Abstract
The mammalian circadian timing system consists of a master pacemaker in the suprachiasmatic nucleus (SCN) and subordinate clocks that disseminate time information to various central and peripheral tissues. While the function of the SCN in circadian rhythm regulation has been extensively studied, we still have limited understanding of how peripheral tissue clock function contributes to the regulation of physiological processes. The adrenal gland plays a special role in this context as adrenal hormones show strong circadian secretion rhythms affecting downstream physiological processes. At the same time, they have been shown to affect clock gene expression in various other tissues, thus mediating systemic entrainment to external zeitgebers and promoting internal circadian alignment. In this review, we discuss the function of circadian clocks in the adrenal gland, how they are reset by the SCN and may further relay time-of-day information to other tissues. Focusing on glucocorticoids, we conclude by outlining the impact of adrenal rhythm disruption on neuropsychiatric, metabolic, immune, and malignant disorders.
The environmental changes associated with the rhythmic succession of night and day have led to the development of endogenous biological timekeepers in most organisms that allow for intrinsic timing of biological processes and behaviors at the appropriate time of day. Circadian clocks (from the Latin
From a chronobiological perspective, the adrenal gland is of particular interest. Adrenal endocrine functions show circadian regulation, and glucocorticoid (GC) rhythms appear to have a key role in synchronizing peripheral clocks downstream of the SCN. Apart from direct effects of rhythmic GC secretion on physiology, which include mediation of stress responses as well as immune and cognitive modulation, GC rhythms may therefore be involved in the adaptation of the circadian system under environmental pressures such as jetlag or shiftwork. Other adrenal hormones such as aldosterone and catecholamines also show time-of-day dependent modulation. The circadian effects of these hormones, however, are less well studied, although they might serve as important regulators of physiological rhythms.
