Abstract
From the perspective of circadian biology, mammalian pregnancy presents an unusual biological scenario in which an entire circadian system (i.e., that of the fetus) is embodied within another (i.e., that of the mother). Moreover, both systems are likely to be influenced at their interface by a third player, the placenta. Successful pregnancy requires major adaptations in maternal physiology, many of which involve circadian changes that support the high metabolic demands of the growing fetus. A functional role for maternal circadian adaptations is implied by the effects of circadian disruption, which result in pregnancy complications including higher risks for miscarriage, preterm labor, and low birth weight. Various aspects of fetal physiology lead to circadian variation, at least in late gestation, but it remains unclear what drives this rhythmicity. It likely involves contributions from the maternal environment and possibly from the placenta and the developing intrinsic molecular clocks within fetal tissues. The role of the placenta is of particular significance because it serves not only to relay signals about the external environment (via the mother) but may also exhibit its own circadian rhythmicity. This review considers how the fetus may be influenced by dynamic circadian signals from the mother and the placenta during gestation, and how, in the face of these changing influences, a new fetal circadian system emerges. Particular emphasis is placed on the role of endocrine signals, most notably melatonin and glucocorticoids, as mediators of maternal-fetal circadian interactions, and on the expression of the clock gene in the 3 compartments. Further study is required to understand how the mother, placenta, and fetus interact across pregnancy to optimize circadian adaptations that support adequate growth and development of the fetus and its transition to postnatal life in a circadian environment.
Introduction
The circadian biology of pregnancy is complex; the mother, the placenta and the fetus may be viewed as distinct circadian entities that interact dynamically across gestation. Major adaptations occur in maternal circadian physiology to support the high metabolic demands of the growing fetus, while, at the same time, the fetus develops its own intrinsic circadian system in readiness for postnatal life (Seron-Ferre et al., 2012; Waddell et al., 2012). At the interface between mother and fetus, the placenta regulates the interaction between maternal and fetal circadian systems and exhibits its own rhythmic function (Wharfe et al., 2011; Waddell et al., 2012). In this review, we consider how the developing fetus is influenced by circadian signals from the mother and placenta throughout gestation, and how, in the face of these changing influences, a new fetal circadian system emerges. Emphasis is placed on the role of endocrine signals as mediators of maternal-fetal circadian interactions, and on the expression of clock genes in key metabolic tissues in the 3 compartments.
The circadian timing system comprises a central clock located in the suprachiasmatic nucleus (SCN) of the hypothalamus, which can influence peripheral clocks located in virtually every tissue (Reppert and Weaver, 2002). The molecular machinery of these clocks comprises a network of ‘clock’ genes, namely Clock, brain and muscle Arnt-like protein (Bmal1), Period (Per1-3) and Cryptochrome (Cry1 and Cry2), which regulate their own expression through a sequence of positive and negative transcriptional-translational feedback loops (Reppert and Weaver, 2002). The positive arm of this oscillatory loop is driven by CLOCK:BMAL1-induced transcription of the Per and Cry genes via binding to their E-box (5’-CACGTG-3’) and E-box-like promoter sequences. PER and CRY proteins dimerize and the resultant PER:CRY complexes translocate to the nucleus and inhibit CLOCK:BMAL1 transcriptional activity, thereby acting as a negative regulator of Per and Cry transcription. An accessory arm adds stability to this system, wherein the CLOCK:BMAL1 dimer also induces the transcription of 2 nuclear receptors, namely reverse erythroblastosis virus α (Reverbα) and retinoic acid receptor-related orphan receptor α (Rorα). Reverbα inhibits Bmal1 transcription via binding to ROR response elements (RORE) on its promotor (Preitner et al., 2002), whereas Rorα aids the positive arm of the system by competing with Reverbα for RORE binding and thus stimulates Bmal1 transcription (Sato et al., 2004). Reverbα also represses Clock transcription (Preitner et al., 2002; Crumbley and Burris, 2011), whereas Rorα does not appear to directly influence Clock (Crumbley and Burris, 2011).
The central SCN clock is entrained to the light-dark cycle via melanopsin-containing, light-sensitive ganglion cells in the retina (Lowrey and Takahashi, 2000) and synchronizes a range of peripheral clocks (e.g., liver, adrenal, adipose) via endocrine, autonomic, and behavioral cues (Hastings et al., 2007; Dickmeis, 2009). The molecular clock network exerts control over specific biological functions by regulating the expression of clock-controlled genes and thus downstream metabolic and physiological pathways (Bozek et al., 2009). Depending on cell type, it is estimated that between 5% and 20% of the transcriptome exhibits circadian rhythmicity (Froy, 2010).
Maternal Adaptation to Pregnancy
Pregnancy is arguably one of the most physiologically challenging states that a mammal encounters across its life cycle. During pregnancy, the mother undergoes a vast array of physiological changes to enable her to meet the requirements of her developing fetus, whilst also preparing for lactation and maintaining her own tissue function. Accordingly, the mother must accommodate the growth and development of a functional placenta, as well as drive widespread adaptations in most of her organ systems. These adaptations extend from marked central modifications in brain function to fundamental changes in respiratory, cardiovascular, and metabolic function. Pregnancy may be considered to have two distinct metabolic phases characterized by anabolism and catabolism, respectively (Hamosh et al., 1970; Knopp et al., 1973). The anabolic phase spans the initial two- thirds of gestation: the first and second trimesters in humans and up to days 10 to 12 in rodents (Knopp et al., 1973; Metcalfe et al., 1988). During the anabolic phase, maternal hyperphagia (Weizenbaum et al., 1979) and enhanced insulin sensitivity facilitate lipid accumulation in adipose tissue. Later in gestation, however, there is a shift to a more catabolic state, which allows for the breakdown of maternal fat stores to provide substrates for fetal growth (Herrera et al., 1988; Palacin et al., 1991; Lain and Catalano, 2007; Herrera and Ortega-Senovilla, 2010). This catabolic phase is facilitated by a late-gestational increase in maternal insulin resistance (Knopp et al., 1970) in conjunction with the action of placental lactogen, progesterone, and glucocorticoids, which induce lipolytic effects in adipose tissue (Williams and Coltart, 1978; Sutter-Dub et al., 1981; Xu et al., 2009). Since glucose is also the major fetal substrate, maternal hepatic glucose production increases in late gestation (Catalano et al., 1992). Despite this increase, circulating maternal glucose decreases towards term (Catalano et al., 1991) due to its rapid uptake by the placenta (Hay, 2006). Recent studies indicate that alterations in maternal lipid and glucose metabolism during pregnancy may be facilitated by the circadian clock, since clock gene expression in the maternal liver and adipose tissue changes substantially across mouse pregnancy (Wharfe et al., 2016b; Wharfe et al., 2016c; see below for details). Moreover, these clock gene changes correspond to gestational adjustments to the rhythmicity of genes regulating lipogenesis and gluconeogenesis in these tissues (Wharfe et al., 2016b; Wharfe et al., 2016c).
The importance of a functional circadian system during pregnancy is further emphasized by evidence that circadian disruption results in numerous pregnancy complications. For example, shift work (particularly overnight) has been associated with a higher risk of miscarriage, pre-term labor, and low birth weight (McDonald et al., 1988; Knutsson, 2003; Zhu et al., 2004). Furthermore, rodent models using altered light cycles to replicate the effects of shift work support this human evidence; pregnant rats exposed to constant light exhibit marked reductions in fetal weight in conjunction with placental abnormalities (Gozeri et al., 2008), and disruptions to maternal melatonin rhythmicity and fetal adrenal development (Mendez et al., 2012). Interestingly, restoration of maternal melatonin rhythmicity via subjective night-time melatonin supplementation completely rescued the fetal growth deficiency associated with constant light exposure (Mendez et al., 2012). Furthermore, pregnant mice exposed to a 6-h phase advance or delay every 5 to 6 days throughout gestation displayed a pronounced decline in pregnancy success rates (Summa et al., 2012), whereas exposure to a 12-h phase shift every 3 to 4 days throughout rat pregnancy resulted in metabolic aberrations across maternal and fetal compartments, including disruptions to rhythmic hormone profiles and altered hepatic expression of clock and metabolic genes (Varcoe et al., 2013). The offspring of mothers exposed to inverted light cycles also experienced programmed disturbances in insulin homeostasis in adulthood, despite being raised in normal lighting conditions themselves (Varcoe et al., 2011); these offspring also showed altered rhythmicity in plasma melatonin, corticosterone, and aldosterone, as well as changes in blood pressure rhythmicity (Mendez et al., 2016). Collectively, these studies suggest that circadian disruption during pregnancy may program disturbances in offspring circadian rhythmicity.
