Boulos et al., 1996b; Boulos et al., 1996c; Boulos et al., 1996d; Fleissner and Fleissner, 1998, 2002; Danilenko et al., 2000; Boulos and Macchi, 2005; Comas and Hut, 2009; Sosniyenko et al., 2009).
In Drosophila melanogaster, simulation of moonlit nights leads to an advance of the morning (M) and a delay of the evening (E) activity peak, which correlates with changes in PER cycling in specific clock neurons (Bachleitner et al., 2007). In addition, the level of nocturnal activity increases significantly in moonlit nights. The latter is independent of a functional clock and is especially mediated by rhodopsin 1 (Rh1) and rhodopsin 6 (Rh6) of the compound eyes (Kempinger et al., 2009; Schlichting et al., 2014). The effect of twilight simulation goes instead in the opposite direction: dawn and dusk provoke a delay of the M and an advance of the E peak, and the flies reduce their nocturnal activity to a minimum (Rieger et al., 2007). In addition, twilight is able to improve the ability to phase-delay the E peak under long photoperiods (Rieger et al., 2012).
To fulfill the difficult sensory task of using light as a zeitgeber, a complex visual system is necessary (Foster and Helfrich-Förster, 2001). To perceive light, Drosophila uses cryptochrome as well as 6 rhodopsins (Stanewsky et al., 1998; Ceriani et al., 1999; Emery et al., 2000; Helfrich-Förster et al., 2001; Rieger et al., 2003; Busza et al., 2004). Cryptochrome is a circadian blue-light photoreceptor, and it is expressed in the compound eyes as well as in several clock neurons (Benito et al., 2008; Yoshii et al., 2008). Rhodopsins are expressed in 7 eye structures: 2 compound eyes, 2 Hofbauer-Buchner (H-B) eyelets, and 3 ocelli (Hofbauer and Buchner, 1989; Salcedo et al., 1999; Rister et al., 2013). This study focuses on the compound eyes, which are the most prominent visual organs of the fly; they consist of about 800 ommatidia, each containing pigment cells as well as 8 photoreceptor cells (R1 to R8). The photoreceptor cells express specific rhodopsins. In particular, the outer photoreceptor cells, R1 to R6, express rhodopsin 1, whereas 70% of the inner photoreceptor cells (R7 and R8) express rhodopsin 4 in R7 and rhodopsin 6 in R8 (yellow subtype), and 30% express rhodopsin 3 in R7 and rhodopsin 5 in R8 (pale subtype) (Rister et al., 2013). Only few specialized ommatidia in the dorsal rim area express only rhodopsin 3 in both inner photoreceptor cells (Wernet and Desplan, 2014).
The first aim of this study was to investigate fly behavior when moonlight and twilight are simulated at the same time. We show that twilight dominates over moonlight in terms of peak timing and nocturnal activity, even though the moonlight-induced nocturnal activity increase was still present. The second aim was to unravel the contribution of the different photoreceptor cells and rhodopsins in perceiving gradual changes in irradiance during twilight. We demonstrate that flies lacking compound eyes fail to adjust their behavior in a normal way and that inner and outer photoreceptor cells cooperate in adapting fly behavior to combined twilight and moonlight with a prominent role of rhodopsin 6 in dim-light detection.
Materials and Methods
Fly Strains and Rearing
Light sensitivity of the circadian clock is known to be influenced by a naturally occurring polymorphism in the timeless (tim) gene (Sandrelli et al., 2007; Tauber et al., 2007). To exclude possible effects of this tim polymorphism, we crossed all photoreceptor mutants into the ls-tim background (Schlichting et al., 2014) and used the wild-type strain WTCantonS with the same ls-tim background as control. In addition, we recorded 2 additional wild-type strains with completely different backgrounds to see whether they responded similarly to the combination of twilight and moonlight. These are WTALA and WTLindelbach, which have been described in Rieger et al. (2012) and Schlichting et al. (2014), respectively. WTLindelbach is s-tim and WTALA carries a mixture of both tim forms.
clieya mutants, which lack the compound eyes but retain the H-B eyelets and the ocelli, served as negative controls (Bonini et al., 1993; Schlichting et al., 2014). To eliminate R1 to R6, we used ninaE17 (neither inactivation nor afterpotential E) mutants (O’Tousa et al., 1985; Kumar and Ready, 1995). To impair R7 function, we used 2 different strains: (1) sevLY3 mutants, in which R7 do not develop and therefore all R8 express rhodopsin 6, and (2) rh31rh41 double mutants, which leave the rhodopsin distribution in R8 unchanged (Benzer, 1967; Chou et al., 1999; Vasiliauskas et al., 2011). To manipulate R8, we used rhodopsin 5 and rhodopsin 6 single mutants (rh52 and rh61) as well as the relevant double mutant (rh52;rh61) (Cook et al., 2003; Yamaguchi et al., 2008). To render both inner photoreceptor cells out of function, rh52;rh31rh41rh61 quadruple mutants were investigated.
All flies were raised on standard Drosophila medium (0.8% agar, 2.2% sugar beet syrup, 8.0% malt extract, 1.8% yeast, 1.0% soy flour, 8.0% corn flour, and 0.3% hydroxybenzoicacid) at 25 °C in LD 12:12.