Control of Adrenal Hormone Rhythms
The adrenal cortex produces 3 classes of hormones: mineralocorticoids (MCs) in the outer
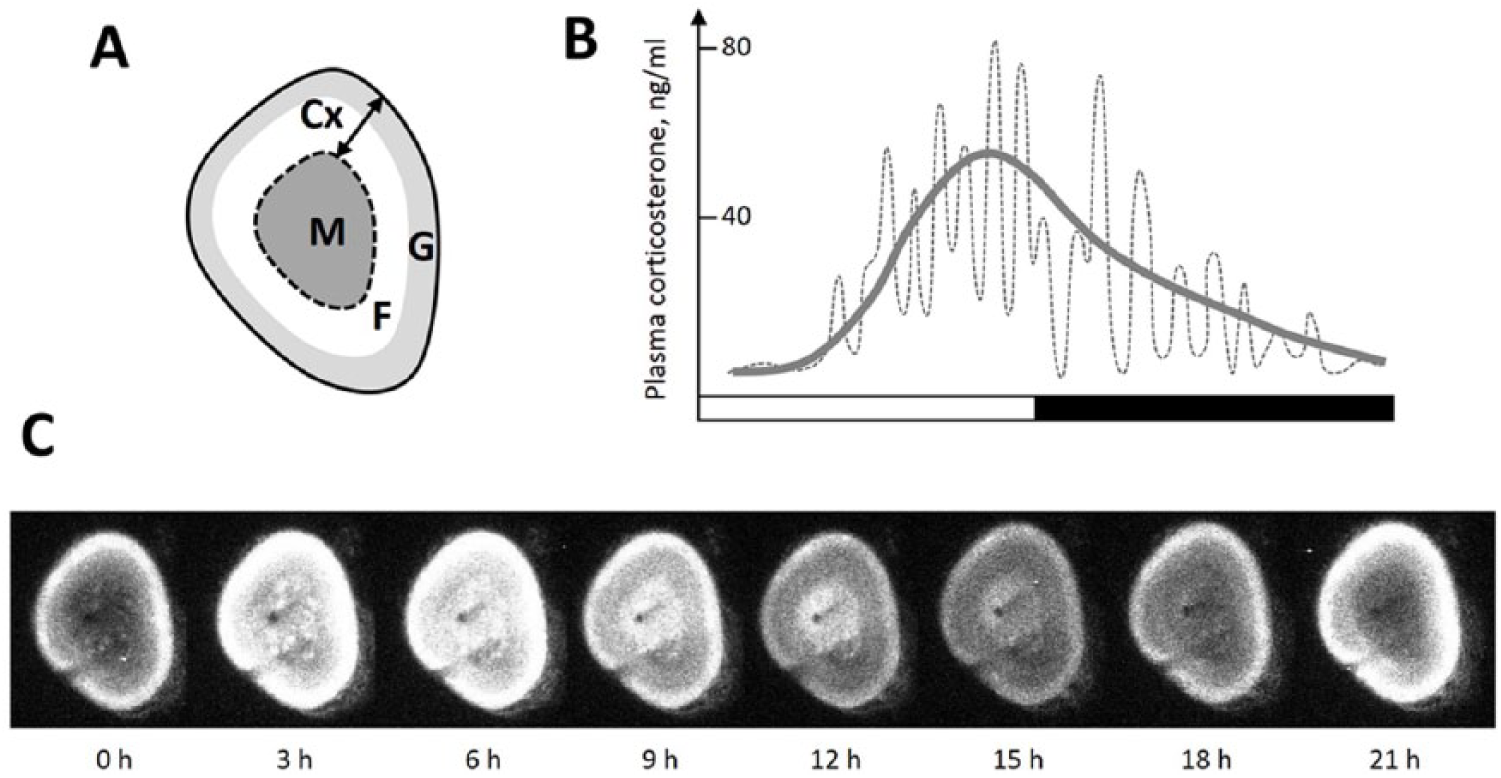
(A) Anatomy of adrenal gland. M = medulla; Cx = cortex; F = zona fasciculata; G = zona glomerulosa. (B) A schematic view of 24-h secretion of plasma corticosterone in the rat (redrawn from Sarabdjitsingh et al., 2010). Circadian variation is represented by a smoothing line. (C) A representative 1-day bioluminescence imaging of a murine PER2::LUC adrenal slice. Note that a peak of the luminescence signal in the adrenal medulla is delayed compared with the cortex.
While pulsatile GC release is controlled by a negative feedback loop involving GR signaling and adrenocorticotropic hormone (ACTH) suppression in the pituitary (Walker et al., 2012), circadian GC rhythms are regulated by 3 main factors: (1) the endocrine hypothalamic-pituitary-adrenal (HPA) axis, (2) SCN-controlled autonomic innervation, and (3) local adrenocortical circadian clocks (Figure 2A). In the HPA axis, circadian information from the SCN (Buijs et al., 1993) and stress-driven signals from the brainstem and the limbic structures of the forebrain (Ulrich-Lai and Herman, 2009) stimulate production of corticotropin-releasing hormone (CRH) and arginine vasopressin (AVP) at the paraventricular nucleus of the hypothalamus (PVN). These signals reach the corticotropes of the anterior pituitary via the blood portal system and stimulate secretion of corticotropin ACTH into circulation. ACTH then activates adrenocortical steroidogenesis via the melanocortin 2 receptor (MC2R) and subsequent cAMP-PKA pathways, which stimulate the transcription of steroidogenic genes such as cholesterol side chain cleavage enzyme (
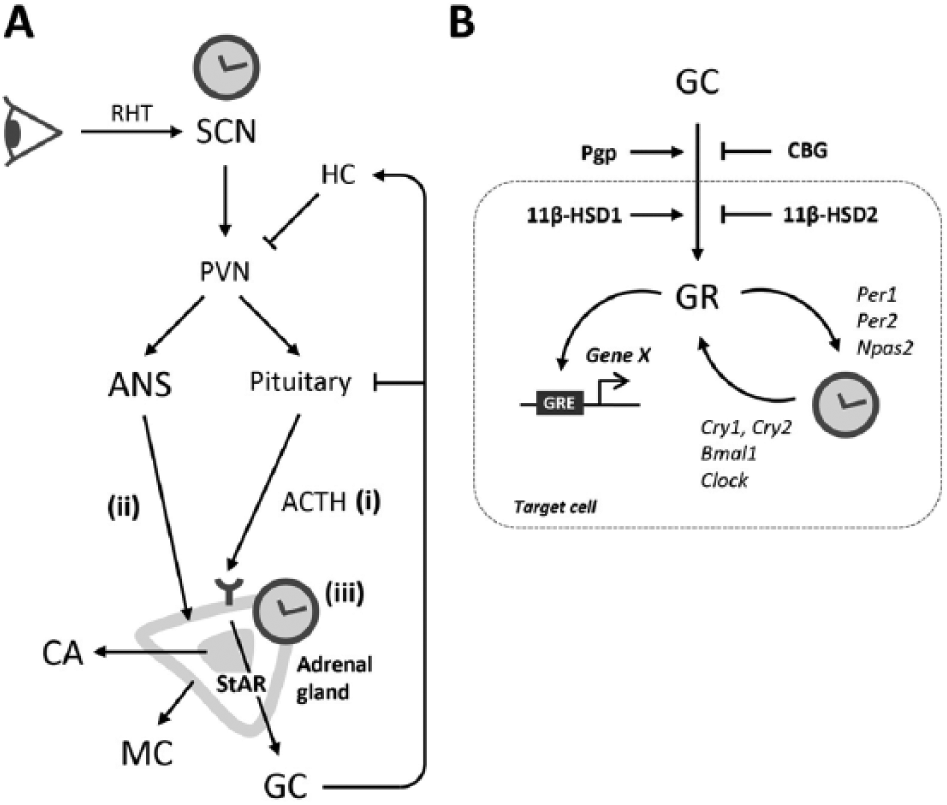