Maternal Circadian Biology in Pregnancy
Endocrine Adaptations
Maternal endocrine function is altered substantially during gestation, with marked changes in ovarian and placental hormone synthesis that promote adaptive changes in maternal physiology. The extent to which these endocrine changes per se and their downstream physiological effects in the mother involve adaptations in circadian biology remains poorly understood. Among the endocrine signals that change during pregnancy, shifts in melatonin and glucocorticoid secretion are likely to be particularly important in relation to the circadian adaptations.
Melatonin
The nocturnally secreted neurohormone melatonin is synthesized by conversion of serotonin in the pineal gland (Kennaway and Wright, 2002). Pineal melatonin synthesis is driven by neuronal outputs from the SCN and is inhibited by light; as such, melatonin exhibits a distinct daily secretion pattern, with peak levels occurring nocturnally in both diurnal and nocturnal species (Pevet and Challet, 2011). During normal human pregnancy, peak maternal melatonin levels increase progressively from 24 weeks of gestation until term, rising ~4-fold over this period and then fall to non-pregnant levels during the post-partum period. Daytime levels of melatonin do not increase during the same period, and thus there is an increased amplitude of melatonin rhythmicity as gestation progresses (Nakamura et al., 2001). Peak maternal melatonin in rats increases ~3-fold from day 7 of gestation to term and then drops to non-pregnant levels (Tamura et al., 2008). Importantly, melatonin is not synthesized by the fetal pineal gland (Deguchi, 1975; Yuwiler et al., 1977; Yellon and Longo, 1988; Nowak et al., 1990; Kennaway et al., 1992), so the fetus must rely on maternal sources of melatonin for time-of-day information. Melatonin of maternal origin crosses the placenta and accesses fetal tissues, as evidenced by studies that show that maternal pinealectomy decreases fetal plasma melatonin levels and abolishes the rhythm in fetal melatonin in the sheep (Yellon and Longo, 1988). The impact of maternal melatonin on the fetal circadian system appears to be organ-specific and dynamic across pregnancy (see below).
The hypothalamic-pituitary-adrenal (HPA) axis
Circadian variation in the HPA axis is a key driver in the overall regulation of adrenal glucocorticoid secretion. Rhythmic variation in maternal glucocorticoid levels (cortisol in most mammals including humans; corticosterone in rodents) is clearly evident for most of human (Patrick et al., 1980), rat (Atkinson and Waddell, 1995), and mouse (Wharfe et al., 2016a) pregnancy; however, in each case, absolute levels increase progressively from mid-gestation. This increase is particularly strong late in mouse pregnancy and coincides with an apparent loss of corticosterone rhythmicity (see Figure 1) (Wharfe et al., 2016a). These HPA axis changes in the mouse occur in conjunction with a gestational shift in clock gene rhythms in the maternal hypothalamus (Wharfe et al., 2016a). The adaptive significance of these changes is thought to relate to metabolic alterations that promote adipose tissue lipolysis and glucose mobilization for fetal growth (see below).
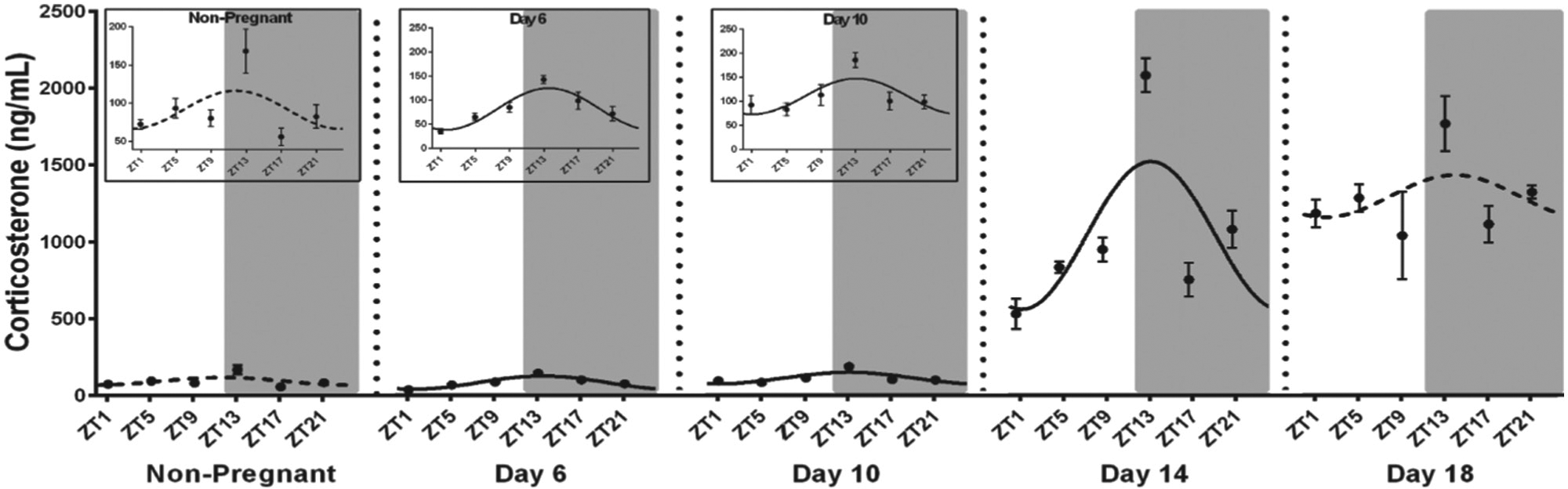
Plasma concentrations (ng/ml) of corticosterone before and across days 6, 10, 14 and 18 of mouse pregnancy. Values are the mean ± SEM. Grey shading represents the dark phase of the light cycle. Insets for non-pregnant and days 6 and 10 of pregnancy show the same data on a reduced y-axis scale. The best-fit curve derived by cosinor analysis is shown as a solid line when significant (P < 0.05) and as a dashed line when non-significant. Modified from Wharfe et al. (2016a).