Behavior Recording and Light Programs
For recording locomotor activity, a homemade system was used (Helfrich-Förster, 1998; Schlichting and Helfrich-Förster, 2015). Two- to 4-day-old single male flies were transferred into photometer cuvettes with food and water supply on one end. On the other end, an infrared beam was installed and a computer measured the number of beam crosses in 1-min intervals. Behavior was recorded in a climate-controlled chamber at 20 °C. As light source, “white” LEDs (Lumitronix LED-Technik GmbH, Jungingen, Germany) were used with additional neutral density filters (Lee Filters Worldwide, Andover, UK) for fine-tuning of light intensity.
To distinguish the effect of simulated dawn and dusk on behavior, we recorded the flies at 4 different light conditions. In the first week, we applied a rectangular light-dark cycle of 12 h light and 12 h darkness (LD 12:12), and in the second week, twilight was simulated using a gradual increase of light intensity in the morning and a gradual decrease in the evening within 1.5 h each (LDR) (Rieger et al., 2007). In the same way, we investigated conditions including moonlight: flies were recorded for 1 week in light-moonlight conditions (LM, moonlight intensity of 0.01 lux), and in the second week, twilight simulation was added (LMR condition). For wild-type flies, we investigated 4 different daylight intensities (10, 100, 1000, and 10,000 lux) to evaluate effects that depended on daylight intensity and to find the optimal intensity for testing the photoreceptor mutants. Experiments were repeated at least twice. Finally, we had 17 to 32 flies for each genotype that survived the entire experiment and could be used to analyze the behavior.
Data Analysis and Statistics
The raw data were plotted as actograms using the ActogramJ plugin for Fiji (available at http://fiji.sc/Downloads; Schmid et al., 2011). In the next step, average activity profiles were analyzed using the last 4 days of each light condition. All activity profiles were normalized to 1 and plotted using the program Qtiplot (version 0.9.8.9; Ion Vasilief, Craiova, Romania). To analyze peak timing, the activity profiles of single flies were smoothened by a moving mean of 30, and the timing of the peak was determined manually as described in Schlichting and Helfrich-Förster (2015). In addition, we calculated the relative nocturnal activity for each experiment by dividing the sum of activity between ZT12 and ZT0 by the whole daily activity. The analysis was done for each single fly, and the values obtained were then averaged.
Statistical analysis was performed using Systat11 (Systat Software, San Jose, CA). Normal distribution was tested using a 1-way Kolmogorov-Smirnov test. In case of normal distributed data, a 1- or 2-way analysis of variance (ANOVA) was applied. For nonnormally distributed data, a Mann-Whitney U test (2 groups) or a Kruskal-Wallis test (more than 2 groups) followed by a Wilcoxon comparison, including Bonferroni adjustment, was used.
Immunocytochemistry and Image Analysis
To assess the distribution of rhodopsin 5 and 6 in the retina, 18-day-old male flies were entrained in LD 12:12 at 20 °C and fixed for 2.5 h in 4% paraformaldehyde (PFA) in phosphate-buffered saline (PBS). After rinsing 4 × 15 min each with PBS containing 0.5% Triton-X (PBST), the retinas were dissected in PBST and the lamina was removed. Retinas were blocked for 30 min in 5% normal goat serum (NGS) in PBST and subsequently incubated in the primary antibody solution consisting of rabbit anti–rhodopsin 6 (1:1000, gift of C. Desplan, New York University, New York, NY; Tahayato et al., 2003) and mouse anti–rhodopsin 5 (1:50, gift of S. Britt, University of Colorado, Aurora, CO; Salcedo et al., 1999) in PBST containing 5% NGS and 0.02% NaN3. Samples were incubated in the primary antibody for 2 nights at room temperature (RT). After rinsing 5 × 20 min each with PBST, the secondary antibody was applied overnight (Alexa Fluor 555 goat anti–mouse and Alexa Fluor 635 goat anti–rabbit, each in a dilution of 1:200 in PBST and 5% NGS). For the following 3 to 5 days, the retinas were rinsed several times per day until the red pigmentation of the eyes was fully washed out to eliminate autofluorescence. Retinas were embedded in Vectashield mounting medium (Vector Laboratories, Burlingame, CA) in a way that the cornea of the eye laid on the glass slide.
The retinas were analyzed using laser scanning confocal microscopy (Leica TCS SPE; Leica, Wetzlar, Germany). To excite the secondary antibody fluorophores, 2 different lasers were sequentially used (532 and 635 nm) and 2-µm-thick stacks were obtained. All images were analyzed using Fiji. We counted manually the number of ommatidia expressing Rh5 or Rh6 in the entire retina and did so for 7 different retinas.