(A) Circadian regulation of GC rhythms. ACTH = adrenocorticotropic hormone; ANS = autonomic nervous system; CA = catecholamines; GC = glucocorticoids; HC = hippocampus; MC = mineralocorticoids; PVN = paraventricular nucleus; RHT = retinohypothalamic tract; SCN = suprachiasmatic nucleus; StAR = steroidogenic acute regulatory protein. See the text for explanations. (B) Circadian regulation of GC action. Prior to binding to glucocorticoid receptor (GR), bioavailability of GCs is regulated by blood levels of corticosteroid-binding globulin (CBG), local activity of GC-converting 11β-hydroxysteroid dehydrogenase (HSD) enzymes in target cells, and transport through the blood-brain barrier by P-glycoprotein (Pgp) (Quax et al., 2013). Notably, circadian variation has been reported in the expression of all these components and of GR, at least in some tissues (Hsu and Kuhn, 1988; Ando et al., 2005; Yang et al., 2006; Buren et al., 2007). Activated GRs modulate gene transcription through binding to glucocorticoid response elements (GRE) in regulatory regions of many genes, including clock genes
The circadian GC peak occurs a few hours before the onset of the active phase. Thus, the timing of GC peaks differs between diurnally and nocturnally active species (Lightman and Conway-Campbell, 2010). Upstream of GC secretion, circulating ACTH shows a similar circadian profile (Henley et al., 2009). CRH expression in the PVN is also rhythmic (Watts et al., 2004), but CRH phasing is not well synchronized with the expression of pro-opiomelanocortin (POMC, the precursor of ACTH) in the pituitary and circulating levels of GCs (Watts et al., 2004; Girotti et al., 2009), and in CRH knockout mice, a constant infusion of CRH is sufficient to rescue diurnal GC rhythms (Muglia et al., 1997). In a similar manner, the rhythmic release of ACTH is not essential for rhythmic GC secretion (Lilley et al., 2012), arguing against a primary regulation of circadian GC rhythms via the HPA axis. In contrast, recent in vitro findings suggest that ACTH is capable of resetting the adrenal clock rhythms (Yoder et al., 2014). Hence, HPA axis activity, although not critical for generation of daily GC rhythms, may contribute to adjustment of circadian phase of hormonal production.
