Abstract
Sleep has strong links to the symptomology of fibromyalgia syndrome (FMS), a diffuse musculoskeletal pain disorder. Information about the involvement of the circadian clock is, however, sparse. In this study, 1548 individuals with FMS completed an online survey containing questions on demographics, stimulant consumption, sleep quality, well-being and subjective pain, chronotype (assessed by the Munich ChronoType Questionnaire, MCTQ), and FMS impact. Chronotype (expressed as the mid-sleep-point on free days, corrected for sleep deficit on workdays, MSFsc) significantly correlated with stress-ratings, so-called “memory failures in everyday life,” fatigue, FMS impact, and depression but not with anxiety. When chronotypes were categorized into 3 groups (early, intermediate, late), significant group differences were found for sum scores of perceived stress, memory failures in everyday life, fatigue, FMS impact, and depression but not anxiety, with late chronotypes being more affected than early chronotypes. Sleepiness ratings were highest in early chronotypes. Challenges of sleep quality and subjective pain were significantly increased in both early and late chronotypes. The results show that according to their reports, late chronotypes are more affected by fibromyalgia.
Fibromyalgia syndrome (FMS) is a musculoskeletal pain disorder that affects 2% to 5% of the population and shows an increased female-to-male ratio (Theadom and Cropley, 2008). Malfunction of central pain processing (Arnold et al., 2011) with abnormal cortisol levels (Riva et al., 2010) and other biomarkers (Geenen and Bijlsma, 2010) is believed to be responsible for increased pain. A study comparing melatonin, cortisol, and core body temperature between female FMS patients and female controls found no differences between the 2 groups (Klerman et al., 2001).
This report presents preliminary results from a large online survey on how FMS patients cope in everyday life, integrating, for the first time, individual circadian phase of entrainment (chronotype; Roenneberg et al., 2003). Between September 2010 and March 2011, 1548 FMS patients (91.5% females) completed (without compensation) the Perceived-Stress-Scale (PSS; Cohen et al., 1983), Everyday-Memory-Questionnaire (EMQ; a measure of so-called “memory failures in everyday life”; Royle and Lincoln, 2008), Fatigue-Severity-Scale (FSS; Krupp et al., 1989), Fibromyalgia-Impact-Questionnaire (FIQR; Burckhardt et al., 1991), Epworth-Sleepiness-Scale (ESS; Johns, 1991), Hospital-Anxiety-and-Depression-Scale (HADS; Zigmond and Snaith, 1983), and the Munich ChronoType Questionnaire (MCTQ; Roenneberg et al., 2003). The study was approved by the University of Surrey Ethics Committee. For 987 participants (mean age 47.16 years ± 10.44; 92% females), we received a complete data set to calculate chronotype (MSFsc, time of mid-sleep on free days, corrected for sleep deficit on workdays). Data from the PSS, EMQ, FSS, FIQR, ESS, and HADS were analyzed using sum scores. Control populations were produced by age and sex matched independent randomized seeds from the general Central European MCTQ database (Roenneberg et al., 2003), showing no significant differences in the chronotype distribution (MSFsc). We divided the chronotype distribution into 3 equally sized groups (early types: n = 329, mean MSFsc 2.1 ± 0.9; intermediate types: n = 330, mean MSFsc 3.6 ± 0.31; and late types: n = 328, mean MSFsc 5.35 ± 1.41). Late chronotypes were significantly younger (F(2,967) = 13.4, main group effect p < 0.001, 1-way ANOVA; mean ages: early types 49.42 years ± 9.79, intermediate types 47.62 years ± 10.25, late types 45.28 years ± 10.48). We therefore used age and sex as covariates in the statistical analysis, in addition to employment status (employed: yes/no) and shift-work status (shift-work: yes/no). Data analysis was performed using PASW Statistics 18.0 (IBM, Somers NY, USA) for Macintosh. Parametric and nonparametric statistical analyses were performed as appropriate.