Results
Wild-Type Behavior Under LD, LDR, LM, and LMR With Different Daylight Intensities
Our previous studies have shown that fly activity pattern depends not only on the presence of twilight or moonlight but also on daylight intensity (Rieger et al., 2007; Schlichting et al., 2014). Therefore, we recorded activity of the wild-type strains under LD, LDR, LM, and LMR at 4 different daylight intensities (10, 100, 1000, and 10,000 lux), respectively. Consistent with previous studies, all 3 wild-type strains showed bimodal activity patterns with M and E peaks, with the phase of the M peak occurring earlier and that of the E peak later with increasing daylight intensity (shown for WTCantonS in Fig. 1A and WTLindelbach and WTALA in Suppl. Fig. S1A,F). Here, we determined only the phase of the E peak and found that it was significantly delayed with increasing light intensity in all wild-types and at all 4 light regimes (LD, LDR, LM, LMR) (p < 0.001; Fig. 1B,C; Suppl. Fig. S1). This delay was most pronounced under moonlight conditions in WTLindelbach (Suppl. Fig. S1C) and the least pronounced in WTCantons (Fig. 1C). In addition, E peak timing depended significantly on the light regime, and this was again similar in all wild-type strains (Fig. 1B,C; Suppl. Fig. S1): simulated twilight advanced the E peak, whereas moonlight delayed it. Consequently, the earliest E peaks were found under LDR (Fig. 1B) and the latest under LM conditions (Fig. 1C; see also Suppl. Fig. S1). Most important, twilight appeared to prevent the E peak from occurring during the night (Fig. 1B,C; Suppl. Fig. S1B,C,G,H). Under LMR, the E peak occurred always during dusk (between 10 and 1000 lux) or directly at lights-off (at 10,000 lux), whereas under moonlight alone (LM), the E peak was after lights-off when daylight intensity exceeded 100 lux. This indicates that advancing the E peak by twilight dominates over delaying the E peak by moonlight.
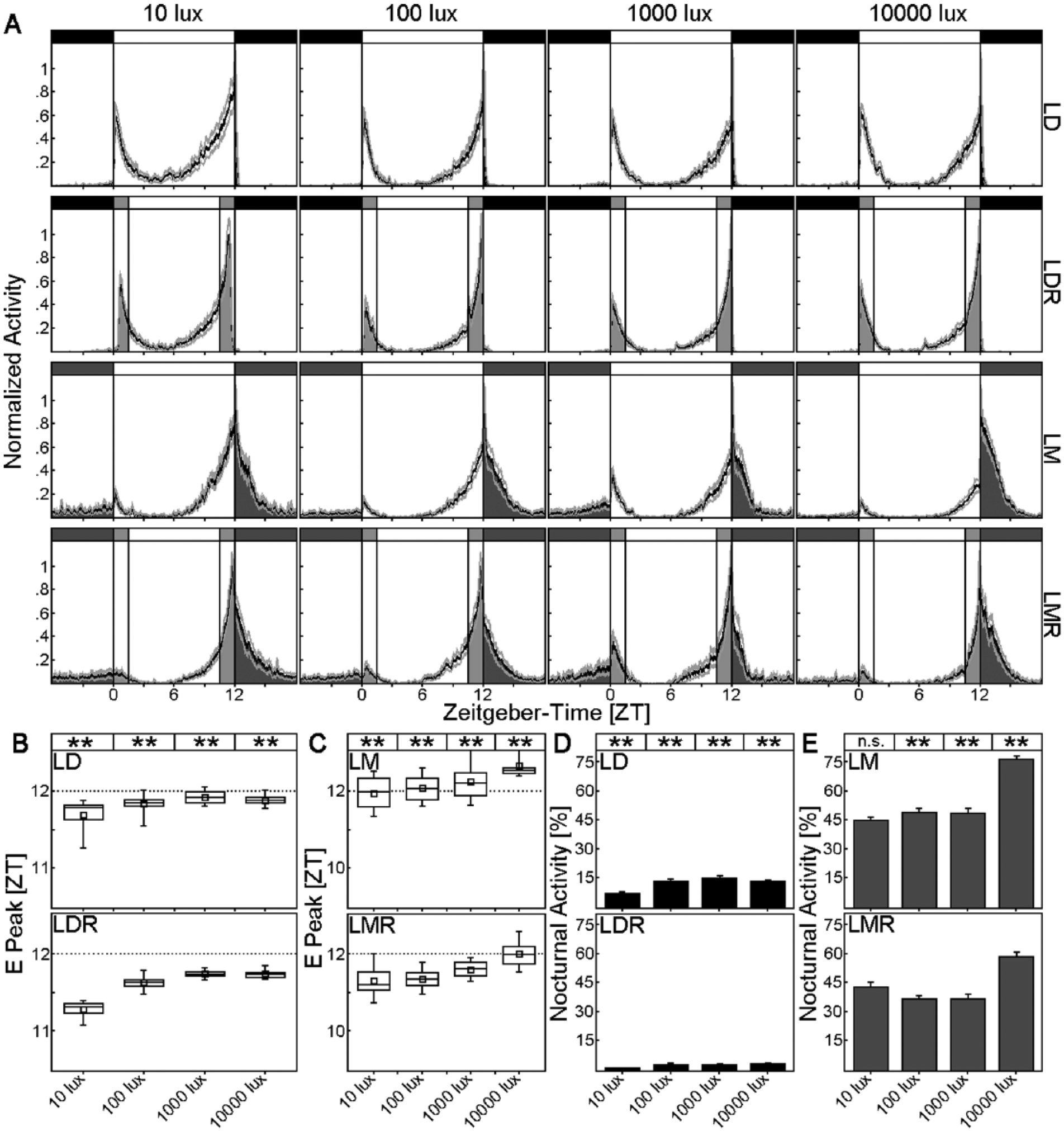
Rhythmic behavior of WTCantonS flies under 4 different light regimes: light-dark cycles (LD, D indicated by black), light-dark cycles with simulated twilight (LDR, twilight [R] indicated by light gray), light-moonlight cycles (LM, M indicated by dark gray), and light-moonlight cycles with simulated twilight (LMR, twilight [R] indicated by light gray, M indicated by dark gray). (A) Average activity profiles under the 4 light regimes with daylight intensity increasing from 10 to 10,000 lux (left to right). Black lines represent the mean of at least 17 flies; the gray lines represent the SEM. (B) Timing of the E peak under LD (upper panel) and LDR (lower panel). Dotted lines indicate lights-off (at ZT12). Timing of the E peak depended on daylight intensity (the higher the light intensity, the later the E peak). Furthermore, the E peak occurred significantly earlier under LDR than under LD at all 4 daylight intensities (p < 0.001) (as indicated by asterisks on top of B). (C) Timing of the E peak under LM (upper panel) and LMR (lower panel). Again, the E peak occurred earlier in LMR compared to LM (p < 0.001), indicating a dominant role of twilight for timing of the E peak. Labeling as in B. (D) Percentage of nocturnal activity (from whole-day activity) in LD and LDR. Under LDR, nocturnal activity was significantly lower than under LD (p < 0.001) as indicated by the asterisks on top. (E) Percentage of nocturnal activity (from whole-day activity) in LM and LMR. Moonlight generally increased nocturnal activity, and this effect was significantly higher under LM than under LMR, at least between 100 and 10,000 lux (see asterisks on top). n.s., not significant; **p < 0.001.