A second communication pathway from the SCN to the adrenal gland runs via the autonomic nervous system (ANS). Since hypophysectomy only ablates GC rhythms in combination with adrenal denervation (Meier, 1976; Ottenweller and Meier, 1982), this may explain the above-mentioned dissociation between GC rhythms and upstream components of the HPA axis. SCN-derived ANS signals travel via preautonomic PVN neurons and sympathetic preganglionic intermediolateral neurons of the spinal cord, and they finally reach the adrenal gland, which is innervated by the splanchnic nerve (Buijs et al., 1999). Splanchnic nerve transsection in rats reduces the peak of plasma GC rhythms compared to sham-operated animals (Ulrich-Lai et al., 2006; Wotus et al., 2013). This autonomic innervation of the adrenal gland also provides a rapid photic input to the adrenal, with a 30-min light pulse being able to stimulate adrenal
Although the SCN is clearly essential for the rhythmic secretion of adrenal hormones in vivo, already in the 1960s, isolated and cultured Syrian hamster adrenal glands were demonstrated to have robust circadian rhythms of respiration and steroid secretion (Andrews and Folk, 1964). More recently, this property was also shown in adrenocortical Y-1 cells (Son et al., 2008). This initial observation occurred before the discovery of the molecular adrenal clockwork, but it is now well understood that clock gene expression in the rodent and primate adrenal gland, particularly in the adrenal cortex, is highly rhythmic (Bittman et al., 2003; Lemos et al., 2006; Oster et al., 2006a, 2006b; Fahrenkrug et al., 2008; Valenzuela et al., 2008) (Figure 1C), and a circadian pattern of expression applies to approximately 10% of the adrenal transcriptome, including genes involved in cholesterol uptake, regulators of steroidogenesis, and ACTH signaling (Oster et al., 2006a). In particular, the key steroidogenesis rate-limiting cholesterol transporter StAR is rhythmically expressed under the direct regulation of adrenal clock proteins (Ulrich-Lai et al., 2006; Son et al., 2008). Furthermore, sensitivity to ACTH appears to be regulated by a circadian gating mechanism in the adrenal clock (Oster et al., 2006b). In line with this, hypocortisolism and diminished expression of transcripts important for steroidogenesis and cholesterol transport were observed in
The molecular clock also controls the sympathoadrenal function and the release of catecholamines—norepinephrine and epinephrine—from the adrenal medulla (Curtis et al., 2007). Plasma and urinary catecholamine levels follow moderate diurnal oscillations, with elevated levels during the active phase, and their production is severely reduced in the
Rhythmic release has also been reported for the mineralocorticoid aldosterone (Wolfe et al., 1966; Doi et al., 2010). A critical role of the adrenocortical clock in regulation of blood pressure has been demonstrated in
Clinical Implications of Adrenal GC Rhythms
Rapid changes in the light-dark cycle or sleep rhythm disruption as occur during jetlag or shift work lead to a misalignment of internal circadian clocks and external time. In addition, conflicting zeitgeber information (e.g., by mistimed food intake) may further disrupt internal circadian synchrony. Such external and internal misalignment is associated with several health disorders, and GC rhythms play an important role in this context due to their capacity to reset tissue clocks via GR signaling (Balsalobre et al., 2000; So et al., 2009; Lamia et al., 2011) (Figure 2B). While the SCN pacemaker is insulated from direct GC feedback due to a lack of GR expression, GCs exert a stabilizing effect on peripheral tissue rhythms against external zeitgeber influence (Le Minh et al., 2001; Oishi et al., 2005; Kiessling et al., 2010; Pezuk et al., 2012).