The results showed significant positive correlations between chronotype (MSFsc) and the following parameters: PSS (r = 0.218, p < 0.001); EMQ (r = 0.158, p < 0.001); FSS (r = 0.091, p = 0.014); FIQR (r = 0.174, p < 0.001); depression (HADS; r = 0.227, p < 0.001; 2-tailed Spearman correlations). Chronotype grouping revealed significant group-differences, again for the same set of parameters: PSS (p < 0.001, Fig. 1B); EMQ (p < 0.001); FSS (p = 0.004); FIQR (p < 0.001, Fig. 1A); depression (HADS, p < 0.001, Fig. 1C). These results indicate that late chronotypes are more affected by FMS in these parameters than early chronotypes. Sleepiness (ESS; p = 0.039) was highest in early chronotypes, and sleep quality was lowest in both early and late chronotypes (p < 0.001). Subjective pain was significantly higher in early and late chronotypes (p < 0.001). Table I shows sum scores in the 3 chronotype groups (all Kruskal-Wallis test). Notably, anxiety (HADS) did not differ between chronotypes, either for individual MSFsc values or between chronotype groups.
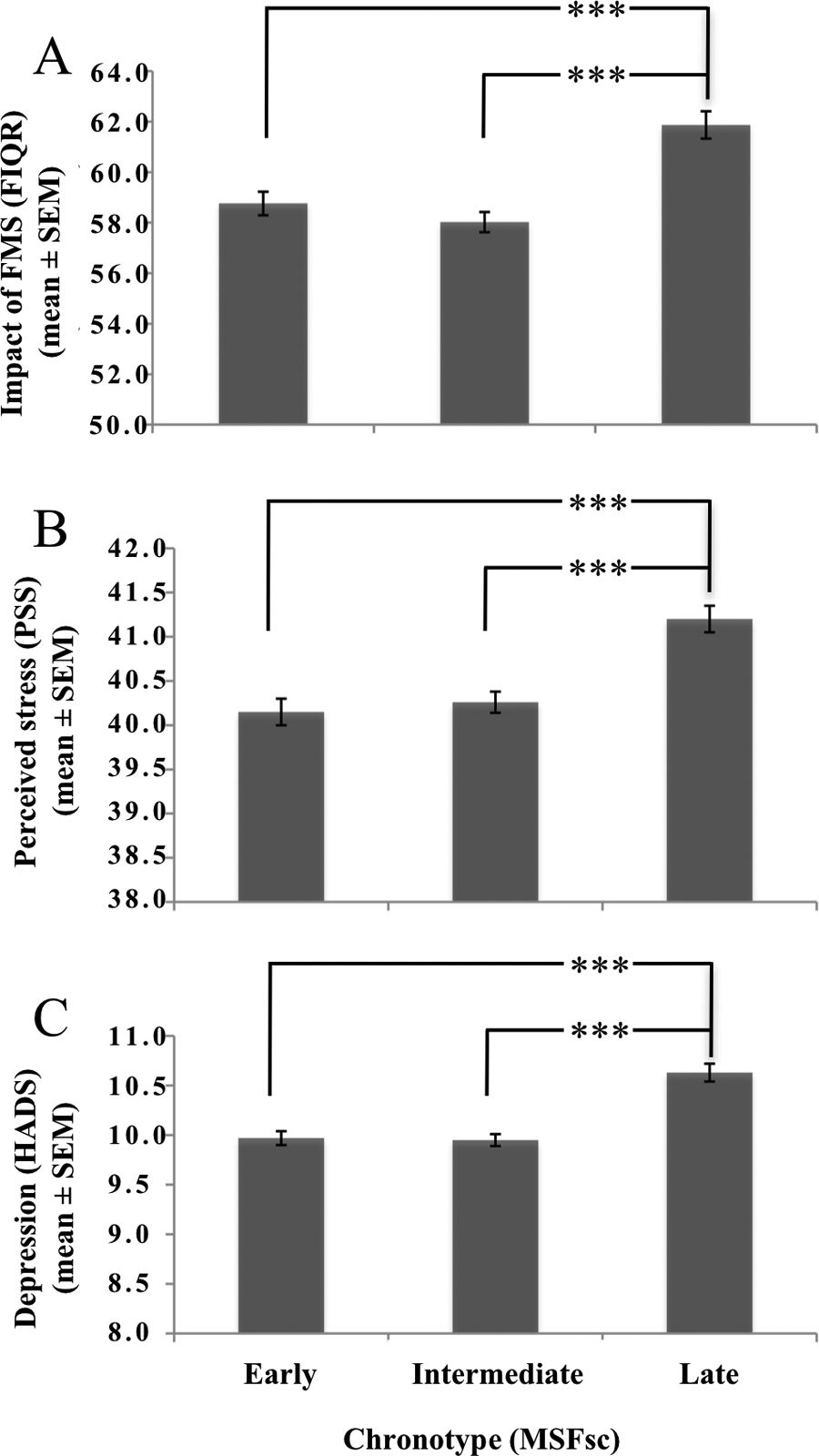
Mean ratings (± SEM) of the 3 chronotype groups (329 early chronotypes with MSFsc ≤ 3.03, 330 intermediate chronotypes with MSFsc 3.04 to 4.11, 328 late chronotypes with MSFsc ≥ 4.12) for the (A) Fibromyalgia Impact Questionnaire (FIQR), (B) Perceived Stress Scale (PSS), and (C) Hospital Anxiety and Depression Scale (HADS), for depression. ***p < 0.001 (post hoc analysis using Mann-Whitney U test, corrected for multiple testing).