Sex steroids
Progesterone is essential for pregnancy success; it provides trophic support for the placenta and fetus (Ogle et al., 1990; Mark et al., 2006), moderates maternal immunology to facilitate implantation (Joachim et al., 2003), and maintains uterine quiescence across gestation (for review see Brown et al., 2004). Progesterone levels increase steadily over human gestation due to increased ovarian (luteal) and then placental production, and there is evidence that this occurs in a circadian manner, possibly due to circadian regulation of placental steroidogenesis (Serón-Ferré et al., 1993). Indeed, numerous studies have reported daily variation of progesterone in human and non-human primate pregnancy, with a majority showing elevated progesterone levels during the dark hours (Walsh et al., 1984; Wilson et al., 1991; Magiakou et al., 1996; Giussani et al., 2000); although, not all studies detected progesterone rhythmicity (Darne et al., 1989). Recent studies also show daily changes in maternal plasma progesterone during late gestation in the rat (Crew et al., 2016) and mouse (Wharfe et al., 2016c); but, unlike the nocturnal peaks observed in human pregnancy, maternal progesterone falls by 20% to 50% during the dark phase. In rodents, the corpus luteum is the predominant source of progesterone throughout gestation, and maternal plasma levels increase markedly from early- to mid-gestation before declining near term as the corpus luteum regresses (Pepe and Rothchild, 1974; Waddell et al., 1989). Daily variation in progesterone secretion in rodents may be linked to the rhythmic expression of ovarian clock genes, which are thought to regulate circadian aspects of fertility and luteal cell function (Boden et al., 2013; Sellix, 2015). Indeed, downregulation of Bmal1 attenuates expression of steroidogenic genes and thus progesterone synthesis in rat granulosa cells (Chen et al., 2013). Moreover, mice with a Bmal1 deletion specific to ovarian steroidogenic cells have markedly reduced progesterone secretion and complete pregnancy failure. This effect was attributed to implantation failure, since copulation and ovulation rates in Bmal1-null mice were comparable to those in control animals (Liu et al., 2014). Collectively, these studies indicate that circadian disruption alters ovarian progesterone synthesis and, thus, fertility outcomes; however, it is unknown how the circadian regulation of ovarian progesterone synthesis may be moderated across gestation, and what impact this may have on pregnancy outcomes. Estrogen levels also exhibit a nocturnal surge 4 h after lights off during late gestation in baboons (Wilson et al., 1991; Giussani et al., 2000) and are higher in the early morning than in the afternoon or evening in the rhesus macaque (Hess et al., 1981; Walsh et al., 1984). Because the placenta is a major source of estrogen for most of primate pregnancy (via aromatization of fetal adrenal androgens; Walsh et al., 1984), rhythmicity in maternal estrogen levels likely reflects elevated nocturnal production of dehydroepiandrosterone by the fetal adrenal (Challis et al., 1980b). Pregnant women also exhibit daily changes in estrogen in the third trimester, with concentrations lower at night (2330 h to 0330 h) than during the day (Challis et al., 1980a).
Metabolic Adaptations
The mother faces major metabolic challenges in pregnancy, including an initial anabolic period of anticipatory adjustment when energy is stored in readiness for the dramatic rise in fetal demand that drives the maternal catabolic phase later in pregnancy. Recently, we characterized the expression of clock genes in the maternal liver (Wharfe et al., 2016b) and adipose tissue (Wharfe et al., 2016c) across mouse pregnancy to assess their possible impact on these phases of metabolic adaptation. In the liver, the key finding was that, although daily variation in clock genes was largely maintained across pregnancy, the amplitude of this rhythmic expression was curtailed (see Figure 2). Moreover, these changes were associated with a similar reduction in the daily variation of hepatic genes involved in glucose homeostasis (see Figure 2), possibly an adaptive response to ensure a sustained supply of glucose to meet the high demands of fetal growth in late gestation (Wharfe et al, 2016b). Interestingly, the molecular clock appeared more stable in adipose tissue across pregnancy, with relatively little change in the characteristics of clock gene rhythms (Wharfe et al, 2016c). But there was an apparent decoupling of the molecular clock from some downstream metabolic genes, since the clearly rhythmic expression patterns of adipose Lpl, Lipe, and Pnpla2 were lost with the onset of pregnancy. Again, this may be part of the maternal adaptive response, as the continuous expression of lipolytic enzymes, such as Lipe and Pnpla2, may ensure constant mobilization of fatty acids to meet the continuous demands of the rapidly growing fetus (Wharfe et al., 2016c).
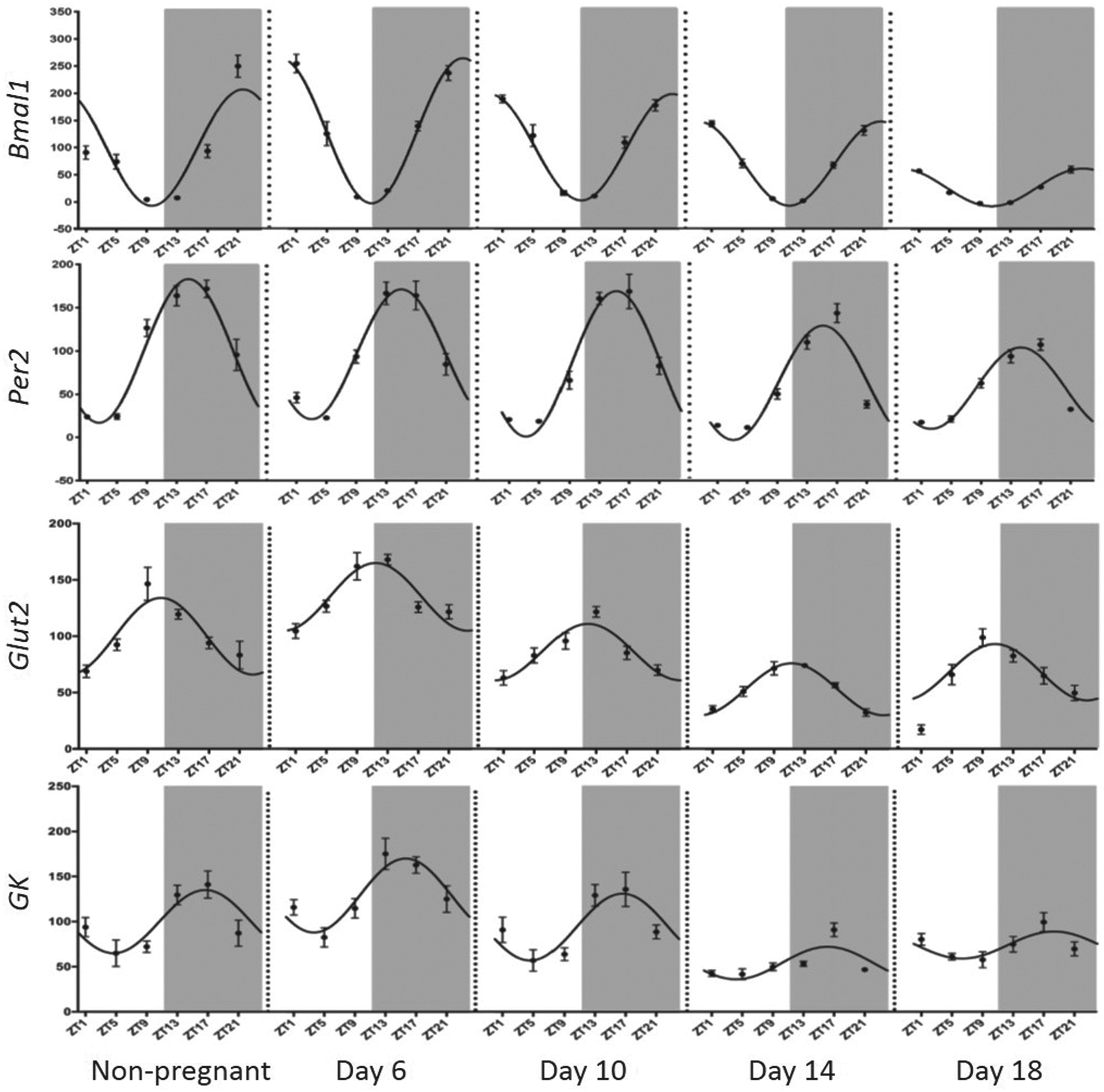
Hepatic expression of Bmal1, Per2, Glut2, and Gk before and across days 6, 10, 14, and 18 of mouse pregnancy. Values are ± SEM and are expressed relative to the mesor in non-pregnant mice (adjusted to 100). Grey shading represents the dark phase of the light cycle. The best-fit curve derived by cosinor analysis is shown as a solid line when significant (P < 0.05) and as a dashed line when non-significant. Modified from Wharfe et al. (2016b).
