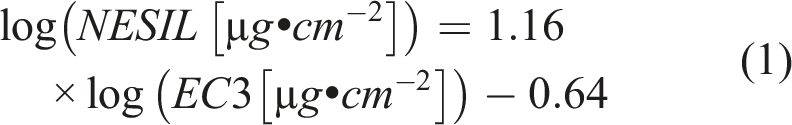Abstract
Flexible polyurethane foams (PUF) are used in many consumer products. PUF may contain trace levels of aromatic diamine impurities that could represent a potential health risk. The risk associated with sleeping on a PUF mattress was evaluated. Toxicity benchmarks for sensitization and non-cancer endpoints were derived from the respective points-of-departure using standard assessment factors. For the cancer endpoints, toxicity benchmarks were derived from the 25th-percentile values of animal studies. Recently published emission and migration data allowed to link exposure with the CertiPURTM voluntary quality limits of ≤5 mg.kg−1 for 2,4-toluene diamine and 4,4’-methylene dianiline in PUF. Using conservative exposure scenarios, lifetime-average daily internal doses from the combined inhalation and dermal exposures were calculated. Margins of safety for non-cancer and sensitization endpoints were >104. The theoretical excess cancer risk was ≤1.5 × 10−7. It is concluded that sleeping on a mattress that satisfies the CertiPUR limit value does not pose undue risk to consumers.
Keywords
Introduction
Flexible Polyurethane Foam (PUF) is produced from the reaction of long-chain polyols with diisocyanates in the presence of water, catalysts, and additives. The polyols and diisocyanates make up ca. 95% of the formulation (Brereton et al., 2019; Szycher, 2012). The polyol is typically a polyether polyol and the diisocyanates are either toluene diisocyanate (TDI) or methylenediphenyl diisocyanate (MDI). PUF is manufactured in industrial settings for subsequent use in consumer products such as furniture, automotive interiors, bedding, and carpet underlay.
The main polymerization reaction takes place between the hydroxyl groups of the polyols and the isocyanate (NCO) groups of TDI or MDI, forming the polyurethane backbone of the PUF. Some of the NCO groups react with water to produce CO2, which facilitates foaming. This hydrolysis process converts the NCO groups into transient amino groups. Since the reactivity of NCO with amino groups is approximately 104 times greater than with hydroxyl groups (Schupp and Plehiers, 2022), various polyurea species are formed and become part of the PUF.
Because of the greater reactivity of NCO groups with amino groups compared to hydroxyl groups, it was once assumed that no residual unbound diamines would be present in PUF (Hall et al., 2001; Hillier et al., 2003). However, it is now generally accepted that, under certain circumstances, residual trace quantities of toluene diamine (TDA) and methylene dianiline (MDA) may be present in fresh PUF at detectable (low mg.kg−1) levels. The residual diamine concentrations have been found to decay over time in normal conditions (Krämer-Lucas et al., 2001).
Since 2,4-TDA and 4,4’-MDA meet the classification requirements in the United Nations Globally Harmonized System (GHS) (UN, 2021) of Class 1B carcinogens (EU Commission, 2008), the CertiPURTM voluntary industry certification scheme was introduced and set a limit for the presence of these diamines in PUF at ≤5 mg.kg−1 (EUROPUR, 2021). This limit was based on risk assessment (RA) work done in the early 2000s. Analytical methodology has been developed that enables establishing relationships between the diamines content and emission and migration rates from PUF (Karlsson, 2023; Karlsson et al., 2023).
In this work, the RA approach taken by Arnold et al. (2012) was followed. Their work addressed concerns of potential health effects from residual TDI in PUF associated with sleeping on a PUF mattress. In that work, toxicity benchmarks for key cancer and non-cancer endpoints (e.g., sensitization) were derived from human and animal data and were used to evaluate migration and emission of TDI from PUF based on data by Vangronsveld et al. (2013) in an “extreme case” exposure scenario for sleeping on a PU foam mattress. Upper-bound inhalation exposure to TDI as well as a lifetime-average daily dose to TDI from dermal exposure were calculated. Arnold et al. (2012) concluded that sleeping on a PU foam mattress does not pose TDI-related health risks to consumers.
Using the same principles of deriving toxicological benchmark values, using current RA approaches, and assessing experimental emission and migration data, this work provides a rigorous evaluation of the 5-mg.kg−1 limits for MDA and TDA in the CertiPUR PUF voluntary certification scheme.
Methods
Risk is a function of both hazard and exposure. RAs are generally performed in four steps (NRC, 1983): hazard, dose-response, exposure, and risk assessment. The sections delineate each of these steps, the data used, and the assumptions made for the RA scenario.
Substances and hazard classification
TDI for manufacturing PUF is in most cases an 80:20 mixture of the 2,4- and 2,6-TDI isomers. In rare instances, a 65:35 mixture is used. Within the scope of this investigation, the corresponding diamines are 2,4- and 2,6-TDA, both of which are classified as skin sensitizers (ECHA, 2023a; ECHA 2023b). For 2,4-TDA (CASRN 95-80-7), a chronic toxicity study by oral administration in rats and mice is available (Cardy, 1979; NCI, 1979), on the basis of which 2,4-TDA can be classified as Carcinogen Category 1B (BAuA, 2008). 2,4-TDA is treated as a genotoxic carcinogen. In another chronic study by oral administration in rats and mice (NCI, 1980), 2,6-TDA (CASRN 823-40-5) was found not to be carcinogenic. Tumors were found but their incidence was not significantly increased compared to controls. Although 2,6-TDA displayed both negative and positive results in in vivo genotoxicity studies, based on the 2-year cancer study, 2,6-TDA is taken to be non-carcinogenic (EPA, 2005a).
