Abstract
Diisocyanates have long been a leading cause of occupational asthma. As control often relies on personal protective equipment and there is the potential for skin uptake, biological monitoring is often used to assess worker exposure. Current routine biological monitoring methods do not distinguish between a diisocyanate and the corresponding diamine exposure in urine samples; therefore, a specific urinary biomarker is desirable. Urine samples were obtained from a group of workers exposed to methylenediphenyl diisocyanate (MDI) where aerosol generation was unlikely. Lysine conjugates of MDI were extracted from urine by solid phase extraction; analysis was performed by liquid chromatography tandem mass spectrometry. Acetylated MDI-lysine (acMDI-Lys) conjugates were detected in 73% of samples tested from persons with exposure to MDI compared to 93% of samples that were positive for methylene dianiline (MDA) in hydrolysed urine. There was a weak but significant positive correlation between the two biomarkers (r2 = 0.377). This is the first report detecting and quantifying acMDI-Lys in the urine of workers exposed to MDI, and acMDI-Lys may be a useful non-invasive biomarker in discriminating between MDI and MDA exposures.
Keywords
Introduction
Diisocyanates are a group of chemicals with two highly reactive isocyanate (N = C = O) groups that react readily with polyols to form polyurethanes. Because of their reactivity, they are classified as both respiratory and skin sensitisers in Great Britain (HSE, 2020) and have been a leading cause of occupational asthma for many years (HSE, 2021). Diisocyanates have a wide range of uses including in polyurethane foams, spray paints, surface coatings and adhesives. Methylenediphenyl diisocyanate (MDI) and toluene diisocyanate (TDI) make up approximately 90% of the total worldwide isocyanate market (Allport et al., 2003). Common methods for the biological monitoring of exposure to diisocyanates are based upon the analysis of the amine derivatives in urine or blood samples after hydrolysis (Cocker, 2007; Tinnerberg et al., 2014). The use of hydrolysed diamines in urine as biomarkers of diisocyanates has been widely adopted by international bodies setting guidance values (ACGIH, 2022; DFG, 2021; HSE, 2020). This so called ‘total body burden’ measurement does not distinguish between direct diamine exposure and diisocyanate exposure as analysis is based on detection of the free diamine after hydrolysis of any diisocyanate conjugates. Because of the potential for confounding, this method may over-estimate diisocyanate exposures because of other sources of the free diamine. For example, in the case of MDI, methylene dianiline (MDA) is both a starting material for the production of MDI (Van Den Berg et al., 2012) and is an industrial chemical in its own right, used as a cross-linking agent for epoxy resins, as a corrosion inhibitor, as an antioxidant and curative agent in rubber, and to prepare azo dyes (NTP, 2021). In addition, MDA exposure can also result from atmospheric hydrolysis of MDI during aerosol generating activities such as spray painting and foam blowing (Jones et al., 2017).
The metabolism of diisocyanates is not fully understood at present; however, MDI conjugation to human albumin in vitro has been reported (Wisnewski et al., 2013); a recent review (Schupp and Plehiers, 2022) has collated work in this area, outlining the known mechanisms of MDI absorption, distribution, metabolism and excretion and detected biomarkers (both in vitro and in vivo). Lysine residues have been reported as the predominant binding sites on human serum albumin (HSA) for TDI and MDI (Hettick and Siegel, 2012; Hettick et al., 2012; Mhike et al., 2013). This knowledge has led to research into the discovery of new biomarkers of diisocyanate exposure. A method to extract and quantify isocyanate-specific conjugates with albumin was first reported in rats (Kumar et al., 2009). Sabbioni and co-workers built on this work and reported MDI specific HSA conjugates in plasma from exposed workers; acetylated MDI-Lysine (acMDI-Lys) and MDI-Lysine (MDI-Lys) conjugates were detected in exposed workers from a chemical company and a construction site. MDI-Lys conjugates were found in over 60% of samples tested in both companies (Sabbioni et al., 2010). The presence of the MDI-lysine conjugates in albumin isolated from human blood plasma samples of persons undergoing Specific Inhalation Challenge (SIC) for MDI was also reported (Sabbioni et al., 2016). A separate study also reported MDI-Lys conjugates that were detected in MDI workers’ sera samples after SIC for MDI (Luna et al., 2014).