Daylight intensity and light regime influenced also nocturnal activity. The flies tended to reduce diurnal and increase nocturnal activity with increasing daylight intensity under all light regimes. As already found for the daylight intensity–dependent delay of the E peak, this tendency was slightly different in the 3 wild-type strains and the least pronounced, although still significant, in WTCantonS (Fig. 1D,E). WTLindelbach flies that carry the s-tim allele showed a linear and steep increase of nocturnal activity with increasing daylight intensity, whereas WTALA flies that carry a mixture of s-tim and ls-tim behaved in between WTCantonS and WTLindelbach flies (Suppl. Fig. S1).
In all strains, the highest nocturnal activity was present under LM conditions, followed by LMR, LD, and LDR (Fig. 1D,E; Suppl. Fig. S1D,E,I,F), and these differences were significant (p ≤ 0.004). Thus, moonlight could even shift activity into the night in the presence of twilight, although nocturnal activity in LMR was lower compared to LM alone (Fig. 1E; Suppl. Fig. S1I,F).
Photoreceptors of the Compound Eyes Mediate E Peak Timing and Nocturnal Activity
To elucidate the contribution of the different compound eye rhodopsins to the adaptation of fly activity patterns to the 4 light regimes, we chose a light intensity of 100 lux, because the WTCantonS control flies strongly responded to twilight and moonlight at this light intensity.
We found that E peak timing and nocturnal activity levels depended significantly on the compound eyes under all 4 light regimes (p < 0.001) (Figs. 2, 3). The more photoreceptor cells or rhodopsins were absent, the earlier the evening peak and the lower the nocturnal activity. This can be best seen in Figure 3, where we arranged the photoreceptor mutants according to their phenotype severity, with the mutants lacking most photoreceptors to the right.