MDI used in flexible polyurethane foam production may contain the three isomers (4,4’-, 2,4’-, and 2,2’-MDI). The corresponding diamines of relevance in this investigation are 4,4’-, 2,4’-, and 2,2’-MDA. The 4,4’-MDA isomer (CASRN 101-77-9) is classified as a skin sensitizer (ECHA, 2023c). For lack of data, the 2,4’- and 2,2’-MDA isomers are assumed to exhibit the same property. A chronic toxicity study by oral administration in rats is available for 4,4’-MDA (Lamb et al., 1986; NTP, 1983; Weisburger et al., 1984), on the basis of which 4,4’-MDA was classified as Carcinogen Category 1B (ECB, 2001). 4,4’-MDA is treated as a genotoxic carcinogen. No chronic study for 2,2’-MDA (CASRN 6582-52-1) is available and its hazard potential was conservatively assumed to be the same as that of 4,4’-MDA. There is no chronic study available for 2,4’-MDA (CASRN 1208-52-2) either. Even though 2,4’-MDA may not be genotoxic based on negative bacterial reverse mutation (OECD, 2020), chromosomal aberration (OECD, 2016a), and in vitro gene mutation (HPRT) (OECD, 2016b) tests (Unterberger-Henig, 2022), the hazard profile of 4,4’-MDA was conservatively assumed to apply to 2,4’-MDA as well.
Dose-response assessment and derivation of toxicological benchmarks
Carcinogenicity
The most relevant and sensitive systemic effect for 2,4-TDA and 4,4’-MDA is carcinogenicity. The derivation of the toxicological benchmark was based on the T25 from the respective 2-year oral chronic animal studies. T25 is the dose at which 25% of test animals would exhibit the effect, that is, tumor formation. The oral T25 can be taken as the basis for estimating risk provided it is compared with the total absorbed dose from all routes of exposure. An allometric scaling factor of 4 (rat) was applied, assuming a human body weight of 70 kg. The resulting human equivalent T25 values were then adjusted to an excess cancer risk of 10−6 for consumers (general population) using linear extrapolation to zero, as commonly done for a presumed genotoxic mode of action (BAuA, 2008, 2010; ECHA, 2015).
The use of benchmark dose software (BMDS V.3.3.2, US Environmental Protection Agency, Washington DC, USA) as an alternative to the T25-based approach was explored but ultimately abandoned after careful consideration of the results of the chronic studies and the objections formulated by BAuA (2010: 12) and DECOS (2015: 19). While no overt signs thereof were apparent in the MDA study with rats, the high-dose groups in both the MDA study with mice and the TDA studies with either species were clearly overdosed (early body weight loss, intermediate dosing corrections, premature sacrifice of the animals, significant hepato-nephro-toxicity as well as liver degeneration were noted). With that many uncertainties regarding one of only two non-zero dose levels, the results of the low-dose groups appeared to be the more reliable starting point for deriving benchmark values.
Repeated dose toxicity
The 2,6-TDA isomer was shown not to be carcinogenic in a chronic animal study (NCI, 1980). Reduced body weight was identified as the lowest point-of-departure (POD) and used to derive a toxicity benchmark for this RA. The standard uncertainty factors (UF) for database deficiencies (10), interspecies (10) and intraspecies variability (10) were applied (aggregate UF = 1000; ECHA, 2012; EPA, 2005b).
Skin sensitization (local effects)
For skin sensitization, the Local Lymph Node Assay (LLNA) is the preferred method for deriving quantitative dose-response data (Gerberick et al., 2007; Roberts et al., 2016). Where LLNA data were available for the substances considered, they were used to calculate a human-equivalent No Expected Sensitization Induction Level (NESIL) according to equation (1) (Safford, 2008)
The EC3-value is the concentration that led to a stimulation index of 3 compared to controls. In equation (1), it is converted into a surface loading (EC3-potency). In the absence of LLNA results, the GHS classification boundary of 1% for Category 1B dermal sensitizers in mixtures (UN, 2021), corresponding to an EC3 of 250 μg.cm−2, was used as a surrogate input to equation (1) for estimating the NESIL. The NESIL was then converted to an Acceptable Exposure Level (AEL) using UFs of 1 for interspecies (Basketter et al., 2018) and 10 for intraspecies variability, and 3 for matrix effects (Basketter and Safford, 2016) (aggregate UF = 30).
Experimental data and exposure assessment
To link the emission (inhalation) and migration (dermal) exposure potentials with the CertiPURTM quality limit (EUROPUR, 2021), data are required from foams that have defined concentrations of TDA or MDA. This exposure assessment made use of the consistent set of emission and migration data generated by Karlsson et al. (2023).
