Abstract
The human circadian system regulates internal 24-h rhythmicity and plays an important role in many aspects of human health and behavior. To investigate properties of the human circadian pacemaker such as intrinsic period and light sensitivity, experimental researchers have developed forced desynchrony (FD) protocols in which manipulations of the light-dark (LD) cycle are used to desynchronize the intrinsic circadian rhythm from the rest-activity cycle. FD protocols have typically been based on exposure to long LD cycles, but recently, ultradian FD protocols with short LD cycles have been proposed as a new methodology for assessing intrinsic circadian period. However, the effects of ultradian FD protocol design, including light intensity or study duration, on estimates of intrinsic circadian period have not, to our knowledge, been systematically studied. To address this gap, we applied a light-sensitive, dynamic mathematical model of the human circadian pacemaker to simulate ultradian FD protocols and analyze the effects of protocol design on estimates of intrinsic circadian period. We found that optimal estimates were obtained using protocols with low light intensities, at least 10 d of exposure to ultradian cycling, and a 7-h LD cycle duration that facilitated uniform light exposure across all circadian phases. Our results establish a theoretical framework for ultradian FD protocols that can be used to provide insights into data obtained under existing protocols and to optimize protocols for future experiments.
The human circadian pacemaker in the suprachiasmatic nucleus (SCN) maintains an ~24-h intrinsic rhythm, entrains to the exactly 24-h day based on light and other environmental inputs, and coordinates circadian rhythms throughout the body (Czeisler et al., 1999; Duffy and Wright, 2005). The intrinsic period (τ) has been estimated to range from 23.8 to 25 h in healthy, adult humans (Scheer et al., 2007), and τ may vary on the basis of sex, age, and race/ethnicity (Duffy and Czeisler, 2002; Smith et al., 2009; Duffy et al., 2011; Eastman et al., 2012). Although the difference in average τ between groups may be small (e.g., 0.2 h for adults v. adolescents; Carskadon et al., 1999; Czeisler et al., 1999), it may drive key differences in behavior and health. Variability in τ may underlie differential responses to normal, entrained conditions, such as phase angle of entrainment between sleep and dim light melatonin secretion, as well as to challenges to the circadian system such as travel across time zones. In addition, there is an increasing appreciation for the role that circadian rhythms play in many different aspects of health, including circadian rhythm sleep disorders, metabolism, cardiovascular function, and immune function (Campbell and Murphy, 2007, Sahar and Sassone-Corsi, 2009, Depner et al., 2014; Altman, 2016; Micic et al., 2016; Truong et al., 2016; Albrecht, 2017). Furthermore, interventions that consider and/or target the circadian system have been developed for treatment in acute situations such as intensive care units (Madrid-Navarro et al., 2015; Ritchie et al., 2015) as well as in everyday life. These interventions have included direct environmental modifications, such as changes to ambient lighting or light therapy; direct treatment modifications, such as timing drug administration to increase efficacy or minimize side effects (Levi et al., 2010); and policy changes that indirectly affect the circadian system, such as delaying school start times for adolescents (Carskadon et al., 1998; Owens et al., 2010). To optimally exploit circadian biology to improve health outcomes, improved understanding of the human circadian system in individuals, groups, and disease states is vital.
Typically, experiments to assess features of human circadian rhythms are very time and resource intensive, often requiring participants to stay in highly controlled laboratory conditions for extended periods of time. Because light is a major influence on the circadian clock, consideration of light conditions is essential for circadian research. To investigate properties of the human circadian clock such as intrinsic period (τ) and light sensitivity, experimental researchers have developed forced desynchrony (FD) protocols in which manipulations of the light-dark (LD) cycle are used to desynchronize the intrinsic circadian rhythm from the rest-activity cycle. To achieve this desynchronization, at least 4 different experimental paradigms have been described: temporal isolation, extended day FD, abbreviated day FD, and ultradian FD (Wyatt et al., 1999, Burgess and Eastman, 2008). In temporal isolation conditions, such as the early circadian experiments conducted in caves or bunkers (Aschoff, 1965; Wever, 1979), participants were isolated from external time cues, but they were able to self-select the timing of wake and sleep, thereby influencing the timing of their exposure to light. By contrast, under extended day, abbreviated day, or ultradian FD protocols, participants are scheduled to regular LD cycles with periods longer or shorter, respectively, than the range of entrainment of the intrinsic pacemaker (Czeisler et al., 1999; Wyatt et al., 1999; Scheer et al., 2007; Burgess and Eastman, 2008). Previous work has established that the human circadian pacemaker cannot entrain to LD cycles shorter than 21 h or longer than 26 h, although the exact relationship between the circadian pacemaker and its limits of entrainment depend on factors such as the intrinsic period of the oscillator and light intensity during light exposure (Klerman et al., 1996; Wright et al., 2001; Scheer et al., 2007).
Extended day FD protocols, typically involving a 28-h LD cycle, represent a well-established methodology for conducting controlled laboratory studies of the human circadian system (Dijk and Czeisler, 1994, 1995; Czeisler et al., 1999). These protocols have been applied to probe both the properties of the system and the underlying circadian rhythmicity of measures such as the propensity for sleep (Carskadon et al., 1999; Czeisler et al., 1999; Wright et al., 2001; Duffy and Czeisler, 2002; Khalsa et al., 2002; Wright et al., 2002; Scheer et al., 2007; Duffy et al., 2011). To optimize the design of extended day FD protocols, Klerman and colleagues (1996) applied a mathematical model of the human circadian pacemaker to simulate protocols under different experimental conditions including variable light intensities and study durations. Their results determined optimal conditions for estimating the intrinsic period using extended day FD protocols and established a theoretical framework for interpretation of data collected using these methods.
Desychronization of sleep and circadian rhythms can also be induced by exposure to LD cycles with periods shorter than 21 h. Historically, abbreviated-day protocols with 20-h LD cycles have been used (Wyatt et al., 1999; Koorengevel et al., 2002), but recently, investigators have developed ultradian FD protocols that use very short (typically 1-5 h) LD cycles (Kripke et al., 2005; Burgess and Eastman, 2008; Smith et al., 2009; Burgess et al., 2010; Eastman et al., 2012; Revell et al., 2012; Eastman et al., 2015; Micic et al., 2016). In addition to their utility for estimating intrinsic period, ultradian FD protocols with short LD cycles have also been used to dissociate circadian and sleep/wake–dependent features of electroencephalographic activity (Knoblauch et al., 2002; Munch et al., 2005). Ultradian FD protocols are less resource intensive and require less time in the lab compared with extended or abbreviated day FD protocols. However, to our knowledge, the effects of protocol design on experimental outcomes such as estimated intrinsic periods have not been formally quantified in ultradian FD protocols. For example, it is not known if estimates of intrinsic period obtained under ultradian FD protocols will depend on light intensity or study duration (Burgess and Eastman, 2008). Furthermore, some ultradian FD protocols, such as those with a 4-h LD cycle over several days, have not been designed to distribute sleep/wake behavior and/or light exposure equally across all circadian phases for pacemakers with τ near 24 h. Nonuniform light exposure may bias estimates of intrinsic period and make estimates obtained under these protocols more susceptible to protocol effects compared with those obtained using extended day FD protocols.
Although FD protocols could be optimized through systematic experimentation, this approach would be extremely resource-intensive (Czeisler et al., 1999). Alternatively, we used a light-sensitive, dynamic mathematical model of the human circadian pacemaker to simulate ultradian FD protocols and analyze protocol effects on estimates of intrinsic circadian period obtained from simulated data. We considered a range of light intensities, LD periods, and study durations to optimize protocol design for assessing physiologically relevant intrinsic circadian periods. Our results establish a theoretical framework for ultradian FD protocols that can be used to provide insights into data obtained under existing protocols and to optimize protocols for future experiments.