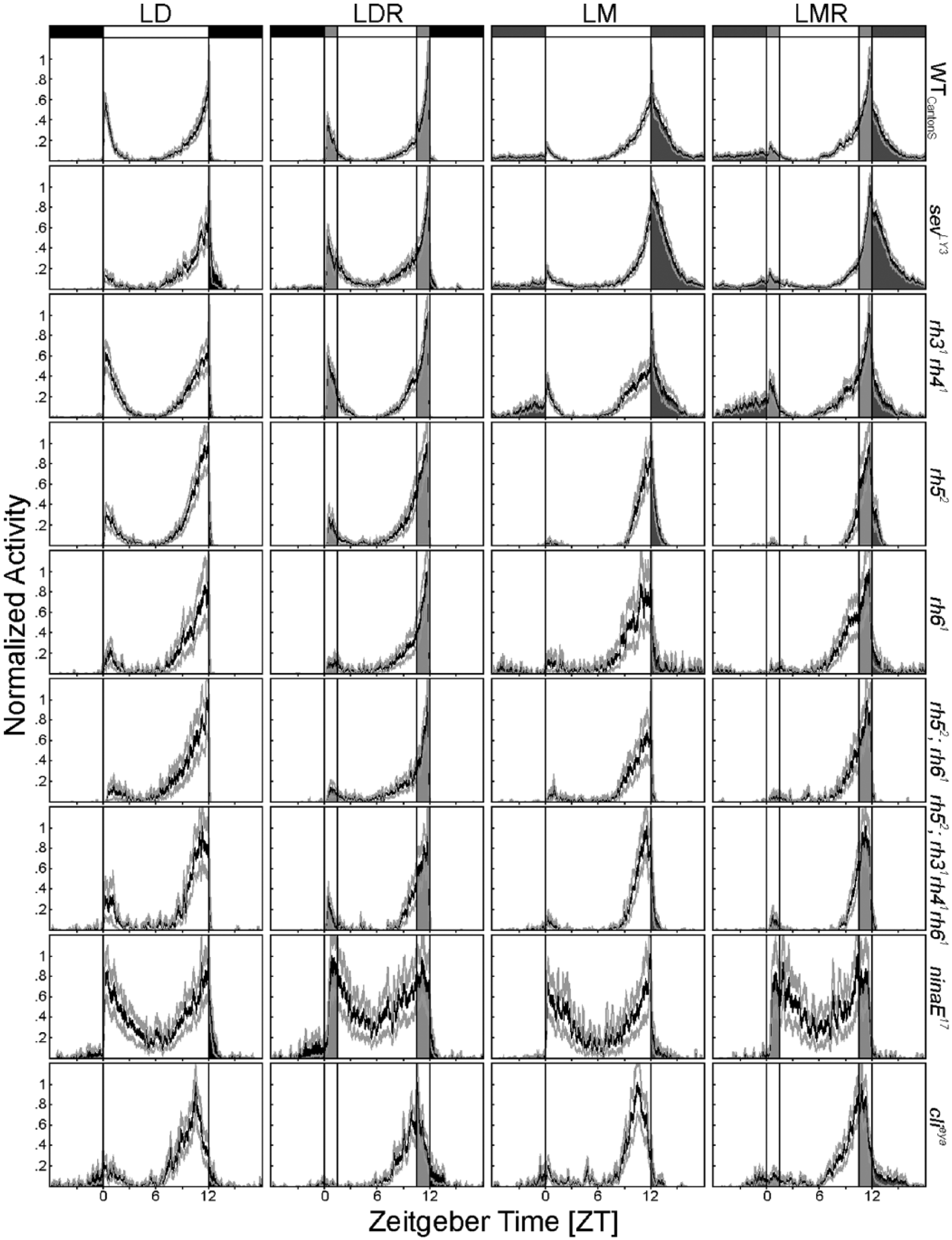
Average activity profiles of WTCantonS flies and all investigated photoreceptor mutants in LD, LDR, LM, and LMR (from left to right) at a daylight intensity of 100 lux. As in Figure 1A, the average values of at least 17 flies (±SEM) are depicted in each diagram. Activity in completely dark nights is represented in black, during moonlight in dark gray, during dawn and dusk in light gray, and in times of maximal light intensity in white. All flies were able to entrain to the investigated light conditions with a prominent E peak and a smaller M peak. Nocturnal activity and the timing of the E peak were clearly influenced by the absence of certain photoreceptor cells or rhodopsins. The highest nocturnal activity was present in sevLY3 mutants, while nocturnal activity was wild-type like in rh31rh41 mutants and reduced in all other mutants. The E peak occurred earlier, with increasing severity of the photoreceptor mutation being the earliest in complete eyeless flies (clieya). In addition, E peak timing and nocturnal activity depended on the light regime. For quantification of E peak timing and nocturnal activity levels, see Figure 3.
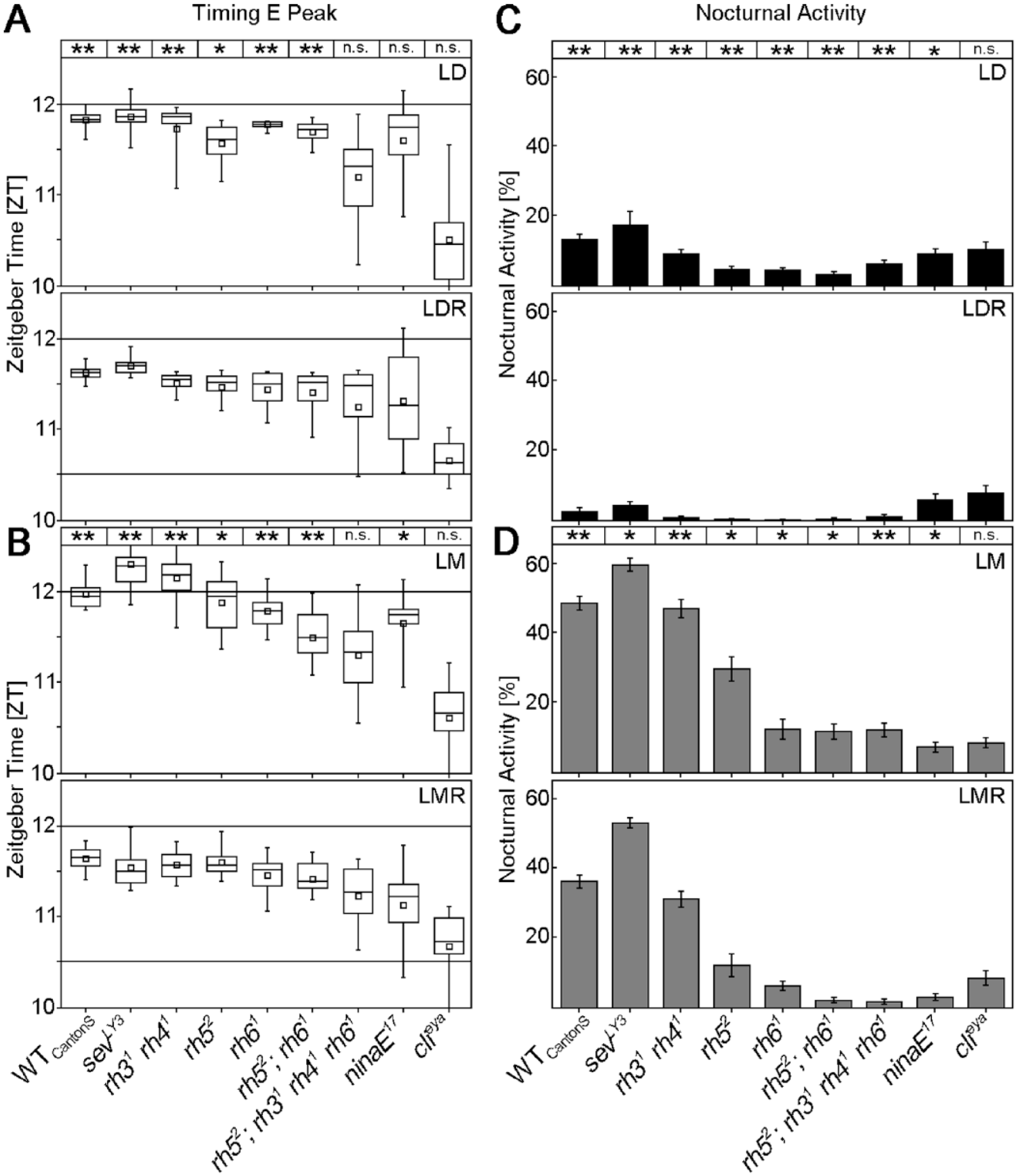
Timing of E peak and levels of nocturnal activity in WTCantonS flies and all investigated photoreceptor mutants. Values are calculated from the data shown in Figure 2. E peak timing and nocturnal activity are compared between LD and LDR (A, C, respectively) and between LM and LMR (B, D, respectively). Significant differences between the 2 conditions are indicated as asterisks on top. n.s., not significant; *p < 0.05, **p < 0.001. For details, see the text.