Importantly, rhythmic GC secretion contributes to the homeostatic regulation of various biological processes, including long-term memory formation in mice and humans (Wagner and Born, 2008; Liston et al., 2013), hippocampal neurogenesis (Dickmeis and Foulkes, 2011), T-cell levels in human blood (Dimitrov et al., 2009), proliferation of murine hematopoietic progenitor cells (Kollet et al., 2013), and cell cycle activity in zebrafish (Dickmeis et al., 2007). Hence, the disruption of daily GC oscillations might be deeply linked with pathology. In the following sections, we will consider potential connections between circadian rhythms of GC production or GC sensitivity and a number of diseases (summarized in Table 1) and suggest therapeutic approaches to incorporate GC rhythm manipulation in the clinic.
GC Production and Signaling in Disease.

CBG = corticosteroid-binding globulin; EAE = experimental autoimmune encephalomyelitis; GC = glucocorticoid; OS = obese strain.
GC Rhythms and Mood Disorders
Several lines of evidence suggest a bidirectional link between the circadian clock, mood regulation, and mood disorders (reviewed in Wulff et al., 2010; Albrecht, 2013; McClung, 2013; Karatsoreos, 2014; LeGates et al., 2014). Disrupted circadian rhythms (e.g., as a result of shift work) facilitate the development of mood disorders and are associated with the disease severity in some susceptible individuals (Albrecht, 2013). Similarly, mice exposed to aberrant light conditions (an extremely short 7-h light-dark cycle) develop a depressive phenotype and suffer from hypercortisolism (LeGates et al., 2012). On the other hand, patients with mood disorders benefit from daily routines that include adherence to a strict bedtime (Frank et al., 2000). Polymorphisms in human clock genes and several clock gene deficiencies in mice are associated with sleep abnormalities and depressive or manic behavior (Kennaway, 2010; Albrecht, 2013). For instance, circadian clock-deficient mice, such as
HPA axis hyperactivity due to impaired negative feedback is one of the best documented clinical findings in patients with major depressive disorder (MDD) (Holsboer, 2000). Studies of circadian rhythms of GC hormones in depressive patients yielded somewhat mixed results (see Jarcho et al., 2013, for discussion). However, a general agreement is that MDD is associated with a flattening of diurnal GC rhythms (Krieger, 1975; Alesci et al., 2005; Heaney et al., 2010; Jarcho et al., 2013; Li et al., 2013b), which is explained by reduced brain sensitivity to negative GC feedback (de Kloet et al., 1998; Holsboer, 2000). Depressive patients show disruptions of daily rhythms of clock gene expression in blood cells (Li et al., 2013b) and various brain areas (Li et al., 2013a), which could to some extent be explained by altered GC rhythms (Segall et al., 2009). The phenomenon of the switch from depression to the opposite states of mania or hypomania and vice versa distinguishes bipolar disorder (BD) from all other psychiatric disorders (Salvadore et al., 2010). Disruptions of sleep (insomnia or hypersomnia) and circadian rhythms, including abnormal function of HPA axis, are core features of BD (Milhiet et al., 2011). In patients with seasonal affective disorder (SAD; also known as winter depression), early reports suggest that there are no significant changes of circadian GC production and GC-mediated negative feedback (James et al., 1986; Skwerer et al., 1988; Joseph-Vanderpool et al., 1991; Oren et al., 1996). However, using a constant routine protocol, phase-delayed cortisol and body temperature rhythms in SAD patients have been demonstrated (Avery et al., 1997). The normal inhibition of cortisol secretion during early sleep was absent in patients with schizophrenia, although the phasing and amplitude of the morning cortisol peak were not affected (Van Cauter et al., 1991). Additionally, in