Methods
Human Circadian Pacemaker Model
To model the human circadian pacemaker, we implemented an established dynamic modeling formalism based on a modified van der Pol oscillator (Kronauer, 1990; Klerman et al., 1996). This model captures key attributes of the human circadian pacemaker: it is intrinsically oscillatory, and it can be entrained by a periodic light stimulus. The model has been calibrated to human data sets to appropriately represent the pacemaker’s dynamic response to light of varying durations and intensities as summarized by phase response curves (PRCs; Klerman et al., 1996). Furthermore, Klerman and colleagues (1996) applied this model to investigate features of extended day FD protocols.
The human circadian pacemaker model is governed by the following equations (Klerman et al., 1996):
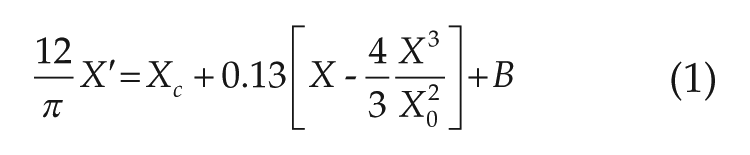
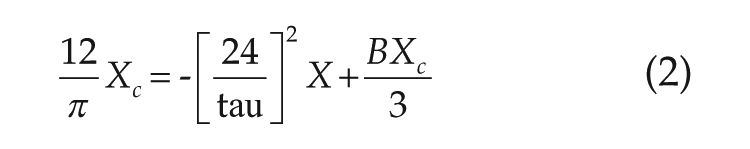
where
Model equations were implemented in MATLAB (Mathworks, Natick, MA) and solved numerically using the built-in MATLAB solver
Simulating Ultradian LD Protocols and Estimating Intrinsic Circadian Period
We systematically investigated the effects of LD period, light intensity, intrinsic period, and study length on the observed period, τobs, where τobs is the estimate of τ obtained from the simulated ultradian FD protocol. The structure of the simulated protocols was based on the structure of published ultradian LD protocols (Burgess and Eastman, 2008; Burgess et al., 2010) and included two parts: an at-home schedule to stabilize circadian rhythms prior to admission to the lab and an in-lab schedule consisting of an adaptation night, baseline night, phase assessment, and period of ultradian LD cycling (Fig. 1). All simulated protocols began with a 14-d at-home schedule in which light intensity was set to 0 lux from 2200 to 0800 h and 150 lux at all other times. The adaptation night and baseline night represent a participant’s transition to the in-lab setting. During this transition, the same timing of LD exposure was maintained, but light intensity during the light period was reduced to 30 lux. During the phase assessment, dim light conditions of 10 lux were imposed from 1800 to 0000 h and 0500 to 1200 h with a period of 0 lux from 0000 to 0500 h. Following the period of phase assessment, a schedule of 4-h LD cycling was initiated and continued for at least 33 d. To assess the sensitivity to phase of ultradian LD cycle onset, we simulated protocols with LD cycling initiated at the standard phase (at 1200 h following the phase assessment), 1 h before the standard phase, and 1 h after the standard phase.
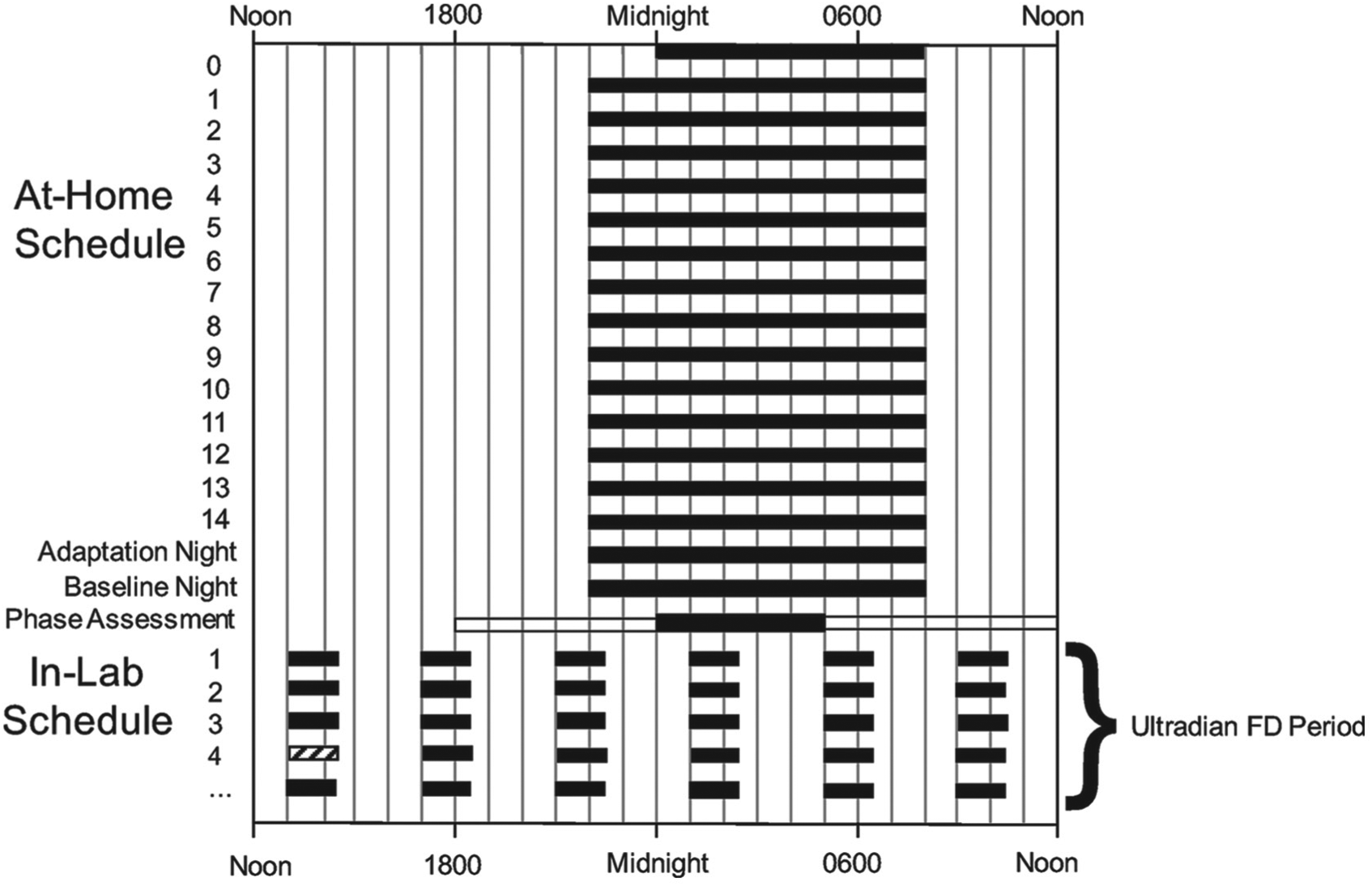
Comprehensive schematic for simulated ultradian forced desynchrony (FD) protocol with 4-h light-dark (LD) cycle. Protocol includes a 14-d at-home portion and an in-lab portion consisting of an adaptation night, a baseline night, a phase assessment, and then exposure to ultradian LD cycling. Black bars denote periods with a light intensity of 0 lux. Light intensities at all other times depend on the part of the protocol: the at-home schedule involves light conditions of 0 lux from 2200 to 0800 h and 150 lux at all other times, the adaptation night and baseline night involve light conditions of 0 lux from 2200 to 0800 and 30 lux at all other times, the phase assessment requires dim light of 10 lux from 1200 0000 h and 0500 to 1200 h with 0 lux from 0000 to 0500 h, and the ultradian FD period involves 4-h LD cycles with 2.5 h of light (variable light intensities depending on protocol) and 1.5 h of 0 lux. The hatched bar denotes the ultradian cycle after which estimates of circadian period are computed.