A brief summary is provided here. Both a representative TDI- and MDI-based PUF were thermally treated to generate extractable levels of diamines close to the CertiPUR limit value of 5 mg.kg−1. Extractable diamines were determined using a method that mirrors the CertiPUR guidance (EUROPUR, 2021) but using 2 instead of the prescribed 4 consecutive extractions with 1% aqueous acetic acid. This could have resulted in a slight underestimation of the extractable diamine content in each foam. When coupled with the fact that measured results were normalized to the 5 mg.kg−1 foam content limit value (as described in the Migration rates section below), the measured extraction values could result in a slight overestimation of the risk and would therefore be considered conservative for the risk assessment evaluation.
Emission rates (inhalation exposure)
The foam samples for emission testing contained up to 9.2 and 6.0 mg.kg−1 of 2,4- and 2,6-TDA, respectively, and up to 27 mg.kg−1 of the summed MDA-isomers (ΣMDA). Emissions from the foams were measured during 8 h using a specifically-designed and validated emission cell (Karlsson, 2023). The limit-of-quantification (LOQ) for the emission rate was 8.5 ng.m−2.h−1 for 2,4-TDA, 28 ng.m−2.h−1 for 2,6-TDA, and 71 ng.m−2.h−1 for ΣMDA. All measured emission rates were less than the respective LOQ.
In the RA, emission for the entire period of use of the mattress was assumed to take place from all surfaces of the mattress at an average rate corresponding to one-half of the LOQ (EPA, 1991) linearly pro-rated to the 5-mg.kg−1 limit value. The rate thus determined was converted into the average external daily inhalation dose as described under “Exposure scenario.”
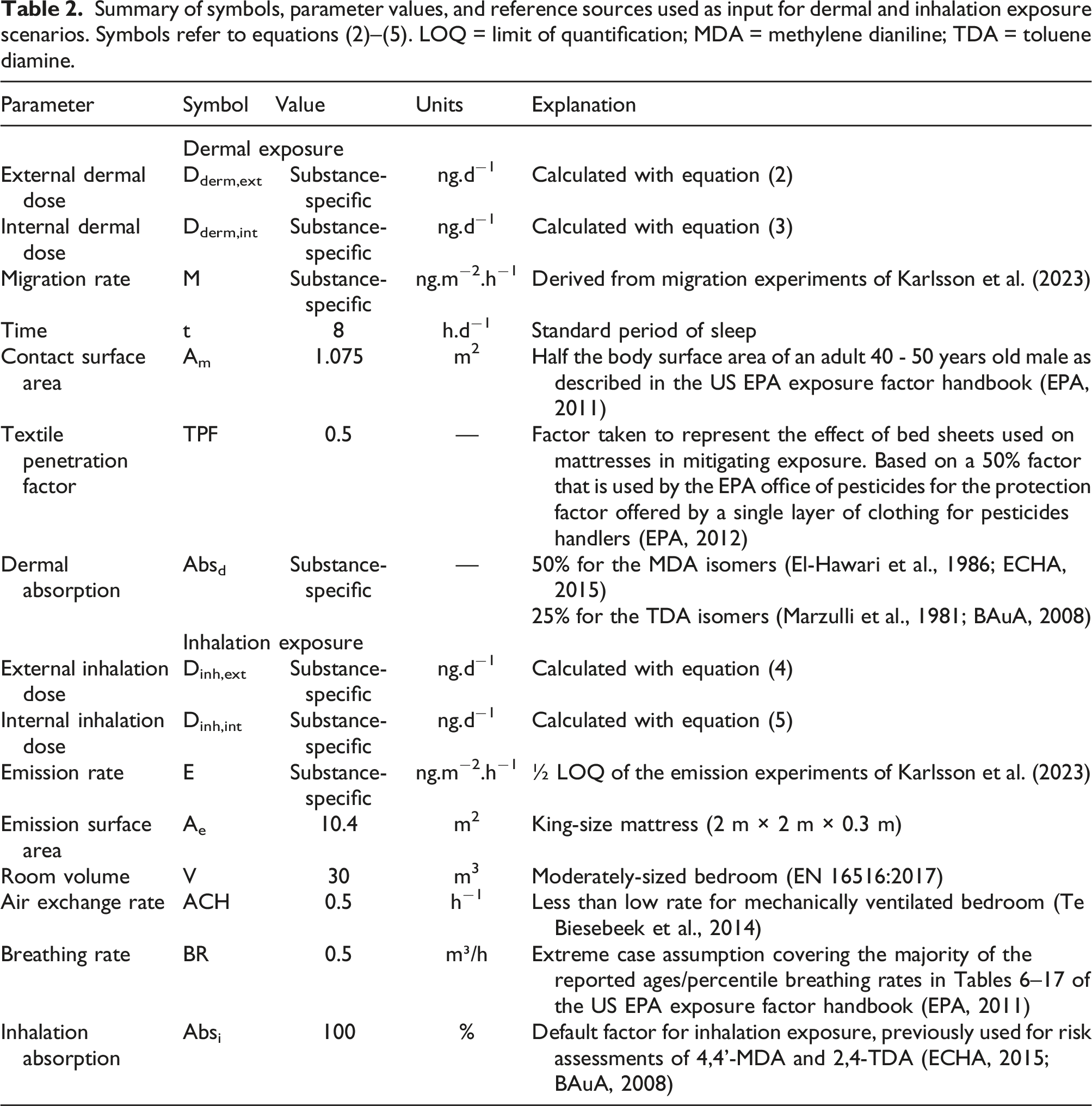
Migration rates (dermal exposure)
Data from Karlsson et al. (2023) and adjustments made for estimating the average daily migration rate. LOQ = limit of quantification; ΣMDA = sum of the three methylene dianiline isomers; TDA = toluene diamine.