Core Body Temperature Changes
In humans, core body temperature (Tc) measured at midday falls with advancing gestation (Hartgill et al., 2011); however, we are not aware of any studies of full circadian profiles of Tc measured across human pregnancy. In non-pregnant, nocturnal animals the daily variation in Tc is characterized by a trough during the light phase and a peak during the dark phase, with an amplitude of approximately 0.75°C (Eliason and Fewell, 1997). Tc rhythms are markedly altered during pregnancy in both rats and mice (Fewell, 1995; Gamo et al., 2013), and we recently observed that although the mesor of the Tc rhythm increases early in mouse gestation, its amplitude falls as early as day 6 and continues to decline thereafter (Wharfe et al., 2016c; see Figure 3). These observations are consistent with the loss of daily variation in Tc in rat pregnancy in which the normal dark phase increase is not observed from day 15 (Fewell, 1995). There is also a late pregnancy reduction in the mesor of Tc in both rats and mice (Fewell, 1995; Gamo et al., 2013; Wharfe et al., 2016c; see Figure 3), which has been proposed to be an adaptive response to heat dissipation across the placenta. The resultant increased heat gradient between the fetus and the mother likely enables the mother to act as a heat sink for the highly metabolically active fetus and placenta, thereby maintaining optimal temperature in utero. Although the stimulus for altered rhythmicity in Tc remains unknown, a role for the placenta and/or the fetus has been proposed due to the rapid restoration of normal rhythmicity immediately after birth (Fewell, 1995). Also, because progesterone can exert thermogenic effects (Freeman et al., 1970), it has been suggested that its preterm decline in rodents may contribute to the late gestation fall in Tc (Fewell, 1995).
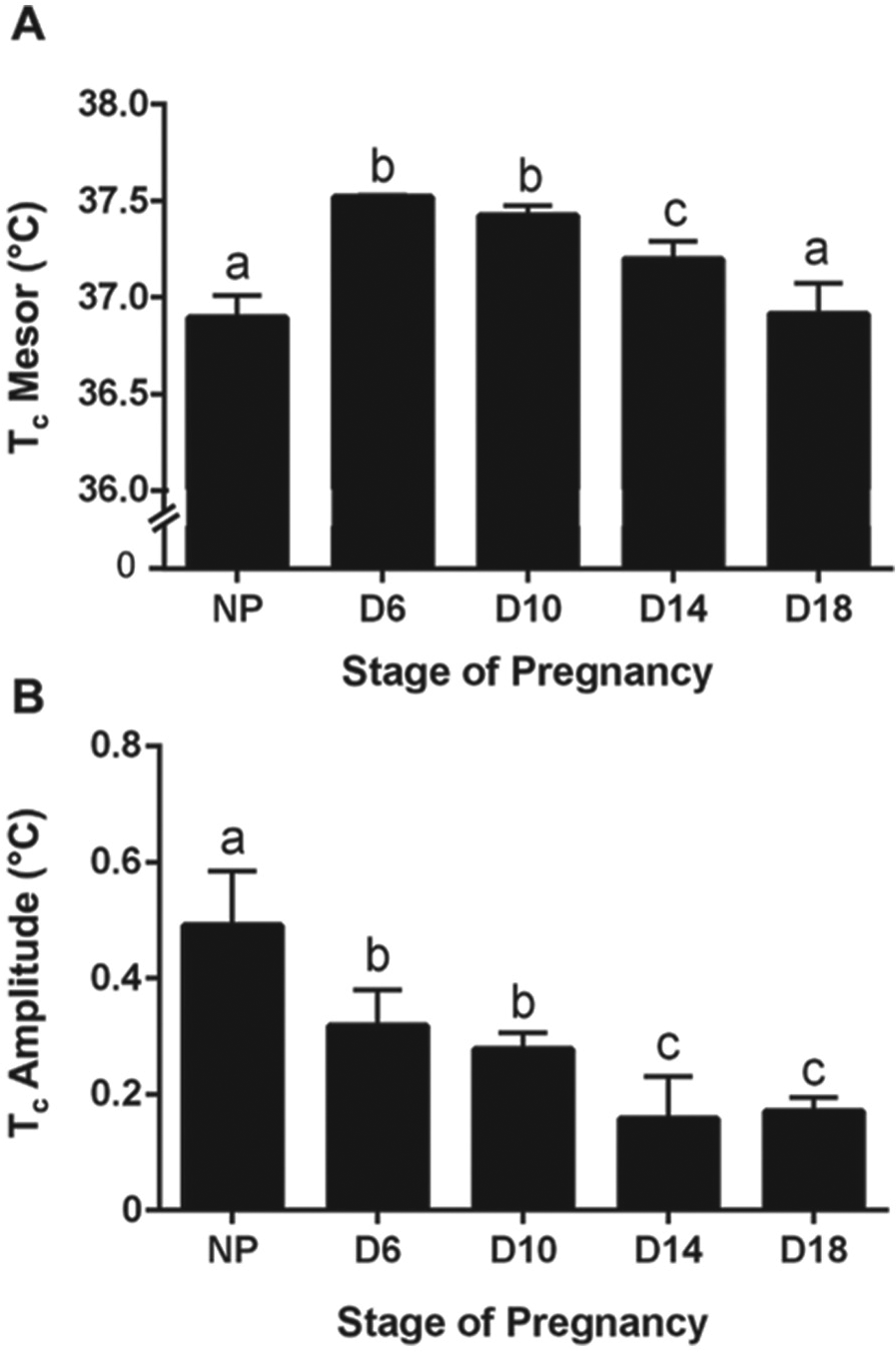
Changes in the (A) mesor and (B) amplitude of core body temperature (Tc) rhythms (derived by cosinor analysis) before (NP; non-pregnant) and at days 6, 10, 14, and 18 of mouse pregnancy. Values are the mean ± SEM. Values without common notation differ significantly (P < 0.05; one-way ANOVA and post hoc Fisher’s LSD test). Modified from Wharfe et al. (2016b).
Maternal Activity and Food Intake
It is well established that pregnancy substantially reduces activity levels in both humans and rodents (Richards, 1966; Rousham et al., 2006; Gamo et al., 2013) but its effect on the daily variation in activity is less well characterized. In non-pregnant rodents, this profile is characterized by low levels of activity during the light phase, which increase substantially with the onset of darkness and progressively decline towards lights on (Kennaway et al., 2003; Gamo et al., 2013). Peak activity count (at 2000 h) is approximately 10 times greater than at the trough (1500 h) in mice (Gamo et al., 2013). Free-running activity rhythms in late gestation were found to be highly variable in rats, with a circadian pattern difficult to detect (Rosenwasser et al., 1987), whereas the amplitude of the activity rhythm was reduced closer to term in hamsters (Scribner and Wynne-Edwards, 1994). This late gestation decrease in activity likely conserves energy for use by the rapidly growing fetus and, as such, may contribute to maternal metabolic adaptations. Interestingly, despite maternal hyperphagia being maintained throughout gestation in rodents, the daily rhythm of food intake does not change appreciably, with most food consumption still occurring exclusively in the dark phase (Hitier et al., 1982; Varcoe et al., 2013).
The Placenta and its Rhythmic Function
Successful pregnancy requires the de novo formation of a placenta that provides a functional interface between the mother and the fetus. Accordingly, the placenta facilitates the transfer of oxygen, nutrients, and waste to ensure meeting the metabolic requirements for the developing fetus. Both human and rodent placentas are hemochorial in structure; that is, maternal blood is in direct contact with the fetal portion of the placenta (as reviewed in Pijnenborg et al., 1981). In humans, this fetal placenta comprises finger-like projections (termed chorionic villi) that are lined externally by the syncytiotrophoblasts and are bathed in maternal blood. Because the chorionic villi contain fetal capillaries, they bring the maternal and fetal blood circulations into proximity and thereby facilitate the exchange of nutrients and gases (Pijnenborg et al., 1981). The fetal portion of the rodent placenta contains 2 morphologically and functionally distinct zones: the junctional zone is primarily responsible for hormone production, whereas the labyrinth zone is the region of maternal-fetal exchange (and is thus analogous to the chorionic villi of the human placenta). The labyrinth zone, as its name suggests, is labyrinthine in structure with maternal blood spaces separated from the fetal vasculature by syncytiotrophoblasts and fetal connective tissue (Soares, 1987).