We simulated ultradian LD cycling involving 4-, 5-, or 7-h LD cycles. The proportion of light and dark in each cycle was held constant across protocols, resulting in 2.5 h L: 1.5 h D; 3.125 h L: 1.875 h D; and 4.375 h L: 2.625 h D for the 4-, 5-, and 7-h LD cycles, respectively. These LD cycle durations were chosen to represent published ultradian FD protocols (Burgess and Eastman, 2008; Burgess et al., 2010) as well as exploratory protocols that used relatively short LD periods, maintained the same proportion of light and dark as the 4h LD cycle, and involved cycle durations that did not evenly divide the 24-h period of the entrained oscillator. We chose to preserve the 2.5:1.5 light:dark ratio when we varied LD cycle duration because this light:dark ratio allowed proportions of active/inactive time similar to those experienced in a typical day. The number of ultradian cycles per 24-h day was determined by the duration of the LD cycle.
We simulated protocols with light intensities of 10, 50, 100, 150, 500, 750, and 1000 lux; intrinsic periods from 23.8 to 25.0 h; and study durations of 3 to 33 d. Light intensities represent a range typically encountered in daily life, and specific intensities were chosen for comparison to previous work (Klerman et al., 1996) and published ultradian FD protocols (Burgess and Eastman, 2008; Burgess et al., 2010). Intrinsic periods were selected to represent the physiological range of τ reported for adult humans (Scheer et al., 2007). Study duration describes the number of 24-h days of ultradian LD cycle exposure measured from 1200 to 1200 h following the phase assessment; study durations were chosen for comparison with published ultradian and extended day FD protocols (Klerman et al., 1996; Burgess and Eastman, 2008; Burgess et al., 2010). In addition, we simulated several protocols for study durations of 100 d to verify that increasing study duration beyond 33 d did not affect estimates of intrinsic circadian period. To determine the effect of constant light on the period of the circadian oscillator, we also simulated a theoretical, nonphysiologic protocol with constant 10 lux light exposure.
For each protocol, the observed circadian period, τobs, was estimated on day
Quantifying Estimates of τobs