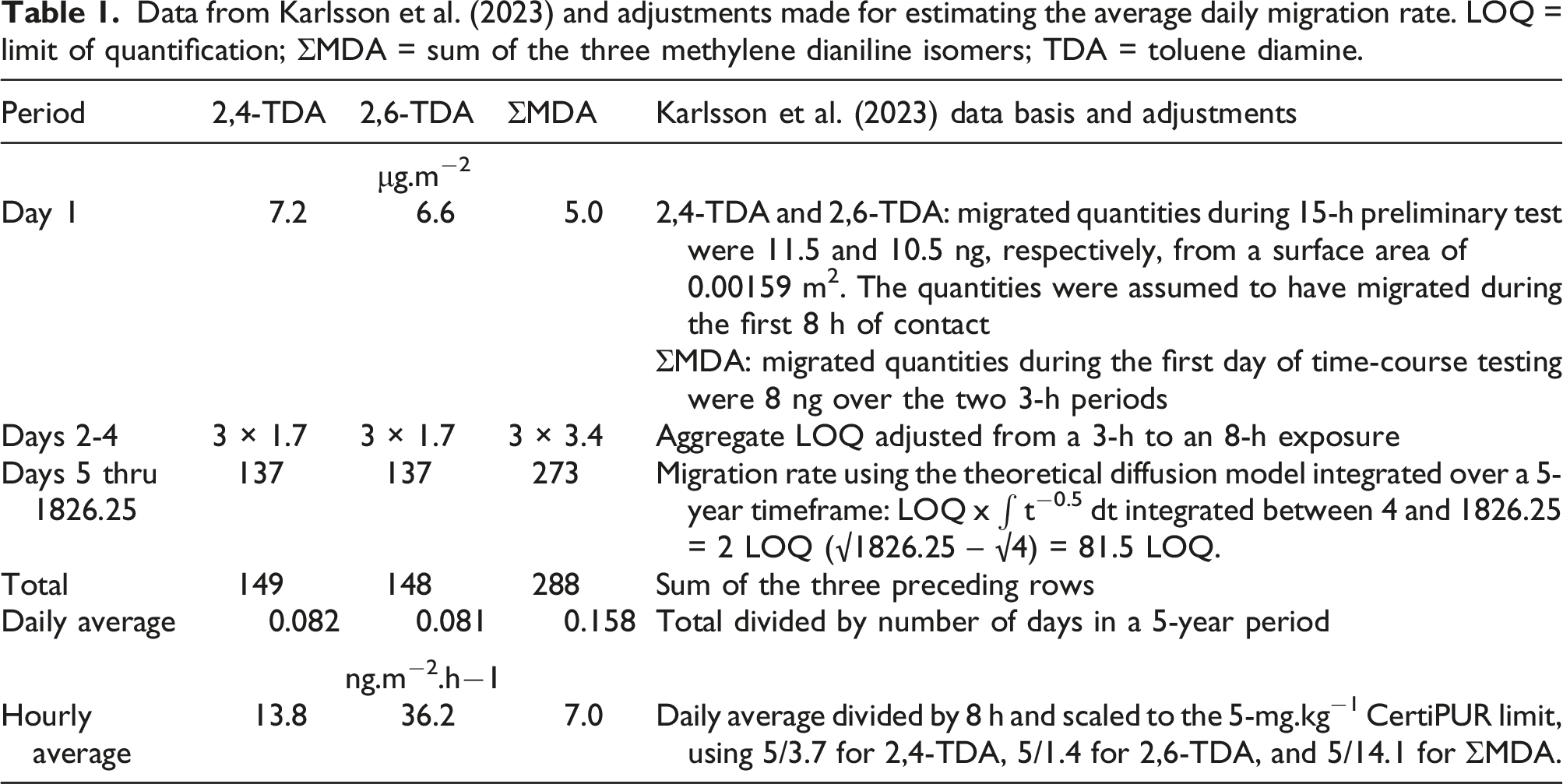
In the RA, migration rates were linearly normalized to the 5 mg.kg−1 foam content limit value from the actual level in the foam sample used and adjusted to an 8-h exposure duration as needed. A mitigation factor of 50% was applied to take into account the presence of a sheet (EPA, 2012) for determining the average external daily dermal dose.
Risk assessment
Exposure scenario
An exposure scenario was constructed representing the reasonable-worst-case scenario of a person sleeping on a foam mattress for 8 h per day, based on the same exposure scenario used by Arnold et al. (2012).