In the following, we will not consider absolute differences in E peak timing and nocturnal activity between controls and mutants but concentrate on the responses to twilight and moonlight of each mutant, since this was the main aim of this study. Eyeless clieya mutants did not respond to moonlight or to twilight; their E peak always occurred at about 1.5 h before lights-off (p > 0.472; Fig. 3A,B), and they always spent about 10% of their daily activity in the night (Fig. 3C,D). ninaE17 mutants that lack the outer photoreceptor cells (R1 to 6) responded to twilight by advancing their E peak in LMR compared to LM (p = 0.002) and showed a tendency to do so in LDR compared to LD (p = 0.062) (Fig. 2A,B). In addition, they slightly but significantly reduced nocturnal activity in response to twilight (LDR and LMR; p ≤ 0.02) (Fig. 2C,D). rh52;rh31rh41rh61 quadruple mutants, in which both inner photoreceptor cells were impaired, always showed their E peak at the same time (~1 h before lights-off; p > 0.386), as did completely eyeless mutants. Nevertheless, these mutants clearly responded to moonlight and twilight with respect to nocturnal activity (Fig. 3C,D). This result indicates that different photoreceptors contribute to the phasing of the E peak and the control of nocturnal activity. The inner photoreceptor cells appear essential for phasing the E peak, while all photoreceptor cells seem involved in adjusting nocturnal activity levels.
Rhodopsin 6 (and Rhodopsin 1) Plays a Special Role in Sensing Moonlight
Next, we aimed to unravel the role of the different rhodopsins in sensing moonlight and twilight. Clearly, all 5 rhodopsins of the compound eyes contributed to the increase of nocturnal activity in response to moonlight, since only completely eyeless flies failed to increase their activity in moonlit nights, whereas all tested single or double mutants were able to do so (Fig. 3D). Similarly, all 4 rhodopsins of the inner photoreceptor cells contributed to the shift of the E peak into twilight, given only quadruple mutants (rh52;rh31rh41rh61), which lack the 4 rhodopsins, failed to shift the E peak into dusk in LDR and LMR. Flies lacking only rhodopsin 3 and 4 (sevLY3 and rh31rh41 mutants) or rhodopsin 5 and/or 6 (rh52, rh61 and rh52;rh61 mutants) were still able to shift their E peak into dusk in LDR (p < 0.011) and LMR (p < 0.004) (Fig. 3A,B).
In spite of the obvious interplay of all rhodopsins in twilight and moonlight detection, rhodopsin 6 (and to some degree also rhodopsin 1) appear to have a special role in detecting moonlight in the presence of twilight. Mutants lacking rhodopsin 6 (rh61, rh52;rh61 and rh52;rh31rh41rh61 mutants) or rhodopsin 1 (ninaE17) barely responded to moonlight when twilight was additionally present (Fig. 4A). sevLY3 mutants were exceptional, because they increased nocturnal activity in response to moonlight more than wild-type flies did (p < 0.001). This may most likely be caused by their high rhodopsin 6 expression (Chou et al., 1999; Vasiliauskas et al., 2011). We found that virtually all R8 cells of our sevLY3 mutants expressed rhodopsin 6 (Fig. 4B,C). In contrast, rh31 rh41 double mutants showed a wild-type rhodopsin 6 distribution (Fig. 4B,C) and also responded to moonlight in a wild-type manner (Fig. 4A).