Given the invasive nature of blood sample collection and ethical considerations, an alternative analytical approach is desirable. Human biomonitoring using urine samples offers many advantages. Urine is a primary excretory pathway, non-invasive, and a suitable matrix for the detection of a wide range of both organic and inorganic compounds and metabolites from occupational and environmental exposure (Polkowska et al., 2004; Smolders et al., 2009). Despite these advantages, care needs to be taken in collecting the sample to avoid contamination (from skin or clothing) and to collect the sample at the right time post-exposure. Specific urinary metabolites of MDI exposure have not yet been reported in humans, although detection of dilysine-MDI in the urine of MDI-exposed mice has been reported (Wisnewski et al., 2019). Based on several reports detecting acMDI-Lys in plasma samples of workers, we have investigated whether this biomarker was excreted in human urine as this would have the potential to provide a less invasive, straightforward method to investigate MDI exposure specifically.
Materials and methods
Samples
Urine samples were obtained from 55 workers using MDI in a casting process. All samples were previously analysed for urinary MDA (uMDA) by gas chromatography mass spectrometry (GCMS) after acid hydrolysis (Cocker et al. 2017). Control samples (n=20) were obtained from non-exposed workers. All samples were collected post-shift and stored at −20°C prior to analysis. All urine samples were provided by workers giving informed consent as per publication HSG167 (HSE Health and Safety Executive, 1997); such consent allowed reuse of samples for method development purposes. Ethical approval to analyse control samples (from pesticide sprayers) was obtained from University of Sheffield Medical School Research Ethics Committee (HSL29) in February 2022.
Chemicals and instrumentation
Custom synthesised acMDI-Lys standard was purchased from HelloBio Ltd (Avonmouth, Bristol, UK). The synthesis route was based on that described previously (Kumar et al., 2009) (see Supplementary Material, S1, for method details and structural confirmation). Deuterated (d8) MDA internal standard was purchased from QMx (Essex, UK). All other reagents were of analytical grade (see Supplementary Material, S1, for details of suppliers).
Analysis was performed on an Applied Biosystems (AB Sciex, Warrington, UK) 4500 QTRAP mass spectrometer interfaced with a HPLC Agilent 1260 (Agilent Technologies, Manchester, UK) system equipped with an autosampler. Solid-phase extraction (SPE) sample clean-up was performed with a Gilson ASPEC (Middleton, WI, USA) GX-271 system using Agilent Bond Elut Plexa PCX cartridges (3 mL, 60 mg).
Sample treatment
Sample aliquots (2 mL) were simply spiked with internal standard solution (100 μL of d8-MDA (50 μ
Statistical analysis
Simple linear regression of acMDI-Lys on uMDA was carried out to estimate the strength of the relationship between the two biomarkers. Bland–Altman analysis was used to investigate the agreement between the two measures by focussing on the differences between them. The Bland–Altman plot summarises this analysis by showing the difference between the two measures plotted against the average of the two measures. The plot indicates whether there is any evidence of a systematic bias in one measure compared to the other, and the estimated agreement interval specifies the interval within which 95% of the differences are expected to occur. The Bland–Altman plot was adjusted for the linear relationship between the paired differences and the paired averages. Stata v17 statistical software (StataCorp. 2021. Stata Statistical Software: Release 17) was used for the analysis.