The average daily external and internal dermal doses (Dderm,ext and Dderm,int) were calculated using equations (2) and (3)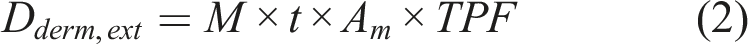
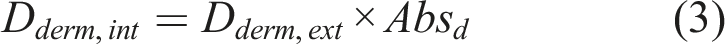
The average daily external and internal inhalation doses (Dinh,ext and Dinh,int) were calculated using equations (4) and (5)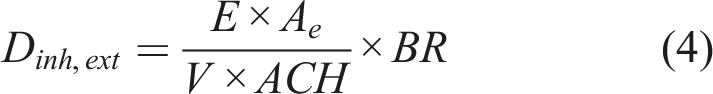
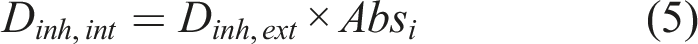
Risk characterization
Deterministic risk evaluations are typically expressed either as the margin of safety (MOS), where MOS equals the toxicological benchmark divided by the average daily total internal dose, or as its inverse, the risk characterization ratio (RCR). A value of MOS >1 or RCR <1 indicated that there is no unacceptable risk to human health.
Sensitivity analyses
It is plausible that an individual may have additional contact with PUF during waking hours, for instance, from upholstered furniture or automotive vehicles. To estimate the effect of this potential additional exposure, a sensitivity analysis was performed whereby the time for the “sleeping on a mattress” scenario was extended to 24 h.
One of the purposes of this work was to probe the adequacy of the 5-mg.kg−1 CertiPUR quality limit for MDA. The main analysis considered the sum of the three MDA isomers. In addition, the effects of a ΣMDA limit of 15 mg.kg−1 and a 5-mg.kg−1 limit for 4,4’-MDA alone were assessed.
Results
Derivation of toxicological benchmarks
Carcinogenicity
4,4'-MDA
The key study for carcinogenicity was performed in F344 rats and B6C3F1 mice (Lamb et al., 1986; NTP, 1983; Weisburger et al., 1984). Both liver and thyroid tumors were observed. The T25(oral, rat) value derived from this study was 9.01 mg. kgbw−1.d−1 (ATSDR, 1998; ECHA, 2015), based on liver tumors in male rats 2 , following correction for control incidence and study duration (actual 103-week vs normal 104-week). From this, a systemic toxicological benchmark of 9 ng. kgbw−1. d−1 was derived. Since no isomer-specific carcinogenicity studies were identified, the systemic toxicological benchmark of 4,4’-MDA was also used for the 2,4’-MDA and 2,2’-MDA isomers
2,4-TDA
The key study for 2,4-TDA was performed in F344 rats and B6C3F1 mice (Cardy, 1979; NCI, 1979). Target organs in rats included both liver and mammary glands. Although the EU RA noted uncertainty in the biological relevance of the mammary gland tumors, they were taken forward as a conservative basis for developing the toxicity benchmark (BAuA, 2008). The T25(oral, rat) valued based on tumorigenesis of the mammary glands was 2 mg.kgbw−1.d−1 (BAuA, 2008), for liver tumors 14 mg.kgbw−1.d−1. From this, a systemic toxicological benchmark of 2 ng. kgbw−1.d−1 was derived.
Repeated dose toxicity
2,6-TDA
2,6-TDA was shown not to be a carcinogen based on a 2-year carcinogenicity study (NCI 1980). Although tumors were observed in both rats and mice dosed with 2,6-TDA hydrochloride, the incidences were not statistically increased from controls (NCI 1980). This study provided adequate data to derive benchmarks based on non-cancer endpoints that can be used for RA. Specifically, reduced body weight in rats was not considered adverse, so the high dose of 25 mg. kgbw−1.d−1 in male rats (32 mg. kgbw−1.d−1 for females) could be used as POD (EPA, 2005a). Applying the standard uncertainty factor of 1000, a toxicological benchmark of 25,000 ng. kgbw−1.d−1 was obtained, in agreement with the published reference dose (EPA, 2005a).
Sensitization
TDA
For 2,4-TDA, a reliable LLNA study (Vanoirbeek et al., 2009) reported an EC3 value of 19%. The corresponding NESIL was then calculated to be 4216 µg.cm−2. The choice of a safety assessment factor for sensitization is somewhat case-specific as matrix effects that may affect dermal absorption could vary widely depending on the application under consideration. For dermal contact with foams a safety factor of 30 was used, which yielded an AEL of 141 µg.cm−2.
Since no LLNA study was found for 2,6-TDA, the same AEL was applied.
MDA
For 4,4'-MDA, no LLNA study has been reported. Based on a human repeated insult patch test (ECHA 2023d), 4,4'-MDA was classified as a Category 1B dermal sensitizer. Taking the GHS reporting threshold of 1% as a surrogate input, a NESIL of 139 µg/cm2 was derived. Applying the assessment factor of 30 yielded an AEL of 4.6 µg.cm−2. Since no isomer-specific sensitization studies were identified, the AEL of 4,4-MDA was also used for the 2,4’-MDA and 2,2’-MDA isomers.
Exposure estimation
Emission rates (inhalation exposure)
ΣMDA
The emission test was conducted with a 150-mm diameter exposed foam surface (0.0177 m2), a test time duration of 8 h, and a ΣMDA LOQ of 71 ng.m−2.h−1. No emission was detected under these circumstances. The emission rate based on half the LOQ (½ × 71) was 35 ng.m−2.h−1. Since the foam samples contained up to 27 mg.kg−1 of ΣMDA, scaling to the 5-mg.kg−1 CertiPUR limit value resulted in an adjusted emission rate of (35 × 5/27) 6.5 ng.m−2.h−1.