In light of the association between altered GC rhythms and mood disorders, the following question arises: how may disturbed circadian rhythms of GCs contribute to the pathogenesis of these conditions? Several pathways might be involved (for a review, see McClung, 2013). (1) GC rhythms regulate circadian gene expression in the dentate gyrus and are necessary for the proliferation of neural progenitor cells in response to antidepressant treatment (Huang and Herbert, 2006; Gilhooley et al., 2011). Moreover, brain-derived neurotrophic factor (BDNF) and its receptor, TrkB, are rhythmically expressed in the hippocampus and are important for the action of antidepressant medications (Schaaf et al., 2000; Dolci et al., 2003). Importantly, BDNF loses its proliferative effects in the absence of daily rhythms in corticosterone (Pinnock and Herbert, 2008). (2) Disrupted GC rhythms may affect monoamine signaling in the brain. GCs modulate extracellular levels of monoamine neurotransmitters, including serotonin, noradrenaline, and dopamine, major targets of current antidepressant therapy (Mora et al., 2012). (3) Altered GC rhythms may affect mood via deregulation of metabolism. Indeed, epidemiological studies report that MDD often correlates with obesity, insulin resistance, and diabetes (Onyike et al., 2003; Pearson et al., 2010). Similarly, hypercortisolic Cushing’s patients with central obesity also have an increased risk of developing MDD (Pereira et al., 2010). (4) Further GC effects may be mediated via their suppressive effect on the immune system. Reduced sensitivity to inhibitory GC action, as seen in depressive patients, promotes secretion of proinflammatory cytokines (Alesci et al., 2005), which may in turn exacerbate depressive symptoms. In conclusion, although clinical data extensively document altered GC rhythms in psychiatric patients, we ought to note that the molecular pathways and causative links underlying the hormonal disruption are rather speculative and should be hence scrutinized in further studies using animal models.
GC Rhythms and Metabolic Disorders
Complex interactions are found between the circadian clock and metabolic pathways, and GC rhythms play an important role in this context (reviewed in Kovac et al., 2009; Nader et al., 2010; Bass, 2012). Human obesity is associated with a flattening of diurnal cortisol rhythms, although changes in overall GC levels are less consistent between studies. Elevated trough corticosteroid levels and dampened or disrupted GC rhythmicity have been reported in obese adults (Ljung et al., 1996; Rosmond et al., 1998; Ljung et al., 2000; Walker et al., 2000), genetically obese Zucker rats (Martin et al., 1978; Walker et al., 1992), and
GC Rhythms and Inflammatory Disorders
GCs play an essential role in the regulation of inflammation and are widely used as immunosuppressive agents for treating chronic inflammatory disorders, including autoimmune pathology (Busillo and Cidlowski, 2013). Proinflammatory cytokines, such as interleukin (IL)-6, tumor necrosis factor (TNF)-α, and IL-1β, potentiate the activity of HPA axis, exerting a feedback inhibition of immune responses and therefore minimizing excessive damage of host tissues (Wick et al., 1993). Daily rhythmic changes occur in basal activity of the immune system and are, at least partially, controlled by GC rhythms (reviewed in Lange et al., 2010; Cermakian et al., 2013). Corticosterone responses to an acute inflammatory stimulus (lipopolysaccharide injection) in rodents follow diurnal variations with a peak of sensitivity in the morning (Guo et al., 1996; Kalsbeek et al., 2012). Diurnal variation in immune reactivity is often preserved in autoimmune diseases; however, deregulated interactions along the immune-HPA axis lead to compromised ability of GCs to suppress pathological inflammation (Wick et al., 1993). Indeed, many patients with asthma and rheumatoid arthritis (RA) have worsening of symptoms in early morning (i.e., close to the peak of cortisol secretion) (reviewed in Cutolo et al., 2006; Burioka et al., 2010; Haus et al., 2012). For instance, it has been demonstrated that a peak of IL-6 secretion in arthritic parents precedes that of cortisol by less than 1 h (Perry et al., 2009). This paradoxical failure of GCs to suppress exacerbated inflammatory activity in the morning, at the peak of GC production, can be explained by impaired GC sensitivity of immune cells. Indeed, nocturnal asthma is associated with reduced GR binding activity and decreased responsiveness of blood leukocytes to GCs at early morning (Kraft et al., 1999). RA patients left untreated with GCs show impaired cortisol responses despite preserved ACTH secretion. This is consistent with a “relative adrenal insufficiency” in the presence of sustained inflammatory processes, allowing for excessive cytokine production in the late night (Cutolo et al., 2006). On the other hand, Lechner et al. (2000) found that increased GC production during the light phase in MRL/MP-