To quantify the robustness of τobs across days, we fixed τ = 24.2 h and computed the regression line based on thirty-three 24-h days of ultradian cycling to determine τobs,33, a stable estimate of intrinsic circadian period. Then we compared the maximum, minimum, and average deviations of individual circadian minima on study days
To quantify the accuracy of τobs as a function of study duration, light intensity, and LD cycle duration, we systematically varied these protocol parameters, estimated τobs, and compared estimates to the known intrinsic period τ. To further examine the role of study duration on the accuracy of τobs, we defined a worst-case deviation of τobs,d from τ for τobs,d of study duration
To summarize the effect of LD cycle duration on the deviations of τobs from the intrinsic period τ and the sensitivity of τobs to the phase of ultradian LD cycle onset, we estimated τobs on study day 4 and study day 11 for intrinsic periods τ ranging from 23.5 to 25 h under 4-, 5-, or 7-h LD cycles with different phases of LD cycle onset. Study days 4 and 11 were selected to reflect a typical study duration of 3 d of ultradian LD exposure and a study duration that produces a stable value of τobs, respectively.
Assessing the Distribution of Light Exposure over the Circadian Cycle
For ultradian FD protocols with 4-, 5-, or 7-h LD cycles, we quantified the distribution of light exposure over the 24-h clock time that is associated with running ultradian cycling over 4 or 10 d of 24 h. We partitioned the 24-h day into 0.1-h bins and determined the light exposure for each bin with fixed light intensity
Results
To investigate the effects of protocol parameters on estimated circadian periods, we used a mathematical model of the human circadian pacemaker to simulate ultradian FD protocols with varying LD cycles, light intensities, and study durations. For each simulated protocol, the timing of the minimum of the simulated circadian oscillation was determined, and the progression of the minimum over each 24-h cycle indicated that the oscillator was unable to entrain to any of the ultradian cycles. For τ = 24.2 h, the timing of the minimum of the simulated circadian oscillation showed the expected rightward progression under a protocol involving 4- (Fig. 2), 5- (Suppl. Fig. 1), or 7-h (Suppl. Fig. 2) LD cycles. We computed τobs to be the slope of the linear regression on the minima as described in the Methods section.