TDA
The TDI-based foam samples contained up to 9.2 and 6 mg.kg−1 2,4-TDA and 2,6-TDA, respectively. No emission was detected at the respective LOQ of 8.5 and 28 ng.m−2.h−1. Emission rates based on half the LOQ were 4.2 and 14 ng.m−2.h−1 for 2,4- and 2,6-TDA, respectively. Scaled to the 5-mg.kg−1 limit, the adjusted emission rates were 2.3 ng.m−2.h−1 for 2,4-TDA and 12 ng.m−2.h−1 for 2,6-TDA.
Migration rates (dermal exposure)
The results of Karlsson et al. (2023) showed that migration occurred primarily during Day 1 and that little to no replenishment of TDA and MDA from the interior to the surface of the foam took place. Average daily migration rates were calculated as shown in Table 1. The migration tests were conducted with a 45-mm diameter exposed foam surface (0.00159 m2). For Day 1, the maximum measured value was taken into account. For Days 2 through 4, the aggregate LOQ was taken as a basis. For the remainder of the mattress use period, the migration rate was integrated using the theoretical t−0.5 model (Karlsson et al., 2023). The average hourly values were scaled to the 5-mg.kg−1 CertiPUR limit using the measured concentration of diamine in the foam samples.
ΣMDA
For ΣMDA, an average adjusted hourly migration rate of 7.0 ng.m−2.h−1 was obtained.
TDA
For 2,4-TDA, an average adjusted hourly migration rate of 13.8 ng.m−2.h−1 was calculated. For 2,6-TDA, the adjusted hourly migration rate was 36.2 ng.m−2.h−1.
The adjusted average hourly emission and migration rates were carried forward into the exposure scenario and RA scheme as described in Methods (equations (2)–(5) and Table 2). Detailed calculations and the risk characterization are available in Supplemental Information.
Risk characterization
Carcinogenicity
2,4-TDA
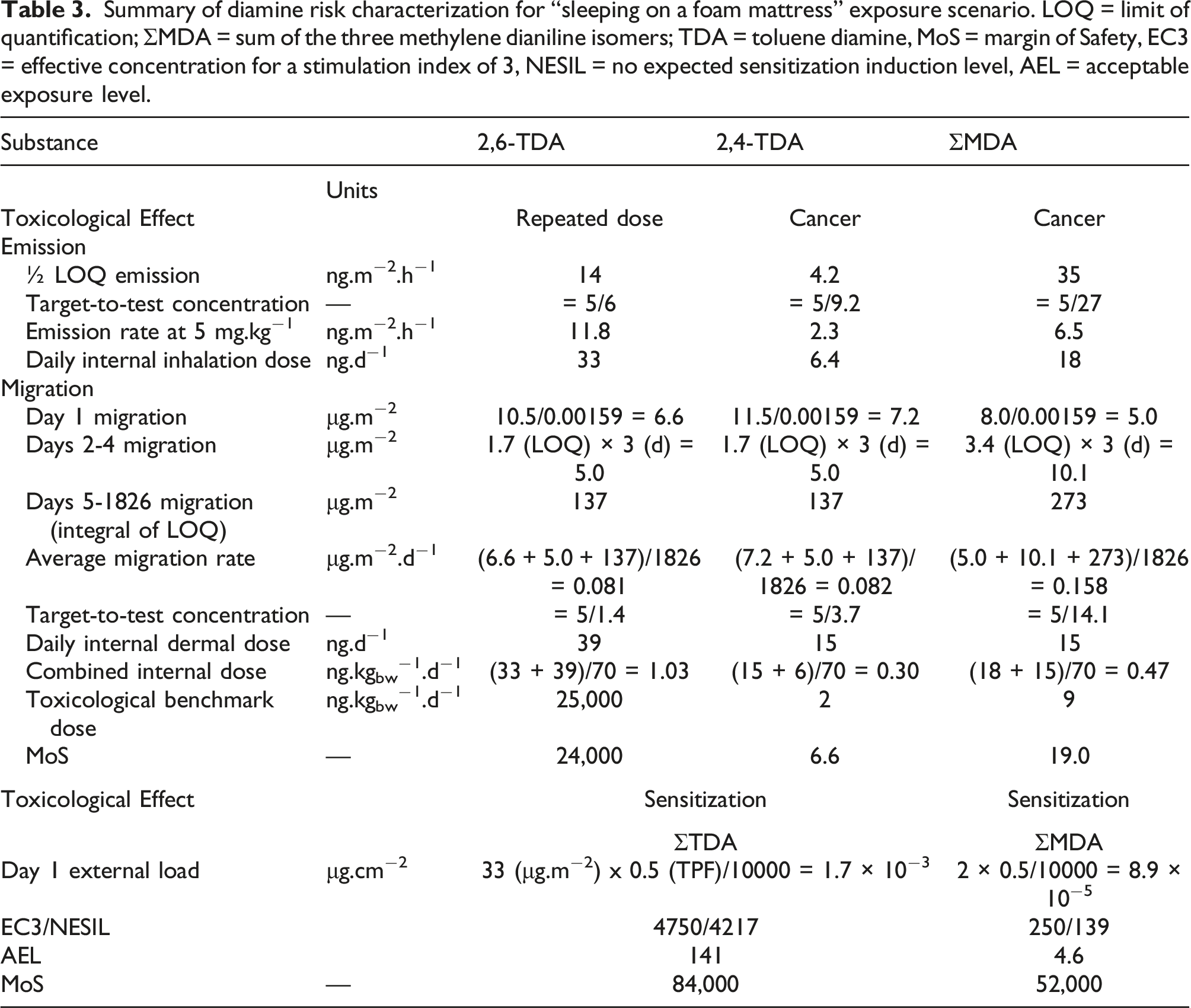
A total systemic uptake of 21 ng.d−1 and a corresponding internal dose of 0.3 ng. kgbw−1. d−1 were calculated. Compared to the toxicological benchmark of 2.0 ng. kgbw−1. d−1, the MOS against a 10−6 risk level was 6.6.