Placental Rhythmicity
In addition to regulating the exchange of nutrients, gases, waste, and hormones between the mother and the fetus, the placenta is a highly metabolic, multifunctional organ. As such, it can potentially influence fetal circadian biology by transmitting rhythmic maternal signals and via its own rhythmic function. Frigato et al., (2009) first observed the rhythmic expression of clock genes in the human extravillous trophoblast cell line HTR-8/SVneo. Subsequent studies demonstrated clock gene expression in the placenta of rodents (Ratajczak et al., 2010; Wharfe et al., 2011) and humans (Perez et al., 2015) but the extent to which this expression is rhythmic in vivo remains unclear. For example, although marginal time-of-day variations were observed for Bmal1, Per1, and Per2 in the labyrinth zone of the rat placenta, the conventional anti-phase expression of Bmal1 and Per1/Per2 was not apparent (Wharfe et al., 2011). More recently, Papacleovoulou and colleagues (2017) reported the rhythmic expression of Bmal1, Clock, and Reverb genes in whole mouse placenta but the Bmal1 pattern was opposite to that observed in maternal tissues (liver and muscle) from the same pregnancies. Placental clock gene rhythms were also reported recently for the human placenta but, oddly, their period varied markedly among clock genes and there was no evidence of an anti-phase relationship between BMAL1 and PER2 (Perez et al., 2015). Collectively, these observations indicate that the positive and negative transcriptional feedback interactions among placental clock genes (and their protein products) differ from those in adult tissues, similar to the interactions among clock genes described below for some fetal tissues.
Despite the relatively stable expression of placental clock genes, other aspects of placental function appear to follow a rhythmic pattern (for review see Waddell et al., 2012). For example, maternal plasma human chorionic gonadotrophin (hCG) levels show a daily rhythm, particularly early in gestation when placental hCG expression is maximal (Díaz-Cueto et al., 1994). Rhythmic expression is also evident for rat placental lactogen II (rPL-II) in the placental junctional zone, with peak expression occurring late in the dark phase (Lee et al., 2003). Intriguingly, the inverse rhythmic pattern in rPL-II expression was observed in the neighboring labyrinth zone, possibly related to opposite rhythms in melatonin receptor expression in these placental zones (Lee et al., 2003). These observations highlight the likely importance of maternal circadian signals (in this case, melatonin) in driving rhythmic placental function, and the need for the separate characterization of expression profiles in the distinct tissue types among peripheral oscillators. Interestingly, the placenta also produces its own melatonin but, like other non-pineal sources, this is not thought to vary in a circadian fashion (for review see Reiter et al., 2014).
Fetal Circadian Development
Various aspects of fetal physiology, including heart rate, body movement, and hormone levels, exhibit circadian variation in late gestation (for reviews see Davis and Reppert, 2001; Serón-Ferré et al., 2007). These fetal rhythms are likely driven, at least in part, by the rhythmic function of the fetal SCN that was first observed by Reppert and Schwartz (1984). What is less clear is the underlying drive for this rhythmicity and, specifically, the extent to which it reflects functional, intrinsic molecular clocks in fetal tissues comparable to those described above for adult tissues. This is because rhythmic fetal functionality may be driven by extrinsic signals from the mother and/or the placenta. Teasing apart the relative importance of intrinsic v. extrinsic rhythmicity in the fetus is complicated by species differences; in altricial species (e.g., laboratory rodents) circadian development occurs primarily in the postnatal period (Davis and Reppert, 2001; Sladek et al., 2007), whereas, in precocial species with a longer gestation (e.g., humans, non-human primates, and sheep) there appears to be more substantial circadian development in utero (Serón-Ferré et al., 2001b).
Clock gene expression in fetal (and embryonic) tissues has been investigated predominantly in rodent models. Whole mouse embryos express clock genes from day 10 of gestation and, while this expression increases up to day 19, importantly, it does not appear to be rhythmic at any prenatal stage (Dolatshad et al., 2010). Explants of fetal mouse SCN obtained from gestational day 14 onwards display robust rhythmic Per2 expression in vitro (Wreschnig et al., 2014) but whether this oscillatory expression occurs in vivo is not clear. In the rat fetus, clock genes are expressed in the SCN by gestational days 19 to 20 but their rhythmicity is not fully developed until postnatal day 10 (Sládek et al., 2004; Kováčiková et al., 2006). Nevertheless, the fetal rat SCN at gestational day 19 exhibits a 24-h rhythm in both metabolic activity (as indicated by glucose utilization) (Reppert and Schwartz, 1984) and in expression of other genes, including c-Fos and Avp (Houdek and Sumova, 2014), presumably in response to cyclic maternal signals (see below) (Sumova et al., 2008; Seron-Ferre et al., 2012).
While clock genes are also expressed in the fetal liver, this appears to be largely arrhythmic (Wharfe et al., 2011). For example, whereas time-of-day variation is evident in fetal hepatic expression of Per2, Per3, and Cry1 at day 21, the expression profiles do not exhibit the typical anti-phase relationships characteristic of intrinsic molecular clocks (Wharfe et al., 2011). Daily variation in hepatic clock gene expression gradually develops in the postnatal period in rats, such that all appear fully rhythmic by postnatal day 30 (Sladek et al., 2007). Thus, substantial development of peripheral metabolic clocks occurs during lactation, and newborn rat offspring separated from their mothers (i.e., food restricted) during the light phase show a complete inversion of SCN rhythms by postnatal day 6 (Ohta et al., 2003). This highlights the importance of postnatal feeding cues for central circadian development in rodents. Hepatic clock gene expression in rat offspring also changes as feeding patterns shift from the relatively constant daily nursing behavior to rhythmic patterns in nocturnal food consumption after weaning (Yamazaki et al., 2009).
In contrast to the fetal SCN and liver, the fetal adrenal clock appears to be functional late in rat pregnancy (Torres-Farfan et al., 2011). Specifically, the day-18 fetal adrenal displays anti-phase expression of Bmal1 and Per2 (see Figure 4) and is associated with daily rhythms in corticosterone content and steroidogenic acute regulatory protein expression (Torres-Farfan et al., 2011). Similarly, the fetal adrenal of the Capuchin monkey shows anti-phase rhythms of Bmal1 and Per2 at 0.9 gestation but, remarkably, these rhythms are not under the control of the fetal SCN (reviewed in Seron-Ferre et al., 2012). This observation highlights the importance of maternal and/or placental signals in regulating fetal rhythms.