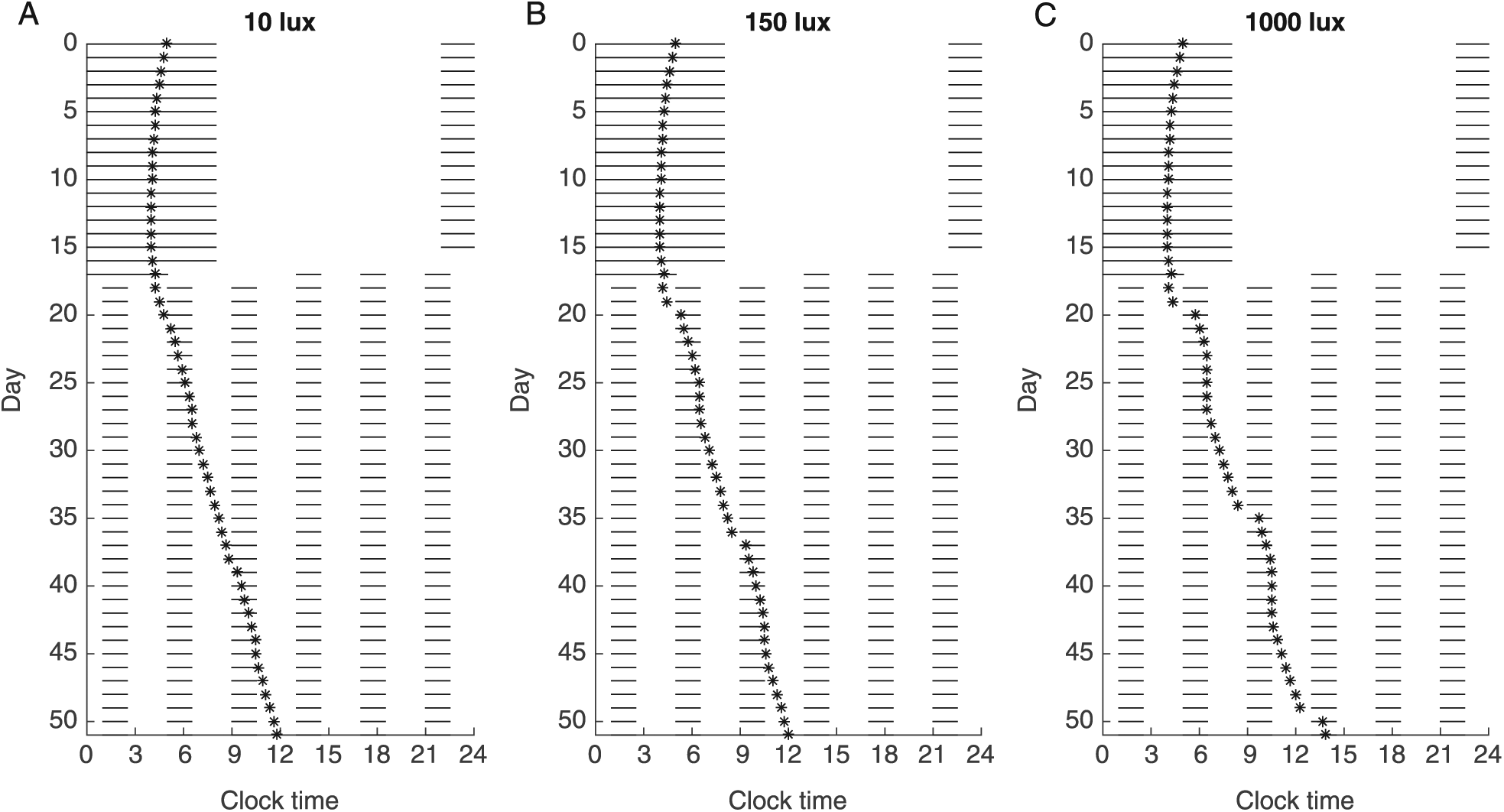
Minimums of
Accuracy of τobs Depends on Light Intensity and Study Duration
For all protocols, τobs,33 provided a stable estimate of τ and deviated less than 0.05 h = 3 min from the known τ. The daily progression of the minimum of
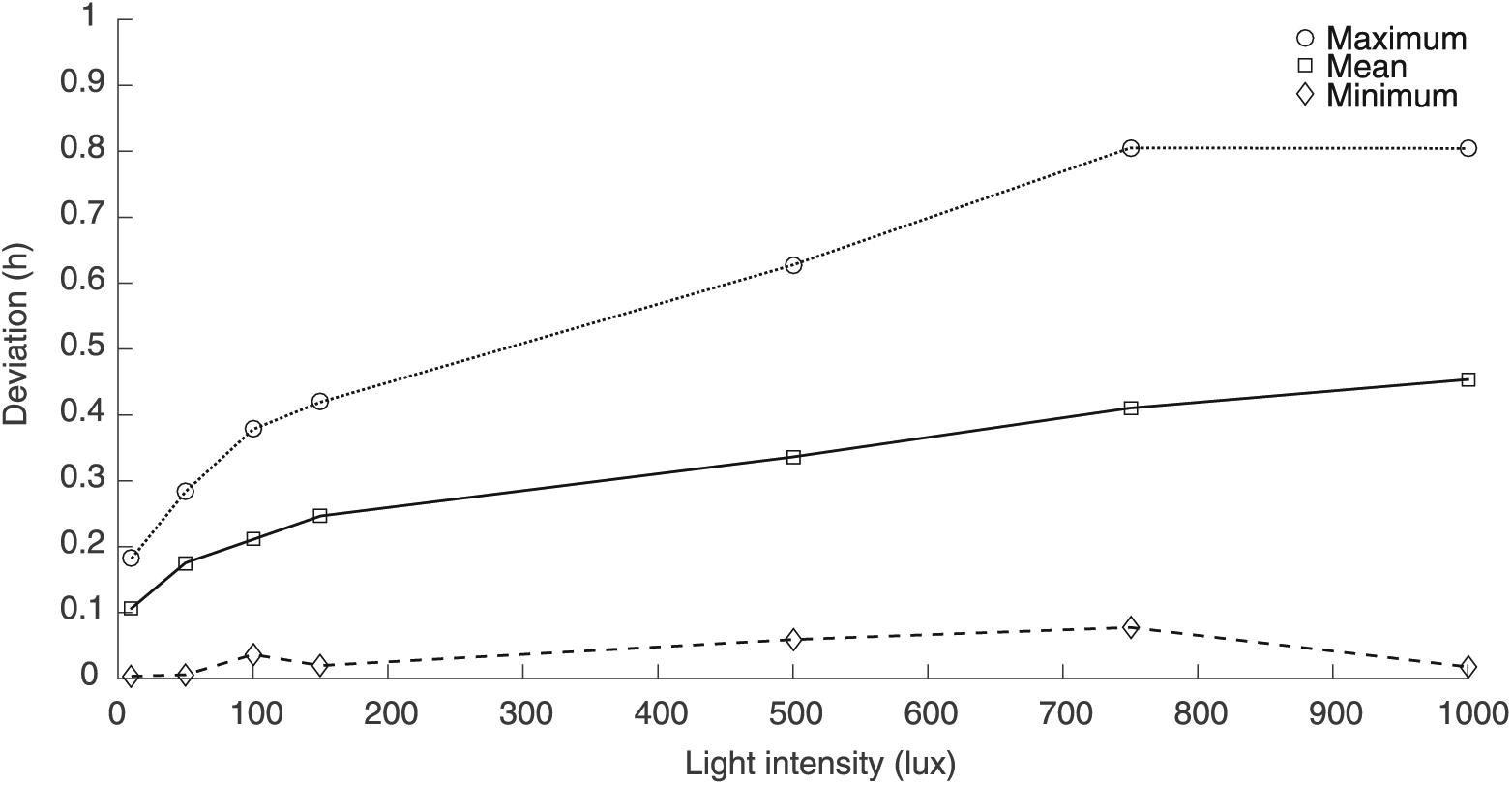
Robustness of τobs was reduced at high light intensities. The deviations of the minimums of
To investigate the effect of protocol duration and light intensity on τobs, we simulated protocols with variable numbers of study days and light intensities with τ ranging from 23.8 to 25 h in the circadian pacemaker model. We report results for τ = 23.8, 24.2, 24.6, and 25.0 h. We found that estimates of intrinsic period under ultradian FD protocols with 4- (Fig. 4), 5- (Suppl. Fig. S5), and 7-h (Suppl. Fig. S6) LD cycles depended on the intrinsic period τ, light intensity
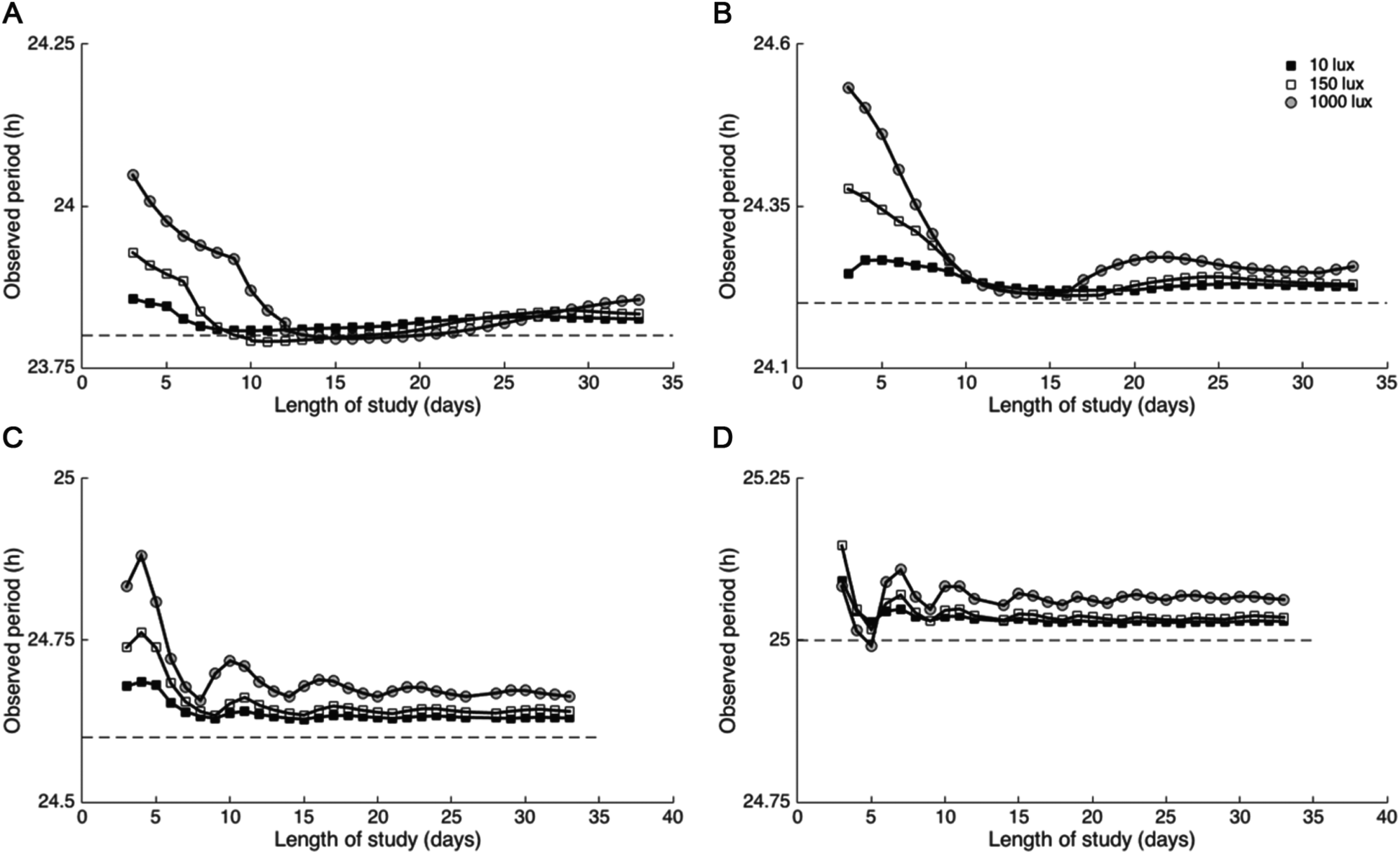
Estimates of intrinsic period improve with longer studies and lower light levels independent of intrinsic period τ. For τ = 23.8 (A), 24.2 (B), 24.6 (C), and 25.0 h (D), higher light intensities were associated with greater deviations of τobs from τ. For all simulations, τobs generally converged within a neighborhood of the intrinsic period with sufficiently long exposure to 4-h light-dark cycling with light intensities of 10, 150, or 1000 lux.
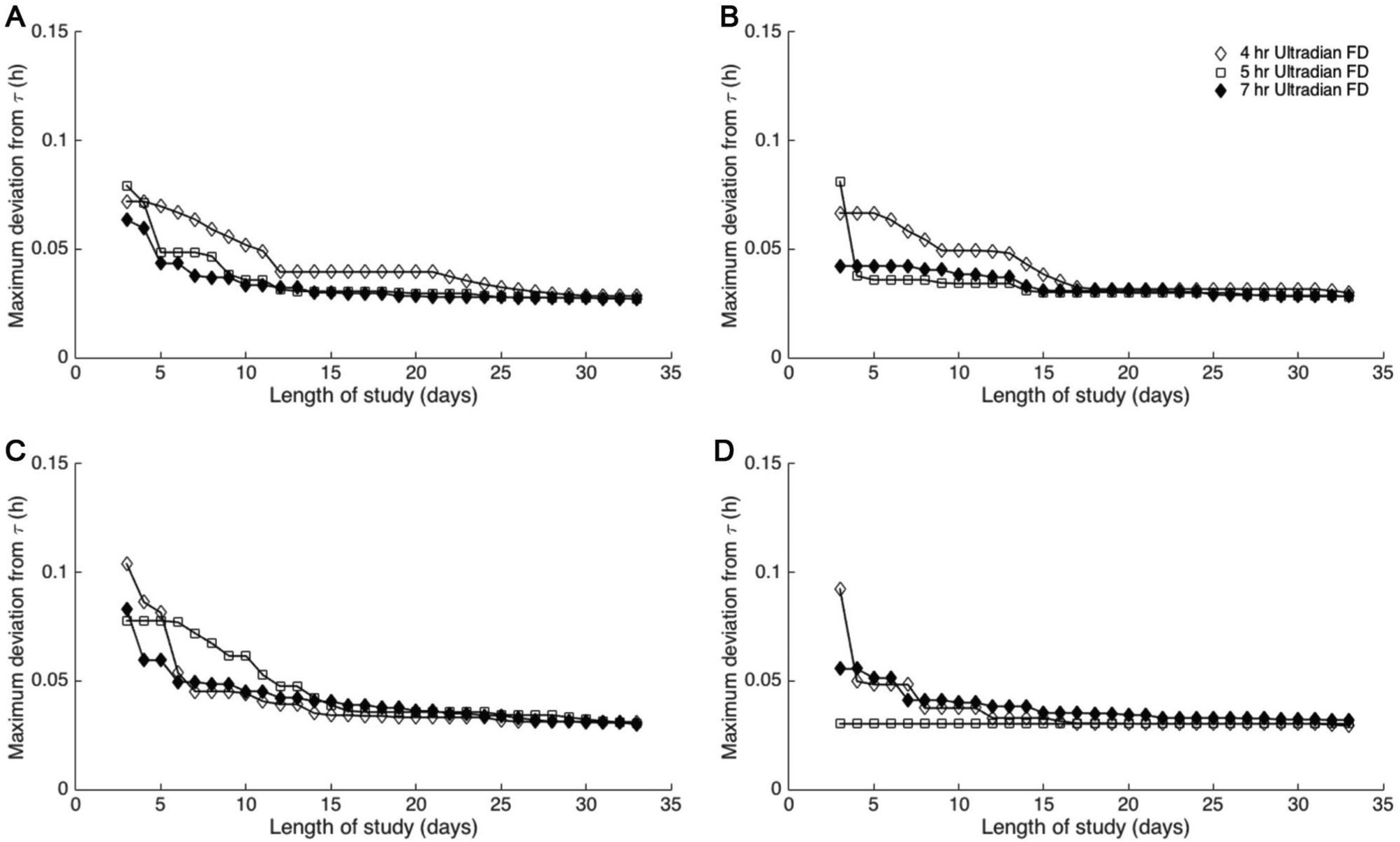
Circadian periods estimated using ultradian forced desynchrony protocols with 4-, 5-, and 7-h light-dark cycles and a light intensity of 10 lux converge to the intrinsic period. For τ = 23.8 (A), 24.2 (B), 24.6 (C), and 25.0 h (D), the maximum deviation of τobs computed on day
Based on this evidence that τobs,10 stably estimated τ, we quantified the robustness of τobs,10 relative to τobs,d for 11 ≤
To assess effects of constant light on estimated circadian period, we simulated a protocol of constant 0 and 10 lux light conditions for τ = 24.2 h. We found minimal effect on the estimated period with τobs, 33 = 24.2314 h at 0 lux and τobs, 33 = 24.2346 h at 10 lux. This validates the use of the parameter τ as the intrinsic circadian period of the model.