ΣMDA
A total systemic uptake of 33 ng.d−1 and a corresponding dose of 0.5 ng.kgbw−1.d−1 were calculated. Compared to the toxicological benchmark of 9.0 ng.kgbw−1. d−1, the MOS against a 10−6 risk level was 19.
For both 2,4-TDA and ΣMDA, sleeping on a PUF mattress that satisfies the CertiPUR quality standard of ≤5 mg.kg−1 does not constitute an unacceptable risk to consumers.
Repeated dose toxicity
2,6-TDA
A total systemic uptake of 72 ng.d−1 and a corresponding internal dose of 1.0 ng. kgbw−1. d−1 were calculated. Compared to the toxicological benchmark of 25,000 ng. kgbw−1. d−1, the MOS was 2.4 × 104, indicating that there is no systemic adverse health risk resulting from exposure to 2,6-TDA.
Sensitization
For local effects, the relevant metric is the highest single dermal dose, which would occur on the first day. The Day 1 migration rates were 33 μg.m−2 for ΣTDA and 2 μg.m−2 for ΣMDA. The AEL were 141 μg.cm−2 for the ΣTDA and 4.6 μg.cm−2 for ΣMDA. This resulted in MOS of 8.4 × 104 for the ΣTDA and of 5.2 × 104 for ΣMDA. The sensitization risk resulting from exposure to the diamines is very low, even when considering “peak” exposures during the first day of mattress use.
Sensitivity analyses
When the contact and residence time for the “sleeping on a mattress” exposure scenario was extended to 24 h (discussed in more detail below), the combined internal dose increased to 0.9, 3.1, and 1.4 ng.kg−1. d−1 resulting in MOS of 2.2, 8100 and 6.3 for 2,4-TDA, 2,6-TDA, ΣMDA, respectively.
When the RA was conducted considering only the 4,4’-MDA isomer (i.e., assuming that the other two isomers would not be carcinogens), an MOS of 9.0 resulted (Supplemental Information). When the ΣMDA concentration in the foam was increased to 15 mg.kg−1, the combined internal dose became 1.4 ng.kg −1. d −1 resulting in an MOS of 6.3.
Discussion
Summary of diamine risk characterization for “sleeping on a foam mattress” exposure scenario. LOQ = limit of quantification; ΣMDA = sum of the three methylene dianiline isomers; TDA = toluene diamine, MoS = margin of Safety, EC3 = effective concentration for a stimulation index of 3, NESIL = no expected sensitization induction level, AEL = acceptable exposure level.
The calculated systemic internal inhalation dose was approximately 30% of the total internal dose for 2,4-TDA and 55% for ΣMDA. One-half of the respective LOQ was used as model input. No decrease in emission rates was assumed over the lifetime of the mattress. This is a conservative assumption since it is expected that emissions of substances from PUF would decrease with time, either by depletion of the substance by emission itself (Hillier et al., 2003; OECD, 2009) or—in the case of diamines—by reaction with the PU matrix (Krämer-Lucas et al., 2001).
The scenario parameters (Table 2) can generally be considered conservative as well. A king-size mattress (2 m × 2 m x 0.3 m) was considered. The room volume (30 m3) was in the range indicated by regulatory bodies (16 m3 (Te Biesebeek et al., 2014) to 41 m3 (Jennings et al., 1987) and matched the reference room size (4 m × 3 m × 2.5 m) used for evaluating construction products (EN 16516, 2017; Oppl, 2014). The ventilation rate (0.5 h−1) was less than the smallest factor published by the Dutch National Institute for Public Health and Environment (RIVM) (Te Biesebeek et al., 2014) (0.6 h−1 for a bedroom with mechanical ventilation). The assumed breathing rate while sleeping was greater than the 95th-percentile reported for all age categories by EPA (2011) and the standard rate used by the Netherlands National Institute for Public Health and the Environment (RIVM) (0.45 m3.h−1, Te Biesebeek et al., 2014).
On balance, the parameters and emission rates used for estimating the internal inhalation dose can be considered a reasonable-worst-case model for sleeping on a mattress. However, it can be argued that 8 h of sleep per day is not necessarily conservative and that longer residence times in the room would be possible or that exposure could occur in other places that contain PUF. This was the subject of the sensitivity analyses discussed below.