Results and discussion
AcMDI-Lys tuned well in both positive and negative electrospray. Here, we present results in positive electrospray as this enabled the use of d8-MDA as an internal standard. Two transitions for acMDI-Lys were used; m/z 413.1/241.4 for quantitation and m/z 413.1/106.0 for confirmation. Calibration curves (see example in Supplementary material S2) were linear over the range 10–160 nmol/L (least squares regression coefficient >0.99). The intra-assay and inter-assay coefficients of variation (CV) for acMDI-Lys were 5.5% (n = 10) and 16.9% (n=38 over four runs), respectively, at a nominal 40 nmol/L. The limit of quantitation (LoQ) was set at 5 nM based on observable peaks at a 10:1 signal-to-noise ratio. The acMDI-Lys adduct was not detected in the control samples (n=20), which positively suggested the specificity of the method to detect the target exposure (MDI). Spiked samples were stable for at least three days at all storage conditions tested (ambient, refrigerated (+4°C), and frozen (−20°C)). Representative chromatograms of a standard and a sample from an exposed worker are shown in Figure 1. Chromatograms for a calibration standard (A) and an actual sample (B) showing peaks for d8-MDA internal standard (18.3 min), and acetyl-MDI-lysine (18.7 min).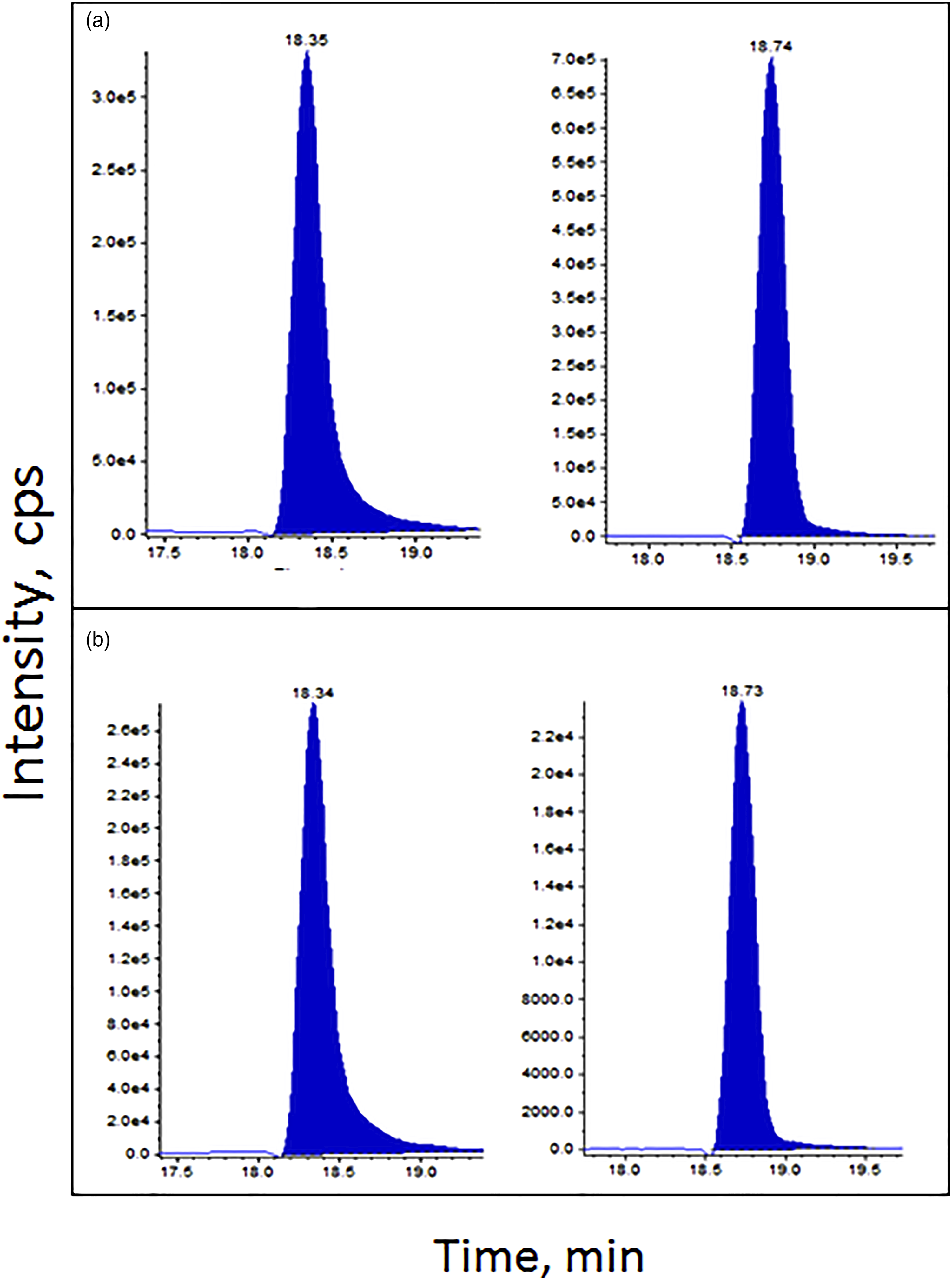
Lysine conjugates were detected as acMDI-Lys in 40 out of 55 (73%) samples from the MDI-exposed workers, while uMDA was detected in 51 samples out of 55 samples (93%) above the limit of quantification (5 nmol/L). Concurrent air monitoring data were not available but previous measurements (performed according to MDHS25/4, (HSE, 2014)) indicated total MDI exposures in the range of 0.01–1.46 μg/m3 (expressed as total NCO), that is, well within the Workplace Exposure Limit for isocyanates (20 μg/m3, 8 h-TWA, (HSE, 2020)). The high detection frequency in urine, despite the low airborne concentrations, might indicate for the possibility of skin absorption. In this study, the samples were obtained from workers handling MDI as part of a casting process. We do not have further details from this site but previous work investigating diisocyanate exposure in two other companies using MDI in casting processes found no evidence of exposure to aerosols or in situ formation of MDA, indicating that the workers of the present study were likely only exposed to MDI vapours (Jones et al., 2017).