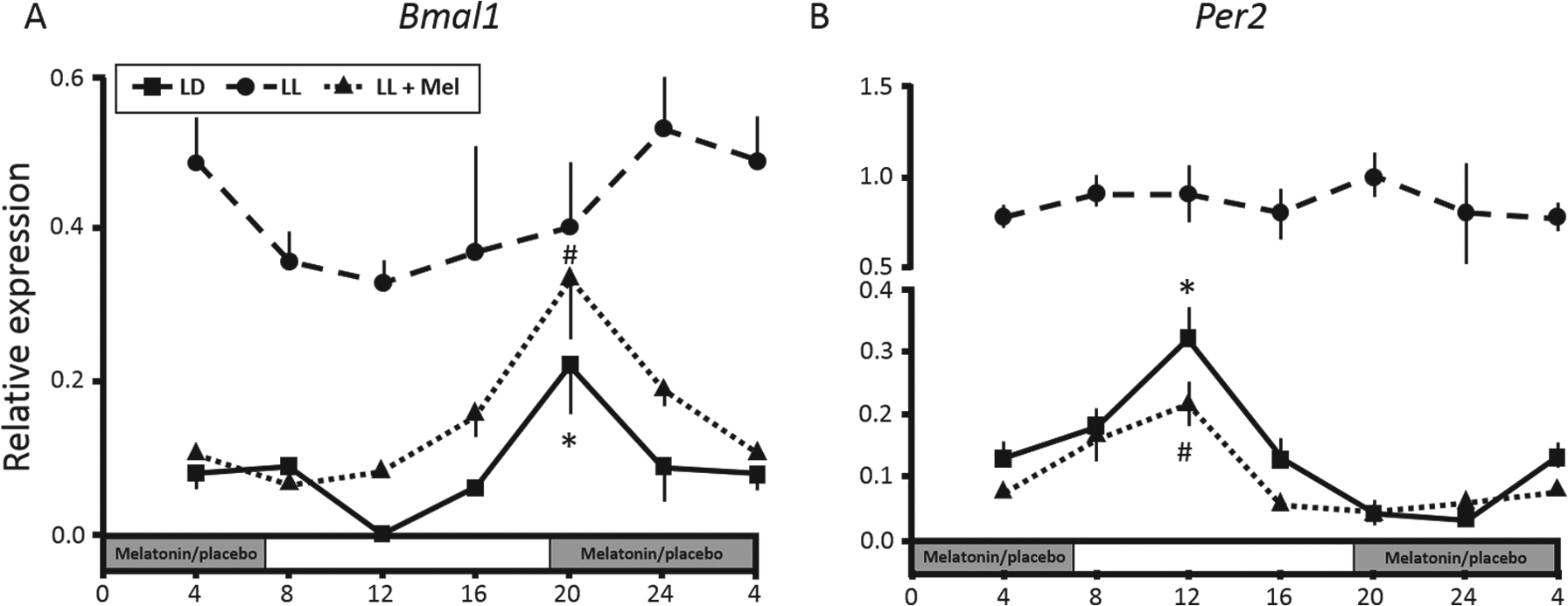
Fetal adrenal expression of (A) Bmal1 and (B) Per2 in rats maintained in either a normal light-dark cycle (LD) or under constant light with (LL+Mel) or without (LL) maternal melatonin replacement. Values are the mean ± SEM; grey shading represents the dark phase of the light-dark cycle and of melatonin replacement or placebo during subjective night. *P < 0.05 compared with other time points for LD; #P < 0.05 compared with other time points for LL+Mel. Modified from Mendez et al. (2012).
Fetal Exposure to Maternal Rhythmic Signals
The context of mammalian pregnancy provides an unusual biological circumstance whereby an entire circadian system (i.e., that of the fetus) may be directly influenced by the organism within which it is embodied (i.e., the mother). Indeed, it has been proposed that the fetus is a peripheral oscillator within the maternal system; that is, rather than peripheral circadian clocks in the fetus being entrained by the fetal SCN, they are largely controlled by the maternal circadian system (Serón-Ferré et al., 2007; Seron-Ferre et al., 2012). In relation to the early evidence supporting maternal regulation of fetal rhythmic function, the reader is directed to the comprehensive review by Davis and Reppert (2001). This review includes the detailed consideration of the crucial transition from maternal entrainment of the fetal SCN in utero to entrainment of the postnatal SCN by environmental cues.
Among the maternal factors likely to impact fetal rhythmicity, maternal endocrine signals, most notably melatonin and glucocorticoids, appear to play a central role, and there may also be a role for other maternal hormones that exhibit rhythmic profiles, such as leptin and progesterone. An important caveat here is that the influence of any maternal endocrine rhythm on the fetus can change across gestation due to either alterations in maternal levels or the action of the placenta. In some cases, such placental “intervention” may be viewed as a means of “weaning” the developing fetus off rhythmic maternal signals in late gestation, particularly in precocial species. Evidence supporting possible time-signaling roles for each of the major maternal players is presented below.
Melatonin
As discussed above, the synchrony of maternal and fetal melatonin rhythms is thought to reflect transplacental passage of maternal melatonin to the fetus. Accordingly, melatonin is considered a key maternal zeitgeber that conveys time-of-day information to the fetus (McMillen and Nowak, 1989; Kennaway et al., 1992; Seron-Ferre et al., 2012). In a series of critical studies involving manipulation of maternal melatonin in pregnancy, Seron-Ferre and colleagues have identified important regulatory effects of melatonin on the fetal SCN and adrenal clocks, as well as highlighting species differences (Torres-Farfan et al., 2006; Torres-Farfan et al., 2011; Mendez et al., 2012; Seron-Ferre et al., 2012). For example, in the Capuchin monkey, maternal melatonin appears to regulate clock gene expression in the fetal SCN but has no influence on fetal adrenal rhythmicity (Torres-Farfan et al., 2006). This is consistent with previous studies showing that the administration of exogenous melatonin to pregnant hamsters with SCN lesions can entrain rhythmicity in the offspring at weaning (Davis and Mannion, 1988). More recently, Houdek et al. (2015) showed that exogenous melatonin synchronizes clock gene expression in the fetal SCN but not in the fetal liver. In rats, maternal melatonin appears to directly influence the fetal adrenal clock, since anti-phase Bmal1 and Per2 rhythms are abolished by suppression of maternal melatonin secretion under constant light (Mendez et al., 2012; see Figure 4). This effect was reversed by melatonin replacement, as were the suppressive effects of constant light on adrenal corticosterone rhythms (Mendez et al., 2012).
Glucocorticoids
Maternal glucocorticoids can also traverse the placenta and so they too may provide a temporal signal to the fetus. Importantly, this signal changes markedly across gestation for 2 reasons: first, maternal glucocorticoid levels increase toward term in most species (e.g., Wharfe et al., 2016a); and second, glucocorticoid passage from the mother to the fetus varies with gestational age due to the dynamic nature of the placental glucocorticoid barrier. The latter is primarily a metabolic gatekeeper, whereby the enzyme 11β-hydroxysteroid dehydrogenase type 2 (11β-HSD2) converts biologically active glucocorticoids (cortisol or corticosterone) to their biologically inert metabolites (cortisone and 11-dehydrocorticosterone (11-DHC), respectively). Intriguingly, placental 11β-HSD2 expression increases with advancing gestation in primates (Pepe and Albrecht, 1998; McTernan et al., 2001) but decreases during late pregnancy in rodents (Burton et al., 1996; Waddell et al., 1998). As a result, the passage of maternal glucocorticoids to the fetus falls towards term in primates but increases in rodents (Burton and Waddell, 1999; Mark et al., 2009). We recently characterized rhythmic patterns of corticosterone and 11-DHC in the mother and fetus of late-pregnant rats and identified an unexpected asynchrony in maternal and fetal corticosterone rhythms. Specifically, whereas the fetal corticosterone peak occurred around 8 h earlier than the maternal peak, maternal and fetal 11-DHC rhythms were synchronous over the same period (see Figure 5). These data suggest that the fetal HPA axis (and thus adrenal corticosterone secretion) is activated in response to the falling delivery of corticosterone during the nadir of the maternal rhythm. Interestingly, umbilical artery (but not umbilical vein) cortisol levels show a similar rhythmic profile in the human fetus near term (Serón-Ferré et al., 2001a) but this more likely reflects an intrinsic fetal HPA rhythm, since the placental glucocorticoid barrier is more complete in human and non-human primates at term (Pepe and Albrecht, 1998; Burton and Waddell, 1999). This contention is further supported by the presence of distinct glucocorticoid rhythms in newborn human infants (Iwata et al., 2013). Regardless of how the fetal glucocorticoid rhythm is generated, it may provide an important prelude to endogenous rhythmicity in postnatal life, as glucocorticoids are key players in the entrainment of peripheral clocks (Balsalobre et al., 2000).