GC Rhythms and Cancer
Altered circadian rhythms, including deregulated GC rhythmicity, may promote cancer development (Mormont and Levi, 1997; Levi et al., 2010). Epidemiological studies have shown the increased cancer risk in shift workers with prolonged exposure to night-shift work (Schernhammer et al., 2001; Megdal et al., 2005; Stevens, 2009; Stevens et al., 2011). Similarly, experimental circadian disruption resulting from chronic jet lag, SCN lesion, or
In summary, disruption of daily GC rhythms, especially due to elevated trough levels, is commonly observed in various pathological conditions (Table 1). Further studies are necessary to dissect the contribution of central and peripheral (adrenocortical) clocks in this context.
GC Chronotherapy
Pharmacological correction of diurnal GC rhythmicity can be achieved by timed administration of exogenous GCs or by time-of-day-dependent suppression of abnormal overproduction of adrenal hormones. GC substitution is the mainstay of treatment in most hypocortisolemic disorders, regardless of their etiology (i.e., primary adrenal failure vs. secondary pituitary corticotrope or hypothalamic anatomical damage or functional impairment) (Coursin and Wood, 2002; Husebye et al., 2014). Major goals of chronic GC replacement are to substitute as much hydrocortisone (HC) as necessary to guarantee adequate stress reactivity and to avoid symptoms of chronic adrenal insufficiency (e.g., fatigue, nausea, or weight loss) (Husebye et al., 2014). Simultaneously, care must be taken to administer as little HC as possible to avert detrimental side effects of GC excess, such as weight gain (Huscher et al., 2009), neuropsychiatric symptoms (Dubovsky et al., 2012), osteoporosis (Lovas et al., 2009), and impaired glucose tolerance (Kwon and Hermayer, 2013). With respect to cumulative physiological cortisol secretion of approximately 3 to 12 mg/m2 per 24 h (Kraan et al., 1998; Purnell et al., 2004) and to the endogenous circadian rhythm of adrenal cortisol secretion, typical current clinical dosing regimens aim to substitute between 15 and 25 mg of HC per day in 3 divided doses. Roughly two-thirds of the cumulative HC dose is administered in the morning (e.g., 15 mg at 0700 h), while the remainder is split between 2 doses (e.g., 5 mg at 1200 h and 1700 h, respectively) (Husebye et al., 2014). Timing the last HC dose of the day sufficiently early (i.e., at least 5-6 h before bedtime) effectively prevents sleep disruption as a consequence of supra-physiological bedtime cortisol concentrations. The need for thrice-daily HC administration represents a significant challenge to therapy adherence. Furthermore, cortisol spikes after the midday and afternoon doses in addition to subphysiological cortisol concentrations preceding the administration of the next dose likely contribute to the excess mortality in patients with adrenal insufficiency on GC replacement therapy (Lovas et al., 2003; Debono et al., 2009). An oral dual-release HC preparation is being tested in the treatment of adrenal insufficiency (Lovas and Husebye, 2007; Johannsson et al., 2009; Johannsson et al., 2012). This preparation combines an outer coating layer that provides an immediate release of the drug and an extended-release HC core (Nilsson et al., 2014). This formulation has been shown to be safe and effective in more efficiently mimicking the natural circadian cortisol rhythm in patients with adrenal insufficiency (Johannsson et al., 2009; Johannsson et al., 2012). This improved approximation of the physiological circadian cortisol rhythm translated into improved quality