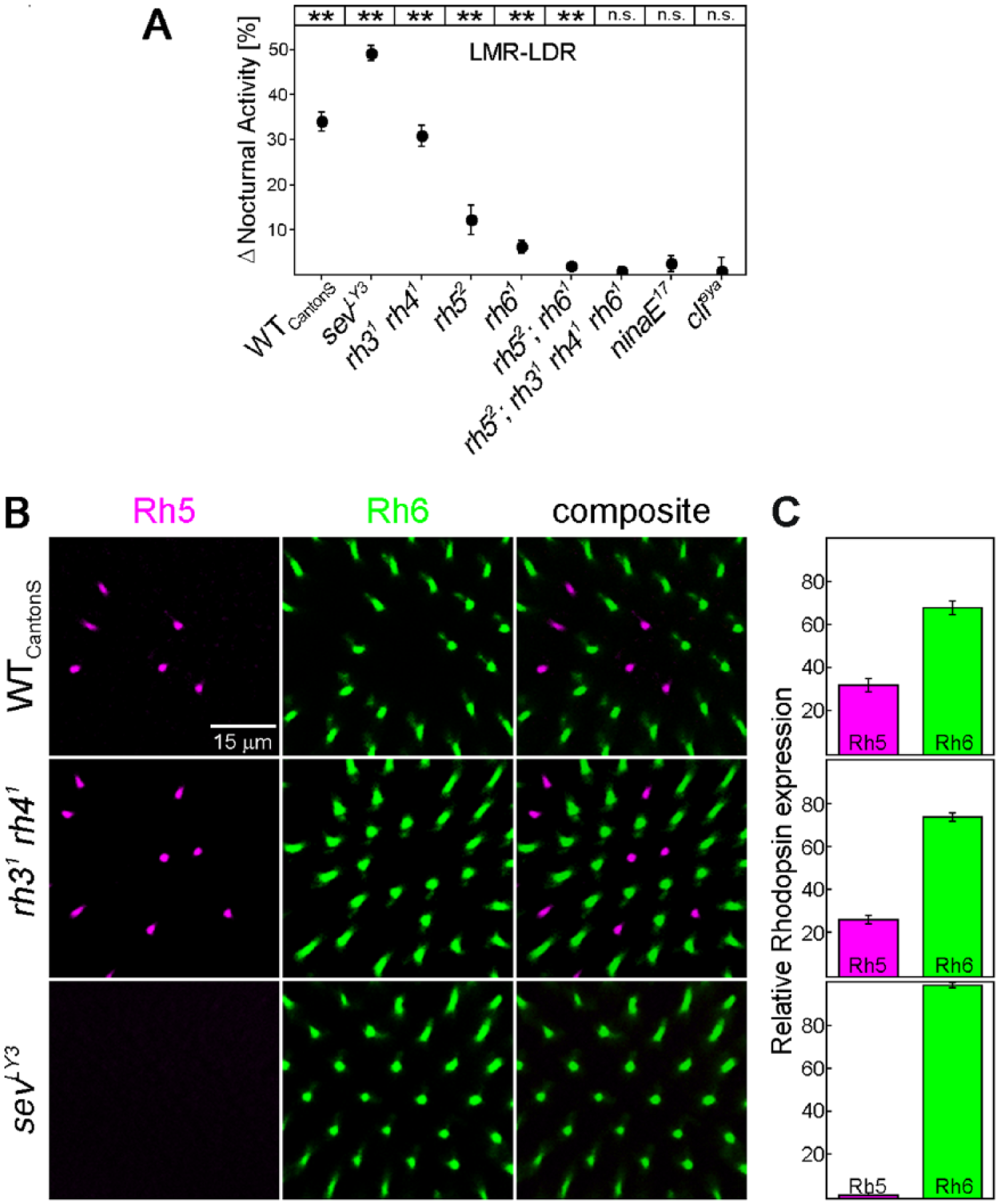
Effect of the different rhodopsins on moonlight detection in the presence of twilight plus rhodopsin 5/6 distribution in the retina of selected mutants. (A) Differences in nocturnal activity between LMR and LDR were calculated for WTCantonS flies and all investigated mutants (±SEM). Asterisks on top of the diagrams indicate significant effects of moonlight in the presence of twilight. n.s., not significant; * p < 0.05, ** p < 0.001. sevLY3 mutants strongly increased nocturnal activity in LMR compared to LDR, whereas rh31rh41 mutants behaved wild-type like. (B) Distribution of rhodopsin 5 and 6 in the retina of 18-day-old WTCantonS flies and rh31rh41 and sevLY3 mutants. sevLY3 mutants almost exclusively expressed rhodopsin 6 in photoreceptor cell R8, whereas rh31rh41 mutants were wild-type like. The scale bar applies to all images. (C) Calculated percentage of rhodopsin 5 and rhodopsin 6 expression in R8. WTCantonS flies and rh31rh41 mutants showed the expected ratio of 30:70, whereas sevLY3 mutants expressed rhodopsin 6 in virtually 100% of R8 cells.
Discussion
The main aim of the present study was to reveal the impact of combined twilight and moonlight on daily activity patterns of fruit flies. We tested the behavior of 3 wild-type strains to unravel the principal effects of the applied light regimes as well as the behavior of 8 photoreceptor mutants to reveal the role of compound eyes, different photoreceptor cells, and rhodopsins.
Twilight Dominates Over Moonlight
The presence of twilight significantly reduced fly nocturnal activity in moonlit nights, hence making their activity pattern look more similar to that observed under outdoor conditions (Vanin et al., 2012). Under natural light and temperature conditions, flies were not nocturnal, nor were they in full-moon nights. Our results indicate that the lack of nocturnal activity in nature is caused not only by lower temperatures at night but also by the presence of twilight that dominates over the moonlight effects even under lab conditions. Most interestingly, all tested wild-type strains behaved in a very similar way. Slight differences were evident in the shift of the E peak into the night and the increase of nocturnal activity under moonlight conditions with increasing daylight intensity. Here, the s-tim flies, such as WTLindelbach, responded most sensitively to an increase of daylight intensity, whereas the ls-tim flies, such as WTCantonS, showed from the beginning onward a rather late E peak and high nocturnal activity. E peak timing and nocturnal activity level were only moderately altered when daylight intensity increased. WTALA flies carrying a mixture of both tim alleles behaved intermediate to the two others. This is consistent with previous studies showing that the clock of s-tim flies is more light sensitive compared to ls-tim flies (Sandrelli et al., 2007; Tauber et al., 2007). It is very likely that also other still unknown gene polymorphisms contribute to fly responses to twilight and moonlight. Nevertheless, for the present study, it is most important to note that the here reported principal responses are very similar in all 3 investigated wild-type strains. Thus, these seem rather independent of the genetic background.