From Figure 2, a weak, but statistically significant (p < 0.001) positive correlation (R2 = 0.377) was observed between acMDI-Lys and uMDA. Although it might be expected that acMDI-Lys would be completely hydrolysed to MDA during the GCMS sample preparation for uMDA, the presence of other conjugates and metabolites (for example, acetylated MDA (Schütze et al., 1995), dilysine-methylenediphenyl diisocyanate (Wisnewski et al., 2019)) and inter-individual variability in these might confound the correlation. Our samples were also analysed for MDI-lysine (without acetylation) but the levels were very low (data not shown), indicating that this was not a significant biomarker, at least in these particular samples. The variability in human acetylator phenotypes (Kadlubar et al., 1992; Salazar-González et al., 2019) will impact the extent of formation of acMDI-Lys in individual workers. It is also possible that different routes of exposure (skin uptake versus inhalation) may impact the metabolite profile. In addition, the expected extended half-life for albumin-bound acMDI-Lys (given that HSA has a half-life of ∼19 days, (Mishra and Heath, 2021) compared to MDA may also reduce the likelihood of a strong correlation. The half-life of excretion in urine will need to be investigated further before an optimised sampling strategy can be proposed. Linear regression correlation of urinary methylene dianiline (uMDA) and urinary acetyl-MDI-lysine (acMDI-Lys) (line: y = 0.0941x + 4.2655, R2 = 0.377, N=55). Results below the limit of quantitation (LOQ) represented as half the LOQ (2.5 nmol/L).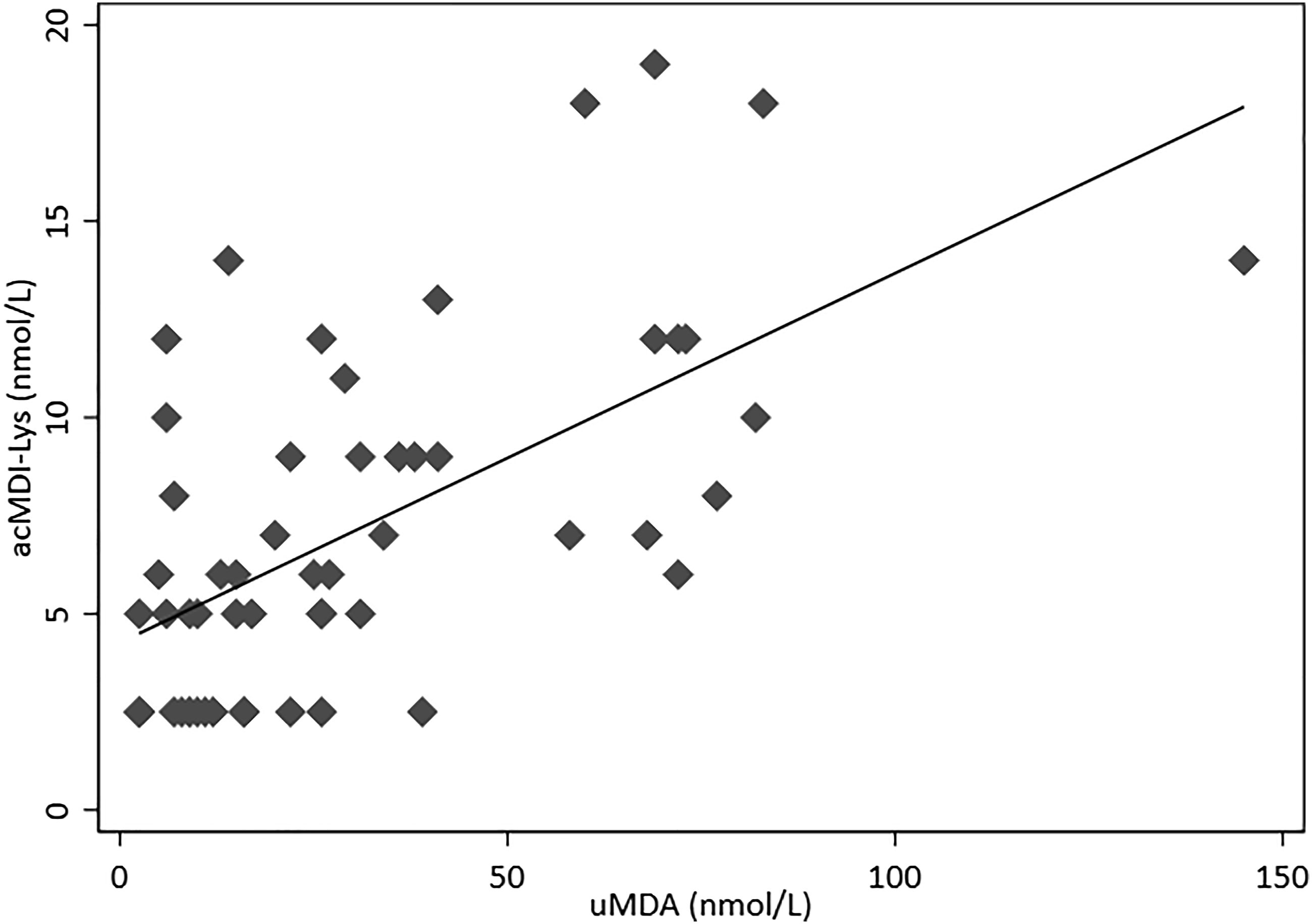