of life, reduced body weight, decreased blood pressure, and improved glucose metabolism in a study involving 64 patients with primary adrenal insufficiency (Johannsson et al., 2012). A small study involving 7 patients with primary adrenal insufficiency moreover demonstrated that continuous subcutaneous HC infusion by programmable infusion pumps was sufficient to imitate the physiological circadian cortisol secretory rhythm, thus allowing for a substantial reduction in HC substitution requirements without adverse effects (Lovas and Husebye, 2007). Refining this paradigm, a pulsatile subcutaneous HC infusion pump has been validated to more closely mimic ultradian and circadian cortisol secretory pulsatility and showed that at comparable overall GC exposure, the pulsatile versus smooth pattern of cortisol secretion shapes a particular gene transcription pattern and subsequently specific neuroendocrine responses (Russell et al., 2014). This approach, although still experimental, may be especially beneficial in patients with pronounced morning fatigue, who are speculated to be more sensitive to the absence of the physiological cortisol peak shortly before awakening, and inherently cannot be addressed by oral HC substitution.
Desynchrony between external time and endogenous circadian clocks, as observed during jetlag or shiftwork, has been associated with fatigue and sleep disorders (Weingarten and Collop, 2013), an increased incidence of malignancy (Haus and Smolensky, 2013), and psychiatric disturbances (Katz, 2011). As mentioned above, numerous cell and animal studies suggest a pivotal role of adrenal GCs in the entrainment of central and peripheral rhythms and clocks (Balsalobre et al., 1998; Balsalobre et al., 2000; Sage et al., 2004; Kiessling et al., 2010). Investigating the impact of desynchronized secretory GC rhythms on human glucose metabolism, Plat et al. (1999) found a significant increase in glucose and insulin levels when HC levels were artificially elevated at night. To examine the impact of modulating the adrenal glucocorticoid secretory rhythm on recovery from jetlag, Kiessling et al. (2010) induced a phase shift in GC secretion by timed administration of the steroidogenesis inhibitor metyrapone in mice. Depending on the direction of the induced shift relative to the external time shift, metyrapone treatment either accelerated or decelerated the duration of jet lag, as manifested by the speed of reentrainment of behavioral and clock gene transcriptional rhythms (Kiessling et al., 2010). This evidence complements the Plat et al. (1999) study, which demonstrated such GC shifting to be feasible in humans. Thus, the capacity of adrenal glucocorticoids to stabilize or reentrain circadian rhythms represents an exciting therapeutic approach to develop treatment strategies for human jetlag and circadian disruption-associated morbidity.
Conclusion
Adrenal clocks and adrenal-derived endocrine rhythms have important functions in the regulation of physiological processes, and adrenal circadian disruption may be an important step in the development of a number of heterogeneous diseases. While we have gained some insight into the regulation of adrenal rhythms by the SCN, it will be important to better describe the interaction of circadian and noncircadian factors in the control of adrenal activity. On the other hand, the role of the adrenal hormones as resetting signals for other tissues deserves further attention, particularly with regard to the development of complex pathologies such as major depression or various immune disorders. Furthermore, the better accessibility of peripheral clocks may actually make them more attractive targets for chronotherapeutic approaches than the SCN pacemaker itself.
Footnotes
Acknowledgements
HO is a Lichtenberg fellow of the Volkswagen Foundation. This work was supported by a project grant of the German Research Foundation (DFG). VO is supported by a grant from the German Ministry of Education and Science (BMBF).
Author Contributions
AL performed experiments. AL & HO analysed the data. AL, RD, VO and HO wrote the manuscript.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