Simultaneous Simulation of Twilight and Moonlight Combines Masking and Clock Effects in Behavior
Generally, light has 2 principal effects on fly activity: (1) direct effects (inhibiting or promoting activity) and (2) phase-shifting (entraining) effects on the circadian clock that controls fly activity rhythm. The direct light effects on activity are also known as “masking” because they may hide the clock-mediated effects. Most important, both light effects have the adaptive value of confining animals to their temporal niche (Redlin, 2001). Masking often complements the circadian clock in fine-tuning activity patterns in response to environmental stimuli. In D. melanogaster clock neurons, cryptochrome can integrate photons over time and effectively phase-shift the clock (Tang et al., 2010; Kistenpfennig et al., 2012; Vinayak et al., 2013; Guo et al., 2014). In contrast, the compound eyes mediate mainly masking effects of light, although they seem to be also involved in entrainment, especially under long photoperiods (Rieger et al., 2003; Helfrich-Förster, 2014).
Moonlight provokes clear masking effects as it stimulates nocturnal activity. This stimulation depends on rhodopsin 1 and rhodopsin 6 in the compound eyes (Schlichting et al., 2014), and it is completely independent of a functional clock (Kempinger et al., 2009). On the other hand, moonlight affects the phase in PER cycling of certain clock neurons, leading to an advance of the M peak and a delay of the E peak compared to LD conditions (Bachleitner et al., 2007). These phase changes are mediated by several rhodopsins in the outer and inner photoreceptor cells, but again rhodopsin 6 played a prominent role (Schlichting et al., 2014).
For twilight, the fraction of masking and clock-mediated effects on fly activity has not been evaluated yet. Simulated twilight (LDR) clearly affects the clock because it facilitates entrainment to long photoperiods (Rieger et al., 2012). In addition, the gradual increase/decrease of light may directly stimulate fly activity, leading to the sharp activity peaks at dawn and dusk. The fact that these sharp peaks take place at an irradiance of ~5 lux, which coincides with the irradiance at which flies are preferably active (Rieger et al., 2007; Kempinger et al., 2009), strongly suggest that the sharp peaks are masking effects. Here, we applied irradiances between 10 and 10,000 lux and found that the timing of the E peak delays with increasing irradiance. This finding is consistent with the results of Rieger et al. (2007) and with the idea that the sharp E peak always takes place at the same dim-light intensity, which occurs later during dusk at higher daylight intensities (since twilight duration was kept constant at 1.5 h).
Furthermore, LMR advances the E peak into dusk compared to LM conditions, where the E peak occurs in the night. This effect cannot be regarded as pure masking of twilight, because the E peak occurs significantly later under LMR than under LDR. Thus, the clock-dependent delay of moonlight on the E peak is also present.
Inner Photoreceptor Cells R7 and R8 Are Essential for the Response to Twilight, Whereas All Photoreceptor Cells Contribute to the Responses to Moonlight
Our results from the different photoreceptor mutants show that the inner photoreceptor cells R7 and R8 are essential for phase advancing the E peak in response to twilight. The phase-delaying effect of moonlight in the presence of twilight (LMR-LDR) was mainly dependent on rhodopsin 6, which fits perfectly to the results obtained from LM alone (Schlichting et al., 2014). The nocturnal activity stimulating (masking) effect of moonlight in the presence of twilight (LMR-LDR) was also mediated by several photoreceptors, but again rhodopsin 6—and, in this case, also rhodopsin 1—was important. Once more, this result fits to the effects of pure moonlight on nocturnal activity (Schlichting et al., 2014). Most important, sevLY3 mutants with a higher amount of rhodopsin 6 showed significantly more nocturnal activity in LMR than wild-type flies, which again points to the prominent role of rhodopsin 6.
Concluding Remarks
At present, we are not able to clearly distinguish masking effects from entraining effects of twilight and moonlight as well as the role of the different photoreceptor cells and rhodopsins in masking and entrainment. Nevertheless, we demonstrate a prominent role of the compound eyes and their rhodopsins in fine-tuning fly activity pattern to twilight and moonlight, with a special influence of rhodopsin 6. Future studies with clockless mutants will have to reveal whether the sharp activity peaks seen under simulated twilight are indeed pure masking effects of light. In addition, the universal phase-delaying effect of photoreceptors in the compound eyes on the E peak observed under all light regimes should be further investigated.
Footnotes
Acknowledgements
We thank Nina Vogt, Claude Desplan, Steve Britt, Erich Buchner, and Christopher Schnaitmann for providing fly lines and Steve Britt and Claude Desplan for providing antibodies. We are very grateful to Marta Beauchamp for language corrections. Further, we thank the Graduate School for Life Sciences Würzburg for supporting Rudi Grebler and Matthias Schlichting. Matthias Schlichting was further sponsored by a Hanns-Seidel-Foundation excellence grant funded by the BMBF (German Ministry for Education and Research). The study was funded by the Deutsche Forschungsgemeinschaft (DFG), collaborative research center SFB 1047 “Insect timing,” Project A1 and A2.