Sum scores for the outcome measures (±SD) for 239 early, 258 intermediate, and 228 late chronotypes
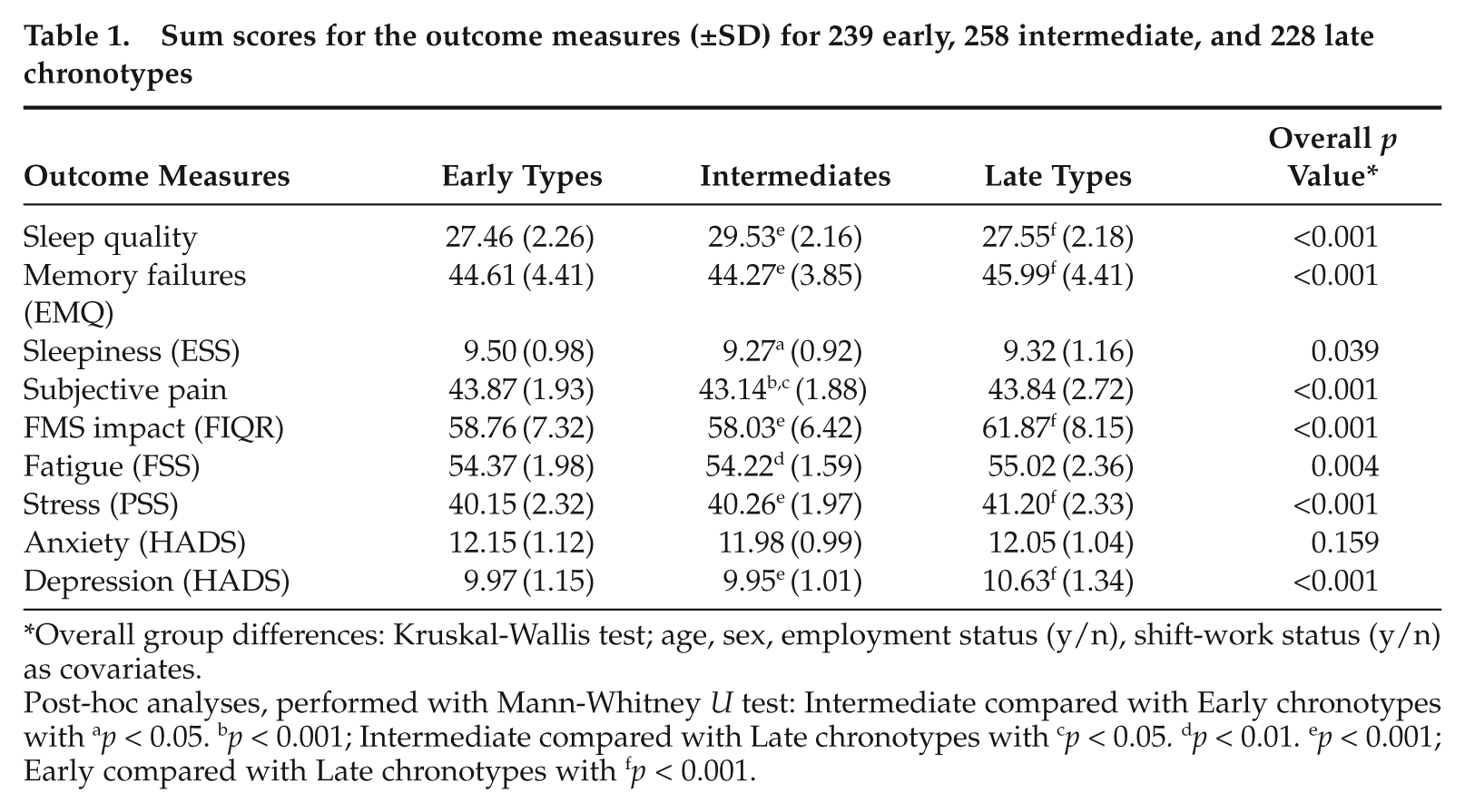
Overall group differences: Kruskal-Wallis test; age, sex, employment status (y/n), shift-work status (y/n) as covariates.