Accuracy of τobs Depends on LD Cycle Duration and Resulting Pattern of Light Exposure
To investigate the dependence of the observed circadian period on LD cycle duration, we compared estimates of τobs,3 and τobs,10 computed for ultradian FD protocols with 4-, 5-, or 7-h LD cycle durations for τ ranging from 23.8 to 25 h. In addition, we considered the sensitivity of τobs to the phase of LD cycle onset by simulating the protocols with the onset of the ultradian LD cycles offset by 1-h shifts. We found that the deviation of observed period from τ varied with the phase of protocol onset for all protocols, and relative differences depended on τ (Fig. 6). For 4-, 5-, and 7-h LD cycles, τobs,3 and τobs,10 generally overestimated τ independent of phase of protocol onset, although there were values of τ for which τobs,3 provided underestimates. This phase dependence was attenuated for τobs,10 compared with τobs,3. However, variability for τobs,10 was greatest for τ near 24 h for protocols with 4-h LD cycles and greatest for τ near 25 h for protocols with 5-h LD cycles. In addition, variability of τobs,3 and τobs,10 with phase of LD cycle onset was minimized for protocols with 7-h LD cycles compared with 4- or 5-h LD cycles across intrinsic periods. For τobs,10, the deviation of observed period from intrinsic period was less than 0.1 h for all protocols and all values of τ; this deviation was less than 0.055 h for the ultradian FD protocol with 7-h LD cycle durations.
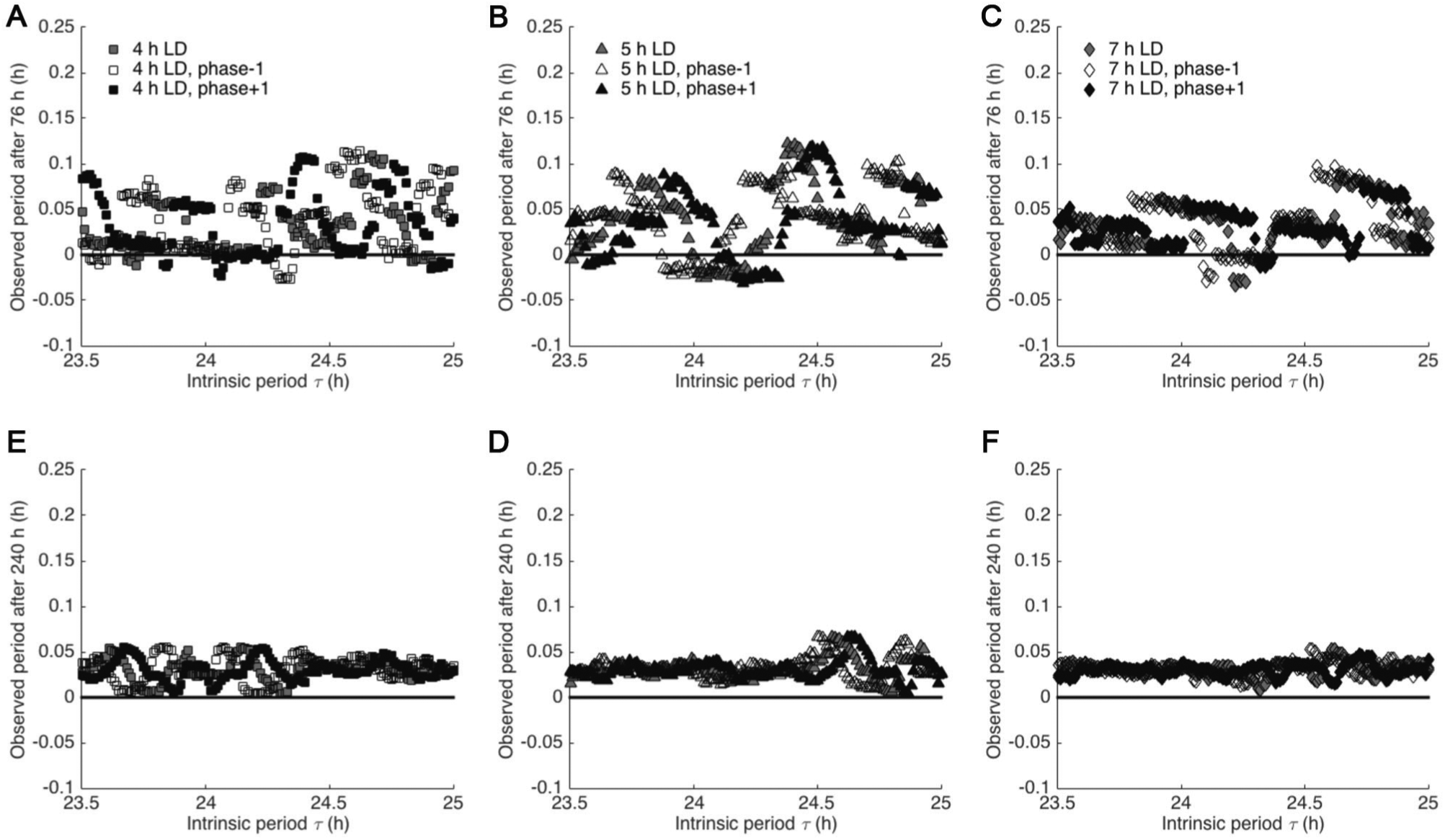
Circadian periods estimated using ultradian forced desynchrony protocols with 4-, 5-, and 7-h light-dark (LD) cycles depended on phase of protocol onset and generally overestimated true circadian period. Shifting the phase of ultradian cycling onset to begin 1 h earlier or later than the baseline phase of protocol onset revealed that protocols with LD cycles of 4 (A, D) or 5 h (B, E) were more sensitive to the phase of protocol onset compared with the protocol with 7-h LD cycles (C, F). Both the phase dependence and the variability in τobs were decreased with a study duration of 11 d (D, E, F) compared with estimates with a study duration of 4 d (A, B, C).
We hypothesized that the decreased sensitivity to onset phase in the ultradian protocol with 7-h LD cycles compared with protocols with 4- or 5-h LD cycles was caused by protocol-associated differences in the timing of light exposure. To test this hypothesis, we computed light exposure at each clock time across the 24-h day for each protocol (Fig. 7). Although light exposure across circadian phase will vary with τ, computing light exposure as a function of clock time approximates the distribution of light exposure for values of τ near 24 h, particularly for short study durations. We found that patterns of light exposure associated with 4-h LD cycles were less uniform than those associated with 7-h LD cycles, and this difference was more pronounced in the 4-d protocol compared with the 10-d protocol.