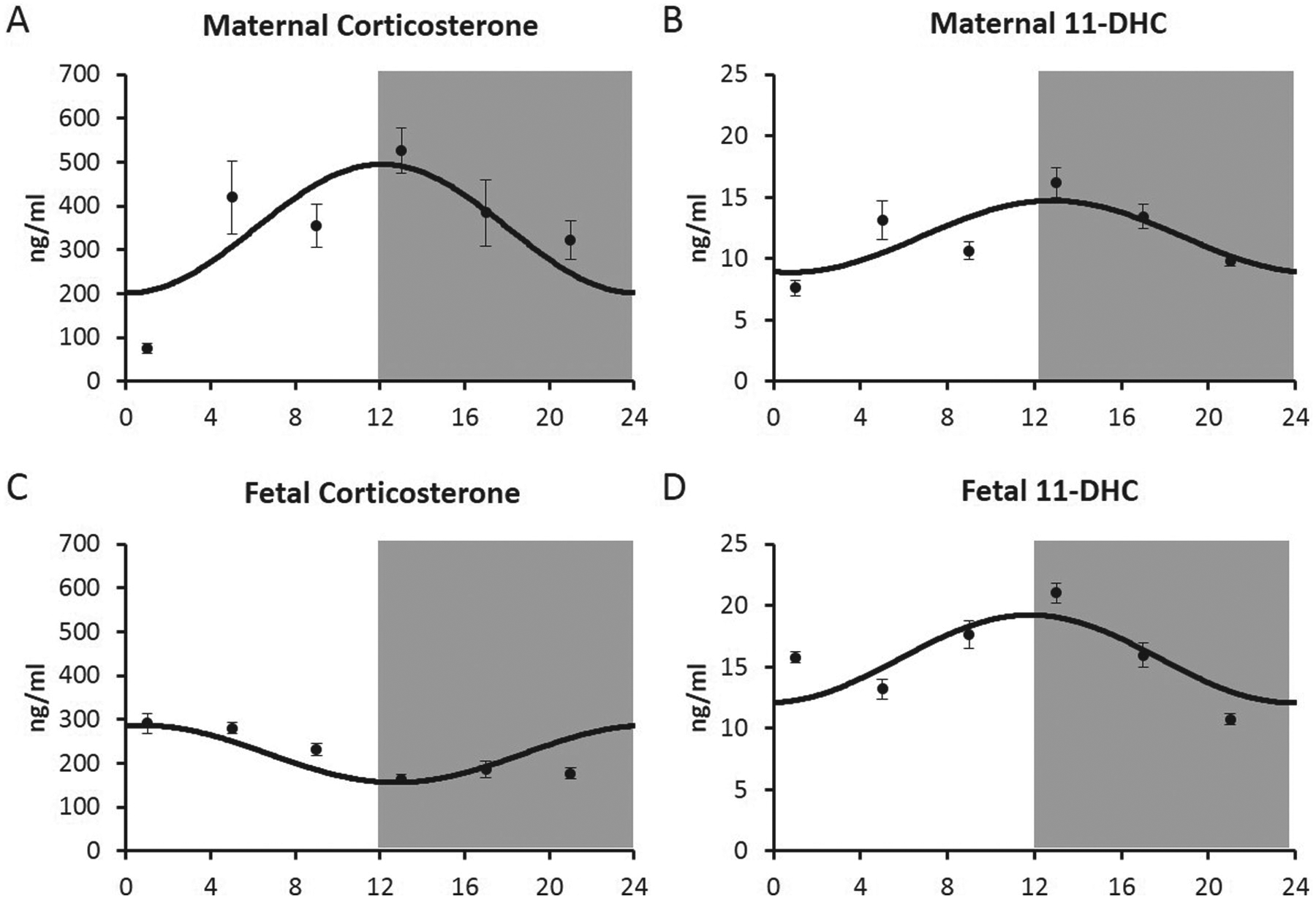
Rhythmic (cosinor) profiles of (A, C) plasma corticosterone and (B, D) 11-dehydrocorticosterone (11-DHC) in (A, B) maternal and (C, D) male fetal plasma across days 21 to 22 of rat pregnancy. Values are the mean ± SEM; grey shading represents the dark phase of the light-dark cycle. Note that while the maternal and fetal 11-DHC rhythms are synchronous, those of corticosterone are not. Modified from Crew et al. (2016).
Other Maternal Signals
Patterns of maternal feeding have also been shown to influence fetal rhythms, specifically rat fetal SCN AVP and c-Fos expression. Thus, Novakova and colleagues (2010) showed that, although AVP and c-Fos SCN rhythms were unaffected by restricted feeding in the normal LD cycle, the abolition of these fetal SCN rhythms by exposure to constant light was reversed by restricted feeding. Because constant light alters a range of other maternal signals, including melatonin, the specific mechanisms underlying these effects remain obscure. Nevertheless, they do raise the possibility that maternal signals that are influenced by feeding, such as leptin and insulin production, could potentially affect fetal rhythms either directly or via effects on placental function. Indeed, most fetal leptin in rodents is thought to be maternal in origin (Smith and Waddell, 2003b; Smith and Waddell, 2003a), and we recently observed strong cosinor rhythmicity in fetal plasma leptin levels in the rat; albeit, with relatively low amplitude (unpublished observations). Interestingly, however, in this case, the fetal leptin rhythm was not accompanied by similar variation in the mother, raising the possibility that transplacental passage of leptin may show daily variation, as has been suggested previously for nutrient passage (Waddell et al., 2012). In any event, daily variation in fetal leptin is likely to influence various aspects of fetal physiology, and again this may provide a prelude to the postnatal leptin rhythms that will subsequently develop. Finally, maternal progesterone may also signal time-of-day information to the fetus, with synchronous rhythmic profiles of progesterone in the mother and fetus recently reported in the rat (Crew et al., 2016), a species in which the maternal ovary (specifically, the corpus luteum) is the major source of progesterone. Given that a significant proportion of maternal progesterone reaches the fetus intact (Benbow and Waddell, 1995), the daily variation observed for fetal progesterone (see Figure 6) is most likely to be maternal in origin. This contrasts with progesterone rhythms in nonhuman primates, which are thought to reflect daily variation in fetal adrenal steroid production (Walsh et al., 1984). Regardless of rhythm origin, because progesterone has been shown to regulate clock gene expression (He et al., 2007; Nakamura et al., 2010) and can counter the actions of glucocorticoids (Patel et al., 2003), its rhythmic variation may well influence fetal circadian biology. Importantly, the normal nocturnal decline in fetal progesterone observed in late pregnancy did not occur in mothers with diet-induced obesity (see Figure 6). This highlights the potential for a maternal pathology, in this case, obesity, to influence fetal circadian biology (Crew et al., 2016).