Post-hoc analyses, performed with Mann-Whitney U test: Intermediate compared with Early chronotypes with ap < 0.05. bp < 0.001; Intermediate compared with Late chronotypes with cp < 0.05. dp < 0.01. ep < 0.001; Early compared with Late chronotypes with fp < 0.001.
Fig. 1 shows the results for FIQR, PSS, and depression (HADS). Post hoc analysis (Mann-Whitney U test, corrected for multiple testing) showed a significant difference for the parameters FIQR, PSS, and depression (HADS) between early and late chronotypes (p < 0.001), and between intermediate and late chronotypes (p < 0.001).
Pain and sleep disturbances have been shown to be central to the FMS symptomology (Theadom and Cropley, 2008), which is corroborated by our results. In addition, we show here for the first time that late chronotypes are most affected by FMS. This result cannot be explained by an age-dependent increase in FMS symptomology (Wolfe et al., 1995), since late chronotypes in our study were significantly younger than early chronotypes. Both early and late chronotypes rated their sleep quality significantly worse than intermediate chronotypes, and early chronotypes also showed higher sleepiness ratings. The fact that subjective pain was indifferent between early and late chronotypes whereas late chronotypes rated highest on most of the other scales could be explained by the higher depression levels in late types, thereby amplifying FMS-related symptoms. Late chronotypes have already been shown to suffer more from bipolar disorders (Wood et al., 2009), headaches (Bruni et al., 2008), seasonal depression (Natale et al., 2005), and depression (Levandovski et al., 2011).
The current study is preliminary and the first to investigate the association between FMS and chronotype. Future studies are necessary to clarify whether late chronotypes per se are more susceptible to FMS-related symp toms or whether the discomfort of FMS challenges sleep quality, thereby delaying sleep onset, as it is the case in restless legs syndrome (Walters and Rye, 2009). Thus, FMS could artificially make affected individuals later chronotypes (note that our study population lacked extreme early types).
Good thermore gulation is important for restorative sleep and health (Sack et al., 2007). Although an earlier study with relatively few participants (Klerman et al., 2001) found no differences in the timing of the core body temperature trough, there is evidence of abnormal skin microcirculation in FMS patients (Jeschonneck et al., 2000). Future studies should therefore definitively include objective temperature measurements (both core and periphery). Current FMS management recommends multilevel treatment including exercise, cognitive-behavioral therapies, and medication (Carville et al. 2008). Sleep timing adjusted more to chronotype than to social time, as well as controlled thermoregulation for maintaining restorative sleep, could be a potentially effective addition to this multilevel treatment.
Although significant, the differences in scores between chronotypes shown here are small, and so far we cannot clearly judge their clinical value. These scores are not tools for clinical diagnostics but rather are designed for early risk identification. Subtle differences in these subjective scores can be given more clinical relevance by objective methods.
Unfortunately, it was impossible to link chronotype to medication use since individual consumption was highly variable (1143 patients, i.e., 73.8%, reported taking more than 1 medicine). The majority (81.2%) of participants reported at least 1 comorbid condition, as is typical for FMS, with arthritis (17%), irritable bowel syndrome (15.2%), depression (12.3%), and asthma (12.1%) being the most frequent. Due to the low number of participants in each condition and chronotype group, it was impossible to associate chronotype and comorbid conditions.
Individualized medicine is highly important for developing more effective treatments, and considering internal time (chronotype) is a simple and promising first step toward this goal. The cross-sectional design of our study is another limitation. Yet, the high number of subjects in our study and the proven validity of the questionnaires strengthen the indication that circadian timing is associated with experiencing the symptom of FMS. Future studies are important to validate the results presented here and especially to further clarify the interplay of subjective and objective pain measurements, sleep timing, and individual chronotype.
Footnotes
Acknowledgements
We thank all study participants and especially our reviewers for their fruitful and helpful comments to our manuscript. T.K. has been funded by DFG (German Research Foundation). A.T. has been co-funded by ABI Rehabilitation Management Ltd, Auckland, New Zealand.
The authors have no conflicts of interest to declare.