The calculated internal dermal dose accounted for approximately 70% of the total internal dose for 2,4-TDA and 45% for ΣMDA. The calculation was based on the measurements performed by Karlsson et al. (2023). For Day 1, the largest measured migration rate was used, that is, for TDA isomers the results of the 15-h experiments and for MDA an aggregate of the measured migration over the two first day 3-h measurements of the time course series. For Days 2 thru 4, while most measurements were <LOQ, migration was assumed to correspond to the aggregate LOQ. For both periods, these are conservative assumptions. For the remainder of the mattress use life, a theoretical model based on Fick’s diffusion law was used (Karlsson et al., 2023). This model is similar to the one described by OECD (2009) for modeling emissions.
The scenario parameters (Table 2) can generally be considered conservative as well. Mattress replacement is assumed to take place every 5 years. Foley (2023) recommends mattresses be replaced every 6–8 years. In The Netherlands, ca. 1.5 million mattresses are discarded every year on a population of 17.4 million (Michel Baumgartner, 15 March 2021; personal communication), suggesting a service life exceeding 11 years. The 8-h contact time is more conservative than for inhalation: while the residence time in the room may be longer, dermal contact is less likely to exceed 8 h.d−1. The contact area of 1.075 m2 (half the body skin surface area) assumes the person lies flat and sunk into the mattress for the entire 8 h of sleeping. The textile protection factor (0.5) is the one recommended by US EPA for the protection offered by a single layer of clothing against pesticides applied directly onto the mattress cover (EPA, 2012). Finally, it should be noted that Karlsson et al. (2023) observed that 80% of the migrated MDA and 35% of TDA were retained on the cloth that was in direct contact with the foam in the presence of an extraction medium more acidic than sweat. In balance, the parameters and migration rates used for estimating the internal dermal dose represent a reasonable-worst-case model.
The risk assessment results reflect adult use of PU foam mattresses. However, it is possible that the use of PU mattresses could start in infancy. Thus, it is reasonable to examine the potential impact of these earlier life exposures on estimated risk from exposure to the primary aromatic amines. The US EPA has developed guidance on how to account for different (higher) susceptibility to cancer from early lifetime exposures (EPA 2005b). That guidance advises that an additional factor of 10 be applied for exposures from birth to age 2 years and a 3-fold factor be applied for the 14 years between age 2 and 16. The overall impact of assuming lifetime exposure (i.e., including the first 16 years of life, with the noted adjustment factors) is to increase RCRs by a factor of approximately 1.66 or to decrease MOS by about 1.66. For 2,4-TDA, for example, the MOS for carcinogenicity (adults' exposure only) of 6.6 would be adjusted to 4.1 for lifetime exposure. This indicates that cancer risk is unlikely to be elevated to unacceptable levels when considering lifetime exposures, and the same reasoning applies to the MDA isomers.
While the purpose of this work was to provide a health-based evaluation of the diamine quality limits included in CertiPUR rather than to provide a definitive lifetime risk assessment of contact with all potential PUF applications, several sensitivity analyses were conducted to assess the impact of additional considerations.
Consideration of prolonged exposure or exposure to a variety of other PUF applications would require making many additional assumptions. However, dermal exposure from sleeping on a mattress (largest contact area, minimal cover considered) is conservative compared to that of other applications. On the other hand, inhalation exposure during waking time (balance of breathing rate and better ventilation) may be greater. To provide an indication of full-day exposure to PUF, the scenario was extended to 24 h, resulting in an MoS of 2 or greater for all substances.
The increase of the ΣMDA concentration to 15 mg.kg−1 resulted in an MOS of 6.4, and conducting the RA solely using the 4,4’-MDA isomer resulted in an MOS of 9.0, demonstrating that the CertiPUR diamine limits remain adequately protective.
Summary and conclusion
The sensitization and non-cancer endpoints lifetime risk for consumers by sleeping on a PUF mattress was found to be very low. For the presumed carcinogens (2,4-TDA and 4,4’-MDA), the CertiPUR voluntary certification program requires their concentrations to be ≤5 mg.kg−1. For 2,4-TDA, this limit was found to be protective (MOS = 6.6). Our results showed that ΣMDA ≤5 mg.kg−1 led to a MOS of 19. It was also shown that the limit of 4,4’-MDA ≤5 mg.kg−1 was conservative (MOS = 9). However, pending further studies on 2,2’- and 2,4’-MDA, it would be scientifically more correct to use a ΣMDA limit ≤15 mg.kg−1 (MOS = 6.4). 3
Supplemental Material
Supplemental Material - Risk assessment - based verification of the CertiPUR
TM
limit values for toluene diamine and methylene dianiline in flexible polyurethane foam
Supplemental Material for Risk assessment - based verification of the CertiPURTM limit values for toluene diamine and methylene dianiline in flexible polyurethane foam by Patrick De Kort, Elke Jensen, Mark W Spence, and Patrick M Plehiers in Toxicology and Industrial Health.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Patrick de Kort is employed by EUROPUR. The trade association is funded by producers of flexible polyurethane foam. Mark W Spence and Patrick M Plehiers are employed by the International Isocyanate Institute, Inc. The Institute is funded by producers of TDI and MDI. Elke Jensen is employed by Dow, a producer of raw material for polyurethane.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the International Isocyanate Institute, Inc. and EUROPUR. Any opinions expressed are those of the authors, not necessarily of the International Isocyanate Institute Inc., EUROPUR, or their member companies.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.