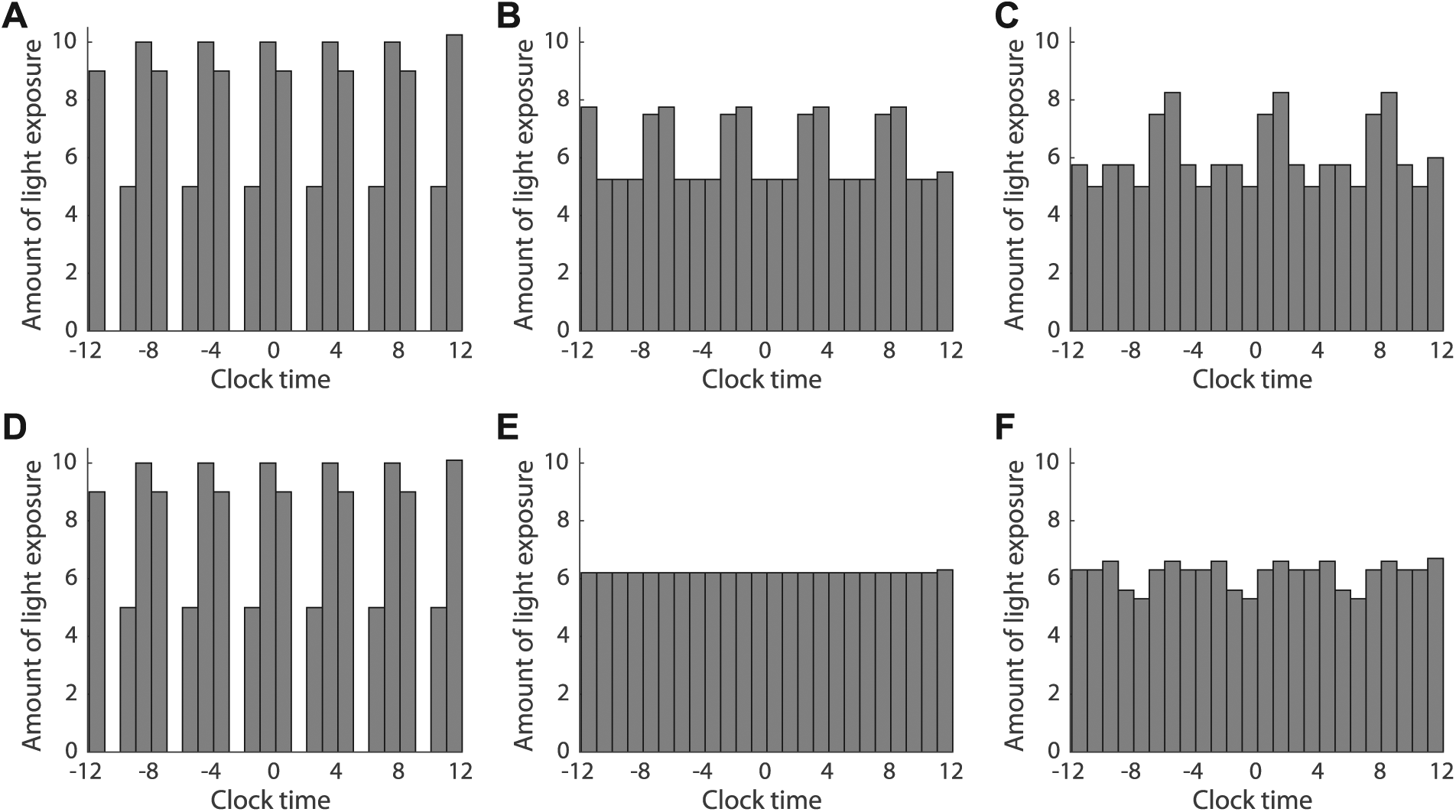
Relative light exposure varies for ultradian forced desynchrony protocols with light-dark (LD) cycles of different durations. Histogram of relative light exposure for an ultradian protocol with 4- (A, D) or 5-h (B, E) LD cycles shows less uniform light exposure compared with protocols with 7-h LD cycles (C, F). Variability in light exposure was decreased with a study duration of 10 d (D, E, F) compared with variability observed in a study duration of 4 d (A, B, C).
Model Dependence Observed in τobs but Not in Protocol Design Features
Because simulation results depend on the choice of human circadian pacemaker model applied, we duplicated all simulations using an alternative pacemaker model with a light response that implements Aschoff’s rule to investigate the potential model dependence of our results. We found that the conclusions regarding ultradian FD protocol design were consistent with simulation results from both models. Specifically, accuracy of τobs was maximized for low (e.g., 10 lux) light intensities, study durations of at least 10 d, and LD cycle durations of 7 h (Suppl. Fig. S7 and S8). Computing τobs using the alternative pacemaker model typically resulted in underestimates of τ independent of phase of protocol onset (Suppl. Fig. S8). Furthermore, the deviations of τobs,10 from τ were greater using the alternative model compared with the original model. Consistent with Aschoff’s rule, simulating a protocol of constant 10 lux light conditions with the alternative model resulted in a decreased estimated period compared with the estimated period associated with 0 lux conditions: for τ = 24.2 h, τobs,33 = 24.1975 h at 0 lux and τobs,33 = 24.1176 h at 10 lux. This complicates the interpretation of deviations from τ in the alternative model.
Discussion
Summary and Interpretation of Results
Intrinsic circadian period, τ, represents a key characteristic of the human circadian system, and current extended or abbreviated day FD experimental protocols used to estimate τ are highly resource intensive. Ultradian FD protocols offer a potential alternative, but less is known about the optimal design of these protocols for obtaining accurate estimates of τ. Using a light-sensitive, dynamic model of the human circadian pacemaker, we simulated ultradian FD protocols and analyzed the effects of light intensities, study durations, LD cycle durations, and intrinsic period on estimated intrinsic circadian period, τobs.
We found that optimal estimates were obtained for protocols with low light intensities (e.g., 10 lux), study durations of at least 10 d, and a 7-h LD cycle duration. For all LD cycle durations tested, ultradian FD protocols produced stable estimates of τobs within 0.07 h of τ with at least 10 d of exposure to ultradian LD cycling, although the rate of convergence of τobs to τ varied for different intrinsic periods and LD cycle durations. Based on the convergence of τobs, our results suggest that extending an ultradian FD study beyond 10 d is unlikely to improve the accuracy of τobs. Across values of τ, estimates of observed period were improved, and the sensitivity to the phase of protocol onset was reduced for protocols using 7-h LD cycle durations compared with 4- or 5-h LD cycle durations. These results provide a theoretical framework for designing future ultradian FD protocols and interpreting data collected under current protocols.
In the lab, intrinsic circadian period is assessed using markers of circadian phase such as core body temperature and salivary or plasma melatonin (Duffy and Dijk, 2002; Wright et al., 2008). When core body temperature is used, it is measured throughout the protocol and fit to a sinusoidal function to determine period (e.g., see Czeisler et al., 1999). When melatonin is used, dim light melatonin onset, determined as an absolute threshold or as 25% of maximum melatonin levels, is computed before and after the protocol (Wright et al., 2008), and intrinsic period is estimated by normalizing the observed shift in phase by the number of days of the protocol (e.g., see Burgess and Eastman, 2008). Although this methodology has been validated, contributions of measurement error, as well as theoretical limitations on accuracy, must be considered when working with experimental data.
Implementation of ultradian FD protocols has varied across labs. Our results suggest that the accuracy of intrinsic period estimates determined using published ultradian FD protocols may vary as a function of τ, study duration, phase of protocol onset, and light intensity. Because the ability to control for factors such as τ and phase of protocol onset is limited, ultradian FD protocols must be designed to provide reliable estimates despite variability in these factors. Although published ultradian FD protocols have typically involved low light intensities, other protocol features, such as the common use of 4-h LD cycles and study durations of 3 to 4 d, are not consistent with predicted optimal protocol design. Given the potential constraints on the accuracy of τobs, caution is warranted when interpreting estimates of intrinsic periods obtained using FD protocols that do not conform to predicted best practices. However, researchers may determine acceptable levels of accuracy of τobs in the context of specific experiments and use these theoretical guidelines to design ultradian FD protocols accordingly.
Designing Ultradian FD Protocols to Produce Uniform Light Exposure
A key feature of extended day FD protocols is a pattern of light scheduling that results in uniform light exposure over all circadian phases. For sufficiently long protocols, this scheduling ensures uniform sampling of the circadian pacemaker’s PRC to light, and it minimizes the biased PRC sampling that occurs under free-running protocols in which participants are able to self-select light exposure. For the model (with τ = 24.2 h), a PRC representing the response to a simulated 5-h pulse of light of 150 lux has been reported and validated against human data (Klerman et al., 1996). The distinct advance and delay regions of the PRC suggest that nonuniform light exposure will result in biased sampling of the PRC, thereby affecting the estimate of observed period.