The Bland–Altman plot (Figure 3) demonstrates that there was no systematic bias and that only three out of the 55 samples (5.5%) were outside the limits of agreement, indicating a good agreement between the two biomarkers. The limits of agreement in the Bland–Altman plot were adjusted for the linear trend between the paired difference and the paired average of the two measures. Bland–Altman plot comparing urinary methylene dianiline (uMDA) and urinary acetyl-MDI-lysine (acMDI-Lys), showing good agreement between the two biomarkers. Agreement between the biomarkers: Mean difference = 6.96 + 1.61•Average; Limits +/− 2.46•(3.31 + 0.08•Average).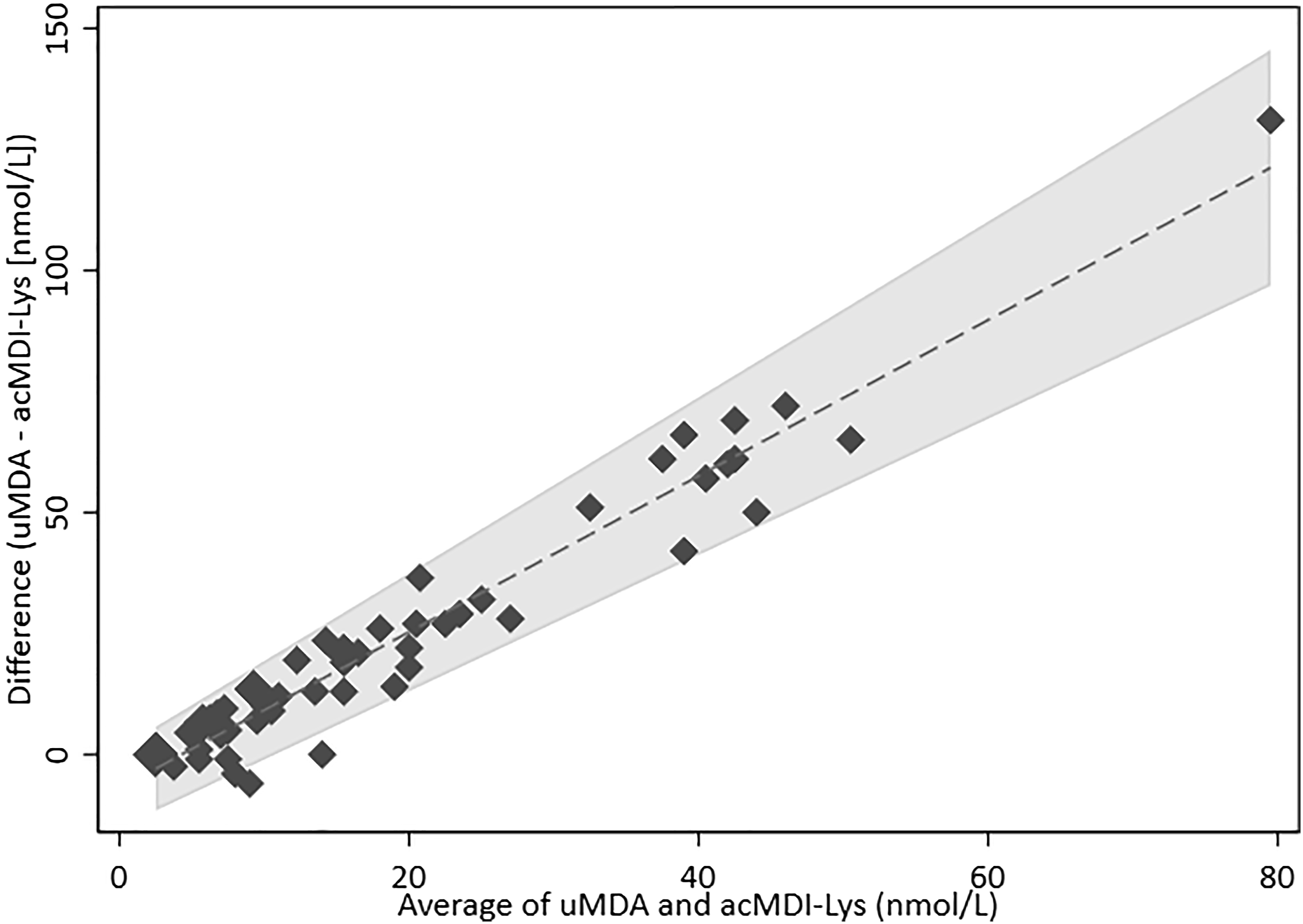
The positive correlation, the good agreement demonstrated by the Bland–Altman plot and the lack of positive acMDI-Lys in control samples provided evidence that urinary acMDI-Lys may be a suitable and specific biomarker for MDI exposure. However, the data we report were from a single company. More evidence is needed reflecting a wider range of workplaces and exposure scenarios, and it is necessary to also determine that MDA-exposed workers do not show significant levels of acMDI-Lys conjugates in order to promote this biomarker as a genuinely specific, non-invasive biomarker for MDI exposure. Whilst the use of hydrolysed diamines will continue to be a valuable tool in occupational exposure assessment for diisocyanates, the additional measurement of acMDI-Lys may also be useful, particularly where there may be confounding from concurrent MDA exposure.
Conclusions
The study has shown for the first time, the detection of acetyl-MDI-lysine conjugates in urine from potentially exposed workers. Previous studies have reported acetyl-MDI-lysine in plasma samples. Despite a weak correlation between acetyl-MDI-lysine and uMDA due to the complex and less understood metabolic process involved in the formation of MDA and conjugates in urine from MDI, the results of this study suggest that urinary acetyl-MDI-lysine may prove to be a specific non-invasive biomarker for MDI and hence provide a better indicator for exposure risk. However, more work is required to support its use as a specific biomarker for MDI exposure, including demonstrating its use in other exposure scenarios, establishing a reference value and defining a sampling strategy.