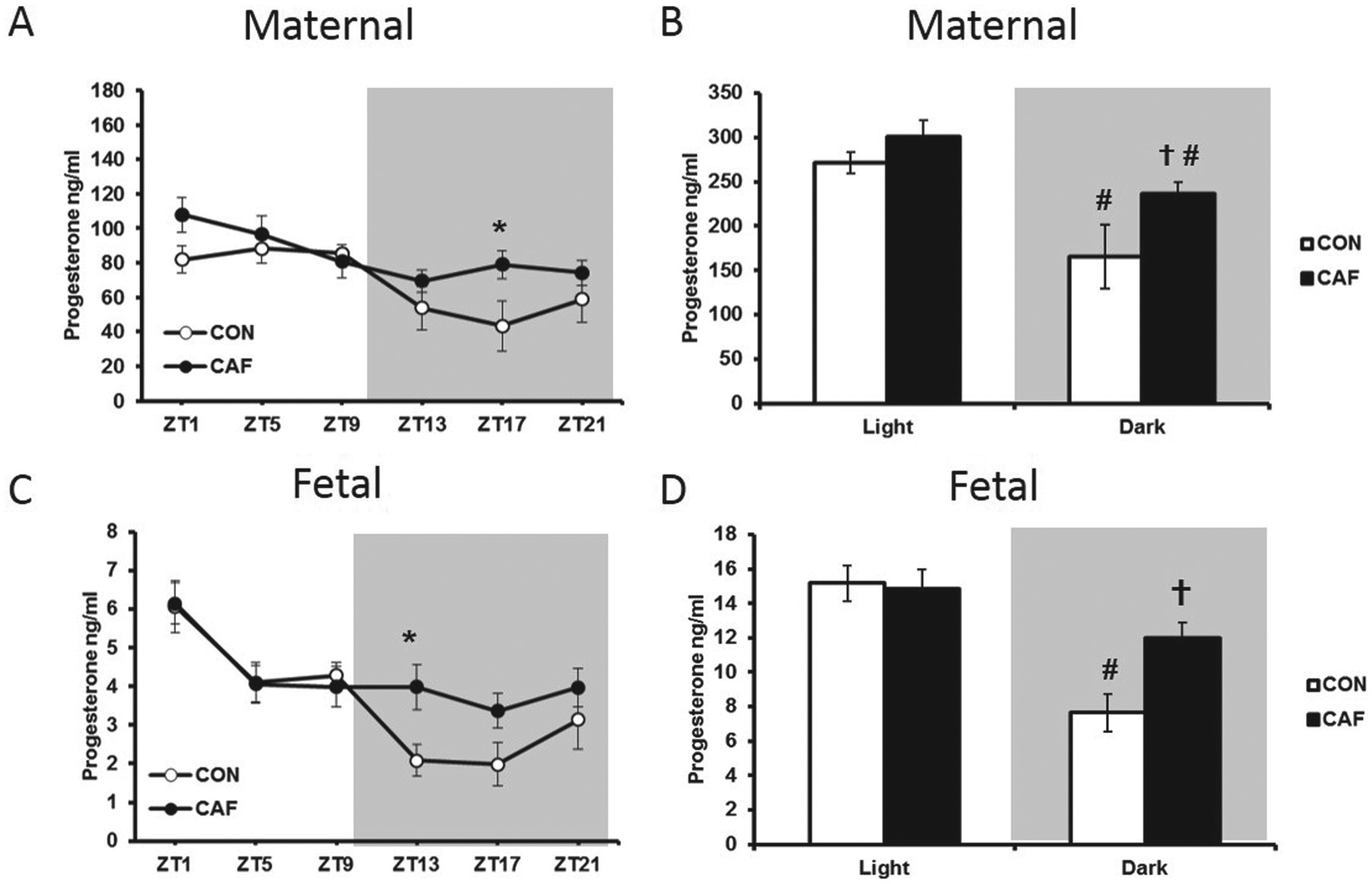
Daily variation in (A) maternal and (C) female fetal progesterone and average progesterone values in (B) maternal and (D) fetal plasma across the ‘light’ and ‘dark’ periods in control (CON) and cafeteria-diet fed (CAF) pregnant rats. Values are the mean ± SEM; grey shading represents the dark phase of the light-dark cycle. *P < 0.05 compared with CON (t test following Diet × Time of Day interaction with two-way ANOVA). P < 0.05 compared with corresponding CON value, #P < 0.05 compared with corresponding light value (t tests following Diet × Light interaction in two-way ANOVA). Modified from Crew et al. (2016).
Concluding Remarks
The interplay among maternal, placental, and fetal circadian systems presents an intriguing and complex biological phenomenon. Not only is this important for the establishment, maintenance, and successful completion of pregnancy, its implications may even extend well into postnatal life given the potential developmental programming effects of circadian disruption. A key challenge we now face is to understand how fetal clocks develop in utero and how they are susceptible to insults that compromise immediate fetal well-being and subsequent health in postnatal life. While animal models have provided considerable insight into how the mother and the placenta influence fetal circadian development and function, these studies have uncovered considerable heterogeneity among fetal tissues, marked species differences (most notably between altricial and precocial species), and maturational changes across gestation. There is also a tendency to assume parallels exist between adult and fetal circadian biology, such as the specifics of how the molecular clock machinery operates and how it responds to external cues. The risk in this assumption is nicely illustrated by comparing peripheral tissue clocks in the mother, the placenta and the fetus within a pregnancy. As shown in Figure 7, there is little similarity in the expression patterns for any of the clock genes in the maternal liver, placenta, or fetal liver late in rat pregnancy (Wharfe et al., 2011). These differences are likely explained by a combination of effects, including exposure of maternal clocks to different SCN-derived signals (e.g., via the autonomic nervous system), modification of endocrine signals (e.g., glucocorticoids) by the placenta, and fundamental differences in the transcriptional control of clock genes among maternal, placental, and fetal tissues. Considerable further study is required to understand how each of these circadian influences interacts throughout pregnancy to optimize maternal adaptations and placental function and thereby support adequate growth and development of the fetus and its transition to postnatal life in a circadian environment.
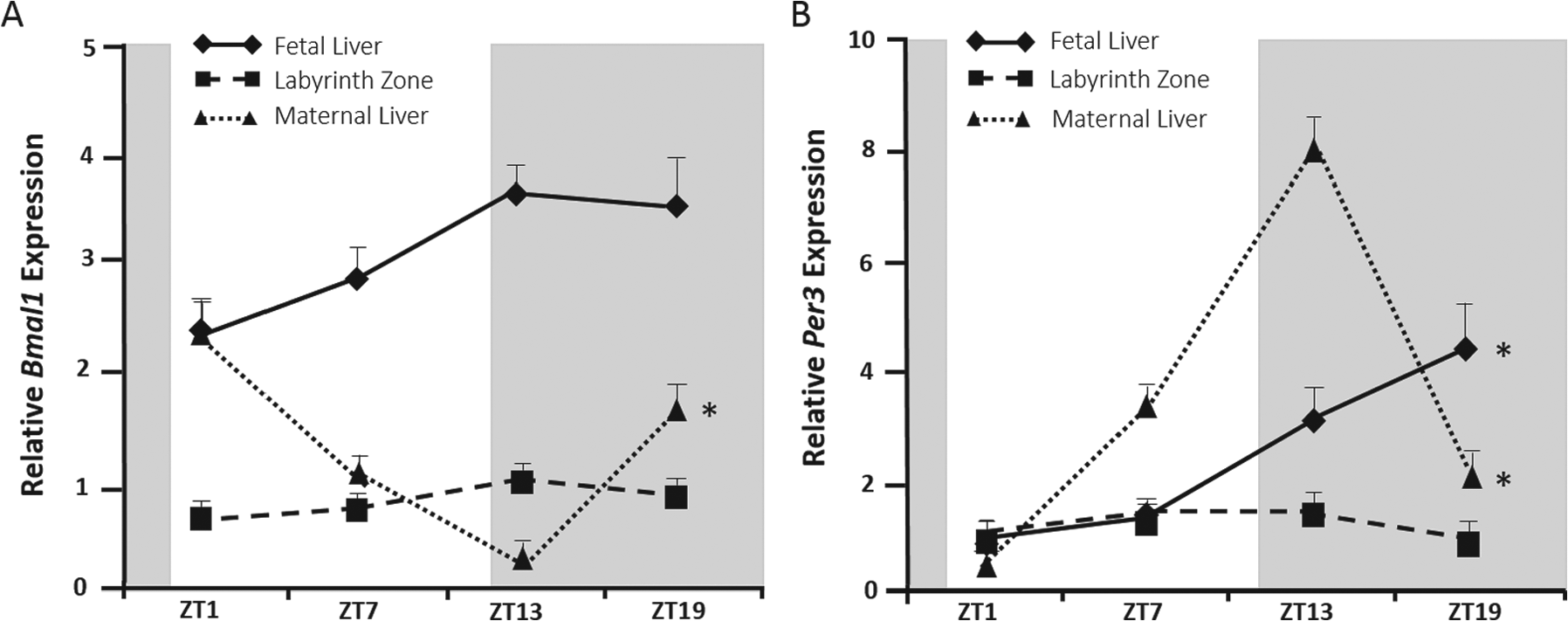
Daily variation in (A) Bmal1 and (B) Per3 expression in the maternal and fetal liver and the placental labyrinth zone over days 21 to 22 of rat pregnancy. Values are the mean ± SEM; grey shading represents the dark phase of the light-dark cycle. * Significant time-of-day variation, P < 0.05 (ANOVA). Modified from Wharfe et al. (2011)
Footnotes
Conflict of Interest Statement
The author(s) have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