Furthermore, when PRC sampling is biased, the phases at which increased light exposure occurs will depend on the phase at which the protocol is initiated, thereby increasing the sensitivity of τobs to protocol effects. For example, for the same individual under the same protocol with a 4-h LD cycle, slight variations in internal circadian phase at protocol onset may produce different estimated τobs values because of shifted patterns of light exposure acting differentially on the PRC to light. Such sensitivity to the phase of protocol onset could also account for increased variability in τobs across a study cohort consistent with observations in our data. Sensitivity to phase of protocol onset is minimized in ultradian FD protocols with 7-h LD cycles and sufficiently long study durations.
Although sufficiently long ultradian FD protocols with 4-h LD cycles would ensure uniform light distribution for τ ≠ 24 h, the number of days required to achieve uniform light exposure increases as τ approaches 24 h. In practice, for τ near 24 h, a 4-h LD cycle results in a pattern of light exposure that is not uniform over the entire circadian cycle, particularly when study durations are short. Given evidence that τ is near 24.2 h for many adult humans, this is an important methodological consideration (Czeisler et al., 1999). For example, for τ =24.2 h, a 3-d protocol with a 4-h LD cycle would result in a drift of approximately 0.6 h = 36 min. Assuming a protocol onset at 1200 h and 2.5 h of light per cycle, the circadian phases associated with the hour from approximately 1230 to 1430 h would receive constant light, and the circadian phases associated with the hour from approximately 1500 h to 1600 h would receive no light throughout the protocol. For τ near 25 h, similar limitations are associated with an ultradian FD protocol with a 5-h LD cycle. The interaction of 4- or 5-h LD cycles with τ is consistent with our results demonstrating that LD cycle durations of 7 h provide improved estimates of τobs compared with 4- or 5-h LD cycle durations across the physiologic range in τ values.
It is important to note that the biased pattern of light exposure associated with the 4-h LD cycle over the 24-h day for τ near 24 h would also occur with an LD cycle of any duration that evenly divides 24 h (e.g., 2-, 3-, 6-, 8-, or 12-h LD cycles). Preliminary evidence suggests that this issue is minimized with very short (e.g., 60 min) LD cycles. This likely reflects decreased magnitudes of advances and delays caused by shorter light exposures (Duffy and Wright, 2005) as well as increased sensitivity of short-bout durations to deviations of τ from 24 h. Design of future ultradian FD protocols should specify LD cycle durations and study durations that ensure a uniform distribution of light across all circadian phases.
Comparison with Extended Day FD Protocols
In previous work, Klerman and colleagues (1996) used this circadian pacemaker model to simulate extended day FD protocols and the effects of protocol design on estimates of intrinsic circadian period. They found that observed period depended on light intensity, LD period, and intrinsic period. Their results suggested that estimates of τ using 28-h FD protocols initially overestimated τ; τobs approached τ when the protocol included at least 3 beat periods (approximately twenty-five 24-h days) and used a light intensity that did not exceed 10 lux. They did not report specific magnitudes of the deviations they observed, although they indicated that the deviation of the observed period from the intrinsic period was considerable at high light intensities, and several specific examples were included.
Consistent with their conclusions, we found that under ultradian FD protocols, τobs typically overestimated τ if study durations were not sufficiently long, and optimal results were obtained under low light intensities. Our results also demonstrated that initial estimates of τobs using ultradian FD protocols were closer to actual intrinsic periods and converged more quickly to intrinsic periods compared with estimates of τobs using extended day FD protocols. In addition, the robustness of estimates of τobs, measured as deviations of individual circadian minima from the regression line, was improved in the ultradian FD protocol compared with the extended day FD protocol. These differences highlight an advantage of ultradian FD protocols: the use of shorter LD cycles reduces the perturbation experienced by the circadian system under the FD protocol. Because this perturbation is particularly pronounced for 28-h FD protocols with short study durations due to highly nonuniform light exposure, much longer study lengths are required to obtain accurate estimates of τobs with 28-h protocols compared with ultradian FD protocols.
Model Dependence in Protocol Effects
A primary limitation of this study is the constraint imposed by the assumptions inherent in the circadian pacemaker model. To facilitate comparison with previous work, this study applied the same human circadian pacemaker model that was used to evaluate protocol effects in extended day FD protocols (Klerman et al., 1996). However, other models of the human circadian pacemaker have been proposed (Kronauer et al., 1982; Achermann and Kunz, 1999; Forger et al., 1999; Jewett et al., 1999), and differences among models may affect simulation outcomes. Most significantly, differences in the model’s response to light, including the PRCs to light of different durations and intensities, may produce different predicted relationships between estimated period and intrinsic period under similar experimental conditions (Kronauer et al., 1982).
To address the issue of model dependence, we duplicated all simulations using an alternative mathematical model of the human circadian pacemaker (Forger et al., 1999). We found that simulations produced by the alternative model supported our previous conclusions regarding light intensity, study duration, and LD cycle duration for optimal design of ultradian FD protocols for estimating intrinsic circadian period. However, our results also suggested that, particularly under conditions in which τobs had not yet converged to τ, individual estimates of τobs demonstrated model dependence. Furthermore, simulations of the original and alternative models resulted in respective over- or underestimation of τ by τobs. The observed model dependence in estimates of τobs is probably caused by differences in light processing between models. In particular, the model proposed by Forger and colleagues (1999) includes an implementation of Aschoff’s rule so intrinsic circadian periods are shorter at higher light intensities (Aschoff, 1960). By simulating a protocol of constant dim light (10 lux), we confirmed that the alternative model demonstrated a light intensity dependence of circadian period that was not present in the original model. The implementation of Aschoff’s rule in the alternative model complicates the interpretation of the deviation of τobs from τ because τ varies as a function of light exposure.
Although preliminary investigations of model dependence did not alter our conclusions regarding the optimal design of ultradian FD protocols for estimating intrinsic circadian period, the evidence for model dependence in individual estimates of τobs suggests that more work is needed to develop robust methods to relate estimated intrinsic circadian period to actual circadian period based on an individual’s response to an ultradian FD protocol. Such methods would have great utility for refining interpretation of existing data sets and enabling comparisons among data collected under different protocols. In addition, circadian pacemaker models do not include explicit interactions with sleep/wake behavior, but sleep state may affect experimental attempts to measure τ. Future work with models that incorporate both sleep and circadian factors may provide additional insight into these interactions (Phillips et al., 2010; Gleit et al., 2013).
Conclusions and Implications
In summary, ultradian FD protocols represent a promising experimental paradigm for addressing key questions in circadian research. In particular, the relative resource- and time-effective features of ultradian FD protocols underscore their utility for studies that require larger numbers of participants or address research questions in pediatric or other populations in which compliance with the time requirements of an extended day FD protocol are particularly challenging. Simulations of a human circadian pacemaker model support the use of well-designed ultradian FD protocols for estimating intrinsic circadian period. In addition, our results highlight the utility of mathematical modeling for optimizing circadian protocols and establish a theoretical framework that provides insights into data obtained under existing protocols and enables design of optimal protocols for future experiments.
Footnotes
Acknowledgements
This study was supported by the National Science Foundation grant DMS 1412571 (to C.D.B.) and National Institute of Mental Health grant MH076969 (M.A.C.).
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