Supplemental Material
Supplemental Material - Methylenediphenyl diisocyanate lysine conjugates in the urine of workers exposed to methylenediphenyl diisocyanate
Supplemental Material for Methylenediphenyl diisocyanate lysine conjugates in the urine of workers exposed to methylenediphenyl diisocyanate by Kenneth C Nwoko, Laura Kenny and Kate Jones in Toxicology and Industrial Health
Supplemental Material
Supplemental Material - Methylenediphenyl diisocyanate lysine conjugates in the urine of workers exposed to methylenediphenyl diisocyanate
Supplemental Material for Methylenediphenyl diisocyanate lysine conjugates in the urine of workers exposed to methylenediphenyl diisocyanate by Kenneth C Nwoko, Laura Kenny and Kate Jones in Toxicology and Industrial Health
Footnotes
Acknowledgements
The authors should like to thank Dr Anne-Helen Harding, HSE for statistical advice and analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication, and the work it describes, was funded by the Health and Safety Executive (HSE) of Great Britain. Its contents, including any opinions and/or conclusions expressed, are those of the authors alone and do not necessarily reflect HSE policy.
Informed Consent
All urine samples were provided by workers for MDI biomarker analysis under written informed consent as per publication HSG167 (HSE 1997); such consent allows reuse of samples for method development purposes. Ethical approval to analyse control samples (from pesticide sprayers) was obtained from University of Sheffield Medical School Research Ethics committee (HSL29) in February 2022.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
